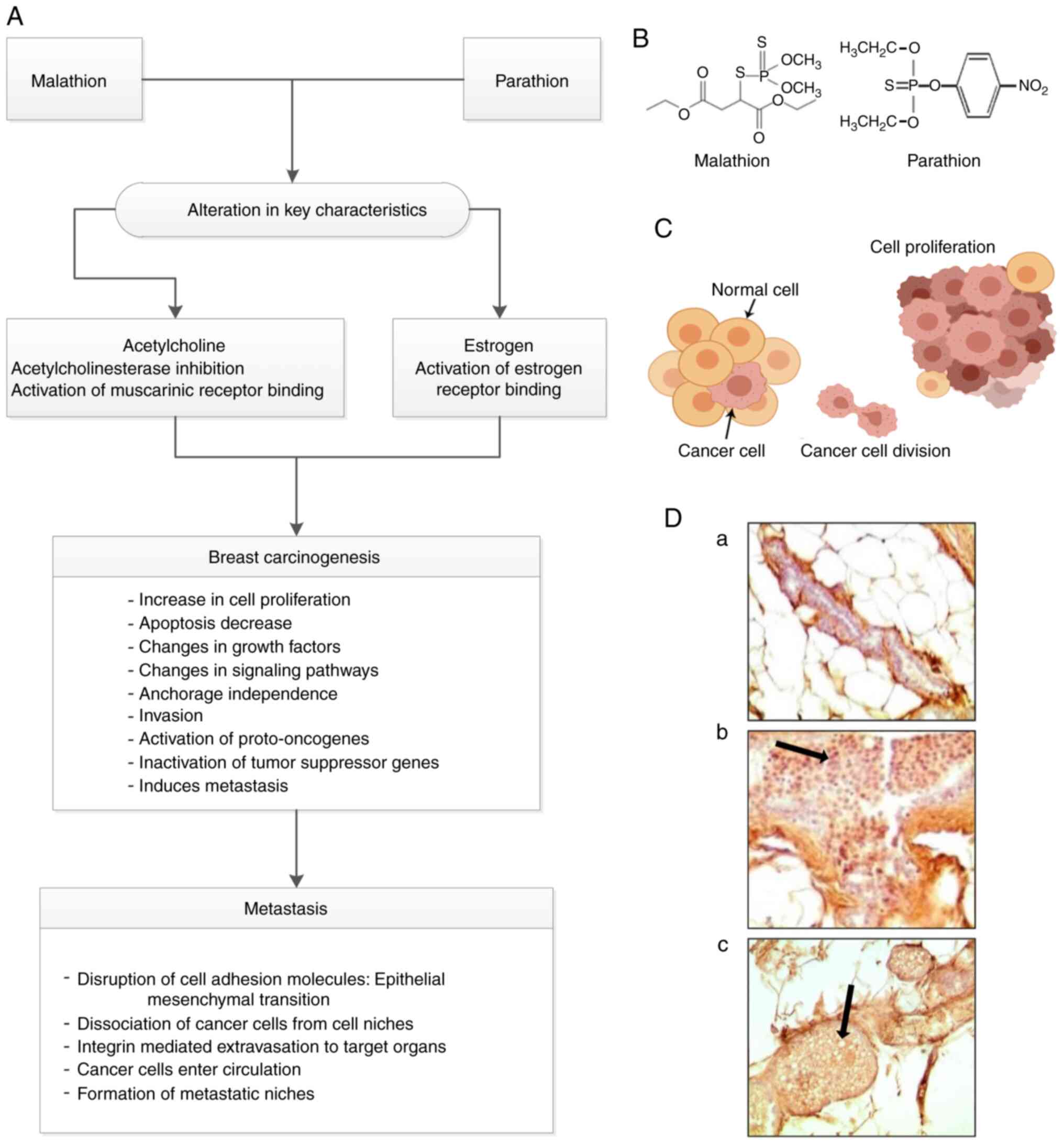
Introduction
Environmental substances appear to be involved in
various human diseases, including breast cancer (1,2). Based on
epidemiological evidence, several studies have found an association
between human cancer and exposure to agricultural pesticides, such
as organophosphorus pesticides (OPs) (3–5).
Generally, OPs can protect agricultural products for decades and
have therefore been widely used, particularly malathion, to control
vectors (6). Parathion has also been
used as a pesticide in agricultural settings. These pesticides,
however, pose a serious threat to multiple organisms, including
humans. For instance, certain pesticides have been associated with
blood diseases, such as non-Hodgkin's lymphoma (7–9) and
leukemia (10,11).
Other pesticides, including organochlorines,
creosote, and sulfallate, have been reported to be carcinogenic in
in vivo studies (12), whereas
dichlorodiphenyltrichloroethane, chlordane, and lindane have been
found to act as tumor promoters (13–15).
However, individual pesticides have only been evaluated in a
limited number of human studies. In addition, certain substances in
commercial pesticide formulations may pose a carcinogenic risk to
humans (15,16). Thus the International Agency for
Research on Cancer (IARC) (17)
classified parathion as ‘possibly carcinogenic’ (Group 2B) and
malathion as ‘probably carcinogenic’ to humans (Group 2A).
Furthermore, experimental studies have proposed that malathion or
its derivatives could be carcinogenic, indicating that impurities
found in commercial malathion, such as malaoxon and isomalathion,
induce DNA damage (18,19).
The etiology of breast cancer remains unclear, and
humans are exposed not only to pesticides but also to a mixture of
estrogenic agents (20). Estrogens
have been implicated in the etiology of breast cancer by
epidemiological and experimental evidence (21–24).
Moreover, the importance of hormones in mammary cancer (25), as well as the effect of a variety of
compounds on this process (26,27), have
been demonstrated. The exposure of human populations to these
substances renders it necessary to consider the effect of
pesticides and estrogens on human health.
Studies using various human epithelial cell lines
have been performed to analyze the cellular and biological
processes involved in transforming a normal cell into a cell with a
malignant phenotype (28,29). Furthermore, the use of experimental
animals and cells in the laboratory has allowed us to determine
whether these environmental substances induce breast cancer
(20,21,30–36).
Table I shows the phenotypic
characteristics of cell lines.
 | Table I.Phenotypic characteristics of cell
lines (20). |
Table I.
Phenotypic characteristics of cell
lines (20).
| Treatment | Anchorage
independent growth assay | Invasion assay |
|---|
| MCF-10F without
treatment | – | – |
| MCF-10F treated
with E2 (10−8 M) | – | – |
| MCF-10F treated
with M (0.5 µg/ml) | + | + |
| MCF-10F treated
with M and E2 | + | + |
| MCF-10F treated
with P (100 ng/ml) | + | + |
| MCF-10F treated
with P and E2 | + | + |
The use of the MCF-10F immortalized normal human
breast epithelial cell line has enabled the detection of the
sensitivity to several substances, such as
7,12-dimethylbenz(a)anthracene (DMBA) and benzo(a)pyrene (BP)
(37), and physical factors, such as
ionizing radiation (38), and the
determination of their carcinogenic properties.
The present study aimed to summarize the in
vitro signs of transformation induced by environmental
substances, such as malathion and parathion, in the presence of an
endogenous substance, such as estrogen, through the use of the
MCF-10F human immortalized breast cell line. This type of cell line
is an important tool in the experimental study of breast
carcinogenesis induction by hormones or transfection with a
c-Ha-ras, or its prevention by antioxidants, such as curcumin
(39–41). Table I
shows the effect of malathion, parathion, and estrogen on anchorage
independence and the invasive capabilities of treated cells. The
MCF-10F cell line treated with malathion or parathion alone and in
combination with estrogen induced anchorage-independent growth and
invasion; however, the same cell line treated with estrogen alone
and the control were negative under the same conditions. A previous
study demonstrated that estrogen exerts its effects when combined
with pesticides in this model, providing an approach to studying
this process (35). A new approach
has emerged for analyzing carcinogens by the IARC and previous
studies; carcinogens were classified based on 10 common
characteristics associated with carcinogenesis (17,42,43).
Data collection
In the present review, a search on MEDLINE (through
PubMed), Web of Science, and SCOPUS was conducted between January
2020 and June 2020 to identify studies examining the in
vitro changes of the normal MCF-10F human breast epithelial
cell line under the effect of pesticides in the presence of
estrogen. The selection was based on cell transformation assays
using the MCF-10F cell line to examine the following: i) Cell
proliferation by the trypan blue exclusion method; ii) cell growth
in a semisolid medium by anchorage-independent assay; iii) cell
invasion by cell invasion assay; iv) oncoprotein by
immunocytochemistry coupled with confocal microscopy; v) gene
expression in several arrays with cell cycle-related key genes;
human drug metabolism in gene array including genes that encode
important receptors and several enzymes involved in drug transport
and phase I and phase II metabolism; and vi) genomic instability in
a human cancer oligo array.
Parathion and malathion increase cell
proliferation
The association between OPs and estrogen was
analyzed in relation to mammary carcinogenic capability. Exposure
to OPs can be considered an important initiator of breast cancer,
as shown by several signs of carcinogenicity detected in
vivo and in vitro. The in vivo studies were based
on morphological and molecular experiments using Sprague-Dawley
rats. Since malathion significantly increased the density of
terminal end buds (TEBs), this research allowed us to obtain a
model of the initiation of mammary gland cancer. The primary
outcome in rats was the increase of mammary cells in TEBs that were
then transformed into proliferative ducts by malathion or
parathion, eventually resulting in ductal mammary carcinomas
morphologically similar to those found in the breast (30,31,44).
When the animal was injected with estrogens, the
formed TEBs were transformed into proliferative lobules full of
secretions, with a decreased density of alveolar buds, resulting in
actively growing tumors (30,31,44); the
pathology of these tumors was of the lobular type. Both the ductal
and lobular mammary carcinomas were similar to those classified by
the World Health Organization. When the animals were exposed to
pesticides and estrogen, both types of structures such as ducts and
lobules were observed. Mammary gland tumors then metastasized to
the bronchi, lungs, and kidneys. The effect of OPs was avoided by
atropine demonstrating an association of atropine with the
muscarinic receptor. In vivo studies showed signs of
carcinogenicity, including cell proliferation leading to tumor
formation and genomic instability (32). The mechanisms for mammary carcinogenic
potential included acetylcholinesterase inhibition, increased
oxidative stress, decreased apoptotic signaling, and
endocrine-disrupting capabilities.
Parathion (33) and
malathion (20) had been previously
found to increase cell proliferation and induce cell transformation
affecting protein expression in the MCF-10F cell line (20,33,34,36,45).
It was found that malathion alone or in the presence of estrogen,
induced anchorage-independent growth, cell invasive capabilities,
altered cell cycle regulation, and increased genomic instability in
the MCF-10F breast cell line in vitro (20,36).
Another study demonstrated that malathion induced changes in gene
expression (45). A scheme of
exposure to OPs, estrogen, and chemical structures of malathion and
parathion is presented in Fig. 1.
When the established model was initially developed,
morphological changes were the first observed signs in
vitro, which included a changing doubling time, colony agar
formation, and invasive capabilities, all indicative of a very
aggressive phenotype, as compared to the control cells (20,21,33,36,38,46–49).
On the other hand, it was observed that atropine, an antagonist of
muscarinic receptors, when combined with any of these pesticides
inhibited all aforementioned effects (33).
The same was observed in another cell line, the
MCF-7 malignant breast cancer cell line, in which estrogen markedly
increased cell proliferation in vitro after 6 days (44). Moreover, it was reported that
sumithrin, a pyrethroid pesticide used to control pests in
agriculture (50), also induced
proliferation in vitro at 10−7 M and
10−5 M doses after 6 days of treatment (51). In addition, in mammalian cells, the
pesticide parathion-methyl increased the number of cells and
changes in the MCF-7 cell line at low concentrations, exerting
toxicity and altering cell-cell interactions in human intestinal
cells; it also had other effects on murine fibroblasts, such as
increased DNA synthesis (52). These
studies demonstrated not only a cell proliferation effect but also
other possible physiological effects with a serious impact on
humans.
It can be hypothesized that one possible mechanism
for breast cancer development is the consequence of excessive
estrogenic stimulation that induces cell proliferation of normal
breast epithelial tissue (53). More
specifically, the malignant phenotype is developed through errors
in cell division (DNA copying errors, translocations); furthermore,
estrogen is known to control the growth of several carcinomas in
experimental animals and humans (53).
Although MCF-10F cells are estrogen receptor
(ER)-negative, they were found to be very sensitive to
17β-estradiol (E2) at 10−8 M since cell proliferation
increased (1.6 fold) after 10 days of culture (53). The addition of E2 and 10−6
M tamoxifen to the MCF-10F cells gave similar results to those of
the malignant carcinomas in vitro since it was found that E2
increased cell proliferation, as compared with the control.
However, tamoxifen alone and tamoxifen plus E2 inhibited cell
proliferation more notably when compared to E2 alone. The
association between cell proliferation and proteins involved in
cell cycle regulation was also investigated (54,55). When
cell cycle control is lost, cells are able to continue dividing
(55). When the cell cycle becomes
deregulated, it can lead to aberrant cell proliferation, eventually
resulting in cancer (56). This
observation prompted the analysis of gene expression during the
cell cycle as well as its regulation and proliferation.
Differential gene expression was studied using the
Oligo GEArray® human cancer microarray (cat. no.
OHS-802) in estrogen- and pesticide-treated MCF-10F breast
epithelial cells. The results indicated that parathion and estrogen
alone, or a combination of the two, induced transcriptional
alterations in 22/96 genes from a cDNA array. These alterations
involved genes associated with the regulation of the cell cycle,
such as cyclins A1, A2, C, D3, G1, G2, and H, cyclin-dependent
kinases (CDKs), including CDK41, and minichromosome
maintenance protein complex (MCM), a 2–7 hexameric helicase,
including MCM2 and MCM3 (20,21).
Regarding cyclins, this family of proteins, particularly D-type
cyclins, form a complex with CDKs, which affects the cell cycle at
the G1 phase (57,58), regulating the cell cycle as a whole
(59). Pesticides have been shown to
have an affinity for CDKs, with OPs exhibiting a particular
affinity for CDK2 and CDK4, which affects cell cycle
regulation in mammalian cells and other pesticides, such as
carbamates and synthetic pyrethroid; these were also evaluated, and
a positive interaction was identified between them and CDKs at low
doses (60).
Previous studies have indicated that the cyclin A2
gene was downregulated by all the substances under study and,
whereas cyclin C and cell division cycle 6 (CDC6) were
upregulated 3-fold by parathion, as compared with the control.
Cyclin D3 gene was upregulated by both estrogen and parathion.
Cyclin-dependent kinase CDKNIA was upregulated 3-fold by
parathion alone, and CDKN2C, which is associated with cell
cycle checkpoint and cell cycle arrest, was downregulated by both
estrogen and parathion (21).
Furthermore, different treatments of pesticides
alone or in combination with estradiol were shown to upregulate the
cyclin D1 and CDK genes. Of note, the resulting proteins
were involved in the phosphorylation of important effectors
associated with different stages of the cell cycle (61–64). A
previous study revealed upregulation by the effect of E2 in
combination with the pesticide compounds, including the
upregulation of cyclin family genes, including keratin 18 (20). These results were in agreement with
previous findings (33).
The MCM2 family of proteins was upregulated by both
malathion and parathion. The MCM family of proteins is known to be
involved in the regulation of DNA replication (65,66). It
has been reported that the expression of MCM proteins increases
during DNA replication (67). The MCM
proteins controlled by E2F transcription factors have been shown to
promote MCM expression (68). The
protein kinase complexes interact with MCM proteins maintaining the
post-replication stage and MCM2/MCM4 serve as substrates for
CDC2/cyclin B (69–71). MCM3 cleavage can be prevented by
caspase inhibitors, resulting in MCM complex inhibition during
apoptosis (72). Furthermore, the
MCM4, MCM6, and MCM7 complexes have been found to be
involved in DNA helicase activity (71,73). In
addition, results indicated that parathion and estrogen upregulated
the MCM6 labeling index, as compared with the control value
(21). Other studies have reported
this index to be correlated with cell proliferation and malignant
behavior in chondrosarcomas (74).
p53 is another gene involved in the
regulation of the cell cycle, serving as a checkpoint for the
G1-S phase (75). At the
same time, the MDM2 gene regulates p53 and is
associated with tumor growth and metastasis (76). It was observed in previous studies
that the combination of parathion and estrogen upregulated
MDM2 (21) and downregulated
p53 (77), thus increasing
tumorigenic capabilities (21).
Similarly, a study analyzed peripheral lymphocyte DNA obtained from
180 workers with long-term exposure to OPs. That study reported
that omethoate, an OP compound, affected the expression of
p53, which in turn had an impact on the length of the
telomere, suggesting a clear influence of pesticides over the cell
cycle and tumor formation (75,78).
Dishevelled (DVL) is a gene that regulates
the migration and proliferation of endothelial cells present in
blood vessels (79,80). Malathion and the combination of
parathion and estrogen upregulated the DVL1 gene and
increased the protein expression in cells treated with parathion,
alone and combined with estrogen, as compared with the controls
(20). The mammalian homologs of the
Drosophila DVL together with DVL proteins are important
molecules in the Wnt signaling pathway (80–82). DVL-2
protein expression was found to be increased by estrogen,
malathion, and parathion, regulating the proliferation of the
MCF-10F cell line, as compared with the control (20).
Other studies have also reported that genes
associated with cell cycle progression, DNA replication, and
checkpoint enzymes were affected by malathion (45,83,84).
Cyclin-dependent kinases regulatory subunit 1 is also fundamental
in cell cycle progression (85), and
associated with genes that particularly affect the G2
phase; G2/M transition was found to be downregulated in
estrogen and parathion treatments (21). These results indicated that pesticides
affected the regulation of the cell cycle with possible effects on
cancer initiation.
E2 at 10−8 M significantly increased cell
proliferation; however, as shown in Table
I, E2 did not induce anchorage-independent growth, anchorage
independence, or invasiveness in Matrigel®. Other
researchers (86) reported the
induction of complete transformation of MCF-10F cells by E2,
confirming its carcinogenicity; however, E2-treated-MCF-10F cells
were trypsinized and seeded in the upper Matrigel-coated invasion
chamber, followed by post-seeding of cells that had crossed the
Matrigel membrane, giving origin to several MCF-10F cell lines. E2
induced complete transformation of the human breast epithelial
MCF-10F cells in vitro, confirming its carcinogenicity and
supporting the concept that this hormone could act as an initiator
of breast cancer in women.
Parathion and malathion modulate epidermal
growth factor receptor and estrogen receptor expression
Previous research has indicated that cell growth is
affected by the epidermal growth factor (EGF) through its
interaction with the EGF receptor (EGFR) (33); since high levels have been found in
the surface of different types of cancer cells (87,88) and
its association with cancer has been confirmed over the years. The
results of a previous study indicated that EGFR protein expression
was increased in cells treated with the pesticide alone or in
combination with the hormone (21).
These results are important, considering that growth factors and
their receptors are proteins associated with cell growth (21,89).
According to previous results, the parathion-treated MCF-10F cell
line induced a higher EGFR/ERBBI protein expression, as compared
with control and parathion plus atropine-treated cell lines
(21). EGFR is a receptor tyrosine
kinase associated with cancer, the overexpression of which is
correlated with poor prognosis, solid tumor growth, cancer
metastasis, and lower survival rate (90,91). Such
results have indicated that pesticides such as parathion induce
EGFR expression.
It has been reported that estradiol increases the
risk of breast cancer in women after long-term exposure since
estrogens increase cell proliferation by activating ER-mediated
transcription; however, this interaction has been shown to induce
genomic instability, chromosomal aberrations, and an increase in
errors during DNA replication (25,26,92,93).
Since the MCF-10F model lacks ER expression, E2
appears to act through ER-independent mechanisms. The ERs are
ligand-inducible transcription factors that belong to the
superfamily of nuclear steroid hormone receptors (94,95). The
transmission of estrogen signaling includes the activation of ERs
and signal transduction, which can be mediated by genomic and
non-genomic signaling pathways. Such classification is based on the
outcome of cellular events, including the modulation of gene
expression or activation of signaling cascades. The classic genomic
pathway is the best-characterized ERa signaling pathway, which is
initiated by the ligand binding to its receptor. The binding
induces a conformational change and dissociation of their
chaperones/nuclear matrix-associated binding proteins (96), forming the E-ER complexes that
translocate to the nucleus and bind to specific DNA sequences;
these are called estrogen response elements (EREs) and are located
in or near the promoters of target genes (97). An ERE-independent signaling pathway
has been reported, where E2-ER complexes can mediate gene
expression through functional interactions with transcription
factors on the DNA (98,99). ERs may interact with many other
proteins, including adaptor proteins, G-proteins, GFRs (EGFR,
IGFR1, and HER2), cytoplasmic kinases [mitogen-activated protein
kinases (MAPKs), PI3K and AKT], and signaling enzymes, which can
eventually lead to indirect changes in gene expression (100).
On the other hand, the influence of ERα-signaling
pathways on epithelial-to-mesenchymal transition-related
transcriptional factors, which are fundamental in the development
of breast cancer, has been reported (34,35,101,102).
Parathion and malathion induce metabolic
alterations
It has been reported that pesticides affect the
human population, due to their long-term exposure and intensity,
and that they alter the detoxification rate by changing the
expression of enzymes associated with the transport and metabolism
of drugs (103). Briefly, the
by-products of drug metabolism are substances that may be
pharmacologically active, inactive, or toxic (104). This process is divided into two
phases, phase I and phase II; the former is mainly associated with
a sophisticated enzymatic complex, known as cytochrome P450 (CYP),
whereas the latter is associated with the addition of polar
moieties to the substrate, to be eliminated by organisms (105).
Previously, genes involved in human drug metabolism
have been analyzed by cDNA microarrays; CYPs, metallothioneins, and
p-glycoproteins were further studied (34). CYPs are an enzymatic complex that
belongs to the family of monooxygenases, which are involved in the
metabolism of endogenous and xenobiotic compounds (106).
According to cDNA microarray, parathion was found to
result in CYP upregulation, whereas estrogen, alone or
combined with parathion, induced the downregulation of
CYP2F1 and CYP4F3; however, there was no change in
the CYP3A7 gene expression following exposure to either
substance (34).
Results have shown that catechol formation is a
major risk factor for breast cancer (107); since it gives rise to reactive
quinones causing DNA damage and redox cycling, which in turn lead
to the generation of reactive oxygen species (ROS), which can cause
oxidative damage (108).
Other important mechanisms involved in carcinogenic
effects, besides the stimulation of cellular proliferation through
their receptor-mediated hormonal activity, are the direct genotoxic
effects exerted by increasing mutation rates through
CYP-mediated metabolic activation. A previous study
(86) demonstrated that estrogens are
carcinogenic in the human breast by testing the natural E2 or its
metabolites, 2-hydroxy, 4-hydroxy and 16-a-hydroxy-estradiol
[2-OH-E(2), 4-OH-E(2) and 16-α-OH E(2), respectively] in an
experimental system, and neoplastic transformation of MCF-10F cells
was observed, to a degree at least similar to that induced by the
BP.
On the other hand, estradiol metabolism may result
in quinone derivatives, which directly replace base pairs from DNA
through depurination, and can also alter the DNA repairing process
(109–111). It has been reported that estrogens
are potent mammary tumor promoters influencing post-initiation
events through epigenetic mechanisms. The upregulation of the
C16α-hydroxylation pathway during E2 biotransformation was
associated with mammary cell transformation. The action of E2
metabolites on tumorigenic transformation was studied in a mammary
epithelial cell line derived from the C57BL mouse strain, where
estrogen or its metabolites were found to function as initiators of
mammary cell transformation demonstrated by increased cell
proliferation, anchorage-independent growth, and alteration of
metabolism (112).
Metallothioneins are proteins with a low molecular
weight that are rich in cysteine domains. These proteins play an
important role in metal homeostasis, particularly the
detoxification of heavy metals (113,114).
Their dysregulated expression has been observed in invasive ductal
breast carcinoma, and they have been proposed for use as a
prognostic biomarker (115).
Metallothionein 2A expression has been found to be associated with
cell proliferation in breast cancer (113,116).
Furthermore, genes associated with metallothioneins have been shown
to be altered by pesticides and estrogen; the only functional gene
upregulated by parathion alone was metallothionein IX, with
estrogen alone and estrogen plus parathion resulting in its
downregulation (34).
In this context, epidemiological studies have found
an association between metabolic enzymes and the age of onset for
sporadic colorectal adenocarcinoma (117,118).
Then, variant alleles of phase II, such as GST, uridine
5′-diphospho-glucoronosyltransferase (UDP), and
glucuronosyltransferase (UGT) can be used as molecular biomarkers
of cancer risk (119). For example,
GSTM(µ)1 was found to be associated with an increased risk of
colorectal, lung, and bladder cancer, and GSTP(π)1 with prostate
cancer (120–123). Furthermore, these enzymes catalyze a
large variety of drugs and endogenous compounds, such as molecules
with sulfo groups in the case of sulfotransferases (124,125),
and are in charge of the biosynthesis of polysaccharides,
oligosaccharides, and conjugates, in the case of
glucosyltransferases (126,127). Previous studies have indicated that
the combination of parathion and estrogen induced the
downregulation of all methyltransferase genes, such as TPMT;
notably, the CHST5, CHST6, and CHST7
(sulfotransferase) genes were upregulated by parathion and
downregulated by estrogen, alone or combined with parathion
(34).
The carbohydrate sulfotransferases play a role in
oxidative stress and the estradiol signaling pathways in
carcinogenesis (128); and have been
investigated in breast cancer and glioma patients (119,129).
On the other hand, several glycosyltransferases (GSTs) have
also been identified; the GSTP1, GSTT2, and microsomal
glutathione s-transferase 1 (MGST1) genes were overexpressed
by parathion and downregulated by estrogen, when compared to the
control, whereas the combination of estrogen and parathion
downregulated MGST1, with no change observed in the
GSTP1 and GSTT2 genes (34).
As previously reported, UDP-UGT is another enzyme
associated with detoxification (130), which was found to be increased by
parathion (UGT1A1 and UGT2B genes) and decreased by
estrogen; however, there was no difference in these genes when the
substances were used together (34).
Clinical studies have shown an increase in the UGT1A1 and
UGT2B gene expression in ovarian cancer (119); therefore, it can be a reliable
molecular biomarker for the risk of cancer. The carcinogenic
activity of 4-hydroxyestradiol was analyzed in a hamster kidney
tumor model of DNA damage by steroidal estrogens through catechol
estrogen metabolites (24,131–133).
It is important to note that the 2-hydroxylation of steroidal
estrogens is the major metabolic oxidation of estrogenic hormones
in most mammalian species (134,135).
Moreover, there are other enzymes with potential
carcinogenic activity in the metabolism of endogenous and exogenous
compounds (136,137); for example, the enzymes comprising
the aldo-keto reductase (AKR) family involved in redox
transformation, with substrates such as glucose and steroids, as
well as environmental pollutants, among others (138). AKR1C1 and AKR1C2 were also
upregulated by another non-organophosphorus pesticide (45). In combination, these examples
indicated an impairment of homeostasis by certain substances,
ultimately leading to carcinogenesis. Another example is the
estrogen-responsive B box protein (139), also upregulated by malathion
(137). This protein belongs to the
tripartite motif protein family, and its upregulation has been
associated with histone acetylation and the transcription of
CYP26A1, which is important in retinoid-resistant cancer cells
(139).
In this context, particularly in cell metabolism
and metabolic pathways, certain studies reported glucose
homeostasis impairment (137) and
metabolic disorders, with certain metabolic changes still present
for a long time even after discontinuing long-term exposure to
malathion (140), which was due to
OPs. These disturbances may have occurred through physiological
stress, oxidative stress, and other mechanisms (141). These results were confirmed by in
vivo studies; for example, malathion induced insulin resistance
biomarkers and reduced insulin sensitivity (140). Other studies reported that, in
general, OPs increased blood glucose (142–144)
and induced glycogen phosphorylase and phosphoenolpyruvate
carboxykinase activity following malathion treatment in rats
(145). Of note, glucose and lipid
metabolism were affected in rats under the influence of malathion
(146,147).
Similarly, neonatal parathion exposure in rats was
found to alter lipid metabolism and induce an inflammatory response
in adipose tissue, parathion alone decreased adiponectin levels and
increased tumor necrosis factor-α (TNF-α) (148). Adiponectin is a monomeric protein
secreted in the circulation with the main purpose of inducing fatty
acid oxidation and inhibiting glucose synthesis in the liver. It
has also been recognized as an anti-inflammatory agent (149), whereas TNF-α is a cytokine
associated with immune homeostasis, inflammation, apoptosis,
angiogenesis, and cell migration (150,151).
These in vivo studies supported the in
vitro results and demonstrated that exposure to OPs induces a
chronic adipose inflammatory response, leading to the emergence of
other diseases, such as diabetes, obesity, and cardiovascular
diseases (148).
Parathion and malathion cause genomic
instability
Genomic instability is known to be induced by
uncontrolled cell proliferation, with pesticides and estrogen found
to increase the risk of genetic damage (152), involving changes in the expression
of oncogenes and the loss or inactivation of tumor-suppressor genes
(153,154); this leads to the accumulation of
abnormalities in cells. It was demonstrated by human cancer
microarray analysis that endogenous and exogenous agents, including
estrogens and OPs, affected 408 genes, 17 of which were involved in
human cancer regulation. Among those genes that were altered are
those associated with cell cycle progression, cell differentiation,
and signal transduction pathways (20). To determine specific genetic changes
and their biological consequences is crucial for understanding
breast carcinogenesis.
Studies have indicated that mutations in the
Ras oncogene observed in cancer cells correspond to the
amino acid substitutions at positions 12, 13, and 61, and it is
important to consider that the oncogenic Ras proteins act
downstream of effector pathways to induce the deregulation of cell
proliferation and abnormal functional properties of cells (155). The results of previous studies have
indicated that allelic imbalance at different chromosomal levels
involved the overexpression of the H-ras oncogene; with the
marker mapped for chromosome 11p14.1 showing microsatellite
instability (MSI) in malathion- and estrogen-treated cells
(156,157). A different study showed that
malathion or parathion, alone and in combination with estrogen,
upregulated H-ras (20). Those
findings indicated a loss of heterozygosity (LOH) in parathion and
estrogen-treated cells, LOH in codon 12 in either malathion- or
estrogen-treated cells, and MSI in codon 61 of malathion- and
estrogen-treated cells (36).
Furthermore, it was shown that chemical carcinogens induced
mutations in codons 12 and 61 of H-ras (158). Another study provided an example of
the genomic instability of the Ras gene in MCF-10F cells
under the influence of the two pesticides and the presence of
estrogen (20). The use of
microsatellite markers can be useful to determine the degrees of
allelic imbalance, LOH or MSI (159–161).
The specific genomic imbalances in microsatellite regions of
specific genes appear to be important in determining the risk of
cancer since tumor pathogenesis is associated with specific
imbalances and disease prognosis (162,163),
and they may serve as specific therapeutic targets. Genomic
instability was observed in parathion-, malathion- and
estrogen-treated MCF-10F cells in the form of LOH and MSI (36). The malignant phenotype was
characterized by an increase in H-ras oncogene expression.
On the other hand, microsatellite markers helped determine that the
malathion- and estrogen-treated cells exhibited MSI with a marker
for H-ras mapped in chromosome 11p14.1, and LOH in the
presence of malathion or estrogen alone (36). Other studies have reported
overexpression of the c-Ha-ras p21 protein in human breast cancer
(164–166), indicating that the expression of
this protein may serve as a marker of breast cancer
progression.
The Trio domains exhibit Rac and Rho
activity (167). Rho, another member
of the RAS superfamily (168), is
present in several cell types and is involved in cell polarity and
motility (169,170). Parathion and estrogen, alone and in
combination, increased Rho-A protein expression, as compared with
the control (21). Kleer et al
(170) reported a higher Rho-A
protein expression in all breast tumor biopsies, as compared with
normal tissues, which was correlated with histological grade; this
suggests a role of this protein in tumor progression and indicates
that it may serve as a prognostic marker in the clinical setting
(21). Rac, a GTP-binding protein of
the Ras superfamily, controls several processes, including cell
proliferation, cell polarity, and cytoskeletal arrangement
(171). Rac was found to be
overexpressed in parathion-treated cells; Rac 3 was particularly
overexpressed in cells treated with parathion combined with
estrogen. In addition, an increase in Trio protein expression was
observed in cells treated with parathion, alone and combined with
estrogen, when compared to the controls (33). The Trio is a multi-domain protein with
two DVL-homology/pleckstrin domains (167,172).
The c-Kit protein has also been found to be overexpressed in breast
cancer (88,173). A previous study demonstrated an
increase of c-Kit protein expression in cells treated with
parathion, alone and in combination with either atropine or
estrogen when compared to their controls (33).
It is known that the activation of tumor-promoting
signaling, such as RAS/MAPK signaling, may promote cancer cell
proliferation and invasion (174).
OPs altered the c-Ha-Ras oncogene and Rho-A, among
others, and estrogen affected ER in the MCF-10F cell line (20,21). Based
on these findings, it is, therefore, possible to hypothesize a
cross-talk between pesticides and estrogen, with the combination of
the two inducing morphological and molecular changes indicative of
cell transformation, which would be completely different in the
absence of estrogen. Therefore, parathion and malathion combined
with estrogen have been found to be involved in breast cell
carcinogenesis.
Growth factor regulators, such as fibroblast growth
factors (FGFs) and their receptors (FGFRs), regulate different
cellular processes, including angiogenesis, metastasis, and tumor
progression; the deregulation of these factors affects signaling
pathways involved in breast cancer (175–178).
Acidic FGF (FGF-1), a member of the fibroblast growth
factor superfamily, has important functions in DNA synthesis, cell
division, and differentiation (179), and it is also critical for the
development of different types of cancer (180–182).
FGF-2 and its ligand, FGFR2, along with FGF-1,
one of the main ligands for FGFR1, have been associated with
tumor progression, regulation of tumor angiogenesis, and metastasis
(183); this is due to their
presence in the tumor microenvironment, which enables them to
mediate the effects of several different pathways, such as MAPL and
PI3K (177,184).
Insulin-like are growth factor-binding proteins
(IGFBPs), among which IGFBP3 and IGFBP5 are important for the
regulation of IGF signaling (185–188).
Exposure to either parathion or malathion upregulated both
IGFBP3 and IGFBP5 gene expression (20). Regarding the interaction of these
compounds with breast cell receptors and their association with an
endocrine-disrupting connotation, a review reported that malathion
induced ER activity and served as a weak ER agonist in the MVLN
human breast carcinoma cell line (189).
The function of the cadherin-catenin system in cell
adhesion and intracellular signaling appears to be the result of
different mechanisms (190,191). Thus, the E-cadherin-catenin complex
is the target of numerous growth factors and hormone-dependent
signaling pathways that regulate its function and expression
(191). In general, β-catenin has
been associated with breast cancer progression due to its invasive
capabilities (46,192–194),
which make it a very sensitive prognostic marker for invasive
breast cancer (190,192–194).
Other OPs have also been evaluated in relation to
cell adhesion. Specific genes significantly altered by OPs were
detected in Caenorhabditis elegans; among them, genes
associated with cell adhesion were affected, including C3C12.5,
mua-6 (ifa-2), and zig-7 (195). Other studies on these types of
organisms have been found to be a good model for investigating the
effects of other substances, due to their similarities with mammals
(196–199). The same study also investigated
genes that were associated with metabolism, including
CYTP450 and UDP-glucosyltransferases (195).
Similarly, MCF-10F cells treated with parathion or
malathion, alone or combined with estrogen, induced changes in cell
adhesion molecules such as CD146 (35), a surface protein also known as
melanoma cell adhesion molecule (200) that is involved in cell adhesion and
other processes, including cell proliferation, migration and
progression, and particularly angiogenesis and vascular
permeability (201–204). The aforementioned treatments also
upregulated keratin 18 expression; another component of epithelial
cells that protects them from external forces, serves as a
biomarker in epithelial cancers and plays other roles in drug
response and tumorigenesis (205–215).
Heat shock proteins (HSPs) are a group of highly
conserved, abundantly expressed proteins with diverse functions
(216), including the assembly and
sequestering of multi-protein complexes, transportation of
polypeptide chains across cellular membranes, regulation of protein
folding (217–221), and certain functions that protect
against stress-induced injury (137). HSPs are known as molecular
chaperones and are organized into six general families according to
their molecular weight and activity: HSP20, HSP40, HSP60, HSP70,
HSP90, and HSP100 (222). Typically,
they are proteins constitutively expressed in the cytoplasm that
co-localize to the nucleus under stress induced by physical and
chemical insult. Among them, HSP90 and HSP27 are associated with
poor prognosis and likely play an important role in drug-resistant
breast cancer (223), as well as
serve as a biomarker due to toxicant exposure (224). A previous study showed that the gene
expression of HSP27 and HSP90 was upregulated by
malathion or parathion, alone or combined with estrogen (20). The overexpression of HSP27 and
HSP90 may suggest an association between them and breast
cancer. Other HSPs such as HSP70 were also upregulated by OPs, an
upregulation positively correlated with ROS generation and
apoptotic cell death, suggesting an association between pesticides
and adverse conditions promoting cell and tissue injury (217–221).
It is well known that the cell cycle involves many
steps that can be positively or negatively regulated by several
factors. The p53 protein is a negative regulator, and its
inactivation by mutation, or its interactions with oncogene
products of DNA tumors, may lead to cancer (225–228).
Furthermore, it has been reported that malathion or parathion,
alone or combined with estrogen, led to the upregulation of the
gene expression of inducible protein TP53. TP53 was
upregulated and the mutant p53 gene expression was higher in
parathion and estrogen-treated cells, as compared with the
controls, while the TP53 gene in Li-Fraumeni syndrome was
upregulated by malathion or estrogen treatment (20). As previously mentioned, molecular
disorders, such as MSI and LOH, are associated with genomic
stability (229); furthermore, the
TP53 gene is located on chromosome 17p13. It was reported
that malathion mixed with estrogen induced MSI at loci 17p13.1
(36). This indicated that the loss
of p53 function can cause the genetic instability of these
transformed cells. Thus, mutant p53 can act as a dominant
oncogene (86). These findings
suggested that OPs and estrogen induced the malignant
transformation of the MCF-10F cell line, as shown by the phenotypic
characteristics and genomic instability indicated by LOH and MSI,
which are considered important events in the process of
carcinogenesis.
Studies have shown that breast cancer is associated
with alterations in the p53 gene in humans (230), and that mutant p53 expression
increases as breast cancer progresses from early in situ to
advanced metastatic lesions, with p53 gene mutations
observed in ~20–50% of human cancers (226,228).
On the other hand, a small number of mutations in p53 have
been found in ductal breast carcinomas (231,232).
The p53 gene was reported to be altered in 17p13.1 during
cell transformation and genotoxic stress (157,233).
p53 was found to be overexpressed in the MCF-10F cell line
when it was exposed to several carcinogens, including DMBA and BP,
and α particle (high LET) radiation in the presence of estrogen,
inducing an allelic imbalance at the respective chromosomal loci
(33,46,47).
In addition, the extent of DNA damage can be
quantitatively measured by tail moment (TM), which has been widely
used in a genotoxic study (234). An
increased TM was observed in human peripheral blood lymphocytes
following malathion and parathion exposure, as determined by a
comet assay, this study also suggested a role of oxidative stress
induced by pesticides in the cytotoxic and genotoxic process
(235). Similar results were
observed in other cell types, such as the HepG2 human liver
carcinoma cells, in which malathion also increased the extent of
DNA damage (236,237).
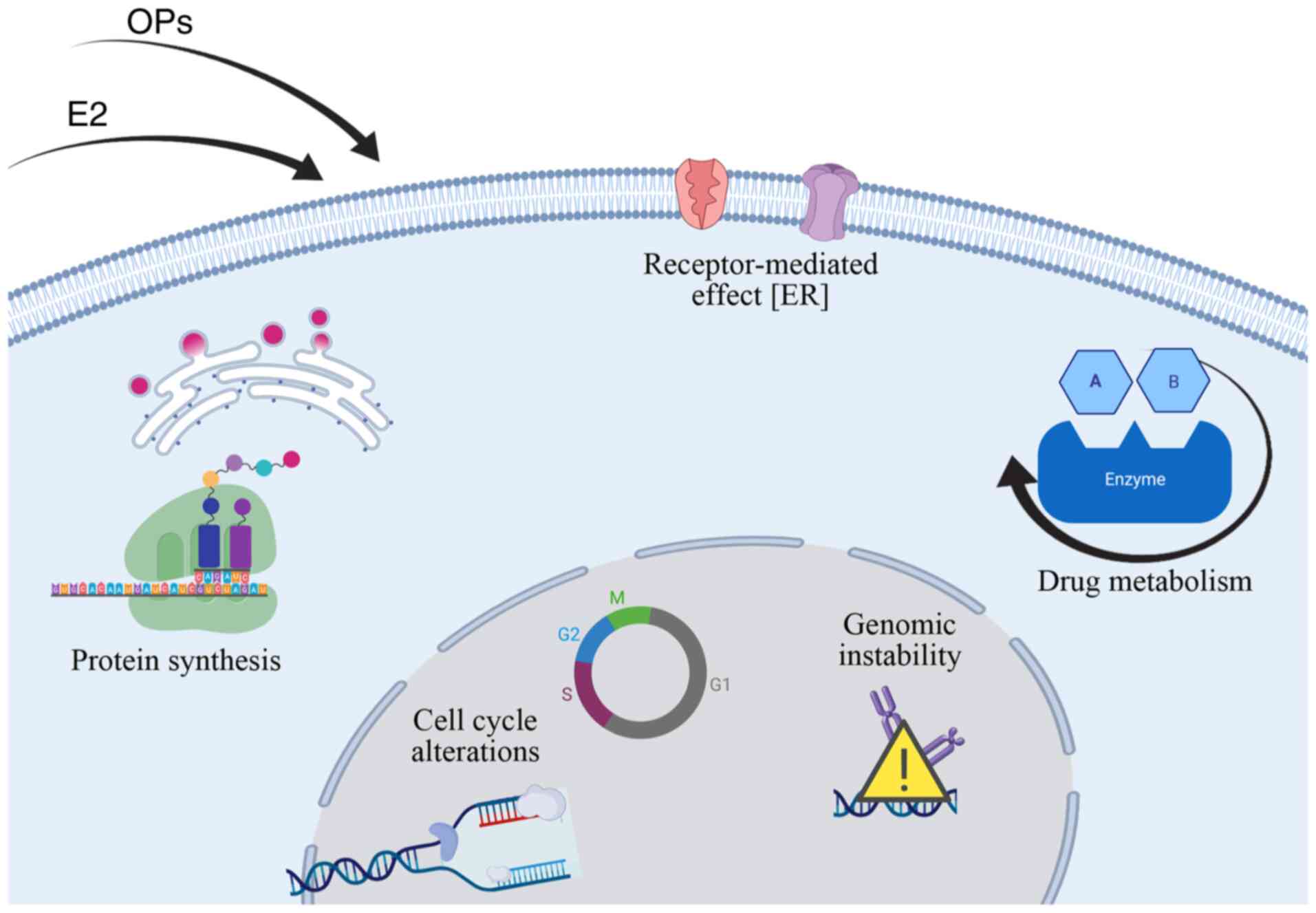
Summary of important findings
A cellular model was presented herein, which was
based on the use of the MCF-10F cell line, a human tissue-derived,
immortalized, a non-tumorigenic cell line that enables a valuable
experimental approach that minimizes extrapolation, thereby
uniquely facilitating the clinical translation of the data. The
cellular and molecular endpoints altered throughout these studies
in response to treatment with estrogen, malathion, and parathion
represent relevant endpoints for tumorigenic transformation. Of
note, this is a unique experimental approach that identifies
mechanistic signs that link OPs with human carcinogenesis.
A non-malignant cell line, MCF-10F, was used to
construct another model, which showed several signs of
carcinogenicity due to malathion and parathion exposure, which
increased cell proliferation and induced cell transformation by
affecting protein expression, promoted anchorage independence,
invasive capabilities, modulation of receptor expression, metabolic
alterations and genomic instability, among others. The mechanisms
underlying the mammary carcinogenic potential involved
acetylcholinesterase inhibition and increased oxidative stress. A
simplified scheme of the molecular changes induced by the effects
of OPs and estrogen is shown in Fig.
2.
Conclusions
Exposure to chemical compounds, such as pesticides,
and endogenous substances, such as estrogens, exert a significant
effect on normal breast cell processes at different levels.
Compounds of natural origin, such as hormones, are closely
associated with hormone-dependent types of cancer, including breast
cancer. The present study provides a comprehensive summary of the
impact of parathion, malathion, and estrogen on breast
carcinogenesis and, specifically, their effects on cell cycle,
signaling pathways linked to EGF and IGF receptors, drug
metabolism, and genomic instability in the ER-negative breast cell
line MCF-10F.
Cancer initiation and progression have been
correlated with an increase in genomic instability identified by
the inactivation of tumor-suppressor genes, and the activation of
oncogenes in the presence of malathion, parathion, and estrogen.
Moreover, advances in science and medicine have helped further
identify and elucidate the functions of useful tumor markers or
signaling molecules, further enhancing our understanding of genetic
changes that are relevant to tumor initiation. Therefore, the signs
of carcinogenicity have been proven to be very useful for analyzing
the main factors involved in breast cancer initiation and may be
used for determining the origin of other types of cancer and the
main contributing factors.
Acknowledgements
The authors would like to thank Mr. Leodán A.
Crispin (Instituto de Alta Investigación, Universidad de Tarapacá)
for providing technical support.
Funding
The present study was funded by the Convenio de
Desempeño UTA1117 (to GMC) grant from Universidad de Tarapacá and
Fondo Nacional de Ciencias grant. no. 1200656 (to GMC).
Availability of data and materials
Not applicable.
Authors' contributions
GMC prepared the original draft. GMC, TCB, and DR
reviewed and edited the manuscript. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Guyton KZ, Loomis D, Grosse Y, El
Ghissassi F, Benbrahim-Tallaa L, Guha N, Scoccianti C, Mattock H
and Straif K; International Agency for Research on Cancer Monograph
Working Group, IARC, Lyon, France, : Carcinogenicity of
tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate.
Lancet Oncol. 16:490–491. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rich JD, Gabriel SM and Schultz-Norton JR:
In vitro effects of herbicides and insecticides on human breast
cells. ISRN Toxicol. 2012:2324612012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lerro CC, Koutros S, Andreotti G, Friesen
MC, Alavanja MC, Blair A, Hoppin JA, Sandler DP, Lubin JH, Ma X, et
al: Organophosphate insecticide use and cancer incidence among
spouses of pesticide applicators in the Agricultural Health Study.
Occup Environ Med. 72:736–744. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Alavanja MC, Ross MK and Bonner MR:
Increased cancer burden among pesticide applicators and others due
to pesticide exposure. CA Cancer J Clin. 63:120–142. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sabarwal A, Kumar K and Singh RP:
Hazardous effects of chemical pesticides on human health-Cancer and
other associated disorders. Environ Toxicol Pharmacol. 63:103–114.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Popp J, Peto K and Nagy J: Pesticide
productivity and food security. A review. Agron Sustain Dev.
33:243–255. 2013. View Article : Google Scholar
|
|
7
|
Cantor KP, Blair A, Everett G, Gibson R,
Burmeister LF, Brown LM, Schuman L and Dick FR: Pesticides and
other agricultural risk factors for non-Hodgkin's lymphoma among
men in Iowa and Minnesota. Cancer Res. 52:2447–2455.
1992.PubMed/NCBI
|
|
8
|
McDuffie HH, Pahwa P, McLaughlin JR,
Spinelli JJ, Fincham S, Dosman JA, Robson D, Skinnider LF and Choi
NW: Non-Hodgkin's lymphoma and specific pesticide exposures in men:
Cross-Canada study of pesticides and health. Cancer Epidemiol
Biomarkers Prev. 10:1155–1163. 2001.PubMed/NCBI
|
|
9
|
Hu L, Luo D, Zhou T, Tao Y, Feng J and Mei
S: The association between non-Hodgkin lymphoma and organophosphate
pesticides exposure: A meta-analysis. Environ Pollut. 231:319–328.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Brown LM, Blair A, Gibson R, Everett GD,
Cantor KP, Schuman LM, Burmeister LF, Van Lier SF and Dick F:
Pesticide exposures and other agricultural risk factors for
leukemia among men in Iowa and Minnesota. Cancer Res. 50:6585–6591.
1990.PubMed/NCBI
|
|
11
|
Soldin OP, Nsouli-Maktabi H, Genkinger JM,
Loffredo CA, Ortega-Garcia JA, Colantino D, Barr DB, Luban NL, Shad
AT and Nelson D: Pediatric acute lymphoblastic leukemia and
exposure to pesticides. Ther Drug Monit. 31:495–501. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Costa LG: Organophosphorus compounds at
80: Some old and new issues. Toxicol Sci. 162:24–35. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shen K and Novak RF: DDT stimulates
c-erbB2, c-met, and STATS tyrosine phosphorylation, Grb2-Sos
association, MAPK phosphorylation, and proliferation of human
breast epithelial cells. Biochem Biophys Res Commun. 231:17–21.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bharathi SP, Raj HM, Jain S, Banerjee BD,
Ahmed T and Arora VK: Role of pesticides in the induction of tumor
angiogenesis. Anticancer Res. 33:231–240. 2013.PubMed/NCBI
|
|
15
|
Dich J, Zahm SH, Hanberg A and Adami HO:
Pesticides and cancer. Cancer Causes Control. 8:420–443. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bassil KL, Vakil C, Sanborn M, Cole DC,
Kaur JS and Kerr KJ: Cancer health effects of pesticides:
Systematic review. Can Fam Physician. 53:1704–1711. 2007.PubMed/NCBI
|
|
17
|
International Agency for Research on
Cancer (IARC), . Some organophosphate insecticides and herbicides.
IARC Working Group on the Evaluation of Carcinogenic Risks to
Humans. Monograph. 112. IARC; Lyon: 2017, https://monographs.iarc.fr/iarc-monographs-on-the-evaluation-of-carcinogenic-risks-to-humans-4/July.
2017
|
|
18
|
Blasiak J, Jaloszynski P, Trzeciak A and
Szyfter K: In vitro studies on the genotoxicity of the
organophosphorus insecticide malathion and its two analogues. Mutat
Res. 445:275–283. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Berkman CE, Quinn DA and Thompson CM:
Interaction of acetylcholinesterase with the enantiomers of
malaoxon and isomalathion. Chem Res Toxicol. 6:724–730. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Calaf GM and Roy D: Cancer genes induced
by malathion and parathion in the presence of estrogen in breast
cells. Int J Mol Med. 21:261–268. 2008.PubMed/NCBI
|
|
21
|
Calaf GM and Roy D: Gene and protein
expressions induced by 17beta-estradiol and parathion in cultured
breast epithelial cells. Mol Med. 13:255–265. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
International Agency for Research on
Cancer (IARC), . Sex Hormones (II). IARC Monographs on the
Evaluation of the Carcinogenic Risk of Chemicals to Humans.
Monograph. 21. IARC; Lyon: 1979, https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Sex-Hormones-II-1979January.
2020
|
|
23
|
International Agency for Research on
Cancer (IARC), . Overall Evaluations of Carcinogenicity: An
Updating of IARC Monographs Volumes 1–42. IARC Monographs on the
Evaluation of Carcinogenic Risks to Humans Supplement. 7. IARC;
Lyon: 1987, https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-Supplements/Overall-Evaluations-Of-Carcinogenicity-An-Updating-Of-IARC-Monographs-Volumes-1%E2%80%9342-1987January.
2020PubMed/NCBI
|
|
24
|
International Agency for Research on
Cancer (IARC), . Hormonal Contraception and Post-menopausal
Hormonal Therapy. IARC Monographs on the Evaluation of Carcinogenic
Risks to Human. Monograph. 72. IARC; Lyon: 1999, https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Hormonal-Contraception-And-Post-menopausal-Hormonal-Therapy-1999January.
2020
|
|
25
|
Bernstein L and Ross RK: Endogenous
hormones and breast cancer risk. Epidemiol Rev. 15:48–65. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Henderson BE, Ross R and Bernstein L:
Estrogens as a cause of human cancer: The richard and hinda
rosenthal foundation award lecture. Cancer Res. 48:246–253.
1988.PubMed/NCBI
|
|
27
|
NRC, . Hormonally Active Agents in the
Environmented. National Research Council Washington (DC): The
National Academies Press; 1999
|
|
28
|
Greenman SB, Rutten MJ, Fowler WM,
Scheffler L, Shortridge LA, Brown B, Sheppard BC, Deveney KE,
Deveney CW and Trunkey DD: Herbicide/pesticide effects on
intestinal epithelial growth. Environ Res. 75:85–93. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Valeron PF, Pestano JJ, Luzardo OP,
Zumbado ML, Almeida M and Boada LD: Differential effects exerted on
human mammary epithelial cells by environmentally relevant
organochlorine pesticides either individually or in combination.
Chem Biol Interact. 180:485–491. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Calaf GM and Garrido F: Catechol estrogens
as biomarkers for mammary gland cancer. Int J Oncol. 39:177–183.
2011.PubMed/NCBI
|
|
31
|
Calaf GM and Echiburu-Chau C: Synergistic
effect of malathion and estrogen on mammary gland carcinogenesis.
Oncol Rep. 28:640–646. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Echiburu-Chau C and Calaf GM: Rat lung
cancer induced by malathion and estrogen. Int J Oncol. 33:603–611.
2008.PubMed/NCBI
|
|
33
|
Calaf GM and Roy D: Gene expression
signature of parathion-transformed human breast epithelial cells.
Int J Mol Med. 19:741–750. 2007.PubMed/NCBI
|
|
34
|
Calaf GM and Roy D: Human drug metabolism
genes in parathion-and estrogen-treated breast cells. Int J Mol
Med. 20:875–881. 2007.PubMed/NCBI
|
|
35
|
Calaf GM and Roy D: Cell adhesion proteins
altered by 17beta estradiol and parathion in breast epithelial
cells. Oncol Rep. 19:165–169. 2008.PubMed/NCBI
|
|
36
|
Calaf GM, Echiburu-Chau C and Roy D:
Organophosphorous pesticides and estrogen induce transformation of
breast cells affecting p53 and c-Ha-ras genes. Int J Oncol.
35:1061–1068. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Calaf G and Russo J: Transformation of
human breast epithelial cells by chemical carcinogens.
Carcinogenesis. 14:483–492. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Calaf GM and Hei TK: Establishment of a
radiation- and estrogen-induced breast cancer model.
Carcinogenesis. 21:769–776. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Gallardo M and Calaf GM: Curcumin and
epithelial-mesenchymal transition in breast cancer cells
transformed by low doses of radiation and estrogen. Int J Oncol.
48:2534–2542. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Calaf GM: Curcumin, oxidative stress and
breast cancer, in Oxidative stress and dietary antioxidants, V. R.
Preedy. Elsevier Inc.; London, UK: pp. 159–169. 2014
|
|
41
|
Calaf GM, Echiburu-Chau C, Roy D, Chai Y,
Wen G and Balajee AS: Protective role of curcumin in oxidative
stress of breast cells. Oncol Rep. 26:1029–1035. 2011.PubMed/NCBI
|
|
42
|
Guyton KZ, Rieswijk L, Wang A, Chiu WA and
Smith MT: Key Characteristics Approach to Carcinogenic Hazard
Identification. Chem Res Toxicol. 31:1290–1292. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Smith MT, Guyton KZ, Gibbons CF, Fritz JM,
Portier CJ, Rusyn I, DeMarini DM, Caldwell JC, Kavlock RJ, Lambert
PF, et al: Key Characteristics of carcinogens as a basis for
organizing data on mechanisms of carcinogenesis. Environ Health
Perspect. 124:713–721. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cabello G, Valenzuela M, Vilaxa A, Duran
V, Rudolph I, Hrepic N and Calaf G: A rat mammary tumor model
induced by the organophosphorous pesticides parathion and
malathion, possibly through acetylcholinesterase inhibition.
Environ Health Perspect. 109:471–479. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gwinn MR, Whipkey DL, Tennant LB and
Weston A: Differential gene expression in normal human mammary
epithelial cells treated with malathion monitored by DNA
microarrays. Environ Health Perspect. 113:1046–10451. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Calaf GM, Alvarado ME and Hei TK: Beta
catenin is associated with breast cancer progression in
vitro. Int J Oncol. 26:913–921. 2005.PubMed/NCBI
|
|
47
|
Calaf G and Hei TK: Oncoprotein expression
in human breast epithelial cells transformed by high-LET radiation.
Int J Radiat Biol. 77:31–40. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Nicolson GL: Cell membrane fluid-mosaic
structure and cancer metastasis. Cancer Res. 75:1169–1176. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Morrissey MA, Hagedorn EJ and Sherwood DR:
Cell invasion through basement membrane: The netrin receptor DCC
guides the way. Worm. 2:e261692013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Naumann K: Synthetic Pyrethroid
Insecticides: Structures and Properties. 1 edition. Chemistry of
Plant Protection. 4. Berlin, Germany: Springer-Verlag Berlin
Heidelberg. XVI, 244. 1990, View Article : Google Scholar
|
|
51
|
Kim IY, Shin JH, Kim HS, Lee SJ, Kang IH,
Kim TS, Moon HJ, Choi KS, Moon A and Han SY: Assessing estrogenic
activity of pyrethroid insecticides using in vitro combination
assays. J Reprod Dev. 50:245–255. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Isoda H, Talorete TP, Han J, Oka S, Abe Y
and Inamori Y: Effects of organophosphorous pesticides used in
china on various mammalian cells. Environ Sci. 12:9–19.
2005.PubMed/NCBI
|
|
53
|
Calaf GM: Susceptibility of human breast
epithelial cells in vitro to hormones and drugs. Int J
Oncol. 28:285–295. 2006.PubMed/NCBI
|
|
54
|
Fernandez PL, Jares P, Rey MJ, Campo E and
Cardesa A: Cell cycle regulators and their abnormalities in breast
cancer. Mol Pathol. 51:305–309. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Tenga MJ and Lazar IM: Proteomic snapshot
of breast cancer cell cycle: G1/S transition point. Proteomics.
13:48–60. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Stopper H, Schmitt E, Gregor C, Mueller SO
and Fischer WH: Increased cell proliferation is associated with
genomic instability: Elevated micronuclei frequencies in
estradiol-treated human ovarian cancer cells. Mutagenesis.
18:243–247. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Deshpande A, Sicinski P and Hinds PW:
Cyclins and cdks in development and cancer: A perspective.
Oncogene. 24:2909–2915. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Hydbring P, Malumbres M and Sicinski P:
Non-canonical functions of cell cycle cyclins and cyclin-dependent
kinases. Nat Rev Mol Cell Biol. 17:280–292. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Petersen BO, Lukas J, Sorensen CS, Bartek
J and Helin K: Phosphorylation of mammalian CDC6 by cyclin A/CDK2
regulates its subcellular localization. EMBO J. 18:396–410. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Saxena P: Comparative prediction of
binding site of organophosphorus, carbamate and synthetic
pyrethroid pesticides on human cyclin-dependent protein kinases
Cdk2 and Cdk4. J Entomol Zoology Stud. 2:106–110. 2014.
|
|
61
|
Okuda T, Cleveland JL and Downing JR:
PCTAIRE-1 and PCTAIRE-3, two members of a novel cdc2/CDC28-related
protein kinase gene family. Oncogene. 7:2249–2258. 1992.PubMed/NCBI
|
|
62
|
Serrano M, Hannon GJ and Beach D: A new
regulatory motif in cell-cycle control causing specific inhibition
of cyclin D/CDK4. Nature. 366:704–707. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Kato JY, Matsuoka M, Strom DK and Sherr
CJ: Regulation of cyclin D-dependent kinase 4 (cdk4) by
cdk4-activating kinase. Mol Cell Biol. 14:2713–2721. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
MacLachlan TK, Sang N and Giordano A:
Cyclins, cyclin-dependent kinases and cdk inhibitors: Implications
in cell cycle control and cancer. Crit Rev Eukaryot Gene Expr.
5:127–156. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Tye BK: MCM proteins in DNA replication.
Annu Rev Biochem. 68:649–486. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Labib K, Tercero JA and Diffley JF:
Uninterrupted MCM2-7 function required for DNA replication fork
progression. Science. 288:1643–1647. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ryu S and Driever W: Minichromosome
maintenance proteins as markers for proliferation zones during
embryogenesis. Cell Cycle. 5:1140–1142. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Ren B, Cam H, Takahashi Y, Volkert T,
Terragni J, Young RA and Dynlacht BD: E2F integrates cell cycle
progression with DNA repair, replication, and G(2)/M checkpoints.
Genes Dev. 16:245–256. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Pereverzeva I, Whitmire E, Khan B and Coue
M: Distinct phosphoisoforms of the Xenopus Mcm4 protein regulate
the function of the Mcm complex. Mol Cell Biol. 20:3667–3676. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ishimi Y, Komamura-Kohno Y, You Z, Omori A
and Kitagawa M: Inhibition of Mcm4,6,7 helicase activity by
phosphorylation with cyclin A/Cdk2. J Biol Chem. 275:16235–16241.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Fujita M, Yamada C, Tsurumi T, Hanaoka F,
Matsuzawa K and Inagaki M: Cell cycle- and chromatin binding
state-dependent phosphorylation of human MCM heterohexameric
complexes. A role for cdc2 kinase. J Biol Chem. 273:17095–17101.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Schories B, Engel K, Dorken B, Gossen M
and Bommert K: Characterization of apoptosis-induced Mcm3 and Cdc6
cleavage reveals a proapoptotic effect for one Mcm3 fragment. Cell
Death Differ. 11:940–942. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Koonin EV: A common set of conserved
motifs in a vast variety of putative nucleic acid-dependent ATPases
including MCM proteins involved in the initiation of eukaryotic DNA
replication. Nucleic Acids Res. 21:2541–2547. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Helfenstein A, Frahm SO, Krams M, Drescher
W, Parwaresch R and Hassenpflug J: Minichromosome maintenance
protein (MCM6) in low-grade chondrosarcoma: Distinction from
enchondroma and identification of progressive tumors. Am J Clin
Pathol. 122:912–918. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Duan X, Yang Y, Wang S, Feng X, Wang T,
Wang P, Liu S, Li L, Yao W, Cui L and Wang W: Changes in the
expression of genes involved in cell cycle regulation and the
relative telomere length in the process of canceration induced by
omethoate. Tumour Biol. 39:10104283177197822017. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Qiu YL, Wang W, Wang T, Liu J, Sun P, Qian
J, Jin L and Xia ZL: Genetic polymorphisms, messenger RNA
expression of p53, p21, and CCND1, and possible links with
chromosomal aberrations in Chinese vinyl chloride-exposed workers.
Cancer Epidemiol Biomarkers Prev. 17:2578–2584. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Lindstrom MS, Jin A, Deisenroth C, White
Wolf G and Zhang Y: Cancer-associated mutations in the MDM2 zinc
finger domain disrupt ribosomal protein interaction and attenuate
MDM2-induced p53 degradation. Mol Cell Biol. 27:1056–1068. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Lee SS, Bohrson C, Pike AM, Wheelan SJ and
Greider CW: ATM kinase is required for telomere elongation in mouse
and human cells. Cell Rep. 13:1623–1632. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Yang-Snyder J, Miller JR, Brown JD, Lai CJ
and Moon RT: A frizzled homolog functions in a vertebrate Wnt
signaling pathway. Curr Biol. 6:1302–1306. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Sharma M, Castro-Piedras I, Simmons GE Jr
and Pruitt K: Dishevelled: A masterful conductor of complex Wnt
signals. Cell Signal. 47:52–64. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Masckauchan TN, Agalliu D, Vorontchikhina
M, Ahn A, Parmalee NL, Li CM, Khoo A, Tycko B, Brown AM and
Kitajewski J: Wnt5a signaling induces proliferation and survival of
endothelial cells in vitro and expression of MMP-1 and Tie-2. Mol
Biol Cell. 17:5163–5172. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Sussman DJ, Klingensmith J, Salinas P,
Adams PS, Nusse R and Perrimon N: Isolation and characterization of
a mouse homolog of the Drosophila segment polarity gene
dishevelled. Dev Biol. 166:73–86. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Testa JR, Zhou JY, Bell DW and Yen TJ:
Chromosomal localization of the genes encoding the kinetochore
proteins CENPE and CENPF to human chromosomes 4q24-->q25 and
1q32-->q41, respectively, by fluorescence in situ hybridization.
Genomics. 23:691–693. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Trinh BN, Ong CN, Coetzee GA, Yu MC and
Laird PW: Thymidylate synthase: A novel genetic determinant of
plasma homocysteine and folate levels. Hum Genet. 111:299–302.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Chang H, Qi X, Trieu Y, Xu W, Reader JC,
Ning Y and Reece D: Multiple myeloma patients with CKS1B gene
amplification have a shorter progression-free survival
post-autologous stem cell transplantation. Br J Haematol.
135:486–491. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Russo J, Tahin Q, Lareef MH, Hu YF and
Russo IH: Neoplastic transformation of human breast epithelial
cells by estrogens and chemical carcinogens. Environ Mol Mutagen.
39:254–263. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Reynolds FH Jr, Todaro GJ, Fryling C and
Stephenson JR: Human transforming growth factors induce tyrosine
phosphorylation of EGF receptors. Nature. 292:259–262. 1981.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Hines SJ, Litz JS and Krystal GW:
Coexpression of c-kit and stem cell factor in breast cancer results
in enhanced sensitivity to members of the EGF family of growth
factors. Breast Cancer Res Treat. 58:1–10. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Wee P and Wang Z: Epidermal growth factor
receptor cell proliferation signaling pathways. Cancers (Basel).
9:522017. View Article : Google Scholar
|
|
90
|
Wang Z: ErbB receptors and cancer. Methods
Mol Biol. 1652:3–35. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Nicholson RI, Gee JM and Harper ME: EGFR
and cancer prognosis. Eur J Cancer. 37 (Suppl 4):S9–S15. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Lippman M, Bolan G and Huff K: The effects
of estrogens and antiestrogens on hormone-responsive human breast
cancer in long-term tissue culture. Cancer Res. 36:4595–4601.
1976.PubMed/NCBI
|
|
93
|
Dickson RB and Lippman ME: Control of
human breast cancer by estrogen, growth factors, and oncogenes.
Cancer Treat Res. 40:119–165. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Yamamoto KR: Steroid receptor regulated
transcription of specific genes and gene networks. Annu Rev Genet.
19:209–252. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Marino M, Galluzzo P and Ascenzi P:
Estrogen signaling multiple pathways to impact gene transcription.
Curr Genomics. 7:497–508. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Pratt WB and Toft DO: Steroid receptor
interactions with heat shock protein and immunophilin chaperones.
Endocr Rev. 18:306–360. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Yasar P, Ayaz G, User SD, Gupur G and
Muyan M: Molecular mechanism of estrogen-estrogen receptor
signaling. Reprod Med Biol. 16:4–20. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Safe S: Transcriptional activation of
genes by 17 beta-estradiol through estrogen receptor-Sp1
interactions. Vitam Horm. 62:231–252. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Bjornstrom L and Sjoberg M: Mutations in
the estrogen receptor DNA-binding domain discriminate between the
classical mechanism of action and cross-talk with Stat5b and
activating protein 1 (AP-1). J Biol Chem. 277:48479–48483. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Garcia-Becerra R, Santos N, Diaz L and
Camacho J: Mechanisms of resistance to endocrine therapy in breast
cancer: Focus on signaling pathways, miRNAs and genetically based
resistance. Int J Mol Sci. 14:108–145. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Felipe Lima J, Nofech-Mozes S, Bayani J
and Bartlett JM: EMT in breast carcinoma-a review. J Clin Med.
5:652016. View Article : Google Scholar
|
|
102
|
Calaf GM, Bleak TC, Munoz JP and Aguayo F:
Markers of epithelial-mesenchymal transition in an experimental
breast cancer model induced by organophosphorous pesticides and
estrogen. Oncol Lett. 20:842020. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Conney AH, Welch RM, Kuntzman R and Burns
JJ: Effects of pesticides on drug and steroid metabolism. Clin
Pharmacol Ther. 8:2–10. 1967. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Taxak N and Bharatam PV: Drug metabolism:
A fascinating link between chemistry and biology. Resonance.
19:259–282. 2014. View Article : Google Scholar
|
|
105
|
Di L and Kerns EH: Metabolic Stability, in
Drug-Like Properties: Concepts, Structure Design and Methods from
ADME to Toxicity Optimization, E. H. K. Li Di. Elsevier; pp.
161–194. 2016
|
|
106
|
Chen TL, Lin CJ and Liu CC: Cytochrome
P450-dependent monooxygenase system and anesthetics. Acta
Anaesthesiol Sin. 33:185–194. 1995.(In Chinese). PubMed/NCBI
|
|
107
|
Bernstein L, Ross RK, Pike MC, Brown JB
and Henderson BE: Hormone levels in older women: A study of
post-menopausal breast cancer patients and healthy population
controls. Br J Cancer. 61:298–302. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Dubey RK and Jackson EK: Estrogen-induced
cardiorenal protection: Potential cellular, biochemical, and
molecular mechanisms. Am J Physiol Renal Physiol. 280:F365–F388.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Yue W, Santen RJ, Wang JP, Li Y, Verderame
MF, Bocchinfuso WP, Korach KS, Devanesan P, Todorovic R, Rogan EG
and Cavalieri EL: Genotoxic metabolites of estradiol in breast:
Potential mechanism of estradiol induced carcinogenesis. J Steroid
Biochem Mol Biol. 86:477–486. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Williamson LM and Lees-Miller SP: Estrogen
receptor alpha-mediated transcription induces cell cycle-dependent
DNA double-strand breaks. Carcinogenesis. 32:279–285. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Yang KJ, Lee J and Park HL:
Organophosphate pesticide exposure and breast cancer risk: A rapid
review of human, animal, and cell-based studies. Int J Environ Res
Public Health. 17:50302020. View Article : Google Scholar
|
|
112
|
Telang NT, Suto A, Wong GY, Osborne MP and
Bradlow HL: Induction by estrogen metabolite 16
alpha-hydroxyestrone of genotoxic damage and aberrant proliferation
in mouse mammary epithelial cells. J Natl Cancer Inst. 84:634–638.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Bay BH, Jin R, Huang J and Tan PH:
Metallothionein as a prognostic biomarker in breast cancer. Exp
Biol Med (Maywood). 231:1516–1521. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Si M and Lang J: The roles of
metallothioneins in carcinogenesis. J Hematol Oncol. 11:1072018.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Lai Y, Yip GW and Bay BH: Targeting
metallothionein for prognosis and treatment of breast cancer.
Recent Pat Anticancer Drug Discov. 6:178–185. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Jin R, Chow VT, Tan PH, Dheen ST, Duan W
and Bay BH: Metallothionein 2A expression is associated with cell
proliferation in breast cancer. Carcinogenesis. 23:81–86. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Nascimento H, Coy CS, Teori MT, Boin IF,
Goes JR, Costa FF and Lima CS: Possible influence of glutathione
S-transferase GSTT1 null genotype on age of onset of sporadic
colorectal adenocarcinoma. Dis Colon Rectum. 46:510–515. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Rebbeck TR: Molecular epidemiology of the
human glutathione S-transferase genotypes GSTM1 and GSTT1 in cancer
susceptibility. Cancer Epidemiol Biomarkers Prev. 6:733–743.
1997.PubMed/NCBI
|
|
119
|
Dalhoff K, Buus Jensen K and Enghusen
Poulsen H: Cancer and molecular biomarkers of phase 2. Methods
Enzymol. 400:618–627. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Slattery ML, Curtin K, Ma K, Schaffer D,
Potter J and Samowitz W: GSTM-1 and NAT2 and genetic alterations in
colon tumors. Cancer Causes Control. 13:527–534. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Liu X, Li Z, Zhang Z, Zhang W, Li W, Xiao
Z, Liu H, Jiao H, Wang Y and Li G: Meta-analysis of GSTM1 null
genotype and lung cancer risk in Asians. Med Sci Monit.
20:1239–1245. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Yu C, Hequn C, Longfei L, Long W, Zhi C,
Feng Z, Jinbo C, Chao L and Xiongbing Z: GSTM1 and GSTT1
polymorphisms are associated with increased bladder cancer risk:
Evidence from updated meta-analysis. Oncotarget. 8:3246–3258. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Martignano F, Gurioli G, Salvi S, Calistri
D, Costantini M, Gunelli R, De Giorgi U, Foca F and Casadio V:
GSTP1 methylation and protein expression in prostate cancer:
Diagnostic implications. Dis Markers. 2016:43582922016. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Gamage N, Barnett A, Hempel N, Duggleby
RG, Windmill KF, Martin JL and McManus ME: Human sulfotransferases
and their role in chemical metabolism. Toxicol Sci. 90:5–22. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Paul P, Suwan J, Liu J, Dordick JS and
Linhardt RJ: Recent advances in sulfotransferase enzyme activity
assays. Anal Bioanal Chem. 403:1491–1500. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Hajishengallis GN and Apostolopoulos AX:
Glucosyltransferase (GTF) and immunization against dental caries in
humans. Odontostomatol Proodos. 43:315–321. 1989.(In Modern Greek).
PubMed/NCBI
|
|
127
|
Breton C, Snajdrova L, Jeanneau C, Koca J
and Imberty A: Structures and mechanisms of glycosyltransferases.
Glycobiology. 16:29R–37R. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Maiti S and Nazmeen A: Impaired redox
regulation of estrogen metabolizing proteins is important
determinant of human breast cancers. Cancer Cell Int. 19:1112019.
View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Subbarayan K and Seliger B:
Tumor-dependent effects of proteoglycans and various
glycosaminoglycan synthesizing enzymes and sulfotransferases on
patients' outcome. Curr Cancer Drug Targets. 19:210–221. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Collier AC, Yamauchi Y, Sato BL, Rougee LR
and Ward MA: UDP-glucuronosyltransferase 1a enzymes are present and
active in the mouse blastocyst. Drug Metab Dispos. 42:1921–1925.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Kirkman H: Estrogen-induced tumors of the
kidney. III. Growth characteristics in the Syrian hamster. Natl
Cancer Inst Monogr. 1:1–57. 1959.PubMed/NCBI
|
|
132
|
Li JJ, Li SA, Klicka JK, Parsons JA and
Lam LK: Relative carcinogenic activity of various synthetic and
natural estrogens in the Syrian hamster kidney. Cancer Res.
43:5200–5204. 1983.PubMed/NCBI
|
|
133
|
Liehr JG, Fang WF, Sirbasku DA and
Ari-Ulubelen A: Carcinogenicity of catechol estrogens in syrian
hamsters. J Steroid Biochem. 24:353–356. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Liehr JG and Ricci MJ: 4-Hydroxylation of
estrogens as marker of human mammary tumors. Proc Natl Acad Sci
USA. 93:3294–3296. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Zhu BT and Conney AH: Functional role of
estrogen metabolism in target cells: Review and perspectives.
Carcinogenesis. 19:1–27. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Palackal NT, Lee SH, Harvey RG, Blair IA
and Penning TM: Activation of polycyclic aromatic hydrocarbon
trans-dihydrodiol proximate carcinogens by human aldo-keto
reductase (AKR1C) enzymes and their functional overexpression in
human lung carcinoma (A549) cells. J Biol Chem. 277:24799–24808.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Satoh T and Gupta RC: Anticholinesterase
Pesticides: Metabolism, Neurotoxicity, and Epidemiologyed. John
Wiley & Sons; pp. 6442011
|
|
138
|
Barski OA, Tipparaju SM and Bhatnagar A:
The aldo-keto reductase superfamily and its role in drug metabolism
and detoxification. Drug Metab Rev. 40:553–624. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Raif A, Marshall GM, Bell JL, Koach J, Tan
O, D'Andreti C, Thomas W, Sekyere E, Norris M, Haber M, et al: The
estrogen-responsive B box protein (EBBP) restores retinoid
sensitivity in retinoid-resistant cancer cells via effects on
histone acetylation. Cancer Lett. 277:82–90. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Lasram MM, Bouzid K, Douib IB, Annabi A,
El Elj N, El Fazaa S, Abdelmoula J and Gharbi N: Lipid metabolism
disturbances contribute to insulin resistance and decrease insulin
sensitivity by malathion exposure in Wistar rat. Drug Chem Toxicol.
38:227–234. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Rahimi R and Abdollahi M: A review on the
mechanisms involved in hyperglycemia induced by organophosphorus
pesticides. Pest Biochem Physiol. 88:115–121. 2007. View Article : Google Scholar
|
|
142
|
Lasram MM, Annabi AB, El Elj N, Selmi S,
Kamoun A, El-Fazaa S and Gharbi N: Metabolic disorders of acute
exposure to malathion in adult Wistar rats. J Hazard Mater.
163:1052–1055. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Lasram MM, Annabi AB, Rezg R, Elj N, Selmi
S, Kamoun A, El-Fazaa S and Gharbi N: Effect of short-time
malathion administration on glucose homeostasis in Wistar rat.
Pestic Biochem Phys. 92:114–119. 2008. View Article : Google Scholar
|
|
144
|
Panahi P, Vosough-Ghanbari S,
Pournourmohammadi S, Ostad SN, Nikfar S, Minaie B and Abdollahi M:
Stimulatory effects of malathion on the key enzymes activities of
insulin secretion in langerhans islets, glutamate dehydrogenase and
glucokinase. Toxicol Mech Methods. 16:161–167. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Abdollahi M, Donyavi M, Pournourmohammadi
S and Saadat M: Hyperglycemia associated with increased hepatic
glycogen phosphorylase and phosphoenolpyruvate carboxykinase in
rats following subchronic exposure to malathion. Comp Biochem
Physiol C Toxicol Pharmacol. 137:343–347. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Kalender S, Uzun FG, Durak D, Demir F and
Kalender Y: Malathion-induced hepatotoxicity in rats: The effects
of vitamins C and E. Food Chem Toxicol. 48:633–638. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Rezg R, Mornagui B, Benahmed M, Chouchane
SG, Belhajhmida N, Abdeladhim M, Kamoun A, El-fazaa S and Gharbi N:
Malathion exposure modulates hypothalamic gene expression and
induces dyslipedemia in Wistar rats. Food Chem Toxicol.
48:1473–1477. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Lassiter TL, Ryde IT, Levin ED, Seidler FJ
and Slotkin TA: Neonatal exposure to parathion alters lipid
metabolism in adulthood: Interactions with dietary fat intake and
implications for neurodevelopmental deficits. Brain Res Bull.
81:85–91. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Fang H and Judd RL: Adiponectin regulation
and function. Compr Physiol. 8:1031–1063. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Josephs SF, Ichim TE, Prince SM, Kesari S,
Marincola FM, Escobedo AR and Jafri A: Unleashing endogenous
TNF-alpha as a cancer immunotherapeutic. J Transl Med. 16:2422018.
View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Wajant H: The role of TNF in cancer.
Results Probl Cell Differ. 49:1–15. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Wang H and Wang WJ: Relationship between
CYP1A1 polymorphisms and invasion and metastasis of breast cancer.
Asian Pac J Trop Med. 6:835–838. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Broustas CG and Lieberman HB: DNA damage
response genes and the development of cancer metastasis. Radiat
Res. 181:111–130. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Elenbaas B, Spirio L, Koerner F, Fleming
MD, Zimonjic DB, Donaher JL, Popescu NC, Hahn WC and Weinberg RA:
Human breast cancer cells generated by oncogenic transformation of
primary mammary epithelial cells. Genes Dev. 15:50–65. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Schubbert S, Shannon K and Bollag G:
Hyperactive Ras in developmental disorders and cancer. Nat Rev
Cancer. 7:295–308. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Roy D, Calaf G and Hei TK: Allelic
imbalance at 11p15.5-15.4 correlated with c-Ha-ras mutation during
radiation-induced neoplastic transformation of human breast
epithelial cells. Int J Cancer. 103:730–737. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Roy D, Calaf G and Hei TK: Frequent
allelic imbalance on chromosome 6 and 17 correlate with
radiation-induced neoplastic transformation of human breast
epithelial cells. Carcinogenesis. 22:1685–1692. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Zhang PL, Calaf G and Russo J: Allele loss
and point mutation in codons 12 and 61 of the c-Ha-ras oncogene in
carcinogen-transformed human breast epithelial cells. Mol Carcinog.
9:46–56. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Lin JC, Wang CC, Jiang RS, Wang WY and Liu
SA: Microsatellite alteration in head and neck squamous cell
carcinoma patients from a betel quid-prevalent region. Sci Rep.
6:226142016. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Hadziavdic V, Pavlovic-Calic N and
Eminovic I: Microsatellite instability and loss of heterozygosity
of tumor suppressor genes in Bosnian patients with sporadic
colorectal cancer. Bosn J Basic Med Sci. 8:313–321. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Chambuso R, Kaambo E, Denny L, Gray CM,
Williamson AL, Migdalska-Sek M, Agenbag G, Rebello G and Ramesar R:
Investigation of cervical tumor biopsies for chromosomal loss of
heterozygosity (LOH) and microsatellite instability (MSI) at the
HLA II locus in HIV-1/HPV Co-infected Women. Front Oncol.
9:9512019. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Boland CR and Goel A: Microsatellite
instability in colorectal cancer. Gastroenterology.
138:2073–2087.e3. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Kim TM and Park PJ: A genome-wide view of
microsatellite instability: Old stories of cancer mutations
revisited with new sequencing technologies. Cancer Res.
74:6377–6382. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Banys-Paluchowski M, Fehm T, Janni W,
Aktas B, Fasching PA, Kasimir-Bauer S, Milde-Langosch K, Pantel K,
Rack B, Riethdorf S, et al: Elevated serum RAS p21 is an
independent prognostic factor in metastatic breast cancer. BMC
Cancer. 18:5412018. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Spandidos DA, Karaiossifidi H, Malliri A,
Linardopoulos S, Vassilaros S, Tsikkinis A and Field JK: Expression
of ras Rb1 and p53 proteins in human breast cancer. Anticancer Res.
12:81–89. 1992.PubMed/NCBI
|
|
166
|
Calaf GM and Abarca-Quinones J: Ras
protein expression as a marker for breast cancer. Oncol Lett.
11:3637–3642. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Didsbury J, Weber RF, Bokoch GM, Evans T
and Snyderman R: Rac, a novel ras-related family of proteins that
are botulinum toxin substrates. J Biol Chem. 264:16378–16382. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Wennerberg K, Rossman KL and Der CJ: The
Ras superfamily at a glance. J Cell Sci. 118:843–846. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Fritz G, Brachetti C, Bahlmann F, Schmidt
M and Kaina B: Rho GTPases in human breast tumours: Expression and
mutation analyses and correlation with clinical parameters. Br J
Cancer. 87:635–644. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Kleer CG, van Golen KL, Zhang Y, Wu ZF,
Rubin MA and Merajver SD: Characterization of RhoC expression in
benign and malignant breast disease: A potential new marker for
small breast carcinomas with metastatic ability. Am J Pathol.
160:579–584. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Bustelo XR, Sauzeau V and Berenjeno IM:
GTP-binding proteins of the Rho/Rac family: Regulation, effectors
and functions in vivo. Bioessays. 29:356–370. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Skowronek KR, Guo F, Zheng Y and Nassar N:
The C-terminal basic tail of RhoG assists the guanine nucleotide
exchange factor trio in binding to phospholipids. J Biol Chem.
279:37895–37907. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Ko CD, Kim JS, Ko BG, Son BH, Kang HJ,
Yoon HS, Cho EY, Gong G and Ahn SH: The meaning of the c-kit
proto-oncogene product in malignant transformation in human mammary
epithelium. Clin Exp Metastasis. 20:593–597. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Najafi M, Ahmadi A and Mortezaee K:
Extracellular-signal-regulated kinase/mitogen-activated protein
kinase signaling as a target for cancer therapy: An updated review.
Cell Biol Int. 43:1206–1222. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Turner N and Grose R: Fibroblast growth
factor signalling: From development to cancer. Nat Rev Cancer.
10:116–129. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Carter EP, Fearon AE and Grose RP:
Careless talk costs lives: Fibroblast growth factor receptor
signalling and the consequences of pathway malfunction. Trends Cell
Biol. 25:221–233. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Akl MR, Nagpal P, Ayoub NM, Tai B, Prabhu
SA, Capac CM, Gliksman M, Goy A and Suh KS: Molecular and clinical
significance of fibroblast growth factor 2 (FGF2/bFGF) in
malignancies of solid and hematological cancers for personalized
therapies. Oncotarget. 7:44735–44762. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Wesche J, Haglund K and Haugsten EM:
Fibroblast growth factors and their receptors in cancer. Biochem J.
437:199–213. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Zakrzewska M, Marcinkowska E and Wiedlocha
A: FGF-1: From biology through engineering to potential medical
applications. Crit Rev Clin Lab Sci. 45:91–135. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Hu Z, Evarts RP, Fujio K, Omori N, Omori
M, Marsden ER and Thorgeirsson SS: Expression of transforming
growth factor alpha/epidermal growth factor receptor, hepatocyte
growth factor/c-met and acidic fibroblast growth factor/fibroblast
growth factor receptors during hepatocarcinogenesis.
Carcinogenesis. 17:931–938. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Chow NH, Cheng KS, Lin PW, Chan SH, Su WC,
Sun YN and Lin XZ: Expression of fibroblast growth factor-1 and
fibroblast growth factor-2 in normal liver and hepatocellular
carcinoma. Dig Dis Sci. 43:2261–2266. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Yamanaka Y, Friess H, Buchler M, Beger HG,
Uchida E, Onda M, Kobrin MS and Korc M: Overexpression of acidic
and basic fibroblast growth factors in human pancreatic cancer
correlates with advanced tumor stage. Cancer Res. 53:5289–5296.
1993.PubMed/NCBI
|
|
183
|
Soufla G, Sifakis S and Spandidos DA: FGF2
transcript levels are positively correlated with EGF and IGF-1 in
the malignant endometrium. Cancer Lett. 259:146–155. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Watnick RS: The role of the tumor
microenvironment in regulating angiogenesis. Cold Spring Harb
Perspect Med. 2:a0066762012. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Schmid C: Insulin-like growth factors.
Cell Biol Int. 19:445–457. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Baxter RC: Insulin-like growth factor
binding proteins as glucoregulators. Metabolism. 44 (10 Suppl
4):S12–S17. 1995. View Article : Google Scholar
|
|
187
|
Kelley KM, Oh Y, Gargosky SE, Gucev Z,
Matsumoto T, Hwa V, Ng L, Simpson DM and Rosenfeld RG: Insulin-like
growth factor-binding proteins (IGFBPs) and their regulatory
dynamics. Int J Biochem Cell Biol. 28:619–637. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Oh Y, Nagalla SR, Yamanaka Y, Kim HS,
Wilson E and Rosenfeld RG: Synthesis and characterization of
insulin-like growth factor-binding protein (IGFBP)-7. Recombinant
human mac25 protein specifically binds IGF-I and -II. J Biol Chem.
271:30322–30325. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Mnif W, Hassine AI, Bouaziz A, Bartegi A,
Thomas O and Roig B: Effect of endocrine disruptor pesticides: A
review. Int J Environ Res Public Health. 8:2265–2303. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Takayama T, Shiozaki H, Shibamoto S, Oka
H, Kimura Y, Tamura S, Inoue M, Monden T, Ito F and Monden M:
Beta-catenin expression in human cancers. Am J Pathol. 148:39–46.
1996.PubMed/NCBI
|
|
191
|
Breen E, Steele G Jr and Mercurio AM: Role
of the E-cadherin/alpha-catenin complex in modulating cell-cell and
cell-matrix adhesive properties of invasive colon carcinoma cells.
Ann Surg Oncol. 2:378–385. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Lim SC and Lee MS: Significance of
E-cadherin/beta-catenin complex and cyclin D1 in breast cancer.
Oncol Rep. 9:915–928. 2002.PubMed/NCBI
|
|
193
|
Nakopoulou L, Gakiopoulou-Givalou H,
Karayiannakis AJ, Giannopoulou I, Keramopoulos A, Davaris P and
Pignatelli M: Abnormal alpha-catenin expression in invasive breast
cancer correlates with poor patient survival. Histopathology.
40:536–546. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Pierceall WE, Woodard AS, Morrow JS, Rimm
D and Fearon ER: Frequent alterations in E-cadherin and alpha- and
beta-catenin expression in human breast cancer cell lines.
Oncogene. 11:1319–1326. 1995.PubMed/NCBI
|
|
195
|
Lewis JA, Szilagyi M, Gehman E, Dennis WE
and Jackson DA: Distinct patterns of gene and protein expression
elicited by organophosphorus pesticides in Caenorhabditis elegans.
BMC Genomics. 10:2022009. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Williams PL and Dusenbery DB: Using the
nematode Caenorhabditis elegans to predict mammalian acute
lethality to metallic salts. Toxicol Ind Health. 4:469–478. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Morgan PG and Sedensky MM: Mutations
affecting sensitivity to ethanol in the nematode, Caenorhabditis
elegans. Alcohol Clin Exp Res. 19:1423–1429. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Bargmann CI: Neurobiology of the
Caenorhabditis elegans genome. Science. 282:2028–2033. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Cole RD, Anderson GL and Williams PL: The
nematode Caenorhabditis elegans as a model of
organophosphate-induced mammalian neurotoxicity. Toxicol Appl
Pharmacol. 194:248–256. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Jin HJ, Kwon JH, Kim M, Bae YK, Choi SJ,
Oh W, Yang YS and Jeon HB: Downregulation of melanoma cell adhesion
molecule (MCAM/CD146) accelerates cellular senescence in human
umbilical cord blood-derived mesenchymal stem cells. Stem Cells
Transl Med. 5:427–439. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Leroyer AS, Blin MG, Bachelier R, Bardin
N, Blot-Chabaud M and Dignat-George F: CD146 (Cluster of
Differentiation 146). Arterioscler Thromb Vasc Biol. 39:1026–1033.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Ye Z, Zhang C, Tu T, Sun M, Liu D, Lu D,
Feng J, Yang D, Liu F and Yan X: Wnt5a uses CD146 as a receptor to
regulate cell motility and convergent extension. Nat Commun.
4:28032013. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Kang Y, Wang F, Feng J, Yang D, Yang X and
Yan X: Knockdown of CD146 reduces the migration and proliferation
of human endothelial cells. Cell Res. 16:313–318. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Ouhtit A, Gaur RL, Abd Elmageed ZY,
Fernando A, Thouta R, Trappey AK, Abdraboh ME, El-Sayyad HI, Rao P
and Raj MG: Towards understanding the mode of action of the
multifaceted cell adhesion receptor CD146. Biochim Biophys Acta.
1795:130–136. 2009.PubMed/NCBI
|
|
205
|
Coulombe PA and Omary MB: ‘Hard’ and
‘soft’ principles defining the structure, function and regulation
of keratin intermediate filaments. Curr Opin Cell Biol. 14:110–122.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Greystoke A, Dean E, Saunders MP, Cummings
J, Hughes A, Ranson M, Dive C and Renehan AG: Multi-level evidence
that circulating CK18 is a biomarker of tumour burden in colorectal
cancer. Br J Cancer. 107:1518–1524. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Schneider J: Tumor markers in detection of
lung cancer. Adv Clin Chem. 42:1–41. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Moll R, Divo M and Langbein L: The human
keratins: Biology and pathology. Histochem Cell Biol. 129:705–733.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Ahn SK, Moon HG, Ko E, Kim HS, Shin HC,
Kim J, You JM, Han W and Noh DY: Preoperative serum tissue
polypeptide-specific antigen is a valuable prognostic marker in
breast cancer. Int J Cancer. 132:875–881. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Escobar-Hoyos LF, Shah R, Roa-Pena L,
Vanner EA, Najafian N, Banach A, Nielsen E, Al-Khalil R, Akalin A,
Talmage D, et al: Keratin-17 promotes p27KIP1 nuclear export and
degradation and offers potential prognostic utility. Cancer Res.
75:3650–3662. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Tan HS, Jiang WH, He Y, Wang DS, Wu ZJ, Wu
DS, Gao L, Bao Y, Shi JZ, Liu B, et al: KRT8 upregulation promotes
tumor metastasis and is predictive of a poor prognosis in clear
cell renal cell carcinoma. Oncotarget. 8:76189–76203. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Cheung KJ, Padmanaban V, Silvestri V,
Schipper K, Cohen JD, Fairchild AN, Gorin MA, Verdone JE, Pienta
KJ, Bader JS and Ewald AJ: Polyclonal breast cancer metastases
arise from collective dissemination of keratin 14-expressing tumor
cell clusters. Proc Natl Acad Sci USA. 113:E854–E863. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Saha SK, Choi HY, Kim BW, Dayem AA, Yang
GM, Kim KS, Yin YF and Cho SG: KRT19 directly interacts with
beta-catenin/RAC1 complex to regulate NUMB-dependent NOTCH
signaling pathway and breast cancer properties. Oncogene.
36:332–349. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Zhang B, Wang J, Liu W, Yin Y, Qian D,
Zhang H, Shi B, Li C, Zhu J, Zhang L, et al: Cytokeratin 18
knockdown decreases cell migration and increases chemosensitivity
in non-small cell lung cancer. J Cancer Res Clin Oncol.
142:2479–2487. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Yin B, Zhang M, Zeng Y, Li Y, Zhang C,
Getzenberg RH and Song Y: Downregulation of cytokeratin 18 is
associated with paclitaxelresistance and tumor aggressiveness in
prostate cancer. Int J Oncol. 48:1730–1736. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Miller DJ and Fort PE: Heat shock proteins
regulatory role in neurodevelopment. Front Neurosci. 12:8212018.
View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Schlesinger MJ, Ashburner M and Tissières
A: Heat Shock: From bacteria to man. Cold Spring Harbor Laboratory;
Cold Spring Harbor, NY: 1982
|
|
218
|
Zeilstra-Ryalls J, Fayet O and
Georgopoulos C: The universally conserved GroE (Hsp60) chaperonins.
Annu Rev Microbiol. 45:301–325. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Georgopoulos C and Welch WJ: Role of the
major heat shock proteins as molecular chaperones. Annu Rev Cell
Biol. 9:601–634. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Ohtsuka K: Cloning of a cDNA for
heat-shock protein hsp40, a human homologue of bacterial DnaJ.
Biochem Biophys Res Commun. 197:235–240. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Todd MJ, Viitanen PV and Lorimer GH:
Dynamics of the chaperonin ATPase cycle: Implications for
facilitated protein folding. Science. 265:659–566. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Meher PK, Sahu TK, Gahoi S and Rao AR:
ir-HSP: Improved recognition of heat shock proteins, their families
and sub-types based On g-Spaced Di-peptide features and support
vector machine. Front Genet. 8:2352017. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Lee CH, Hong HM, Chang YY and Chang WW:
Inhibition of heat shock protein (Hsp) 27 potentiates the
suppressive effect of Hsp90 inhibitors in targeting breast cancer
stem-like cells. Biochimie. 94:1382–1389. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Werner I, Koger CS, Hamm JT and Hinton DE:
Ontogeny of the heat shock protein, hsp70 and hsp60, response and
developmental effects of heat-shock in the teleost, medaka (Oryzias
latipes). Environmental Sci. 8:13–29. 2001.
|
|
225
|
Levine AJ, Momand J and Finlay CA: The p53
tumour suppressor gene. Nature. 351:453–456. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Callahan R: p53 mutations, another breast
cancer prognostic factor. J Natl Cancer Inst. 84:826–827. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Moll UM, Ostermeyer AG, Ahomadegbe JC,
Mathieu MC and Riou G: p53 mediated tumor cell response to
chemotherapeutic DNA damage: A preliminary study in matched pairs
of breast cancer biopsies. Hum Pathol. 26:1293–1301. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Nigro JM, Baker SJ, Preisinger AC, Jessup
JM, Hostetter R, Cleary K, Bigner SH, Davidson N, Baylin S, Devilee
P, et al: Mutations in the p53 gene occur in diverse human tumour
types. Nature. 342:705–708. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Migdalska-Sek M, Pastuszak-Lewandoska D
and Brzezianska E: MSI and LOH in the development and prognosis of
follicular cell-derived thyroid tumours. Endokrynol Pol.
63:126–136. 2012.PubMed/NCBI
|
|
230
|
Kiaris H and Spandidos D: Mutations of ras
genes in human tumors (review). Int J Oncol. 7:413–421.
1995.PubMed/NCBI
|
|
231
|
Samowitz WS, Holden JA, Curtin K, Edwards
SL, Walker AR, Lin HA, Robertson MA, Nichols MF, Gruenthal KM,
Lynch BJ, et al: Inverse relationship between microsatellite
instability and K-ras and p53 gene alterations in colon cancer. Am
J Pathol. 158:1517–1524. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Mendez O, Manas S, Peinado, Fabra A,
Escobedo A, Moreno A and Sierra A: Microsatellite instability is
associated with the loss of apoptosis in ductal breast carcinomas.
Breast Cancer Res Treat. 65:171–177. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Appella E and Anderson CW:
Post-translational modifications and activation of p53 by genotoxic
stresses. Eur J Biochem. 268:2764–2772. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Lee E, Oh E and Lee J, Sul D and Lee J:
Use of the tail moment of the lymphocytes to evaluate DNA damage in
human biomonitoring studies. Toxicol Sci. 81:121–132. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Kumar N, Yadav A, Gulati S, Priya K,
Aggarwal N and Gupta R: Effects of GST polymorphism on ameliorative
effect of curcumin and carvacrol against DNA damage induced by
combined treatment of malathion and parathion. IJT. 10:19–27. 2016.
View Article : Google Scholar
|
|
236
|
Collins AR: The comet assay for DNA damage
and repair: Principles, applications, and limitations. Mol
Biotechnol. 26:249–261. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Moore PD, Yedjou CG and Tchounwou PB:
Malathion-induced oxidative stress, cytotoxicity, and genotoxicity
in human liver carcinoma (HepG2) cells. Environ Toxicol.
25:221–226. 2010. View Article : Google Scholar : PubMed/NCBI
|