Introduction
Cholangiocarcinoma (CCA) is an aggressive malignant
tumor that originates from bile duct epithelial cells (1). This cancer is the second most common
primary hepatic malignancy, after hepatocellular cancer (2). There is a significant geographical
variation in the incidence of CCA, with the highest incidence
occurring in eastern Asian countries including Thailand, Korea,
China and Japan (3,4). Conditions that result in prolonged
biliary inflammation, such as primary sclerosing cholangitis (PSC),
liver fluke infestation, congenital abnormalities, such as
choledochal cysts, and hepatitis B and C, predispose patients to
developing CCA (5). CCA is often
detected at an advanced stage, which has a very poor prognosis, and
a 5-year survival rate of <5–10% (6). Additionally, CCA tends to remain
clinically insidious until it has progressed to a terminal stage.
Despite the limited clinical benefits, chemotherapy represents the
cornerstone of disease management for patients with inoperable CCA
(7). Although novel anticancer
drugs and therapeutic regimens have been extensively explored, few
treatment options have exhibited promising results in improving the
outcome of this lethal disease (8,9).
Therefore, the development of new therapeutic strategies is
urgently needed.
Therapeutic agents, including gemcitabine, cisplatin
or 5-FU, cause major side-effects when given at the maximum
tolerated doses, and CCA fails to adequately respond to these drugs
due to the acquisition of chemoresistance (1). In the current literature, there is no
neoadjuvant therapy that is considered a standard approach for the
treatment of patients with CCA (10). Therefore, we examined whether some
natural medications that possess potent antitumor,
anti-inflammatory and anti-angiogenic functions may be more
efficient in treating CCA.
Thymoquinone (TQ) is a bioactive ingredient derived
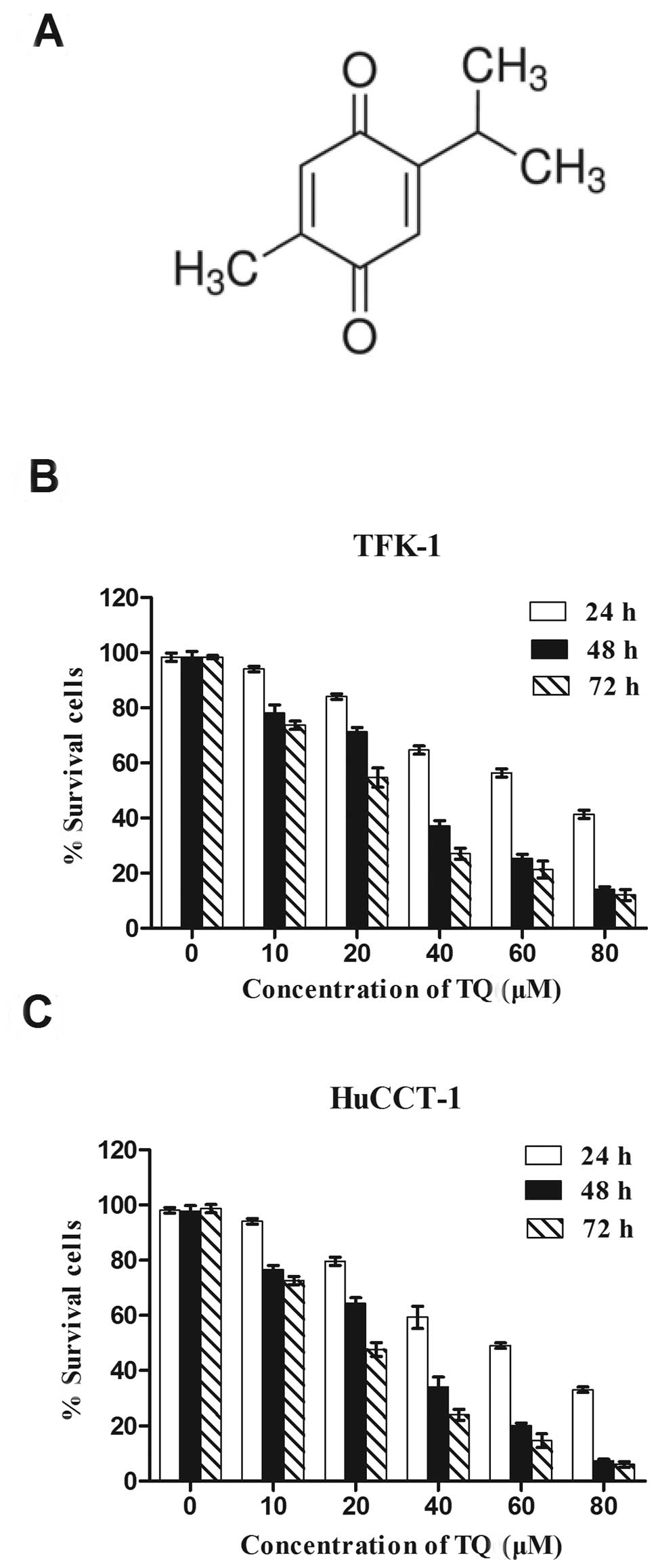
from black seed (Nigella sativa) oil (Fig. 1A) (11,12).
In folk medicine, the seed is reportedly associated with
anti-inflammatory effects and has been shown to protect against
bronchial asthma, dysentery, headache and gastrointestinal problems
(13). Furthermore, TQ has been
reported to exhibit anticancer effects on cell lines derived from
breast, colon, ovarian, larynx and lung cancer, as well as
myeloblastic leukemia and osteosarcomas, without any serious
side-effects (14,15).
To corroborate the anti-inflammatory and antitumor
activity of TQ, we sought to understand the activity of TQ in human
CCA cell lines and in a CCA-inoculated mouse model and to elucidate
the potential mechanism of action. Among the various molecular
defects that allow CCA cells to evade drug-induced apoptotic
signaling, the activation of the AKT and nuclear factor-κB (NF-κB)
pathways is particularly obvious (16–18).
Constitutive activation of the AKT and NF-κB pathways has been
reported to play a key role in cell proliferation, invasion,
angiogenesis, metastasis, suppression of apoptosis and
chemoresistance of CCA (19,20).
However, the potential efficacy of TQ in the human CCA cells and
the possible mechanism of action remain unknown.
Materials and methods
Materials
The p-AKT, AKT, XIAP, COX-2, p65 and cyclin B1
antibodies were obtained from Cell Signaling. The Bcl-2 and Bax
antibodies were purchased from Santa Cruz Biotechnology, and the
anti-β-actin antibody was purchased from Sigma. Thymoquinone
(Sigma) was dissolved in dehydrated alcohol to create a 20 mmol/l
stock solution.
Cell culture
TFK-1 cells, which are moderately differentiated,
and HuCCT1 cells, which are poorly differentiated, were kindly
provided by the Cancer Cell Repository, Tohoku University, Japan.
The cells were grown in monolayers in RPMI-1640 medium,
supplemented with antibiotics, 10% fetal bovine serum (FBS) (all
from Gibco) and 2 mmol/l glutamine at 37°C with 5% CO2
in a humidified atmosphere.
Cell viability assay
To assess cell viability, both the TFK-1 and HuCCT1
cells were seeded in 96-well plates at a density of
3×103 cells/well and allowed to adhere and grow
overnight in RPMI-1640 medium containing 10% heat-inactivated FBS.
Cell Counting Kit-8 (CCK-8; Dojindo Lab-Orgemcitabineries,
Kumamoto, Japan) was used to determine the viability of the cells.
The cells were then cultured with TQ (0, 10, 20, 40, 60, or 80 μM)
for 24, 48 or 72 h. Cell viability was measured using the CCK-8 kit
according to the manufacturer’s instructions, as previously
described (22). Three independent
experiments were performed.
Cell cycle analysis
The CycleTest™ Plus DNA Reagent kit (BD Biosciences,
San Jose, CA, USA) was used to identify the percentage of cells in
the G0–G1, S and G2-M phases of the cell cycle. Subconfluent CCA
cells were plated at a density of 1×106 cells/well in
6-well plates. After treatment with TQ (0–60 μM) for 48 h, the
supernatant was discarded, and the cells were washed with
phosphate-buffered saline (PBS), then centrifuged. The cells were
washed twice with ice-cold PBS, and 3 ml of ice-cold 70% ethanol
was added. The cells were then incubated for 1 h at 4°C. The cells
were washed twice with PBS, and 10 mg/ml RNase A was added.
Propidium iodide (PI) was added to the tubes at a final
concentration of 0.05 mg/ml, and the cells were incubated at 4°C
for 30 min in the dark. Flow cytometric analysis was performed
using FACScan (Becton-Dickinson) to detect the percentage of cells
in the various phases of the cell cycle, as previously described
(22). The experiments were
repeated three times.
Detection of cell apoptosis
The PI/Annexin V-FITC apoptosis detection kit (BD
Biosciences) was used to assess the number of apoptotic CCA cells
after treatment with TQ; the kit was used according to the
manufacturer’s instructions. Briefly, following treatment, the
cells were harvested with trypsin, washed in PBS and counted. The
cells were then resuspended in binding buffer, and 5 ml of Annexin
V and 5 ml of PI were added. The cells were then incubated at room
temperature for at least 15 min in the dark. The percentage of
apoptotic cells was analyzed using flow cytometry (Epics Altra II;
Beckman Coulter, USA) as previously described (23). The experiments were repeated three
times.
Electrophoretic mobility shift assay
(EMSA)
Nuclear extract (5 μg) was incubated with 1 μg of
poly(deoxyinosinic-deoxycytidylic acid) in binding buffer for 30
min at 4°C. DNA-binding activity was confirmed using a
biotin-labeled oligonucleotide bio-NF-κB probe
(5′-AGTTGAGGGGACTTTCCCAGGC-3′) using an EMSA kit according to the
manufacturer’s instructions (Viagene, Beijing, China). The probe
was resolved on a 4% polyacrylamide gel containing 0.25X TBE
(Tris/borate/EDTA) buffer and visualized using a Cool Imager
imaging system (IMGR002; Viagene).
Western blot assay
The method was described previously (24). Briefly, the cells were washed twice
in PBS, sonicated in lysis buffer and homogenized; alternatively,
tumor tissues were excised, minced and homogenized in protein
lysate buffer. Debris was removed by centrifugation. Samples (20
μg) of the total protein lysate were resolved on 12% polyacrylamide
SDS gels and were electrophoretically transferred onto
polyvinylidene difluoride (PVDF) membranes. The membranes were
blocked with 5% skim milk in TBST buffer (TBS plus 0.1% Tween-20),
incubated with the appropriate primary Ab and subsequently
incubated with an alkaline phosphatase-conjugated secondary Ab. The
blots were developed using 5-bromo-4-chloro-3-indolyl
phosphate/nitroblue tetrazolium (Tiangen Biotech Co., Ltd.,
Beijing, China). The membranes were then washed, and the protein
bands were visualized following exposure of the membrane to X-ray
film. β-actin was used as a loading control.
Animal model and treatments
Surgical procedures and administration of drugs to
the animals were in accordance with the Institutional Animal Ethics
Guidelines. Tumors were fixed by subcutaneous injection of
4×106 HuCCT-1 tumor cells into the flanks of the mice.
The tumor volumes were approximated according to the formula:
π/6xa2xb, where a is the short axis, and b is the long
axis. When the tumors reached ~100 mm3 after
approximately two weeks, the mice were randomly assigned to four
groups, each containing seven mice. The mice received either daily
doses of PBS or 2, 4 or 8 mg/mouse TQ given daily by intragastric
intubation. The doses and methods were based on our preliminary
experiments and previous reports (15). The treatments lasted for 20 days,
during which time the sizes of the tumors were documented. The mice
were euthanized three days after the last treatment, and the tumors
were excised, weighed and fixed in 10% buffered formalin for
immunohistochemistry and apoptotic assays.
Immunohistochemistry
Immunohistochemistry analysis was performed using an
anti-Ki67 Ab. Briefly, after deparaffinization, rehydration and
antigen retrieval, the permeabilized sections (4 μm) were blocked
with 3% bovine serum albumin (BSA) for 2 h and incubated overnight
with primary Abs. The sections were subsequently cultivated for 30
min with IgG, mounted and examined under a light microscope or a
fluorescence microscope.
In situ detection of apoptotic cells
Histological analysis of DNA fragmentation was
performed to identify the apoptotic cells. Tumor sections were
stained with the TUNEL reagent (Roche, Shanghai, China), and the
TUNEL-positive cells were counted in 10 randomly selected ×400
high-power fields. The apoptosis index was calculated according to
the following formula: The number of apoptotic cells/total number
of nucleated cells × 100%.
Statistical analysis
The data obtained are expressed as the mean values ±
standard deviation of at least three separate experiments.
Statistical significance was determined using Student’s t-test, and
P<0.05 was considered to indicate a statistically significant
difference.
Results
TQ exerts antiproliferative effects in
CCA cells in vitro
To determine the effects of TQ on cell growth, the
CCA cell lines (TFK-1 and HuCCT1) were treated with varying
concentrations (0–80 μM) of TQ for 24, 48 and 72 h, and cell
viability was assessed using the CCK-8 assay. As shown in Fig. 1B and C, TQ treatment resulted in a
dose- and time-dependent inhibition of cell growth in both the CCA
cell lines tested. The inhibitory concentration IC50
values were ~62.91, 33.47 and 23.17 μM for the TFK-1 cells and
48.27, 27.51 and 20.28 μM for the HuCCT1 cells for the 24, 48 and
72 h treatments, respectively. Of note, TQ inhibited the poorly
differentiated HuCCT1 cell line more than the moderately
differentiated TFK-1 cell line. These results indicate that TQ has
potent antiproliferative effects in CCA cell lines.
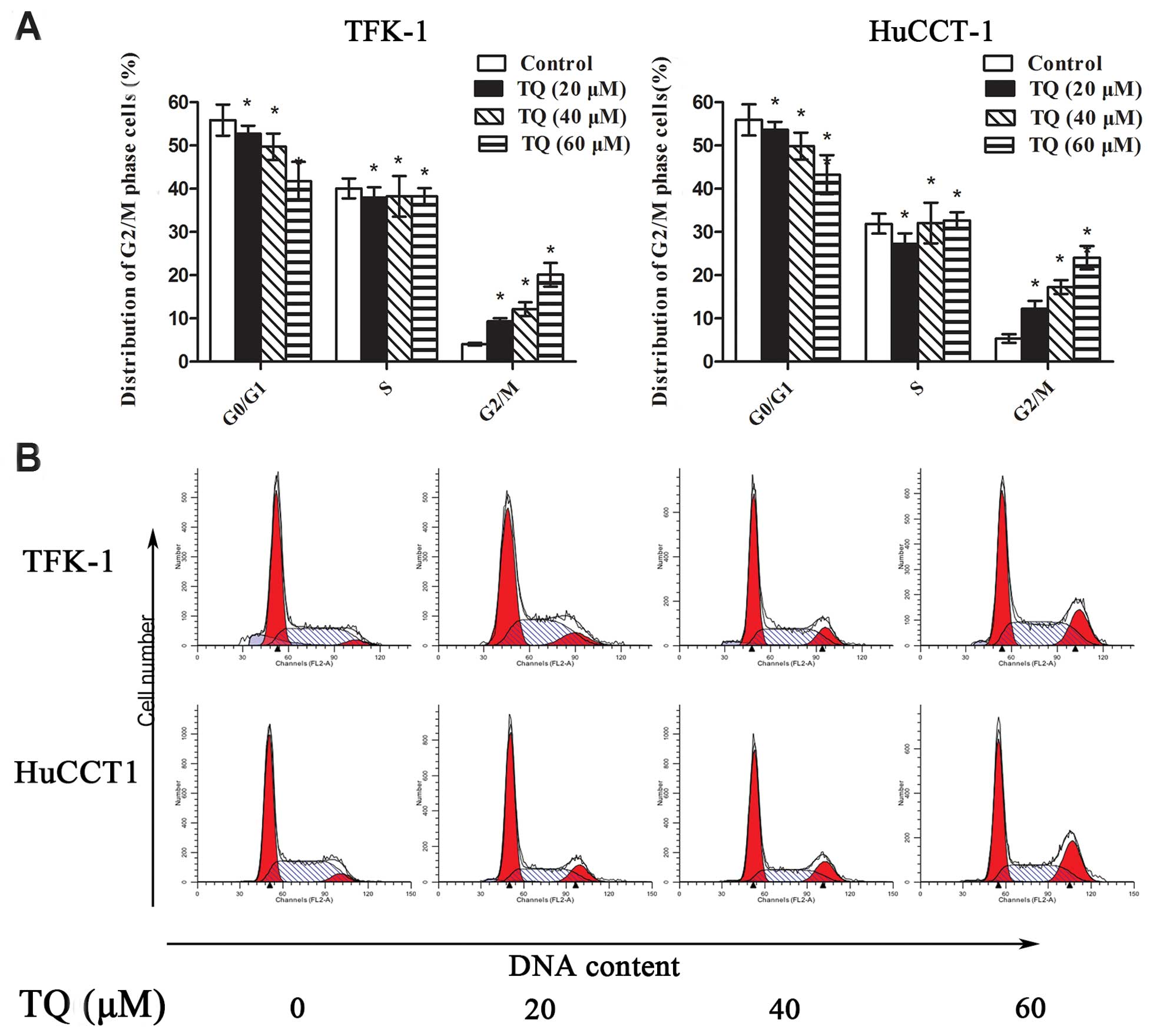
TQ induces G2/M phase cell cycle arrest
and alters the expression of G2/M phase cell cycle-related proteins
in CCA cells
Based on the preliminary assays in which we
evaluated the effect of TQ on the growth of CCA cells, the 0, 20,
40 and 60 μM doses of TQ were selected for further in vitro
mechanistic studies. To explore the underlying mechanism of the
TQ-induced growth inhibition of the cells, the effect of TQ on the
cell cycle distribution was studied using flow cytometric analysis
of the cellular DNA content. As shown in Fig. 2, treatment with TQ for 48 h resulted
in a significant dose-dependent arrest of the CCA cells in the G2/M
phase of the cell cycle. The G2/M phase cell cycle distribution was
4.05, 9.40, 11.76 and 18.55% in the TFK-1 cells (Fig. 2A) and 5.51, 12.25, 16.14 and 24.02%
in the HuCCT1 cells when treated with 0, 20, 40 and 60 μM TQ
(Fig. 2B), respectively. This
increase in the percentage of cells in G2/M phase was accompanied
by a concomitant reduction in the percentage of cells in G0/G1
phase.
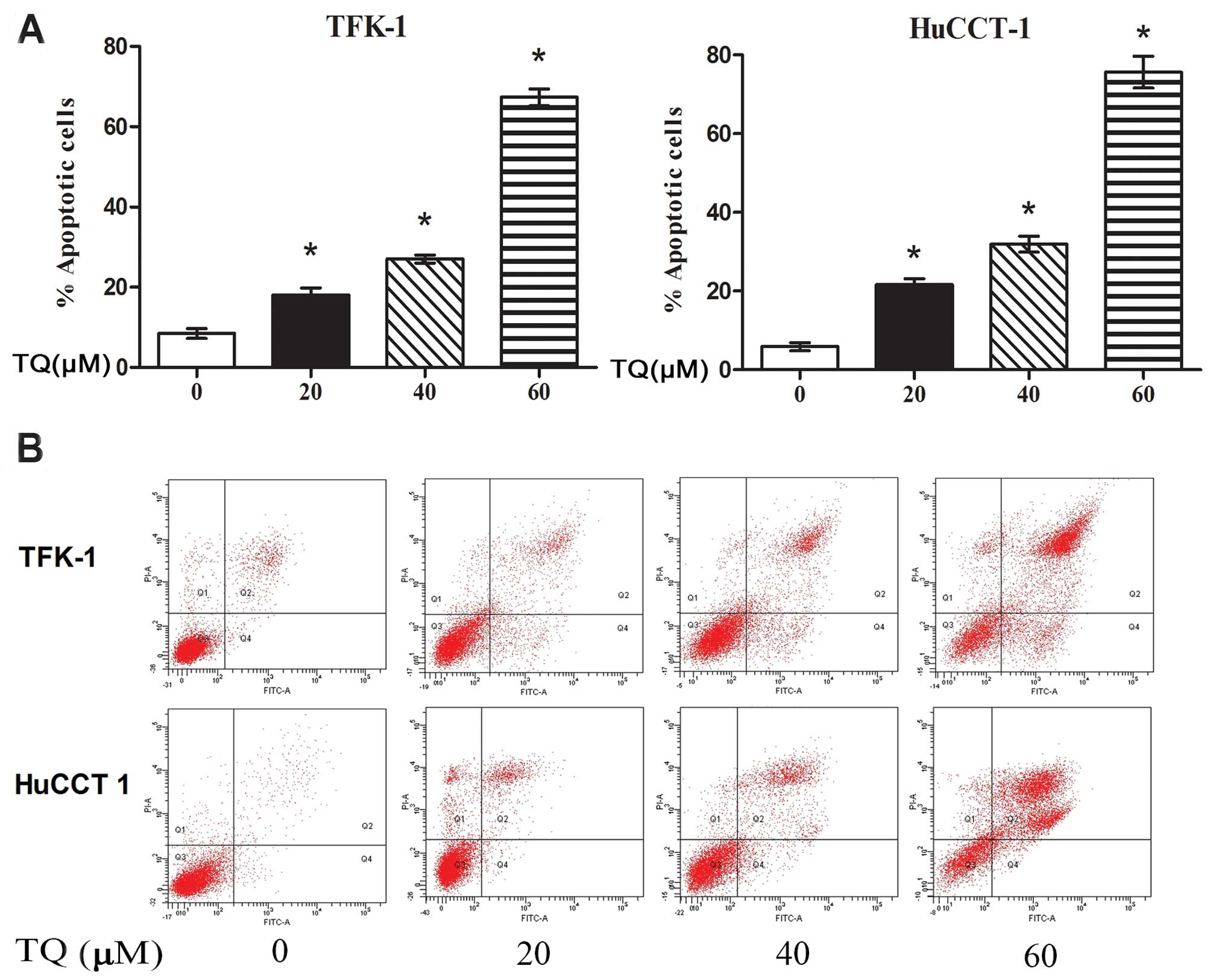
TQ potentiates the apoptosis-inducing
effect in CCA cells in vitro
In addition to the cell cycle arrest, morphological
observation of the TQ-treated CCA cells indicated that the
TQ-induced growth inhibition may also be associated with the
induction of apoptosis. Therefore, the apoptosis-inducing effect of
TQ in CCA cells was assessed. The cells were treated with various
concentrations of TQ (0–60 μM). Then, the cells were stained with
Annexin V/PI and subjected to flow cytometry to determine the
amount of apoptotic cells. As shown in Fig. 3, treatment with TQ for 48 h resulted
in a significant dose-dependent enhancement in both the number of
early and late apoptotic CCA cells. The percentages of apoptotic
cells were 8.5, 18.0, 26.1 and 67.3% in the TFK-1 cells (Fig. 3A) and 5.9, 21.7, 31.9 and 75.7% in
the HuCCT1 cells treated with 0, 20, 40 and 60 μM TQ (Fig. 3B), respectively. Taken together, our
results suggest that the TQ-induced growth inhibition was partially
due to the induction of apoptosis.
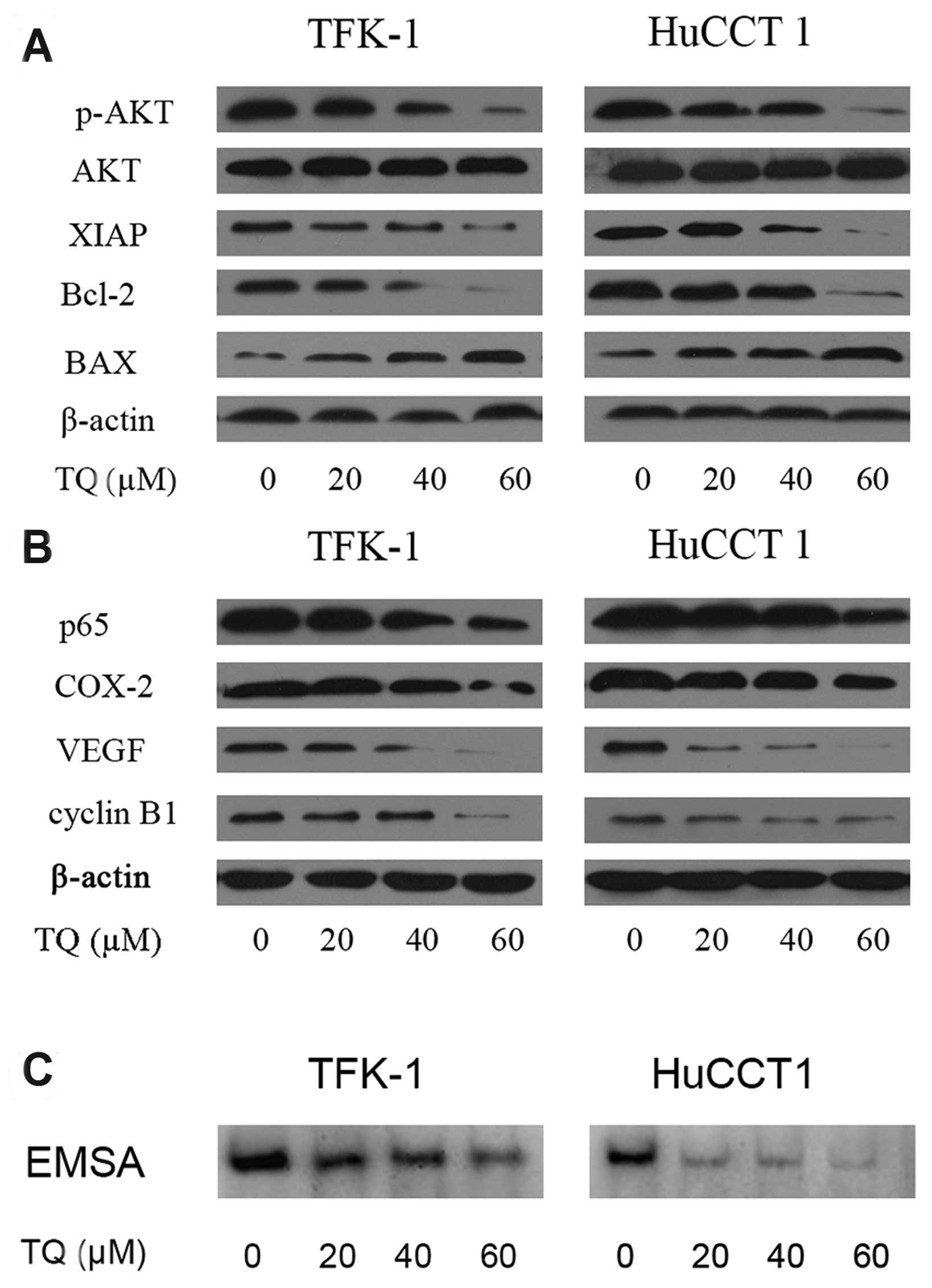
TQ suppresses PI3K and Akt activation in
CCA cells in vitro
To determine whether the antitumor effect was
associated with PI3K/Akt signaling, the TFK-1 and HuCCT-1 cells
were treated as described above for 48 h and were then subjected to
western blot analysis. As shown in Fig.
4, treatment with TQ resulted in the downregulation of p-Akt in
both of the CCA cell lines, whereas the levels of total Akt protein
were not altered (Fig. 4A).
Furthermore, the downregulation of p-Akt was associated with the
downregulation of XIAP and Bcl-2, as well as the upregulation of
BAX, which is further evidence of the induction of apoptosis in
cells exposed to TQ for 48 h (Fig.
4B). These results suggest that PI3K/Akt signaling was, at
least partially, involved in the effect.
TQ inhibits the effects of NF-κB and
NF-κB-regulated gene products in CCA cells in vitro
To investigate whether TQ abrogates the
constitutively active NF-κB in the CCA cells, we used a DNA binding
assay to determine whether NF-κB, a downstream target of the
PI3K/Akt signaling pathway, was also involved in the effect. After
the above treatments, nuclear extracts were obtained and the NF-κB
DNA-binding activity was determined using an EMSA assay;
additionally, total protein extracts were used to determine the
expression of the downstream NF-κB genes by western blot analysis.
As shown in Fig. 4C, treatment with
TQ resulted in constitutive NF-κB activity. In both cell lines, TQ
treatment significantly reduced the DNA-binding activity of NF-κB,
and this decrease was correlated with the increased inhibitory
effect on cell viability and increased apoptosis. To confirm our
hypothesis, we tested whether the inhibitory effect of TQ on NF-κB
may result in the downregulation of NF-κB regulated genes. As shown
in Fig. 4B, in both cell lines, TQ
treatment resulted in significantly decreased expression of COX-2,
VEGF and cyclin B1, which are known to be regulated by NF-κB. These
results are consistent with the increased growth inhibition and
apoptosis-inducing effects, suggesting that in vitro TQ
inhibits NF-κB DNA-binding activity and the expression of its
downstream gene products, which are believed to be partially
responsible for the enhanced cell killing.
TQ exerts antitumor, antiproliferative,
pro-apoptotic and anti-angiogenic effects in HuCCT1 xenografts in
nude mice
To evaluate the role of TQ in tumor proliferation
in vivo, we examined the ability of TQ to suppress the
growth of HuCCT-1 xenografts in nude mice. HuCCT-1-derived
xenograft tumors were allowed to develop and grow to a size of ~100
mm3; once the tumors had reached this size, TQ was
administered at daily doses of 2, 4 or 8 mg/mouse by intragastric
intubation for 20 days. As shown in Fig. 5A, the tumors in the mice treated
with various concentrations of TQ decreased in volume after 20 days
of treatment, and this reduction in volume was significantly
different from that of the control tumors. These results suggested
that TQ may largely inhibit the growth of the tumor xenografts. In
general, the tumors in the control group grew continuously during
the experimental period, whereas tumor growth in the TQ-treated
mice was suppressed significantly (Fig.
5C). However, there was no apparent change in liver (Fig. 5J), spleen (Fig. 5K) or body weight (Fig. 5B) in the animals, indicating that TQ
is a potential therapeutic agent for the treatment of CCA and is
relatively non-toxic to mice. Ki-67 staining for cell proliferation
was performed in the tumors that were removed from the animals. The
relative number of Ki-67-positive tumor cells was substantially
less in the tumors from mice treated with TQ than in the control
tumors (Fig. 5G and H).
Additionally, as shown in the representative images, the tumor
xenografts from the TQ-treated mice showed a marked increase in the
number of TUNEL-positive apoptotic cells compared to the control
group (Fig. 5G and I).
Quantification of TUNEL-stained samples showed a 2- to 3-fold
increase (P<0.05) in the number of TUNEL-positive cells in the
TQ-treated groups compared to the control group.
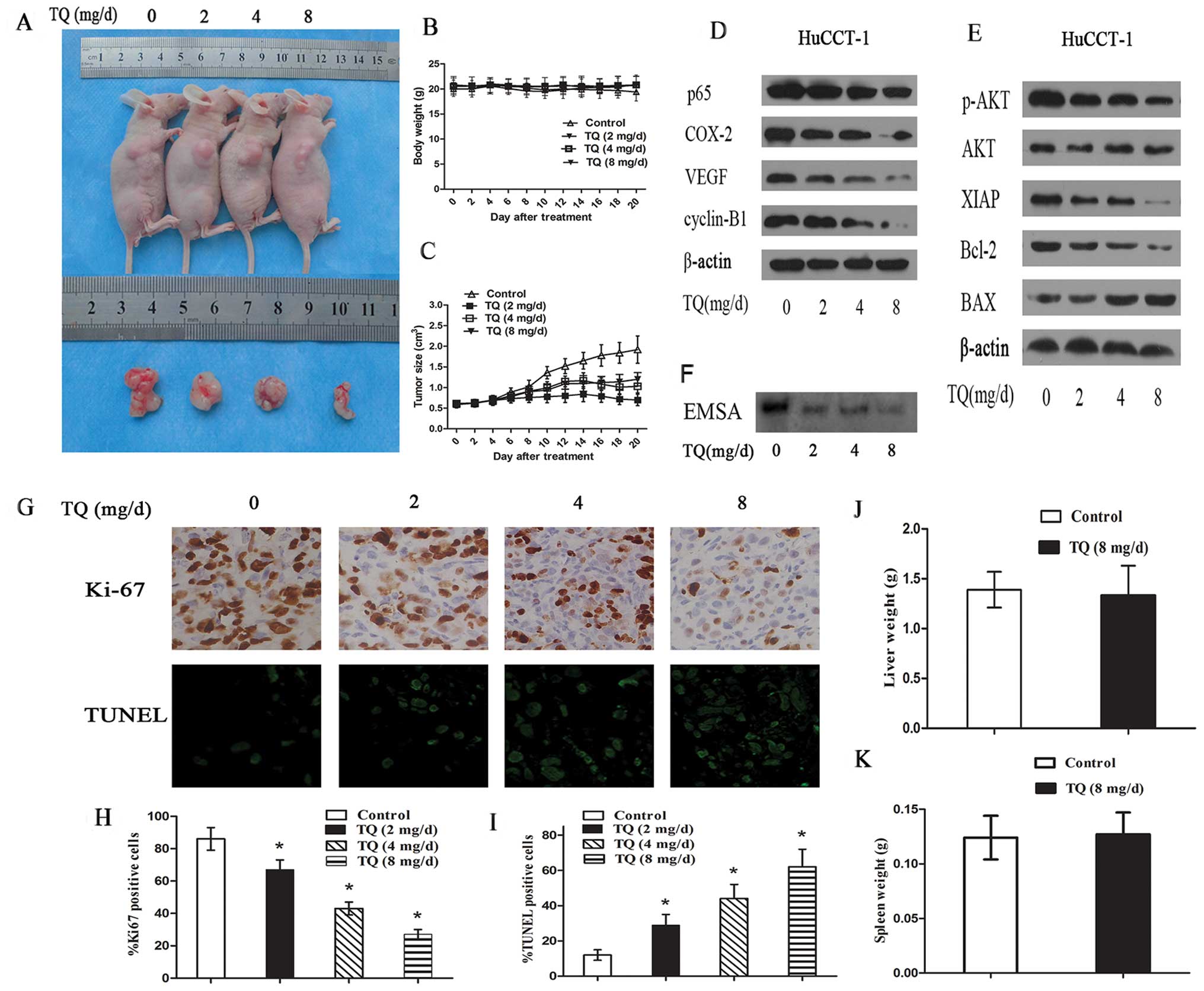 | Figure 5TQ inhibits CCA cancer tumor xenograft
growth in vivo. (A) HuCCT-1 cells were injected to the
flanks of nude mice and palpable tumors were allowed to develop for
14 days. Subsequently, PBS or 2, 4 or 8 mg/mouse TQ were given
daily by intragastric intubation for 20 days. Three days later,
tumors were excised and subjected to further analyses. Tumor
volumes in TQ administered mice were smaller than those of control
mice. (B) There was no significant difference in body weight
between TQ-treated animals and controls. (C) Tumor size was
measured every two days. There was a significant reduction in
relative tumor volume from TQ-treated animals when compared with
untreated controls. (D) Western blot analysis was performed to
detect the expression of p65, COX-2, VEGF, cyclin B1 in tumor
tissues. (E) The expression of AKT, phospho-AKT, XIAP, Bcl-2 and
BAX in CCA tumor tissue was also measured by western blotting, with
β-actin as protein internal control. (F) EMSA analysis of NF-κB
DNA-binding activity in tumor tissues. (G) Immunohistochemical
analysis of Ki-67 for cell proliferation and TUNEL analysis of
apoptotic cells in tumor tissues. (H) Ki-67-positive cells were
counted to calculate the proliferation index. (I) TUNEL-positive
cells were counted to record the apoptosis index. (J) Liver weight
of the nude mice in 8 mg/day TQ-treated and control group. (K)
Spleen weight of the nude mice in 8 mg/day TQ-treated and control
group. *P<0.05, compared with control. TQ,
thymoquinone; CCA, cholangiocarcinoma. |
TQ inhibits PI3K/Akt activation in CCA
xenografts in nude mice
We next evaluated the expression of p-Akt in the
HuCCT-1 xenografts from the control and TQ-treated mice. The
expression of p-Akt and its downstream gene products was assessed
in the tumor tissues using western blot analysis, and the results
showed that TQ treatment resulted in a decrease in the expression
of p-Akt, XIAP and Bcl-2, as well as an increase in the expression
of BAX (Fig. 5E). These results
suggest that the PI3K/Akt signaling pathway is partially involved
in the effects of TQ on CCA cells in vivo.
TQ inhibits NF-κB and its downstream gene
products in nude mice
We investigated whether the antitumor effect of TQ
in mice was associated with the inhibition of NF-κB activation. The
EMSA results showed that TQ had an effect on NF-κB activation in
the tumor samples (Fig. 5F).
Accordingly, we observed the expression of downstream NF-κB target
genes (as mentioned above) in these tumors samples. As shown in
Fig. 5D, TQ treatment resulted in a
significant decrease in the expression of all these proteins in the
HuCCT-1 xenografts compared to the control treatment.
Discussion
CCA is a lethal cancer in Asia and throughout the
world. Patients often suffer from inoperable conditions when the
diagnosis is made at a late stage, and they require systemic
chemotherapy to combat the disease (25). However, no agent has been shown to
provide excellent benefits in controlled clinical trials, and the
treatment outcome remains poor due to the acquisition of drug
resistance and dose-limiting toxicities to normal cells. Emerging
evidence has shown that a loss of viability and the induction of
apoptotic cell death are two major mechanisms by which
chemotherapeutic agents kill cancer (26,27).
In the present study, the results showed that TQ potently inhibits
the proliferation of CCA cells and induces cell cycle arrest and
apoptosis in vitro and in vivo. Additionally, the key
finding from the present study is that TQ exhibits a
chemopreventive potential against human CCA cells by inhibiting the
constitutive activation of proinflammatory transcription factors,
including both PI3K/Akt and NF-κB, and their target genes that are
involved in tumor cell survival and proliferation, both of which
are associated with the pathogenesis and therapeutic resistance of
CCA.
The results of the present study showed that TQ
caused a G2/M phase cell cycle arrest in both TFK1 and HuCCT-1
cells in vitro and resulted in decreased expression of the
G2/M checkpoint protein cyclin B1. However, the cellular and
molecular bases of this phenomenon have not yet been clearly
defined. It is conceivable that TQ prevented the progression of the
cell cycle through G2/M phase, resulting in apoptosis. The results
indicate that the cell cycle arrest is likely one of the major
mechanisms of the anticancer activity of TQ.
With the goal of understanding how TQ inhibits cell
viability and induces apoptosis, we observed that TQ treatment may
effectively induce apoptosis in CCA by downregulating several
anti-apoptotic proteins. The PI3K/Akt signaling pathway is
associated with early carcinogenesis, and disruption of this
pathway results in antiproliferation, anti-survival,
anti-angiogenic and pro-apoptotic effects (28,29).
Upregulation of this pathway, through the phosphorylation of Akt,
plays a major role in carcinogenesis and drug resistance in
numerous types of cancer, including CCA. Notably, the activation of
Akt signaling has been observed in 40–50% of CCA tumors, indicating
the importance of Akt signaling in CCA (30). Thus, the PI3K/Akt pathway is an
attractive target in CCA (31).
However, increasing evidence shows that NF-κB activation is a
result of the underlying inflammation or is a consequence of the
formation of an inflammatory microenvironment during malignant
progression; thus, NF-κB provides a critical link between
inflammation and cancer (32).
Furthermore, previous reports have indicated that PI3K/Akt can
trigger NF-κB activation by enhancing the transcriptional activity
of the p65 subunit (33).
Meanwhile, emerging evidence also indicates that overexpression of
p-AKT, XIAP, Bcl-2, COX-2 and VEGF is associated with poor
prognosis. Additionally, the EMSA results showed that TQ
effectively inhibits the activation of the NF-κB pathway in the
TFK1 and HuCCT-1 cell lines at an estimated concentration of 40 μM.
Consistent with our hypothesis, we present important evidence
documenting a significant reduction in tumor size in vivo,
which is associated with the inhibition of the above anti-apoptotic
proteins. In the present study, a marked suppression of tumor
growth in mice xenografts was observed following TQ treatment.
Additionally, a conspicuous suppression of
proliferation was observed from the Ki-67 and TUNEL immunostaining,
which showed that there were an increasing number of apoptotic
cells in the TQ-treated animals. However, further studies are
needed to confirm and extend the present study to determine whether
TQ can be used as an effective therapy for CCA. These features are
of significant value in predicting the improved therapeutic outcome
and warrant further investigation. In particular, absorption and
analysis of the pharmacokinetic properties of TQ still need to be
investigated in future studies; however, the results of our
preliminary studies indicate that TQ exhibits low toxicity in the
liver and spleen and allowed the mice treated with TQ to maintain a
normal weight gain.
In conclusion, we have presented evidence that TQ
treatment results in the downregulation of anti-apoptotic and
pro-survival proteins that are transcriptionally regulated by the
PI3K/Akt and NF-κB pathways, resulting in a loss of CCA cell
survival and proliferation. Our in vitro findings, along
with the in vivo results, support the further development of
TQ as a novel therapeutic regimen for the targeted inactivation of
the PI3K/Akt and NF-κB pathways for the treatment of human CCA. The
combination of TQ and other conventional chemotherapeutic drugs may
produce a greater therapeutic effect, as well as reduce the
toxicity of the conventional chemotherapeutics. Further studies
should investigate these possibilities.
Acknowledgements
The authors thank Dr Xiang Fang for technical
assistance. The present study was supported in part by grants from
Heilongjiang Postdoctoral Foundation (LBH-Z11066), the China
Postdoctoral Science Foundation (nos. 2012M510990, 2012M520769 and
2013T60387), and the National Natural Scientific Foundation of
China (nos. 81100305 and 81270527).
References
|
1
|
Ramírez-Merino N, Aix SP and Cortés-Funes
H: Chemotherapy for cholangiocarcinoma: an update. World J
Gastrointest Oncol. 5:171–176. 2013.
|
|
2
|
Patel T: Cholangiocarcinoma. Nat Clin
Pract Gastroenterol Hepatol. 3:33–42. 2006. View Article : Google Scholar
|
|
3
|
Randi G, Malvezzi M, Levi F, et al:
Epidemiology of biliary tract cancers: an update. Ann Oncol.
20:146–159. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Shin HR, Oh JK, Masuyer E, et al:
Epidemiology of cholangiocarcinoma: an update focusing on risk
factors. Cancer Sci. 101:579–585. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tyson GL and El-Serag HB: Risk factors for
cholangiocarcinoma. Hepatology. 54:173–184. 2011. View Article : Google Scholar
|
|
6
|
de Groen PC, Gores GJ, LaRusso NF,
Gunderson LL and Nagorney DM: Biliary tract cancers. N Engl J Med.
341:1368–1378. 1999.
|
|
7
|
Hezel AF and Zhu AX: Systemic therapy for
biliary tract cancers. Oncologist. 13:415–423. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sirica AE: Cholangiocarcinoma: molecular
targeting strategies for chemoprevention and therapy. Hepatology.
41:5–15. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Blechacz B and Gores GJ:
Cholangiocarcinoma: advances in pathogenesis, diagnosis, and
treatment. Hepatology. 48:308–321. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Furuse J, Kasuga A, Takasu A, Kitamura H
and Nagashima F: Role of chemotherapy in treatments for biliary
tract cancer. J Hepatobiliary Pancreat Sci. 19:337–341. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gali-Muhtasib H, Roessner A and
Schneider-Stock R: Thymoquinone: a promising anti-cancer drug from
natural sources. Int J Biochem Cell Biol. 38:1249–1253.
2006.PubMed/NCBI
|
|
12
|
Yi T, Cho SG, Yi Z, et al: Thymoquinone
inhibits tumor angiogenesis and tumor growth through suppressing
AKT and extracellular signal-regulated kinase signaling pathways.
Mol Cancer Ther. 7:1789–1796. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Woo CC, Kumar AP, Sethi G and Tan KH:
Thymoquinone: potential cure for inflammatory disorders and cancer.
Biochem Pharmacol. 83:443–451. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Attoub S, Sperandio O, Raza H, et al:
Thymoquinone as an anticancer agent: evidence from inhibition of
cancer cells viability and invasion in vitro and tumor growth in
vivo. Fundam Clin Pharmacol. 27:557–569. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Banerjee S, Kaseb AO, Wang Z, et al:
Antitumor activity of gemcitabine and oxaliplatin is augmented by
thymoquinone in pancreatic cancer. Cancer Res. 69:5575–5583. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shen HM and Tergaonkar V: NFκB signaling
in carcinogenesis and as a potential molecular target for cancer
therapy. Apoptosis. 14:348–363. 2009.
|
|
17
|
Schmitz KJ, Lang H, Wohlschlaeger J, et
al: AKT and ERK1/2 signaling in intrahepatic cholangiocarcinoma.
World J Gastroenterol. 13:6470–6477. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Vivanco I and Sawyers CL: The
phosphatidylinositol 3-kinase AKT pathway in human cancer. Nature
Reviews Cancer. 2:489–501. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Seubwai W, Vaeteewoottacharn K, Hiyoshi M,
et al: Cepharanthine exerts antitumor activity on
cholangiocarcinoma by inhibiting NF-κB. Cancer Sci. 101:1590–1595.
2010.PubMed/NCBI
|
|
20
|
Kim SH, Song SH, Kim SG, et al: Celecoxib
induces apoptosis in cervical cancer cells independent of
cyclooxygenase using NF-κB as a possible target. J Cancer Res Clin
Oncol. 130:551–560. 2004.PubMed/NCBI
|
|
21
|
Jiang H, Ma Y, Chen X, et al: Genistein
synergizes with arsenic trioxide to suppress human hepatocellular
carcinoma. Cancer Sci. 101:975–983. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ma Y, Wang J, Liu L, et al: Genistein
potentiates the effect of arsenic trioxide against human
hepatocellular carcinoma: role of Akt and nuclear factor-κB. Cancer
Lett. 301:75–84. 2011.PubMed/NCBI
|
|
23
|
Liang Y, Zheng T, Song R, et al:
Hypoxia-mediated sorafenib resistance can be overcome by EF24
through Von Hippel-Lindau tumor suppressor-dependent HIF-1α
inhibition in hepatocellular carcinoma. Hepatology. 57:1847–1857.
2013.PubMed/NCBI
|
|
24
|
Sun X, Jiang H, Jiang X, et al: Antisense
hypoxia-inducible factor-1α augments transcatheter arterial
embolization in the treatment of hepatocellular carcinomas in rats.
Hum Gene Ther. 20:314–324. 2009.
|
|
25
|
Khan SA, Toledano MB and Taylor-Robinson
SD: Epidemiology, risk factors, and pathogenesis of
cholangiocarcinoma. HPB. 10:77–82. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Johnstone RW, Ruefli AA and Lowe SW:
Apoptosis: a link between cancer genetics and chemotherapy. Cell.
108:153–164. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tannock IF and Lee C: Evidence against
apoptosis as a major mechanism for reproductive cell death
following treatment of cell lines with anti-cancer drugs. Br J
Cancer. 84:100–105. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sheppard K, Kinross KM, Solomon B, Pearson
RB and Phillips WA: Targeting PI3 kinase/AKT/mTOR signaling in
cancer. Crit Rev Oncog. 17:69–95. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Samuels Y and Ericson K: Oncogenic PI3K
and its role in cancer. Curr Opin Oncol. 18:77–82. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Engelman JA: Targeting PI3K signalling in
cancer: opportunities, challenges and limitations. Nat Rev Cancer.
9:550–562. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ewald F, Grabinski N, Grottke A, et al:
Combined targeting of AKT and mTOR using MK-2206 and RAD001 is
synergistic in the treatment of cholangiocarcinoma. Int J Cancer.
133:2065–2076. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
DiDonato JA, Mercurio F and Karin M: NF-κB
and the link between inflammation and cancer. Immunol Rev.
246:379–400. 2012.
|
|
33
|
Arlt A, Gehrz A, Müerköster S, et al: Role
of NF-κB and Akt/PI3K in the resistance of pancreatic carcinoma
cell lines against gemcitabine-induced cell death. Oncogene.
22:3243–3251. 2003.
|