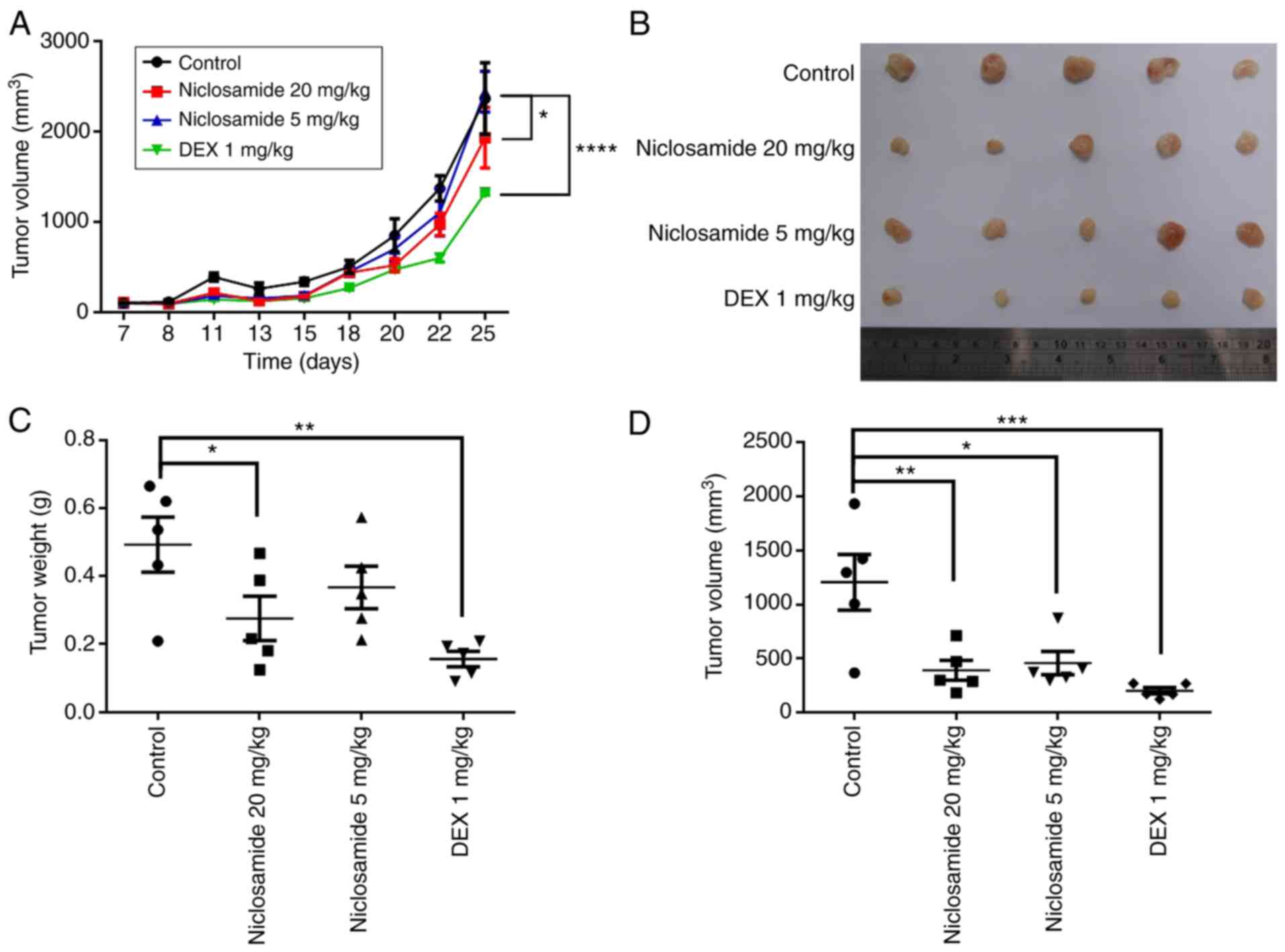
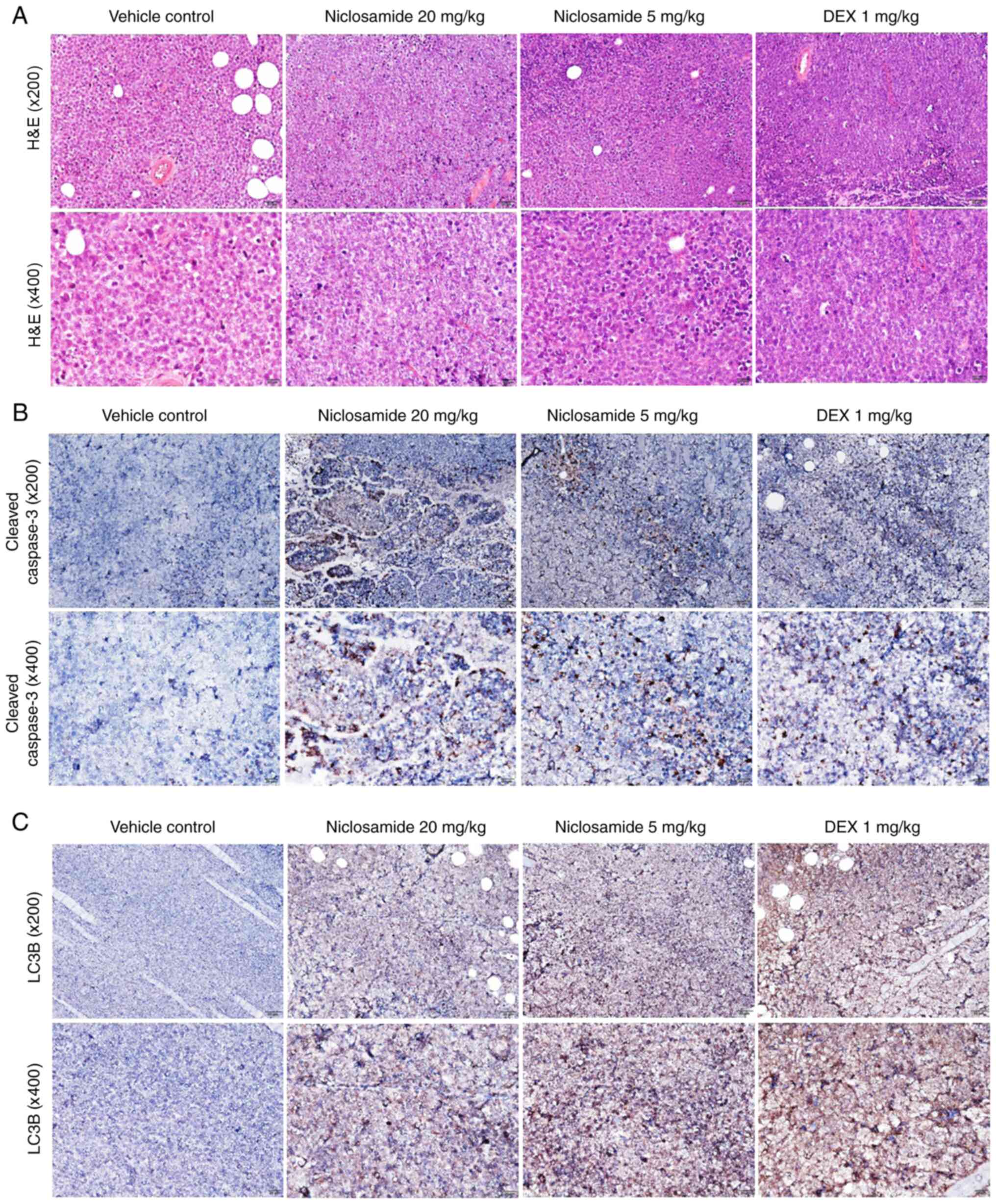
|
1
|
Arber DA, Orazi A, Hasserjian R, Thiele J,
Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M and Vardiman JW:
The 2016 revision to the World Health Organization classification
of myeloid neoplasms and acute leukemia. Blood. 127:2391–2405.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Terwilliger T and Abdul-Hay M: Acute
lymphoblastic leukemia: A comprehensive review and 2017 update.
Blood Cancer J. 7:e5772017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Karrman K and Johansson B: Pediatric
T-cell acute lymphoblastic leukemia. Genes Chromosomes Cancer.
56:89–116. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Corella Aznar EG, Ayerza Casas A, Carboné
Bañeres A, Calvo Escribano MÁC, Labarta Aizpún JI and Samper
Villagrasa P: Quality of life and chronic health conditions in
childhood acute leukaemia survivors. Med Clin (Barc). 152:167–173.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Singh SK, Banerjee S, Acosta EP, Lillard
JW and Singh R: Resveratrol induces cell cycle arrest and apoptosis
with docetaxel in prostate cancer cells via a p53/p21WAF1/CIP1 and
p27KIP1 pathway. Oncotarget. 8:17216–17228. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cai Y, Xia Q, Su Q, Luo R, Sun Y, Shi Y
and Jiang W: mTOR inhibitor RAD001 (everolimus) induces apoptotic,
not autophagic cell death, in human nasopharyngeal carcinoma cells.
Int J Mol Med. 31:904–912. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ciombor KK and Bekaii-Saab T: Selumetinib
for the treatment of cancer. Expert Opin Investig Drugs.
24:111–123. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Huang FL, Liao EC, Li CL, Yen CY and Yu
SJ: Pathogenesis of pediatric B-cell acute lymphoblastic leukemia:
Molecular pathways and disease treatments (Review). Oncol Lett.
20:448–454. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Al-Hadiya BM: Niclosamide: Comprehensive
profile. Profiles Drug Subst Excip Relat Methodol. 32:67–96. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Merschjohann K and Steverding D: In vitro
trypanocidal activity of the anti-helminthic drug niclosamide. Exp
Parasitol. 118:637–640. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hogarth LA and Hall AG: Increased BAX
expression is associated with an increased risk of relapse in
childhood acute lymphocytic leukemia. Blood. 93:2671–2678. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen W, Mook RA Jr, Premont RT and Wang J:
Niclosamide: Beyond an antihelminthic drug. Cell Signal. 41:89–96.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhu Y, Zuo W, Chen L, Bian S, Jing J, Gan
C, Wu X, Liu H, Su X, Hu W, et al: Repurposing of the
anti-helminthic drug niclosamide to treat melanoma and pulmonary
metastasis via the STAT3 signaling pathway. Biochem Pharmacol.
169:1136102019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liao Z, Nan G, Yan Z, Zeng L, Deng Y, Ye
J, Zhang Z, Qiao M, Li R, Denduluri S, et al: The anthelmintic drug
niclosamide inhibits the proliferative activity of human
osteosarcoma cells by targeting multiple signal pathways. Curr
Cancer Drug Targets. 15:726–738. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mathew R, Kongara S, Beaudoin B, Karp CM,
Bray K, Degenhardt K, Chen G, Jin S and White E: Autophagy
suppresses tumor progression by limiting chromosomal instability.
Genes Dev. 21:1367–1381. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mathew R, Karp CM, Beaudoin B, Vuong N,
Chen G, Chen HY, Bray K, Reddy A, Bhanot G, Gelinas C, et al:
Autophagy suppresses tumorigenesis through elimination of p62.
Cell. 137:1062–1075. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Teuffel O, Kuster SP, Hunger SP, Conter V,
Hitzler J, Ethier MC, Shah PS, Beyene J and Sung L: Dexamethasone
versus prednisone for induction therapy in childhood acute
lymphoblastic leukemia: A systematic review and meta-analysis.
Leukemia. 25:1232–1238. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bhadri VA, Trahair TN and Lock RB:
Glucocorticoid resistance in paediatric acute lymphoblastic
leukaemia. J Paediatr Child Health. 48:634–640. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang H, Dong R, Fan WW, Zheng XC, Li AM
and Wang WD: Timosaponin AIII induces autophagy of Tcell acute
lymphoblastic leukemia Jurkat cells via inhibition of the
PI3K/Akt/mTOR pathway. Oncol Rep. 41:2937–2944. 2019.PubMed/NCBI
|
|
20
|
Wang J, Ren XR, Piao H, Zhao S, Osada T,
Premont RT, Mook RA Jr, Morse MA, Lyerly HK and Chen W:
Niclosamide-induced Wnt signaling inhibition in colorectal cancer
is mediated by autophagy. Biochem J. 476:535–546. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jin B, Wang C, Shen Y and Pan J:
Anthelmintic niclosamide suppresses transcription of BCR-ABL fusion
oncogene via disabling Sp1 and induces apoptosis in
imatinib-resistant CML cells harboring T315I mutant. Cell Death
Dis. 9:682018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lu L, Dong J, Wang L, Xia Q, Zhang D, Kim
H, Yin T, Fan S and Shen Q: Activation of STAT3 and Bcl-2 and
reduction of reactive oxygen species (ROS) promote radioresistance
in breast cancer and overcome of radioresistance with niclosamide.
Oncogene. 37:5292–5304. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Satoh K, Zhang L, Zhang Y, Chelluri R,
Boufraqech M, Nilubol N, Patel D, Shen M and Kebebew E:
Identification of niclosamide as a novel anticancer agent for
adrenocortical carcinoma. Clin Cancer Res. 22:3458–3466. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Krause M and Durner J: Harpin inactivates
mitochondria in Arabidopsis suspension cells. Mol Plant Microbe
Interact. 17:131–139. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu WJ, Ye L, Huang WF, Guo LJ, Xu ZG, Wu
HL, Yang C and Liu HF: p62 links the autophagy pathway and the
ubiqutin-proteasome system upon ubiquitinated protein degradation.
Cell Mol Biol Lett. 21:292016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nguyen TD, Shaid S, Vakhrusheva O,
Koschade SE, Klann K, Thölken M, Baker F, Zhang J, Oellerich T,
Sürün D, et al: Loss of the selective autophagy receptor p62
impairs murine myeloid leukemia progression and mitophagy. Blood.
133:168–179. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gong Y, Wu J, Yang R, Zhang L and Ma Z:
Rapamycin-induced autophagy plays a pro-survival role by enhancing
up-regulation of intracellular ferritin expression in acute
lymphoblastic leukemia. Exp Oncol. 42:11–15. 2020.PubMed/NCBI
|
|
28
|
Rothe K, Lin H, Lin KB, Leung A, Wang HM,
Malekesmaeili M, Brinkman RR, Forrest DL, Gorski SM and Jiang X:
The core autophagy protein ATG4B is a potential biomarker and
therapeutic target in CML stem/progenitor cells. Blood.
123:3622–3634. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Baquero P, Dawson A, Mukhopadhyay A, Kuntz
EM, Mitchell R, Olivares O, Ianniciello A, Scott MT, Dunn K,
Nicastri MC, et al: Targeting quiescent leukemic stem cells using
second generation autophagy inhibitors. Leukemia. 33:981–994. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Park SJ, Shin JH, Kang H, Hwang JJ and Cho
DH: Niclosamide induces mitochondria fragmentation and promotes
both apoptotic and autophagic cell death. BMB Rep. 44:517–522.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Torres-López L, Maycotte P, Liñán-Rico A,
Liñán-Rico L, Donis-Maturano L, Delgado-Enciso I, Meza-Robles C,
Vásquez-Jiménez C, Hernández-Cruz A and Dobrovinskaya O: Tamoxifen
induces toxicity, causes autophagy, and partially reverses
dexamethasone resistance in Jurkat T cells. J Leukoc Biol.
105:983–998. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chong SJF, Marchi S, Petroni G, Kroemer G,
Galluzzi L and Pervaiz S: Noncanonical cell fate regulation by
Bcl-2 proteins. Trends Cell Biol. 30:537–555. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pattingre S, Tassa A, Qu X, Garuti R,
Liang XH, Mizushima N, Packer M, Schneider MD and Levine B: Bcl-2
antiapoptotic proteins inhibit Beclin 1-dependent autophagy. Cell.
122:927–939. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Saeki K, Yuo A, Okuma E, Yazaki Y, Susin
SA, Kroemer G and Takaku F: Bcl-2 down-regulation causes autophagy
in a caspase-independent manner in human leukemic HL60 cells. Cell
Death Differ. 7:1263–1269. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ge C, Huang H, Huang F, Yang T, Zhang T,
Wu H, Zhou H, Chen Q, Shi Y, Sun Y, et al: Neurokinin-1 receptor is
an effective target for treating leukemia by inducing oxidative
stress through mitochondrial calcium overload. Proc Natl Acad Sci
USA. 116:19635–19645. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yang L, Li M, Wang F, Zhen C, Luo M, Fang
X, Zhang H, Zhang J, Li Q and Fu L: Ceritinib enhances the efficacy
of substrate chemotherapeutic agent in human ABCB1-Overexpressing
leukemia cells in vitro, in vivo and ex-vivo. Cell Physiol Biochem.
46:2487–2499. 2018. View Article : Google Scholar : PubMed/NCBI
|