Introduction
Myocardial ischemia/reperfusion (I/R) injury begins
with cardiac ischemia; reperfusion leads to increased cell loss and
enlarged infarction area (1).
Restoration of blood flow following infarction increases myocardial
structural and functional damage, resulting in cardiac dysfunction,
myocardial shock and malignant arrhythmia (2,3).
Decreasing myocardial I/R injury can lessen infarct size, thereby
improving long-term cardiac function. Therefore, it is important to
understand the molecular mechanism of I/R injury, find effective
targets and prevent myocardial cell loss following I/R injury.
Autophagy and apoptosis are the two types of
programmed cell death (4).
Inhibiting apoptosis decreases myocardial damage following
infarction. The role of autophagy has been studied in many fields,
such as cancer and cardiovascular disease (5,6).
Autophagy is widespread in myocardial I/R injury (7) and its role may be protective or
destructive (8). In the early stage
of ischemia, autophagy facilitates cardiomyocyte survival, but with
the prolongation of ischemia, the increase of autophagy may cause
cell death and lead to heart failure (9). Controlling the autophagy pathway
during myocardial I/R may serve a protective or damaging role. It
is crucial to selectively block undesirable pathways involved in
autophagy without damaging beneficial pathways. For example,
exosomes derived from mesenchymal stem cells alleviate myocardial
ischemia reperfusion by inducing cardiomyocyte autophagy via
AMPK/mTOR and Akt/mTOR pathways (10). However, miR-103a-3p relieves
apoptosis and autophagy in hypoxia-induced H9c2 cells by targeting
Atg5(11)
Recent studies (12-14)
have found a promising way to relieve or eliminate I/R injury
following myocardial infarction by controlling miRNA expression.
MicroRNAs (miRNAs or miRs) are a class of small non-coding RNA with
21-23 nucleotides that inhibit gene expression by binding to the 3'
untranslated region (UTR) of mRNA (15,16). A
large number of miRNAs and mRNAs constitute a complex regulatory
network (17). A single miRNA may
regulate hundreds of different target mRNAs, while a single mRNA
may also be regulated by several miRNAs (18). miRNAs have been reported to be
involved in the pathological process of cardiovascular disease,
especially in the pathophysiological progress of myocardial
apoptosis, arrhythmia, heart failure and cardiac hypertrophy
(19,20) but they do not have protein-coding
capabilities (21). There is
increasing evidence (9,15) that miRNAs affect autophagy or
apoptosis during myocardial I/R injury. For example, miR-103a-3p
serves a protective role in myocardial ischemia by affecting
autophagy (11) and miR-496
inhibits cardiomyocyte apoptosis and protects ischemic myocardial
tissue (22). miR-590-3p is
produced by its precursor mir-590 and its role in cancer has been
widely reported (14); miR-590-3p
serves an important role in myocardial fibrosis. For example,
miR-590-3p inhibits cardiac fibroblast proliferation,
differentiation, migration and collagen synthesis by targeting zinc
finger E-box binding homeobox 1(23). In addition, immediate injection of
synthetic miR-590-3p lipid formulations on the anterior ventricular
wall following myocardial infarction in mice decreases infarct size
and promotes recovery of myocardial function (24). To the best of our knowledge,
however, the expression of miR-590-3p following cardiac I/R and the
role of miR-590-3p in cardiac I/R injury have not been
reported.
Materials and methods
Isolation of primary rat
cardiomyocytes
A total of 15 Neonatal Sprague-Dawley (SD) rats (1-3
days, sex indeterminable, 5-6 g) were purchased from the
Experimental Animal Center of Bengbu Medical College (Bengbu,
China; lot no. 20180004002485). All animal procedures complied with
the United States National Institutes of Health Guide (25) and were approved by the Animal Ethics
Association of Bengbu Medical College (approval no. 075, 2017).
Primary cardiomyocytes were isolated under sterile conditions, as
previously described (26).
Following anesthesia with 4% chloral hydrate, rats were euthanized
via cervical dislocation. Death was confirmed by pupil dilation and
cessation of heartbeat. The ventricles was removed, washed with
cold PBS, cut into 1-mm3 pieces and digested in 0.1% II
type collagenase at 37˚C for 5 min. The digestion process was
repeated 4-5 times. In order to isolate fibroblasts, cell
suspensions (1x105/ml)were cultured at 37˚C for 1.5 h,
then non-adherent cells were collected and cultured in DMEM F12
containing 10% FBS (both HyClone; Cytiva) in CO2 at
37˚C. A total of 100 µmol/l 5-bromo-2-deoxyuridine was added to the
medium to prevent proliferation of cardiac fibroblasts.
Troponin C (TNNC1) is a specific protein of
cardiomyocytes (27). Anti-cardiac
TNNC1 antibody (1:300; cat. no. BA4613; Wuhan Boster Biological
Technology Ltd.) was used to identify cardiomyocytes by
Immunofluorescence. Cell slides were incubated with Anti-cardiac
TNNC1 overnight at 4˚C after blocking with 5% bovine serum albumin
(BSA; Gibco; Thermo Fisher Scientific, Inc.) for 30 min at 37˚C.
FITC-conjugated secondary antibodies (1:200; cat. no. S0008;
Affinity Biosciences) were added and incubated in 37˚C for 1h. The
nuclei of primary cardiomyocytes were stained with DAPI for 10 min
in 37˚C. Fluorescent images were obtained with fluorescence
microscope camera (OLYMPUSU-HGLGPS).
Experimental groups and
interventions
In order to mimic H/R injury, cells
(1x105/ml) were cultured in a hypoxic incubator with 1%
O2, 94% N2 and 5% CO2 at 37˚C for
6 h. Then, cells were reoxygenated (95% air, 5% CO2) for
6 h. psiCHECK2-HIF-1α plasmid and control psiCHECK2 plasmid were
commercially provided by Hunan Fenghui Biotechnology Co., Ltd. Cell
transfection was performed using Lipofectamine® 2000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) in accordance
with the manufacturer's instruction. Primary cardiomyocytes were
divided into the following groups: Control (untreated primary
cardiomyocytes), H/R (H/R-treated primary cardiomyocytes),
miR-negative control (NC) + H/R (transfection with 100 µg of
miR-590-3p mimic NC in 37˚C for 6 h, then H/R treatment), mimic +
H/R (transfection with 100 µg of miR-590-3p mimic in 37˚C for 6 h,
then H/R treatment), mimic + vector + H/R (co-transfection with 100
µg of miR-590-3p mimic and 0.8 µg of control psiCHECK2 plasmid in
37˚C for 6 h, then H/R treatment) and mimic + HIF-1α + H/R
(co-transfection with 100 µg of miR-590-3p mimic and 0.8 µg of
psiCHECK2-HIF-1α plasmid, then H/R treatment). H/R treatment was
performed 24 h after transfection. The sequence of miR-590-3p mimic
and mimic NC were: miR-590-3p mimic sense,
5'-UAAUUUUAUGUAUAAGCUAGU-3' and antisense,
5'-UUCUCCGAACGUGUCACGUTT-3' and miR-590-3p mimic NC sense,
5'-UAGCUUAUACAUAAAAUUAUU-3' and antisense,
5'-ACGUGACACGUUCGGAGAATT-3'.
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted from primary cardiomyocytes
using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions.
Purity and concentration of total RNA were detected using a
microplate reader (Biotek™ Epoch™; BioTek
Instruments, Inc.). Total RNA (2 µg) was used to synthesize cDNA
using an All-in-One™ miRNA RT-qPCR Detection kit
(GeneCopoeia, Inc.) at 37˚C for 60 min and 85˚C for 5 min. The
primers were as follows: miR-590-3p forward,
5'-CGCTAATTTTATGTATAAGCTAGTAAAA-3' and reverse,
5'-TGGTGTCGTGGAGTCG-3' and U6 forward, 5'-AUAAAUCCCUUUACACCUCTT-3'
and reverse, 5'-AAUAAAUCCCUUUACACCUCTT-3'. The miR-590-3p
expression level was quantified using 20 µl reaction mixture
containing 2.0 cDNA (20 ng/µl), 2.0 each forward reverse primer,
10.0 All-in-One qPCR Mix and 0.4 µl Rox reference dye (from the
kit). The thermocycling conditions were as follows: 95˚C for 10
min, then 40 cycles at 95˚C for 10 sec, 60˚C for 20 sec and 72˚C
for 10 sec. The relative expression of miR-590-3p was analyzed by
2-ΔΔCq value calculation method (28) and normalized to the internal
control, U6. Each experiment was repeated three times.
Detection of cell apoptosis
Annexin V-FITC/PI apoptosis detection kit (Beyotime
Institute of Biotechnology) was used to detect cell apoptosis by
flow cytometry. Following treatment as aforementioned, cells were
collected and resuspended with 1X binding buffer. Cell suspension
was adjusted to a concentration of 1x106 cells/ml.
Subsequently, 5 µl Annexin V-FITC and 10 µl PI were added and
vortexed gently, then incubated at room temperature (20-25˚C) for
15 min in the dark. Cells were analyzed using
FACSCalibur™ (Becton, Dickinson and Company). The
percentage of cells in each quadrant were analyzed using ModFit
software (Verity Software House, Inc.). Each experiment was
repeated three times.
Western blot analysis
Following treatment as aforementioned, cells were
collected and lysed with lysis buffer (Beyotime Institute of
Biotechnology) containing 0.5 mM PMSF (Sigma-Aldrich; Merck KGaA).
Protein concentration was determined by BCA kit (Beyotime Institute
of Biotechnology). Equal amounts of protein (40 µg) was subjected
to 10% SDS-PAGE gels and then transferred to a polyvinylidene
difluoride membrane. The membranes were blocked with 5% free-fat
milk for 2 h at room temperature, and subsequently incubated with
the following primary antibodies at 4˚C overnight: Bcl-2 (1:1,000;
cat. no. AF6139; Affinity Biosciences), Bax (1:1,000; cat. no.
AF0120; Affinity Biosciences), cleaved caspase-3 (1:1,000; cat. no.
AF7022; Affinity Biosciences), light chain (LC)3 (1:1,000; cat. no.
ab51520; Abcam), p62 (1:1,000; cat. no. ab56416; Abcam), Beclin-1
(1:1,000; cat. no. ab62557; Abcam), HIF-1α (1:1,000 cat. no.
ab216842; Abcam) and β-actin (1:3,000, cat. no. AF7018, Affinity
Biosciences). Then the membrane was incubated with HRP-conjugated
secondary antibody (1:10,000; cat. no. BL003A; Biosharp) for 2 h at
room temperature. Proteins were identified by
electrochemiluminescence (Beyotime Institute of Biotechnology) and
imaged using X-ray film. The intensities of protein were quantified
by Bio-Rad Quantity One v4.62 software (Bio-Rad Laboratories,
Inc.). Each experiment was repeated three times.
Dual luciferase reporter assay
TargetScan (29)
database was used to identify miR-590-3p target genes.
Dual-Lumi™ Luciferase Assay kit (Beyotime Institute of
Biotechnology) was used for dual luciferase reporter assay
according to the manufacturer's instruction. Human embryonic kidney
cells (HEK293, 1x105/ml) were cultured in a 24-well
plate in CO2 at 37˚C and then co-transfected with
pGL3-HIF-1α-3'UTR/pGL3-HIF-1α mutant 3'UTR plasmid (Promega
Corporation) and mimic control/miR-590-3p mimic using Lipofectamine
2000 (Invitrogen; Thermo Fisher Scientific, Inc.) according to the
manufacturer's instruction. Following 48 h of incubation, the
luciferase activity was measured using dual luciferase reporter
assay kit (Beyotime Institute of Biotechnology) according to the
manufacturer's protocol. Each experiment was repeated three
times.
Statistical analysis
Data are presented as the mean ± SD. Each experiment
was repeated three times. Data were analyzed by GraphPad Prism
software 7.0 (GraphPad Software, Inc.). Data from two groups were
compared by paired Student's t-test; data from multiple groups were
compared using one-way analysis of variance followed by Tukey's
post hoc test. P<0.05 was considered to indicate a statistically
significant difference.
Results
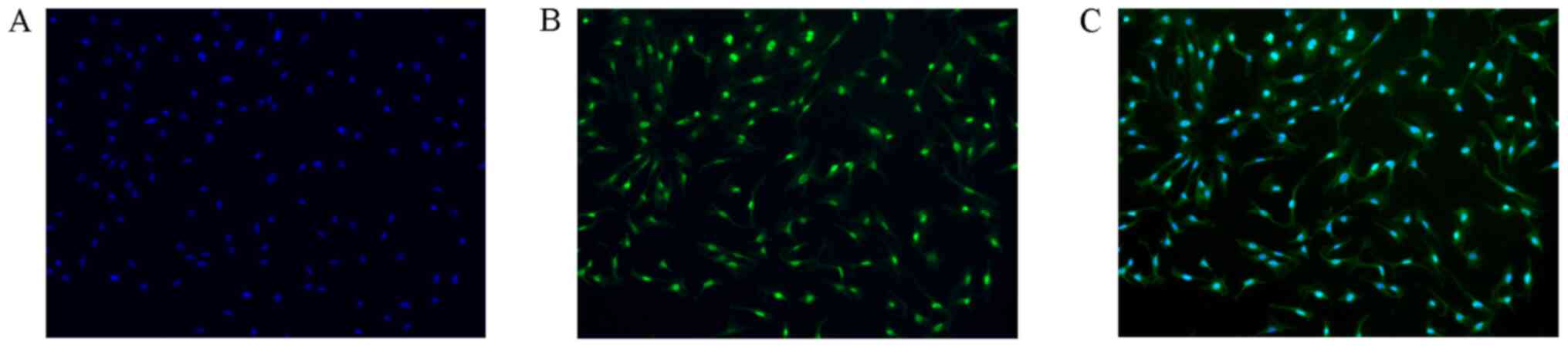
Immunofluorescence identification of
primary cardiomyocytes
Primary cardiomyocytes were observed by fluorescence
microscopy using TNNC1 as a specific cardiomyocyte protein. Green
fluorescence was located in the cytoplasm of cardiomyocytes; blue
fluorescence was located in the nucleus of all cells. The purity of
cultured cardiomyocytes was >95% (Fig. 1).
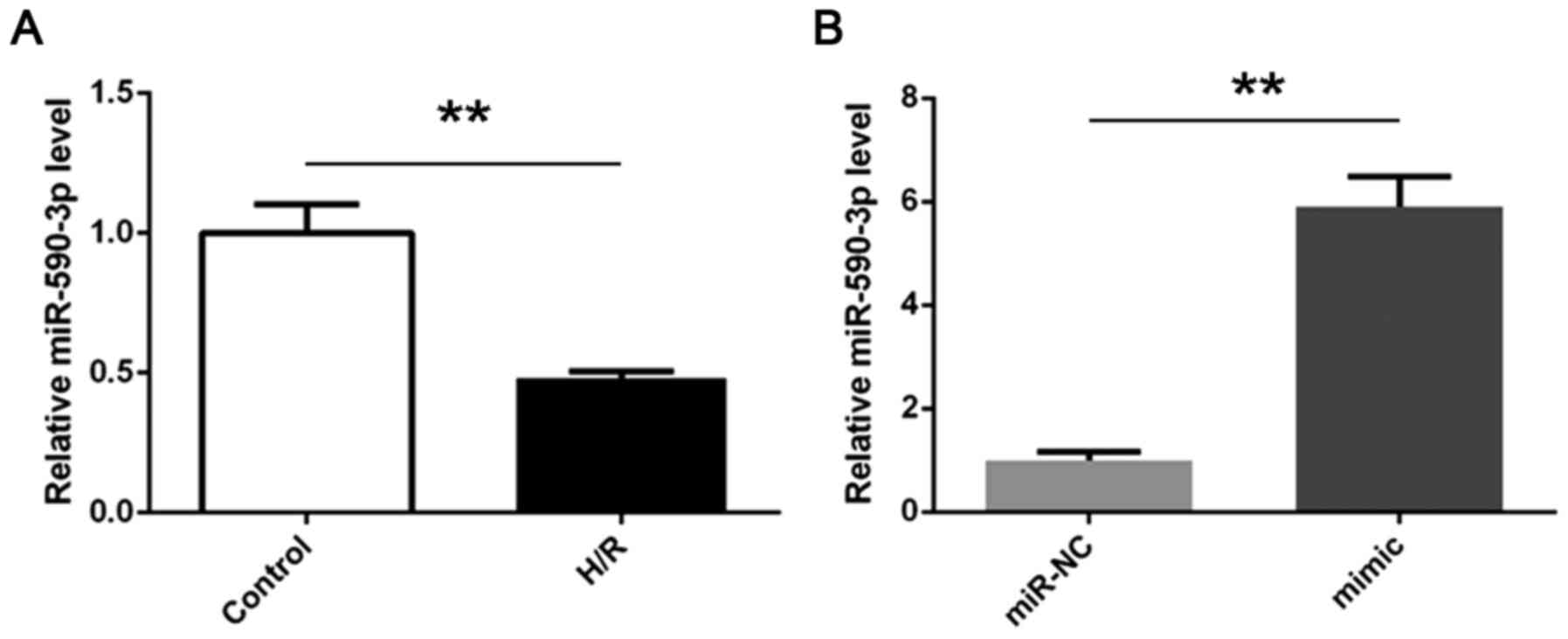
Expression of miR-590-3p decreases in
primary cardiomyocytes following H/R treatment
In order to determine expression of miR-590-3p,
RT-qPCR was performed in H/R and control groups. The results
indicated that compared with the control group, the expression of
miR-590-3p in cardiomyocytes decreased in the H/R group (Fig. 2A). miR-590-3p mimic transfection
significantly improved miR-590-3p expression levels (Fig. 2B).
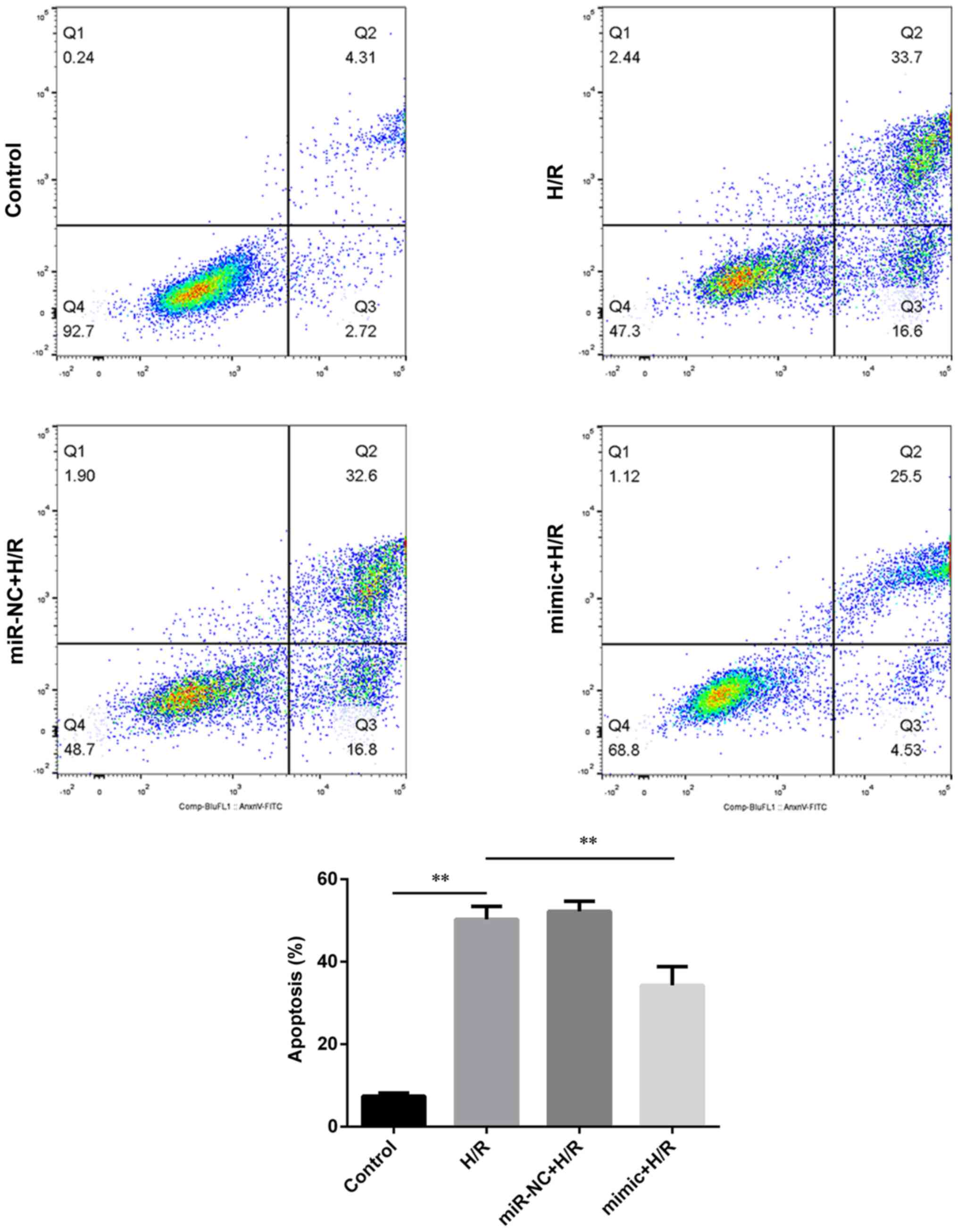
miR-590-3p relieves autophagy and
apoptosis in primary cardiomyocytes during H/R injury
In order to evaluate the role of miR-590-3p in
H/R-treated cardiomyocytes, annexin V-FITC staining and flow
cytometry were used to detect apoptosis rate. The results indicated
that H/R treatment induced apoptosis and this was significantly
reversed by miR-590-3p mimic transfection (Fig. 3). Apoptosis- and
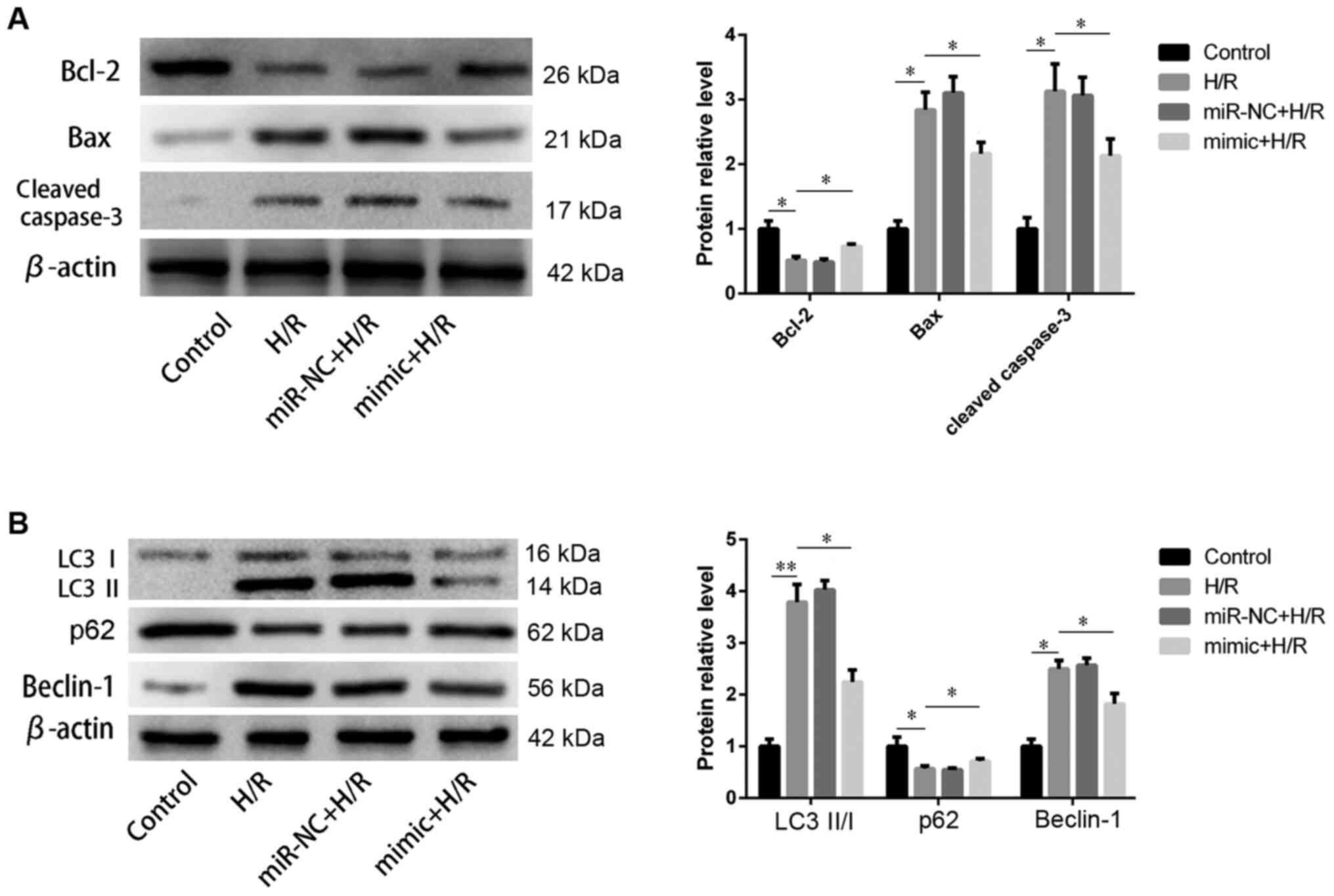
autophagy-associated proteins were detected by western blot
analysis. Levels of apoptotic protein Bcl-2 was decreased but those
of Bax and cleaved caspase-3 were increased following H/R
treatment. miR-590-3p transfection significantly rescued these
changes (Fig. 4A), which was
consistent with flow cytometry analysis. In addition, autophagy was
activated following H/R treatment; this was accompanied by
increased LC3Ⅱ/Ⅰ ratio and Beclin-1 protein levels and decreased
p62 protein level. Transfection of miR-590-3p mimic significantly
reversed the changes caused by H/R (Fig. 4B).
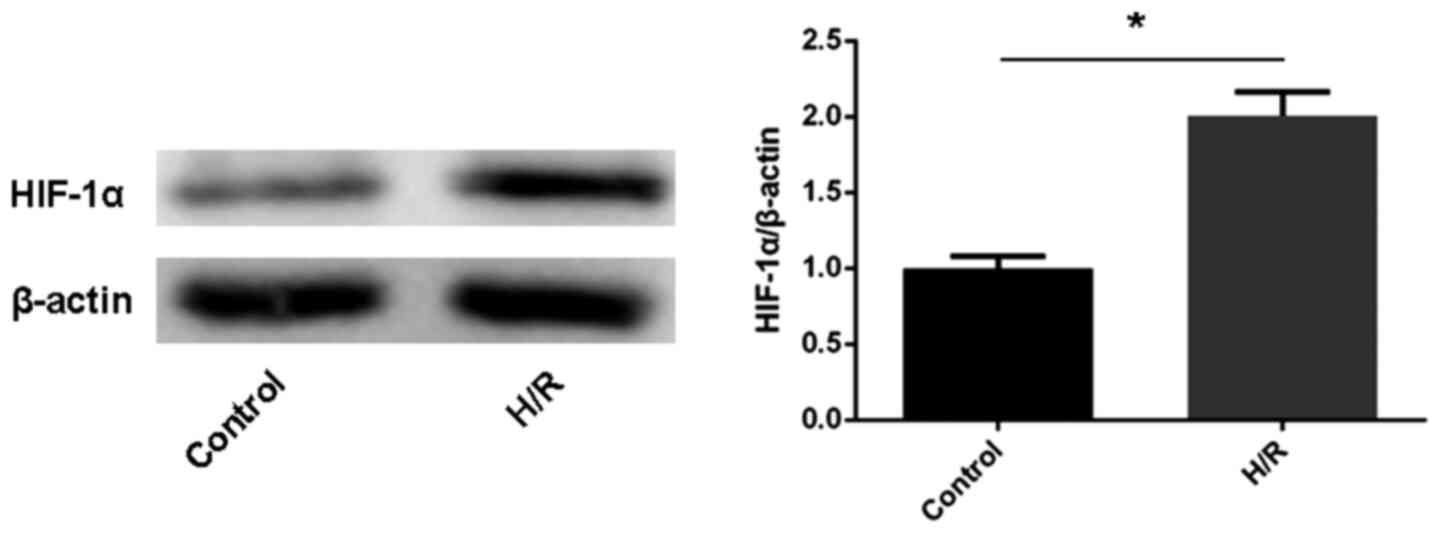
Expression of HIF-1α increases in
primary cardiomyocytes following H/R treatment
In order to assess the protein expression levels of
HIF-1α, western blot analysis was performed in the H/R and control
groups. The results indicated that compared with the control group,
the expression of HIF-1α protein was increased in the H/R group
(Fig. 5).
HIF-1α is a target gene of
miR-590-3p
In order to determine the target gene of miR-590-3p,
bioinformatics analysis was performed using the TargetScan
database. The 3'UTR region of HIF-1α gene contained a binding site
for miR-590-3p. In order to verify whether miR-590-3p regulates the
transcriptional activity of HIF-1α by targeting the 3'UTR of
HIF-1α, luciferase reporter assay was performed. The results
confirmed that HIF-1α was a direct target gene of miR-590-3p.
(Fig. 6).
miR-590-3p inhibits autophagy and
apoptosis via HIF-1α in H/R-treated cardiomyocytes
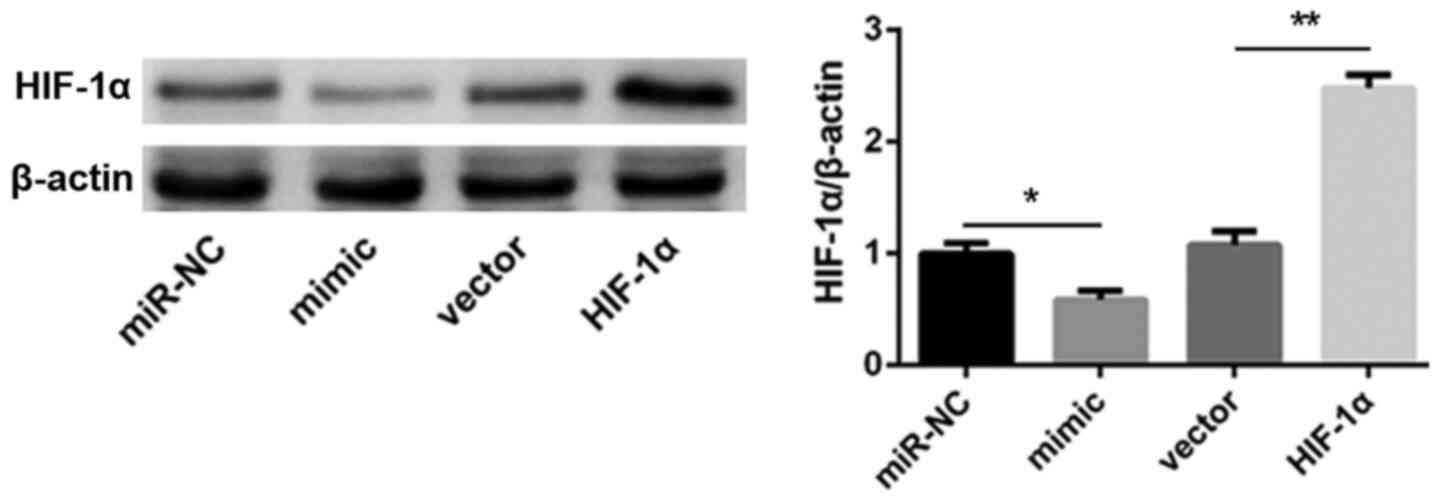
In order to investigate the role of HIF-1α in
miR-590-3p-mediated inhibition of autophagy and apoptosis in
cardiomyocytes, HIF-1α overexpression vector was constructed and
co-transfected with miR-590-3p mimic. Transfection efficiency was
detected by western blot analysis (Fig.
7).
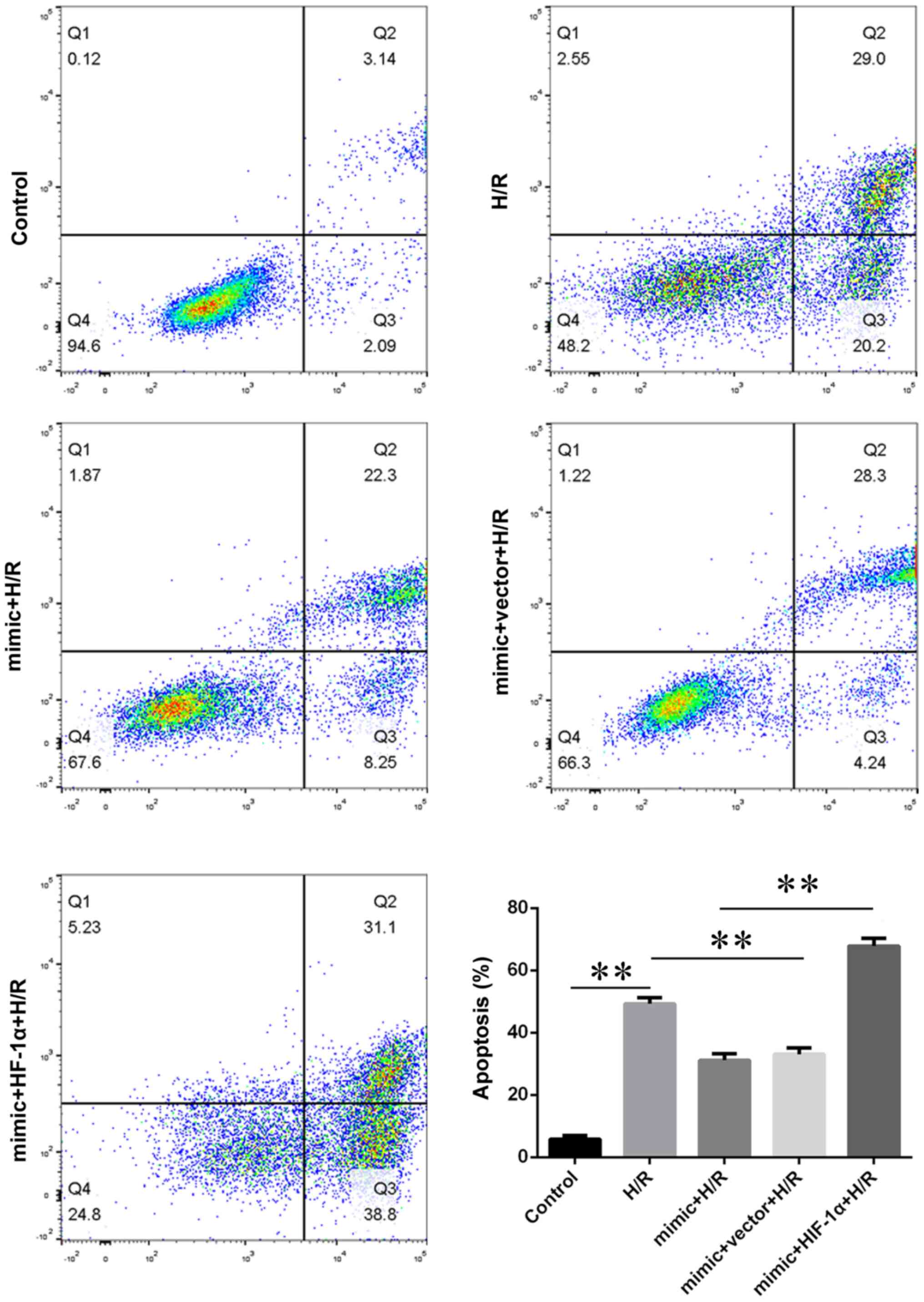
Flow cytometric analysis revealed that HIF-1α
overexpression significantly increased the apoptotic rate in the
HIF-1α + mimic + H/R group compared with mimic + H/R group
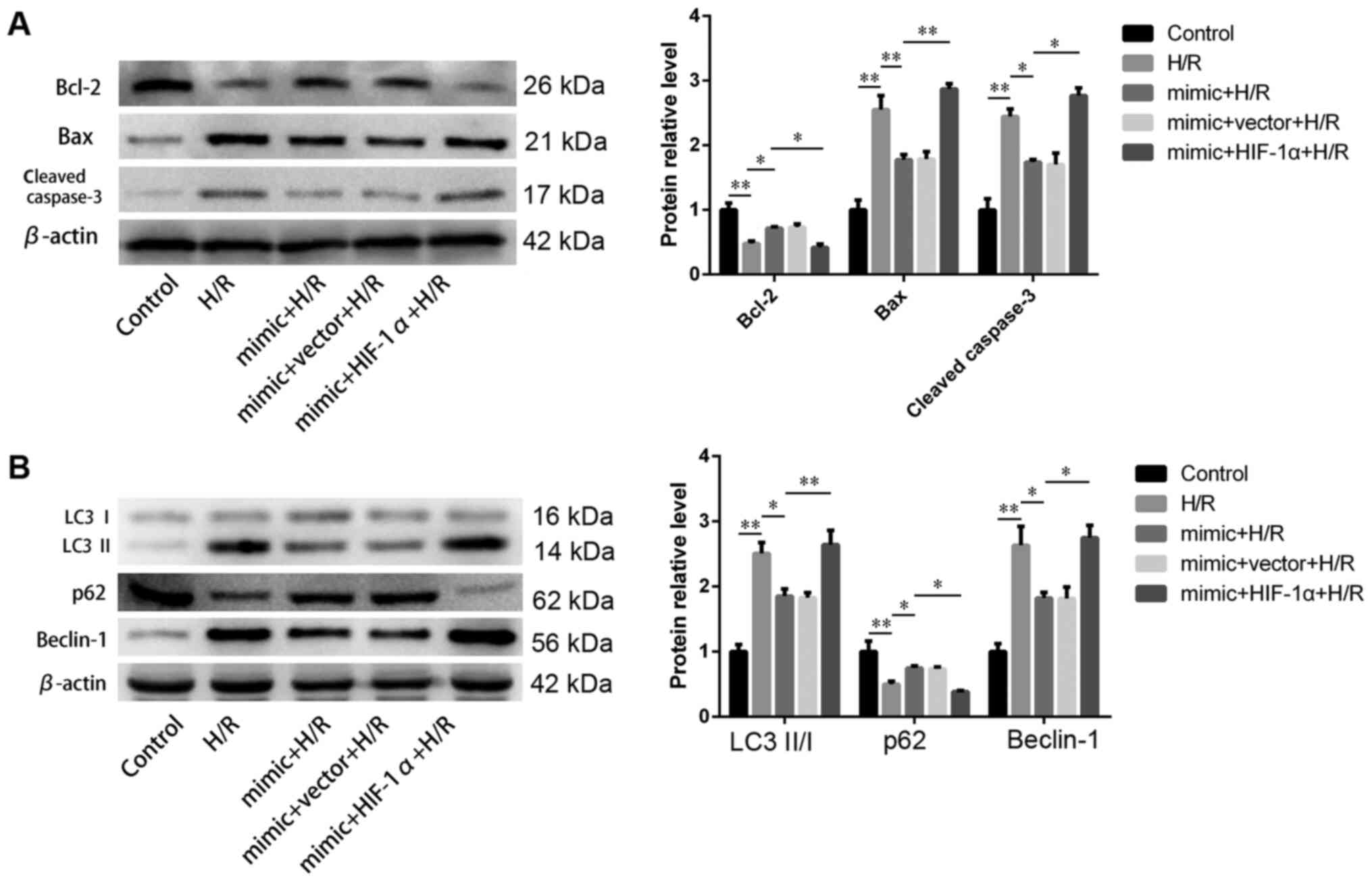
(Fig. 8). Compared with mimic + H/R
group, the expression levels of apoptotic and autophagic proteins
in the mimic + HIF-1α + H/R group were significantly increased
(Fig. 9).
Discussion
The present data suggested that miR-590-3p exhibited
a protective effect on cardiomyocytes following H/R treatment. The
protective effect was due to inhibition of autophagy and apoptosis.
The mechanism may be mediated by inhibition of HIF-1α. The present
study indicated that miR-590-3p relieved I/R injury and confirmed
miR-590-3p as a potential therapeutic target for myocardial
ischemia.
miRNAs are an important medium in the pathological
process of various types of cardiovascular disease and may be a
therapeutic target (30). One of
the challenges in developing miRNAs to treat cardiovascular disease
is to identify such miRNAs. Correcting dysregulated miRNAs can
provide options for clinical treatment of cardiovascular disease
(9). In the present study,
H/R-treated cardiomyocytes were used to simulate myocardial I/R
injury and it was found that expression levels of miR-590-3p were
significantly decreased. Overexpressing miR-590-3p inhibited
H/R-induced cardiomyocyte apoptosis. Apoptosis is a key part of
myocardial I/R injury and is regulated by miRNAs (31). For example, miR-327 attenuates
cardiomyocyte apoptosis induced by myocardial I/R injury (32). Cardiomyocytes are terminally
differentiated cells that can cause irreversible damage to
myocardium following apoptosis (33). miRNAs are involved in the induction
of apoptosis during I/R injury (34). Annexin V-FITC/PI staining
demonstrated that miR-590-3p mimic inhibited cardiomyocyte
apoptosis following H/R, suggesting that miR-590-3p promoted cell
survival. To the best of our knowledge, there are no previous
reports on the protective effect of miR-590-3p in myocardial I/R
injury.
Autophagy is a highly conserved catabolic process
that maintains homeostasis of cells by removing damaged proteins
and organelles (35). However,
under pathological conditions, chronic upregulation of autophagy
may lead to an imbalance of homeostatic conditions (35). In the present study, autophagy was
evaluated by detecting LC3Ⅱ/I ratio and Beclin-1 and p62 protein
levels. Increased LC3Ⅱ/Ⅰ ratio is a sign of increased autophagosome
formation (36). Beclin-1 is a key
molecule in autophagy and is regulated by miRNAs (37). In addition, p62 has been identified
as a substrate that can be degraded by interacting with LC3 via the
autophagy-lysosomal pathway (38).
In the present study, LC3Ⅱ/Ⅰ and levels of Beclin-1 increased,
whereas p62 expression levels decreased in H/R-treated
cardiomyocytes. Following miR-590-3p mimic transfection, LC3Ⅱ and
the ratio of LC3Ⅱ/Ⅰ decreased, Beclin-1 expression levels decreased
and p62 levels increased, which indicated that autophagy increased
following H/R treatment; miR-590-3p reversed the increased
autophagy induced by H/R treatment.
The mechanism of miR-590-3p in alleviating
myocardial I/R injury was further investigated. The target of
miR-590-3p was predicted and it was confirmed that HIF-1α directly
interacts with miR-590-3p. The expression of HIF-1α was increased
in H/R-treated cardiomyocytes, but significantly decreased
following miR-590-3p mimic transfection. These data suggest it is a
promising target for miR-590-3p-mediated protection. HIF-1α is a
key transcription and regulatory factor in hypoxia and is induced
during hypoxia or ischemia (39).
High expression levels of HIF-1α can initiate a series of events,
including apoptosis (40). HIF-1α
is associated with activation of autophagy. Overexpression of
HIF-1α can induce autophagy in numerous types of cell (41,42).
Inhibition of HIF-1α activity decreases I/R and H/R injury in rat
heart and cardiomyocytes (43,44).
In the present study, miR-590-3p inhibits apoptosis and autophagy
by downregulation of HIF-1α, protecting cardiomyocytes from H/R
injury.
In summary, the present study suggested that
increased miR-590-3p expression decreases myocardial autophagy and
apoptosis by specifically targeting HIF-1α, thus decreasing
myocardial I/R injury; this may provide novel therapeutic options
for clinical treatment of myocardial I/R injury.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by grants from the
National Natural Science Foundation of China (grant no. 81770297)
and the Nature Science Major and Key Program of College and
University of Anhui Province (grant no. KJ2018A0212).
Availability of data and materials
All data generated or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
XC and QG designed the study, NG and XY collected
and analyzed the data, and confirm the authenticity of all the raw
data. XL, YJ, CG and SM analyzed the data and drafted the
manuscript and critically revised the manuscript for important
intellectual content. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
All animal procedures complied with the United
States National Institutes of Health Guide and were approved by the
Animal Ethics Association of Bengbu Medical College, China
(approval no. 075, 2017).
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Bochaton T and Ovize M: Circadian rhythm
and ischaemia-reperfusion injury. Lancet. 391:8–9. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hausenloy DJ, Botker HE, Engstrom T,
Erlinge D, Heusch G, Ibanez B, Kloner RA, Ovize M, Yellon DM and
Garcia-Dorado D: Targeting reperfusion injury in patients with
ST-segment elevation myocardial infarction: Trials and
tribulations. Eur Heart J. 38:935–941. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Pagel PS, Sethi P, Freed JK, Boettcher BT
and Hossein Almassi G: A rare complication of cardiopulmonary
resuscitation after mitral valve replacement. J Cardiothorac Vasc
Anesth. 31:770–772. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Nikoletopoulou V, Markaki M, Palikaras K
and Tavernarakis N: Crosstalk between apoptosis, necrosis and
autophagy. Biochim Biophys Acta. 1833:3448–3459. 2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Levine B and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ichimiya T, Yamakawa T, Hirano T, Yokoyama
Y, Hayashi Y, Hirayama D, Wagatsuma K, Itoi T and Nakase H:
Autophagy and autophagy-related diseases: A review. Int J Mol Sci.
21(8974)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xuan F and Jian J: Epigallocatechin
gallate exerts protective effects against myocardial
ischemia/reperfusion injury through the PI3K/Akt pathway-mediated
inhibition of apoptosis and the restoration of the autophagic flux.
Int J Mol Med. 38:328–336. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tanaka Y, Guhde G, Suter A, Eskelinen EL,
Hartmann D, Lüllmann-Rauch R, Janssen PM, Blanz J, von Figura K and
Saftig P: Accumulation of autophagic vacuoles and cardiomyopathy in
LAMP-2-deficient mice. Nature. 406:902–906. 2000.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Sermersheim MA, Park KH, Gumpper K,
Adesanya TM, Song K, Tan T, Ren X, Yang JM and Zhu H: MicroRNA
regulation of autophagy in cardiovascular disease. Front Biosci
(Landmark Ed). 22:48–65. 2017.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Liu L, Jin X, Hu CF, Li R, Zhou Z and Shen
CX: Exosomes derived from mesenchymal stem cells rescue myocardial
ischaemia/reperfusion injury by inducing cardiomyocyte autophagy
Via AMPK and Akt pathways. Cell Physiol Biochem. 43:52–68.
2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Zhang C, Zhang C, Wang H, Qi Y, Kan Y and
Ge Z: Effects of miR103a3p on the autophagy and apoptosis of
cardiomyocytes by regulating Atg5. Int J Mol Med. 43:1951–1960.
2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wehbe N, Nasser SA, Pintus G, Badran A,
Eid AH and Baydoun E: MicroRNAs in cardiac hypertrophy. Int J Mol
Sci. 20(4714)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhang X, Dong S, Jia Q, Zhang A, Li Y, Zhu
Y, Lv S and Zhang J: The microRNA in ventricular remodeling: The
miR-30 family. Biosci Rep. 39(BSR20190788)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Moghaddam AS, Afshari JT, Esmaeili SA,
Saburi E, Joneidi Z and Momtazi-Borojeni AA: Cardioprotective
microRNAs: Lessons from stem cell-derived exosomal microRNAs to
treat cardiovascular disease. Atherosclerosis. 285:1–9.
2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Krol J, Loedige I and Filipowicz W: The
widespread regulation of microRNA biogenesis, function and decay.
Nat Rev Genet. 11:597–610. 2010.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Bartel DP: MicroRNAs: Target recognition
and regulatory functions. Cell. 136:215–233. 2009.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Sohel MMH: Macronutrient modulation of
mRNA and microRNA function in animals: A review. Anim Nutr.
6:258–268. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Seo HH, Lee SY, Lee CY, Kim R, Kim P, Oh
S, Lee H, Lee MY, Kim J, Kim LK, et al: Exogenous miRNA-146a
enhances the therapeutic efficacy of human mesenchymal stem cells
by increasing vascular endothelial growth factor secretion in the
ischemia/reperfusion-injured heart. J Vasc Res. 54:100–108.
2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Hendgen-Cotta UB, Messiha D, Esfeld S,
Deenen R, Rassaf T and Totzeck M: Inorganic nitrite modulates miRNA
signatures in acute myocardial in vivo ischemia/reperfusion. Free
Radic Res. 51:91–102. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhou Y, Chen Q, Lew KS, Richards AM and
Wang P: Discovery of potential therapeutic miRNA targets in cardiac
ischemia-reperfusion injury. J Cardiovasc Pharmacol Ther.
21:296–309. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Correia de Sousa M, Gjorgjieva M, Dolicka
D, Sobolewski C and Foti M: Deciphering miRNAs' action through
miRNA editing. Int J Mol Sci. 20(6249)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Jin Y and Ni S: miR-496 remedies hypoxia
reoxygenation-induced H9c2 cardiomyocyte apoptosis via
Hook3-targeted PI3k/Akt/mTOR signaling pathway activation. J Cell
Biochem. 121:698–712. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yuan X, Pan J, Wen L, Gong B, Li J, Gao H,
Tan W, Liang S, Zhang H and Wang X: MiR-590-3p regulates
proliferation, migration and collagen synthesis of cardiac
fibroblast by targeting ZEB1. J Cell Mol Med. 24:227–237.
2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Lesizza P, Prosdocimo G, Martinelli V,
Sinagra G, Zacchigna S and Giacca M: Single-dose intracardiac
injection of pro-regenerative microRNAs improves cardiac function
after myocardial infarction. Circ Res. 120:1298–1304.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Orlans FB: Regulation of animal
experimentation: United States of America. Acta Physiol Scand
Suppl. 554:138–152. 1986.PubMed/NCBI
|
|
26
|
Louch WE, Sheehan KA and Wolska BM:
Methods in cardiomyocyte isolation, culture, and gene transfer. J
Mol Cell Cardiol. 51:288–298. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Rebuzzini P, Fassina L, Mulas F, Bellazzi
R, Redi CA, Di Liberto R, Magenes G, Adjaye J, Zuccotti M and
Garagna S: Mouse embryonic stem cells irradiated with gamma-rays
differentiate into cardiomyocytes but with altered contractile
properties. Mutat Res. 756:37–45. 2013.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Agarwal V, Bell GW, Nam JW and Bartel DP:
Predicting effective microRNA target sites in mammalian mRNAs.
Elife. 4(e05005)2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wong LL, Rademaker MT, Saw EL, Lew KS,
Ellmers LJ, Charles CJ, Richards AM and Wang P: Identification of
novel microRNAs in the sheep heart and their regulation in heart
failure. Sci Rep. 7(8250)2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Bainey KR and Armstrong PW: Clinical
perspectives on reperfusion injury in acute myocardial infarction.
Am Heart J. 167:637–645. 2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Li Q and Yang J, Zhang J, Liu XW, Yang CJ,
Fan ZX, Wang HB, Yang Y, Zheng T and Yang J: Inhibition of
microRNA-327 ameliorates ischemia/reperfusion injury-induced
cardiomyocytes apoptosis through targeting apoptosis repressor with
caspase recruitment domain. J Cell Physiol. 235:3753–3767.
2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Woodcock EA and Matkovich SJ:
Cardiomyocytes structure, function and associated pathologies. Int
J Biochem Cell Biol. 37:1746–1751. 2005.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Di Y, Lei Y, Yu F, Changfeng F, Song W and
Xuming M: MicroRNAs expression and function in cerebral ischemia
reperfusion injury. J Mol Neurosci. 53:242–250. 2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Ghavami S, Gupta S, Ambrose E, Hnatowich
M, Freed DH and Dixon IM: Autophagy and heart disease: Implications
for cardiac ischemia-reperfusion damage. Curr Mol Med. 14:616–629.
2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Maejima Y, Kyoi S, Zhai P, Liu T, Li H,
Ivessa A, Sciarretta S, Del Re DP, Zablocki DK, Hsu CP, et al: Mst1
inhibits autophagy by promoting the interaction between Beclin1 and
Bcl-2. Nat Med. 19:1478–1488. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
37
|
Maejima Y, Isobe M and Sadoshima J:
Regulation of autophagy by Beclin 1 in the heart. J Mol Cell
Cardiol. 95:19–25. 2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Komatsu M and Ichimura Y: Physiological
significance of selective degradation of p62 by autophagy. FEBS
Lett. 584:1374–1378. 2010.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Mazure NM, Brahimi-Horn MC, Berta MA,
Berta MA, Benizri E, Bilton RL, Dayan F, Ginouvès A, Berra E and
Pouysségur J: HIF-1: Master and commander of the hypoxic world. A
pharmacological approach to its regulation by siRNAs. Biochem
Pharmacol. 68:971–980. 2004.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Greijer AE and van der Wall E: The role of
hypoxia inducible factor 1 (HIF-1) in hypoxia induced apoptosis. J
Clin Pathol. 57:1009–1014. 2004.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Gong G, Hu L, Liu Y, Bai S, Dai X, Yin L,
Sun Y, Wang X and Hou L: Upregulation of HIF-1α protein induces
mitochondrial autophagy in primary cortical cell cultures through
the inhibition of the mTOR pathway. Int J Mol Med. 34:1133–1140.
2014.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Wang IK, Sun KT, Tsai TH, Chen CW, Chang
SS, Yu TM, Yen TH, Lin FY, Huang CC and Li CY: MiR-20a-5p mediates
hypoxia-induced autophagy by targeting ATG16L1 in ischemic kidney
injury. Life Sci. 136:133–141. 2015.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Liu S, Ai Q, Feng K, Li Y and Liu X: The
cardioprotective effect of dihydromyricetin prevents
ischemia-reperfusion-induced apoptosis in vivo and in vitro via the
PI3K/Akt and HIF-1α signaling pathways. Apoptosis. 21:1366–1385.
2016.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wang X, Ma S and Qi G: Effect of
hypoxia-inducible factor 1-alpha on hypoxia/reoxygenation-induced
apoptosis in primary neonatal rat cardiomyocytes. Biochem Biophys
Res Commun. 417:1227–1234. 2012.PubMed/NCBI View Article : Google Scholar
|