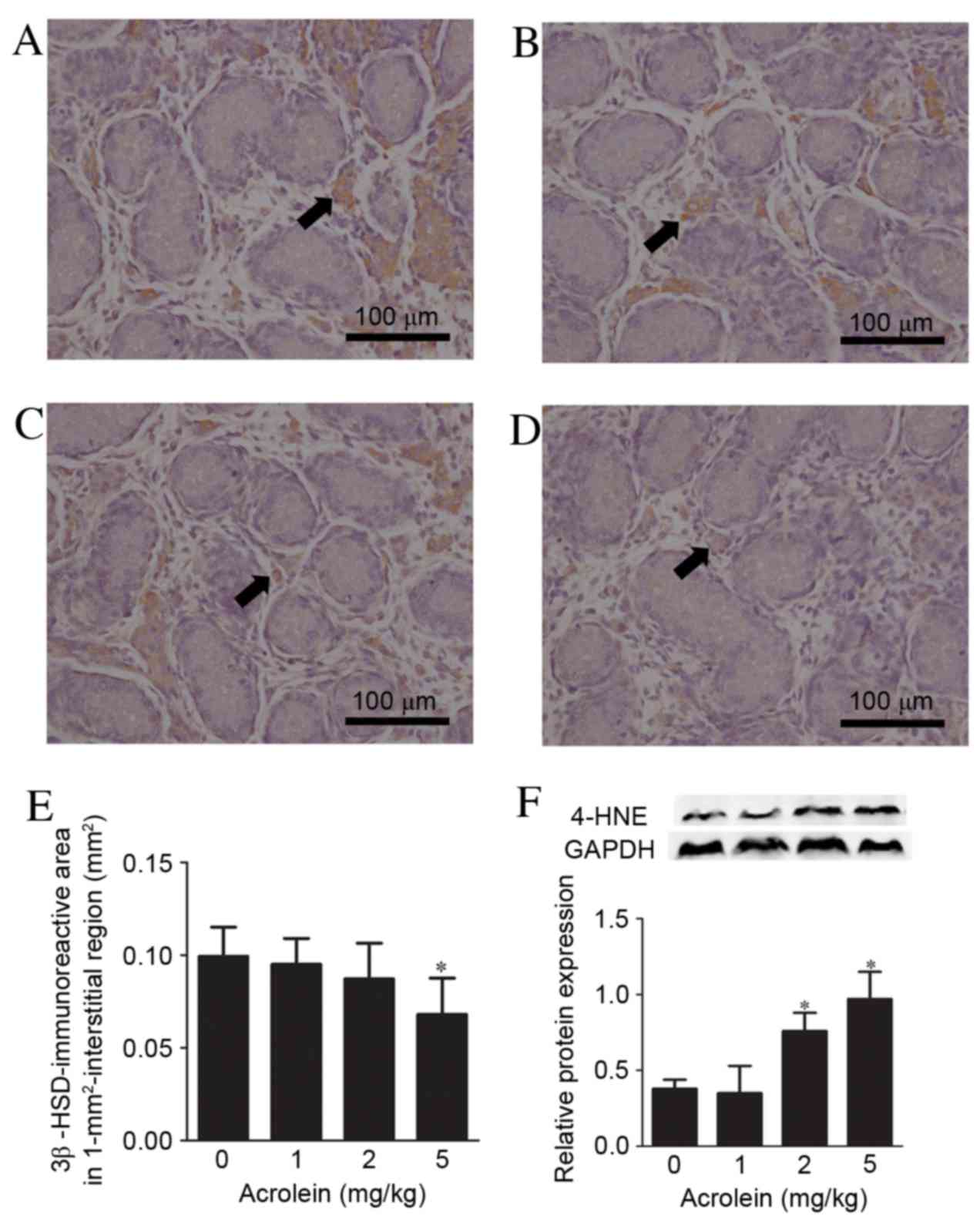
|
1
|
Skakkebaek NE, Rajpert-De Meyts E and Main
KM: Testicular dysgenesis syndrome: An increasingly common
developmental disorder with environmental aspects. Hum Reprod.
16:972–978. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Schnack TH, Poulsen G, Myrup C, Wohlfahrt
J and Melbye M: Familial coaggregation of cryptorchidism,
hypospadias, and testicular germ cell cancer: A nationwide cohort
study. J Natl Cancer Inst. 102:187–192. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sharpe RM and Skakkebaek NE: Testicular
dysgenesis syndrome: Mechanistic insights and potential new
downstream effects. Fertil Steril. 89:(2 Suppl). e33–e38. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Macleod DJ, Sharpe RM, Welsh M, Fisken M,
Scott HM, Hutchison GR, Drake AJ and van den Driesche S: Androgen
action in the masculinization programming window and development of
male reproductive organs. Int J Androl. 33:279–287. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu D, Shen L, Tao Y, Kuang Y, Cai L, Wang
D, He M, Tong X, Zhou S, Sun J, et al: Alterations in gene
expression during sexual differentiation in androgen receptor
knockout mice induced by environmental endocrine disruptors. Int J
Mol Med. 35:399–404. 2015.PubMed/NCBI
|
|
6
|
Araki A, Mitsui T, Miyashita C, Nakajima
T, Naito H, Ito S, Sasaki S, Cho K, Ikeno T, Nonomura K and Kishi
R: Association between maternal exposure to di (2-ethylhexyl)
phthalate and reproductive hormone levels in fetal blood: The
Hokkaido study on environment and children's health. PLoS One.
9:e1090392014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wilson VS, Lambright CR, Furr JR,
Howdeshell KL and Gray Earl L Jr: The herbicide linuron reduces
testosterone production from the fetal rat testis during both in
utero and in vitro exposures. Toxicol Lett. 186:73–77. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Stevens JF and Maier CS: Acrolein:
Sources, metabolism, and biomolecular interactions relevant to
human health and disease. Mol Nutr Food Res. 52:7–25. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cahill TM: Ambient acrolein concentrations
in coastal, remote, and urban regions in California. Environ Sci
Technol. 48:8507–8513. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Logue JM, Price PN, Sherman MH and Singer
BC: A method to estimate the chronic health impact of air
pollutants in U.S. residences. Environ Health Perspect.
120:216–222. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Moghe A, Ghare S, Lamoreau B, Mohammad M,
Barve S, McClain C and Joshi-Barve S: Molecular mechanisms of
acrolein toxicity: Relevance to human disease. Toxicol Sci.
143:242–255. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
DeJarnett N, Conklin DJ, Riggs DW, Myers
JA, O'Toole TE, Hamzeh I, Wagner S, Chugh A, Ramos KS, Srivastava
S, et al: Acrolein exposure is associated with increased
cardiovascular disease risk. J Am Heart Assoc. 3:pii: e000934.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lovell MA, Xie C and Markesbery WR:
Acrolein is increased in Alzheimer's disease brain and is toxic to
primary hippocampal cultures. Neurobiol Aging. 22:187–194. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Golla U, Bandi G and Tomar RS: Molecular
cytotoxicity mechanisms of allyl alcohol (acrolein) in budding
yeast. Chem Res Toxicol. 28:1246–1264. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Slott VL and Hales BF: The embryolethality
and teratogenicity of acrolein in cultured rat embryos. Teratology.
34:155–163. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kenney LB, Laufer MR, Grant FD, Grier H
and Diller L: High risk of infertility and long term gonadal damage
in males treated with high dose cyclophosphamide for sarcoma during
childhood. Cancer. 91:613–621. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Westlind A, Malmebo S, Johansson I, Otter
C, Andersson TB, Ingelman-Sundberg M and Oscarson M: Cloning and
tissue distribution of a novel human cytochrome p450 of the CYP3A
subfamily, CYP3A43. Biochem Biophys Res Commun. 281:1349–1355.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ekhart C, Doodeman VD, Rodenhuis S, Smits
PH, Beijnen JH and Huitema AD: Influence of polymorphisms of drug
metabolizing enzymes (CYP2B6, CYP2C9, CYP2C19, CYP3A4, CYP3A5,
GSTA1, GSTP1, ALDH1A1 and ALDH3A1) on the pharmacokinetics of
cyclophosphamide and 4-hydroxycyclophosphamide. Pharmacogenet
Genomics. 18:515–523. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ji YL, Wang H, Liu P, Zhao XF, Zhang Y,
Wang Q, Zhang H, Zhang C, Duan ZH, Meng C and Xu DX: Effects of
maternal cadmium exposure during late pregnant period on testicular
steroidogenesis in male offspring. Toxicol Lett. 205:69–78. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Auerbach SS, Mahler J, Travlos GS and
Irwin RD: A comparative 90-day toxicity study of allyl acetate,
allyl alcohol and acrolein. Toxicology. 253:79–88. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rashedinia M, Lari P, Abnous K and
Hosseinzadeh H: Proteomic analysis of rat cerebral cortex following
subchronic acrolein toxicity. Toxicol Appl Pharmacol. 272:199–207.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tsukahara S, Nakajima D, Kuroda Y, Hojo R,
Kageyama S and Fujimaki H: Effects of maternal toluene exposure on
testosterone levels in fetal rats. Toxicol Lett. 185:79–84. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu F, Li XL, Lin T, He DW, Wei GH, Liu JH
and Li LS: The cyclophosphamide metabolite, acrolein, induces
cytoskeletal changes and oxidative stress in Sertoli cells. Mol
Biol Rep. 39:493–500. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
He X, Song W, Liu C, Chen S and Hua J:
Rapamycin inhibits acrolein-induced apoptosis by alleviating
ROS-driven mitochondrial dysfunction in male germ cells. Cell
Prolif. 47:161–171. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Griswold SL and Behringer RR: Fetal Leydig
cell origin and development. Sex Dev. 3:1–15. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
O'Shaughnessy PJ, Baker PJ and Johnston H:
The foetal Leydig cell-differentiation, function and regulation.
Int J Androl. 29:90–108. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Habert R, Lejeune H and Saez JM: Origin,
differentiation and regulation of fetal and adult Leydig cells. Mol
Cell Endocrinol. 179:47–74. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhao B, Li L, Liu J, Li H, Zhang C1, Han
P, Zhang Y, Yuan X, Ge RS and Chu Y: Exposure to perfluorooctane
sulfonate in utero reduces testosterone production in rat fetal
Leydig cells. PLoS One. 9:e788882014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lin H, Ge RS, Chen GR, Hu GX, Dong L, Lian
QQ, Hardy DO, Sottas CM, Li XK and Hardy MP: Involvement of
testicular growth factors in fetal Leydig cell aggregation after
exposure to phthalate in utero. Proc Natl Acad Sci USA.
105:7218–7222. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Payne AH and Hales DB: Overview of
steroidogenic enzymes in the pathway from cholesterol to active
steroid hormones. Endocr Rev. 25:947–970. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tyczewska M, Rucinski M, Ziolkowska A,
Trejter M, Szyszka M and Malendowicz LK: Expression of selected
genes involved in steroidogenesis in the course of
enucleation-induced rat adrenal regeneration. Int J Mol Med.
33:613–623. 2014.PubMed/NCBI
|
|
33
|
Chadwick AC, Holme RL, Chen Y, Thomas MJ,
Sorci-Thomas MG, Silverstein RL, Pritchard KA Jr and Sahoo D:
Acrolein impairs the cholesterol transport functions of high
density lipoproteins. PLoS One. 10:e01231382015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Luu-The V: Assessment of steroidogenesis
and steroidogenic enzyme functions. J Steroid Biochem Mol Biol.
137:176–182. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ivell R and Bathgate RA: Reproductive
biology of the relaxin-like factor (RLF/INSL3). Biol Reprod.
67:699–705. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Parent RA, Caravello HE and Hoberman AM:
Reproductive study of acrolein on two generations of rats. Fundam
Appl Toxicol. 19:228–237. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kuwada M, Kawashima R, Nakamura K, Kojima
H, Hasumi H, Maki J and Sugano S: Neonatal exposure to endocrine
disruptors suppresses juvenile testis weight and steroidogenesis
but spermatogenesis is considerably restored during puberty.
Biochem Biophys Res Commun. 295:193–197. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Monroe RK and Halvorsen SW: Environmental
toxicants inhibit neuronal Jak tyrosine kinase by mitochondrial
disruption. Neurotoxicology. 30:589–598. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Shi Z, Feng Y, Wang J, Zhang H, Ding L and
Dai J: Perfluorododecanoic acid-induced steroidogenic inhibition is
associated with steroidogenic acute regulatory protein and reactive
oxygen species in cAMP-stimulated Leydig cells. Toxicol Sci.
114:285–294. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhou L, Beattie MC, Lin CY, Liu J, Traore
K, Papadopoulos V, Zirkin BR and Chen H: Oxidative stress and
phthalate-induced down-regulation of steroidogenesis in MA-10
Leydig cells. Reprod Toxicol. 42:95–101. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ayala A, Muñoz MF and Argüelles S: Lipid
peroxidation: Production, metabolism, and signaling mechanisms of
malondialdehyde and 4-hydroxy-2-nonenal. Oxid Med Cell Longev.
2014:3604382014. View Article : Google Scholar : PubMed/NCBI
|