Introduction
Glaucoma is the second most common eye disease
leading to blindness in the world with high intraocular pressure,
irreversible optic atrophy and visual field defects as the main
features. Clinical treatment mainly aims to reduce intraocular
pressure and maintain the normal range of intraocular pressure,
main treatments include laser, drugs, surgery and other methods
(1). Basic study confirmed that
(2) retinal ganglion cell (RGC)
apoptosis and optic nerve axon degeneration caused by ischemia,
oxidative stress and inflammatory response are important causes of
the occurrence and development of glaucoma. Malondialdehyde (MDA),
nitric oxide (NO) and superoxide dismutase (SOD) are the most
commonly used biochemical markers for the evaluation of glaucoma
animal model and glaucoma patients with oxidative stress disorder.
The use of optic nerve protection drugs such as nerve growth factor
(NGF) can prevent or delay the damage of RGCs, so as to improve
glaucoma symptoms and prognosis (3).
Drug application paths mainly include oral medication, and
intravenous and intramuscular injection, the drug reaches the eyes
through circulation system, which in turn lead to the relative low
effective concentrations of drugs in area around retina and optic
nerve, leading to the low efficacy, at the same time, the incidence
of toxic side effects will be increased (4). Therefore, the development of a more
efficient and safe way of drug administration will definitely
improve the clinical treatment of glaucoma. Drug release mediated
by ultrasound targeted microbubble destruction can achieve accurate
positioning and targeted release, which in turn reduces the dose,
improves clinical results, and reduces systemic adverse reactions
(5). Ginsenoside Rg1 has proven
anti-fatigue, anti-aging, anticancer, lipid-lowering, memory
enhancement, immunity improvement and other pharmacological effects
(6). Based on this, we investigated
the mechanism of treatment of glaucomatous optic nerve damage using
ginsenoside Rg1 mediated by ultrasound targeted microbubble
destruction.
Materials and methods
Experimental materials
Thirty healthy New Zealand white rabbits (2–2.5 kg)
without restriction on sex were purchased from Sangon (Shanghai,
China). Rabbits were raised under normal conditions for 1 week to
be familiar with the environment before the experiment. The study
was approved by the Ethics Committee of Cangzhou Central
Hospital.
Establishment of glaucomatous optic nerve damage
model: Intramuscular injection of sumianxin II (0.2 ml/kg) for
anesthesia, dicaine hydrochloride eye drops was used for topical
anesthesia; aqueous humor (0.2 ml) was extracted from anterior
chamber along one side of corneoscleral in the direction of 9
o'clock, 0.2 ml of compound carbomer solution (0.3%) was injected
into the anterior chamber along the other side of corneoscleral;
ofloxacin eye drops was used. Intraocular pressure measured by
accumen hand-held tonometer >22 mm Hg for 4 weeks indicate the
successfully established model; if intraocular pressure <22
mmHg, drug administration can be repeated 7 days later.
Main reagent: Ginsenoside Rg1 powder (purity ≥98%)
was purchased from Beyotime Biotechnology (Jiangsu, China), NGF
injection was purchased from Beijing Zhongshan Golden Bridge
Biotechnology Co., Ltd. (Beijing, China). Sulfur hexafluoride
microbubbles for injection was purchased from Bracco Inc. (Milan,
Italy), medical ultrasonic coupling agent was from Sigma (St.
Louis, MO, USA), MDA, SOD and NO, enzyme-linked immunosorbent assay
(ELISA) kits were purchased from R&D (Minneapolis, MN,
USA).
Main instruments: ophthalmic microsurgical
instruments were purchased from Beijing Liuyi Instrument Factory
(Beijing, China), pathology slicing machine was from Leica
(Wetzlar, Germany), optical microscope was purchased from Olympus
(Tokyo, Japan), Accupen handheld tonometer was from Accutome Inc.
(Malvern, PA, USA).
Research methods
Rabbits were divided into 5 groups: control group,
model group, model group + intravitreal injection of nerve growth
factor NGF group (NGF group), model group + intravitreal injection
of ginsenoside Rg1 group (Rg1 group), model group + intravitreal
injection of ginsenoside Rg1 + ultrasound microbubble group
(ultrasound group), model group + ultrasound targeted microbubble
destruction (ultrasound group), 6 rabbits in each group. Methods of
intravitreal injection: compound tropicamide eye drops were used
for mydriasis, eyelid was opened with eye speculum, conjunctival
sac was rinsed with 3% iodophor. One milliliter syringe was
injected vertically into vitreous cavity through the position 3 mm
away from corneoscleral limbus to extract 0.1 ml vitreous humor,
then 0.1 ml ginsenoside Rg1 solution (2.5 mg/kg) or NGF solution
(18 µg/0.1 ml) was injected, once per 3 days for 4 weeks. Methods
of ultrasound targeted microbubbles destruction: After the
injection of ginsenoside Rg1 solution or NGF solution, 0.1 ml
microbubble suspension (45 µg/0.1 ml) and coupling agent coating
was performed after the eyes were closed. Ultrasonic probe was
places above the eyes to irradiate the eyes for 60 sec, once per 3
days for 4 weeks.
Observation indicators
Intraocular pressures were compared at 1, 2 and 4
weeks after model establishment. Rabbits were sacrificed 4 weeks
after model establishment to collect retinal tissue for H&E
staining. Histological changes were observed and the retinal
thickness was measured under a light microscope. Contents of MDA,
SOD and NO were measured by ELISA. All operations were performed in
strict accordance with the instructions of kits.
Statistical analysis
Statistical analysis was performed using SPSS 20.0
software (SPSS Inc., Chicago, IL, USA). Measurement data were
expressed as mean ± standard deviation. Single-factor ANOVA
analysis was used for the comparisons among multiple groups, and
LSD-t method was used for the comparisons between two groups,
intraocular pressure within a group was compared with the analysis
of variance of repeated measure data. P<0.05 was considered to
be statistically significant.
Results
Comparison of intraocular
pressure
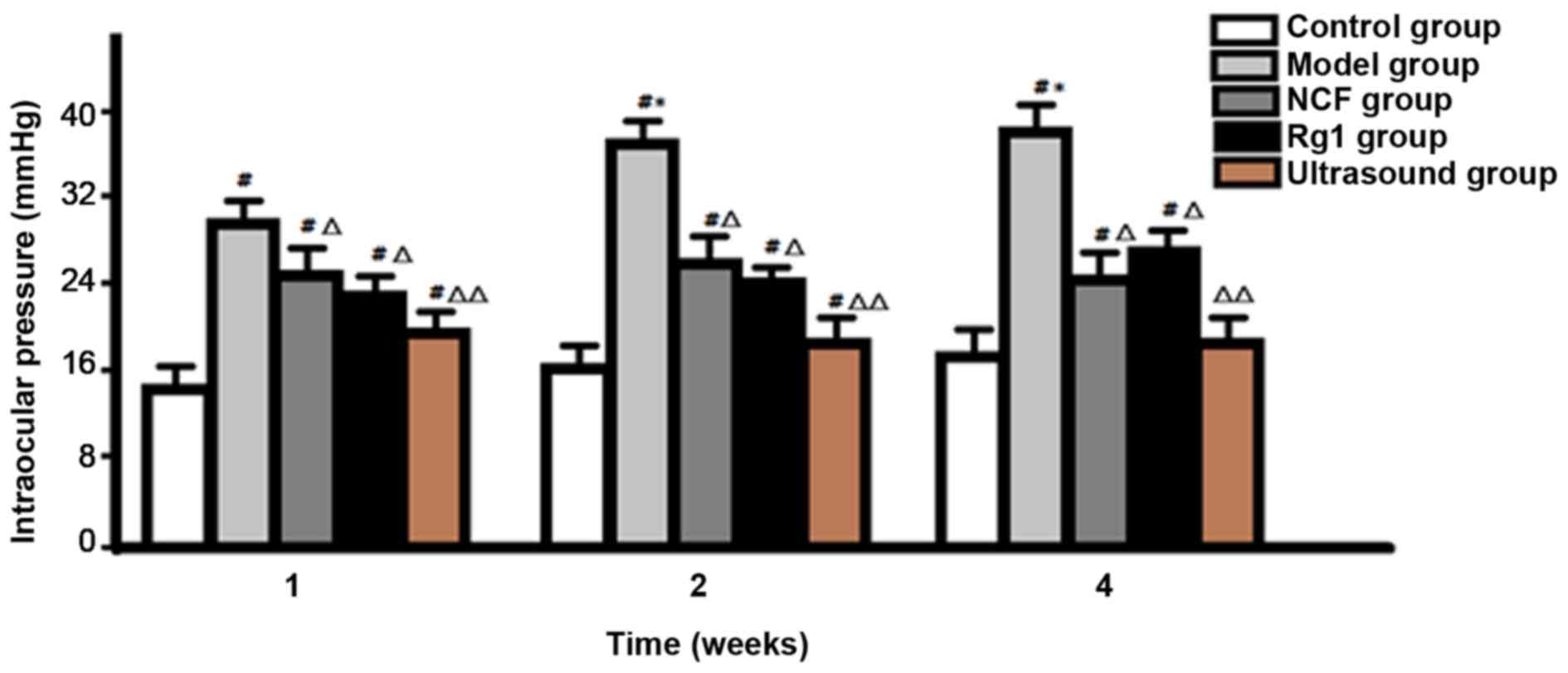
Intraocular pressure was significantly higher in the
model group than in the control group at 1 week after model
establishment, and the peak was reached at 2 weeks after model
establishment. Intraocular pressure of NGF group and Rg1 group was
significantly lower than that of model group (P<0.05), but no
significant difference was found between NGF group and Rg1 group
(P>0.05). Intraocular pressure was significantly lower in
ultrasound group than in NGF group and Rg1 group at all time-points
(P<0.05), and no significant difference was found between
ultrasound group and control group at 4 weeks after model
establishment (P>0.05) (Fig.
1).
Comparison of histomorphologic
structure and retinal thickness
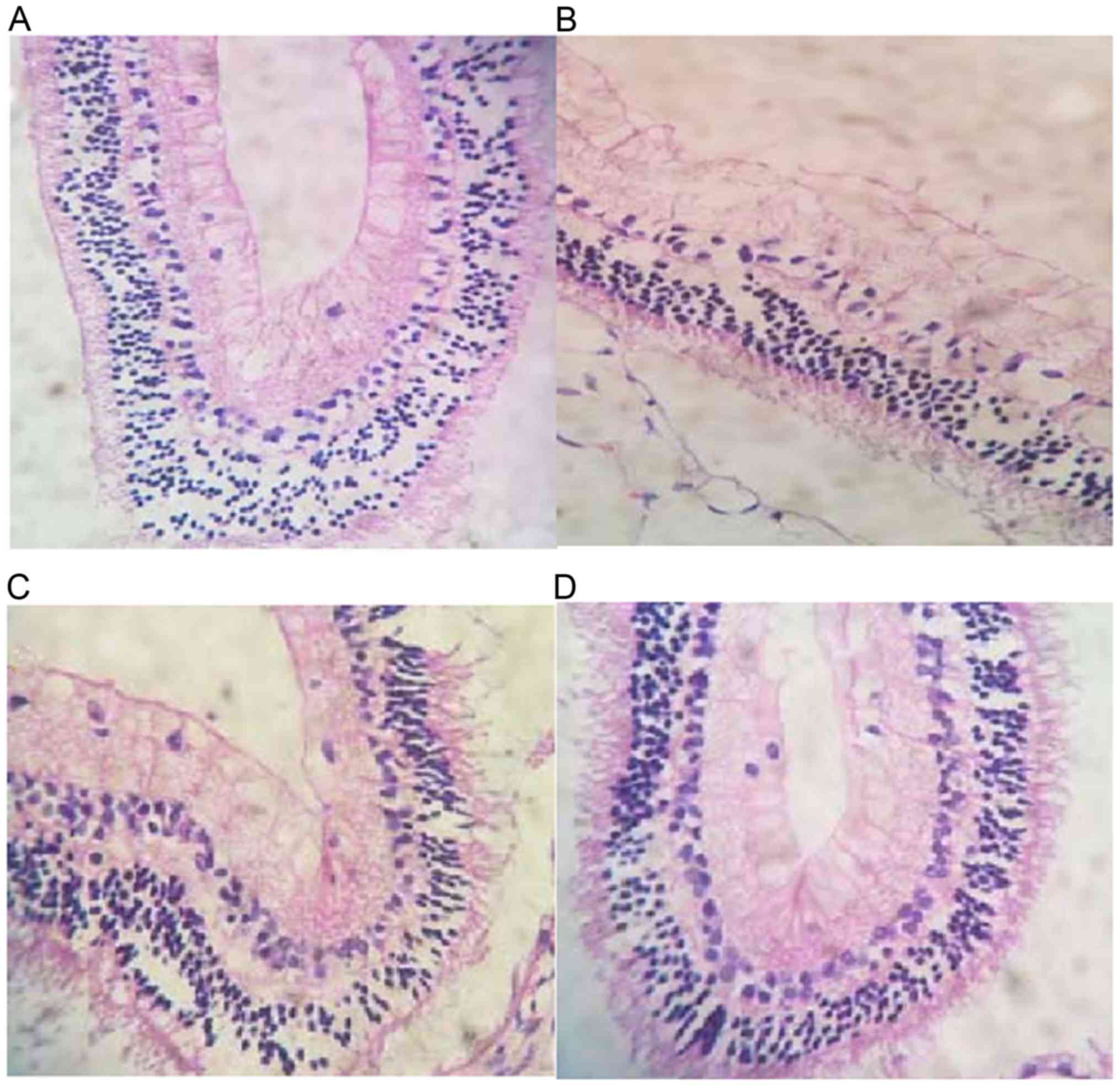
Retinal histomorphology: H&E staining showed
that the RGCs in control group were arranged in a single layer with
different sizes, deep nuclear staining, clear boundary and neat
arrangement, relative more cells with small nucleus and deep
staining were found in outer nuclear layer, cells with neat
arrangement, big nucleus and deep staining were found in the inner
layer, inner and outer plexus layers showed obvious network
structure; in model group, cell vacuolization was common, inner
layer of retina was thinned, the nucleolus was dissolved, the
nuclei staining was light, color of cytoplasm was light, number of
cells in inner and outer nuclear layers was decreased, the gap was
widened, cells were arranged sparsely without clear boundaries,
inner plexus layers was significantly thinner or even disappeared;
number of RGCs in NGF group and Rg1 was reduced but higher than
model group, cell vacuolization and karyolysis were rare, cells in
inner and outer nuclear layers were arranged sparsely, and inner
and outer plexus layers were slightly thinned; there was no
decrease in RGCs was found in the ultrasound group, and most of the
nuclei were present, disorder in the arrangement of cells in inner
and outer nuclear layers was not obvious (Fig. 2).
As shown in Fig. 2A
in the control group, cells were arranged in a single layer with
different sizes, deep nuclear staining, clear boundary and neat
arrangement, relative more cells with small nucleus and deep
staining were found in outer nuclear layer, cells with neat
arrangement, big nucleus and deep staining were found in inner
layer, inner and outer plexus layers showed obvious network
structure. In Fig. 2B in the model
group, number of RGCs was significantly reduced, cell vacuolization
was common, inner layer of retina was thinned, karyolysis occurred,
the nuclei staining was light, color of cytoplasm was light, number
of cells in inner and outer nuclear layers was decreased, the gap
was widened, cells were arranged sparsely without clear boundaries,
inner plexus layers was significantly thinner or even disappeared.
In Fig. 2C in the NGF group and Rg1
group, number of RGCs was reduced but higher than model group, cell
vacuolization and karyolysis were rare, cells in inner and outer
nuclear layers were arranged sparsely, and inner and outer plexus
layers were slightly thinned. No decrease in RGCs was found, most
of the nuclei were present, disorder in the arrangement of cells in
inner and outer nuclear layers was not obvious (Fig. 2D).
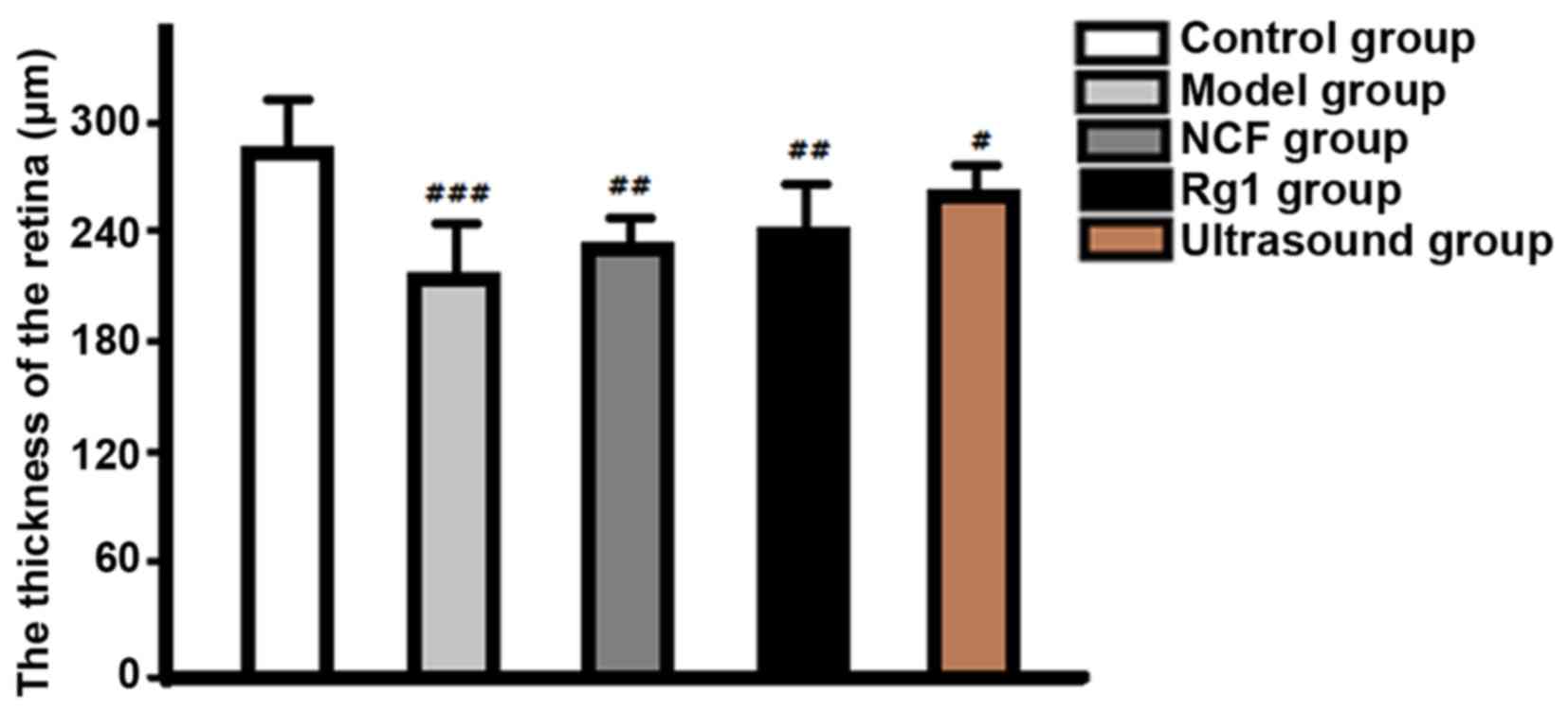
Retinal thickness: The retina in control group was
significantly thicker than that in ultrasound group (P<0.05),
followed by NGF group and Rg1 group, the model group was the
thinnest (Fig. 3).
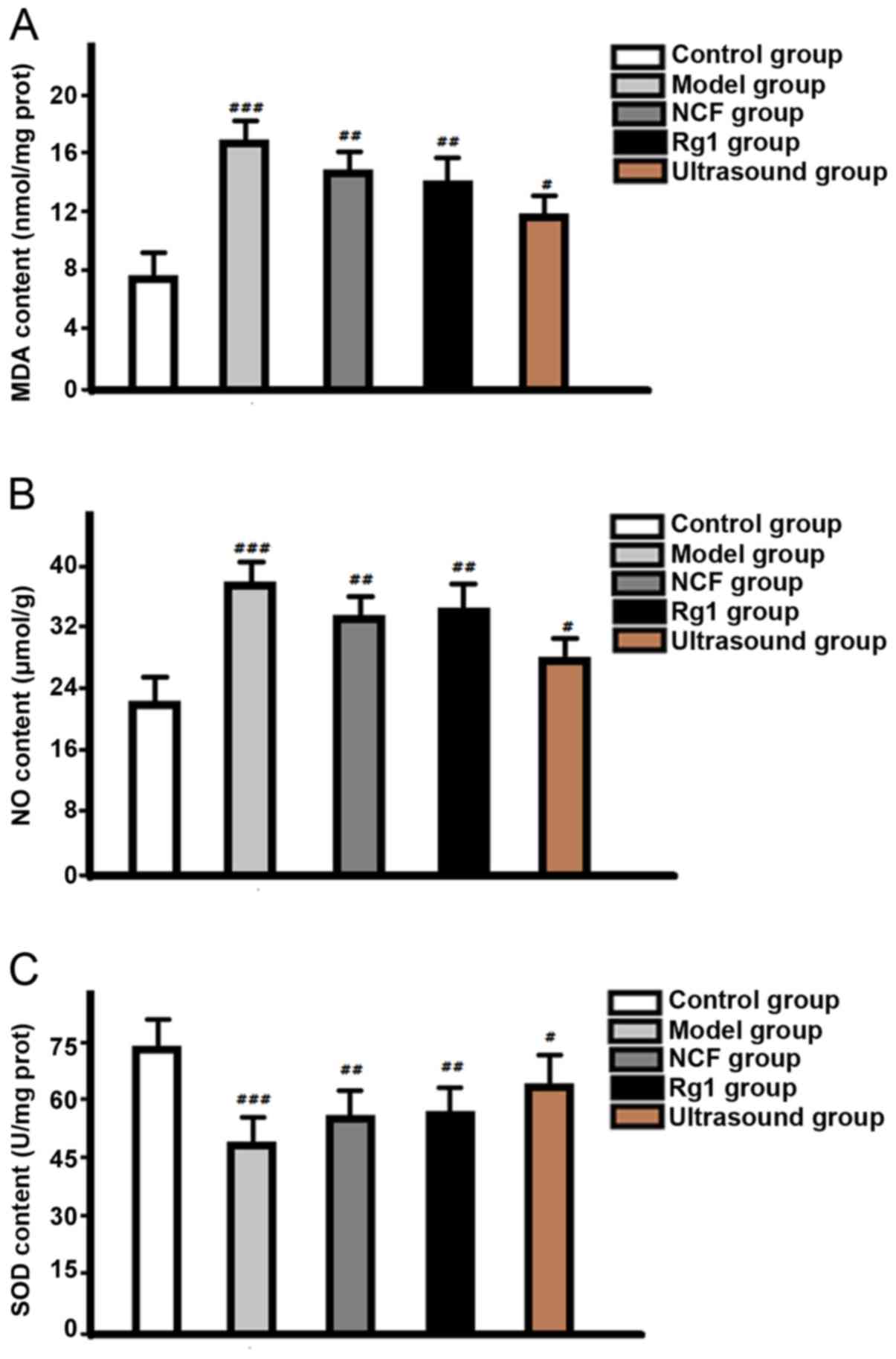
Comparison of MDA, SOD and NO contents
among groups
Levels of MDA and NO in retina of model group were
significantly higher than those in NGF group and Rg1 group, and
control group was the lowest. SOD content in control group was
higher than that in ultrasound group, followed by NGF group and Rg1
group, and the differences were all statistically significant
(P<0.05) (Fig. 4).
Discussion
In this study, the compound carbomer-induced rabbit
glaucoma model was used. Results showed that the average
intraocular pressure at 1 week after model establishment was 30
mmHg, and the peak (38 mmHg) was reached at 2 weeks after model
establishment; retinal histomorphological changes were significant
and the thickness was significantly decreased, indicating that the
intraocular pressure of the rabbit model was significantly
increased after carbomer treatment, the duration is long. This
method is easy for operation and control, so it is an ideal model
for the studies on glaucomatous optic nerve damage and
anti-glaucoma drug screening. As the key cytokine involved in optic
nerve development, NGF regulates the growth, differentiation and
apoptosis of RGCs (7). Different
concentrations of NGF added to neonatal rat RGC cell culture medium
can promote the survival of RGCs and optic nerve axon elongation,
so as to achieve a certain therapeutic effect (8). Recent studies have found that
ginsenoside Rg1 can play a neuroprotective by antagonizing dopamine
to alleviate the excitotoxicity of neuronal amino acids and reduce
neuronal amino acid excitotoxicity, and reduce the toxic effects of
phenylfenapyridine (9,10). Rg1 can upregulate the expression of
glial cell derived and brain-derived NGF and other genes (11), promote axonal regeneration, increase
the expression of Bcl-2 protein and reduce the expression of Bax
protein (12), improve mitochondrial
structure and dysfunction, inhibit optic neuronal apoptosis, and
block Ca2+ influx (13).
Results showed that, compared with model group, NGF and Rg1 can
significantly reduce intraocular pressure, improve retinal tissue
morphologic structure, increase the thickness of the retina, reduce
MDA and NO content, and increase SOD content.
Ultrasound microbubbles achieved great success in
the diagnosis and treatment of neoplastic diseases (14), ultrasonic microbubbles were formed by
two-part shell and gas inside. After ultrasonic irradiation,
drug-containing microbubbles will be destroyed to release drug and
cause reversible or irreversible pores to increase the permeability
of the cell membrane, which in turn achieve targeted drug
administration (14,15). In the field of ophthalmology,
ultrasound microbubbles are mainly used for gene transfection, such
as ultrasound microbubble technology to improve the transfection
rate DNA plasmid in transfecting retinoblastoma cells (15). Results of this study showed that
ultrasound group can further reduce the intraocular pressure,
improve retinal histomorphological changes and increase retinal
thickness compared with drug treatment alone (NGF group and Rg1
group). In addition, content of MDA and NO in retinal tissue of
ultrasound group was lower than that of NGF group and Rg1 group,
and the content of SOD was higher than that of NGF group and Rg1
group, and the differences were statistically significant,
indicating that the use of ultrasound microbubbles can achieve
targeted drug administration to increase the drug concentration in
eyes. So, ginsenoside Rg1 and NGF mediated by ultrasound targeted
microbubble destruction is effective in the treatment of
glaucomatous optic nerve damage, which may be related to the
changed contents of NO, MDA and SOD (16–18).
This study provided references for the studies on mechanism of the
role of oxidative stress injury in the occurrence and development
of glaucoma.
References
|
1
|
Quigley HA: Understanding Glaucomatous
Optic Neuropathy: The synergy between clinical observation and
investigation. Annu Rev Vis Sci. 2:235–254. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen X and Zhao Y: Diagnostic performance
of isolated-check visual evoked potential versus retinal ganglion
cell-inner plexiform layer analysis in early primary open-angle
glaucoma. BMC Ophthalmol. 17:772017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Oddone F, Roberti G, Micera A, Busanello
A, Bonini S, Quaranta L, Agnifili L and Manni G: Exploring serum
levels of brain derived neurotrophic factor and nerve growth factor
across glaucoma stages. PLoS One. 12:e01685652017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mesentier-Louro LA, De Nicolò S, Rosso P,
De Vitis LA, Castoldi V, Leocani L, Mendez-Otero R, Santiago MF,
Tirassa P, Rama P, et al: Time-dependent nerve growth factor
signaling changes in the rat retina during optic nerve
crush-induced degeneration of retinal ganglion cells. Int J Mol
Sci. 18:E982017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hussein F, Antonescu C and Karshafian R:
Ultrasound and microbubble induced release from intracellular
compartments. BMC Biotechnol. 17:452017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Luo H, Huang WX, Yang C, Zhao JQ, Liu S,
Xu YS and Liu CW: Therapeutic efficacy and mechanism of action of
ginsenoside Rg1 in treating acute hepatic failure in mice. Zhonghua
Gan Zang Bing Za Zhi. 25:217–222. 2017.(In Chinese). PubMed/NCBI
|
|
7
|
Garcia TB, Hollborn M and Bringmann A:
Expression and signaling of NGF in the healthy and injured retina.
Cytokine Growth Factor Rev. 34:43–57. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kimura A, Namekata K, Guo X, Harada C and
Harada T: Neuroprotection, growth factors and BDNF-TrkB signalling
in retinal degeneration. Int J Mol Sci. 17:E15842016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sun ZG, Chen LP, Wang FW, Xu CY and Geng
M: Protective effects of ginsenoside Rg1 against hydrogen
peroxide-induced injury in human neuroblastoma cells. Neural Regen
Res. 11:1159–1164. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang L, Liu LF, Liu J, Dou L, Wang GY,
Liu XQ and Yuan QL: Ginsenoside Rg1 protects against
neurodegeneration by inducing neurite outgrowth in cultured
hippocampal neurons. Neural Regen Res. 11:319–325. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Huo DS, Zhang M, Cai ZP, Dong CX, Wang H
and Yang ZJ: The role of nerve growth factor in ginsenoside
Rg1-induced regeneration of injured rat sciatic nerve. J Toxicol
Environ Health A. 78:1328–1337. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang B, He L, Cui B and Lv H: Protection
of ginsenoside Rg1 on central nerve cell damage and the influence
on neuron apoptosis. Pak J Pharm Sci. 27 Suppl 6:2035–2040.
2014.PubMed/NCBI
|
|
13
|
Li YB, Wang Y, Tang JP, Chen D and Wang
SL: Neuroprotective effects of ginsenoside Rg1-induced neural stem
cell transplantation on hypoxic-ischemic encephalopathy. Neural
Regen Res. 10:753–759. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guo X, Guo S, Pan L, Ruan L, Gu Y and Lai
J: Anti-microRNA-21/221 and microRNA-199a transfected by ultrasound
microbubbles induces the apoptosis of human hepatoma HepG2 cells.
Oncol Lett. 13:3669–3675. 2017.PubMed/NCBI
|
|
15
|
Luo J, Zhou X, Diao L and Wang Z:
Experimental research on wild-type p53 plasmid transfected into
retinoblastoma cells and tissues using an ultrasound microbubble
intensifier. J Int Med Res. 38:1005–1015. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rokicki W, Zalejska-Fiolka J,
Pojda-Wilczek D, Hampel A, Majewski W, Ogultekin S and
Mrukwa-Kominek E: Differences in serum oxidative status between
glaucomatous and nonglaucomatous cataract patients. BMC Ophthalmol.
17:132017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Panchal SS, Patidar RK, Jha AB, Allam AA,
Ajarem J and Butani SB: Anti-inflammatory and antioxidative stress
effects of oryzanol in glaucomatous rabbits. J Ophthalmol.
2017:14687162017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mumcu UY, Kocer I, Ates O and Alp HH:
Decreased paraoxonase1 activity and increased malondialdehyde and
oxidative DNA damage levels in primary open angle glaucoma. Int J
Ophthalmol. 9:1518–1520. 2016.PubMed/NCBI
|