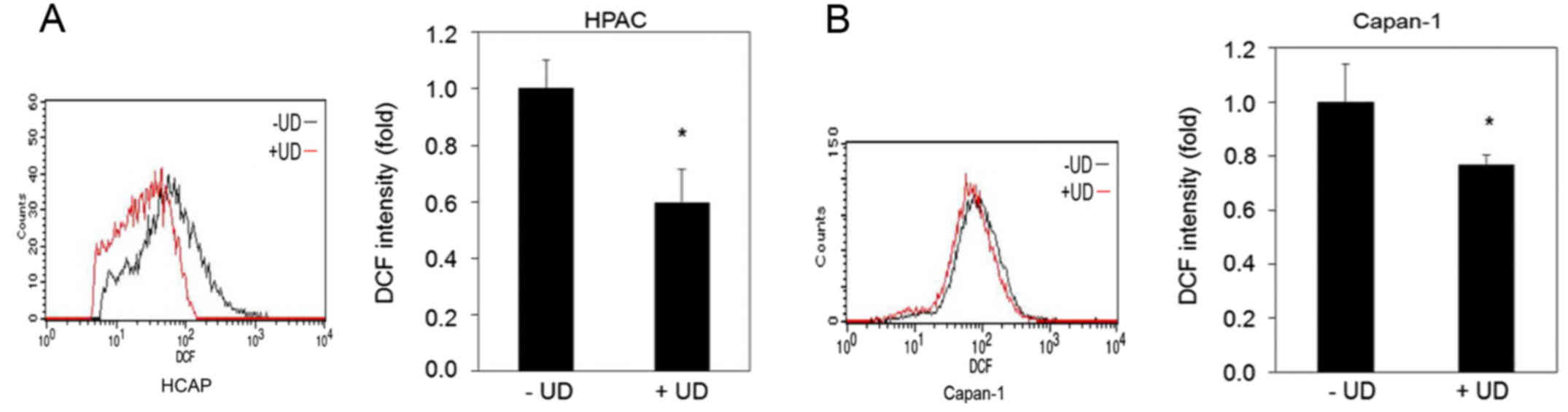
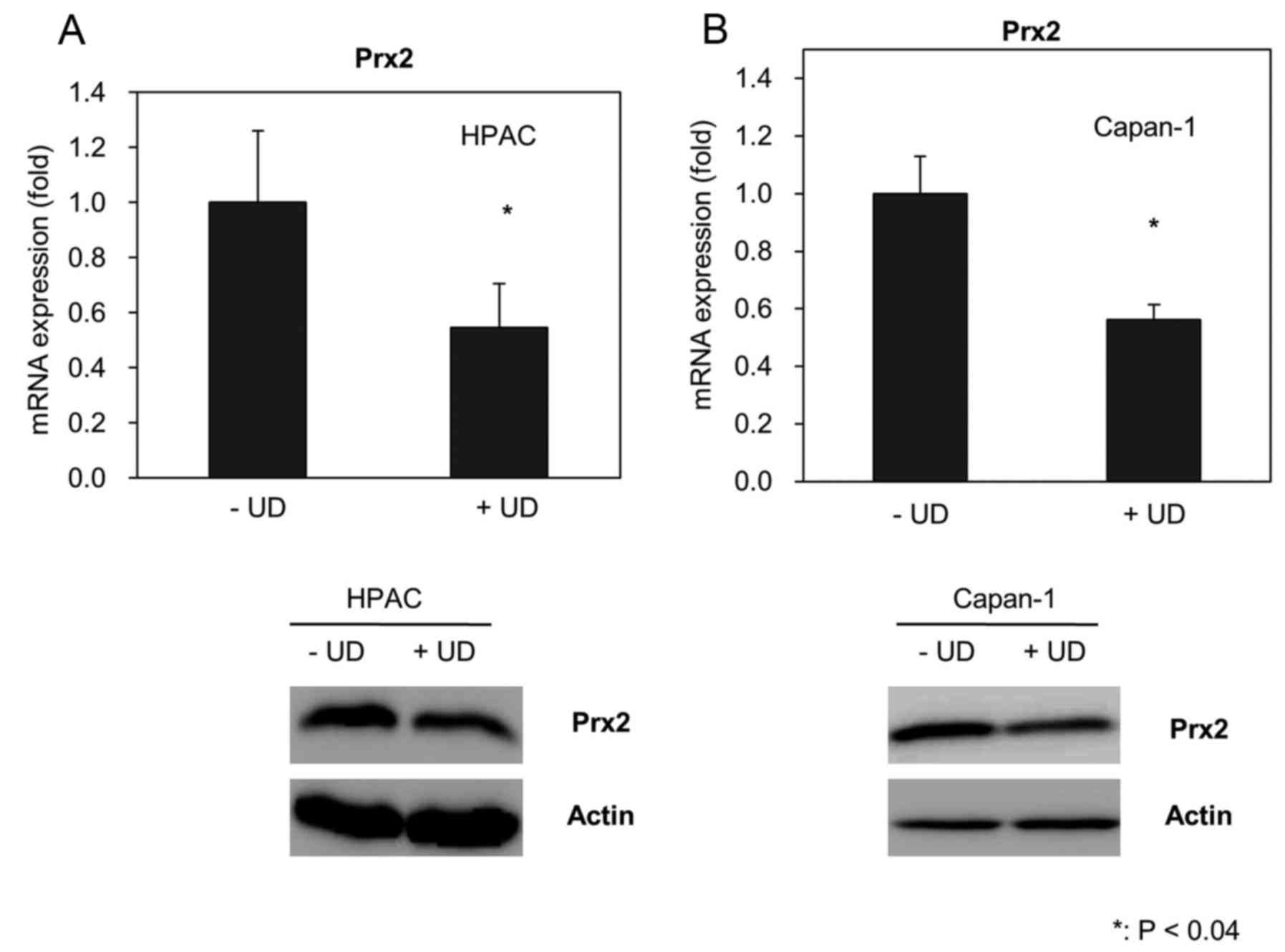
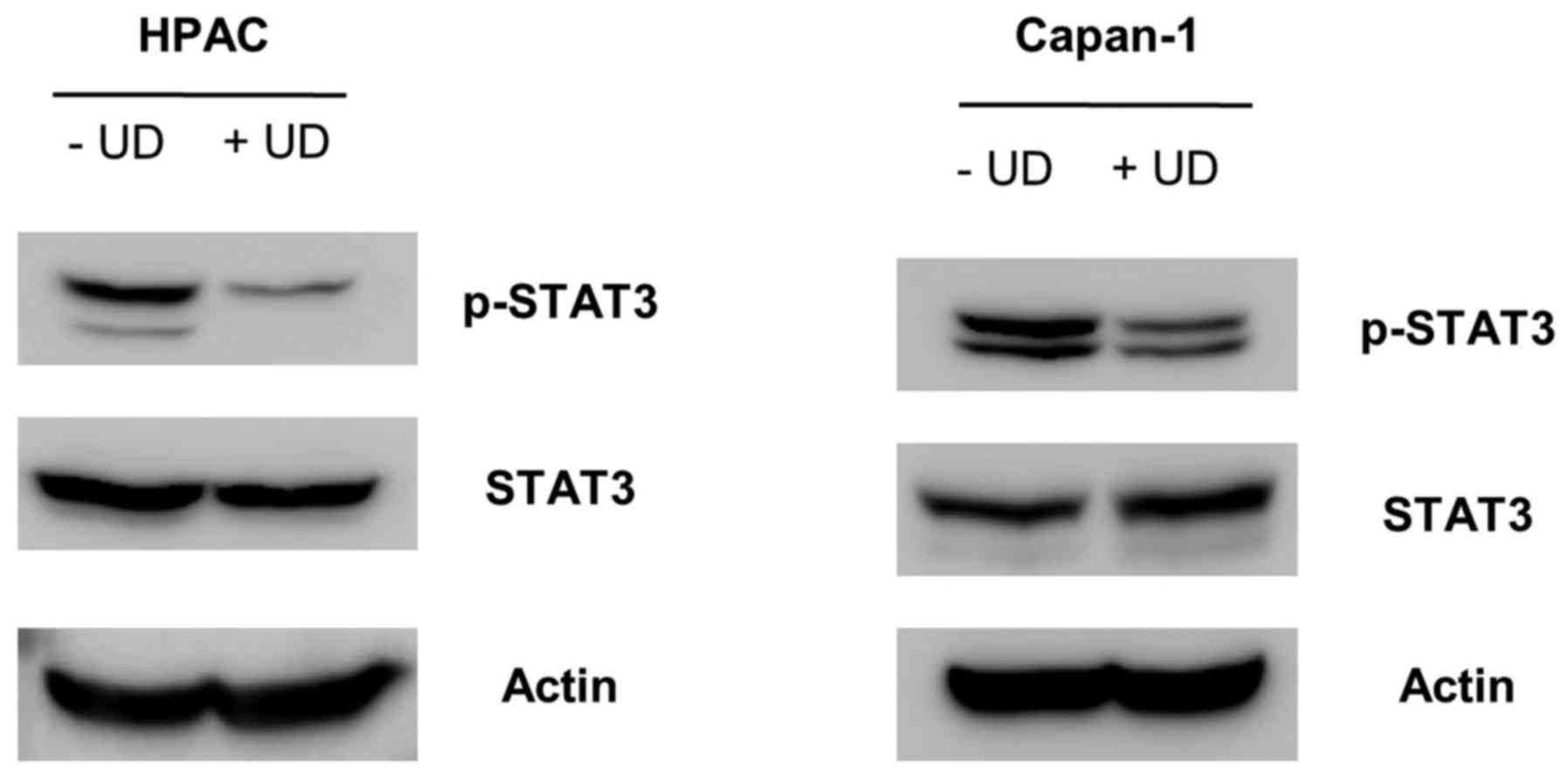
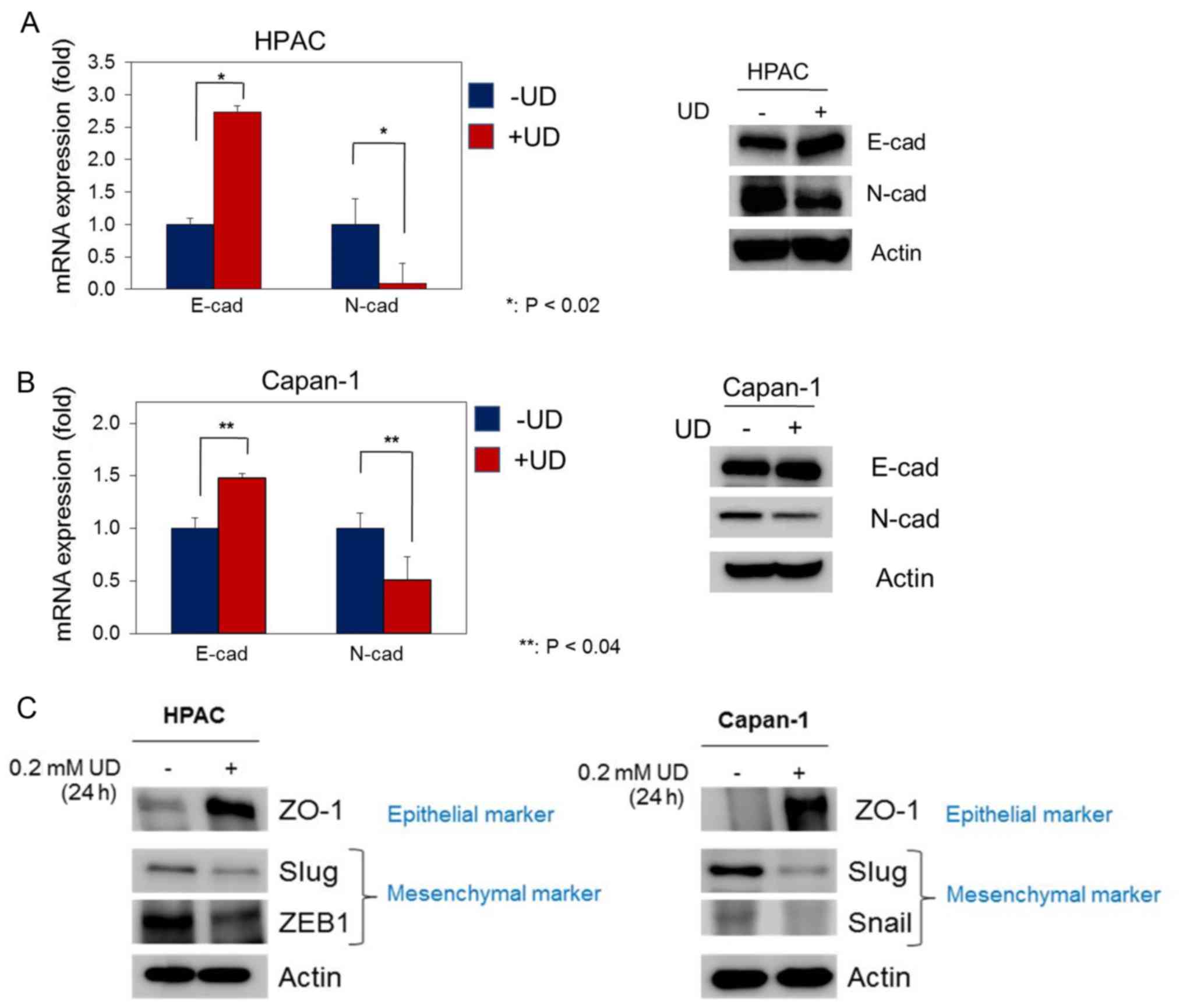
|
1
|
Knudsen ES, Balaji U, Mannakee B, Vail P,
Eslinger C, Moxom C, Mansour J and Witkiewicz AK: Pancreatic cancer
cell lines as patient-derived avatars: Genetic characterisation and
functional utility. Gut. Jan 10–2017.(Epub ahead of print). doi:
10.1136/gutjnl-2016-313133. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Stathis A and Moore MJ: Advanced
pancreatic carcinoma: Current treatment and future challenges. Nat
Rev Clin Oncol. 7:163–172. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kleger A, Perkhofer L and Seufferlein T:
Smarter drugs emerging in pancreatic cancer therapy. Ann Oncol.
25:1260–1270. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Feng W, Zhang B, Cai D and Zou X:
Therapeutic potential of histone deacetylase inhibitors in
pancreatic cancer. Cancer Lett. 347:183–190. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wong HH and Lemoine NR: Pancreatic cancer:
Molecular pathogenesis and new therapeutic targets. Nat Rev
Gastroenterol Hepatol. 6:412–422. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Trachootham D, Alexandre J and Huang P:
Targeting cancer cells by ROS-mediated mechanisms: A radical
therapeutic approach? Nat Rev Drug Discov. 8:579–591. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gupta A, Rosenberger SF and Bowden GT:
Increased ROS levels contribute to elevated transcription factor
and MAP kinase activities in malignantly progressed mouse
keratinocyte cell lines. Carcinogenesis. 20:2063–2073. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shi X, Zhang Y, Zheng J and Pan J:
Reactive oxygen species in cancer stem cells. Antioxid Redox
Signal. 16:1215–1228. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Diehn M, Cho RW, Lobo NA, Kalisky T, Dorie
MJ, Kulp AN, Qian D, Lam JS, Ailles LE, Wong M, et al: Association
of reactive oxygen species levels and radioresistance in cancer
stem cells. Nature. 458:780–783. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ishimoto T, Nagano O, Yae T, Tamada M,
Motohara T, Oshima H, Oshima M, Ikeda T, Asaba R, Yagi H, et al:
CD44 variant regulates redox status in cancer cells by stabilizing
the xCT subunit of system xc− and thereby promotes tumor
growth. Cancer Cell. 19:387–400. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sabharwal SS and Schumacker PT:
Mitochondrial ROS in cancer: Initiators, amplifiers or an Achilles'
heel? Nat Rev Cancer. 14:709–721. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Xu Y, Luo Q, Lin T, Zeng Z, Wang G, Zeng
D, Ding R, Sun C, Zhang XK and Chen H: U12, a UDCA derivative, acts
as an anti-hepatoma drug lead and inhibits the mTOR/S6K1 and
cyclin/CDK complex pathways. PLoS One. 9:e1134792014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wali RK, Stoiber D, Nguyen L, Hart J,
Sitrin MD, Brasitus T and Bissonnette M: Ursodeoxycholic acid
inhibits the initiation and postinitiation phases of
azoxymethane-induced colonic tumor development. Cancer Epidemiol
Biomarkers Prev. 11:1316–1321. 2002.PubMed/NCBI
|
|
15
|
Loddenkemper C, Keller S, Hanski ML, Cao
M, Jahreis G, Stein H, Zeitz M and Hanski C: Prevention of
colitis-associated carcinogenesis in a mouse model by diet
supplementation with ursodeoxycholic acid. Int J Cancer.
118:2750–2757. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Serfaty L: Chemoprevention of colorectal
cancer with ursodeoxycholic acid: Pro. Clin Res Hepatol
Gastroenterol. 36 Suppl 1:S53–S60. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
van Heumen BW, Roelofs HM, Morsche Te RH,
Marian B, Nagengast FM and Peters WH: Celecoxib and
tauro-ursodeoxycholic acid co-treatment inhibits cell growth in
familial adenomatous polyposis derived LT97 colon adenoma cells.
Exp Cell Res. 318:819–827. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Benz C, Angermüller S, Töx U,
Klöters-Plachky P, Riedel HD, Sauer P, Stremmel W and Stiehl A:
Effect of tauroursodeoxycholic acid on bile-acid-induced apoptosis
and cytolysis in rat hepatocytes. J Hepatol. 28:99–106. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rodrigues CM, Fan G, Ma X, Kren BT and
Steer CJ: A novel role for ursodeoxycholic acid in inhibiting
apoptosis by modulating mitochondrial membrane perturbation. J Clin
Invest. 101:2790–2799. 1998. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Benz C, Angermüller S, Otto G, Sauer P,
Stremmel W and Stiehl A: Effect of tauroursodeoxycholic acid on
bile acid-induced apoptosis in primary human hepatocytes. Eur J
Clin Invest. 30:203–209. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lindor K: Ursodeoxycholic acid for the
treatment of primary biliary cirrhosis. N Engl J Med.
357:1524–1529. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Paumgartner G and Beuers U:
Ursodeoxycholic acid in cholestatic liver disease: Mechanisms of
action and therapeutic use revisited. Hepatology. 36:525–531. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Siegel JL, Jorgensen R, Angulo P and
Lindor KD: Treatment with ursodeoxycholic acid is associated with
weight gain in patients with primary biliary cirrhosis. J Clin
Gastroenterol. 37:183–185. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Perez MJ and Briz O: Bile-acid-induced
cell injury and protection. World J Gastroenterol. 15:1677–1689.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ray PD, Huang BW and Tsuji Y: Reactive
oxygen species (ROS) homeostasis and redox regulation in cellular
signaling. Cell Signal. 24:981–990. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schieber M and Chandel NS: ROS function in
redox signaling and oxidative stress. Curr Biol. 24:R453–R462.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Rhee SG, Woo HA, Kil IS and Bae SH:
Peroxiredoxin functions as a peroxidase and a regulator and sensor
of local peroxides. J Biol Chem. 287:4403–4410. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Poljsak B, Šuput D and Milisav I:
Achieving the balance between ROS and antioxidants: When to use the
synthetic antioxidants. Oxid Med Cell Longev. 2013:9567922013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Espinosa-Diez C, Miguel V, Mennerich D,
Kietzmann T, Sánchez-Pérez P, Cadenas S and Lamas S: Antioxidant
responses and cellular adjustments to oxidative stress. Redox Biol.
6:183–197. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wood ZA, Schröder E, Harris Robin J and
Poole LB: Structure, mechanism and regulation of peroxiredoxins.
Trends Biochem Sci. 28:32–40. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cerda MB, Lloyd R, Batalla M, Giannoni F,
Casal M and Policastro L: Silencing peroxiredoxin-2 sensitizes
human colorectal cancer cells to ionizing radiation and
oxaliplatin. Cancer Lett. 388:312–319. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Latimer HR and Veal EA: Peroxiredoxins in
regulation of MAPK signalling pathways; sensors and barriers to
signal transduction. Mol Cells. 39:40–45. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Moon JC, Hah YS, Kim WY, Jung BG, Jang HH,
Lee JR, Kim SY, Lee YM, Jeon MG, Kim CW, et al: Oxidative
stress-dependent structural and functional switching of a human
2-Cys peroxiredoxin isotype II that enhances HeLa cell resistance
to H2O2-induced cell death. J Biol Chem.
280:28775–28784. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Qu D, Rashidian J, Mount MP, Aleyasin H,
Parsanejad M, Lira A, Haque E, Zhang Y, Callaghan S, Daigle M, et
al: Role of Cdk5-mediated phosphorylation of Prx2 in MPTP toxicity
and Parkinson's disease. Neuron. 55:37–52. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Park H, Noh AL, Kang JH, Sim JS, Lee DS
and Yim M: Peroxiredoxin II negatively regulates
lipopolysaccharide-induced osteoclast formation and bone loss via
JNK and STAT3. Antioxid Redox Signal. 22:63–77. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sobotta MC, Liou W, Stöcker S, Talwar D,
Oehler M, Ruppert T, Scharf AN and Dick TP: Peroxiredoxin-2 and
STAT3 form a redox relay for H2O2 signaling.
Nat Chem Biol. 11:64–70. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kwon T, Bak Y, Park YH, Jang GB, Nam JS,
Yoo JE, Park YN, Bak IS, Kim JM, Yoon DY, et al: Peroxiredoxin II
is essential for maintaining stemness by redox regulation in liver
cancer cells. Stem Cells. 34:1188–1197. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hawkins K, Mohamet L, Ritson S, Merry CL
and Ward CM: E-cadherin and, in its absence, N-cadherin promotes
Nanog expression in mouse embryonic stem cells via STAT3
phosphorylation. Stem Cells. 30:1842–1851. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Xiong H, Hong J, Du W, Lin YW, Ren LL,
Wang YC, Su WY, Wang JL, Cui Y, Wang ZH, et al: Roles of STAT3 and
ZEB1 proteins in E-cadherin down-regulation and human colorectal
cancer epithelial-mesenchymal transition. J Biol Chem.
287:5819–5832. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhang C, Guo F, Xu G, Ma J and Shao F:
STAT3 cooperates with Twist to mediate epithelial-mesenchymal
transition in human hepatocellular carcinoma cells. Oncol Rep.
33:1872–1882. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Foshay KM and Gallicano GI: Regulation of
Sox2 by STAT3 initiates commitment to the neural precursor cell
fate. Stem Cells Dev. 17:269–278. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gao H, Teng C, Huang W, Peng J and Wang C:
SOX2 Promotes the epithelial to mesenchymal transition of
esophageal squamous cells by modulating slug expression through the
activation of STAT3/HIF-α Signaling. Int J Mol Sci. 16:21643–21657.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang H, Cai HB, Chen LL, Zhao WJ, Li P,
Wang ZQ and Li Z: STAT3 correlates with stem cell-related
transcription factors in cervical cancer. J Huazhong Univ Sci
Technolog Med Sci. 35:891–897. 2015. View Article : Google Scholar : PubMed/NCBI
|