Introduction
Endometrial cancer (EC), the most common
gynecological malignant cancer, can be clinically classified into 2
types (1,2). Type I, the most common clinically, is
most likely caused by the long-term stimulation of estrogen,
obesity, hypertension and diabetes. Generally, the Type I EC
population is younger with a higher degree of tumor differentiation
and better prognosis (3). By
contrast, type II EC is usually observed in women aged >65
years, who have experienced pregnancy (2,4).
Although most EC cases are diagnosed at an early stage, which is
correlated with a better outcome, women at an intermediate and late
stage of EC are at a highest risk of recurrence and poor prognosis.
In addition, the lowly differentiated ones can hardly be cured,
resulting in death rates of ~20%. In particular, EC cases with
regional or distal metastasis tend to recur, and the effect of
systemic therapies is limited (5–8).
Therefore, it is important to further explore the molecular
mechanisms responsible for EC tumorigenesis and progression, and to
develop novel diagnostic and therapeutic strategies.
According to The Cancer Genome Atlas (TCGA)
datasets, solute carrier organic anion transporter family member
4C1 (SLCO4C1) upregulation was revealed to be strongly associated
with the survival time and clinical stage of patients with EC.
SLCO4C1 is a human kidney-specific organic anion transporting
polypeptide (9,10). In addition, SLCO4C1 was revealed to
excrete uremic toxins, which led to reduced blood pressure and
decreased kidney inflammation (11,12).
In addition, SLCO4C1 also served as a tumor suppressor gene in head
and neck cancer (HNC) (13). These
results revealed that SLCO4C1 is involved in tumorigenesis,
however, there is no study on the relationship between SLCO4C1 and
EC.
In the present study, it was revealed that SLCO4C1
interference regulated biological behaviors of EC cells, such as
proliferation, apoptosis, sphere formation, migration, invasion and
EMT phenotype by inactivating the PI3K/Akt signaling pathway. These
results shed lights on the mechanisms underlying EC occurrence and
development, and provided evidence that the SLCO4C1/PI3K/Akt
signaling pathway may be a potential therapeutic target for the
treatment of EC.
Materials and methods
SLCO4C1 expression analysis in the
TCGA dataset
TCGA_UCEC_exp_RNA-seq, a TCGA dataset containing 177
Uterine Corpus Endometrial Carcinoma and 24 para-tumor tissue
specimens was downloaded from https://tcga-data.nci.nih.gov/tcga/. The clinical
features of patients, including survival time and clinical stage,
were collected retrospectively, based on the records of patients.
The significance threshold of the differentially expressed mRNA was
set at P<0.05 following t-tests. The correlation of clinical
characteristics with SLCO4C1 expression was analyzed using
χ2 test. The prognostic value of SLCO4C1 in EC was
analyzed using ggsurv R package (http://www.sthda.com/english/rpkgs/survminer/), the
clinical stage of SLCO4C1 in EC was assessed using Spearman's rank
correlation coefficient method.
Gene ontology (GO) enrichment
analysis
GO analysis has been recognized as a helpful
approach to annotating genes and gene sets that possess relevant
biological features for high-throughput genomic or transcriptomic
data. The Database for Annotation, Visualization and Integrated
Discovery (DAVID) has provided simple and efficient annotation
tools for researchers, aiding them in identifying the biological
meanings of different genes. In the present study, GO enrichment
analysis of the differentially expressed genes (DEGs) was performed
using the aforementioned DAVID database, and FDR<0.05 was deemed
as the threshold.
Patients and tissues
A total of 20 EC and 10 normal endometrial tissues
were obtained from female Chinese patients who had undergone
surgical treatment from January 2017 to December 2019 at the
Shanghai First Maternity and Infant Hospital (Shanghai, China). No
patient had undergone endocrine therapy, radiotherapy or
chemotherapy prior to surgery. The present research was approved by
the Human Investigation Ethics Committee of the Shanghai First
Maternity and Infant Hospital. Samples were collected from patients
after receiving their written informed consent.
Immunohistochemistry (IHC)
Endometrial tissues were sliced into 5-µm thick
sections and embedded in paraffin. Next, the sections were mounted
on poly-L-lysine-coated slides and then deparaffinized in xylene
and dehydrated with gradient ethanol. Antigen retrieval was carried
out in 10 mM citrate buffer (pH 6.0) for 10 min. Endogenous
peroxidase activity was blocked with 3% hydrogen peroxide for 10
min at room temperature. Following blocking with 1% horse serum
albumin for 20 min at room temperature, the sections were incubated
with rabbit polyclonal antibody against SLCO4C1 (cat. no.
203261-T10; 1:100 dilution; Sino Biological, Inc.) overnight at 4°C
and then incubated with rabbit polyclonal secondary antibody (cat.
no. VC003; 1:200 dilution; R&D Systems, Inc.) at 37°C for 30
min. The sections were stained with DAB for 5 min and re-stained
with hematoxylin for 2 min.
Cell culture
Several human EC cell lines, including HEC-1A,
HEC-1B, Ishikawa 3H12, RL95-2 and KLE, were provided by the
Shanghai Institute of Cell Biology, Chinese Academy of Sciences.
HEC-1A cells were cultured in McCoy'5A (Thermo Fisher Scientific,
Inc.) containing 10% fetal bovine serum (FBS; GE Healthcare) and 1%
penicillin/streptomycin (P/S; Thermo Fisher Scientific, Inc.).
HEC-1B, Ishikawa 3H12 and RL95-2 cells were cultured in Dulbecco's
Modified Eagle's Medium (DMEM)/Nutrient Mixture F-12 (Biological
Industries) containing 10% FBS and 1% P/S. KLE cells were cultured
in high-glucose DMEM (Thermo Fisher Scientific, Inc.) containing
10% FBS as well as 1% P/S. Cells were placed in an incubator
supplemented with 5% CO2 at 37°C.
Small interfering RNA (siRNA)
transfection
The siRNAs targeting SLCO4C1 (siSLCO4C1-1 and
siSLCO4C1-2) and the negative control (siNC) were purchased from
Hanbio Biotechnology Co., Ltd. The control (Ctrl) was a blank
control group. HEC-1A and RL95-2 cells were transfected with the
siRNAs, siNC or Ctrl in Opti-MEM (Thermo Fisher Scientific, Inc.)
using Lipofectamine 3000 (Thermo Fisher Scientific, Inc.),
according to the manufacturer's instructions. The concentration of
siRNA transfected in the cells reached 1 nM in a 12-well plate. The
sequences of the RNA oligonucleotides were as follows: siSLCO4C1-1
forward, 5′-CAGGCUCAUCAGAGUAAUAdTd-3′ and reverse,
3′-UAUUACUCUGAUGAGCCUGdTd-5′; siSLCO4C1-2 forward,
5′-CAUAUGCUAGUAGCCAUAAdTd-3′ and reverse,
3′-UUAUGGCUACUAGCAUAUGdTd-5′; siNC forward,
5′-UUCUCCGAACGUGUCACGUdTd-3′ and reverse,
3′-ACGUGACACGUUCGGAGAAdTd-5.
Cell viability assay
Cell viability was assessed by CCK-8 assay (Dojindo
Molecular Technologies, Inc.). HEC-1A and RL95-2 cells were
inoculated into 96-well plates (4×103 cells/well) for 24
h, followed by transfection with siNC, siSLCO4C1-1 or siSLCO4C1-2.
In the rescue experiment, an equal number of cells from the
siSLCO4C1-1 or siSLCO4C1-2 group was treated with 740 Y-P (PI3K
signaling activator, 100 µg/ml and incubation for 24 h;
MedChemExpress), comprising groups siSLCO4C1-1+740 Y-P or
siSLCO4C1-2+740 Y-P, respectively. After 24, 48 and 72 h, 10 µl
CCK-8 solution was added to each well, followed by further
incubation of the plates at 37°C for 4 h. Subsequently, a
microplate reader was used to measure the ultraviolet absorbance of
all samples at 450 nm.
Apoptosis assay
Apoptosis was detected using the Annexin V-FITC
apoptosis detection kit I (BD Biosciences). HEC-1A and RL95-2 cells
were washed with cold phosphate-buffered saline (PBS; Biological
Industries) twice and resuspended in Annexin V binding buffer at a
density of 0.5×107 cells/ml. Subsequently, 100 µl cell
suspension was transferred into the 1 ml test tube, followed by the
addition of Annexin V- FITC (5 µl) and propidium iodide solution
(10 µl). Then, cells were subjected to gentle vortex and 15 min of
incubation in the dark under ambient temperature (25°C).
Subsequently, Annexin V binding buffer (400 µl) was added to each
tube, followed by flow cytometry (Accuri C6; BD Biosciences) in 1
h. Early apoptotic cells were stained Annexin V-positive and
PI-negative, whereas late apoptotic cells were stained both Annexin
V- and PI-positive. The sum ratio of early and late apoptosis
represented in Q3 and Q2 quadrants, respectively, was calculated
for apoptosis rate analysis.
Sphere-formation assay
A total of 1,000 single cells were inoculated into
each well of the 24-well ultra-low attachment plate (Corning Inc.),
which was filled with serum-free high-glucose DMEM. The N2 plus
media supplement (Thermo Fisher Scientific, Inc.), epidermal growth
factor (20 ng/ml; Thermo Fisher Scientific, Inc.), basic fibroblast
growth factor (20 ng/ml; Thermo Fisher Scientific, Inc.), and
heparin (4 mg/ml; Merck KGaA) were also added to the medium. The
number of formed spheres >50 µm was calculated 10 days after
culture using the Leica DMi8 inverted microscope (magnification,
×200; Leica Microsystems GmbH).
Wound healing assay
HEC-1A and RL95-2 cells (from the Ctrl, siNC,
siSLCO4C1-1, siSLCO4C1-2, siSLCO4C1-1+740 Y-P and siSLCO4C1-2+740
Y-P groups) were inoculated into the 6-well plate until they
reached full confluence. A 200-µl pipette tip was used to create
scratch wounds on the cells. Cells were further cultured in the
serum-free high-glucose DMEM, and images of the scratches were
captured using the Leica DMi8 inverted microscope (magnification,
×50) at 0 and 48 h respectively.
Cell invasion assay
HEC-1A and RL95-2 cells were grown until they had
reached 50–70% convergence, followed by transfection as
aforementioned. The transfected cells were inoculated into the
upper Boyden chamber covered with Matrigel 24 h later, as
previously described (14). Next,
24 h after incubation, the cells that had not invaded the Matrigel
were gently eliminated using a cotton swab. The invasive cells on
the lower chamber surface were stained using Giemsa (prepared by
mixing 10 drops of Giemsa stock solution in 10 ml of PBS; Leagene
Biotechnology) at room temperature for 15 min and counted under the
Leica DMi8 inverted microscope (magnification, ×200). The relative
cell invasion activity was indicated as the fold change relative to
that of the respective controls.
RNA extraction and RT-qPCR
TRIzol reagent (Thermo Fisher Scientific, Inc.) was
used to extract the total RNA from cells. Next, the SYBR FAST
one-Step RT-qPCR kit (Merck KGaA) was used for RT-qPCR assays with
the CFX96 Real Time-PCR Detection System (Bio-Rad Laboratories,
Inc.). RT-qPCR was performed with following cycling conditions: 30
sec at 95°C; 40 cycles of 5 sec at 95°C, 20 sec at 60°C and 10 sec
at 72°C; 1 min at 95°C, 30 sec at 60°C; and 30 sec at 95°C. The
primer sequences used were as follows: SLCO4C1 (128 bp) forward,
5′-CAGACATGAAGAGCGCCAAA-3′ and reverse, 5′-AAGCTCCTGTGGCTGAGAAT-3′;
GAPDH (124 bp) forward, 5′-ACCCAGAAGACTGTGGATGG-3′ and reverse,
5′-TCAGCTCAGGGATGACCTTG-3′. The SLCO4C1 expression was normalized
to that of GAPDH, and the relative miRNA expression level was
measured using the 2−ΔΔCq method (15).
Western blotting
RIPA buffer (Beyotime Institute of Biotechnology)
was used to extract the total protein from HEC-1A and RL95-2 cells
in the Ctrl, siNC, siSLCO4C1-1, siSLCO4C1-2, siSLCO4C1-1+740 Y-P
and siSLCO4C1-2+740 Y-P groups. The concentration of the extracted
protein was determined using BCA Protein Assay Kit (Thermo Fisher
Scientific, Inc.). The protein samples were mixed with loading
buffer and then boiled for 10 min. Total 20 µg of protein were
loaded onto each lane of an 10% SDS-PAGE gel for protein
separation, and transferred onto polyvinylidene fluoride membranes
(EMD Millipore). The membranes were blocked with 5% bovine serum
albumin for 2 h and incubated with antibodies against SLCO4C1 (cat.
no. 24584-1-AP; 1:600 dilution; ProteinTech Group, Inc.), GAPDH
(product code ab9485; 1:2,500 dilution; Abcam), E-cadherin (product
no. 3195; 1:1,000 dilution), vimentin (product no. 5741; 1:1,000
dilution) and N-cadherin (product no. 13116; 1:1,000 dilution; all
from Cell Signaling Technology, Inc.), Ki67 (product code ab243878;
1:1,000 dilution) and cleaved caspase-3 (product code ab2302;
1:1,000 dilution) and cleaved caspase-9 (product code ab32539;
1:1,000 dilution; all from Abcam), p-PI3K (product no. 4228;
1:1,000 dilution), PI3K (product no. 4257; 1:1,000 dilution), p-Akt
(product no. 4060; 1:1,000 dilution) and Akt (product no. 9272;
1:1,000 dilution; all from Cell Signaling Technology, Inc.) at 4°C
overnight. The membranes were incubated with peroxidase-linked
secondary anti-rabbit antibodies (product no. 7074; 1:2,000
dilution; Cell Signaling Technology, Inc.) for 1 h at room
temperature to detect the bound primary antibodies, and the blotted
proteins were visualized using an enhanced chemiluminescence kit
(Thermo Fisher Scientific, Inc.). The intensity of the protein
bands was quantified using ImageJ software (v1.8.0; National
Institutes of Health). The relative expression of target proteins
was described as a ratio relative to the expression of GAPDH, and
statistical data from at least three experiments were graphed.
Statistical analysis
In the present study, SPSS 19.0 (IBM Corp.) was used
for all statistical analysis, and images were edited using GraphPad
Prism 6.0 version (GraphPad Software, Inc.). A Student's t-test was
used for pairwise comparisons of differences, whereas one-way
analysis of variance (ANOVA) and Tukey's post hoc test were used to
analyze the difference among groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
Expression of DEGs in EC based on TCGA
datasets
The TCGA RNA-seq dataset of EC patients was
selected, and DEGs were analyzed using Deseq2. First, the RNA-seq
was analyzed to perform the volcano plot. Analysis of RNA-seq
expression profiling data from the TCGA patient cohort was plotted
into upregulated (red dots) or downregulated genes (blue dots) to
compare EC to normal cohorts, as revealed in Fig. 1A. Subsequently, 6,634 upregulated
and 4,623 downregulated genes were examined, and their expression
levels were determined and presented in a heatmap; it was revealed
that these genes were well clustered into the non-endometrial and
EC tissues. The significantly altered genes (fold change ≥2 or ≤-2
and P<0.01) were defined as DEGs (Fig. 1B). The prognostic value and clinical
stage of these DEGs in EC were analyzed using ggsurv R package and
Spearman's rank correlation coefficient, respectively, in order to
identify the molecular signatures affecting the survival time and
status of EC. In addition, Venn analysis was used to obtain the
intersection of the DEG profiles, as revealed in Fig. 1C. A total of 57 DEGs were revealed
to be strongly associated with survival time and clinical stage in
EC.
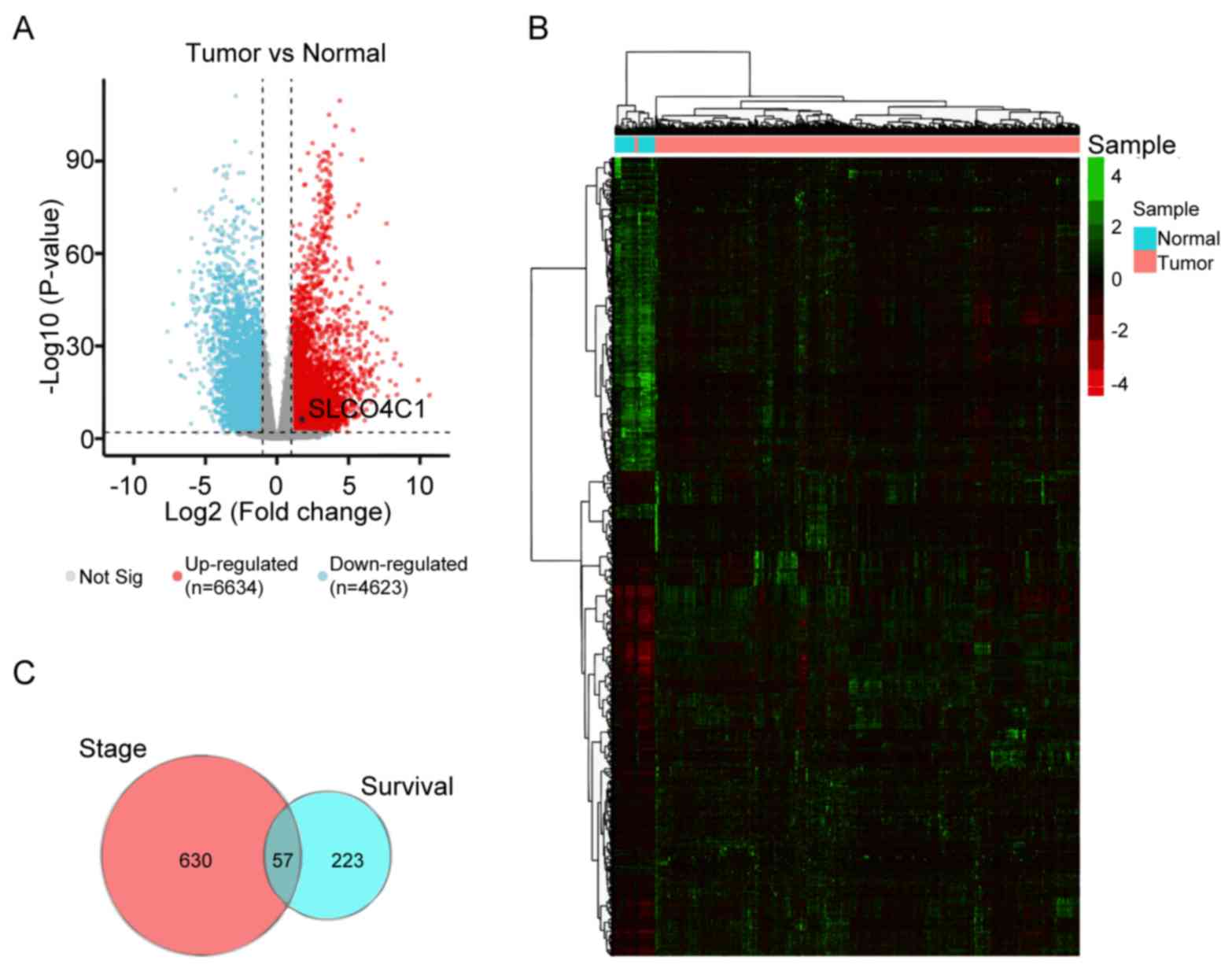 | Figure 1.DEG expression in EC based on TCGA
datasets. (A) Volcano plot showing the gene expression differences
between normal and EC patient cohorts. Blue dots, downregulated
genes. Red dots, upregulated genes. Black dot, SLCO4C1. (B) Heatmap
of the expression of 6,634 upregulated and 4,623 downregulated
genes, when comparing normal to EC tissues from TCGA database. (C)
Venn analysis of 57 overlapping genes associated with clinical
stage and survival time. DEG, differentially expressed genes; EC,
endometrial cancer; TCGA, The Cancer Genome Atlas; SLCO4C1, solute
carrier organic anion transporter family member 4C1. |
Identifying the specific gene
signatures and the key SLCO4C1 gene
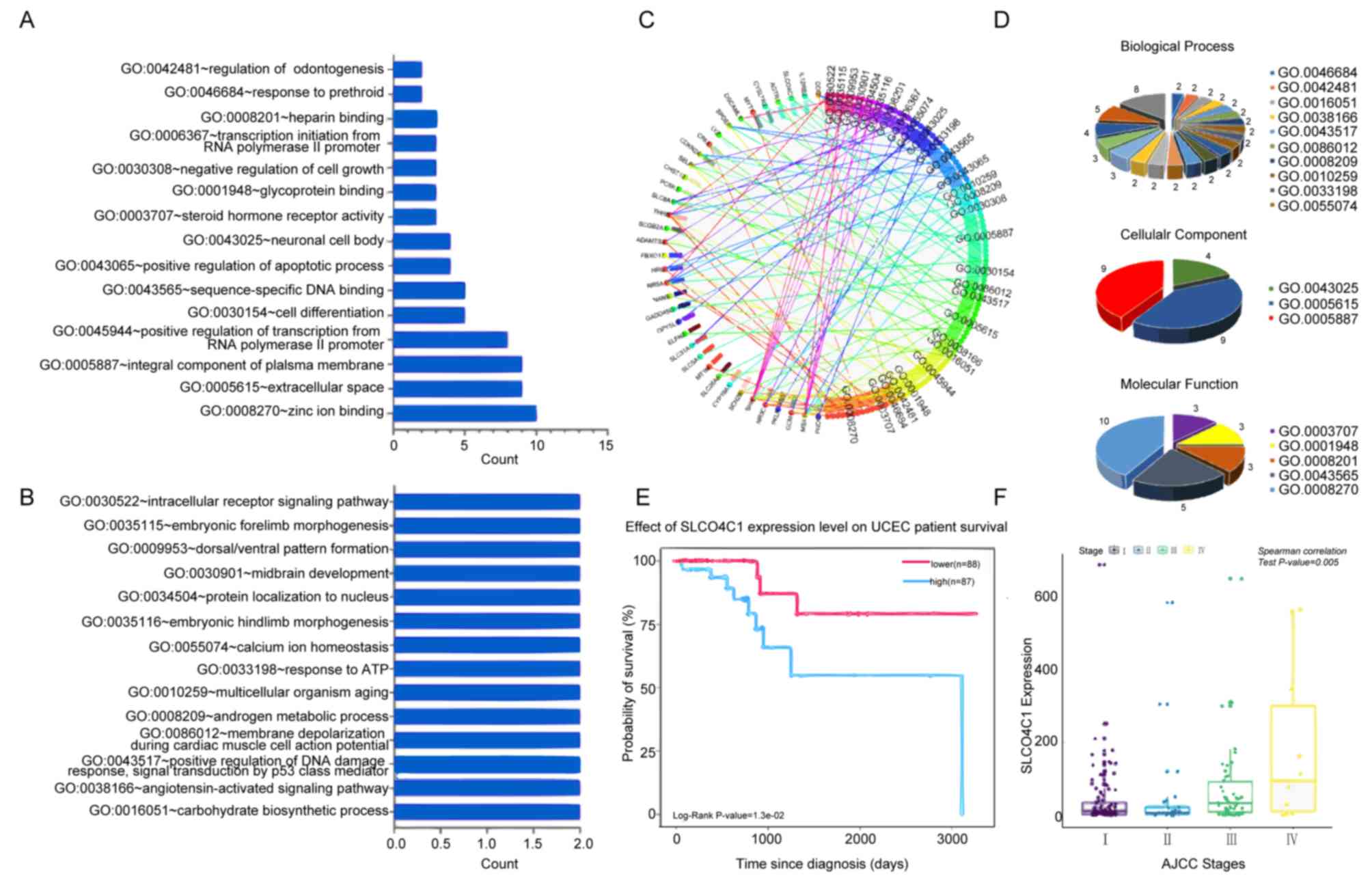
To predict the functional category of these 57 DEGs,
GO analysis was performed and it was revealed that 57 DEGs were
associated with various biological processes, molecular functions
and cellular components. Among them, 37 DEGs were found to mainly
participate in 18 significant GOs (P<0.05; Fig. 2A-D). Specific genes were revealed to
participate in zinc ion binding, integral component of plasma
membrane, extracellular space, positive transcriptional regulation
promoted by RNA polymerase II, sequence-specific DNA binding and
cell differentiation. Notably, SLCO4C1 was significantly highly
expressed in 2 terms, including integral component of plasma
membrane and cell differentiation, which was correlated with poor
prognosis and advanced clinical stage in EC (Fig. 2E and F). Thus, the function of
SLCO4C1 and its molecular mechanism were next explored.
SLCO4C1 is highly expressed in EC
tissues
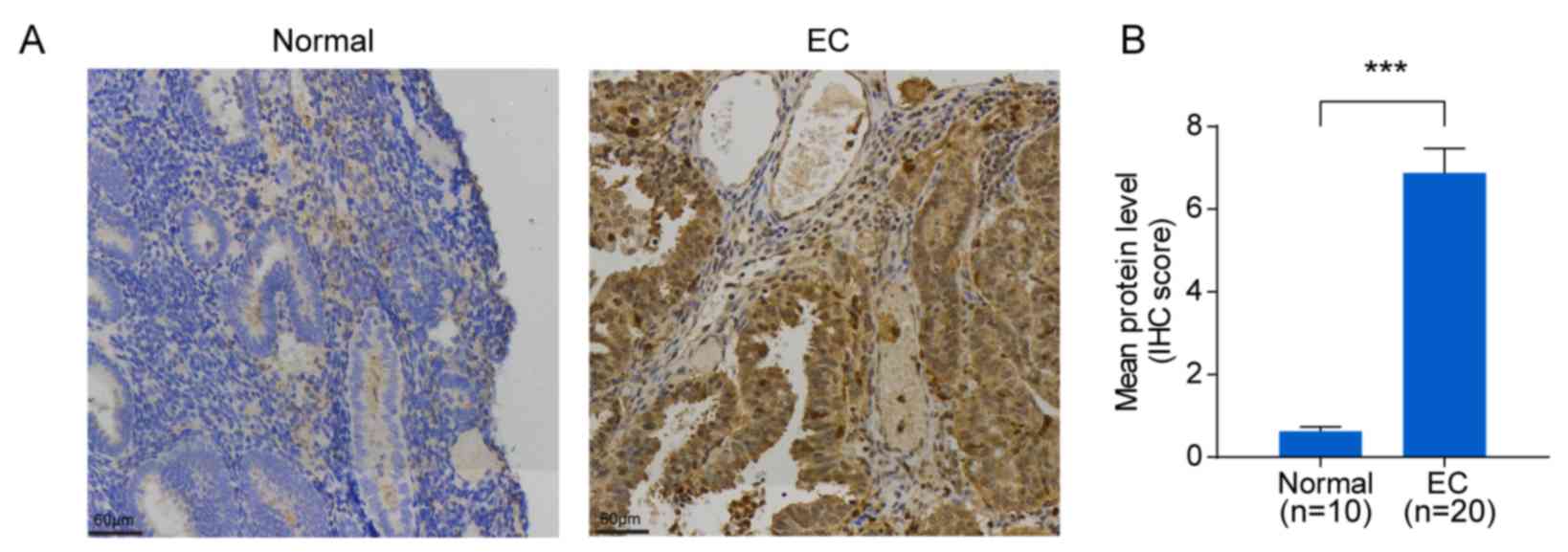
To verify SLCO4C1 expression in EC tissues,
immunochemistry was carried out in EC (n=20) and normal endometrium
(n=10) tissues. Immunochemistry revealed that the SLCO4C1 protein
was predominantly localized in the membrane and cytoplasm of
endometrial epithelial cells. Only weak or no staining was observed
in normal endometria, whereas strong SLCO4C1 immunostaining was
observed in EC tissues (P<0.001; Fig. 3). These data indicated that SLCO4C1
expression was significantly higher in EC tissues.
SLCO4C1 downregulation inhibits
proliferation and sphere formation, and induces apoptosis in HEC-1A
and RL95-2 cells
A high SLCO4C1 expression was detected in the EC
cell lines, as verified by RT-qPCR and western blotting (Fig. 4A and B). HEC-1A and RL95-2 cells
with a relatively higher SLCO4C1 expression were used for
subsequent experiments. siSLCO4C1-1 and siSLCO4C1-2 were
successfully transfected into HEC-1A and RL95-2 cells, respectively
(Fig. 4C and D). To examine whether
SLCO4C1 would affect cell proliferation, apoptosis and sphere
formation in EC, a CCK-8 assay, flow cytometry and a sphere
formation assay were performed. As revealed in Fig. 4E, the proliferation of HEC-1A and
RL95-2 cells was significantly decreased by SLCO4C1 interference at
72 h. Concurrently, HEC-1A and RL95-2 cells transfected with
siSLCO4C1-1 and siSLCO4C1-2 were associated with increased
apoptosis, compared to Ctrl and siNC cells (Fig. 4F). The downregulation of SLCO4C1
could inhibit the sphere forming ability of HEC-1A and RL95-2 cells
while suspending their growth (Fig.
4G). In addition, the knockdown of SLCO4C1 significantly
downregulated the expression of proliferation-related protein Ki67
and enhanced the expression of apoptotic-related protein cleaved
caspase-3/9, as revealed by western blotting (Fig. 4H).
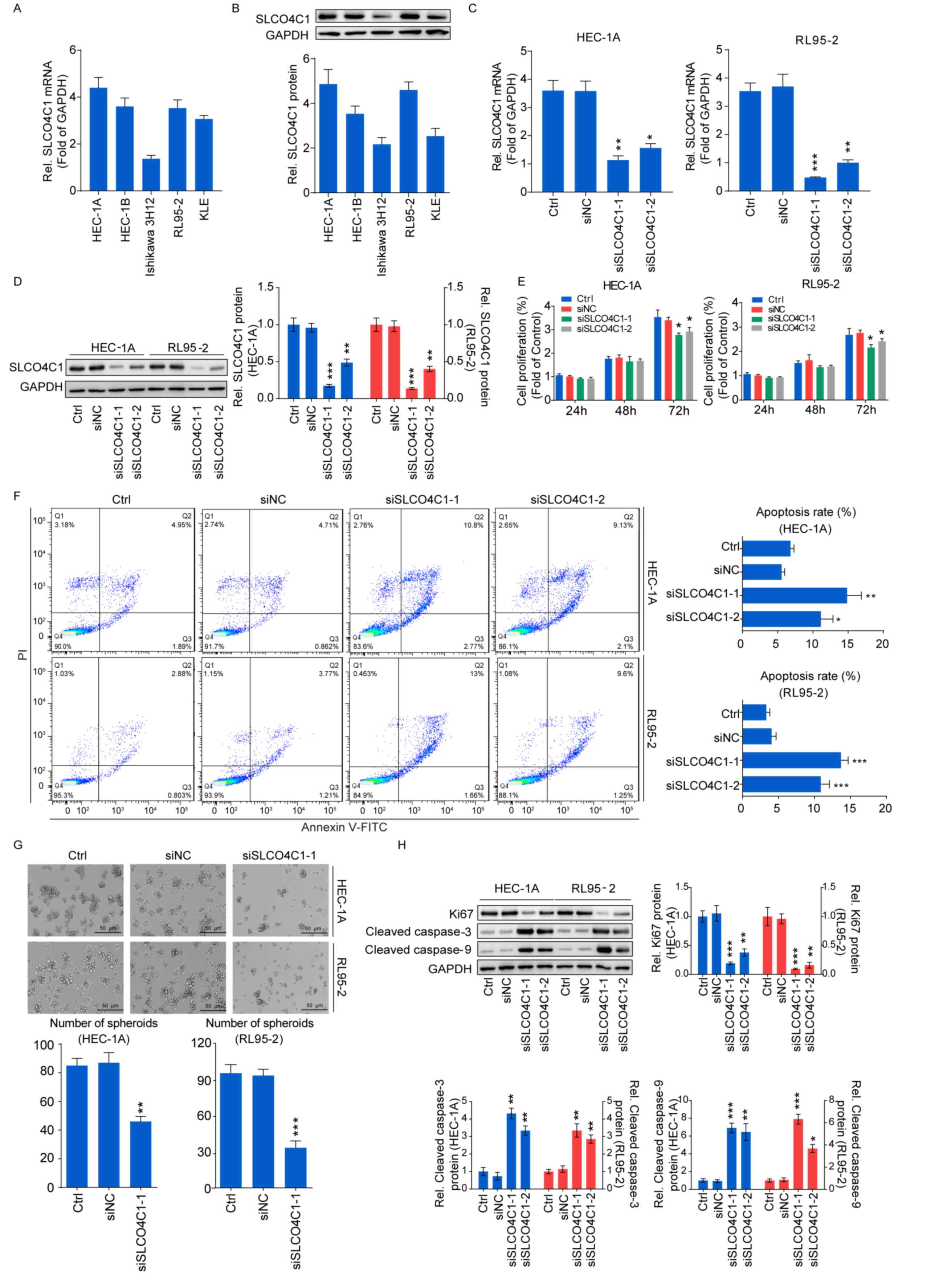 | Figure 4.Downregulation of SLCO4C1 affects the
proliferation, apoptosis and sphere forming abilities of HEC-1A and
RL95-2 cells. (A) The expression of SLCO4C1 in EC cell lines was
detected by RT-qPCR. (B) The expression of SLCO4C1 in EC cell lines
was detected by western blotting. (C) The expression of SLCO4C1 in
HEC-1A and RL95-2 cells transfected with siNC, siSLCO4C1-1 or
siSLCO4C1-2 was detected by RT-qPCR analysis. (D) The expression of
SLCO4C1 in HEC-1A and RL95-2 cells transfected with siNC,
siSLCO4C1-1 or siSLCO4C1-2 was detected by western blotting. (E)
Cell proliferation was detected by CCK-8 assay in HEC-1A and RL95-2
cells transfected with siNC, siSLCO4C1-1 or siSLCO4C1-2. (F)
Apoptosis analysis of HEC-1A and RL95-2 cells transfected with
siNC, siSLCO4C1-1 or siSLCO4C1-2 by flow cytometry. (G) Sphere
forming ability of HEC-1A and RL95-2 cells transfected with siNC or
siSLCO4C1-1 by sphere-formation assay. (H) Expression of
proliferation-related protein Ki67 and apoptotic-related proteins
cleaved caspase-3/9 in HEC-1A and RL95-2 cells transfected with
siNC, siSLCO4C1-1 or siSLCO4C1-2 by western blotting. GAPDH was
used as a loading control. The results were determined from
triplicate experiments and the error bars represent the mean ± SD.
*P<0.05, **P<0.01, ***P<0.001 vs. the Ctrl group and siNC
group. SLCO4C1, solute carrier organic anion transporter family
member 4C1; EC, endometrial cancer. |
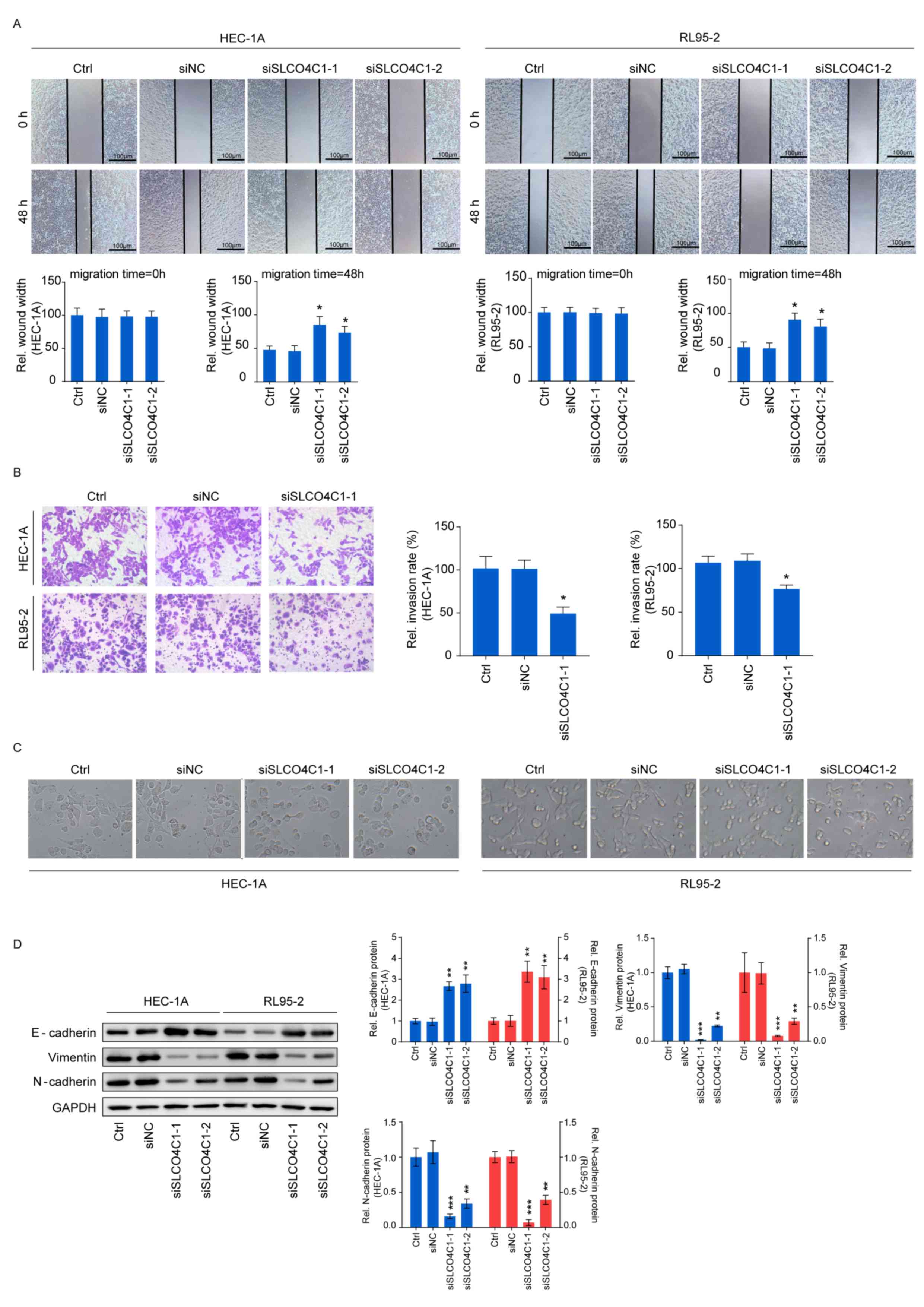
Downregulation of SLCO4C1 suppresses
the migration and invasion capacities of HEC-1A and RL95-2 cells
and affects the epithelial-mesenchymal transition (EMT) phenotype
in EC cells
Wound healing and Transwell assays were carried out
in order to determine cell migration and invasion, thus further
exploring the influence of SLCO4C1 on the behavior of EC cells. As
revealed in Fig. 5A and B, the
migration ability of HEC-1A and RL95-2 cells was significantly
reduced by SLCO4C1 interference at 48 h, while these cells
transfected with siSLCO4C1-1 displayed a markedly decreased
invasion ability, compared to Ctrl and siNC cells. The functions of
SLCO4C1 in migration and invasion prompted us to examine whether
SLCO4C1 knockdown could induce morphological changes. siSLCO4C1-1
and siSLCO4C1-2 cells were morphologically transformed toward
epithelial, compared to Ctrl and siNC cells (Fig. 5C). This change was characterized by
a transition from the spindle-like morphology of the parental cells
to a more differentiated keratinocyte-like morphology, indicating a
phenotypic transition from mesenchymal to epithelial. Processes
involved in EMT are closely correlated with cancer metastasis. To
further determine if this transformation represented an EMT, the
levels of several characteristic epithelial and mesenchymal
proteins were analyzed by western blotting in Ctrl, siNC,
siSLCO4C1-1 and siSLCO4C1-2 cells. As revealed in Fig. 5D, the expression of E-cadherin was
significantly upregulated while that of vimentin and N-cadherin was
significantly downregulated in HEC-1A and RL95-2 cells transfected
with siSLCO4C1-1 and siSLCO4C1-2, compared to Ctrl and siNC cells.
Collectively, these findings indicated that SLCO4C1 knockdown was
accompanied by the loss of mesenchymal and gain of epithelial
markers, and that SLCO4C1 is likely to play a crucial role in the
regulation of EMT and metastasis in EC cells.
SLCO4C1 regulates the behavior of EC
through the PI3K/Akt signaling pathway in HEC-1A and RL95-2
cells
To explore the molecular mechanism underlying
SLCO4C1, western blotting was performed in Ctrl, siNC, siSLCO4C1-1
and siSLCO4C1-2 cells, in order to detect the markers of the
PI3K/Akt signaling pathway. It was revealed that SLCO4C1
interference could significantly inactivate the PI3K/Akt signaling
pathway by decreasing the phosphorylated levels of PI3K and Akt
proteins (Fig. 6A). In order to
further verify the association between SLCO4C1 and the PI3K/Akt
signaling pathway, an equal number of siSLCO4C1-1 or siSLCO4C1-2
cells were treated with 740 Y-P (PI3K signaling activator) for a
rescue assay. The inhibition of proliferation caused by the
knockdown of SLCO4C1 was reversed following 740 Y-P stimulation
(Fig. 6B). The 740 Y-P treatment
also prevented the enhancement of apoptosis mediated by siSLCO4C1
in HEC-1A and RL95-2 cells (Fig.
6C). In addition, the reduction of migration mediated by
siSLCO4C1 was rescued with treatment of 740 Y-P (Fig. 6D). Furthermore, the PI3K/Akt
signaling pathway was rescued with treatment of 740 Y-P in the
presence of SLCO4C1 knockdown (Fig.
6E). These results indicated that SLCO4C1 played a pivotal role
in EC through the PI3K/Akt signaling pathway.
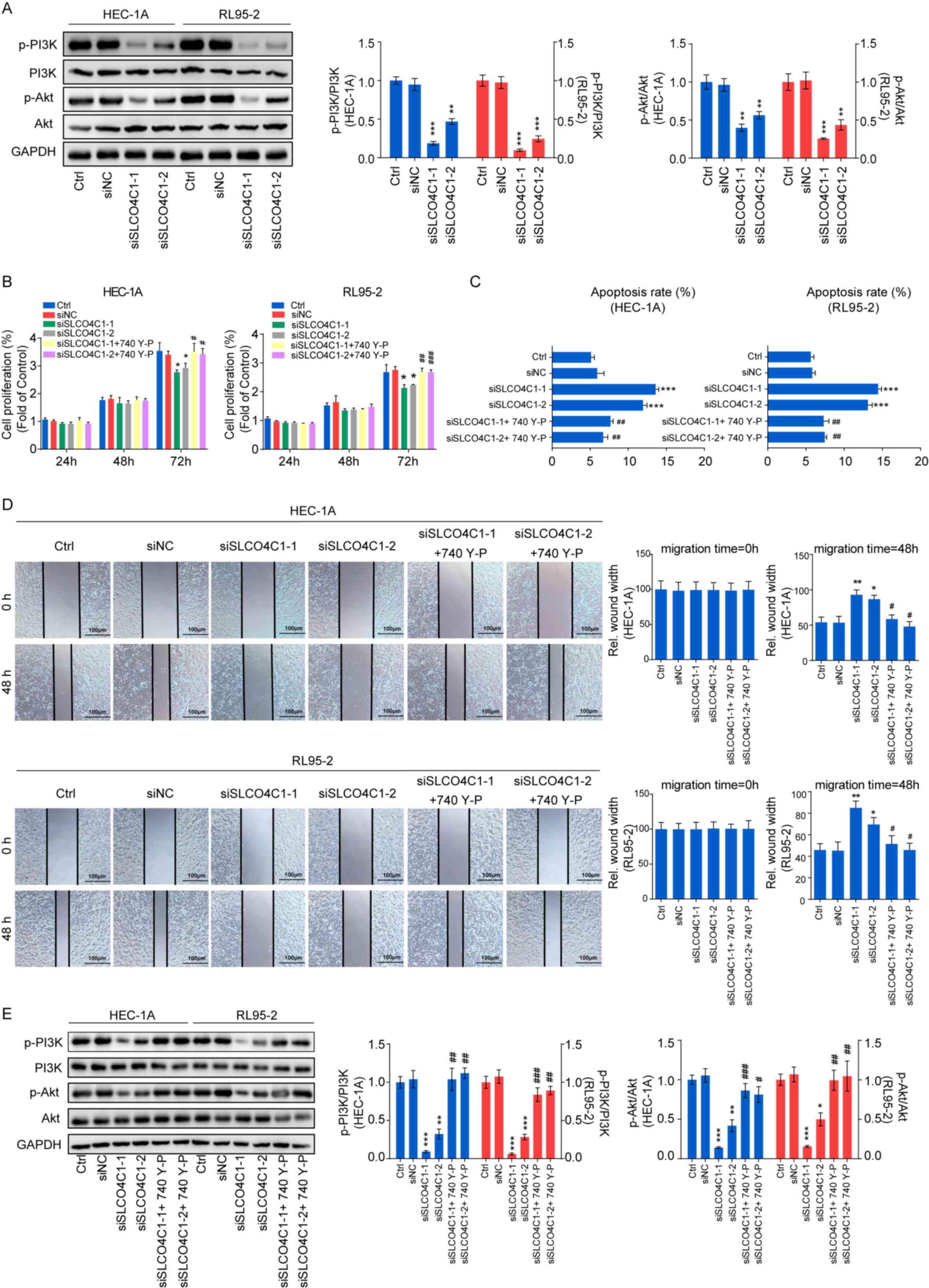 | Figure 6.Downregulation of SLCO4C1 regulates
the behavior of EC via the PI3K/Akt signaling pathway in HEC-1A and
RL95-2 cells. (A) Expression of p-PI3K, PI3K, p-Akt and Akt in
HEC-1A and RL95-2 cells from the Ctrl, siNC, siSLCO4C1-1 or
siSLCO4C1-2 groups, as determined by western blotting. (B) Cell
proliferation was detected by CCK-8 assay in HEC-1A and RL95-2
cells from the Ctrl, siNC, siSLCO4C1-1, siSLCO4C1-2,
siSLCO4C1-1+740 Y-P (PI3K signaling activator) or siSLCO4C1-2+740
Y-P groups. (C) Cell apoptosis was detected by flow cytometry in
HEC-1A and RL95-2 cells from the Ctrl, siNC, siSLCO4C1-1,
siSLCO4C1-2, siSLCO4C1-1+740 Y-P or siSLCO4C1-2+740 Y-P groups. (D)
Cell migration was detected by wound healing assay at 0 and 48 h in
HEC-1A and RL95-2 cells from the Ctrl, siNC, siSLCO4C1-1,
siSLCO4C1-2, siSLCO4C1-1+740 Y-P or siSLCO4C1-2+740 Y-P groups. (E)
Expression of p-PI3K, PI3K, p-Akt and Akt in HEC-1A and RL95-2
cells from the Ctrl, siNC, siSLCO4C1-1, siSLCO4C1-2,
siSLCO4C1-1+740 Y-P or siSLCO4C1-2+740 Y-P groups, as determined by
western blotting. GAPDH was used as a loading control. The results
were determined from triplicate experiments and the error bars
represent the mean ± SD. Asterisks indicated a difference between
siSLCO4C1 and siNC, whereas hashtags indicated a difference between
siSLCO4C1+740 Y-P and siSLCO4C1. */#P<0.05,
**/##P<0.01, ***/###P<0.001 vs. the
Ctrl group and siNC group. SLCO4C1, solute carrier organic anion
transporter family member 4C1; EC, endometrial cancer; p-PI3K,
phosphorylated-PI3K; p-Akt, phosphorylated-Akt. |
Discussion
Tumorigenesis and cancer development can be
regulated by multiple genes, environmental factors and pathways in
multiple ways. Certain genes have formed a complex regulatory
network to induce pathogenesis (16). Each cancer type possesses a spectrum
of tumor-specific molecular characteristics, including somatic
mutations, copy number variations, changes in gene expression
profiles and epigenetic changes. The life science field has entered
the era of big data, which is characterized by mass multivariate
omics data (17,18). In the present study, the newly
sorted EC mRNA expression data, along with the corresponding
clinical information, was downloaded from TCGA database, and 6,634
upregulated and 4,623 downregulated genes were detected in
endometrial samples. According to the TCGA database and GO
analysis, 57 DEGs were revealed to be associated with the survival
time and clinical stage of tumor patients, and SLCO4C1 was revealed
to be involved in cell differentiation and the composition of the
cell membrane. Moreover, it has been reported in literature to be
closely correlated with kidney disease, head and neck cancer and
reproductive development (9–13).
Therefore, the association of SLCO4C1 with EC was specifically
investigated in the present study. Bioinformatics analysis results
indicated that SLCO4C1 was highly expressed and the survival rate
was reduced, accompanied by cancer procession. A high SLCO4C1
expression was also detected in EC tissues and cell lines.
Consequently, the SLCO4C1 expression in HEC-1A and
RL95-2 cells was knocked down through siRNA transfection. In
addition, proliferation, apoptosis, migration and invasion were
detected by CCK-8, flow cytometry, and sphere-formation, wound
healing and Transwell assays. The present results revealed that
SLCO4C1 knockdown could suppress cell viability and promote
apoptosis. It could also reduce the sphere formation, migration and
invasion abilities of cells, indicating that SLCO4C1 could actually
affect the biological behaviors of EC.
Cell proliferation and apoptosis are dynamically
balanced processes, and an imbalance between them leads to the
malignant transformation of cells and tumorigenesis (19). Ki67 is a proliferation-related
protein; cleaved caspase-9 is the apoptotic initiator and cleaved
caspase-3 is the executor of apoptosis (20,21).
The present findings indicated that, following the knockdown of
SLCO4C1, proliferation was inhibited and apoptosis was induced.
Ki67 expression in cells was significantly downregulated, while
cleaved caspase-3/9 expression was significantly upregulated,
indicating that SLCO4C1 participated in the dynamic balance between
cell proliferation and apoptosis in EC.
Cancer cells that disseminate from the primary tumor
and invade distant organs are the leading causes of mortality in
EC. EMT has been revealed to be of vital importance in the early
events of cancer cell metastatic dissemination by endowing cells
with a more motile, invasive potential (22). E-cadherin, vimentin and N-cadherin
are the recognized molecular markers participating in EMT (23). During migration and invasion, the
expression of E-cadherin is reduced while that of vimentin and
N-cadherin is increased (24). The
present study demonstrated that a reduction in SLCO4C1 expression
could suppress migration and invasion, increase E-cadherin
expression and reduce vimentin and N-cadherin expression,
indicating that SLCO4C1 was responsible for the EMT process.
The associated molecular mechanism was further
explored. Biological behaviors of tumor cells such as
proliferation, apoptosis, migration and invasion are a result of
the joint effect of multiple genes, and certain key proteins have
been revealed to play crucial regulatory roles during these
processes (25). The PI3K/Akt
signaling pathway has been extensively identified among cancer
cells and is an important signaling pathway involved in cancer cell
proliferation, apoptosis, migration and invasion (25,26).
Previous studies have identified that the activation of the
PI3K/Akt signaling pathway is involved in EMT and processes of
tumor progression and metastasis (26,27).
PI3K-activated products can bind to the Akt PH domain, which can
not only mediate the membrane translocation of Akt from the
cytoplasm, but also promote its conformational change. Typically,
such a process can expose the Ser473 and Thr308 phosphorylation
sites of Akt, thus activating Akt (28,29).
The activated Akt can further regulate cancer cell proliferation,
apoptosis, migration and invasion (30). The present study revealed that
SLCO4C1 knockdown could markedly suppress the phosphorylation
levels of PI3K and Akt, indicating that decreasing SLCO4C1
expression could inactivate the signaling pathway. The rescue
experiments with PI3K signaling activator 740 Y-P verified that the
PI3K/Akt signaling pathway was essential in tumor progression and
EMT mediated by SLCO4C1. Therefore, the present findings were the
first, to the best of our knowledge, to demonstrate the role of
SLCO4C1 interference in affecting apoptosis, proliferation,
migration, invasion and the EMT phenotype in EC cells by
alleviating the activation of the PI3K/Akt signaling pathway.
In conclusion, the present study revealed that
SLCO4C1 is highly expressed in EC tissues, as compared to normal
tissues. In addition, SLCO4C1 interference could promote apoptosis
and suppress cell proliferation and metastasis in EC cells in
vitro by inactivating the PI3K/Akt signaling pathway.
Therefore, clinical diagnosis may benefit from SLCO4C1 assessment
and the SLCO4C1/PI3K/Akt signaling pathway could serve as a
potential therapeutic target for the treatment of EC.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant nos. 81472427, 81672574
and 81702547), the Shanghai Sailing Program (grant no.
17YF1415300), the Shanghai New Frontier Technology Project (grant
no. SHDC12015110) and the Shanghai Municipal Medical and Health
Discipline Construction Projects (grant no. 2017ZZ02015).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XH and YB wrote the original manuscript and analyzed
the data. XH and TH undertook most of the experiments, manuscript
rewriting or revising as well as figure and data processing. XH,
TH, YB, HT and XWe performed the experiments and data analysis. YL
and XWa designed the study and revised the manuscript critically
for important intellectual content. All authors read and approved
the manuscript and agree to be accountable for all aspects of the
research in ensuring that the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
This research was approved by the Human
Investigation Ethics Committee of the Shanghai First Maternity and
Infant Hospital. Samples were collected from patients after
receiving their written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mazzocca A, Schönauer LM, De Nola R,
Lippolis A, Marrano T, Loverro M, Sabbà C and Di Naro E: Autotaxin
is a novel molecular identifier of type I endometrial cancer. Med
Oncol. 35:1572018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Setiawan VW, Yang HP, Pike MC, McCann SE,
Yu H, Xiang YB, Wolk A, Wentzensen N, Weiss NS, Webb PM, et al:
Type I and II endometrial cancers: Have they different risk
factors? J Clin Oncol. 31:2607–2618. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhou M, Liu C, Cao G, Gao H and Zhang Z:
Expression of polymeric immunoglobulin receptor and its biological
function in endometrial adenocarcinoma. J Cancer Res Ther.
15:420–425. 2019.PubMed/NCBI
|
|
4
|
Schlumbrecht M, Baeker Bispo JA, Balise
RR, Huang M, Slomovitz B and Kobetz E: Variation in type II
endometrial cancer risk by Hispanic subpopulation: An exploratory
analysis. Gynecol Oncol. 147:329–333. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kiyohara T, Nakamaru S, Miyamoto M,
Shijimaya T, Nagano N, Makimura K and Tanimura H: Metaplastic
rectal adenocarcinoma into metastatic mucinous carcinoma on the
buttock: The efficacy of immunohistochemistry. J Dermatol.
46:e337–e339. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Luo C, Cen S, Ding G and Wu W: Mucinous
colorectal adenocarcinoma: Clinical pathology and treatment
options. Cancer Commun (Lond). 39:132019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Maeda H, Yokomizo H, Okayama S, Yamada Y,
Ida A, Satake M, Yano Y, Asaka S, Usui T, Shiozawa S, et al: A case
of low-grade appendiceal mucinous neoplasm with cecum cancer. Gan
To Kagaku Ryoho. 46:518–520. 2019.(In Japanese). PubMed/NCBI
|
|
8
|
Pan J and Fan Z: Mucinous adenocarcinoma
of the abdomen. Int Wound J. 16:1045–1046. 2019.PubMed/NCBI
|
|
9
|
Jang H, Choi Y, Yoo I, Han J, Kim M and Ka
H: Expression and regulation of prostaglandin transporters,
ATP-binding cassette, subfamily C, member 1 and 9, and solute
carrier organic anion transporter family, member 2A1 and 5A1 in the
uterine endometrium during the estrous cycle and pregnancy in pigs.
Asian-Australas J Anim Sci. 30:643–652. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Taghikhani E, Maas R, Fromm MF and Konig
J: The renal transport protein OATP4C1 mediates uptake of the
uremic toxin asymmetric dimethylarginine (ADMA) and efflux of
cardioprotective L-homoarginine. PLoS One. 14:e02137472019.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Suzuki T, Toyohara T, Akiyama Y, Takeuchi
Y, Mishima E, Suzuki C, Ito S, Soga T and Abe T: Transcriptional
regulation of organic anion transporting polypeptide SLCO4C1 as a
new therapeutic modality to prevent chronic kidney disease. J Pharm
Sci. 100:3696–3707. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Toyohara T, Suzuki T, Morimoto R, Akiyama
Y, Souma T, Shiwaku HO, Takeuchi Y, Mishima E, Abe M, Tanemoto M,
et al: SLCO4C1 transporter eliminates uremic toxins and attenuates
hypertension and renal inflammation. J Am Soc Nephrol.
20:2546–2555. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Guerrero-Preston R, Michailidi C,
Marchionni L, Pickering CR, Frederick MJ, Myers JN,
Yegnasubramanian S, Hadar T, Noordhuis MG, Zizkova V, et al: Key
tumor suppressor genes inactivated by ‘greater promoter’
methylation and somatic mutations in head and neck cancer.
Epigenetics. 9:1031–1046. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li LY, Yin KM, Bai YH, Zhang ZG, Di W and
Zhang S: CTHRC1 promotes M2-like macrophage recruitment and
myometrial invasion in endometrial carcinoma by integrin-Akt
signaling pathway. Clin Exp Metastasis. 36:351–363. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Boyango I, Barash U, Fux L, Naroditsky I,
Ilan N and Vlodavsky I: Targeting heparanase to the mammary
epithelium enhances mammary gland development and promotes tumor
growth and metastasis. Matrix Biol. 65:91–103. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li X, Li MW, Zhang YN and Xu HM: Common
cancer genetic analysis methods and application study based on TCGA
database. Yi Chuan. 41:234–242. 2019.PubMed/NCBI
|
|
18
|
Liu Y, Nan F, Lu K and Wang Y, Liu Y, Wei
S, Wu R and Wang Y: Identification of key genes in endometrioid
endometrial adenocarcinoma via TCGA database. Cancer Biomark.
21:11–21. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Çıkla-Süzgün P and Küçükgüzel ŞG: Recent
advances in apoptosis: The role of hydrazones. Mini Rev Med Chem.
19:1427–1442. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wan J, Zou S, Hu M, Zhu R, Xu J, Jiao Y
and Fan S: Thoc1 inhibits cell growth via induction of cell cycle
arrest and apoptosis in lung cancer cells. Mol Med Rep.
9:2321–2327. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rivera M, Ramos Y, Rodríguez-Valentín M,
López-Acevedo S, Cubano LA, Zou J, Zhang Q, Wang G and Boukli NM:
Targeting multiple pro-apoptotic signaling pathways with curcumin
in prostate cancer cells. PLoS One. 12:e01795872017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Baek SH, Ko JH, Lee JH, Kim C, Lee H, Nam
D, Lee J, Lee SG, Yang WM, Um JY, et al: Ginkgolic acid inhibits
invasion and migration and TGF-β-induced EMT of lung cancer cells
through PI3K/Akt/mTOR inactivation. J Cell Physiol. 232:346–354.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shen M, Xu Z, Xu W, Jiang K, Zhang F, Ding
Q, Xu Z and Chen Y: Inhibition of ATM reverses EMT and decreases
metastatic potential of cisplatin-resistant lung cancer cells
through JAK/STAT3/PD-L1 pathway. J Exp Clin Cancer Res. 38:1492019.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lee GA, Hwang KA and Choi KC: Roles of
dietary phytoestrogens on the regulation of epithelial-mesenchymal
transition in diverse cancer metastasis. Toxins (Basel).
8:E1622016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li Y, Xiao X, Bossé Y, Gorlova O, Gorlov
I, Han Y, Byun J, Leighl N, Johansen JS, Barnett M, et al: Genetic
interaction analysis among oncogenesis-related genes revealed novel
genes and networks in lung cancer development. Oncotarget.
10:1760–1774. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lv Q, Wang G, Zhang Y, Han X, Li H, Le W,
Zhang M, Ma C, Wang P and Ding Q: FABP5 regulates the proliferation
of clear cell renal cell carcinoma cells via the PI3K/AKT signaling
pathway. Int J Oncol. 54:1221–1232. 2019.PubMed/NCBI
|
|
27
|
Sharma N, Nanta R, Sharma J, Gunewardena
S, Singh KP, Shankar S and Srivastava RK: PI3K/AKT/mTOR and sonic
hedgehog pathways cooperate together to inhibit human pancreatic
cancer stem cell characteristics and tumor growth. Oncotarget.
6:32039–32060. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Haodang L, Lianmei Q, Ranhui L, Liesong C,
Jun H, Yihua Z, Cuiming Z, Yimou W and Xiaoxing Y: HO-1 mediates
the anti-inflammatory actions of Sulforaphane in monocytes
stimulated with a mycoplasmal lipopeptide. Chem Biol Interact.
306:10–18. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zong HY, Wang EL, Han YM, Wang QJ, Wang JL
and Wang Z: Effect of miR-29b on rats with gestational diabetes
mellitus by targeting PI3K/Akt signal. Eur Rev Med Pharmacol Sci.
23:2325–2331. 2019.PubMed/NCBI
|
|
30
|
Zhang W, Liang X, Gong Y, Xiao C, Guo B
and Yang T: The signal transducer and activator of transcription 5B
(STAT5B) gene promotes proliferation and drug resistance of human
mantle cell lymphoma cells by activating the Akt signaling pathway.
Med Sci Monit. 25:2599–2608. 2019. View Article : Google Scholar : PubMed/NCBI
|