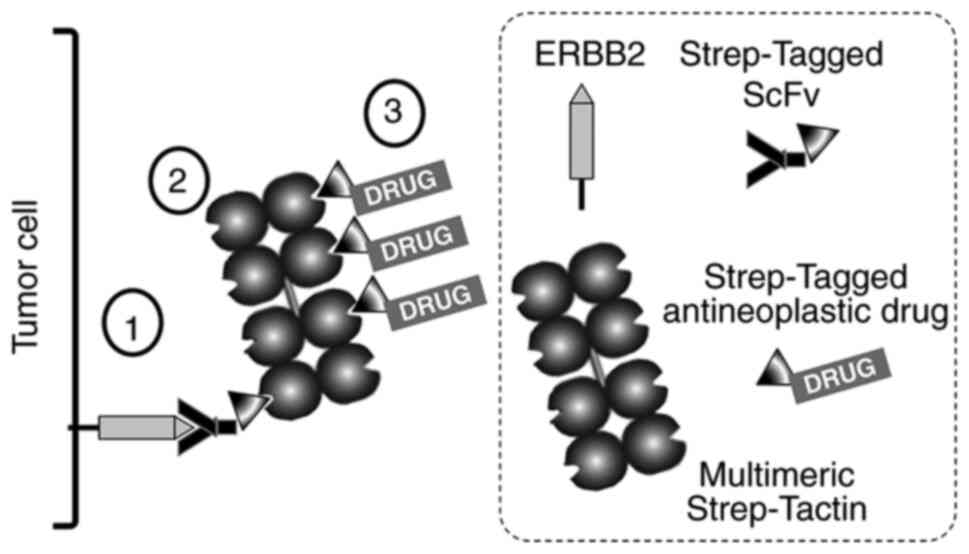
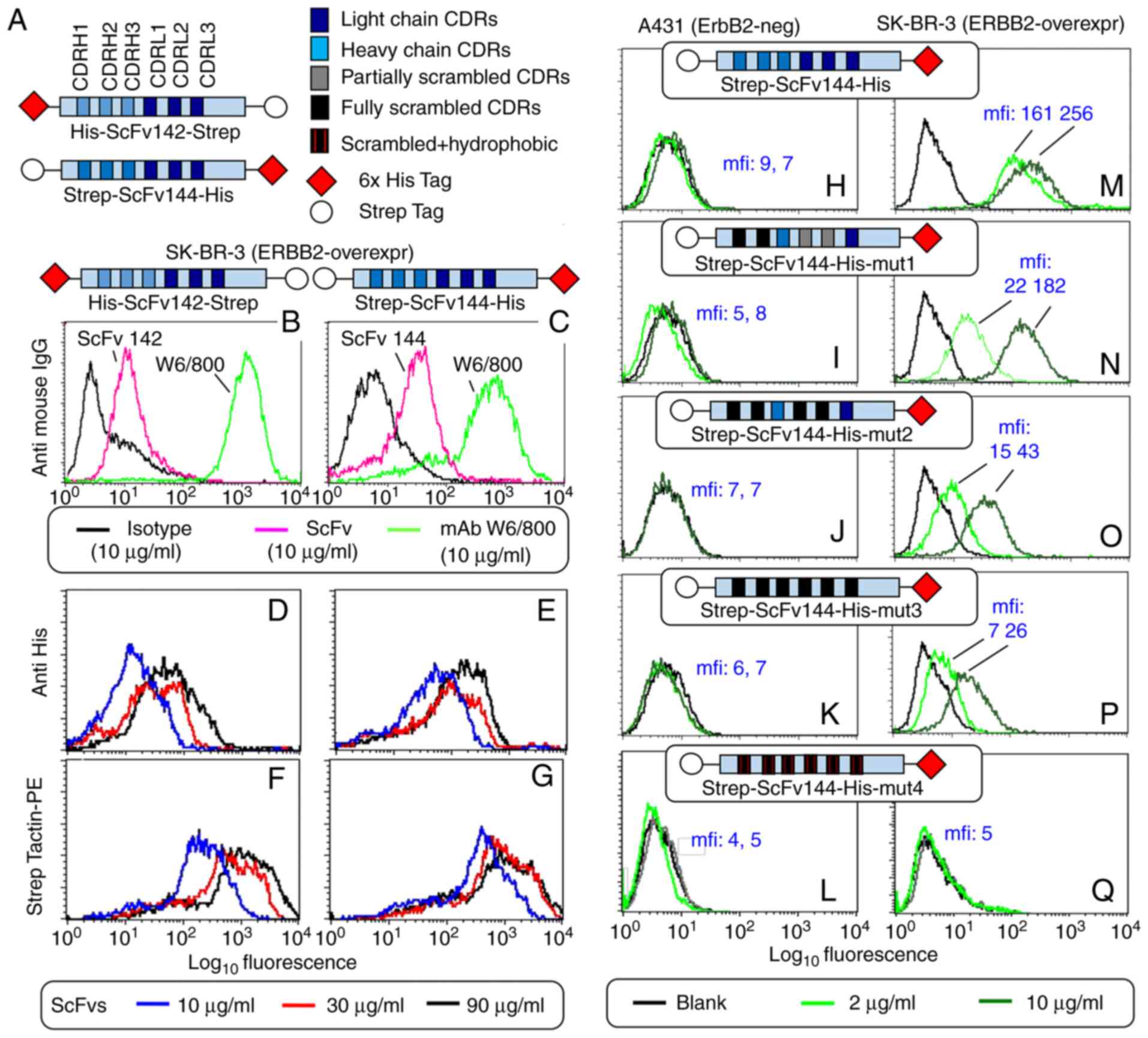
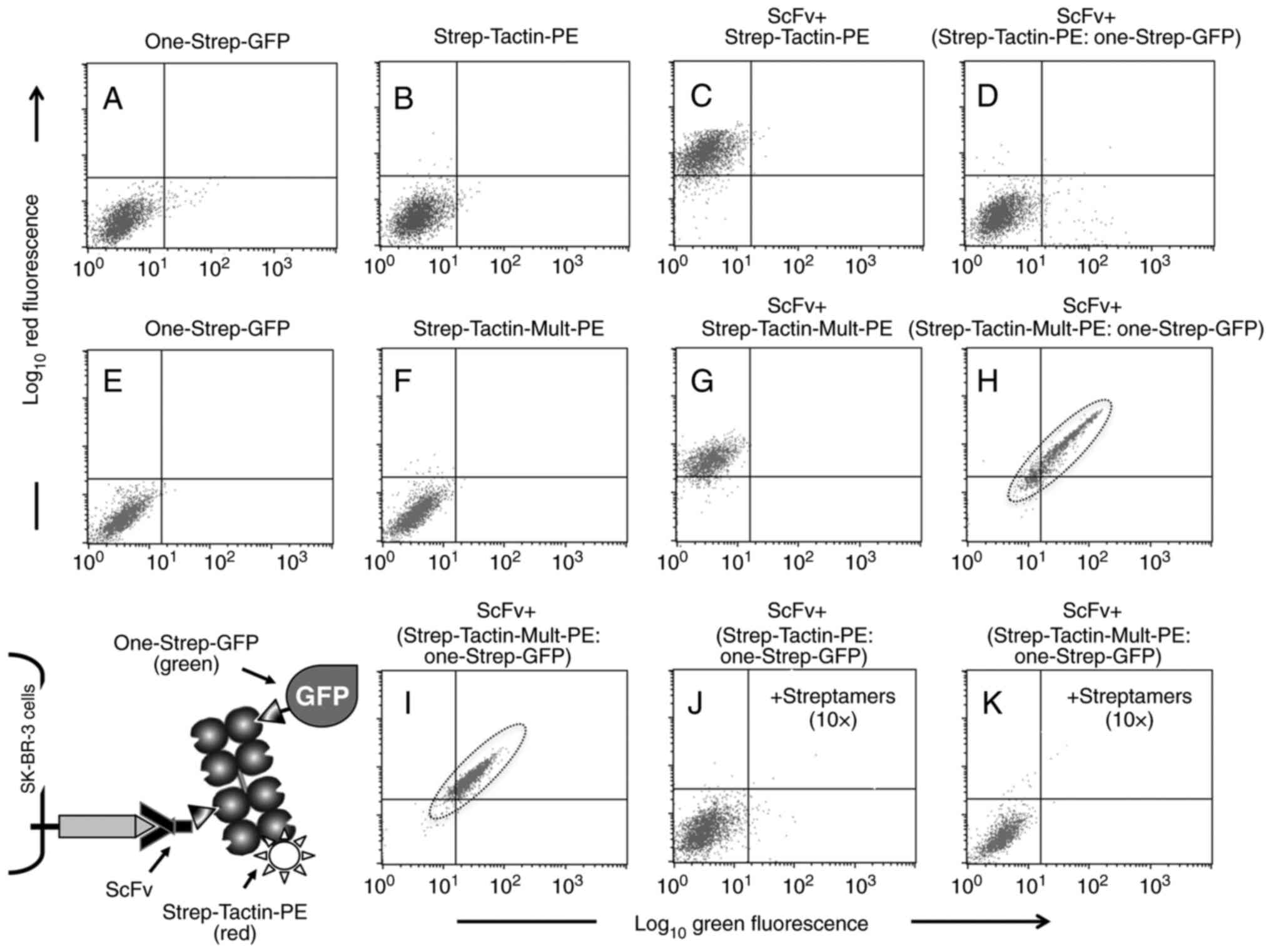
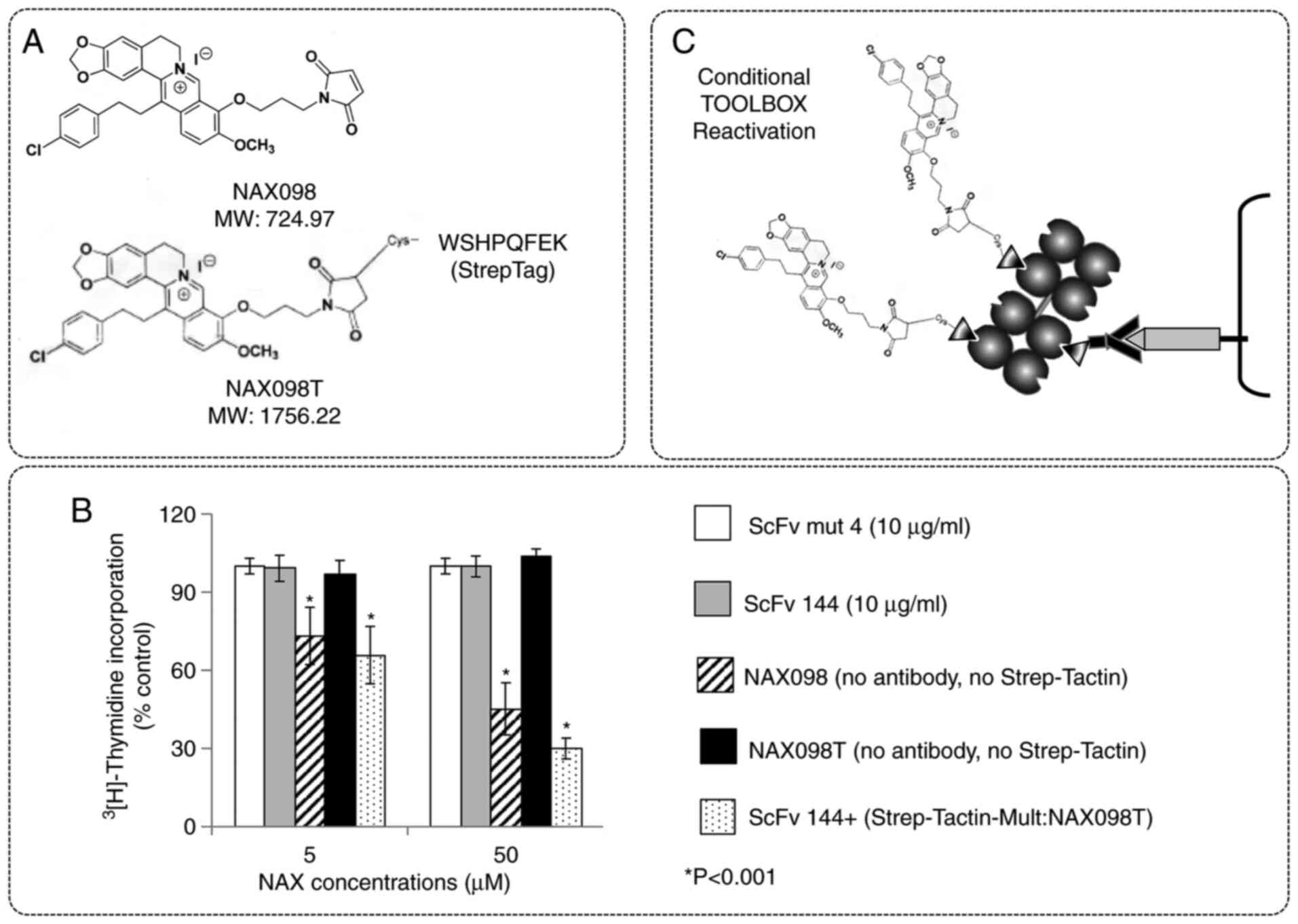
|
1
|
de Goeij BE and Lambert JM: New
developments for antibody-drug conjugate-based therapeutic
approaches. Curr Opin Immunol. 40:14–23. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Verma S, Miles D, Gianni L, Krop IE,
Welslau M, Baselga J, Pegram M, Oh DY, Diéras V, Guardino E, et al:
Trastuzumab emtansine for HER2-positive advanced breast cancer. N
Engl J Med. 367:1783–1791. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
von Minckwitz G, Huang CS, Mano MS, Loibl
S, Mamounas EP, Untch M, Wolmark N, Rastogi P, Schneeweiss A,
Redondo A, et al: Trastuzumab emtansine for residual invasive
HER2-positive breast cancer. N Engl J Med. 380:617–628. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Baselga J, Lewis Phillips GD, Verma S, Ro
J, Huober J, Guardino AE, Samant MK, Olsen S, de Haas SL and Pegram
MD: Relationship between tumor biomarkers and efficacy in EMILIA, a
phase iii study of trastuzumab emtansine in HER2-positive
metastatic breast cancer. Clin Cancer Res. 22:3755–3763. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Amadori D, Bertoni L, Flamigni A, Savini
S, De Giovanni C, Casanova S, De Paola F, Amadori A, Giulotto E and
Zoli W: Establishment and characterization of a new cell line from
primary human breast carcinoma. Breast Cancer Res Treat.
28:251–260. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chavez KJ, Garimella SV and Lipkowitz S:
Triple negative breast cancer cell lines: One tool in the search
for better treatment of triple negative breast cancer. Breast Dis.
32:35–48. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mayer IA, Abramson VG, Lehmann BD and
Pietenpol JA: New strategies for triple-negative breast
cancer-deciphering the heterogeneity. Clin Cancer Res. 20:782–790.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Giacomini P, Giorda E, Pera C and Ferrara
GB: An ID card for tumour cell lines: HLA typing can help. Lancet
Oncol. 2:6582001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Digiesi G, Giacomini P, Fraioli R, Mariani
M, Nicotra MR, Segatto O and Natali PG: Production and
characterization of murine mAbs to the extracellular domain of
human neu oncogene product GP185HER2. Hybridoma. 11:519–527. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Galeffi P, Lombardi A, Pietraforte I,
Novelli F, Di Donato M, Sperandei M, Tornambé A, Fraioli R,
Martayan A, Natali PG, et al: Functional expression of a
single-chain antibody to ErbB-2 in plants and cell-free systems. J
Transl Med. 4:392006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pierpaoli E, Fiorillo G, Lombardi P,
Salvatore C, Geroni C, Piacenza F and Provinciali M: Antitumor
activity of NAX060: A novel semisynthetic berberine derivative in
breast cancer cells. Biofactors. 44:443–452. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Schonthal A, Herrlich P, Rahmsdorf HJ and
Ponta H: Requirement for fos gene expression in the transcriptional
activation of collagenase by other oncogenes and phorbol esters.
Cell. 54:325–334. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Knutti N, Kuepper M and Friedrich K:
Soluble extracellular matrix metalloproteinase inducer (EMMPRIN,
EMN) regulates cancer-related cellular functions by homotypic
interactions with surface CD147. FEBS J. 282:4187–4200. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Stacer AC, Nyati S, Moudgil P, Iyengar R,
Luker KE, Rehemtulla A and Luker GD: NanoLuc reporter for dual
luciferase imaging in living animals. Mol Imaging. 12:1–13. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bravo R, Burckhardt J, Curran T and Muller
R: Stimulation and inhibition of growth by EGF in different A431
cell clones is accompanied by the rapid induction of c-fos and
c-myc proto-oncogenes. EMBO J. 4:1193–1197. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Daemen A and Manning G: HER2 is not a
cancer subtype but rather a pan-cancer event and is highly enriched
in AR-driven breast tumors. Breast Cancer Res. 20:82018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Modi S, Saura C, Yamashita T, Park YH, Kim
SB, Tamura K, Andre F, Iwata H, Ito Y, Tsurutani J, et al:
Trastuzumab deruxtecan in previously treated HER2-positive breast
cancer. N Engl J Med. 382:610–621. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Falvo E, Tremante E, Arcovito A, Papi M,
Elad N, Boffi A, Morea V, Conti G, Toffoli G, Fracasso G, et al:
Improved doxorubicin encapsulation and pharmacokinetics of
ferritin-fusion protein nanocarriers bearing proline, serine, and
alanine elements. Biomacromolecules. 17:514–522. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Robert B, Guillaume P, Luescher I, Romero
P and Mach JP: Antibody-conjugated MHC class I tetramers can target
tumor cells for specific lysis by T lymphocytes. Eur J Immunol.
30:3165–3170. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ogg GS, Dunbar PR, Cerundolo V, McMichael
AJ, Lemoine NR and Savage P: Sensitization of tumour cells to lysis
by virus-specific CTL using antibody-targeted MHC class I/peptide
complexes. Br J Cancer. 82:1058–1062. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Metz S, Haas AK, Daub K, Croasdale R,
Stracke J, Lau W, Georges G, Josel HP, Dziadek S, Hopfner KP, et
al: Bispecific digoxigenin-binding antibodies for targeted payload
delivery. Proc Natl Acad Sci USA. 108:8194–8199. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Stefan D, Claudio S and Ulrich B:
Engineered hapten-binding antibody derivatives for modulation of
pharmacokinetic properties of small molecules and targeted payload
delivery. Immunol Rev. 270:165–177. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Allegretti M, Fabi A, Buglioni S, Martayan
A, Conti L, Pescarmona E, Ciliberto G and Giacomini P: Tearing down
the walls: FDA approves next generation sequencing (NGS) assays for
actionable cancer genomic aberrations. J Exp Clin Cancer Res.
37:472018. View Article : Google Scholar : PubMed/NCBI
|