Introduction
With the recent advances in chemotherapy, the
prognosis for patients with metastatic colorectal cancer (CRC) has
been considerably improved (1).
However, CRC is still one of the most common malignancies worldwide
and is a major cause of cancer-related deaths (2). Prognosis of this disease depends on
tumor stage. The 5-year overall survival rates range from 93% for
stage I patients to 8% for stage I patients (3). Thus, early detection of CRC is
important to reduce the mortality of this disease.
Representative tumor markers for CRC,
carcinoembryonic antigen (CEA) and carbohydrate antigen (CA) 19-9
are not reliable in early detection of CRC. A previous study showed
that serum p53 antibody test was a more sensitive tumor marker to
detect early stage of CRC rather than CEA and CA19-9 (4). However, its feasibility as a
screening test remained to be explored. Fecal occult blood test
(FOBT) is employed as a screening test to detect CRC, but only
50–60% of early CRC is positive for FOBT. Currently, CRC is
primarily diagnosed through colonoscopy. However, this procedure is
invasive and expensive, and additionally requires bowel
preparation, and may be associated with medical complications.
Therefore, an optimal screening test that is easy to perform,
noninvasive, acceptable, and can select those who have neoplastic
lesions is required to improve the detection of CRC.
MicroRNAs (miRNAs) are newly discovered class of 22
nucleotide noncoding RNA molecules that regulate the translation
and stability of specific target mRNAs through base-printing to
partially complementary sites on target mRNAs that usually reside
within the 3′ untranslated regions (5). miRNAs have been considered to play an
important role in the multistep processes of carcinogenesis either
by oncogenic or tumor suppressive function (6). The association with tumorigenesis
indicates their potential as diagnostic markers (7).
Circulating miRNAs are stably detected in plasma or
serum and serve as biomarkers for several diseases, making them
potentially useful noninvasive markers for early diagnosis or in
monitoring of cancer progression (8,9).
Previous studies revealed that miRNAs were potential diagnostic or
prognostic tools for CRC. For example, miR-92 or miR-21 is
significantly elevated in plasma of CRC patients, and can be a
potential noninvasive molecular marker for CRC detection (10,11).
We have recently found that serum miR-199a-3p is significantly
higher in CRC patients than non-cancer patients, suggesting that
circulating miR-199a-3p could be a biomarker for CRC (12). In this study, we explored other
novel circulating miRNAs in CRC patients and evaluated its
feasibility as a noninvasive diagnostic test for efficient
detection of CRC.
Materials and methods
Patients and samples
Informed consent was obtained from CRC patients and
non-cancer patients for the use of their blood samples. Venous
blood samples were collected from the CRC patients (n=114) and
non-cancer patients (n=32) from April 2011 to June 2013. In 30 of
the CRC patients, blood samples were taken before and 7th day after
surgery. No cancer patients received chemotherapy or radiotherapy
before blood sampling. The blood samples were obtained from Osaka
University, and its associated hospitals. Whole blood was
collected, centrifuged at 1,000 rpm at 4°C for 15 min. The
supernatant fluids were centrifuged at 15,000 rpm at 4°C for 10
min. The supernatant fluids were stored at −80°C until RNA
extraction. This study was conducted under the supervision of the
ethics board of Osaka University Hospital.
RNA extraction
Small RNA was enriched from all serum samples using
the mirVana PARIS RNA isolation kit (Ambion, Austin, TX, USA),
following the manufacturer's instructions. Briefly, 400 μl of serum
was thawed on ice and centrifuged at 15,000 rpm for 15 min to
remove cell debris. Next, 300 μl of supernatant was lysed with an
equal volume of 2X denaturing solution. For normalization of
sample-to-sample variation during the RNA isolation procedures, 20
fmol of synthetic C. elegans miRNA cel-miR-39 was added to
each denatured sample. Small RNAs were then enriched and purified
following manufacturer's protocol. The concentration of all RNA
samples were quantified by Nano Drop ND-1000 (NanoDrop
Technologies, Wilmington, DE, USA).
miRNA microarray analysis
miRNA microarray experiments were carried out by
using Agilent human miRNA microarray cataloged in the Sanger
database version 12.0 (design ID 021827). About ten nanogram
aliquots of total RNA with cel-miR-39 was used for making miRNA
probes according to the Agilent protocol (version 2.3). Microarrays
were performed for paired pre- and post-operative serum from 10 CRC
patients. Briefly, total RNA was dephosphorylated with calf
intestine alkaline phosphatase, denatured with dimethyl sulfoxide,
and labeled with pCp-Cy3 using T4 RNA ligase using the miRNA
Labeling Reagent and Hybridization kit. Probes were hybridized at
55°C for 20 h with rotation. Then the slides were washed by Gene
Expression Wash Buffer 1 at room temperature for 5 min and by Gene
Expression Wash Buffer 2 at 37°C for 5 min. After hybridization and
washing, the slides were scanned using an Agilent scanner (G2505C).
Images were extracted using Agilent Feature Extraction software
(version 10.7.3.1) and Agilent GeneSpring GX software (version
10.0.2). Differences in miRNA expression between the 10 pairs was
determined if the fold change of cel-miR-39 normalized expression
values was >2.0 and the P-value was <0.05 using paired t-test
for further analysis. The microarray raw data are available in Gene
Expression Omnibus (GEO;http://www.ncbi.nlm.nih.gov/geo) under accession
number GSE55139.
qRT-PCR
For microRNA based RT-PCR assays, 2.5 μl of enriched
small RNAs from serum samples were reverse transcribed using the
TaqMan MicroRNA Reverse Transcription kit (Applied Biosystems, San
Diego, CA, USA) according to manufacturer's instructions in a total
reaction volume of 7.5 μl. A 1:20 dilution of RT products was used
as template for the PCR stage. PCR reaction was performed in
triplicates using TaqMan 2X Universal PCR Master Mix according to
the manufacturer's instructions. Each reaction was performed in a
final column of 20 μl containing 1.33 μl of the cDNA and 1 μl of
Taqman miRNA assay mix. The amplification profile was: denaturation
at 95°C for 10 min, followed by 40 cycles of 95°C for 15 sec and
60°C for 60 sec. Each sample was run in triplicates for analysis.
The cycle threshold (Ct) is defined as the number of cycles
required for the fluorescent signal to cross the threshold in qPCR.
The 7900 Sequence Detection System 2.3 (Applied Biosystems)
software was used to compute the relative change in RNA expression
by the 2−ΔΔCt method with 95% confidence intervals.
Primers
The miRNA-specific primer sequences, including
miRNA-103, miR-720, cel-miR-39, and RNU6B were designed based on
the miRNA sequences obtained from the miRBase. The primer sequences
are: hsa-miR-103, 5′-AGCAGCAUUGUACAGGGCUAUGA-3′; hsa-miR-720,
5′-UCUCGCUGGGGCCUCCA-3′; hsa-miR-21, 5′-UAGCUUAUCAGACUGAUGUUGA-3′;
cel-miR-39, 5′-UCACCGGGUGUAAAUCAGCUUG-3′; RNU6B,
5′-CGCAAGGATGACACGCAAATTCGTGAAGCGTTCCATATTTTT-3′.
Statistical analysis
The significance of serum miRNA level was determined
by Mann-Whitney test, Wilcoxon test and χ2 test where
appropriate using the Graph Pad Prism 6 (San Diego, CA, USA). The
sensitivity, specificity, and accuracy were calculated according to
the standard formulas. Receiver operating characteristic (ROC)
curves and area under the ROC curve (AUC) were established for
discriminating patients with CRC. P-value of <0.05 was
considered statistically significant.
Results
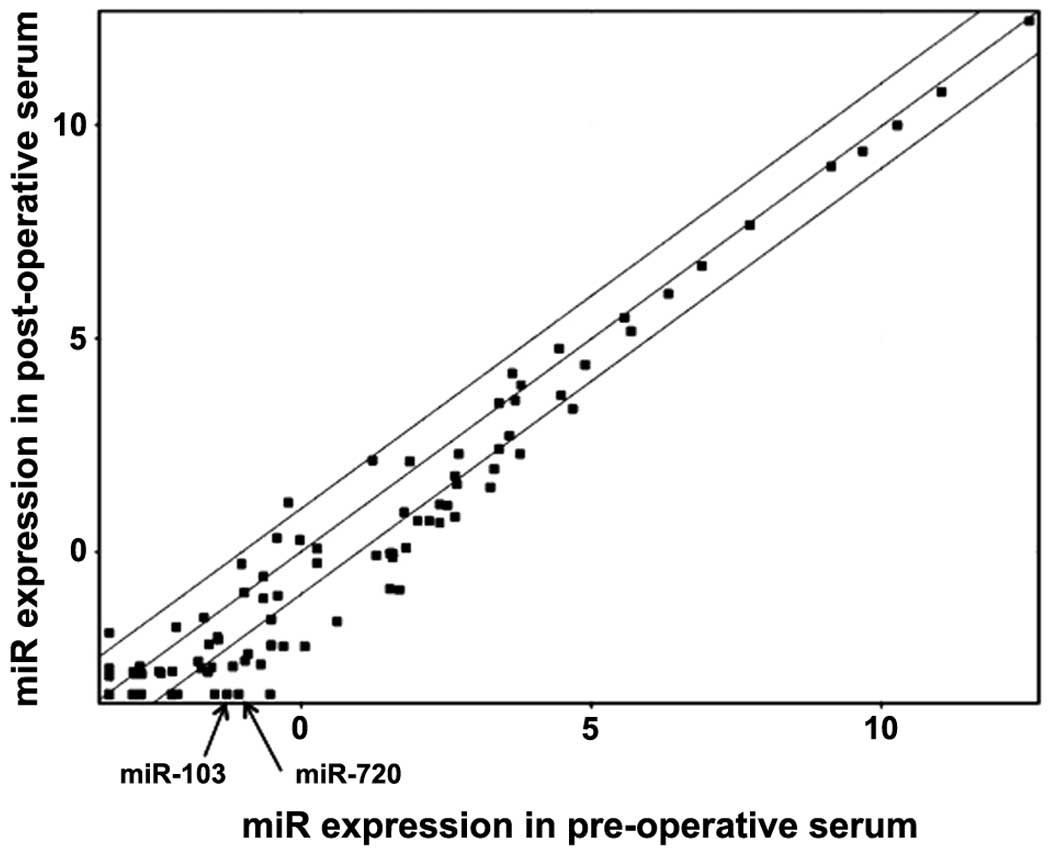
Results of miRNA microarray analysis
In comparison of CRC patient serum between pre- and
post-operation (n=10; 4 stage II CRCs and 6 stage III CRCs) by the
miRNA array, we identified miRNAs, most of which showed a decrease
in the post-operative serum (Fig.
1). Among them, we focused on the two miRNAs, miR-103 and
miR-720 whose expression showed a large decrease after surgery with
significant P-value (4.09-fold and 4.68-fold decrease; P=0.037 and
P=0.037, respectively, Fig.
1).
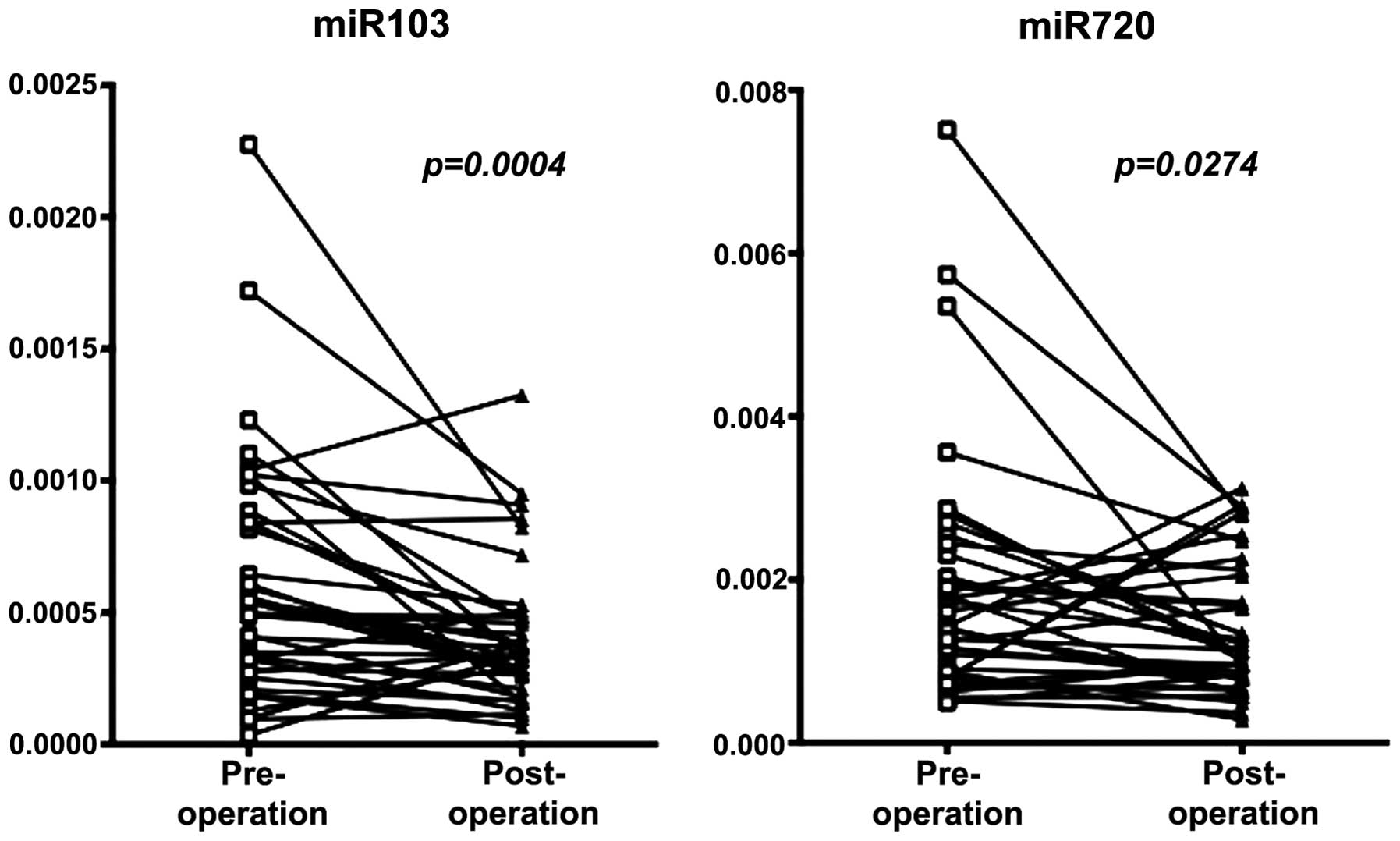
Confirmation of results obtained by miRNA
array by qRT-PCR
We then attempted to confirm the results obtained by
miRNA array by qRT-PCR in extended samples of CRC patients (n=30).
As shown in Fig. 2, significant
decrease in miRNA levels was noted in the post-operative serum for
both miRNAs; P=0.0004 for miR-103, and P=0.027 for miR-720.
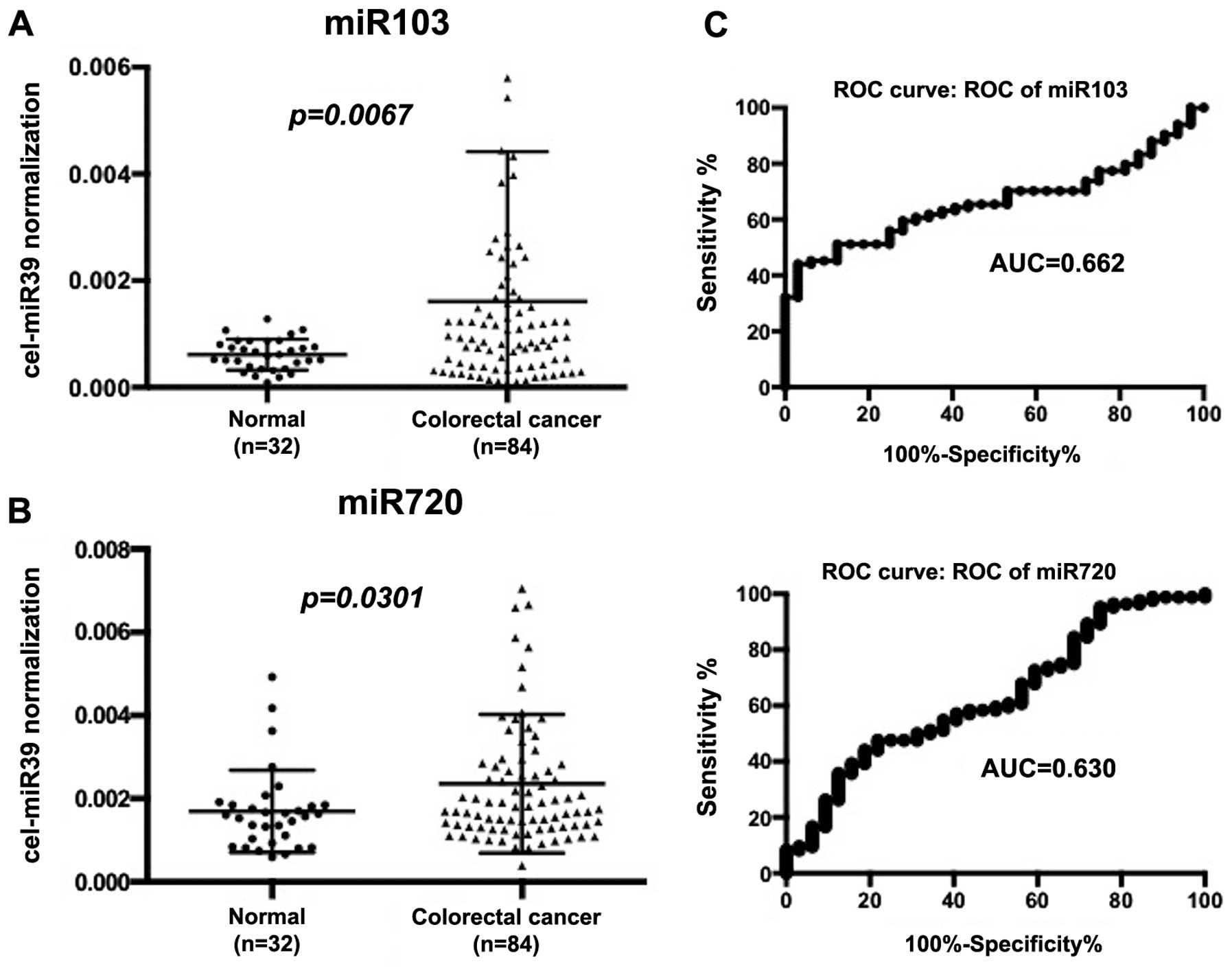
Expression of miRNAs in serum of normal
and CRC patients
We examined serum miR-103 and miR-720 levels in 32
non-cancer patients and 84 CRC patients. As shown in Fig. 3, both miRNAs were significantly
higher in CRC patients than non-cancer patients (P=0.0067 for
miR-103, and P=0.030 for miR-720, respectively). ROC curve was
drawn for serum miR-103 and miR-720, which yielded 0.662 for
miR-103 and 0.630 for miR-720 as a value of AUC (Fig. 3). In discriminating CRC from
non-tumor control subjects, the sensitivity and specificity of
miR-103 were 55.9% and 75.0% at a cut-off point of 0.00081; these
values for miR-720 were 58.3% and 56.3% at a cut-off point of
0.0016.
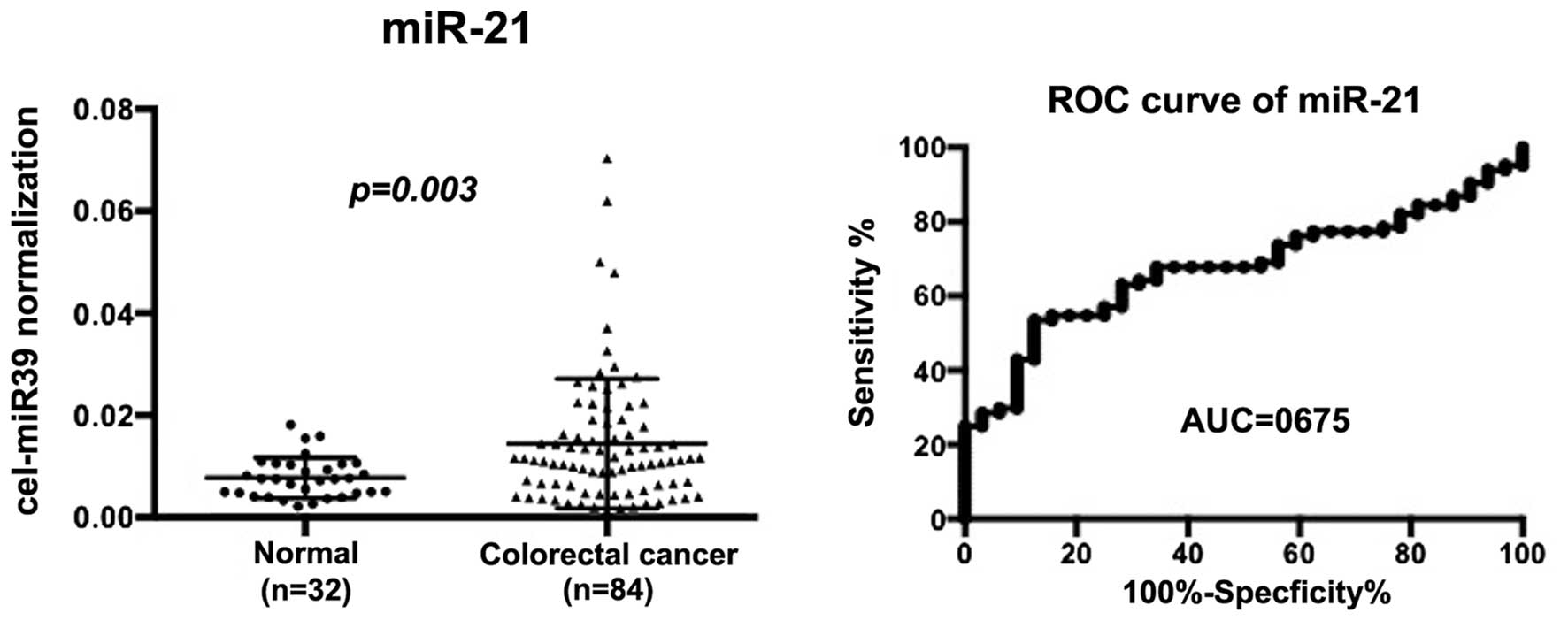
Serum miR-21 expression in non-cancer and
CRC patients
As a reference we examined miR-21 as a putative
circulating miRNA. Using the same serum sets of normal and CRC
patients, we found that serum miR-21 levels increased significantly
in CRC patients as compared to non-tumor control patients
(P=0.003). AUC of the ROC curves was 0.675. When a cut-off point
was set at 0.0107, the sensitivity was 54.7% and the specificity
was 84.4% in discriminating CRC from non-tumor control subjects
(Fig. 4).
Expression of miR-103 and miR-720 in
patients serum and clinicopathological characteristics
CRC patients were divided into two groups by a
median expression. Clinical and pathological survey indicated that
high expression of miR-103 was significantly associated with poor
differentiation and lymphatic invasion as compared to low miR-103
expression group (P=0.044 and P=0.040, respectively; Table I). High expression of miR-720 was
significantly associated with male gender and lymph node metastasis
as compared to low miR-720 expression group (P=0.001 and P=0.048,
respectively; Table I).
 | Table IRelation between miR-103, miR-720 and
clinico-pathological features. |
Table I
Relation between miR-103, miR-720 and
clinico-pathological features.
| miR-103 | miR-720 |
|---|
|
|
|
|---|
| Factors | High | Low | P-value | High | Low | P-value |
|---|
| Gender | | | 0.165 | | | 0.001a |
| Male | 31 | 25 | | 35 | 21 | |
| Female | 11 | 17 | | 7 | 21 | |
| Differentiation | | | 0.044a | | | 0.503 |
| tub1 | 12 | 21 | | 15 | 18 | |
| tub2 por muc | 30 | 21 | | 27 | 24 | |
| Tumor size | | | 1.000 | | | 1.000 |
| ≥35 mm | 22 | 22 | | 22 | 22 | |
| <35 mm | 20 | 20 | | 20 | 20 | |
| Serosal invasion | | | 0.172 | | | 1.000 |
| Positive | 30 | 24 | | 15 | 27 | |
| Negative | 12 | 18 | | 15 | 27 | |
| Lymph node
metastatis | | | 0.820 | | | 0.048a |
| Positive | 19 | 18 | | 23 | 14 | |
| Negative | 23 | 24 | | 19 | 28 | |
| Lymphatic
invasion | | | 0.040a | | | 0.827 |
| Positive | 25 | 18 | | 21 | 22 | |
| Negative | 13 | 24 | | 21 | 20 | |
| Venous
invasion | | | 0.474 | | | 0.095 |
| Positive | 14 | 11 | | 16 | 9 | |
| Negative | 28 | 31 | | 26 | 33 | |
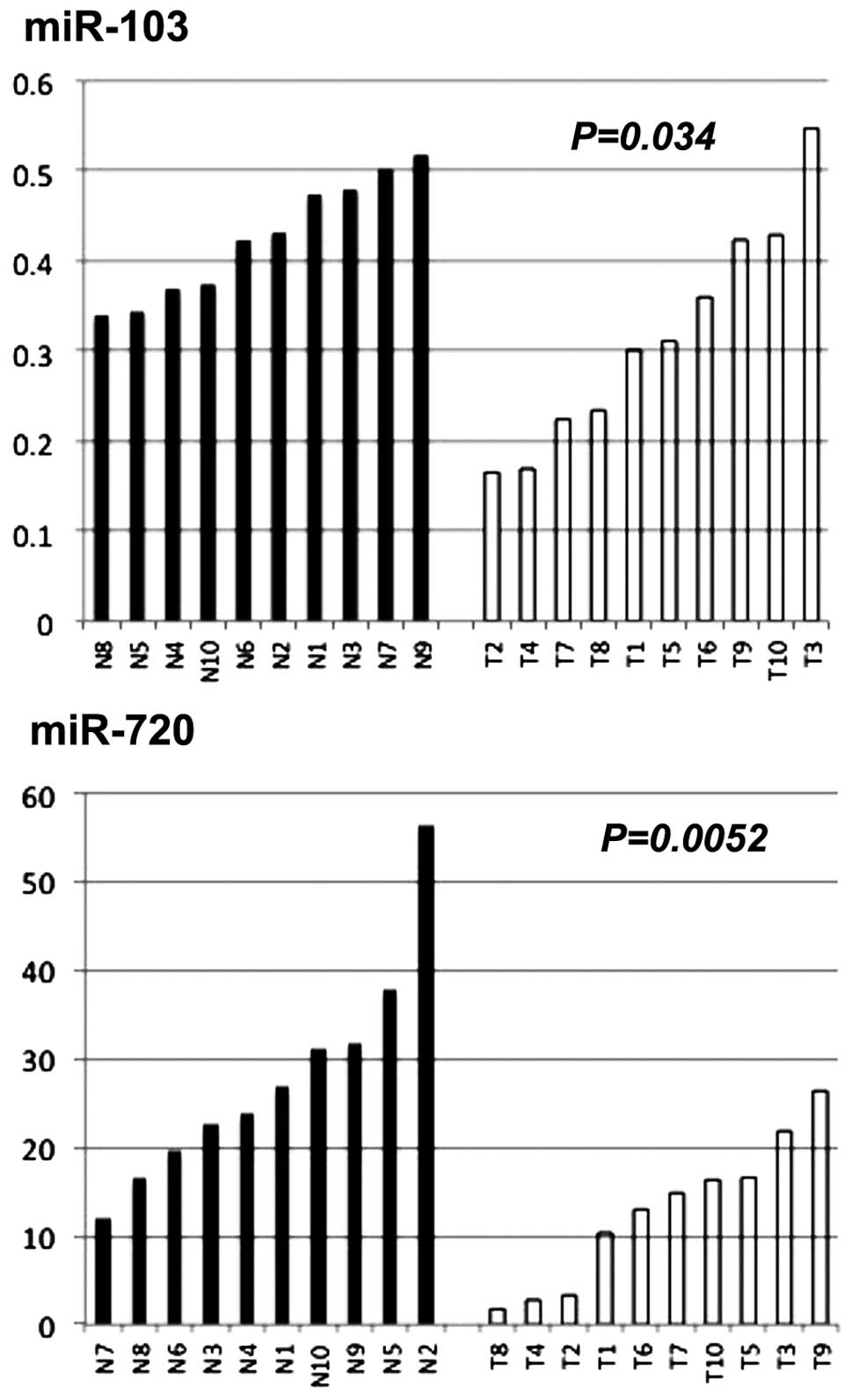
Expression of miR-103 and miR-720 in
normal mucosa and CRC tissue samples
We examined miR-103 and miR-720 expression in normal
colonic mucosa (n=10) and CRC tissue samples (n=10). Both miRs were
significantly lower in CRC tissue samples than in normal colonic
mucosa (P=0.034 for miR-103, and P=0.0052 for miR-720) (Fig. 5).
Discussion
In this study, we explored to identify a novel serum
marker for CRC by comparison of patient serum before and after
operation, using microarray analysis of miRNAs. miRNAs were mostly
reduced after surgery to various degrees (Fig. 1). Among them, miR-103 and miR-720
showed relatively large reduction after surgery. We validated the
results by qRT-PCR in extended number of CRC patients (Fig. 2). These findings suggest that the
two miRNAs could be derived from main CRC tumors, either directly
or indirectly and that they may be useful to monitor the disease
progression.
We demonstrated that the serum levels of miR-103 and
miR-720 increased in CRC patients compared with non-cancer patients
(Fig. 3). The ROC curves of
miR-103 and miR-720 to distinguish cancer patients from non-cancer
patients appeared to have limitation in sensitivity and
specificity. To estimate its value, we examined serum miR-21 in the
same series of non-tumor and CRC patients. Serum miR-21 is well
known as a serum biomarker for CRC (13). We found that diagnostic power of
miR-103 was similar to that of miR-21 in our series but that
miR-720 showed limited specificity as compared to miR-21. Thus,
miR-103 might be better biomarker for CRC than miR-720.
Clinicopathological survey showed that high
expression of serum miR-103 was associated with poor
differentiation and lymphatic invasion. Chen et al showed
that, miR-103 promoted metastasis of CRC by targeting the
metastasis suppressors DAPK and KLF4. This study also showed that
miR-103 had a role in downregulating E-cadherin, claudin-3, and
occludin, which sensitized tumor cells to EMT-inducing signals, and
led to local invasion (14).
Additionally, we found that high expression of serum miR-720 was
associated with lymph node metastasis. The finding is consistent
with a report by Wang et al that high level of miR-720 in
CRC tissues correlated with the tumor size, lymphatic metastasis,
distant metastasis, and poor prognosis (15).
In analysis of miR-103 and miR-720 derived from the
tissue samples, we found that the expression levels of the two
miRNAs, especially miR-720 were significantly lower in CRC tissue
samples than in normal colonic mucosa (Fig. 5). The contradictory miRNA
expression levels between the tumor tissues and the serum have been
demonstrated as to several miRNAs, but this phenomenon remains
unexplained and needs to be further explored (16,17).
A previous in vitro study showed that miRNA profiles in a
conditioned medium were different from those in cells, and it is
thus suggested that the secreted miRNAs represent a class of
signaling molecules in mediating intercellular communication
(18). The precise mechanism of
miRNA packaging and secretion is largely unknown, and investigation
of the mechanism is a future issue of miRNA research (19).
In conclusion, we identified miR-103 and miR-720
from differential expression profile between pre- and
post-operation as novel serum biomarkers for CRC. Further
investigation of serum miR-103 and miR-720 in patient prognosis and
monitoring therapeutic efficacy of chemotherapy is essential.
Acknowledgements
This study was supported by a Grant-in-Aid for
Scientific Research (B) (24390315 to H.Y.).
Abbreviations:
|
CRC
|
colorectal cancer
|
|
miR, miRNA
|
microRNA
|
|
qRT-PCR
|
quantitative reverse
transcription-polymerase chain reaction
|
References
|
1
|
Inoue Y and Kusunoki M: Advances and
directions in chemotherapy using implantable port systems for
colorectal cancer: A historical review. Surg Today. 44:1406–1414.
2014. View Article : Google Scholar :
|
|
2
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hewitson P, Glasziou P, Watson E, Towler B
and Irwig L: Cochrane systematic review of colorectal cancer
screening using the fecal occult blood test (hemoccult): An update.
Am J Gastroenterol. 103:1541–1549. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ochiai H, Ohishi T, Osumi K, Tokuyama J,
Urakami H, Seki S, Shimada A, Matsui A, Isobe Y, Murata Y, et al:
Reevaluation of serum p53 antibody as a tumor marker in colorectal
cancer patients. Surg Today. 42:164–168. 2012. View Article : Google Scholar
|
|
5
|
Mendell JT and Olson EN: MicroRNAs in
stress signaling and human disease. Cell. 148:1172–1187. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schetter AJ, Leung SY, Sohn JJ, Zanetti
KA, Bowman ED, Yanaihara N, Yuen ST, Chan TL, Kwong DL, Au GK, et
al: MicroRNA expression profiles associated with prognosis and
therapeutic outcome in colon adenocarcinoma. JAMA. 299:425–436.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lu J, Getz G, Miska EA, Alvarez-Saavedra
E, Lamb J, Peck D, Sweet-Cordero A, Ebert BL, Mak RH, Ferrando AA,
et al: MicroRNA expression profiles classify human cancers. Nature.
435:834–838. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen X, Ba Y, Ma L, Cai X, Yin Y, Wang K,
Guo J, Zhang Y, Chen J, Guo X, et al: Characterization of microRNAs
in serum: A novel class of biomarkers for diagnosis of cancer and
other diseases. Cell Res. 18:997–1006. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fabbri M: miRNAs as molecular biomarkers
of cancer. Expert Rev Mol Diagn. 10:435–444. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ng EK, Chong WW, Jin H, Lam EK, Shin VY,
Yu J, Poon TC, Ng SS and Sung JJ: Differential expression of
microRNAs in plasma of patients with colorectal cancer: A potential
marker for colorectal cancer screening. Gut. 58:1375–1381. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kanaan Z, Rai SN, Eichenberger MR, Roberts
H, Keskey B, Pan J and Galandiuk S: Plasma miR-21: A potential
diagnostic marker of colorectal cancer. Ann Surg. 256:544–551.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nonaka R, Nishimura J, Kagawa Y, Osawa H,
Hasegawa J, Murata K, Okamura S, Ota H, Uemura M, Hata T, et al:
Circulating miR-199a-3p as a novel serum biomarker for colorectal
cancer. Oncol Rep. 32:2354–2358. 2014.PubMed/NCBI
|
|
13
|
Toiyama Y, Takahashi M, Hur K, Nagasaka T,
Tanaka K, Inoue Y, Kusunoki M, Boland CR and Goel A: Serum miR-21
as a diagnostic and prognostic biomarker in colorectal cancer. J
Natl Cancer Inst. 105:849–859. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen HY, Lin YM, Chung HC, Lang YD, Lin
CJ, Huang J, Wang WC, Lin FM, Chen Z, Huang HD, et al: miR-103/107
promote metastasis of colorectal cancer by targeting the metastasis
suppressors DAPK and KLF4. Cancer Res. 72:3631–3641. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang X, Kuang Y, Shen X, Zhou H, Chen Y,
Han Y, Yuan B, Zhou J, Zhao H, Zhi Q and Xue X: Evaluation of
miR-720 prognostic significance in patients with colorectal cancer.
Tumour Biol. 36:719–727. 2015. View Article : Google Scholar
|
|
16
|
Yang IP, Tsai HL, Huang CW, Huang MY, Hou
MF, Juo SH and Wang JY: The functional significance of microRNA-29c
in patients with colorectal cancer: A potential circulating
biomarker for predicting early relapse. PLoS One. 8:e668422013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Heegaard NH, Schetter AJ, Welsh JA, Yoneda
M, Bowman ED and Harris CC: Circulating micro-RNA expression
profiles in early stage nonsmall cell lung cancer. Int J Cancer.
130:1378–1386. 2012. View Article : Google Scholar :
|
|
18
|
Zhang Y, Liu D, Chen X, Li J, Li L, Bian
Z, Sun F, Lu J, Yin Y, Cai X, et al: Secreted monocytic miR-150
enhances targeted endothelial cell migration. Mol Cell. 39:133–144.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cheng H, Zhang L, Cogdell DE, Zheng H,
Schetter AJ, Nykter M, Harris CC, Chen K, Hamilton SR and Zhang W:
Circulating plasma MiR-141 is a novel biomarker for metastatic
colon cancer and predicts poor prognosis. PLoS One. 6:e177452011.
View Article : Google Scholar : PubMed/NCBI
|