Introduction
Acute lung injury (ALI) is characterized by high
morbidity and mortality. Neonates are prone to ALI, particularly
when they have infection, hypoxia or shock (1). The primary clinical manifestations of
ALI are progressive hypoxemia and respiratory distress. ALI may
lead to multiple organ dysfunction syndrome, multiple system organ
failure and other life-threatening complications (2). Acute respiratory distress syndrome
(ARDS) is a severe form of ALI (3).
ARDS accounts for approximately 1-4% of all admissions to pediatric
intensive care units, and 8-10% of patients with ARDS require
mechanical ventilation worldwide. Despite progress in management of
ARDS in pediatric intensive care units, ARDS still has a high
mortality of 20-75% (4) worldwide.
Currently, there is no specific method in clinical practice for the
treatment of ALI, though it is usually treated using mechanical
ventilation and treatment methods for secondary organ injury and
primary underlying diseases, and prognosis in patients is poor
(5). Therefore, development of a
more effective and safe treatment would be highly beneficial for
children with ALI.
Intravenous immunoglobulin (IVIG) is a plasma
product extracted from healthy human blood, with abundant
immunoglobulin G (IgG) antibodies, which has an immunosuppressive
and anti-inflammatory effect. It has been widely used in the
treatment of various autoimmune and inflammatory diseases (6,7), such
as Kawasaki disease, sepsis and viral encephalitis (8-10).
However, few studies have been conducted on the application of IVIG
in neonates with ALI (11,12). Currently, the pathogenesis of ALI is
under investigation. An inflammatory cascade reaction triggered by
lung injury can activate a variety of inflammatory cells to release
a large number of inflammatory cytokines, including interleukin-6
(IL-6) and tumor necrosis factor-α (TNF-α). These inflammatory
cytokines may damage pulmonary alveoli and pulmonary capillary
structure by binding with receptors on endothelial cells in lung
capillaries and alveolar epithelial cells. Therefore, a number of
studies have suggested that inflammatory responses play an
important role and are involved in the occurrence and development
of ALI (13-15).
Currently, research into the treatment of ALI in
neonates mainly focuses on mechanical ventilation and pulmonary
surfactants and few studies have reported treatment with IVIG
(16). Therefore, the present study
was designed to explore the efficacy of IVIG and its effects on
serum inflammatory cytokines in neonates with ALI.
Materials and methods
Research subjects
The research subjects were 140 neonates with ALI who
were admitted to the Neonatal Intensive Care Unit of Longhua
People's Hospital (Shenzhen, China) between May 2016 and December
2018. Subjects were randomly assigned to the control group (COG) or
the study group (STG), with 70 patients in each group. After 30
days' treatment, there were 60 patients alive in the COG and 66
patients alive in STG. The inclusion criteria were that all
patients met the diagnostic criteria for ALI (17). Patients with congenital heart
disease or congenital lung development defects, patients whose
families disagreed to their participation or whose etiology was
unclear, and patients with a length of labor >1 day were
excluded. The parents or guardians of the patients signed the
informed consent forms after understanding the study, and the
experiment was approved by the Medical Ethics Committee of Longhua
People's Hospital (Shenzhen, China; approval no. 20180713).
Treatment methods
The patients all underwent chest X-ray, oxygenation
index and oxygen partial pressure examinations. Patients in the COG
were treated with the following routine treatment scheme: At 0, 12,
24 and 36 h, each of the patients was hooked up to a ventilator in
pressure support ventilation and synchronized intermittent
mandatory ventilation mode [exhaled tidal volume, 6 ml/kg; arterial
partial pressure of oxygen (PaO2), >8.24 kPa;
arterial carbon dioxide partial pressure (PaCO2),
<5.34 kPa), and they were also provided water and treatment to
ensure the same electrolyte balance, anti-infection and nutritional
support at the same time. Patients in the STG were treated with
IVIG (cat. no. S10980061; Shanghai RAAS Blood Products Co., Ltd.)
in addition to the treatment used for the COG. IVIG was
administered by intravenous drip at 1 g/kg weight /day for 2 days
consecutively. Standard treatment was administered for the
subsequent 5 days. Both groups were treated for 7 days.
Observation indices
The PaO2 and fraction of inspired oxygen
(FIO2) at 1 h before treatment and the mechanical
ventilation status and hospitalization time of patients in the two
groups were recorded and their 30-day survival rates were
analyzed.
Determination of inflammatory cytokine
levels
Venous blood from the elbow of the arm (3 ml) was
sampled from each patient in the two groups at 1 h before and at
12, 24 and 36 h after treatment, respectively. The clotted sampled
blood was centrifuged at 300 x g to extract the supernatant, which
was stored in a refrigerator at -20˚C for subsequent analysis.
ELISA was used to determine the serum IL-6 and TNF-α
levels of patients from the two groups in accordance with the
manufacturer's instructions [human IL-6 ELISA (cat. no. KIT10395A;
Sino Biological Inc.) and human TNF-α ELISA (cat. no. FK-R0122;
Shanghai FKBIO Co., Ltd.)]. A well for samples to be determined, a
standard well and a blank well were set. No enzyme-labeled reagent
or sample was added into the blank well, and 100 µl of samples and
100 µl of standards were added into the well for samples to be
determined and standard well, respectively, and mixed well. Both
samples to be determined and standard wells were covered with a
film and incubated at 37˚C for 2 h. The liquid was discarded and
the plate was patted dry, and 100 µl each of working fluid A and
fluid B was added. The optical density of each well at a wavelength
of 450 nm was measured using an enzyme mark instrument (Wuhan USCN
Business Co., Ltd.), and the cytokine concentration of each sample
was calculated.
Statistical analysis
The data were statistically analyzed using SPSS 21.0
(IBM Corp.) and visualized in figures using GraphPad Prism 7
(GraphPad Software, Inc.). Patient numbers are presented as [n(%)],
and differences in rates were analyzed using χ2 test.
Measurement data are presented as the mean ± SD. Measurement data
between two groups were analyzed using an independent samples
Student's t-test. Two-way ANOVA was employed to analyze the data in
Fig. 1. Comparison between the
groups was performed by analyzing the data with Bonferroni's post
hoc test.
Repeated measures data were analyzed using a
repeated measures ANOVA with Bonferroni's post hoc test. The
Kaplan-Meier method was used to draw survival curves of the
patients and log-rank test was adopted for analysis. P<0.05 was
considered to indicate a statistically significant difference.
Results
Comparison of general data between the
two groups
The COG consisted of 40 male and 30 female neonates,
with a mean age of 7.92±3.36 h, a mean fetal age of 36.23±2.21
gestational weeks and a mean weight of 2.79±0.33 kg. In terms of
primary diseases, the COG had 29 patients with asphyxia, 18 with
pneumonia, 13 with septicemia and 10 with meconium aspiration
syndrome. In terms of delivery mode, 31 patients were delivered
through vaginal and 39 through cesarean delivery. The STG consisted
of 32 male and 38 female neonates, with a mean average age of
8.64±2.88 h, a mean fetal age of 35.81±2.68 gestational weeks and a
mean weight of 2.87±0.41 kg. In terms of primary diseases, the COG
had 21 patients with asphyxia, 25 with pneumonia, 9 with septicemia
and 15 with meconium aspiration syndrome. In terms of delivery
mode, 26 patients were delivered through vaginal delivery and 44
through cesarean. General data, including sex, age, fetal age,
weight, primary diseases and delivery mode were not significantly
different between the two groups (all P>0.05; Table I).
 | Table IComparison of general data between the
two groups. |
Table I
Comparison of general data between the
two groups.
| Characteristic | Control group
(n=70) | Study group
(n=70) |
χ2/t-statistic | P-value |
|---|
| Sex | | | 1.830 | 0.176 |
|
Male | 40 (57.14) | 32 (45.71) | | |
|
Female | 30 (42.86) | 38 (54.29) | | |
| Mean age, days | 0.33±0.14 | 0.36±0.12 | 1.361 | 0.176 |
| Fetal age,
weeks | 36.23±2.21 | 35.81±2.68 | 1.012 | 0.314 |
| Average weight,
kg | 2.79±0.33 | 2.87±0.41 | 1.478 | 0.142 |
| Primary
diseases | | | 2.935 | 0.231 |
|
Asphyxia | 29 (41.43) | 21 (30.00) | | |
|
Pneumonia | 18 (25.71) | 25 (35.71) | | |
|
Septicemia | 13 (18.57) | 9 (12.86) | | |
|
Meconium
aspiration syndrome | 10 (14.29) | 15 (21.43) | | |
| Delivery mode | | | 0.740 | 0.390 |
|
Vaginal
delivery | 31 (44.29) | 26 (37.14) | | |
|
Cesarean
delivery | 39 (55.71) | 44 (62.86) | | |
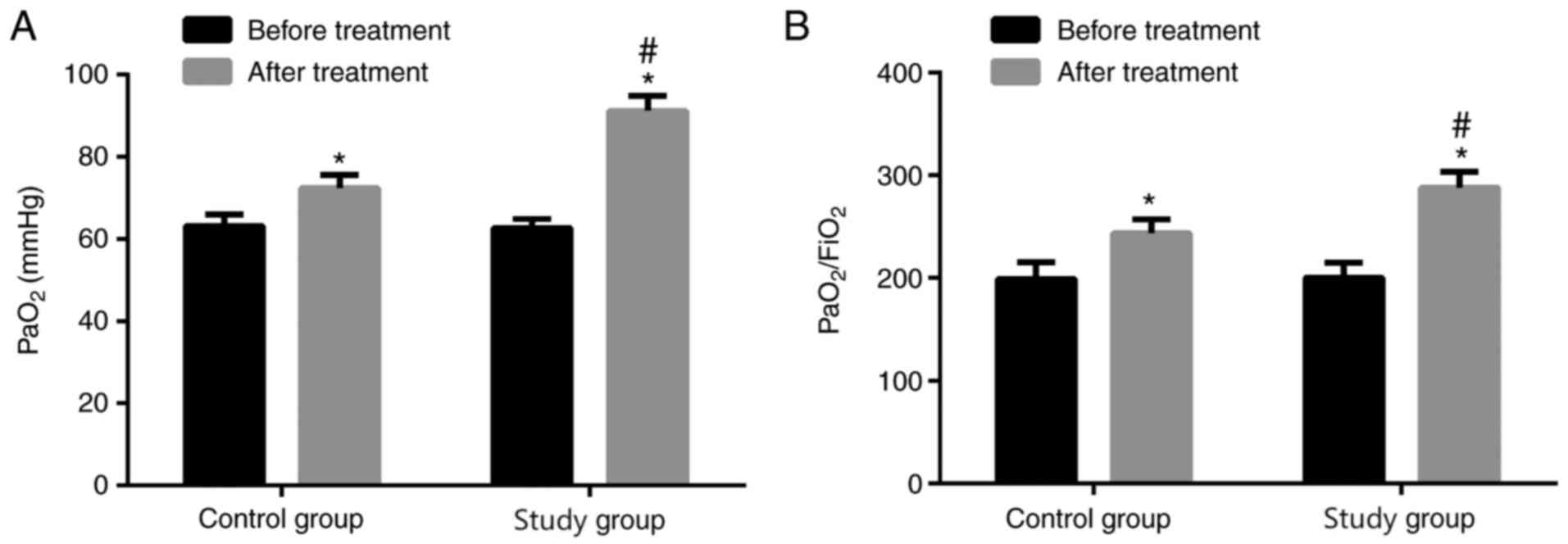
Comparison of PaO2 and
PaO2/FIO2 before and after treatment between
the two groups
PaO2 and PaO2/FIO2
were not significantly different before treatment between the two
groups (P>0.05), whereas PaO2 and
PaO2/FIO2 significantly increased after
treatment (both P<0.05). PaO2 and
PaO2/FIO2 in the STG were higher than those
in the COG (both P<0.05; Fig.
1).
Comparison of mechanical ventilation
and hospitalization time between the two groups
The mechanical ventilation and hospitalization times
of the STG patients were 54.53±10.17 h and 17.46±3.76 days, and of
the COG patients 75.45±13.30 h and 3.64±4.27 days. Thus, the STG
patients experienced significantly shorter durations of mechanical
ventilation and hospitalization time than the COG patients (both
P<0.001; Table II).
 | Table IIComparison of mechanical ventilation
and hospitalization time between the two groups. |
Table II
Comparison of mechanical ventilation
and hospitalization time between the two groups.
| Group | Mechanical
ventilation time, h | t-statistic | P-value | Hospitalization
time, days | t-statistic | P-value |
|---|
| Control group
(n=70) | 75.45±13.30 | 10.454 | <0.001 | 23.64±4.27 | 9.088 | <0.001 |
| Study group
(n=70) | 54.53±10.17 | | | 17.46±3.76 | | |
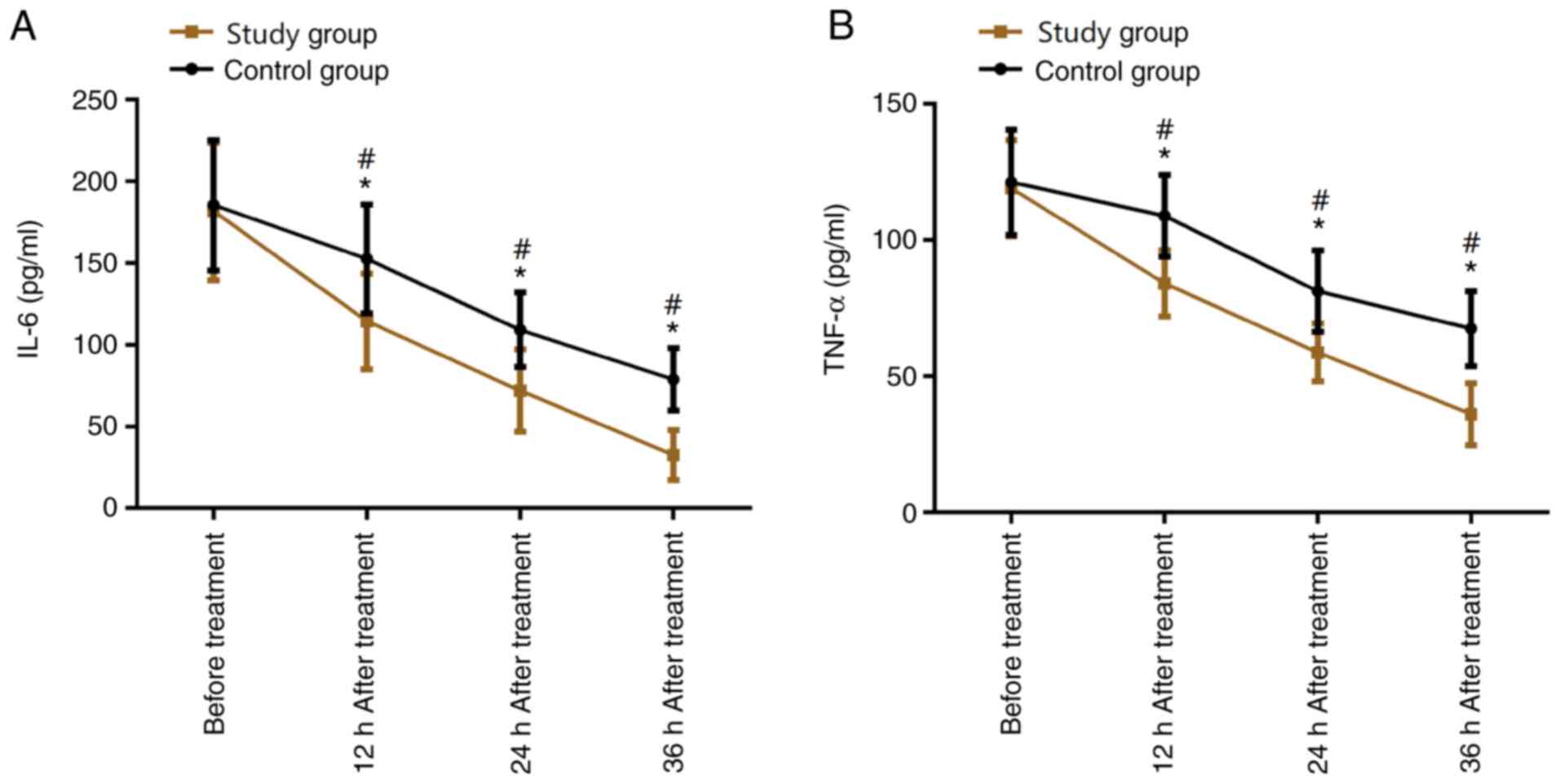
Comparison of serum IL-6 and TNF-α
levels before and after treatment between the two groups
The serum IL-6 and TNF-α levels at 12, 24 and 36 h
after treatment in the two groups were significantly lower than
those before treatment (all P<0.05). The serum IL-6 and TNF-α
levels at 24 and 36 h after treatment were lower than those at 12 h
after treatment (both P<0.05). The serum IL-6 and TNF-α levels
at 36 h after treatment were lower than those at 24 h after
treatment (both P<0.05). The serum IL-6 and TNF-α levels before
treatment were not significantly different between the two groups
(both P>0.05), whereas the serum IL-6 and TNF-α levels at 12, 24
and 36 h after treatment in the STG were significantly lower than
those in the COG (P<0.05; Fig.
2).
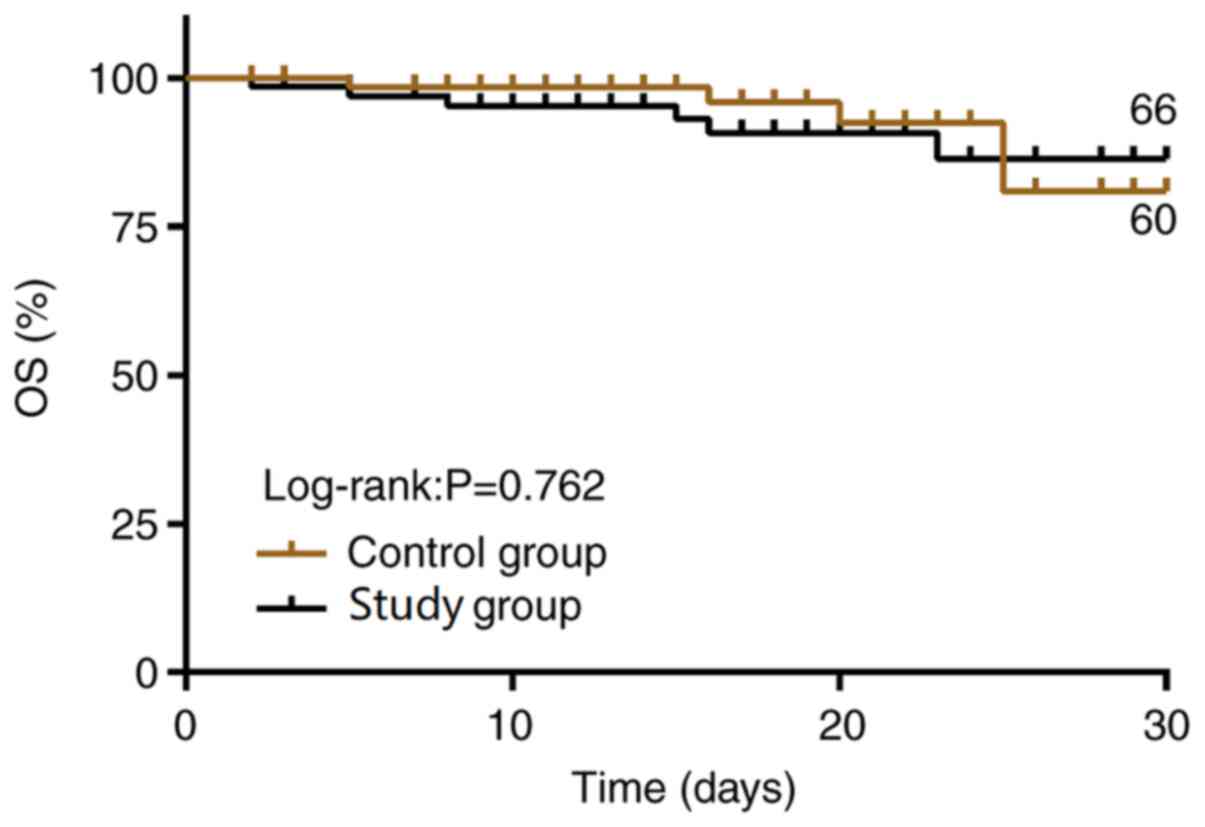
Comparison of 30-day survival rate
after treatment between the two groups
The follow-up results showed that the 30-day
survival rate was not different between the COG and STG patients
[94.28% (66/70) vs. 85.71% (60/70); P>0.05; Fig. 3].
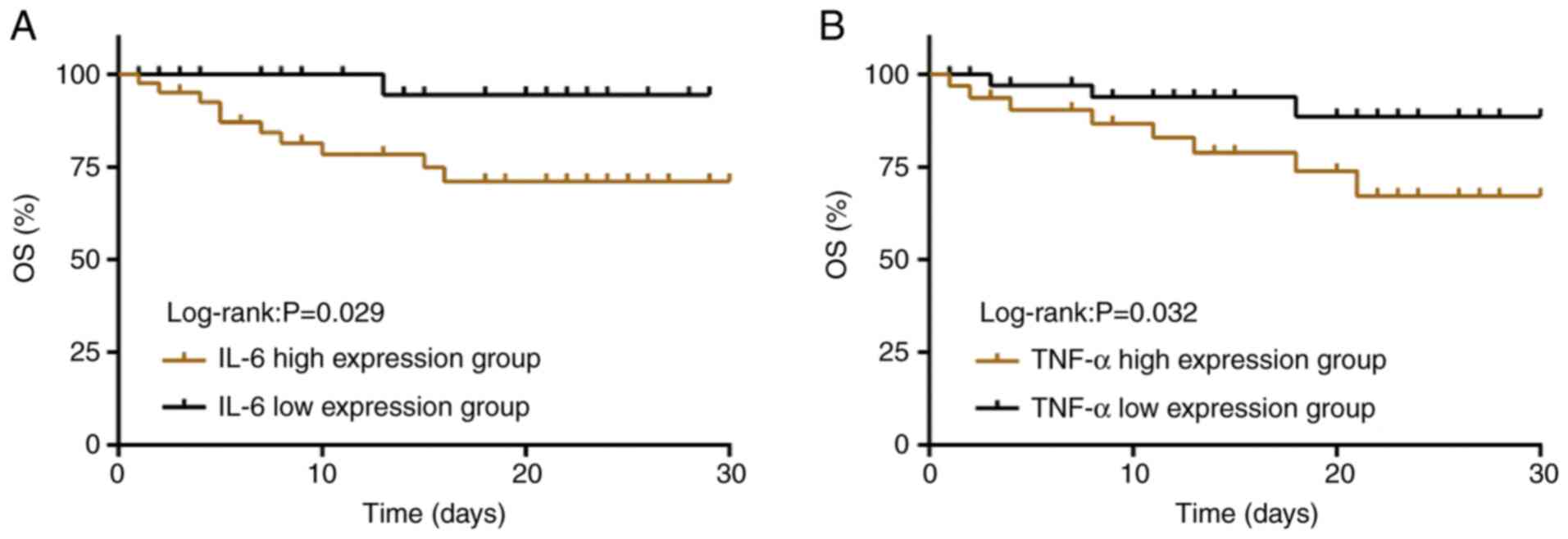
Effects of IL-6 and TNF-α levels on
the survival rate of patients
The patients in the COG were divided into high
(n=41; expression ≥185.45 pg/ml) and low (n=29; expression
<185.45 pg/ml) IL-6 expression groups based on their mean serum
IL-6 level before treatment. They were also divided into high
(n=32; expression ≥121.13 pg/ml) and low (n=38; expression
<121.13 pg/ml) TNF-α expression groups based on their mean serum
TNF-α level before treatment. The 30-day survival rate of the high
IL-6 expression group was significantly lower than that in the low
IL-6 expression group [68.29% (28/41) vs. 96.55% (28/29);
P<0.05] and that in the high TNF-α expression group was also
lower than that in the low TNF-α expression group [65.63% (21/32)
vs. 92.11% (35/38); P<0.05; Fig.
4].
Discussion
The pathological basis of ALI is that damaged lung
capillary endothelial cells and alveolar epithelial cells cause
edema in pulmonary alveoli and the pulmonary interstitium, leading
to acute hypoxic respiratory insufficiency or failure and
pathophysiological characteristics, such as lung volume reduction,
lung compliance decline and severe ventilation/blood flow
disproportion (18). ALI is usually
treated with respiratory support measures in clinical practice. In
addition, with the continuous progress of studies on ALI, most
researchers support the hypothesis that anti-inflammatory treatment
can be beneficially applied in ALI (19,20).
Clinically, nonspecific anti-inflammatory treatment is administered
with use of glucocorticoid, immunoglobulin and non-steroidal
anti-inflammatory preparations (21). However, glucocorticoid and
non-steroidal anti-inflammatory preparations can lead to various
noxious toxic side effects when used in neonates; thus, they are
not suitable for the treatment of ALI in this group of patients
(21). Therefore, the present study
adopted IVIG combined with mechanical ventilation to treat neonates
with ALI.
With the ability to regulate macrophage activity,
IVIG not only can inhibit macrophage overactivation but can also
suppress T lymphocyte and natural killer cell activity, thus
lowering autoimmune reactions (22). In addition, IVIG contains a large
number of specific antibodies that can regulate synthesis and
release of inflammatory cytokines; hence, it has a strong
anti-inflammatory effect (23,24).
The efficacy of respiratory support is judged on monitored
respiratory mechanics. PaO2 can directly reflect
hypoxemia and its severity, and PaO2/FIO2 can
reflect the extent of damage to pulmonary vessels and alveoli
(25). Therefore, the present study
selected PaO2 and PaO2/FIO2 as
evaluation indices for pulmonary function of neonates with ALI. In
addition to measures of mechanical ventilation and PS, the present
study additionally used IVIG through intravenous injection to
neonates with ALI. Subsequently, PaO2 and
PaO2/FIO2 in the STG were significantly
higher than those in the COG patients, and the STG patients
experienced significantly shorter mechanical ventilation and
hospitalization times than the COG patients, suggesting that
mechanical ventilation and intravenous injection of IVIG could
improve pulmonary gas exchange in neonates with ALI. Subsequently,
the 30-day survival rate of neonates with ALI in the two groups was
analyzed, and no significant differences were found between the two
groups, indicating that though mechanical ventilation and
intravenous injection of IVIG appeared to improve pulmonary gas
exchange in neonates with ALI it had no effect on their short-term
survival rate.
ALI is a stage of systemic inflammatory response
syndrome and an earlier study indicated that the inflammatory
response is one of the main reasons for progression of ALI into
ARDS (26). Due to ALI,
inflammatory cells are activated, and these cells release a large
number of pro-inflammatory cytokines, causing cell metabolism
dysfunction and aggregation of a large number of neutrophil cells
to inflammatory sites, where they then release a large number of
oxygen free radicals, causing oxidative stress injuries (27,28).
IL-6, a pro-inflammatory and immunomodulatory cytokine, can help
the host defend against infection and tissue damage, and its
abnormal expression in the human body can lead to various diseases,
such as dyslipidemia, hyperinsulinemia, diabetes, hypertension and
cardiovascular diseases (29).
TNF-α is a protein produced by activated macrophages and monocytes,
which participates in the human inflammatory and immune response,
and is crucial in maintaining homeostasis (30). Lowering TNF-α levels or blocking
TNF-α binding to its receptors can alleviate inflammatory injury
(31). A study by Chu et al
(32) showed that TNF-α and IL-6
were highly expressed in ALI mouse models and that their expression
levels decreased after treatment. A study by Wang et al
(33) indicated that children with
ALI treated with probiotics had significantly decreased serum IL-6
and TNF-α levels than control patients, which were negatively
correlated with pulmonary artery pressure. The results of the
present study showed that in both groups there was a significant
decrease in serum IL-6 and TNF-α levels after treatment when
compared with pre-treatment levels, and the STG showed a more
significant decrease, similarly to the above cited study results
(33), suggesting that IVIG can
effectively reduce inflammatory response in neonates with ALI. If
large doses of IVIG are used frequently, IVIG may inhibit the
production of immunoglobulins in autoimmune cells. However, IVIG is
an allergen to the human body and excessive use can cause allergic
reactions (34).
In the present study, the mean serum IL-6 and TNF-α
levels in patients in the COG before treatment were taken into
consideration to analyze the effects of their high and low
expression levels on the survival rate. Patients with high
expression levels of serum IL-6 and TNF-α showed a significantly
lower 30-day survival rate than those with low expressions,
suggesting that the probable survival of neonates with ALI can be
evaluated by determining their serum IL-6 and TNF-α
expressions.
The research subjects in this study were selected in
strict accordance with inclusion and exclusion criteria, and no
significant differences were found between the two groups in
general data, including sex, age, fetal age, weight, primary
diseases, and delivery mode, which reduced effects of other factors
on the study results and ensured the preciseness of the study.
However, this study has some limitations. The survival rate of
patients was only recorded for 30 days after treatment and
long-term follow-up to understand their long-term survival rate was
not performed. In addition, the mechanism of action of IVIG in
treating neonates with ALI was neither thoroughly or
comprehensively explored as it was not possible to assess the
optimal dosage, treatment course and applicable conditions of IVIG.
Inflammatory chemokines, such as monocyte chemoattractant protein-1
(MCP-1) are also important in ALI (35) and were not assessed in the present
study. These limitations will be addressed in the future.
In summary, IVIG treatment may improve the pulmonary
gas exchange of neonates with ALI, shorten their course of disease
and reduce the inflammatory response. However, few studies have
reported on IVIG as treatment for neonates with ALI and therefore
more research is needed.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported by a grant from the Key Project
of Science and Technology Innovation Bureau of Futian District,
Shenzhen, 2019 (grant no. FTWS2019004).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SW and ZW designed the study, SW wrote the draft and
ZW reviewed the manuscript. ZT, XZ and JD analyzed the data and
performed the statistical analysis. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
The parents or guardians of patients signed informed
consent forms after understanding the study, and the experiment was
approved by the Medical Ethics Committee of Longhua People's
Hospital (Shenzhen, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Li Y, Wu R, Tian Y, Yu M, Tang Y, Cheng H
and Tian Z: RAGE/NF-κB signaling mediates lipopolysaccharide
induced acute lung injury in neonate rat model. Int J Clin Exp Med.
8:13371–13376. 2015.PubMed/NCBI
|
|
2
|
Jin Z, Chun Suen K and Ma D: Perioperative
‘remote’ acute lung injury: Recent update. J Biomed Res.
31:197–212. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Rajasekaran S, Pattarayan D, Rajaguru P,
Sudhakar Gandhi PS and Thimmulappa RK: MicroRNA regulation of acute
lung injury and acute respiratory distress syndrome. J Cell
Physiol. 231:2097–2106. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gupta S, Sankar J, Lodha R and Kabra SK:
Comparison of prevalence and outcomes of pediatric acute
respiratory distress syndrome using Pediatric Acute Lung Injury
Consensus Conference criteria and Berlin definition. Front Pediatr.
6(93)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Perl M, Chung C-S, Perl U, Lomas-Neira J,
de Paepe M, Cioffi WG and Ayala A: Fas-induced pulmonary apoptosis
and inflammation during indirect acute lung injury. Am J Respir
Crit Care Med. 176:591–601. 2007.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Perez EE, Orange JS, Bonilla F, Chinen J,
Chinn IK, Dorsey M, El-Gamal Y, Harville TO, Hossny E, Mazer B, et
al: Update on the use of immunoglobulin in human disease: A review
of evidence. J Allergy Clin Immunol. 139:S1–S46. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang Y and Scott DE: Development of a
complement-mediated hemolysis assay for Immune globulin intravenous
products and characterization of lots associated with clinical
hemolysis. 2017.
|
|
8
|
Shulman ST: Intravenous immunoglobulin for
the treatment of Kawasaki disease. Pediatr Ann. 46:e25–e28.
2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tagami T, Matsui H, Fushimi K and Yasunaga
H: Intravenous immunoglobulin use in septic shock patients after
emergency laparotomy. J Infect. 71:158–166. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Rayamajhi A, Nightingale S, Bhatta NK,
Singh R, Kneen R, Ledger E, Bista KP, Lewthwaite P, Mahaseth C,
Turtle L, et al: A preliminary randomized double blind
placebo-controlled trial of intravenous immunoglobulin for Japanese
encephalitis in Nepal. PLoS One. 10(e0122608)2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Fakhari Z, Farsaei S and Sabzghabaee AM:
Predicting factors for the pattern of intravenous immunoglobulin
utilization in a Middle Eastern University Hospital. J Res Pharm
Pract. 7:188–194. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hartung HP, Mouthon L, Ahmed R, Jordan S,
Laupland KB and Jolles S: Clinical applications of intravenous
immunoglobulins (IVIg) - beyond immunodeficiencies and neurology.
Clin Exp Immunol. 158 (Suppl 1):23–33. 2009.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Jing W, Chunhua M and Shumin W: Effects of
acteoside on lipopolysaccharide-induced inflammation in acute lung
injury via regulation of NF-κB pathway in vivo and in vitro.
Toxicol Appl Pharmacol. 285:128–135. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Jiang W, Luo F, Lu Q, Liu J, Li P, Wang X,
Fu Y, Hao K, Yan T and Ding X: The protective effect of Trillin
LPS-induced acute lung injury by the regulations of inflammation
and oxidative state. Chem Biol Interact. 243:127–134.
2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Mokra D and Kosutova P: Biomarkers in
acute lung injury. Respir Physiol Neurobiol. 209:52–58.
2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Altirkawi K: Surfactant therapy: The
current practice and the future trends. Sudan J Paediatr. 13:11–22.
2013.PubMed/NCBI
|
|
17
|
Demirakça S, Dötsch J, Knothe C, Magsaam
J, Reiter HL, Bauer J and Kuehl PG: Inhaled nitric oxide in
neonatal and pediatric acute respiratory distress syndrome: Dose
response, prolonged inhalation, and weaning. Crit Care Med.
24:1913–1919. 1996.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Lai D, Xia J, Wang J, Wei X, Qian J, Lou
Q, Ren X and Huang X: The effect of paraquat on voltage-dependent
anion channel and caspase-3, 8, 9 in the mitochondria of rat lung.
Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi. 33:363–365.
2015.PubMed/NCBI(In Chinese).
|
|
19
|
Zimmermann KK, Spassov SG, Strosing KM,
Ihle PM, Engelstaedter H, Hoetzel A and Faller S: Hydrogen sulfide
exerts anti-oxidative and anti-inflammatory effects in acute lung
injury. Inflammation. 41:249–259. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Li SJ, Wang XJ, Hu JB, Kang XQ, Chen L, Xu
XL, Ying XY, Jiang SP and Du YZ: Targeting delivery of simvastatin
using ICAM-1 antibody-conjugated nanostructured lipid carriers for
acute lung injury therapy. Drug Deliv. 24:402–413. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Becker DE: Basic and clinical pharmacology
of glucocorticosteroids. Anesth Prog. 60:25–31; quiz 32.
2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Navegantes KC, de Souza Gomes R, Pereira
PA, Czaikoski PG, Azevedo CH and Monteiro MC: Immune modulation of
some autoimmune diseases: The critical role of macrophages and
neutrophils in the innate and adaptive immunity. J Transl Med.
15(36)2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Gadian J, Kirk E, Holliday K, Lim M and
Absoud M: Systematic review of immunoglobulin use in paediatric
neurological and neurodevelopmental disorders. Dev Med Child
Neurol. 59:136–144. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ma XY, Li Z, Wang XJ, Ye JJ, Ma YP and Li
Y: Clinical efficacy of different doses of gamma globulin combined
with glucocorticoid in treatment of moderate/severe acute
Guillain-Barré syndrome in children: A comparative analysis.
Zhongguo Dang Dai Er Ke Za Zhi. 18:1286–1290. 2016.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
25
|
Viasus D, Garcia-Vidal C, Simonetti AF,
Dorca J, Llopis F, Mestre M, Morandeira-Rego F and Carratalà J: The
effect of simvastatin on inflammatory cytokines in
community-acquired pneumonia: A randomised, double-blind,
placebo-controlled trial. BMJ Open. 5(e006251)2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Fang Y, Xu P, Gu C, Wang Y, Fu XJ, Yu WR
and Yao M: Ulinastatin improves pulmonary function in severe
burn-induced acute lung injury by attenuating inflammatory
response. J Trauma. 71:1297–1304. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Gao M, Xie B, Gu C, Li H, Zhang F and Yu
Y: Targeting the proinflammatory cytokine tumor necrosis factor-α
to alleviate cardiopulmonary bypass-induced lung injury (review).
Mol Med Rep. 11:2373–2378. 2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Massey VL, Poole LG, Siow DL, Torres E,
Warner NL, Schmidt RH, Ritzenthaler JD, Roman J and Arteel GE:
Chronic alcohol exposure enhances lipopolysaccharide-induced lung
injury in mice: Potential role of systemic tumor necrosis
factor-alpha. Alcohol Clin Exp Res. 39:1978–1988. 2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kang S, Tanaka T and Kishimoto T:
Therapeutic uses of anti-interleukin-6 receptor antibody. Int
Immunol. 27:21–29. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wang L, Chen Q, Shi C, Lv H, Xu X and Yu
L: Changes of serum TNF-α, IL-5 and IgE levels in the patients of
mycoplasma pneumonia infection with or without bronchial asthma.
Int J Clin Exp Med. 8:3901–3906. 2015.PubMed/NCBI
|
|
31
|
Han CL and Zhao SL: Intravenous
immunoglobulin gamma (IVIG) versus IVIG plus infliximab in young
children with Kawasaki disease. Med Sci Monit. 24:7264–7270.
2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Chu L, Zhu F, Zhou W, Du Z, Li J, Wang X,
Wang L and Liu A: Baicalein attenuates acute lung injury induced by
intestinal ischemia/reperfusion via inhibition of nuclear factor-κB
pathway in mice. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 29:228–232.
2017.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
33
|
Wang Y, Gao L, Yang Z, Chen F and Zhang Y:
Effects of probiotics on ghrelin and lungs in children with acute
lung injury: A double-blind randomized, controlled trial. Pediatr
Pulmonol. 53:197–203. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Galli SJ, Tsai M and Piliponsky AM: The
development of allergic inflammation. Nature. 454:445–454.
2008.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Deshmane SL, Kremlev S, Amini S and Sawaya
BE: Monocyte chemoattractant protein-1 (MCP-1): An overview. J
Interferon Cytokine Res. 29:313–326. 2009.PubMed/NCBI View Article : Google Scholar
|