Introduction
Cancer is a leading cause of death worldwide
(1). Current conventional
anticancer treatments incorporate the use of chemotherapy, targeted
therapies and radiation treatment. The utilization of these
treatments is often limited by severe adverse effects, which often
lead to dose reductions and treatment delays. It is therefore
important to search for therapies which can reduce the side effects
of anticancer treatments without altering their efficacy or
increasing toxicity. Such therapies would not only improve the
quality of life of patients with cancer but would also help
patients complete their anti-cancer regimen (2).
LCS101 is a botanical compound developed for the
treatment of patients with solid cancers, based on the principles
of traditional Chinese medicine (TCM). The formula contains
concentrated extracts from the following herbs: Astragalus
membranaceus, Atractylodes macrocephala, Citrus reticulate, Glehnia
littoralis, Ligustrum lucidum, Lycium chinense, Milletia
reticulata, Oldenlandia diffusa, Ophiopogon japonicus, Paeonia
lactiflora, Paeonia obovata, Poriae cocos, Prunella vulgaris
and Scutellaria barbata. Extracts of these compound are
manufactured in accordance with good manufacturing practice (GMP)
conditions, and are imported under license (Zen Herbs Inc.,
Rehovot, Israel), in accordance with the regulations of the Israel
Ministry of Health. All batches of the compound are analyzed and
certified to be free of heavy metals, microbial contamination,
pesticide residues and mycotoxins. The herbal components of LCS101
are considered to be safe for human consumption, and have not been
found to alter the bioavailability or efficacy of anticancer drugs
(3).
In earlier preclinical research, LCS101 was shown
in vitro to inhibit breast cancer cell survival in a
dose-dependent manner on human breast carcinoma cell lines MDA-231,
MDA-453 and T47D. The compound has also shown dose-dependent
inhibition of cell growth (T47D cell line), as well as a
dose-dependent increase in cell apoptosis, as demonstrated by
sub-diploid DNA content (4). In
vivo studies have shown that LCS101 increases peripheral
neutrophil counts in doxo-rubicin-treated mice with breast cancer,
preserving splenic erythrocyte and leucocyte counts (unpublished
data). LCS101 has also been shown to have indirect anticancer
effects, with immunomodulating effects which include the promotion
of T-cell proliferation, NK cell activation and cytokine (TNFα)
activity, as well as the correction of reduced IFN-γ following
exposure to doxorubicin (5). In
clinical studies, LCS101 was shown to reduce anemia and neutropenia
in female patients with locally advanced breast cancer undergoing
anthracycline and taxane-based treatments. In clinical practice and
research LCS101 was found to be both safe and well-tolerated by
patients (6).
The purpose of the present study was to examine the
selectivity of the anticancer effects of LCS101 on cancer cells,
and to investigate its impact on the anticancer activity of the
chemotherapeutic agents doxorubicin and 5-fluorouracil.
Materials and methods
Antibodies and reagents
Primary antibodies: rabbit anti-human PARP-1
(polyclonal, 1:1,000) and rabbit anti-human caspase-3 (monoclonal,
1:1,000) were from Cell Signaling Technologies (Boston, MA, USA).
Mouse anti-human α-tubulin (monoclonal, 1:1,000) was from
Sigma-Aldrich (St. Louis, MO, USA). Goat anti-human actin
(polyclonal, 1:1,000) and mouse anti-human GAPDH (monoclonal,
1:100,000) were from Santa Cruz Biotechnology (Dallas, TX, USA).
Secondary antibodies: peroxidase-conjugated goat anti-rabbit, goat
anti-mouse and rabbit anti-goat IgG (H+L) antibodies were from
Jackson (Baltimore Pike West Grove, PA, USA). Propidium iodide,
MG132 (Z-Leu-Leu-Leu-al), insulin, doxorubicin, 5-fluorouracil,
cholera toxin and hydrocortisone were from Sigma-Aldrich. DMEM
F/12, high glucose DMEM, L-glutamine, donor horse serum, fetal
bovine serum, recombinant human EGF, trypsin and PBS were from
Biological Industries (Beit-Ha-Emek, Israel). Prostate epithelial
growth medium was from Lonza (Walkersville, MD, USA).
Cell culture
Human MCF10A non-tumorigenic breast epithelial cells
were propagated in DMEM F/12 medium supplemented with 5% horse
serum, 2 ng/ml epidermal growth factor, 100 ng/ml cholera toxin, 50
ng/ml hydrocortisone and 10 μg/ml insulin. Human HCT116 colorectal
carcinoma, PC-3 prostate adenocarcinoma, DU-145 prostate carcinoma,
MCF7 breast adenocarcinoma and MDA-MB-231 breast adenocarcinoma
cells were propagated in high glucose DMEM supplemented with 10%
fetal bovine serum and 2 mM L-glutamine. All cell lines were from
American Type Tissue Collection (ATCC, USA) and were authenticated
using STR analysis. Human EP#2 non-tumorigenic normal prostate
epithelial cells were donated by Dr Orit Leshem (7) and propagated in prostate epithelial
growth medium. All cells were propagated in a 37°C humidified
incubator with 5% CO2.
Study compound
A dry extract powder of the formula (Zen Herbs Inc.)
was dissolved in PBS at a concentration of 100 mg/ml. The solution
was then centrifuged at 4300 g for 5 min, with the supernatant
filtered through a 0.45-μM Millex PVDF filter (Merck Millipore,
Tullagreen, Ireland). Solubility was estimated by cryophilization
and weighting of the pellet, and was estimated to be ~50%. For
convenience, the final stock concentration was designated at 100
mg/ml (w/v concentration of crude powder in PBS), enabling the
comparison of the individual herbal components with their variable
solubilities with the formula in its entirety.
XTT viability assay
Breast, prostate and colorectal cells were plated in
triplicate into 96-well plates (MCF10A at 6×103/w; MCF7,
DU-145 and HCT116 at 3×103/w; PC-3 and MDA-MB-231 at
4×103/w; EP#2 - 8×103/w), and allowed to
attach and grow overnight. The medium was replaced with a fresh
treatment-containing medium, and the cells were propagated for an
additional 72 h. Cell viability was determined by XTT cell
proliferation kit (Biological Industries) by replacing the medium
with a fresh medium (in order to prevent interference of treatment
color with XTT signal), and the addition of an XTT for 2–3 h. The
resulting signal was measured by Power Wave × 340-I ELISA reader
(Biotek Instruments, Winooski, VT, USA), with each cell line tested
in at least three independent assays.
FACS analysis
Both cancer and non-tumorigenic cells were plated at
a density of 0.6-1×106/10 cm plate, and then treated the
following day. The cells were collected by trypsinization into
their own medium to prevent loss of dead cells, with each sample
divided into two aliquots. The first aliquot was analyzed for
necrosis following exposure to a free propidium iodide (PI) influx
for 15 min. The second aliquot was fixed with 70% ice-cold ethanol,
stained with PI and used for cell cycle and apoptosis analysis.
Cell sorting was performed on a BD FACSCalibur flow cytometer (BD
Biosciences, San Jose, CA, USA). Cells were resolved on an FL-2
logarithmic scale for necrosis analysis and on an FL-2 linear scale
for apoptosis (cell cycle) analysis, and later analyzed using a
WinMDI 2.9 program (Purdue University Cytometry Laboratories, West
Lafayette, IN, USA).
Immunoblotting
Cancer and non-tumorigenic cells were plated at a
density of 6×105/10 cm plate. On the following day cells
were exposed to LCS101 treatments as indicated in the legends.
After 24–72 h, the cells were collected by scraping, washed with
cold PBS and lysed with RIPA (150 mM NaCl, 1% NP-40, 0.5%
deoxycholic acid, 0.1% SDS, 0.5 M Tris pH 8.0), supplemented with
complete mini protease inhibitor cocktail (Roche Diagnostics GmbH,
Mannheim, Germany). Protein concentration was determined with
Pierce BCA protein assay kit (Thermo Scientific, Rockford, IL,
USA). Samples (50 μg) were resolved by 8% SDS-PAGE, transferred to
Protran BA-83 0.2 μm nitrocellulose membrane (Whatman, Piscataway,
NJ, USA), blocked with 5% skimmed milk and immunoblotted with
appropriate antibodies. The membrane was then washed thrice with
TBST, incubated with corresponding HRP-conjugated secondary
antibodies, probed with EZ-ECL enhanced chemiluminescence detection
kit (Biological Industries) according to the manufacturer’s
instructions and then exposed to Fuji Super RX film (Fujifilm,
Tokyo, Japan).
RT-qPCR
Breast cancer cells were plated at a density of
6×105/10-cm plates and treated the following day with 3
mg/ ml LCS101. After 24 h the cells were collected by scrapping and
washed with cold PBS. Total RNA was isolated using an RNeasy Mini
kit (Qiagen, GmbH, Hilden, Germany). RNA concentration and quality
were determined by optic density measurement (260, and 280 nm). The
quality of the samples was further verified by electrophoresis on
1% agarose gel, stained with ethidium bromide to visualize the 18S
and 28S rRNA bands. Complementary DNA (cDNA) was prepared using
random primers and a High Capacity cDNA Reverse Transcription kit
(AB Applied Biosystems, Foster City, CA, USA). cDNA was subjected
to RT-qPCR on a StepOnePlus Real-Time PCR System using a Power SYBR
Green PCR Master Mix (AB Applied Biosystems). The RT-qPCR was
performed according to the manufacturer’s instructions using the
following primer sets: PARP-1 forward, 5′-AAGCTCT
ATCGAGTCGAGTACG-3′; reverse, 5-GAAGCTCAGAGA ACCCATCC-3. GADPH
forward, 5-TGGACCTCATGG CCCACA-3; reverse,
5-TCAAGGGGTCTACATGGCAA-3. The expression levels of PARP-1 from
triplicate reactions was determined by normalization to GAPDH
according to the manufacturer’s instructions.
Statistical methods
The mean ± standard deviations were calculated in
each experiment, which were performed in triplicate. The data were
collated and analyzed in a Microsoft Excel 2007 program.
Results
LCS101 selectively induces cell death in
cancer cells
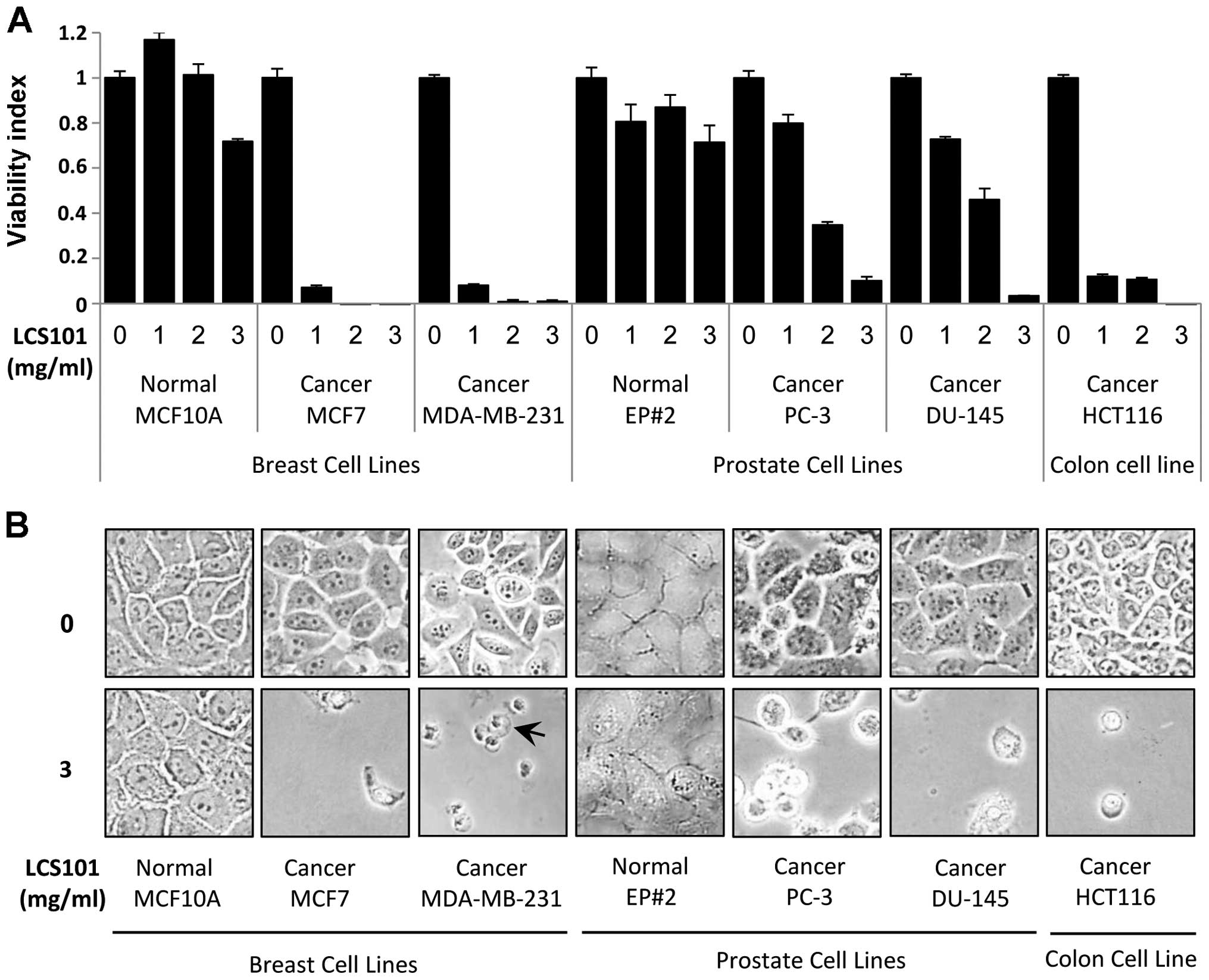
Initially we treated the different tumor cell lines
and non-tumorigenic human cell lines with the LCS101 compound.
Exposure of the cultured tumor cells to the compound led to a
dose-dependent reduction in cell viability, with cell death
observed in >90% of cells, as measured by XTT assay. This
phenomenon was observed at concentrations of 1 mg/ml for breast and
colon cancer cell lines, and at 3 mg/ml for prostate cancer cell
lines. At the same time, the non-tumorigenic human epithelial cell
lines MCF10A (breast) and EP#2 (prostate) demonstrated a reduction
in viability of <30% following exposure to the botanical
compound (Fig. 1A). The
non-tumorigenic human luminal breast cell line HB-2 also displayed
an attenuated response to LCS101 exposure (not shown). Light
microscopy showed increased cell death of all cancer cells
following exposure to LCS101 treatment, with some of the treated
cancer cells demonstrating swelled morphology indicative of a
necrotic process. In contrast, non-tumorigenic human epithelial
cells exhibited normal density and morphology following exposure to
the compound (Fig. 1B).
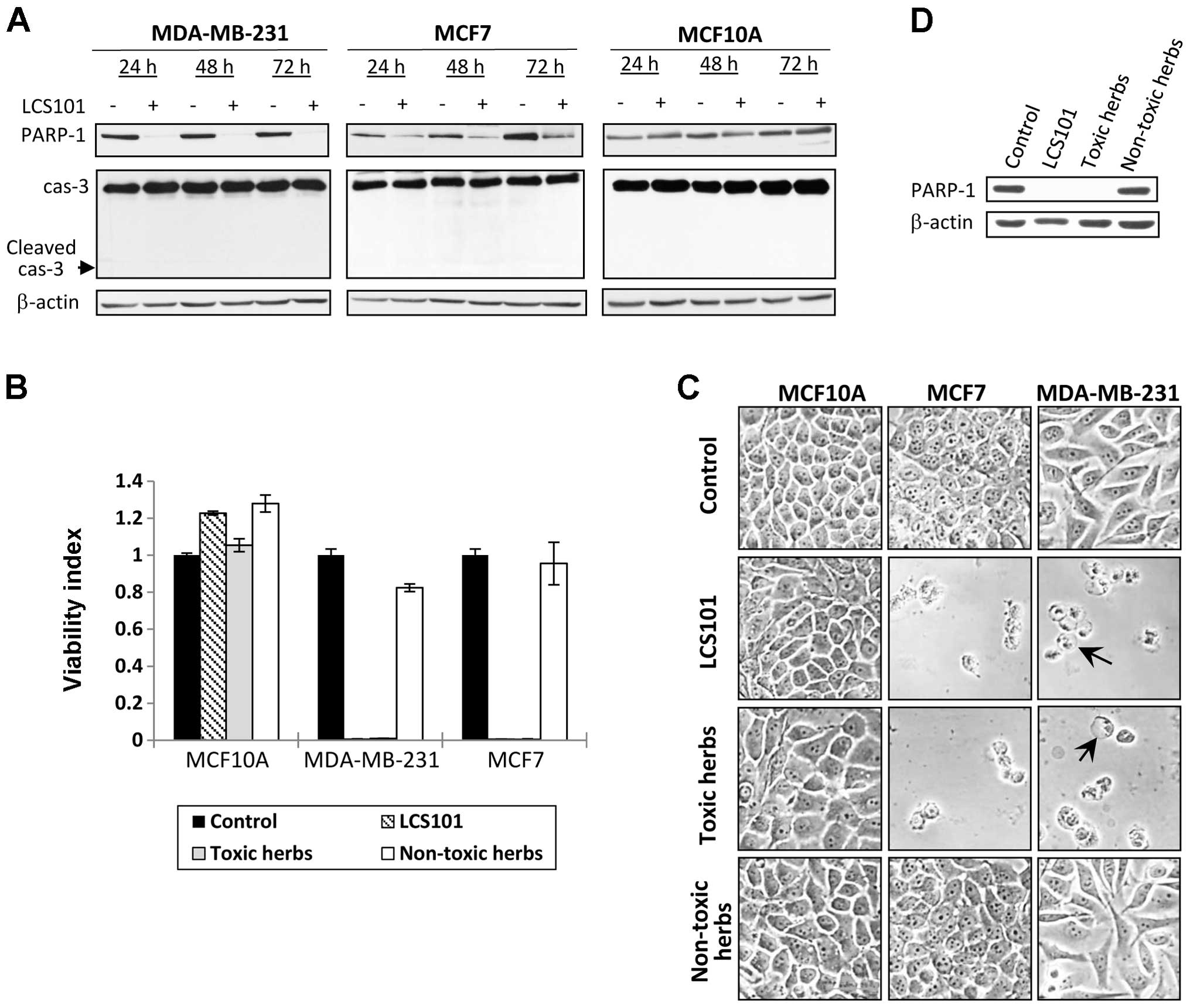
LCS101 reduces PARP-1 expression in
breast cancer cell lines
To address the mechanisms underlying the anticancer
activity of LCS101, MDA-MB-231, MCF7 and MCF10A breast cells were
treated with LCS101, and expression of apoptosis markers caspase-3
(cas-3) and poly(ADP-ribose) polymerase 1 (PARP-1) were examined.
In classic apoptosis both of these proteins undergo cleavage, which
is considered as hallmark of apoptosis. Surprisingly, no cleavage
of caspase-3 and PARP-1 was detected, though a significant
reduction in the level of PARP-1 protein was observed in both of
these cancer cell lines. In contrast, the non-tumorigenic human
epithelial breast MCF10A cells exposed to LCS101 showed no
reduction in PARP-1 levels (Fig.
2A).
LCS101-induced toxicity correlates with
reduced PARP-1 levels
In order to better understand the cytotoxic effects
of the LCS101 formulature, 6 of the 14 herbal components were
isolated and selected: Ligustrum lucidum, Milletia reticulata,
Paeonia lactiflora, Paeonia obovata, Prunella vulgaris and
Scutellaria barbata. These herbs were found to display
greater toxic effects towards MDA-MB-231 cancer cells, without any
harmful effects on the non-tumorigenic human epithelial breast
MCF10A cells (not shown). Following these findings, the LCS101
formula was divided into the ‘toxic formula’ (the above-mentioned 6
components) and the ‘non-toxic formula’ (the remaining 8
components). Exposure to the toxic formula resulted in a
significant increase in cell death in MCF7 and MDA-MB-231 breast
cancer lines, compared to no cytotoxic effect in the non-toxic
formula (Fig. 2B and C). Following
exposure to the toxic formula, cell swelling was observed in
treated MDA-MB-231 cells (Fig. 2C,
arrows). Both the toxic and non-toxic formulas had no effect on the
non-tumorigenic human epithelial breast MCF10A cells (Fig. 2B and C). Finally, while the toxic
formula reduced PARP-1 levels in MDA-MB-231 cells, no such effect
was observed with the non-toxic components of the formula (Fig. 2D).
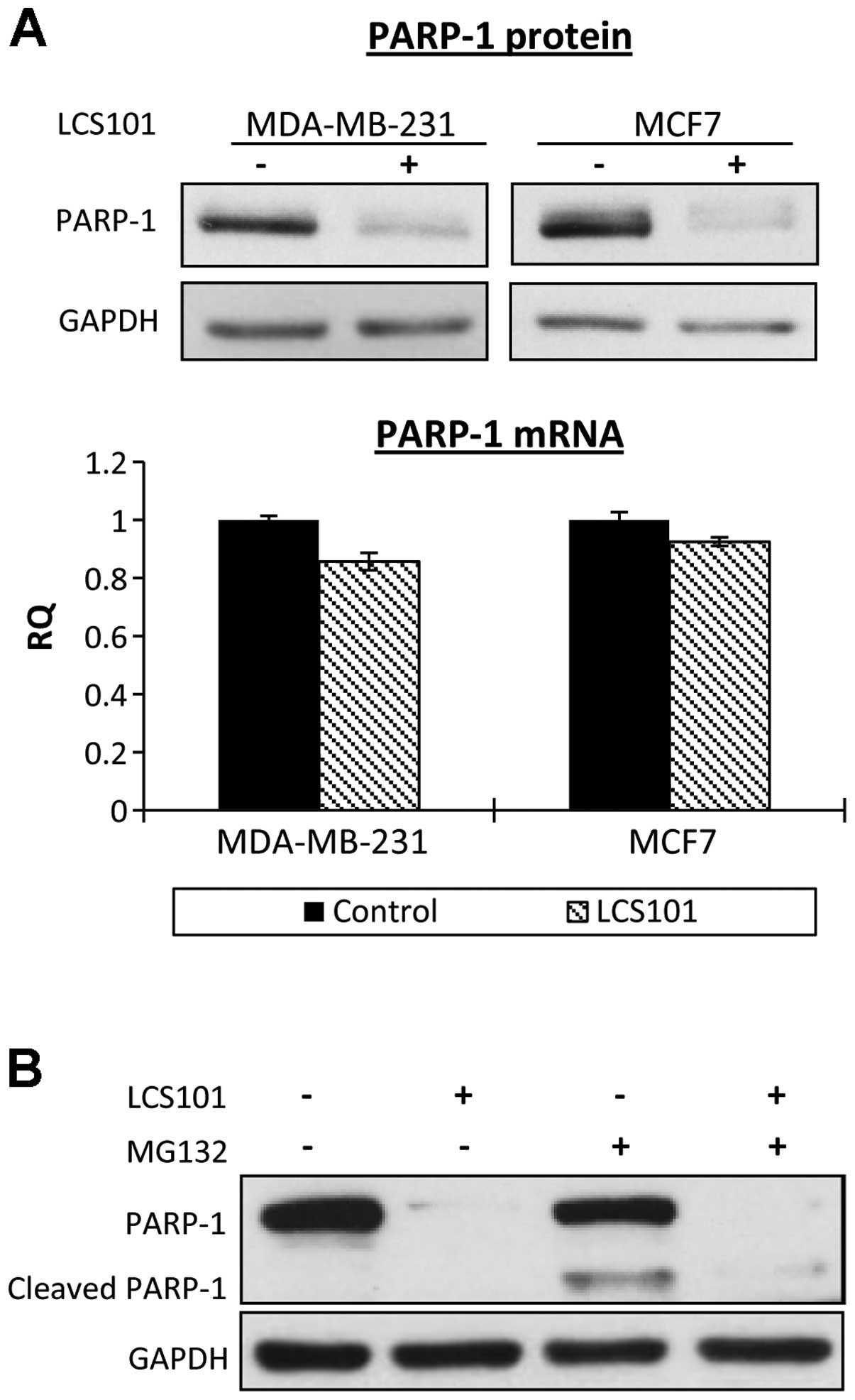
LCS101-induced PARP-1 reduction is
unrelated to mRNA or proteasomal degradation
To further address the mechanism of PARP-1 protein
reduction, we tested PARP-1 mRNA level in LCS101-treated cells
using RT-PCR. PARP-1 mRNA levels in exposed MCF7 and MDA-MB-231
cells were similar to those found in controls (Fig. 3A). We then examined PARP-1 protein
degradation using the proteasome inhibitor MG132 on MDA-MB-231
cells. After 24 h of treatment with MG132, which was necessary for
LCS101-induced PARP-1 reduction, massive cell death and complete
PARP-1 cleavage, characteristic of apoptosis, were observed.
However, the examination of MCF7 cells, which apparently were more
resistant to MG132-induced apoptosis, found that the MG132 failed
to prevent PARP-1 elimination following exposure to the botanical
formula (Fig. 3B).
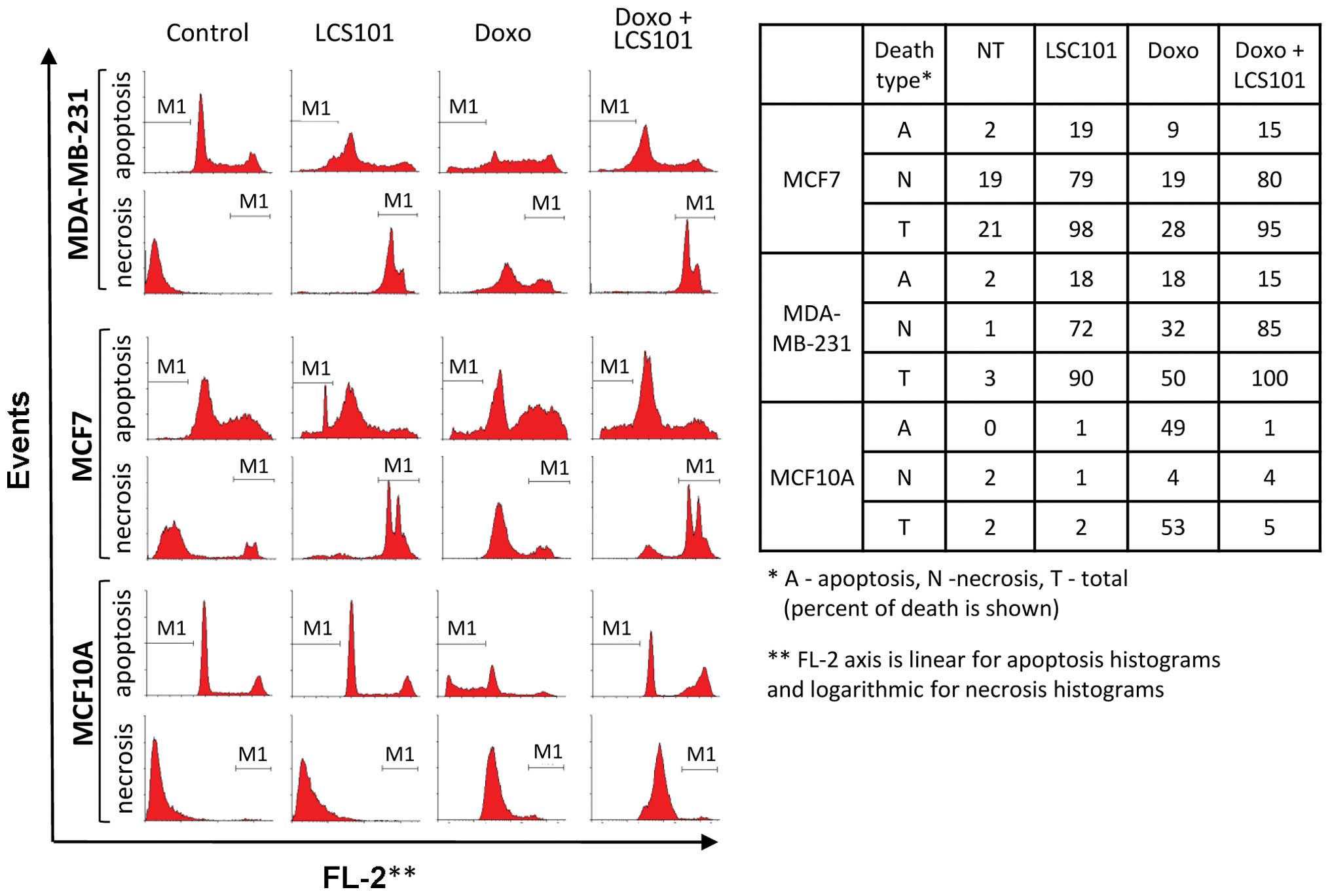
LCS101-induced cell death exhibits
necrosis-like features
LCS101-treated cells were analyzed using FACS in
order to distinguish between apoptotic and necrotic features. For
this purpose, MCF7, MDA-MB-231 and MCF10A cells were treated with
LCS101 for 72 h, with each sample divided into two aliquots for
FACS analysis. One aliquot was fixed and stained with PI to assess
the sub-G1 population, which contained cells with degraded DNA,
characteristic of apoptotic death. The second aliquot was used to
evaluate the percentage of cells with ruptured membranes, typical
of necrotic death, using free PI influx by live unfixed cells.
Ruptured membrane of necrotic cells allows free PI uptake which
causes the necrotic population to appear very bright on logarithmic
PI scale. Following exposure to LCS101, the pattern of the vast
majority of the cancer cell lines MCF7 and MDA-MB-231 moved far to
the right upon free PI uptake, indicating that most of the cells
possessed a necrosis-like ruptured membrane (Fig. 4). Morphologically, a number of the
affected cells exhibited significant swelling, typical of a
necrotic process as well (Fig.
1B). At the same time, only 20% of the cells exhibited sub-G1
DNA content, a typical indicator of apoptosis (Fig. 4). LCS101 did not induce cell death
in the non-tumorigenic human epithelial breast MCF10A cells
(Fig. 4).
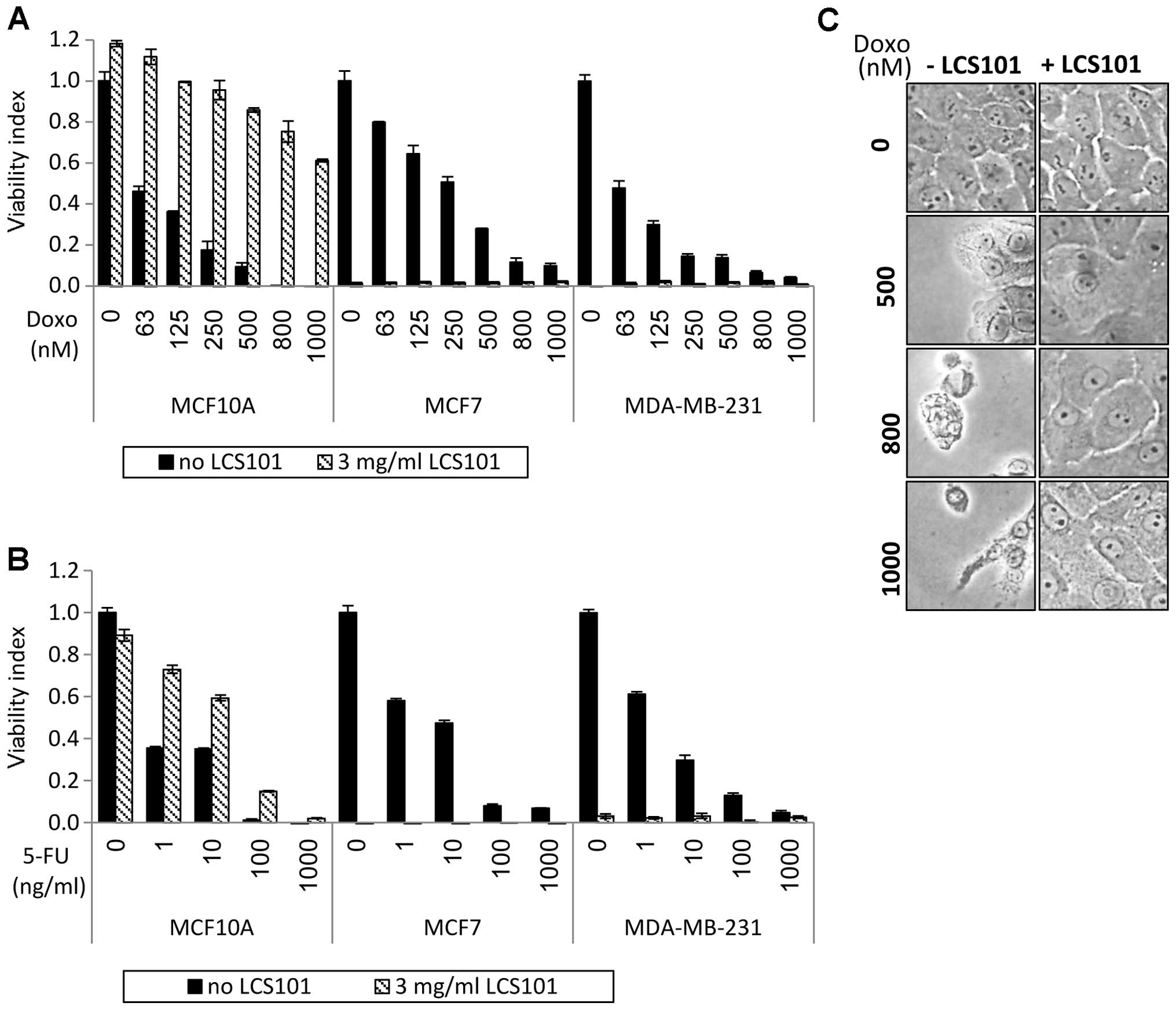
LCS101 selectively protects
non-tumorigenic cells from doxorubicin and 5-FU
Chemotherapy-induced cell death was observed in all
three breast cell lines (MCF-10A, MCF-7 and MDA-MB-231) following
exposure to the chemotherapy agent doxorubicin (Fig. 5A). When introduced at a
concentration of 3 mg/ml, LCS101 augmented the tumoricidal effects
of the chemotherapy in MCF7 and MDA-MB-231 cells. In contrast, the
addition of the botanical formula to the non-tumorigenic human
epithelial breast MCF10A cells greatly reduced cell death (Fig. 5A and C). A similar selective effect
was seen in 5-FU-treated cell lines, with LCS101 increasing the
tumoricidal effect of the chemotherapy agent on MCF7 and MDA-MB-231
cells, while reducing cell death in the non-tumorigenic human
epithelial breast MCF10A cells (Fig.
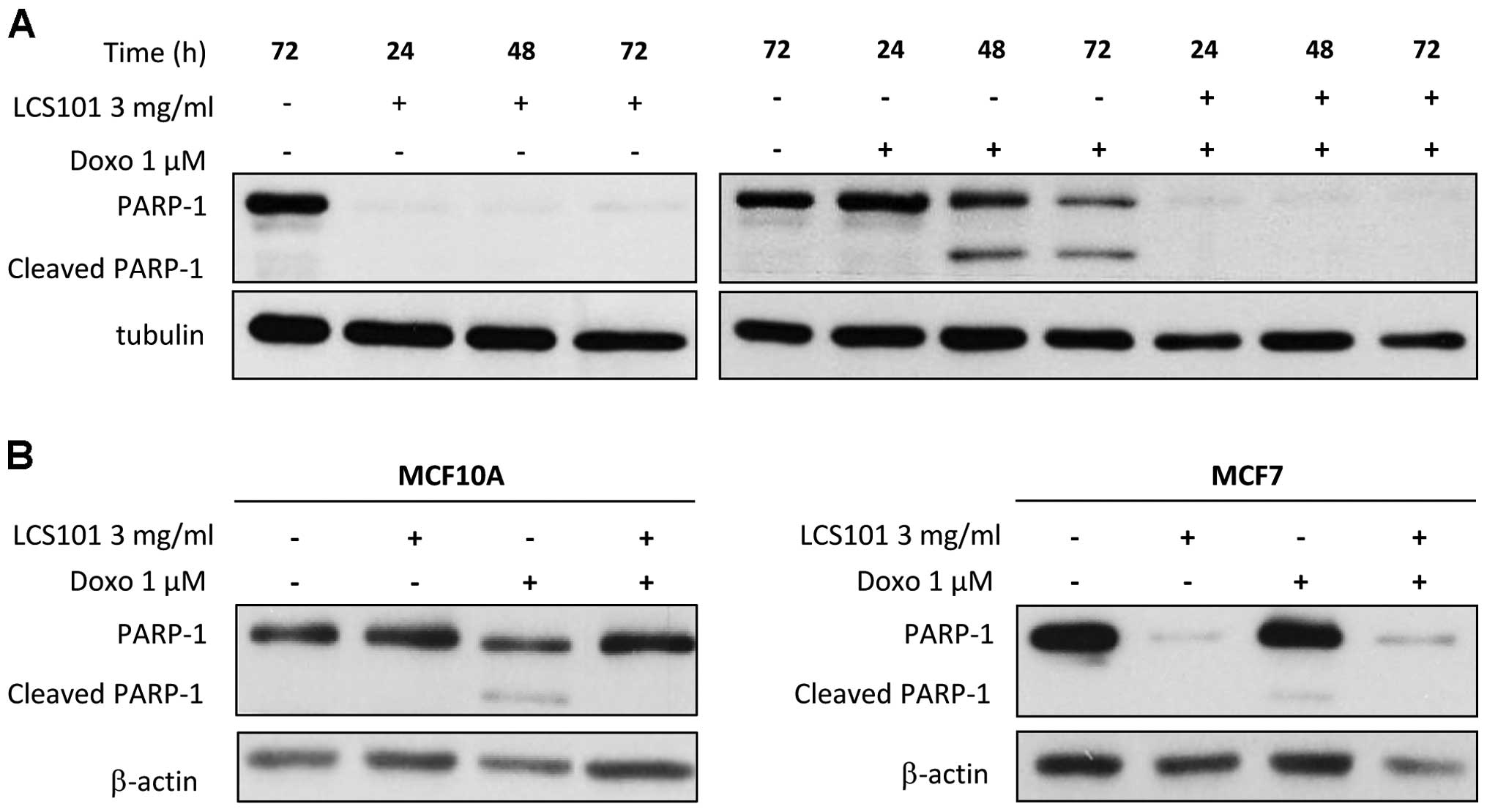
5B). In addition to the above findings, MDA-MB-231 cells
treated with doxorubicin demonstrated PARP-1 cleavage typical of
apoptosis, at 48 and at 72 h (Fig.
6A). A less prominent but still clearly detectable PARP-1
cleavage was observed in MCF10A and MCF7 cells at 72-h treatment
with doxorubicin (Fig. 6B). The
addition of LCS101 to doxo-rubicin-treated MDA-MB-231 and MCF7
cells, however, led to the disappearance of PARP-1. In
non-tumorigenic human epithelial breast MCF10A cells, LCS101
prevented doxorubicin-induced PARP-1 cleavage altogether,
supporting our previous findings showing protective effect of
LCS101 in normal cells.
In order to confirm the above findings, we performed
FACS analysis of MDA-MB-231, MCF7 and MCF10A cells, which were
treated with both doxorubicin and LCS101 (Fig. 4). Our findings were consistent with
those above regarding PARP-1 cleavage, with typical apoptosis
observed in all three cell lines treated with doxorubicin alone and
a clearly demarked sub-G1 apoptotic population. At the same time,
the addition of LCS101 reduced apoptosis in non-tumorigenic human
epithelial breast MCF10A cells, while increasing cell death in the
two cancer cell lines (Fig. 4). In
cancer cells treated with both LCS101 and doxorubicin, cell death
exhibited necrotic features, as described above. This indicates
that the increase in doxorubicin-induced cell death in cancer cell
lines treated with LCS101 results from a necrosis-like process. At
the same time, LCS101 offers a protective effect on non-tumorigenic
cells exposed to the chemotherapy agent.
Discussion
The treatment of patients with cancer presents a
number of challenges to oncologists. Anticancer therapies, whether
chemotherapy or personalized and targeted biological agents, are
often only partially effective and are invariably accompanied by
debilitating adverse effects which can compromise the treatment
regimen. Many tumors are aggressive and resistant to conventional
treatments, which themselves can impair the body’s immunity and
increase susceptibility to infection. The use of additional
chemotherapy agents to established regimens can further increase
tumor response, though this positive effect is offset by increased
toxicity (8).
Botanical medicine has been in use for thousands of
years, with pre-clinical and clinical research demonstrating a
number of positive effects of many of the herbal compounds being
used for the treatment of cancer, with reduction of disease
activity and treatment-related symptoms. In the present study we
found the botanical compound LCS101 demonstrated a dose-dependent
induction of cell death in breast, prostate and colorectal cancer
cells. At the same time, LCS101 exhibited no cytotoxic effects on
non-tumorigenic human epithelial breast MCF10A cells. The cytotoxic
effects of many of the individual LCS101 components have been
reported elsewhere in the scientific literature (Table I). Little is known, however, about
the selectivity of these effects, and the potential for negative
effects on non-tumorigenic cells has limited their use in clinical
practice. In light of this, the findings of the present study may
have significant implications regarding the incorporation of
botanical products into standard anticancer care.
 | Table IAnticancer effects of LCS101 herbal
components. |
Table I
Anticancer effects of LCS101 herbal
components.
| Herbal component | Anticancer
effects |
|---|
| Astragalus
membranaceus | Suppression of C6
glioma cells, in vitro and in vivo (9) |
| Atractylodes
macrocephala | Mediation of reactive
oxygen species apoptosis in human leukemia cells (10) |
| Citrus
reticulate | Induction of
apoptosis in SNU-C4 human colon cancer cells (11)
Induction of apoptosis in human gastric cancer cells (cas-3
pathway) (12) |
| Ligustrum
lucidum | Induction of human
glioma cell death through regulation of Akt/mTOR pathway in
vitro and reduction of glioma tumor growth in U87MG xenograph
mouse model (13) |
| Oldenlandia
diffusa | Augmentation of
oxidative burst in macrophages and inhibited tumor growth (14)
Selective anticancer in vitro effects in B16-F10 mice lung
cancer and Renca renal carcinoma models (15) |
| Paeonia
lactiflora | Inhibition of bladder
cancer growth in a rat model involving phosphorylation of Chk2,
in vitro and in vivo (16) |
| Prunella
vulgaris | Chemoprevention of
non-small cell lung cancer (NSCLC) via promotion of apoptosis and
regulation of the cell cycle (17)
Suppression of PMA-induced tumor cell invasion and metastasis via
inhibition of NF-κB-dependent MMP-9 expression (18) |
| Scutellaria
barbata | Induction of
oxidative stress damage with redistribution of metabolic fluxes in
breast cancer cells (19)
Selective cytotoxic activity on breast cancer cells (20)
Augmentation of oxidative burst in macrophages and inhibited tumor
growth (14)
Modulation of apoptosis and cell survival in murine and human
prostate cancer cells and tumor development in TRAMP mice (21) |
LCS101-induced cancer cell death was manifest as
both rupturing of the cell membrane and, in some cases, cell
swelling. Both phenomena are demonstrative of a necrotic pathway.
The absence of caspase-3 cleavage and lack of DNA degradation
despite the massive cell death observed provides further evidence
supporting the understanding that this was a manifestation of
necrotic cell death. At the same time, however, LCS101-induced
cancer cell death was associated with a drastic drop in PARP-1
protein levels, a phenomenon not reported elsewhere in the
literature, to the best of our knowledge. This was also observed in
the correlation between PARP-1 reduction and cytotoxic effects of
the toxic and non-toxic sub-formulas of the botanical compounds.
The reduction in PARP-1 levels was not related to either reduction
in mRNA expression or proteasomal degradation. Further research is
needed in order to understand the implications and mechanisms of
these effects on PARP-1 pathways.
We also evaluated the effects of LCS101 on cells
treated with the chemotherapy agents doxorubicin and 5-FU. As
expected, these agents led to significantly reduced survival in all
cell lines. However, while cell death was significantly increased
in breast cancer cell lines MCF-7 and MDA-MB-231 following the
addition of the botanical compound, non-tumorigenic human
epithelial breast MCF-10A cells were protected from
doxorubicin-induced apoptosis. These findings further support the
results observed in earlier clinical trials, in which LCS101 was
found to be safe and non-toxic when administered to patients with
cancer.
TCM employs a holistic, personalized approach to the
treatment of disease. The use of herbal formulas which combine a
number of herbal products, each with its own effects on the body
acting in harmony with each another, enhances the therapeutic
process and promotes well-being. We believe that for this reason,
the toxic components need to be supplemented by the non-toxic
components in order to promote healing. Many of the LCS101
components have indeed been shown to have anticancer and
immunomodulatory affects, as well as demonstrating protective
effects against chemotherapy and reactive oxygen species (Table I).
In conclusion, our findings strongly support our
previous data suggesting that LCS101 has a cytotoxic effect on
cancer cell lines. Furthermore, we show that LCS101 cytotoxicity is
selective, with no deleterious effects on non-tumorigenic
epithelial cells. LCS101-induced cancer cell death resembles
necrosis, though further research is needed in order to better
understand this mechanism. In addition, LCS101 provides a
selectively protective effect on non-tumorigenic epithelial cells
exposed to the chemotherapy agents doxorubicin and 5-FU, while at
the same time augmenting their cytotoxic effects on cancer cell
lines. Further research is needed to support these findings, as
well as understand the clinical implications of this particular
botanical compound on anti-cancer therapy.
Acknowledgments
Conflict of interest statement: Dr Yair Maimon is a
shareholder of LifeBiotics Ltd.
References
|
1
|
World Health Organization fact sheet No.
297. http://www.who.int/mediacentre/factsheets/fs297/en/index.html:.
Accessed December 28, 2013
|
|
2
|
Liu SH and Cheng YC: Old formula, new Rx:
the journey of PHY906 as cancer adjuvant therapy. J Ethnopharmacol.
140:614–623. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Samuels N, Maimon Y and Zisk-Rony RY:
Effect of the botanical compound LCS101 on chemotherapy-induced
symptoms in patients with breast cancer: a case series report.
Integr Med Insights. 8:1–8. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Maimon Y, Karaush V, Yaal-Hahoshen N,
Ben-Yosef R, Ron I, Vexler A and Lev-Ari S: Effect of Chinese
herbal therapy on breast cancer adenocarcinoma cell lines. J Int
Med Res. 38:2033–2039. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rachmut IH, Samuels N, Melnick SJ,
Ramachandran C, Sharabi Y, Pavlovsky A, Maimon Y and Shoham J:
Immunomodulatory effects of the botanical compound LCS101:
implications for cancer treatment. Onco Targets Ther. 6:437–445.
2013.PubMed/NCBI
|
|
6
|
Yaal-Hahoshen N, Maimon Y,
Siegelmann-Danieli N, Lev-Ari S, Ron IG, Sperber F, Samuels N,
Shoham J and Merimsky O: A prospective, controlled study of the
botanical compound mixture LCS101 for chemotherapy-induced
hematological complications in breast cancer. Oncologist.
16:1197–1202. 2011. View Article : Google Scholar
|
|
7
|
Leshem O, Madar S, Kogan-Sakin I, Kamer I,
Goldstein I, Brosh R, Cohen Y, Jacob-Hirsch J, Ehrlich M,
Ben-Sasson S, Goldfinger N, Loewenthal R, Gazit E, Rotter V and
Berger R: TMPRSS2/ERG promotes epithelial to mesenchymal transition
through the ZEB1/ZEB2 axis in a prostate cancer model. PLoS One.
6:e216502011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Butters DJ, Ghersi D, Wilcken N, Kirk SJ
and Mallon PT: Addition of drug/s to a chemotherapy regimen for
metastatic breast cancer. Cochrane Database Syst Rev. 2010.CD003368
View Article : Google Scholar
|
|
9
|
Sun JY, Yang H, Miao S, Li JP, Wang SW,
Zhu MZ, Xie YH, Wang JB, Liu Z and Yang Q: Suppressive effects of
swainsonine on C6 glioma cell in vitro and in vivo. Phytomedicine.
16:1070–1074. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang HL, Chen CC, Yeh CY and Huang RL:
Reactive oxygen species mediation of baizhu-induced apoptosis in
human leukemia cells. J Ethnopharmacol. 97:21–29. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kang SA, Park HJ, Kim MJ, Lee SY, Han SW
and Leem KH: Citri Reticulatae Viride Pericarpium extract
induced apoptosis in SNU-C4, human colon cancer cells. J
Ethnopharmacol. 97:231–235. 2005. View Article : Google Scholar
|
|
12
|
Kim MJ, Park HJ, Hong MS, Park HJ, Kim MS,
Leem KH, Kim JB, Kim YJ and Kim HK: Citrus Reticulata blanco
induces apoptosis in human gastric cancer cells SNU-668. Nutr
Cancer. 51:78–82. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jeong JC, Kim JW, Kwon CH, Kim TH and Kim
YK: Fructus ligustri lucidi extracts induce human glioma cell death
through regulation of Akt/mTOR pathway in vitro and reduce glioma
tumor growth in U87MG xenograft mouse model. Phytother Res.
25:429–434. 2011. View
Article : Google Scholar
|
|
14
|
Wong BY, Lau BH, Jia TY and Wan CP:
Oldenlandia diffusa and Scutellaria barbata augment macrophage
oxidative burst and inhibit tumor growth. Cancer Biother
Radiopharm. 11:51–56. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gupta S, Zhang D, Yi J and Shao J:
Anticancer activities of Oldenlandia diffusa. J Herb Pharmacother.
4:21–33. 2004. View Article : Google Scholar
|
|
16
|
Ou TT, Wu CH, Hsu JD, Chyau CC, Lee HJ and
Wang CJ: Paeonia lactiflora Pall inhibits bladder cancer
growth involving phosphorylation of Chk2 in vitro and in vivo. J
Ethnopharmacol. 135:162–172. 2011. View Article : Google Scholar
|
|
17
|
Feng L, Jia X, Zhu M, Chen Y and Shi F:
Chemoprevention by Prunella vulgaris L. extract of non-small
cell lung cancer via promoting apoptosis and regulating the cell
cycle. Asian Pac J Cancer Prev. 11:1355–1358. 2010.
|
|
18
|
Choi JH, Han EH, Hwang YP, Choi JM, Choi
CY, Chung YC, Seo JK and Jeong HG: Suppression of PMA-induced tumor
cell invasion and metastasis by aqueous extract isolated from
Prunella vulgaris via the inhibition of NF-kappaB-dependent
MMP-9 expression. Food Chem Toxicol. 48:564–571. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Klawitter J, Klawitter J, Gurshtein J,
Corby K, Fong S, Tagliaferri M, Quattrochi L, Cohen I, Shtivelman E
and Christians U: Bezielle (BZL101)-induced oxidative stress damage
followed by redistribution of metabolic fluxes in breast cancer
cells: a combined proteomic and metabolomic study. Int J Cancer.
129:2945–2957. 2011. View Article : Google Scholar
|
|
20
|
Fong S, Shoemaker M, Cadaoas J, Lo A, Liao
W, Tagliaferri M, Cohen I and Shtivelman E: Molecular mechanisms
underlying selective cytotoxic activity of BZL101, an extract of
Scutellaria barbata, towards breast cancer cells. Cancer Biol Ther.
7:577–586. 2008. View Article : Google Scholar
|
|
21
|
Wong BY, Nguyen DL, Lin T, Wong HH,
Cavalcante A, Greenberg NM, Hausted RP and Zheng J: Chinese
medicinal herb Scutellaria barbata modulates apoptosis and cell
survival in murine and human prostate cancer cells and tumor
development in TRAMP mice. Eur J Cancer Prev. 18:331–341. 2009.
View Article : Google Scholar : PubMed/NCBI
|