Introduction
Osteosarcoma (OS) is the most frequent primary bone
malignancy comprising almost 60% of all bone sarcomas and a leading
cause of cancer-related death among children, adolescents and young
adults (1). Despite modern
multimodality therapies, long-term survival rates of ∼70% can be
achieved only for those patients with resectable primary tumors and
non-metastatic disease at initial diagnosis (2). OS is believed to originate from
undifferentiated mesenchymal cells and consists of osteoblastic,
chondroblastic and fibroblastic cells or their combination. These
histological features suggest that OS may arise from impaired
differentiation of these immature cells into more mature types,
thus it has been recently suggested that OS can be regarded as a
differentiation disease. Restoring defective differentiation and/or
correction of this impairment may be able to regulate
tumorigenicity or reduce malignancy and increase the efficacy of
chemotherapy. Therefore, differentiation induction holds great
potential as a new modality of cancer therapy (3,4).
Bone morphogenetic proteins (BMPs), with >30
different isoforms in a variety of organisms, belong to the
transforming growth factor (TGF)-β superfamily known to regulate
cell proliferation, differentiation, apoptosis, chemotaxis,
angiogenesis and to participate in the development of most tissues
and organ in vertebrates (5).
BMP-2, a member of this large family of proteins, has been
originally identified for its ability to induce bone and cartilage
formation when implanted at non-bony sites in vivo (6). Similar to TGF-β, BMP-2 exerts its
effect via specific serine-threonine kinase receptors, type 1A
(BMPR1A), type 1B (BMPR1B) and type 2 (BMPR2). BMP receptor type 2
is activated upon BMP-2 binding and subsequently induces
oligomerization of the receptor complex, resulting in
phosphorylation of the type 1 receptor and recruitment of
downstream signaling Sma- and Mad-related proteins (Smad1, Smad5
and Smad8). The phosphorylated Smad1/5/8 can bind to a common
mediator Smad4 in a heterodimeric complex that is translocated to
the nucleus where it induces the expression of responsive genes
such as Runx-2 that mediate the osteogenic activity of BMP-2
(7,8). In addition to the canonical Smad
pathway, non-Smad pathways mitogen-activated protein kinase (MAPK)
pathways including p38, c-jun-N-terminal kinase (JNK) and
extracellular signal-regulated kinase (ERK1/2) pathway, may also
play important roles in cell proliferation and differentiation
(9,10).
Recently, several lines of evidence have revealed
BMP-2 signaling in cancer cells. Expressions of BMP-2 and BMP
receptors have been found to be altered in many tumor types
(11–17). Bioengineered recombinant human
BMP-2 (rhBMP-2) has been demonstrated to increase tumor growth of
lung carcinoma (18), pancreatic
carcinoma (13) and prostate
cancers cells in the absence of androgen (19). However, the effect of rhBMP-2 on
cancer cells remains controversial. Some studies have shown rhBMP-2
exerts inhibitory effects on many tumor cells including breast
cancer, myeloma, gastric cancer, colon cancer and prostate cancer
(19–24). We have recently reported that
rhBMP-2 inhibits the tumorigenicity of cancer stem cells with high
aldehyde dehydrogenase (ALDH) activity (ALDHbr cells)
derived from human OS xenografts (25). We also reported that rhBMP-2
inhibits tumor growth and induces bone formation in human renal
cell carcinoma cells (26). These
results led us to examine the possibility that BMP-2 induces bone
formation in human OS cells.
In the present study, we sought to more extensively
explore the effect of BMP-2 on heterogeneous population of
ALDHbr cells and their progenies with low ALDH activity
(ALDHlo cells) derived from human OS xenografts. Our
findings that BMP-2 differentially induces the expression of
osteogenic marker genes in ALDHbr and ALDHlo
cells mediated by Smad signaling pathway provide a striking
implication with the use of BMP-2 to restrict human OS
expansion.
Materials and methods
Human OS cell culture
Human OS OS99-1 cell line originally derived from a
highly aggressive primary human OS (27) was a generous gift from Dr Sheila M.
Nielsen-Preiss (Montana State University, Bozeman, MT, USA). Cells
were routinely cultured in Dulbecco’s modified Eagle’s medium
(DMEM)/F12 medium (Gibco, Carlsbad, CA, USA) supplemented with 10%
fetal bovine serum (Gibco) in a humidified atmosphere of 5%
CO2 in air at 37°C and used when in the log phase of
growth.
Xenografts, tumor dissociation, ALDEfluor
cell analysis and flow cytometry
Immunodeficient non-obese diabetic (NOD)/severe
combined immunodeficient (SCID) (NOD/SCID) mice (5- to 6-week-old)
were purchased from Harlan Laboratories (Harlan Laboratories,
Indianapolis, IN, USA). All animal studies were performed according
to protocol approved by the Institutional Animal Care and Use
Committee of the University of Michigan. Xenografts and basic
experimental procedures for tumor dissociation, ALDEfluor cell
analysis and flow cytometry to isolate ALDHbr cells and
ALDHlo cells were detailed elsewhere (25,28).
Semi-quantitative real-time polymerase
chain reaction (PCR)
To test the expression of BMP receptors in
ALDHbr cells and ALDHlo cells, total RNA was
extracted and semi-quantitative PCR was run as described previously
(25,28).
Quantitative real-time polymerase chain
reaction (qPCR)
To further compare the expression of BMP receptors
in freshly sorted ALDHbr cells and ALDHlo
cells, quantitative real-time PCR of BMPR1A (Hs01034913_g1), BMPR1B
(Hs00176144_m1) and BMPR2 (Hs00176148_m1) and β-actin gene
expression were run in triplicate using Eppendorf Mastercycler
Realplex Detection System (Eppendorf, Germany). All primers were
designed and purchased from Applied Biosystems (Life Technologies
Corp., Carlsbad, CA, USA). To test the expression of osteogenic
markers in sorted ALDHbr and ALDHlo cells in
response to BMP-2 (GenScript Corp., Piscataway, NJ, USA), freshly
sorted cells were washed and cultured for expansion and then
inoculated in a 6-well culture plate. After 24-h incubation, the
medium was replaced with 1% serum-containing medium for 24 h and
then replaced with 0 and 300 ng/ml BMP-2 diluted in 1%
serum-containing medium. After 48 h total RNA was extracted as
described above. Quantitative real-time PCR of osteogenic markers
Runx-2 (Hs00231692_m1), Osx (Hs018666874_s1), ALP (Hs01029144_m1)
and collagen type I (Hs00164004_m1) and β-actin gene expression
were run in triplicate as described previously (25,28).
Western blot analysis
Freshly sorted cells were washed and cultured for
expansion and then inoculated, at 5×105 cells, in a
10-cm culture dish and grown to ∼80–90% confluence. The medium was
replaced with 1% serum-containing medium for 24 h and then replaced
with 0 and 300 ng/ml BMP-2 diluted in 1% serum-containing medium
for the time indicated. Cells were lysed as previously described
(26). The protein concentrations
were then measured using BCA protein assay kit (Thermo Scientific,
Pittsburgh, PA, USA). Next, the protein lysates were separated by
sodium dodecyl sulfate-polyacrylamide (SDS-PAGE) gel
electrophoresis and then transferred onto hybond-C pure
nitrocellulose membrane (Amersham, Piscataway, NJ, USA). Membranes
were blocked with TBS containing 0.1% Tween-20 containing 5%
non-fat dry milk and then incubated with primary antibody
overnight. The primary antibodies were as follows:
anti-phospho-Smad1/5/8, anti-phospho-ERK1/2,
anti-anti-phospho-p38MAPK and anti-GAPDH (Cell Signaling
Technology, Danvers, MA, USA). After washing with TBS with
Tween-20, the secondary antibodies were added. Finally, the
proteins were visualized with the ECL chemiluminescence system
(Amersham).
In vivo co-treatment experiments
Freshly sorted ALDHbr cells and
ALDHlo cells (1×104) treated with BMP-2 or
vehicle control were subcutaneously injected into right and left
lower abdominal area of NOD/SCID mice. A more detailed description
of this procedure can be found in Wang et al (26). Tumor growth was monitored weekly
for 12 weeks. Tumors formed were removed and a portion of each
tumor was processed for histological analysis.
Bone formation analysis
The mice were sacrificed and specimens harvested 12
weeks after implantation. Radiographs were obtained using Faxitron
X-ray unit (Field Emission Corp., McMinniville, OR, USA). For
microcomputed tomograpgy (micro-CT) analysis, specimens were
scanned at 8.93 μm voxel resolution on a micro-CT scanner
(EVS Corp.), with a total of 667 slices per scan. GEMS MicroView
software (GE Healthcare Biosciences) was used to make a
three-dimensional reconstruction from the set of scans. Three
samples per treatment were assessed. A cylindrical region of
interest (ROI) was concentrically positioned over the defect site
and kept constant for all the samples. The total volume of
newly-formed bone within the ROI was measured by assigning a
predetermined threshold and bone mineral content and was recorded
as previously described (29).
Histological analysis
For histomorphometry, specimens were stained with
haematoxylin and eosin (H&E) and Masson’s trichrome staining to
show collagen type I protein in the newly formed bone.
Undecalcified sections were stained with von-Kossa staining to
identify the calcification during osteogenesis in the tumor.
Statistical analysis
Data were expressed as mean ± SD. Statistically
significant differences were determined by two-tailed Student’s
t-test and defined as P<0.05.
Results
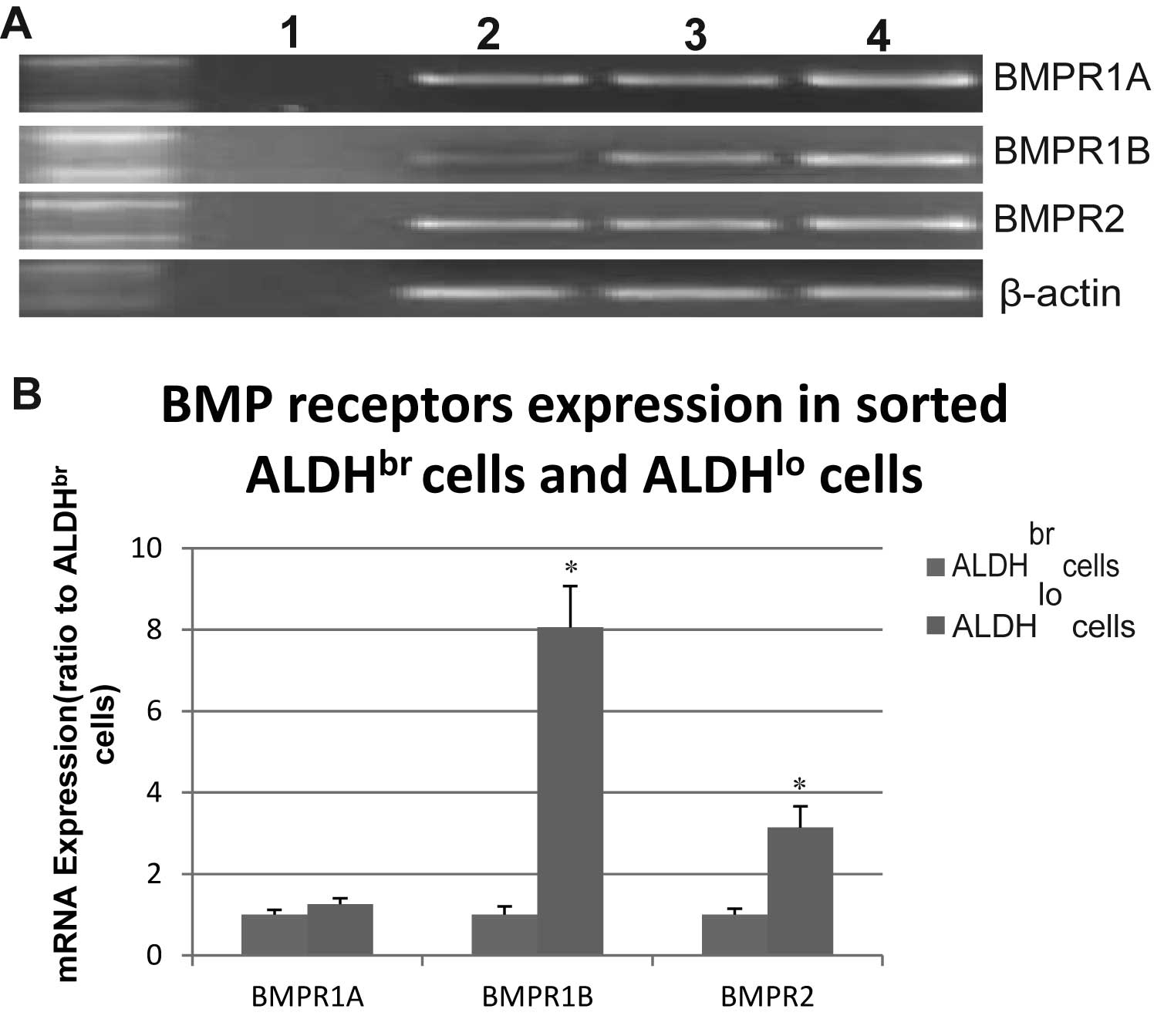
BMP receptor mRNA expression in freshly
sorted ALDHbr and ALDHlo cells
We first examined the mRNA expression of the BMP
type 1 and 2 receptors in freshly sorted ALDHbr and
ALDHlo cells derived from OS99-1 xenografts. As shown in
Fig. 1, all BMP-2 receptors were
expressed in ALDHbr and ALDHlo cells.
Quantitative RT-PCR revealed that there was significantly higher
mRNA expression of BMPR1B and BMPR2 in ALDHlo cells
compared with that in ALDHbr cells (P<0.05) (Fig. 1B) and the BMPR1B expression in
ALDHlo cells was around 8-fold higher than that in
ALDHbr cells. The mRNA expression of BMPR1A was higher
in ALDHlo cells compared with that in ALDHbr
cells, but there was no significant difference between the two cell
types (Fig. 1B).
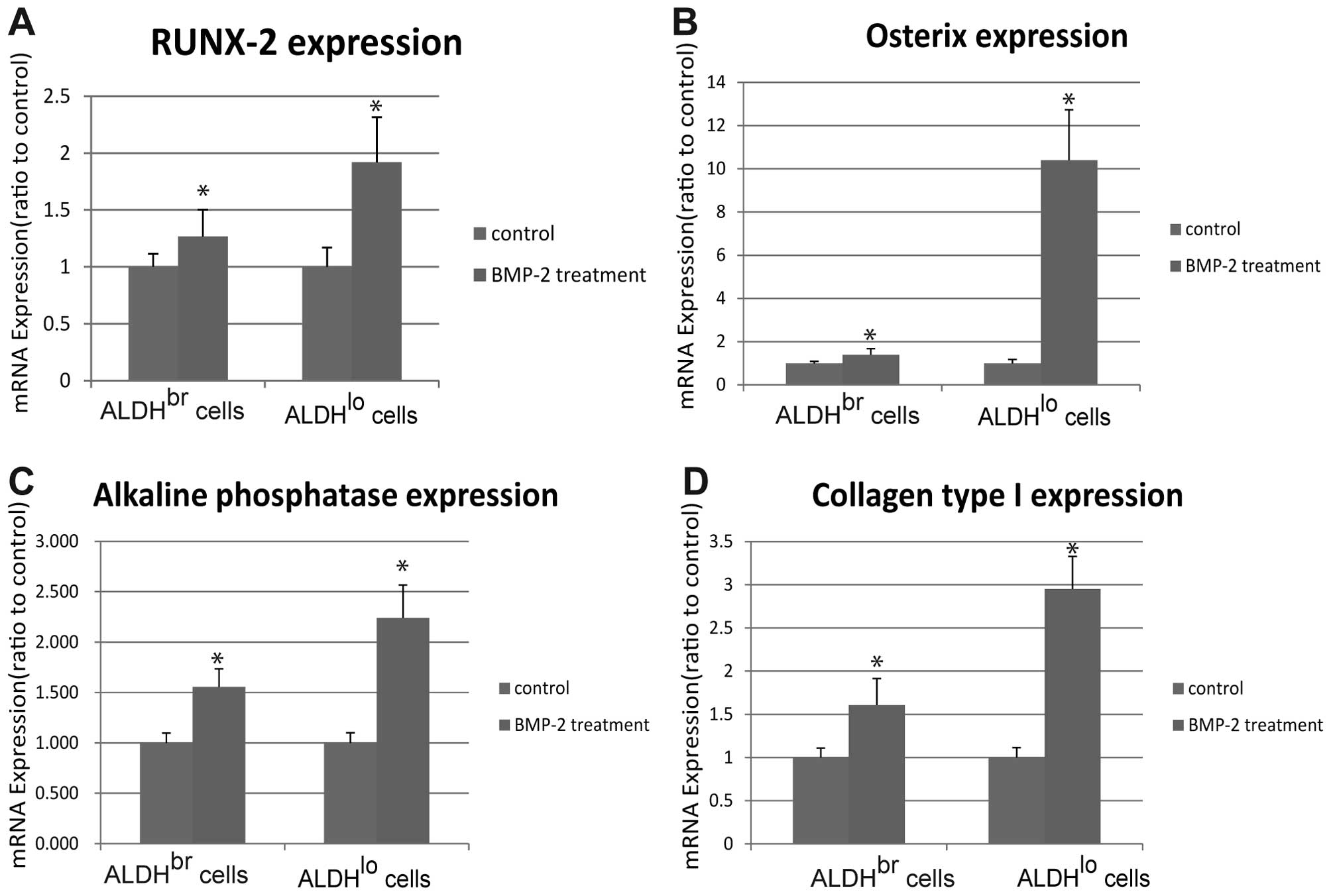
Expression of osteogenic marker genes
induced by BMP-2
BMP-2 has been shown to act as a potent inducer of
osteogenic differentiation (30).
Based on our previous report that BMP-2 significantly inhibits the
growth of ALDHbr cells at 300 ng/ml for 48 h (25), we chose to treat ALDHbr
and ALDHlo cells with BMP-2 at the same dose for the
following experiment. As shown in Fig.
2, ALDHbr and ALDHlo cells treated with
BMP-2 had significantly higher expression of Runx-2, Osx, ALP and
collagen type I than the cells treated with the same volume of
vehicle (P<0.05). In addition, BMP-2 induced higher
transcription of osteogenic markers in ALDHlo cells than
in ALDHbr cells and Osx expression in ALDHlo
cells treated with BMP-2 showed around 10-fold higher than that in
ALDHlo cells treated with vehicle control (Fig. 2B).
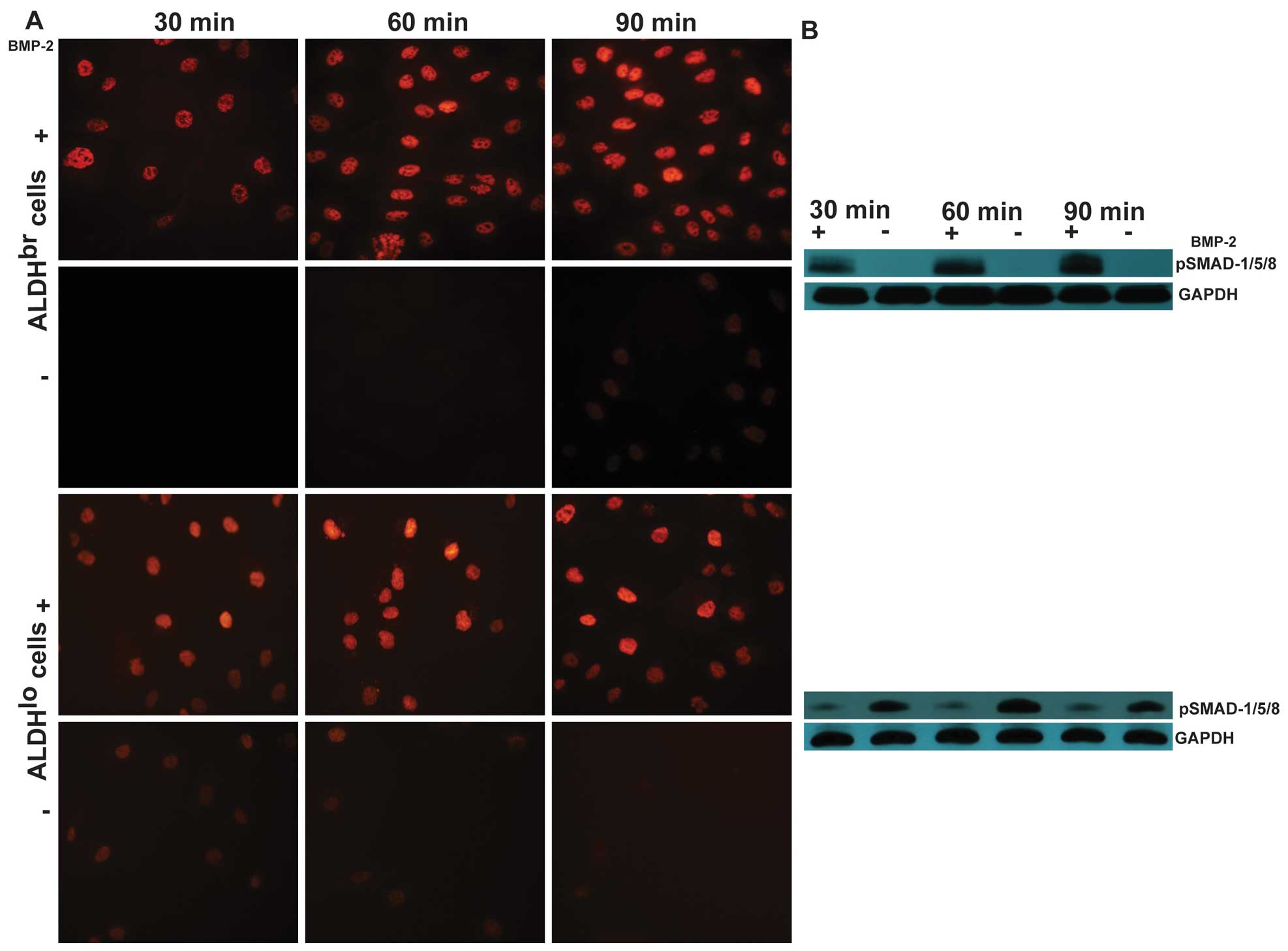
BMP-2 activates Smad pathway in sorted
ALDHbr and ALDHlo cells
To determine whether BMP-2 signaling pathways were
functional in ALDHbr and ALDHlo cells, we
first stimulated cells with BMP-2 and examined the phosphorylation
and nuclear translocation of Smad1/5/8, since BMP-2 is thought to
predominantly act through the activation of these transcription
factors (31). Both
ALDHbr cells and ALDHlo cells responded to
BMP-2 treatment in cell culture. Immunofluorescence staining of
phosphorylated Smad1/5/8 using an antibody which specifically
recognizes the phosphorylated forms demonstrated activated Smad
proteins were clearly located in the nuclei of ALDHbr
and ALDHlo cells treated with BMP-2 at 300 ng/ml for 30,
60 and 90 min, respectively (Fig.
3A). The ability of BMP-2 to phosphorylate Smad1/5/8 was then
confirmed by western blot analysis after cells were treated with
BMP-2 or vehicle control. As shown in Fig. 3B, western blot analysis of
phosphorylation of Smad1/5/8 revealed the highest levels of
activated Smad proteins in ALDHbr cells at 90 min, while
in ALDHlo cells at 60 min. These results revealed that
BMP receptors are functional and BMP-2 can induce a classical Smad
signaling pathway in ALDHbr and ALDHlo
cells.
BMP-2 activates ERK and p38 MAPKS
pathways in sorted ALDHlo cells
BMP-2 has also been shown to induce osteoblastic
differentiation through extracellular signal-regulated kinase 1/2
(ERK1/2) and p38 mitogen-activated protein kinase (MAPK) pathways
in human osteoblast cells (32).
To determine if BMP-2 stimulation leads to MAPK activation in
sorted ALDHbr and ALDHlo cells, we examined
the phosphorylation of p38 and ERK1/2 using a specific antibody
that recognizes phosphorylated serine sites. No BMP-2 induced
phosphorylation of p38 and ERK1/2 was observed in sorted
ALDHbr cells (not shown). However, in ALDHlo
cells, constitutive phosphorylation of ERK and p38 MARKs was
visible and a slight transient increase was induced in the
cytoplasm after 10 min of BMP-2 treatment (Fig. 4).
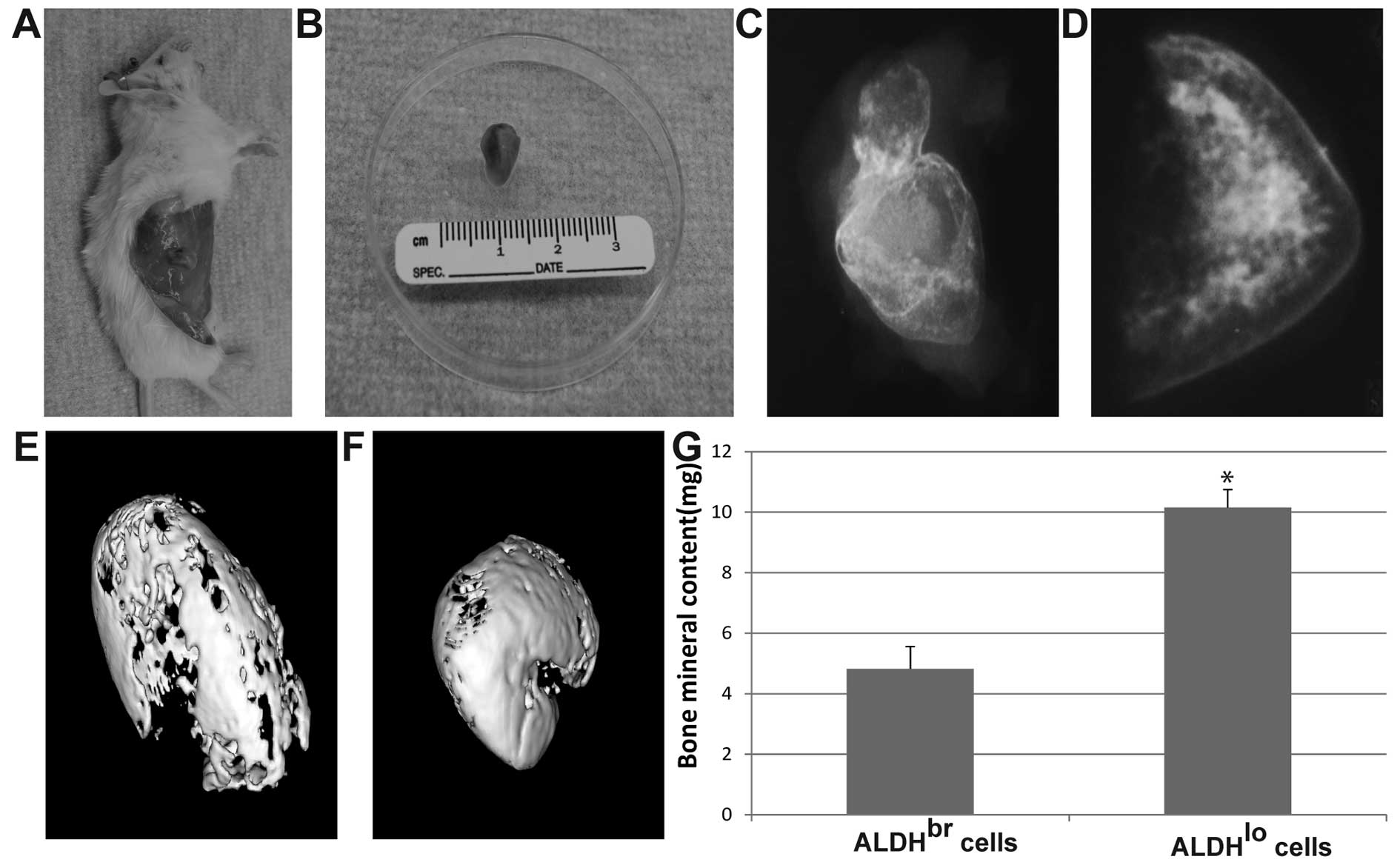
BMP-2 induces bone formation in sorted
ALDHbr cells and ALDHlo cells in vivo
We next injected freshly sorted ALDHbr
and ALDHlo cells with BMP-2 treatment or vehicle control
subcutaneously into NOD/SCID mice. Both ALDHbr cells and
ALDHlo cells were induced to form bone in the mouse
ectopic subcutaneous model. The bone formation induced by the
addition of 30 μg/animal of BMP-2 with ALDHlo
cells was palpable within 1–2 weeks after injection (Fig. 5A and B), while the bone formation
induced by ALDHbr cells was palpable at 7–8 weeks. This
was verified by 2D X-ray analyses (Fig. 5C and D) and 3D micro-CT (Fig. 5E and F). We chose a pre-determined
threshold that resembles denser, cortical bone as this would be of
direct clinical relevance. Bone formation induced by BMP-2 in
ALDHlo cells showed significantly more bone mineral
content compared to that in ALDHbr cells (P<0.05)
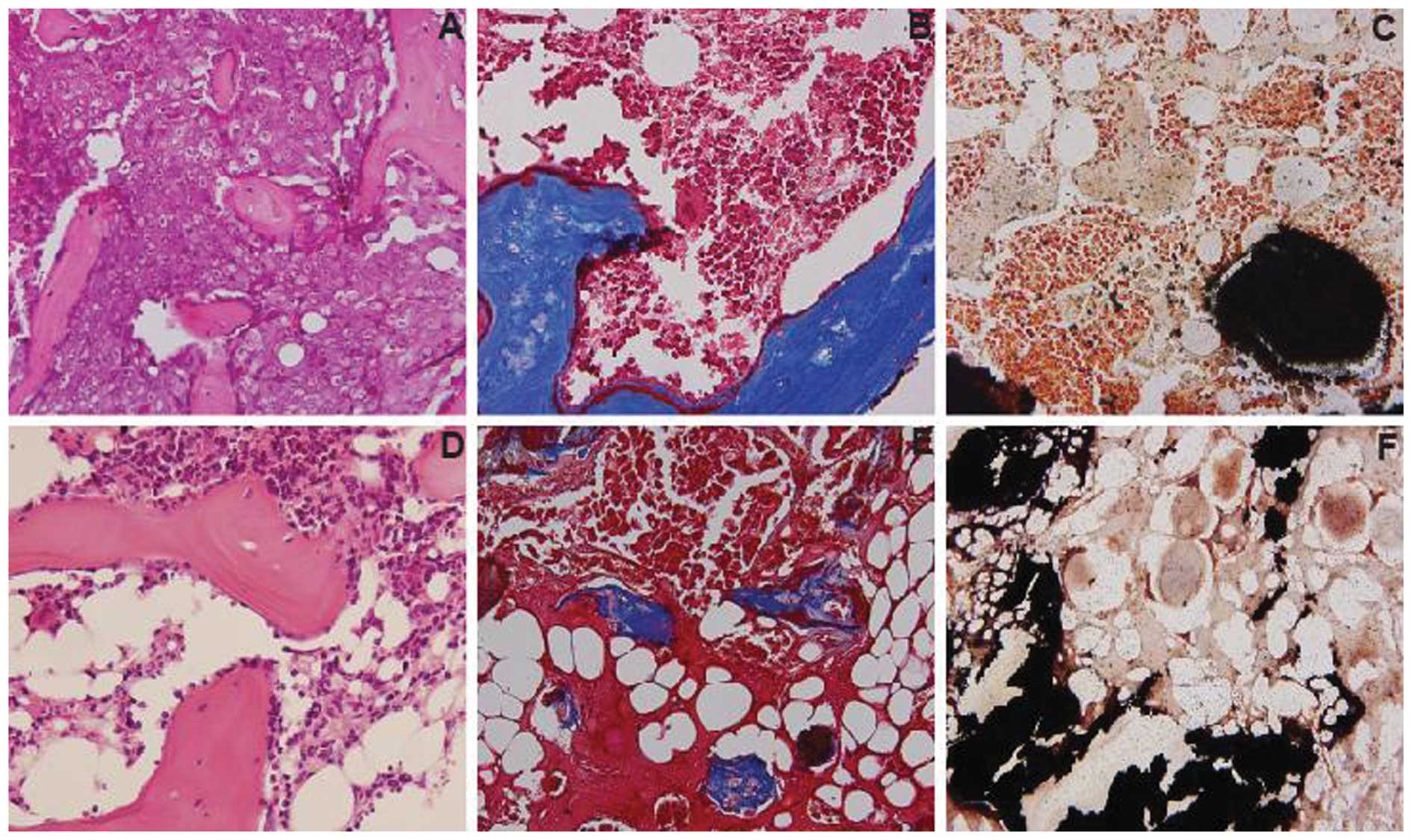
(Fig. 5G). Hematoxylin and eosin
staining revealed that bone marrow which included blood vessels,
fat and hematopoietic cells was observed in the bone formation
induced in ALDHlo cells, which was also confirmed by
Masson’s trichrome staining and von-Kossa staining (Fig. 6D–F). However, no marrow was found
in the bone formation induced in ALDHbr cells (Fig. 6A–C).
Discussion
OS is the most common malignant tumor of the bone in
the pediatric age group, with an incidence of 8.7 per million in
children and adolescent under the age of 20 years (33). OS is a clinically and molecularly
heterogeneous group of malignancies characterized by varying
degrees of mesenchymal differentiation. It has been proposed to be
a differentiation-flawed disease and believed to arise from
mesenchymal stem cells or osteoprogenitor cells resulting from a
disruption in the osteoblast differentiation (34). Although modern multi-modality
therapies have improved the 5-year survival rate of OS patients,
recurrent and/or metastatic OS tumors are more aggressive and
usually resistant to conventional cancer therapies. Identification
of the critical differentiation defects in OS tumors may lead to a
rational design of therapeutic strategies that can induce terminal
differentiation of OS cells through alternative differentiation
pathways and/or bypassing the differentiation defects (34).
Bone morphogenetic proteins (BMPs) were originally
identified as osteoinductive cytokines to induce the entire cascade
of cartilage and bone formation in vivo (35). There are >30 isotypes BMPs, with
BMP-2 and BMP-4 having 92% of homology. Studies on a variety of
human cancer cells revealed that BMPs produce a complex set of
effects in cancer, in which they can function as either
protumorigenic oncogene or antitumorigenic tumor suppressor,
depending on the stage of disease (36). BMP-4 has been reported to induce
differentiation of brain tumor stem cells in vivo (37). More recently, we have demonstrated
that treatment of human OS-derived tumor-initiating cells with
BMP-2 inhibits cell proliferation and importantly, reduces the
ability to form tumors in immunodeficient mice (25). We also found that BMP-2 has an
inhibitory effect on human renal cell carcinoma cells and induces
bone formation (26). BMP-2 has
also been reported to induce in vitro differentiation of
canine osteosarcoma cells (38).
However, little is known about bone formation of BMP-2 in human OS
cells.
It has been reported that BMP signaling for the
growth and differentiation of normal or neoplastic cells is
dependent on its receptors (39).
There are currently 3 characterized BMP receptors: BMPR1A, BMPR1B
and BMPRR2. Activation of the BMP receptor complex initiates
intracellular signaling transduction (6). In the present study, using regular
RT-PCR, we determined that all 3 types of BMP receptors were
expressed in freshly sorted ALDHbr and ALDHlo
cells derived from OS99-1 xenografts, suggesting that BMP-2 could
bind to its receptors and activate cell signaling to affect
osteosarcoma cell activities. By using quantitative RT-PCR, we
further demonstrated that there was significantly higher mRNA
expression of BMPR1B and BMPR2 in ALDHlo cells compared
with that in ALDHbr cells and the BMPR1B expression in
ALDHlo cells showed ∼8-fold higher than that in
ALDHbr cells. These results indicate higher expression
of BMPR1B in ALDHlo cells might correlate with its
differentiated property as we have previously shown that
ALDHbr cells from human osteosarcoma OS99-1 xenografts
has been identified as cancer stem cells and have the capability to
produce differentiated progeny ALDHlo cells (28). In agreement with this view,
previous studies have shown that the expression of BMPR1B and BMPR2
in benign ovarian tumors and normal ovarian tissue was
significantly higher than those in ovarian cancer tissue (40). In addition, Hall and Miller
(41) revealed that increased
expression of BMPR1B in response to BMP2/4 promotes neuronal and
astrocytic differentiation of neural stem cell. Forced expression
of BMPR1B either by transgene expression or demethylation of the
promoter restores differentiation capabilities and induces loss of
their tumorigenicity in glioblastoma tumor initiating cells
(42). Thus, our findings further
support that non-tumorigenic ALDHlo cells are more
differentiated progeny cells compared with tumorigenic
ALDHbr cells. Higher expression of BMPR1B and BMPR2 in
non-tumorigenic ALDHlo cells suggests that BMP-2 may act
through its main receptors, BMPR1B and BMPR2, to induce osteogenic
differentiation in non-tumorigenic ALDHlo cells.
BMP-2 has been shown to play important roles in the
regulation of differentiation of many different cell types along
osteoblastic pathways (43,44).
BMP-2 target genes include a growing number of tissue-determining
transcription factors that promote differentiation of different
cell types toward the osseous cell phenotypes (45). The runt homology domain factor
Runx-2 (Cbfa1) and Osx have been widely accepted as
osteoblast-specific transcriptional factors along osteoblastic
pathways since neither Runx-2 nor Osx null mice form mature
osteoblasts (46). Osx expression
is more restricted to osteoblasts than Runx-2 (46). ALP is an early osteoblast marker
and collagen type I comprises 85–90% of the total bone matrix
(47). Runx-2 and collagen type I
are known to be upregulated by BMP-2 in human prostate cancer
cells, osteosarcoma cells renal carcinoma cells (21,25,26).
Therefore, using quantitative RT-PCR, we found that Runx-2, Osx,
ALP and collagen type I were significantly upregulated in sorted
ALDHbr and ALDHlo cells treated with BMP-2 at
a concentration of 300 ng/ml for 48 h when compared with untreated
controls. BMP-2 induced higher transcription of osteogenic markers
in ALDHlo cells than in ALDHbr cells and
especially Osx expression in ALDHlo cells treated with
BMP-2 showed ∼10-fold higher than that in ALDHlo cells
treated with vehicle control. These results indicated that
non-tumorigenic ALDHlo cells are more likely to be
induced to express higher level of osteogenic markers than
tumorigenic ALDHbr cells. Our findings suggested that
BMP-2 might induce human osteosarcoma cells to express an
osteoblastic phenotype and thus activate osteogenic differentiation
to form bone.
BMP-2 is thought to predominantly exert their effect
via binding to 2 types of receptors, leading to downstream
transduction of the BMP signal through phosphorylation of specific
intracellular proteins called Smads (48). To further explore whether or not
BMP-2 signaling pathways were functional in ALDHbr and
ALDHlo cells, the phosphorylation and nuclear
translocation of Smad1/5/8 were detected by immunostaining and
western blot analysis using an antibody which specifically
recognizes the phosphorylated Smad1/5/8. Our results demonstrated
that the highest level of phosphorylation of Smad1/5/8 in
ALDHbr cells at 90 min and in ALDHlo cells at
60 min. The elevation of Smad activation at the 60- or 90-min
time-point, before osteogenic markers Runx-2, Osx, ALP and collagen
type I were significantly upregulated after 48 h of treatment,
suggests that in transcription regulation of bone-related
osteogenic markers gene expression by BMP-2 might be mediated via
the Smads transducer. In addition, BMP-2 was shown to activate
ERK1/2 and p38 MAPK pathways in ALDHlo cells, but not in
ALDHbr cells. This difference in the activation of MAPK
pathways between ALDHlo cells and ALDHbr
cells may be due to the type of receptors expressed by the cells.
Therefore, activation of MAPK pathways in ALDHlo cells
by BMP-2 might have a synergetic effect in the induction of higher
Osx mRNA expression since BMP-2 has been demonstrated to induce Osx
expression through phosphorylation of p38 (45). Further experiments are therefore
required to understand the different gene expression between
ALDHlo and ALDHbr cells induced by BMP-2.
Determining the bone formation of BMP-2 in
osteosarcoma cells in vivo is essential for determining the
potential use of BMP-2 clinically because in vitro analysis
does not always reflect exactly the in vivo situation. In
the present study, BMP-2 was shown to induce bone formation in both
tumorigenic ALDHbr cells and non-tumorigenic
ALDHlo cells. All animals receiving non-tumorigenic
ALDHlo (1×104) cells treated with 30
μg BMP-2 per animal resulted in significant bone formation
within 1–2 weeks after injection in NOD/SCID mice. The bone
formation was further confirmed by radiograph and micro-CT and
histopathological analysis of sections from bony tissues formed by
ALDHbr and ALDHlo cells treated with BMP-2.
These findings are in agreement with previous results indicating
that viral vector-induced expression of BMP-2 in a breast cancer
cell line and a colon cancer cell line induces calcification of
tumors to arrest tumor growth (49). By contrast, Luo et al
(30) reported that osteogenic
BMPs promote tumor growth of human osteosarcoma. They infected
osteosarcoma cells with adenoviral vectors to secret BMP-2 and
BMP-9. Factors such as the concentration and distribution of these
endogenous BMP compared to exogenous application of BMP-2 as done
in this study may be the possible explanation for the proliferation
of osteosarcoma cells observed in that investigation.
Our study is the first to provide evidence
indicating that exogenous BMP-2 induces bone formation in human
osteosarcoma cells. However, the use of only one cell line provides
limited evidence. Further research using more cell lines and
primary tumors is therefore necessary to confirm the findings of
this study.
In conclusion, our findings suggest that BMP-2 can
upregulate osteogenic markers Runx-2, Osx, ALP and collagen type I
gene expression in both ALDHbr cells and
ALDHlo cells and this osteoinductive effect may be
mediated by Smad signaling pathway. Subsequently, we demonstrated
that BMP-2 induced bone formation in vivo. Our findings
present evidence on a potential therapeutic application of
exogenous BMP-2 on human osteosarcoma by inducing differentiation
of tumorigenic cells along an osteogenic pathway.
Acknowledgements
The authors gratefully acknowledge
funding support provided by the National Institutes of Health. This
study was partially supported by grant no. R01 AR056649 from
NIAMS/NIH and the pilot research fund of the Department of
Neurosurgery, University of Michigan. The authors would also like
to thank Dr Sheila M. Nielsen-Preiss from Montana State University
for the generous gift of human osteosarcoma cell line OS99-1, Mrs.
Holly Wagner for assistance in the preparation of the manuscript
and Mr. Martin J. White for flow cytometry.
References
|
1.
|
Ottaviani G and Jaffe N: The epidemiology
of osteosarcoma. Cancer Treat Res. 152:3–13. 2009. View Article : Google Scholar
|
|
2.
|
Gorlick R and Khanna C: Osteosarcoma. J
Bone Miner Res. 25:683–691. 2010. View
Article : Google Scholar
|
|
3.
|
Shimo T, Matsumura S, Ibaragi S, et al:
Specific inhibitor of MEK-mediated cross-talk between ERK and p38
MAPK during differentiation of human osteosarcoma cells. J Cell
Commun Signal. 1:103–111. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Luk F, Yu Y, Dong HT, Walsh WR and Yang
JL: New gene groups associated with dissimilar osteoblastic
differentiation are linked to osteosarcomagenesis. Cancer Genomics
Proteomics. 8:65–75. 2011.PubMed/NCBI
|
|
5.
|
Hruska KA, Mathew S and Saab G: Bone
morphogenetic proteins in vascular calcification. Circ Res.
97:105–114. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6.
|
Barboza E, Caula A and Machado F:
Potential of recombinant human bone morphogenetic protein-2 in bone
regeneration. Implant Dent. 8:360–367. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Hay E, Lemonnier J, Fromigue O, Guenou H
and Marie PJ: Bone morphogenetic protein receptor IB signaling
mediates apoptosis independently of differentiation in osteoblastic
cells. J Biol Chem. 279:1650–1658. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8.
|
Wrana JL: Regulation of Smad activity.
Cell. 100:189–192. 2000. View Article : Google Scholar
|
|
9.
|
Moustakas A and Heldin CH: Non-Smad
TGF-beta signals. J Cell Sci. 118:3573–3584. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10.
|
Guicheux J, Lemonnier J, Ghayor C, Suzuki
A, Palmer G and Caverzasio J: Activation of p38 mitogen-activated
protein kinase and c-Jun-NH2-terminal kinase by BMP-2 and their
implication in the stimulation of osteoblastic cell
differentiation. J Bone Miner Res. 18:2060–2068. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Arnold SF, Tims E and McGrath BE:
Identification of bone morphogenetic proteins and their receptors
in human breast cancer cell lines: importance of BMP2. Cytokine.
11:1031–1037. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Hardwick JC, Van Den Brink GR, Bleuming
SA, et al: Bone morphogenetic protein 2 is expressed by and acts
upon, mature epithelial cells in the colon. Gastroenterology.
126:111–121. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13.
|
Kleeff J, Maruyama H, Ishiwata T, et al:
Bone morphogenetic protein 2 exerts diverse effects on cell growth
in vitro and is expressed in human pancreatic cancer in vivo.
Gastroenterology. 116:1202–1216. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Soares AF, Xavier RL, da Costa Miguel MC,
de Souza LB and Pinto LP: Bone morphogenetic protein-2/4 and bone
morphogenetic protein receptor type IA expression in metastatic and
nonmetastatic oral squamous cell carcinoma. Am J Otolaryngol.
31:266–271. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Kim IY, Lee DH, Lee DK, et al: Decreased
expression of bone morphogenetic protein (BMP) receptor type II
correlates with insensitivity to BMP-6 in human renal cell
carcinoma cells. Clin Cancer Res. 9:6046–6051. 2003.PubMed/NCBI
|
|
16.
|
Katoh M and Terada M: Overexpression of
bone morphogenic protein (BMP)-4 mRNA in gastric cancer cell lines
of poorly differentiated type. J Gastroenterol. 31:137–139. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Langenfeld EM, Calvano SE, Abou-Nukta F,
Lowry SF, Amenta P and Langenfeld J: The mature bone morphogenetic
protein-2 is aberrantly expressed in non-small cell lung carcinomas
and stimulates tumor growth of A549 cells. Carcinogenesis.
24:1445–1454. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Langenfeld EM, Kong Y and Langenfeld J:
Bone morphogenetic protein 2 stimulation of tumor growth involves
the activation of Smad-1/5. Oncogene. 25:685–692. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Ide H, Yoshida T, Matsumoto N, et al:
Growth regulation of human prostate cancer cells by bone
morphogenetic protein-2. Cancer Res. 57:5022–5027. 1997.PubMed/NCBI
|
|
20.
|
Beck SE, Jung BH, Fiorino A, et al: Bone
morphogenetic protein signaling and growth suppression in colon
cancer. Am J Physiol Gastrointest Liver Physiol. 291:G135–G145.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Brubaker KD, Corey E, Brown LG and
Vessella RL: Bone morphogenetic protein signaling in prostate
cancer cell lines. J Cell Biochem. 91:151–160. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Ghosh-Choudhury N, Woodruff K, Qi W,
Celeste A, Abboud SL and Ghosh Choudhury G: Bone morphogenetic
protein-2 blocks MDA MB 231 human breast cancer cell proliferation
by inhibiting cyclin-dependent kinase-mediated retinoblastoma
protein phosphorylation. Biochem Biophys Res Commun. 272:705–711.
2000. View Article : Google Scholar
|
|
23.
|
Kawamura C, Kizaki M, Yamato K, et al:
Bone morphogenetic protein-2 induces apoptosis in human myeloma
cells with modulation of STAT3. Blood. 96:2005–2011.
2000.PubMed/NCBI
|
|
24.
|
Wen XZ, Miyake S, Akiyama Y and Yuasa Y:
BMP-2 modulates the proliferation and differentiation of normal and
cancerous gastric cells. Biochem Biophys Res Commun. 316:100–106.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Wang L, Park P, Zhang H, et al: BMP-2
inhibits the tumorigenicity of cancer stem cells in human
osteosarcoma OS99-1 cell line. Cancer Biol Ther. 11:457–463. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Wang L, Park P, Zhang H, et al: BMP-2
inhibits tumor growth of human renal cell carcinoma and induces
bone formation. Int J Cancer. 131:1941–1950. 2012. View Article : Google Scholar
|
|
27.
|
Gillette JM, Gibbs CP and Nielsen-Preiss
SM: Establishment and characterization of OS 99-1, a cell line
derived from a highly aggressive primary human osteosarcoma. In
Vitro Cell Dev Biol Anim. 44:87–95. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Wang L, Park P, Zhang H, La Marca F and
Lin CY: Prospective identification of tumorigenic osteosarcoma
cancer stem cells in OS99-1 cells based on high aldehyde
dehydrogenase activity. Int J Cancer. 128:294–303. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Eguchi Y, Wakitani S, Imai Y, et al:
Antitumor necrotic factor agent promotes BMP-2-induced ectopic bone
formation. J Bone Miner Metab. 28:157–164. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Luo X, Chen J, Song WX, et al: Osteogenic
BMPs promote tumor growth of human osteosarcomas that harbor
differentiation defects. Lab Invest. 88:1264–1277. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Le Page C, Puiffe ML, Meunier L, et al:
BMP-2 signaling in ovarian cancer and its association with poor
prognosis. J Ovarian Res. 2:42009.PubMed/NCBI
|
|
32.
|
Kuo PL, Hsu YL, Chang CH and Chang JK:
Osthole-mediated cell differentiation through bone morphogenetic
protein-2/p38 and extracellular signal-regulated kinase 1/2 pathway
in human osteoblast cells. J Pharmacol Exp Ther. 314:1290–1299.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Mirabello L, Troisi RJ and Savage SA:
International osteosarcoma incidence patterns in children and
adolescents, middle ages and elderly persons. Int J Cancer.
125:229–234. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Tang N, Song WX, Luo J, Haydon RC and He
TC: Osteosarcoma development and stem cell differentiation. Clin
Orthop Relat Res. 466:2114–2130. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Wozney JM, Rosen V, Celeste AJ, et al:
Novel regulators of bone formation: molecular clones and
activities. Science. 242:1528–1534. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
36.
|
Katsuno Y, Hanyu A, Kanda H, et al: Bone
morphogenetic protein signaling enhances invasion and bone
metastasis of breast cancer cells through Smad pathway. Oncogene.
27:6322–6333. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Piccirillo SG, Reynolds BA, Zanetti N, et
al: Bone morphogenetic proteins inhibit the tumorigenic potential
of human brain tumour-initiating cells. Nature. 444:761–765. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Nozaki K, Kadosawa T, Nishimura R,
Mochizuki M, Takahashi K and Sasaki N: 1,25-Dihydroxyvitamin D3,
recombinant human transforming growth factor-beta 1 and recombinant
human bone morphogenetic protein-2 induce in vitro differentiation
of canine osteosarcoma cells. J Vet Med Sci. 61:649–656. 1999.
View Article : Google Scholar
|
|
39.
|
Orui H, Imaizumi S, Ogino T and Motoyama
T: Effects of bone morphogenetic protein-2 on human tumor cell
growth and differentiation: a preliminary report. J Orthop Sci.
5:600–604. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Ma Y, Ma L, Guo Q and Zhang S: Expression
of bone morphogenetic protein-2 and its receptors in epithelial
ovarian cancer and their influence on the prognosis of ovarian
cancer patients. J Exp Clin Cancer Res. 29:852010. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Hall AK and Miller RH: Emerging roles for
bone morphogenetic proteins in central nervous system glial
biology. J Neurosci Res. 76:1–8. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Lee J, Son MJ, Woolard K, et al:
Epigenetic-mediated dysfunction of the bone morphogenetic protein
pathway inhibits differentiation of glioblastoma-initiating cells.
Cancer Cell. 13:69–80. 2008. View Article : Google Scholar
|
|
43.
|
Yamate J, Kotera T, Kuwamura M and Kotani
T: Potential osteogenic differentiation of cisplatin-resistant rat
malignant fibrous histiocytoma-derived cell lines. Exp Toxicol
Pathol. 58:299–309. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44.
|
Reddi AH: Bone morphogenetic proteins:
from basic science to clinical applications. J Bone Joint Surg Am.
83-A(Suppl 1): S1–S6. 2001.PubMed/NCBI
|
|
45.
|
Ulsamer A, Ortuno MJ, Ruiz S, et al: BMP-2
induces Osterix expression through up-regulation of Dlx5 and its
phosphorylation by p38. J Biol Chem. 283:3816–3826. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
46.
|
Nakashima K, Zhou X, Kunkel G, et al: The
novel zinc finger-containing transcription factor osterix is
required for osteoblast differentiation and bone formation. Cell.
108:17–29. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
47.
|
zur Nieden NI, Kempka G and Ahr HJ: In
vitro differentiation of embryonic stem cells into mineralized
osteoblasts. Differentiation. 71:18–27. 2003.PubMed/NCBI
|
|
48.
|
Nohe A, Keating E, Knaus P and Petersen
NO: Signal transduction of bone morphogenetic protein receptors.
Cell Signal. 16:291–299. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
49.
|
Fong S, Chan MK, Fong A, Bowers WJ and
Kelly KJ: Viral vector-induced expression of bone morphogenetic
protein 2 produces inhibition of tumor growth and bone
differentiation of stem cells. Cancer Gene Ther. 17:80–85. 2010.
View Article : Google Scholar : PubMed/NCBI
|