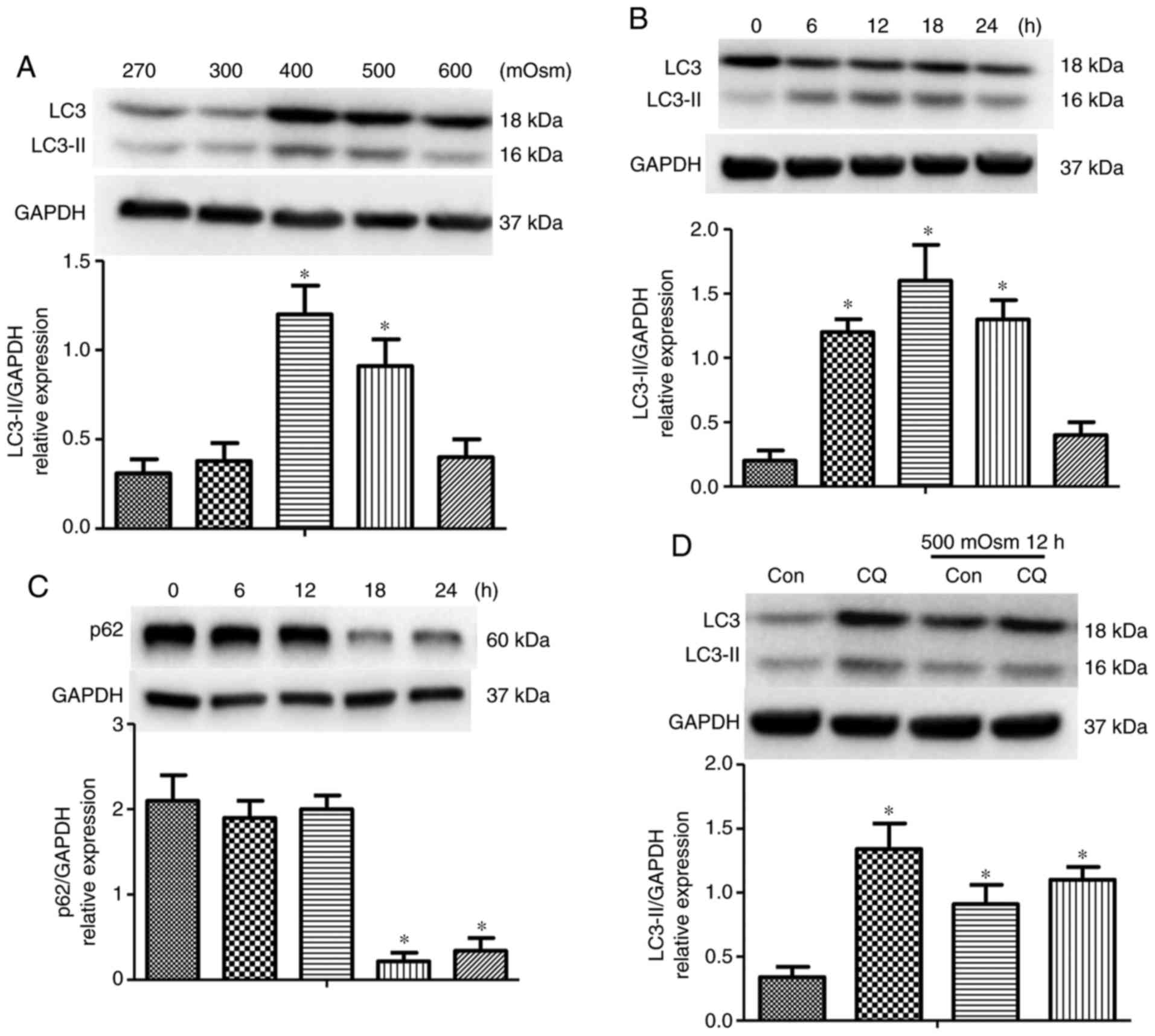
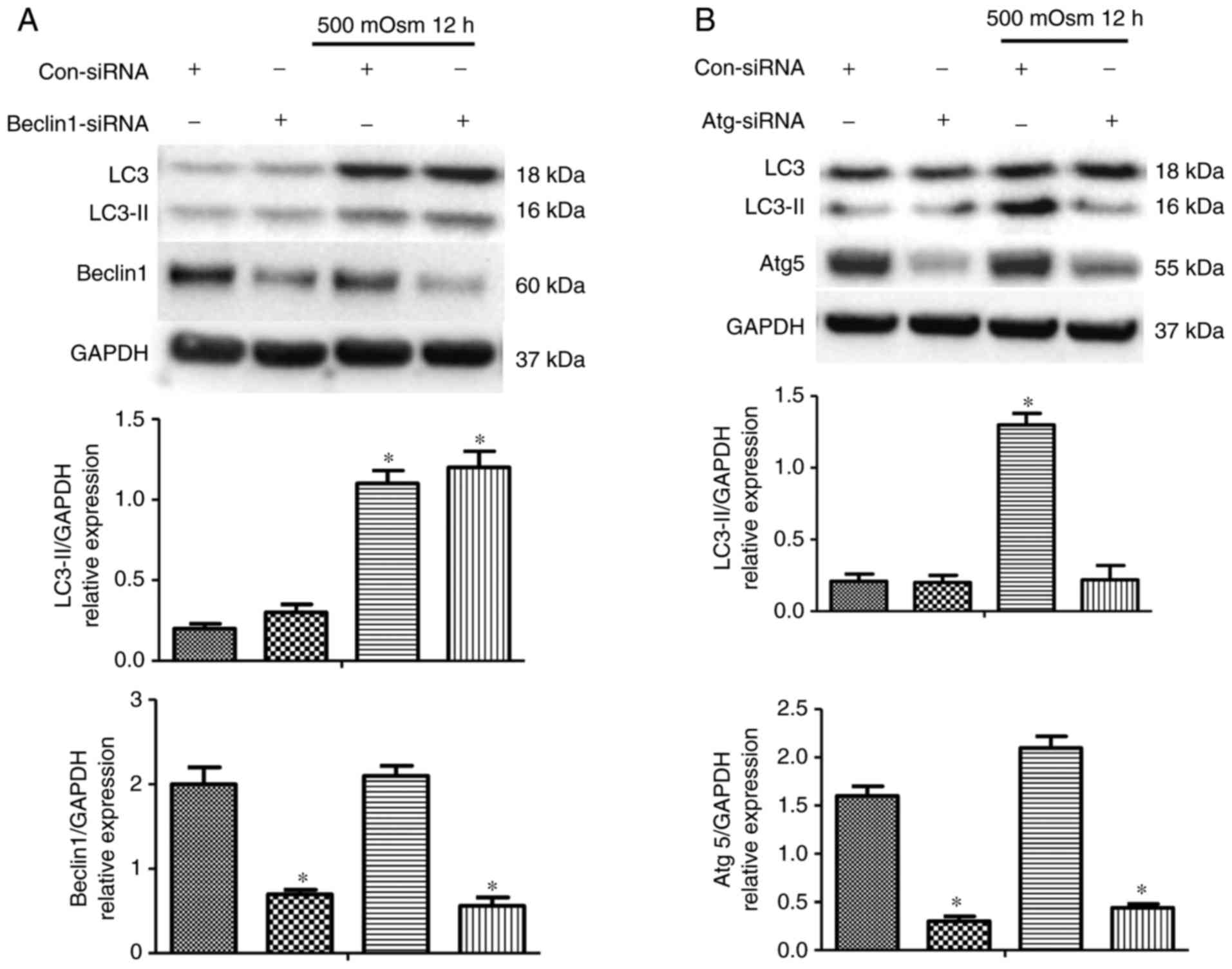
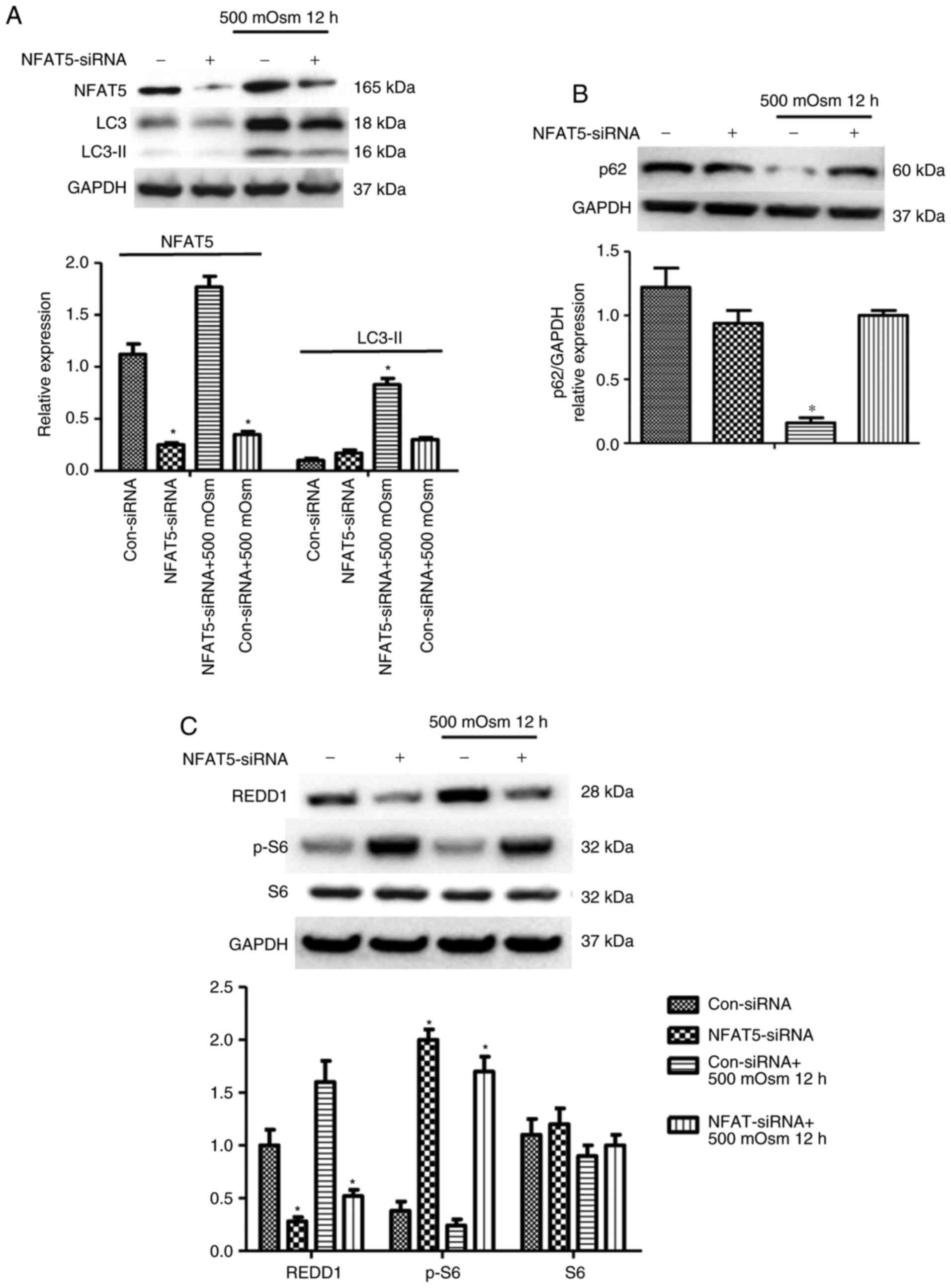
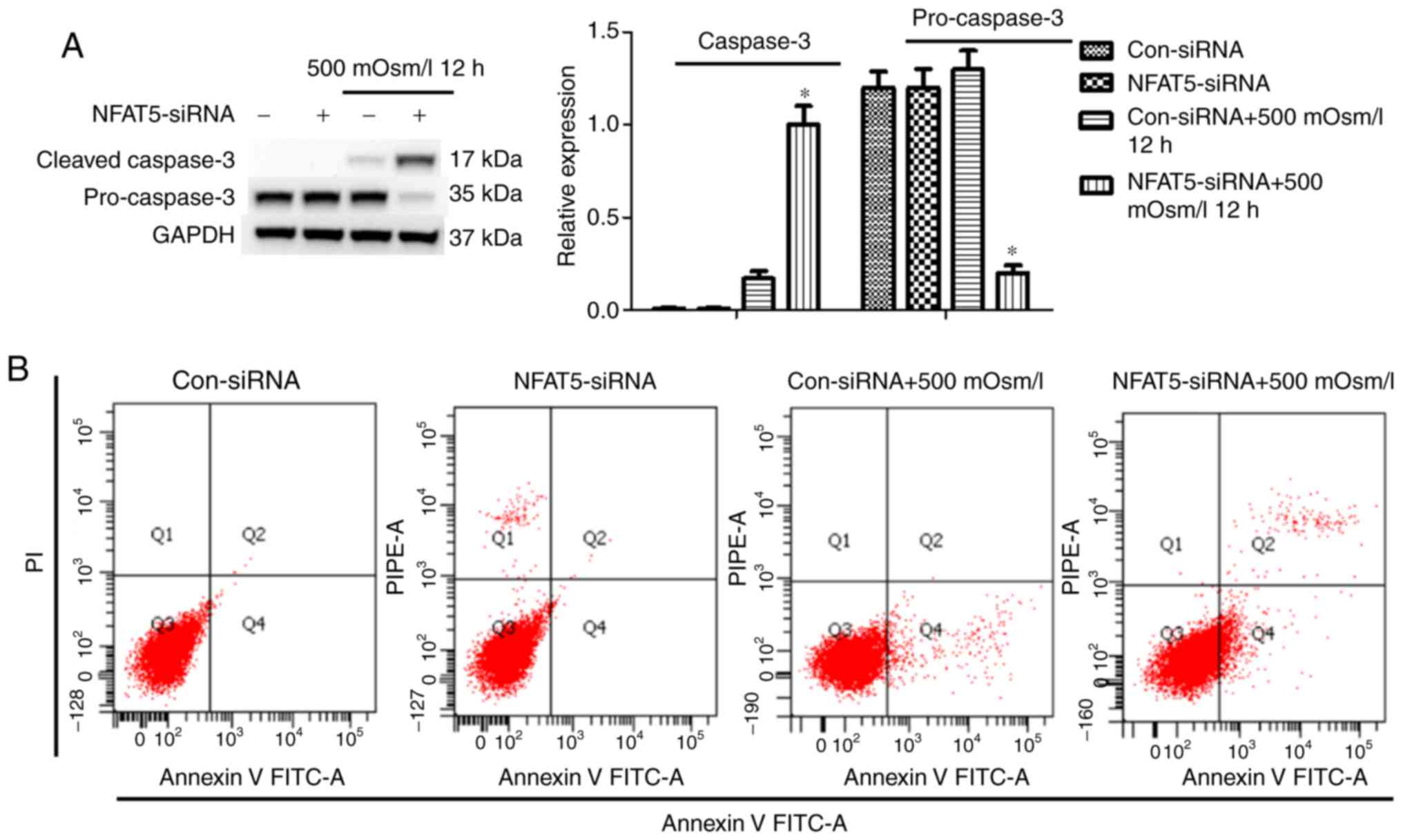
|
1
|
Wright AR and Rees SA: Cardiac cell
volume: Crystal clear or murky waters? A comparison with other cell
types. Pharmacol Ther. 80:89–121. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Galvez A, Morales MP, Eltit JM, Ocaranza
P, Carrasco L, Campos X, Sapag-Hagar M, Díaz-Araya G and Lavandero
S: A rapid and strong apoptotic process is triggered by
hyperosmotic stress in cultured rat cardiac myocytes. Cell Tissue
Res. 304:279–285. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kaur J and Debnath J: Autophagy at the
crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol.
16:461–472. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tanaka K and Matsuda N: Proteostasis and
neurodegeneration: The roles of proteasomal degradation and
autophagy. Biochim Biophys Acta. 1843:197–204. 2014. View Article : Google Scholar
|
|
5
|
Mazure NM and Pouysségur J:
Hypoxia-induced autophagy: Cell death or cell survival? Curr Opin
Cell Biol. 22:177–180. 2010. View Article : Google Scholar
|
|
6
|
Galluzzi L, Pietrocola F, Levine B and
Kroemer G: Metabolic control of autophagy. Cell. 159:1263–1276.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Russell RC, Yuan HX and Guan KL: Autophagy
regulation by nutrient signaling. Cell Res. 24:42–57. 2014.
View Article : Google Scholar :
|
|
8
|
Levine B and Deretic V: Unveiling the
roles of autophagy in innate and adaptive immunity. Nat Rev
Immunol. 7:767–777. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Klionsky DJ, Abdelmohsen K, Abe A, Abedin
MJ, Abeliovich H, Acevedo Arozena A, Adachi H, Adams CM, Adams PD,
Adeli K, et al: Guidelines for the use and interpretation of assays
for monitoring autophagy (3rd edition). Autophagy. 12:1–222. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Klionsky DJ, Abdalla FC, Abeliovich H,
Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M,
Agostinis P, Aguirre-Ghiso JA, et al: Guidelines for the use and
interpretation of assays for monitoring autophagy. Autophagy.
8:445–544. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen C, Deng M, Sun Q, Loughran P, Billiar
TR and Scott MJ: Lipopolysaccharide stimulates p62-dependent
autophagy-like aggregate clearance in hepatocytes. Biomed Res Int.
2014:2673502014.PubMed/NCBI
|
|
12
|
Nishida Y, Arakawa S, Fujitani K,
Yamaguchi H, Mizuta T, Kanaseki T, Komatsu M, Otsu K, Tsujimoto Y
and Shimizu S: Discovery of Atg5/Atg7-independent alternative
macroau-tophagy. Nature. 461:6542009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Johnson ZI, Shapiro IM and Risbud MV:
Extracellular osmo-larity regulates matrix homeostasis in the
intervertebral disc and articular cartilage: Evolving role of
TonEBP. Matrix Biol. 40:10–16. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Woo SK, Lee S and Kwon MH: TonEBP
transcriptional activator in the cellular response to increased
osmolality. Pflugers Arch. 444:579–585. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Woo SK and Kwon HM: Adaptation of kidney
medulla to hypertonicity: Role of the transcription factor TonEBP.
Int Rev Cytol. 215:189–202. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ortells MC, Morancho B, Drews-Elger K,
Viollet B, Laderoute KR, López-Rodríguez C and Aramburu J:
Transcriptional regulation of gene expression during osmotic stress
responses by the mammalian target of rapamycin. Nucleic Acids Res.
40:4368–4384. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou Y, Wang Q, Weiss HL and Evers BM:
Nuclear factor of activated T-cells 5 increases intestinal goblet
cell differentiation through an mTOR/Notch signaling pathway. Mol
Biol Cell. 25:2882–2890. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
National Research Council: Guide for the
care and use of laboratory animals. 8th edition. National Academies
Press; Washington, DC: 2011
|
|
19
|
Pursiheimo JP, Rantanen K, Heikkinen PT,
Johansen T and Jaakkola PM: Hypoxia-activated autophagy accelerates
degradation of SQSTM1/p62. Oncogene. 28:334–344. 2009. View Article : Google Scholar
|
|
20
|
Beth L and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008. View Article : Google Scholar
|
|
21
|
Burg MB, Ferraris JD and Dmitrieva NI:
Cellular response to hyperosmotic stresses. Physiol Rev.
87:1441–1474. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hoover HE, Thuerauf DJ, Martindale JJ and
Glembotski CC: Alpha B-crystallin gene induction and
phosphorylation by MKK6-activated p38 A potential role for alpha
B-crystallin as a target of the p38 branch of the cardiac stress
response. J Biol Chem. 275:825–833. 2000. View Article : Google Scholar
|
|
23
|
Takatani T, Takahashi K, Uozumi Y, Matsuda
T, Ito T, Schaffer SW, Fujio Y and Azuma J: Taurine prevents the
ischemia-induced apoptosis in cultured neonatal rat cardio-myocytes
through Akt/caspase-9 pathway. Biochem Biophys Res Commun.
316:484–489. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Takatani T, Takahashi K, Uozumi Y, Shikata
E, Yamamoto Y, Ito T, Matsuda T, Schaffer SW, Fujio Y and Azuma J:
Taurine inhibits apoptosis by preventing formation of the
Apaf-1/caspase-9 apoptosome. Am J Physiol Cell Physiol.
287:C949–C953. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Nunes P, Ernandez T, Roth I, Qiao X,
Strebel D, Bouley R, Charollais A, Ramadori P, Foti M, Meda P, et
al: Hypertonic stress promotes autophagy and microtubule-dependent
autopha-gosomal clusters. Autophagy. 9:550–567. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Biswas K, Khongsngi JL, Häussinger D and
Saha N: Influence of cell volume changes on autophagic proteolysis
in the perfused liver of air-breathing walking catfish (Clarias
batrachus). J Exp Zool A Ecol Genet Physiol. 311:115–124. 2009.
View Article : Google Scholar
|
|
27
|
Liu Y, Xiong Y and Bassham DC: Autophagy
is required for tolerance of drought and salt stress in plants.
Autophagy. 5:954–963. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Marchenko ND, Zaika A and Moll UM: Death
signal-induced localization of p53 protein to mitochondria a
potential role in apoptotic signaling. J Biol Chem.
275:16202–16212. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Mihara M, Erster S, Zaika A, Petrenko O,
Chittenden T, Pancoska P and Moll UM: p53 has a direct apoptogenic
role at the mitochondria. Mol Cell. 11:577–590. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chipuk JE, Kuwana T, Bouchier-Hayes L,
Droin NM, Newmeyer DD, Schuler M and Green DR: Direct activation of
Bax by p53 mediates mitochondrial membrane permeabilization and
apoptosis. Science. 303:1010–1014. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Leu JJ, Dumont P, Hafey M, Murphy ME and
George DL: Mitochondrial p53 activates Bak and causes disruption of
a Bak-Mcl1 complex. Nat Cell Biol. 6:443–450. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou F, Yang Y and Xing D: Bcl-2 and
Bcl-xL play important roles in the crosstalk between autophagy and
apoptosis. FEBS J. 278:403–413. 2011. View Article : Google Scholar
|
|
33
|
Strasser A: The role of BH3-only proteins
in the immune system. Nat Rev Immunol. 5:189–200. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Olson JM and Hallahan AR: p38 MAP kinase:
A convergence point in cancer therapy. Trends Mol Med. 10:125–129.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Irarrazabal CE, Liu JC, Burg MB and
Ferraris JD: ATM, a DNA damage-inducible kinase, contributes to
activation by high NaCl of the transcription factor TonEBP/OREBP.
Proc Natl Acad Sci USA. 101:8809–8814. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ko BC, Lam AK, Kapus A, Fan L, Chung SK
and Chung SS: Fyn and p38 signaling are both required for maximal
hypertonic activation of the osmotic response element-binding
protein/tonicity-responsive enhancer-binding protein
(OREBP/TonEBP). J Biol Chem. 277:46085–46092. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ferraris JD, Williams CK, Persaud P, Zhang
Z, Chen Y and Burg MB: Activity of the TonEBP/OREBP transactivation
domain varies directly with extracellular NaCl concentration. Proc
Natl Acad Sci USA. 99:739–744. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jauliac S, López-Rodriguez C, Shaw LM,
Brown LF, Rao A and Toker A: The role of NFAT transcription factors
in integrin-mediated carcinoma invasion. Nat Cell Biol. 4:540–544.
2002. View
Article : Google Scholar : PubMed/NCBI
|
|
39
|
O'Connor RS, Mills ST, Jones KA, Ho SN and
Pavlath GK: A combinatorial role for NFAT5 in both myoblast
migration and differentiation during skeletal muscle myogenesis. J
Cell Sci. 120:149–159. 2007. View Article : Google Scholar
|
|
40
|
Go WY, Liu X, Roti MA, Liu F and Ho SN:
NFAT5/TonEBP mutant mice define osmotic stress as a critical
feature of the lymphoid microenvironment. Proc Natl Acad Sci USA.
101:10673–10678. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Halterman JA, Kwon HM and Wamhoff BR:
Tonicity-independent regulation of the osmosensitive transcription
factor TonEBP (NFAT5). Am J Physiol Cell Physiol. 302:C1–C8. 2012.
View Article : Google Scholar :
|
|
42
|
Cuadrado A and Nebreda AR: Mechanisms and
functions of p38 MAPK signalling. Biochem J. 429:403–417. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Paillas S, Causse A, Marzi L, de Medina P,
Poirot M, Denis V, Vezzio-Vie N, Espert L, Arzouk H, Coquelle A, et
al: MAPK14/p38α confers irinotecan resistance to TP53-defective
cells by inducing survival autophagy. Autophagy. 8:1098–1112. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ye YC, Yu L, Wang HJ, Tashiro S, Onodera S
and Ikejima T: TNFα-induced necroptosis and autophagy via
supression of the p38-NF-κB survival pathway in L929 cells. J
Pharmacol Sci. 117:160–169. 2011. View Article : Google Scholar
|
|
45
|
Keil E, Höcker R, Schuster M, Essmann F,
Ueffing N, Hoffman B, Liebermann DA, Pfeffer K, Schulze-Osthoff K
and Schmitz I: Phosphorylation of Atg5 by the Gadd45β-MEKK4-p38
pathway inhibits autophagy. Cell death and differentiation.
20:3212013. View Article : Google Scholar
|