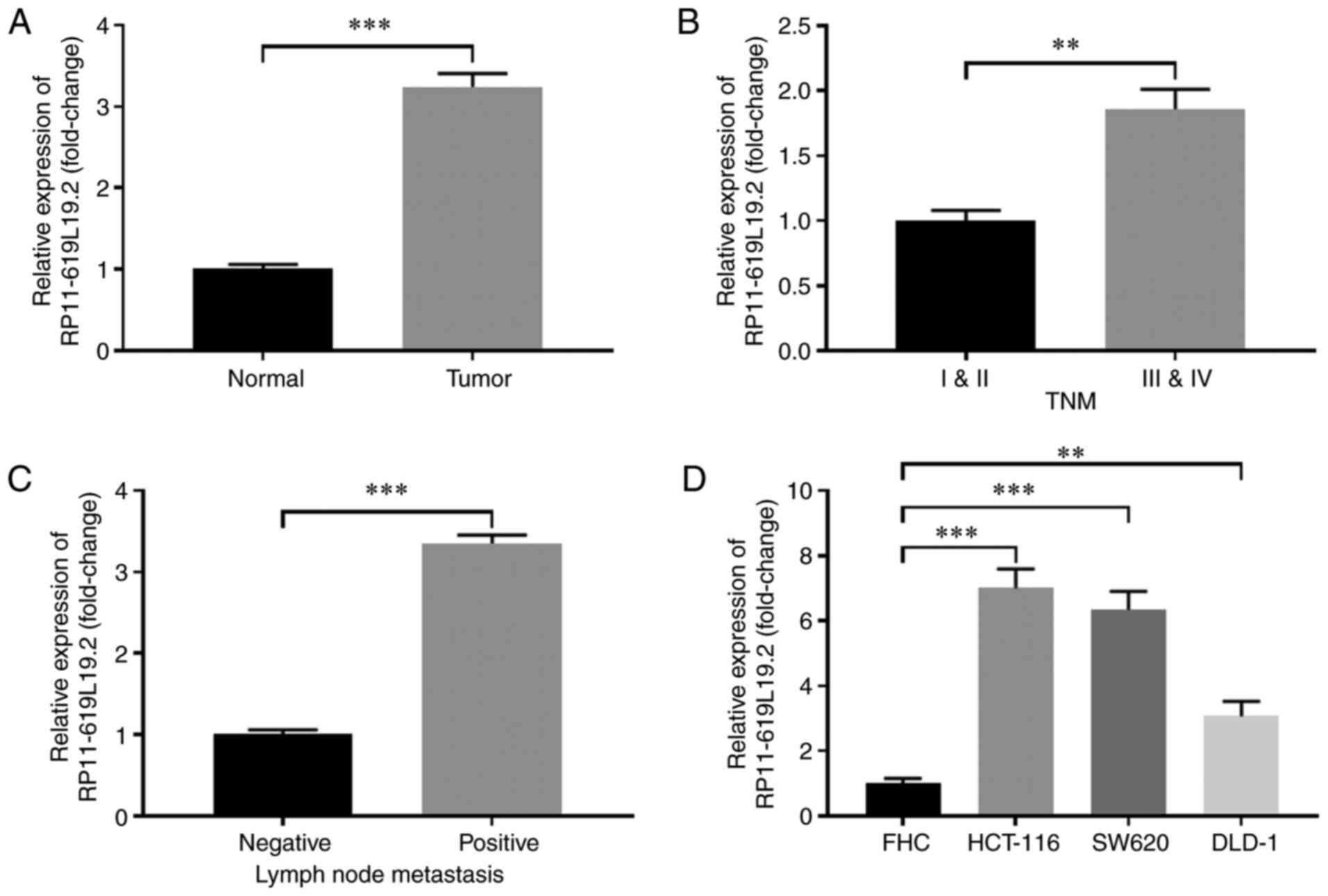
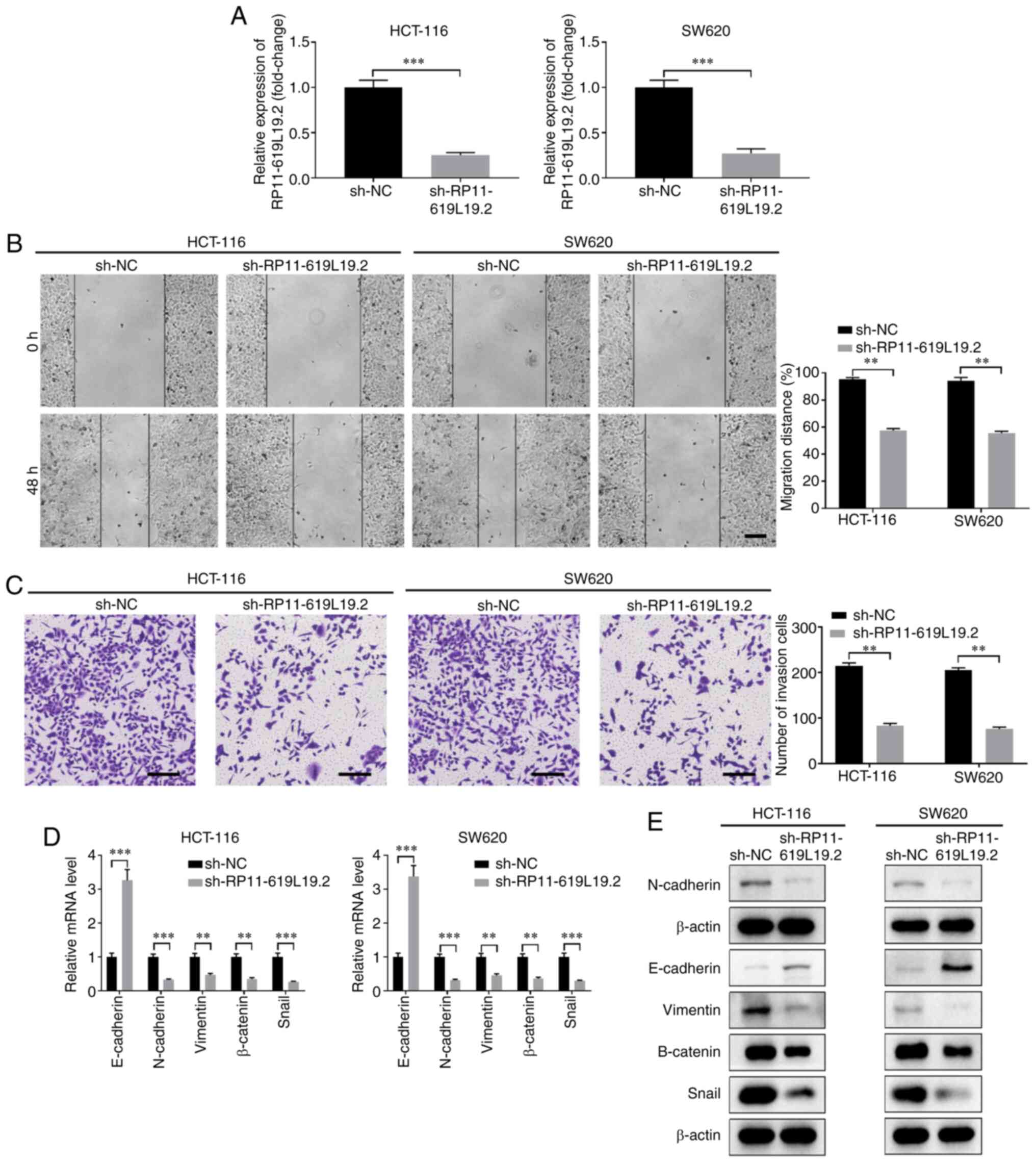
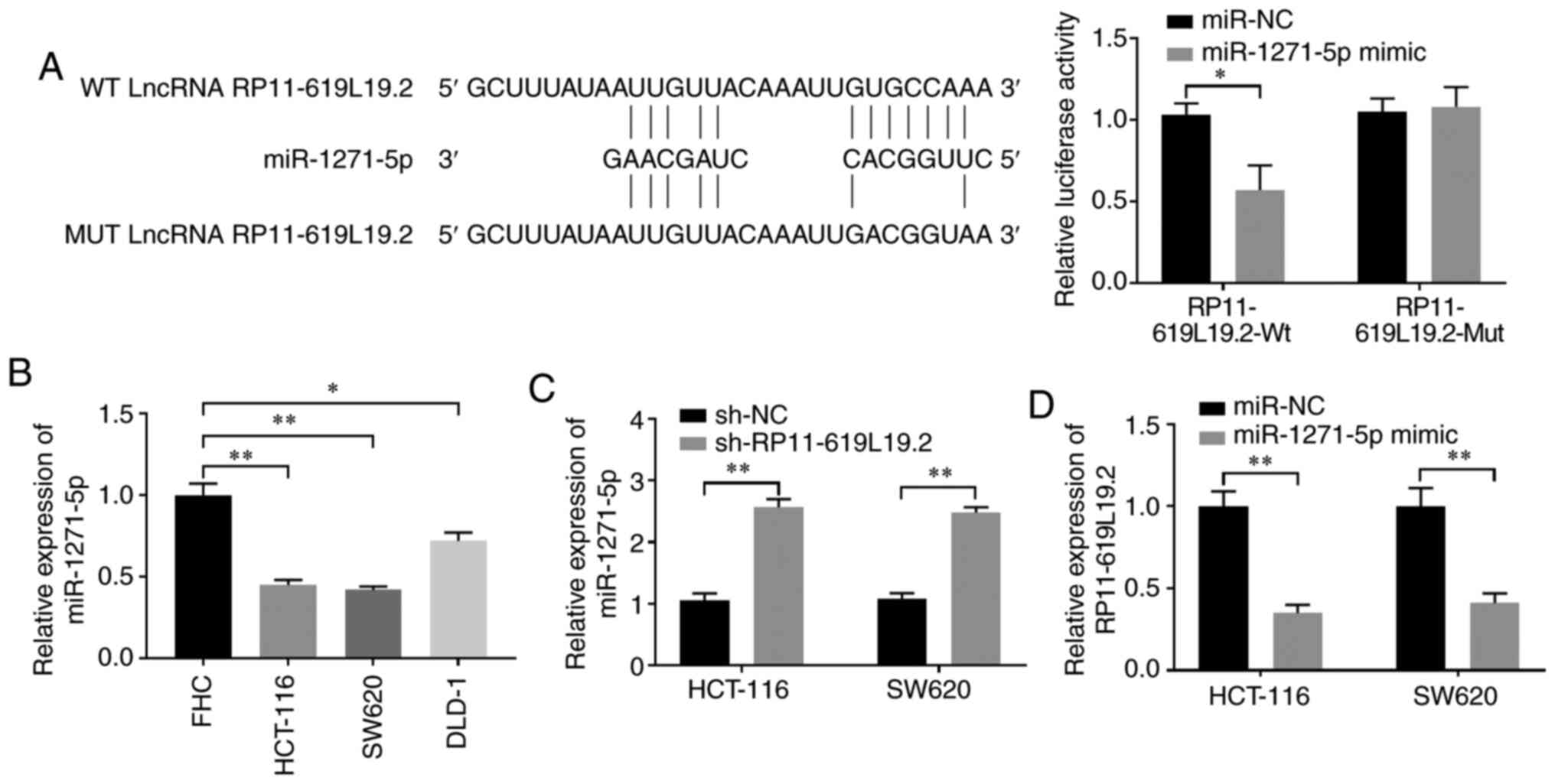
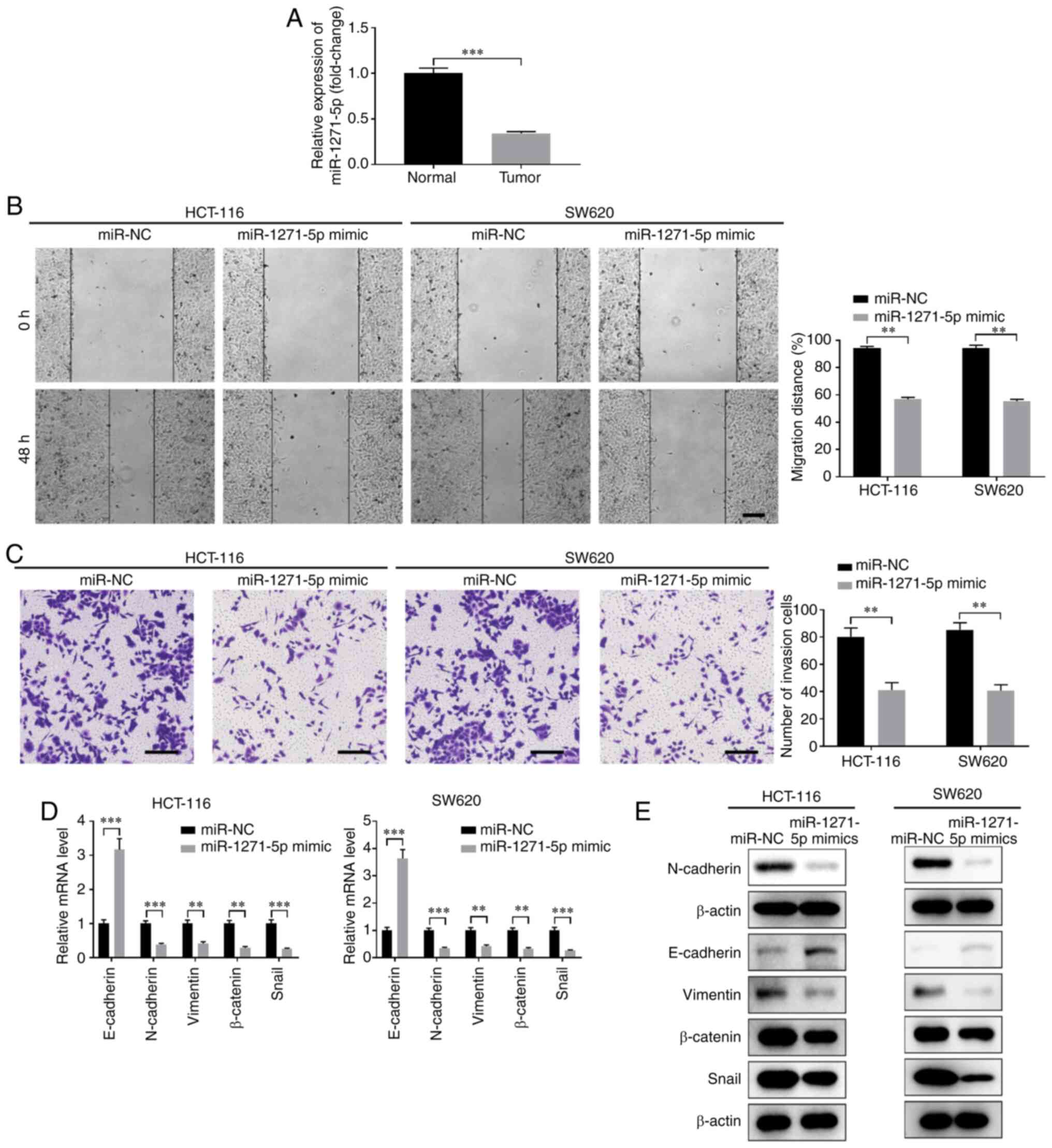
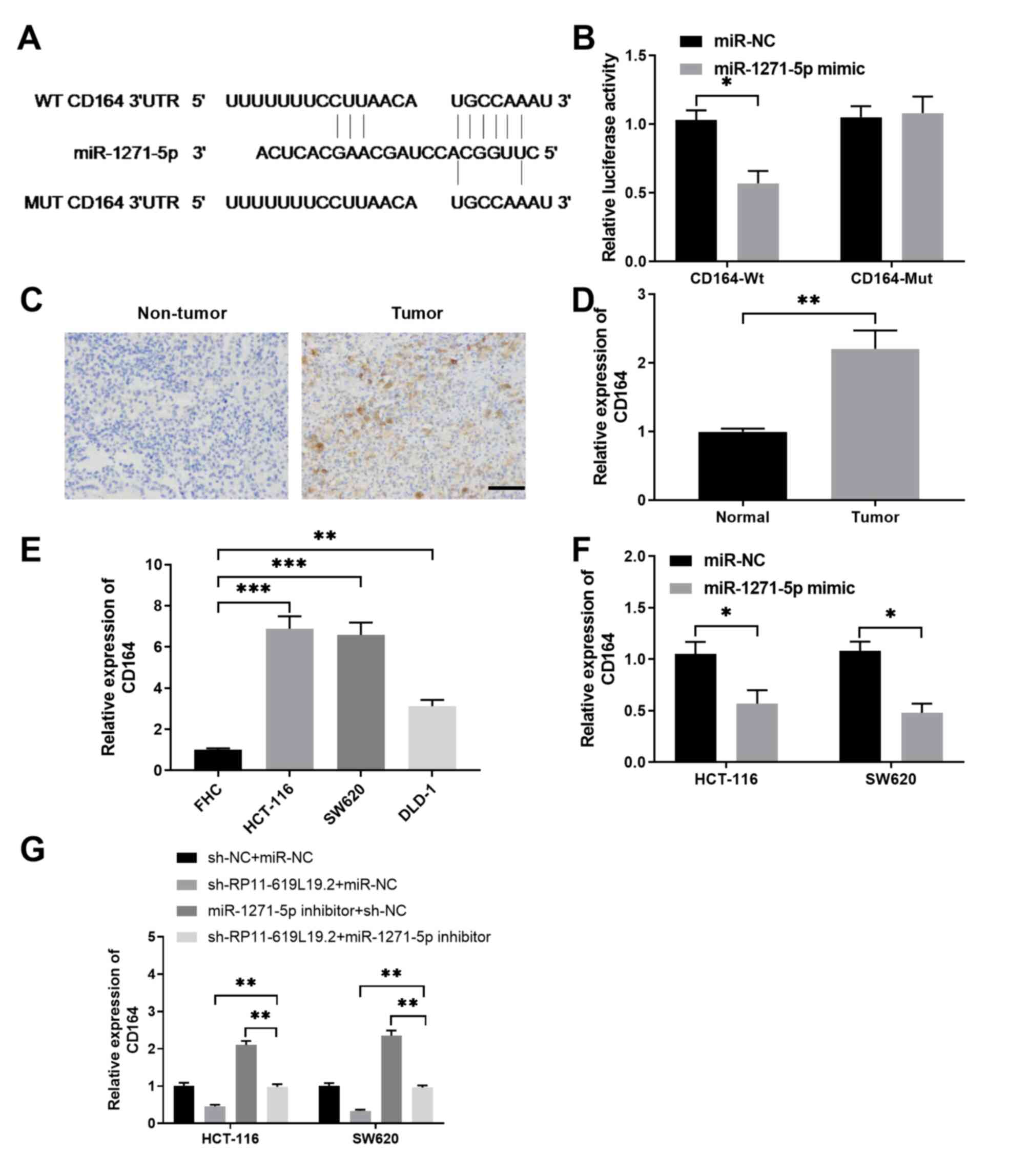
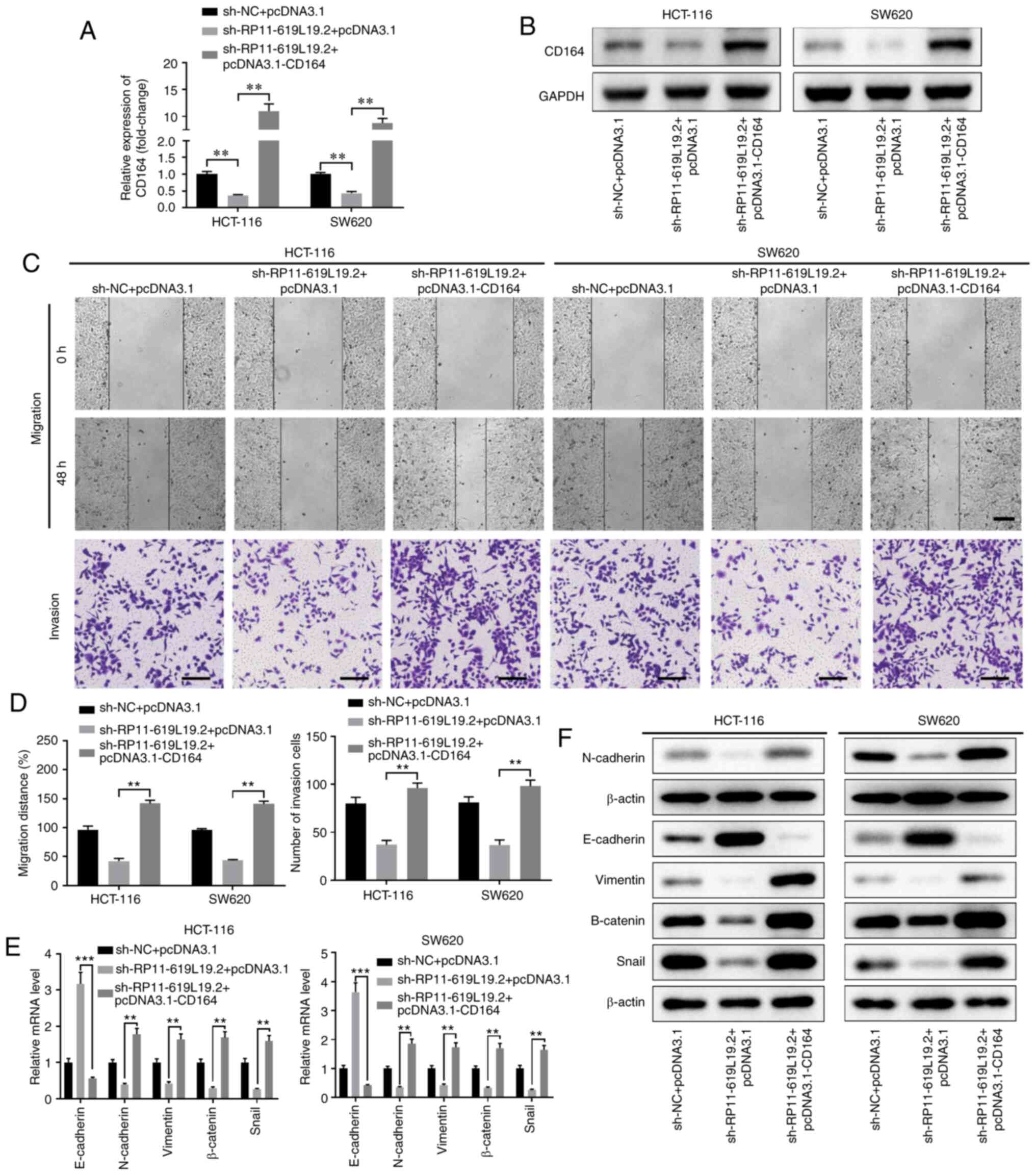
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Favoriti P, Carbone G, Greco M, Pirozzi F,
Pirozzi RE and Corcione F: Worldwide burden of colorectal cancer: A
review. Updates Surg. 68:7–11. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang ML, Peng P, Wu CX, Gong YM, Zhang
SW, Chen WQ and Bao PP: Report of breast cancer incidence and
mortality in China registry regions, 2008–2012. Zhonghua Zhong Liu
Za Zhi. 41:315–320. 2019.(In Chinese). PubMed/NCBI
|
|
4
|
Telang NT, Li G and Katdare M: Prevention
of early-onset familial/hereditary colon cancer: New models and
mechanistic biomarkers (R-eview). Int J Oncol. 28:1523–1529.
2006.PubMed/NCBI
|
|
5
|
Yang S, Sun Z, Zhou Q, Wang W, Wang G,
Song J, Li Z, Zhang Z, Chang Y, Xia K, et al: MicroRNAs, long
noncoding RNAs, and circular RNAs: Potential tumor biomarkers and
targets for colorectal cancer. Cancer Manag Res. 10:2249–2257.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Xue M, Zhuo Y and Shan B: MicroRNAs, long
noncoding RNAs, and their functions in human disease. Methods Mol
Biol. 1617:1–25. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huarte M: The emerging role of lncRNAs in
cancer. Nat Med. 21:1253–1261. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Renganathan A and Felley-Bosco E: Long
Noncoding RNAs in Cancer and Therapeutic Potential. Adv Exp Med
Biol. 1008:199–222. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Peng Y and Croce CM: The role of MicroRNAs
in human cancer. Signal Transduct Target Ther. 1:150042016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang G, Pian C, Chen Z, Zhang J, Xu M,
Zhang L and Chen Y: Identification of cancer-related miRNA-lncRNA
biomarkers using a basic miRNA-lncRNA network. PLoS One.
13:e01966812018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hon KW, Abu N, Ab Mutalib NS and Jamal R:
miRNAs and lncRNAs as predictive biomarkers of response to FOLFOX
therapy in colorectal cancer. Front Pharmacol. 9:8462018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shao M and Li W: Transcriptional factor
regulation network and competitive endogenous RNA (ceRNA) network
determining response of esophageal squamous cell carcinomas to
neoadjuvant chemoradiotherapy. PeerJ. 7:e66682019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu J, Li H, Zheng B, Sun L, Yuan Y and
Xing C: Competitive endogenous RNA (ceRNA) regulation network of
lncRNA-miRNA-mRNA in colorectal carcinogenesis. Dig Dis Sci.
64:1868–1877. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhou RS, Zhang EX, Sun QF, Ye ZJ, Liu JW,
Zhou DH and Tang Y: Integrated analysis of lncRNA-miRNA-mRNA ceRNA
network in squamous cell carcinoma of tongue. BMC Cancer.
19:7792019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cheng Y, Geng L, Wang K, Sun J, Xu W, Gong
S and Zhu Y: Long noncoding RNA expression signatures of colon
cancer based on the ceRNA network and their prognostic value. Dis
Markers. 2019:76367572019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gao Z, Fu P, Yu Z, Zhen F and Gu Y:
Comprehensive Analysis of lncRNA-miRNA- mRNA network ascertains
prognostic factors in patients with colon cancer. Technol Cancer
Res Treat. 18:15330338198532372019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tang XJ, Wang W and Hann SS: Interactions
among lncRNAs, miRNAs and mRNA in colorectal cancer. Biochimie.
163:58–72. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shi L, Hong X, Ba L, He X, Xiong Y, Ding
Q, Yang S and Peng G: Long non-coding RNA ZNFX1-AS1 promotes the
tumor progression and metastasis of colorectal cancer by acting as
a competing endogenous RNA of miR-144 to regulate EZH2 expression.
Cell Death Dis. 10:1502019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu Y, Nangia-Makker P, Farhana L and
Majumdar APN: A novel mechanism of lncRNA and miRNA interaction:
CCAT2 regulates miR-145 expression by suppressing its maturation
process in colon cancer cells. Mol Cancer. 16:1552017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang AF, Chen MW, Huang SM, Kao CL, Lai
HC and Chan JY: CD164 regulates the tumorigenesis of ovarian
surface epithelial cells through the SDF-1α/CXCR4 axis. Mol Cancer.
12:1152013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Heerboth S, Housman G, Leary M, Longacre
M, Byler S, Lapinska K, Willbanks A and Sarkar S: EMT and tumor
metastasis. Clin Transl Med. 4:62015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Paraskevopoulou MD, Georgakilas G,
Kostoulas N, Reczko M, Maragkakis M, Dalamagas TM and Hatzigeorgiou
AG: DIANA-LncBase: Experimentally verified and computationally
predicted microRNA targets on long non-coding RNAs. Nucleic Acids
Res. 41:D239–D245. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Marisa L, de Reynies A, Duval A, Selves J,
Gaub MP, Vescovo L, Etienne-Grimaldi MC, Schiappa R, Guenot D,
Ayadi M, et al: Gene expression classification of colon cancer into
molecular subtypes: Characterization, validation, and prognostic
value. PLoS Med. 10:e10014532013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen H, Xu J, Hong J, Tang R, Zhang X and
Fang JY: Long noncoding RNA profiles identify five distinct
molecular subtypes of colorectal cancer with clinical relevance.
Mol Oncol. 8:1393–1403. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen X, Yang S, Zeng J and Chen M:
miR12715p inhibits cell proliferation and induces apoptosis in
acute myeloid leukemia by targeting ZIC2. Mol Med Rep. 19:508–514.
2019.PubMed/NCBI
|
|
26
|
Lin MF, Yang YF, Peng ZP, Zhang MF, Liang
JY, Chen W, Liu XH and Zheng YL: FOXK2, regulted by miR-1271-5p,
promotes cell growth and indicates unfavorable prognosis in
hepatocellular carcinoma. Int J Biochem Cell Biol. 88:155–161.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang Y and Chen L: Downregulation of
lncRNA UCA1 facilitates apoptosis and reduces proliferation in
multiple myeloma via regulation of the miR-1271-5p/HGF axis. J Chin
Med Assoc. 82:699–709. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Doyonnas R, Yi-Hsin Chan J, Butler LH,
Rappold I, Lee-Prudhoe JE, Zannettino AC, Simmons PJ, Bühring HJ,
Levesque JP and Watt SM: CD164 monoclonal antibodies that block
hemopoietic progenitor cell adhesion and proliferation interact
with the first mucin domain of the CD164 receptor. J Immunol.
165:840–851. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lin J, Xu K, Wei J, Heimberger AB, Roth JA
and Ji L: MicroRNA-124 suppresses tumor cell proliferation and
invasion by targeting CD164 signaling pathway in non-small cell
lung cancer. J Gene Ther. 2:62016.PubMed/NCBI
|
|
30
|
Tang J, Zhang L, She X, Zhou G, Yu F,
Xiang J and Li G: Inhibiting CD164 expression in colon cancer cell
line HCT116 leads to reduced cancer cell proliferation, mobility,
and metastasis in vitro and in vivo. Cancer Invest. 30:380–389.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Coustan-Smith E, Song G, Clark C, Key L,
Liu P, Mehrpooya M, Stow P, Su X, Shurtleff S, Pui CH, et al: New
markers for minimal residual disease detection in acute
lymphoblastic leukemia. Blood. 117:6267–6276. 2011. View Article : Google Scholar : PubMed/NCBI
|