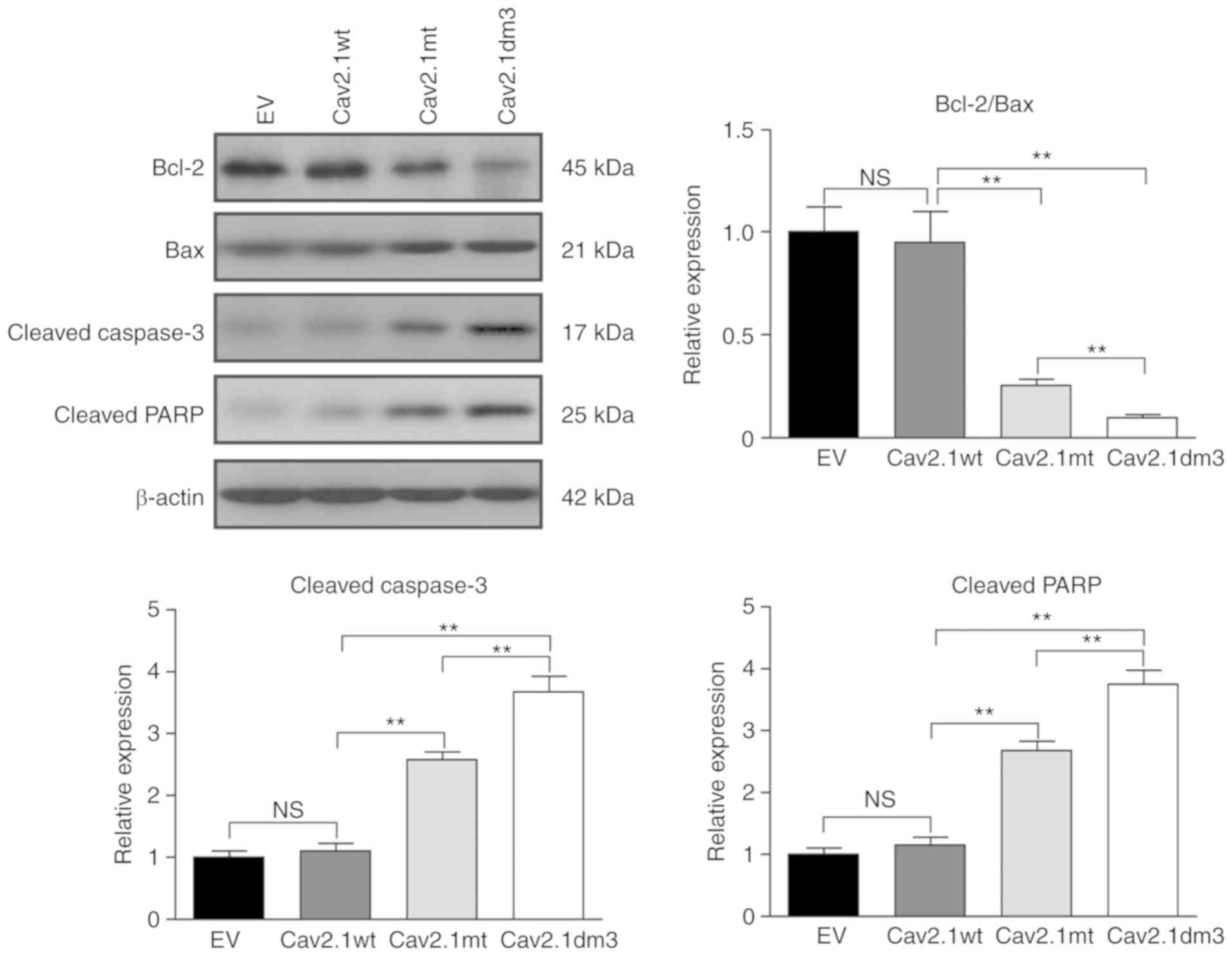
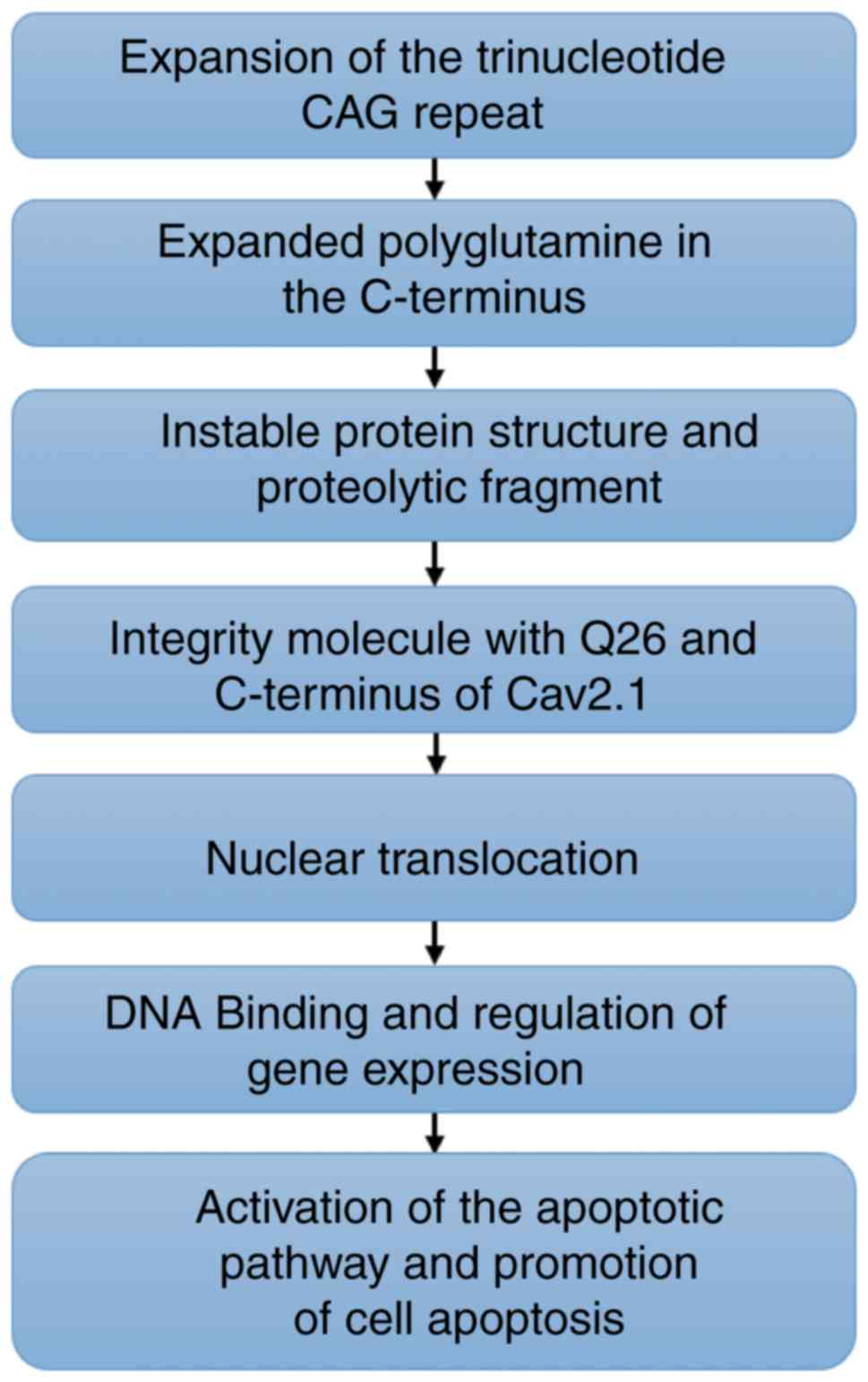
|
1
|
Ophoff RA, Terwindt GM, Vergouwe MN, van
Eijk R, Oefner PJ, Hoffman SM, Lamerdin JE, Mohrenweiser HW, Bulman
DE, Ferrari M, et al: Familial hemiplegic migraine and episodic
ataxia type-2 are caused by mutations in the Ca2+ channel gene
CACNL1A4. Cell. 87:543–552. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lea RA, Curtain RP, Hutchins C, Brimage PJ
and Griffiths LR: Investigation of the CACNA1A gene as a candidate
for typical migraine susceptibility. Am J Med Genet. 105:707–712.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Petrovicova A, Brozman M, Kurca E, Gobo T,
Dluha J, Kalmarova K, Nosal V, Hikkelova M, Krajciova A,
Burjanivova T and Sivak S: Novel missense variant of CACNA1A gene
in a Slovak family with episodic ataxia type 2. Biomed Pap Med Fac
Univ Palacky Olomouc Czech Repub. 161:107–110. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bavassano C, Eigentler A, Stanika R,
Obermair GJ, Boesch S, Dechant G and Nat R: Bicistronic CACNA1A
gene expression in neurons derived from spinocerebellar ataxia type
6 patient-induced pluripotent stem cells. Stem Cells Dev.
26:1612–1625. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Khaiboullina SF, Mendelevich EG, Shigapova
LH, Shagimardanova E, Gazizova G, Nikitin A, Martynova E, Davidyuk
YN, Bogdanov EI, Gusev O, et al: Cerebellar atrophy and changes in
cytokines associated with the CACNA1A R583Q mutation in a russian
familial hemiplegic migraine type 1 family. Front Cell Neurosci.
11:2632017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Grieco GS, Gagliardi S, Ricca I, Pansarasa
O, Neri M, Gualandi F, Nappi G, Ferlini A and Cereda C: New CACNA1A
deletions are associated to migraine phenotypes. J Headache Pain.
19:752018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lv Y, Wang Z, Liu C and Cui L:
Identification of a novel CACNA1A mutation in a Chinese family with
autosomal recessive progressive myoclonic epilepsy. Neuropsychiatr
Dis Treat. 13:2631–2636. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang Q, Chen ZS, An Y, Liu H, Hou Y, Li
W, Lau KF, Koon AC, Ngo JCK and Chan HYE: A peptidylic inhibitor
for neutralizing expanded CAG RNA-induced nucleolar stress in
polyglutamine diseases. RNA. 24:486–498. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Roshan R, Choudhary A, Bhambri A, Bakshi
B, Ghosh T and Pillai B: MicroRNA dysregulation in polyglutamine
toxicity of TATA-box binding protein is mediated through STAT1 in
mouse neuronal cells. J Neuroinflammation. 14:1552017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
von Niederhausern N, Ducray A, Zielinski
J, Murbach M and Mevissen M: Effects of radiofrequency
electromagnetic field exposure on neuronal differentiation and
mitochondrial function in SH-SY5Y cells. Toxicol In Vitro.
61:1046092019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Karikari TK, Nagel DA, Grainger A,
Clarke-Bland C, Crowe J, Hill EJ and Moffat KG: Distinct
conformations, aggregation and cellular internalization of
different tau strains. Front Cell Neurosci. 13:2962019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu M, Bai X, Yu S, Zhao W, Qiao J, Liu Y,
Zhao D, Wang J and Wang S: Ginsenoside Re inhibits ROS/ASK-1
dependent mitochondrial apoptosis pathway and activation of
Nrf2-Antioxidant response in beta-amyloid-challenged SH-SY5Y cells.
Molecules. 24:E26872019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kordasiewicz HB, Thompson RM, Clark HB and
Gomez CM: C-termini of P/Q-type Ca2+ channel alpha1A subunits
translocate to nuclei and promote polyglutamine-mediated toxicity.
Hum Mol Genet. 15:1587–1599. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsoi H, Lau TC, Tsang SY, Lau KF and Chan
HY: CAG expansion induces nucleolar stress in polyglutamine
diseases. Proc Natl Acad Sci USA. 109:13428–13433. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mizuguchi T, Suzuki T, Abe C, Umemura A,
Tokunaga K, Kawai Y, Nakamura M, Nagasaki M, Kinoshita K, Okamura
Y, et al: A 12-kb structural variation in progressive myoclonic
epilepsy was newly identified by long-read whole-genome sequencing.
J Hum Genet. 64:359–368. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tonin R, Catarzi S, Caciotti A, Procopio
E, Marini C, Guerrini R and Morrone A: Progressive myoclonus
epilepsy in gaucher disease due to a new Gly-Gly mutation causing
loss of an exonic splicing enhancer. J Neurol. 266:92–101. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
He J, Lin H, Li JJ, Su HZ, Wang DN, Lin Y,
Wang N and Chen WJ: Identification of a novel homozygous
splice-site mutation in SCARB2 that causes progressive myoclonus
epilepsy with or without renal failure. Chin Med J (Engl).
131:1575–1583. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Algahtani H, Shirah B, Algahtani R,
Al-Qahtani MH, Abdulkareem AA and Naseer MI: A novel mutation in
CACNA1A gene in a Saudi female with episodic ataxia type 2 with no
response to acetazolamide or 4-aminopyridine. Intractable Rare Dis
Res. 8:67–71. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gerhardstein BL, Gao T, Bunemann M, Puri
TS, Adair A, Ma H and Hosey MM: Proteolytic processing of the C
terminus of the alpha(1C) subunit of L-type calcium channels and
the role of a proline-rich domain in membrane tethering of
proteolytic fragments. J Biol Chem. 275:8556–8563. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gomez-Ospina N, Tsuruta F, Barreto-Chang
O, Hu L and Dolmetsch R: The C terminus of the L-type voltage-gated
calcium channel Ca(V)1.2 encodes a transcription factor. Cell.
127:591–606. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Park D, Kim SH, Lee YJ, Song GJ and Park
JS: A novel CACNA1A mutation associated with episodic ataxia 2
presenting with periodic paralysis. Acta Neurol Belg. 118:137–139.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Walker FO: Huntington's disease. Lancet.
369:218–228. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Miyata R, Hayashi M, Tanuma N, Shioda K,
Fukatsu R and Mizutani S: Oxidative stress in neurodegeneration in
dentatorubral-pallidoluysian atrophy. J Neurol Sci. 264:133–139.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sanchez I, Xu CJ, Juo P, Kakizaka A,
Blenis J and Yuan J: Caspase-8 is required for cell death induced
by expanded polyglutamine repeats. Neuron. 22:623–633. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tien CL, Wen FC and Hsieh M: The
polyglutamine-expanded protein ataxin-3 decreases bcl-2 mRNA
stability. Biochem Biophys Res Commun. 365:232–238. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Maiuri T, Suart CE, Hung CLK, Graham KJ,
Barba Bazan CA and Truant R: DNA damage repair in huntington's
disease and other neurodegenerative diseases. Neurotherapeutics.
16:948–956. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hwang JY, Park JH, Kim MJ, Kim WJ, Ha KT,
Choi BT, Lee SY and Shin HK: Isolinderalactone regulates the
BCL-2/caspase-3/PARP pathway and suppresses tumor growth in a human
glioblastoma multiforme xenograft mouse model. Cancer Lett.
443:25–33. 2019. View Article : Google Scholar : PubMed/NCBI
|