Introduction
Pre-existing cardiovascular comorbidities are
important risk factors for patients with severe acute respiratory
syndrome coronavirus-2 (SARS-CoV-2). A recent study reported a
20-fold higher mortality rate in patients with cardiovascular
diseases and SARS-CoV-2 infection, with a >10 times higher risk
of being admitted to the intensive care unit (1). Heart failure, coronary artery disease
and arrhythmias are risk factors for coronavirus disease 2019
(COVID-19) progression and they are associated with poor outcome
(2).
However, one study showed that acute cardiovascular
events during COVID-19 hospitalization are frequent. Among the
3,011 hospitalized patients from The Netherlands over a 3-month
period, cardiac complications occurred in 11.6%, atrial
fibrillation being the most common, followed by heart failure,
acute coronary syndrome, ventricular arrhythmia, bacterial
endocarditis, myocarditis, pericarditis and pulmonary embolism
(3).
During the pandemic, a total of 920 COVID-19
patients were admitted to the Department of Infectious Disease in
the Clinical Infectious Diseases Hospital (Constanta, Romania)
between March 2020 and March 2021. The patients exhibited different
stages of the disease, with most of them having moderate or severe
forms. Cardiovascular complications were diagnosed in 103 patients
(11.20%), with cardiac rhythm and conduction abnormalities being
the most frequent, including atrial fibrillation (n=12), atrial
flutter (n=2), other supraventricular tachycardias (n=19),
bradycardia (n=16), bundle branch block (n=6) and a prolonged QT
interval (n=16). The incidence of pulmonary embolism was 0.87%
(n=8) and of deep vein thrombosis 0.65% (n=6). Acute renal artery
occlusion caused by systemic thromboembolism occurred in two cases.
The incidence of other cardiovascular complications was 1.20% for
pericarditis (n=11), 0.33% for stroke (n=3), 0.11% for myocarditis
(n=1) and 0.11% for bacterial endocarditis (n=1). Most of the
complications occurred in men, with a median age of 69 years, male
sex and advanced age (>55 years old) being two important
cardiovascular factors. SARS-COV2-infected patients have a high
risk of developing cardiovascular complications such as bacterial
endocarditis. A complete routine echocardiography, in patients with
severe SARS-COV-2 pneumonia admitted to intensive care units may be
required for the early diagnosis and treatment of infective
endocarditis.
Case report
A 73-year-old woman was admitted in May 2021 to the
Department of Infectious Disease in the Clinical Infectious
Diseases Hospital with a cough, shortness of breath and fatigue
that was found to be due to severe SARS-CoV-2 pneumonia by
nasopharyngeal swab. The patient had multiple cardiovascular risk
factors, including arterial hypertension, type 2 diabetes mellitus,
obesity (36.7 BMI) and a history of coronary heart disease for a
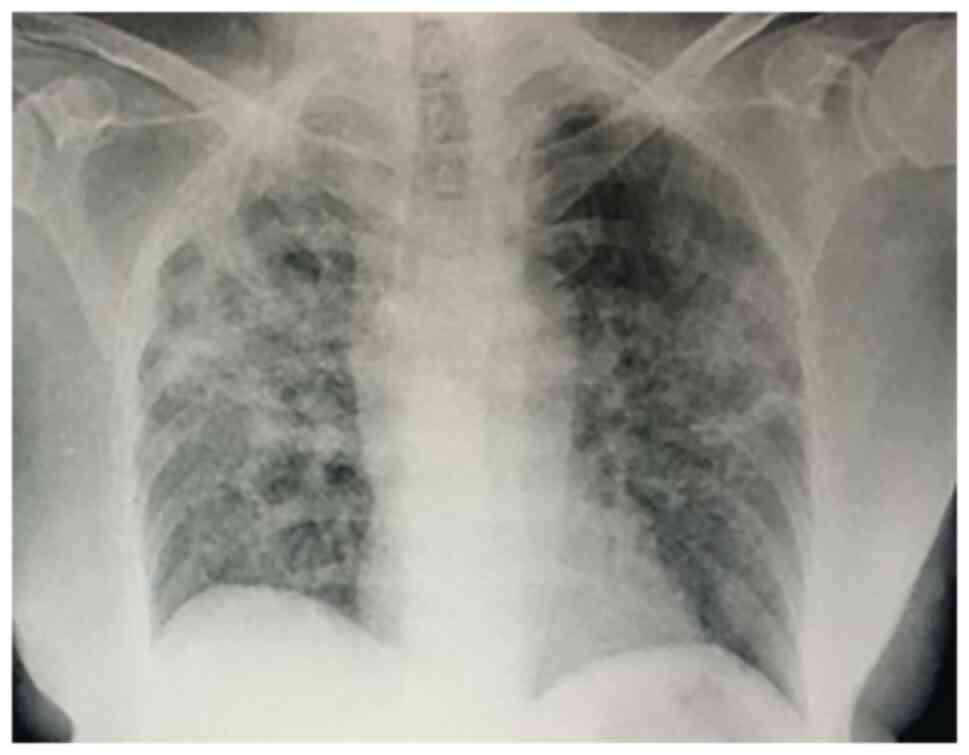
number of years. Chest X-rays showed bilateral pneumonia with
peripheral consolidations and a reticular pattern due to septal
thickening with ground-glass opacities (Fig. 1). From admission, the patient
exhibited dyspnea, with acute respiratory failure (mixed venous
oxygen saturation, 78%), and blood tests showed inflammatory
syndrome [elevated erythrocyte sedimentation rate (80 mm/h;
cut-off, 3-9 mm/h), and fibrinogen (820 mg/dl; cut-off, 196-372
mg/dl), C reactive protein (120 mg/l; cut-off, 0-5 mg/dl), ferritin
(1,206 ng/ml; cut-off, 70-435 ng/ml) and IL-6 (276 pg/ml; cut-off,
0-7 pg/ml) levels]. The serum level of procalcitonin was normal.
The patient received corticosteroid therapy (8 mg dexamethasone
twice daily) from admission, as well as tocilizumab (2x800 mg twice
daily), remdesivir (200 mg on day 1, then 100 mg daily for 5 days)
and prophylactic low molecular weight heparin (4,000 IU enoxaparin
daily). The clinical and biological evolution was favorable during
the first hospitalization period, but on the 14th day, the patient
developed a fever (38.5˚C temperature) and their general condition
worsened. Laboratory tests revealed a high procalcitonin level of
4.1 ng/ml (cut-off <0.5 ng/ml), mildly elevated liver
transaminase levels (ALT, 120 u/l; cut-off, 4-36 U/l; AST, 163 U/l;
cut-off, 5-40 U/l) and a decreased estimated glomerular filtration
rate (41 ml/min/1.73 m2). The hs-troponin (152 ng/l;
cut-off, 4.9-50 ng/l) and D-Dimer (1.78 µg FEU/ml; cut-off, 0-0.5
µg FEU/ml) levels were also elevated. The patient developed
respiratory distress (arterial oxygen partial pressure/fractional
inspired oxygen ratio <100) that required non-invasive
continuous positive airway pressure ventilation. A pulmonary
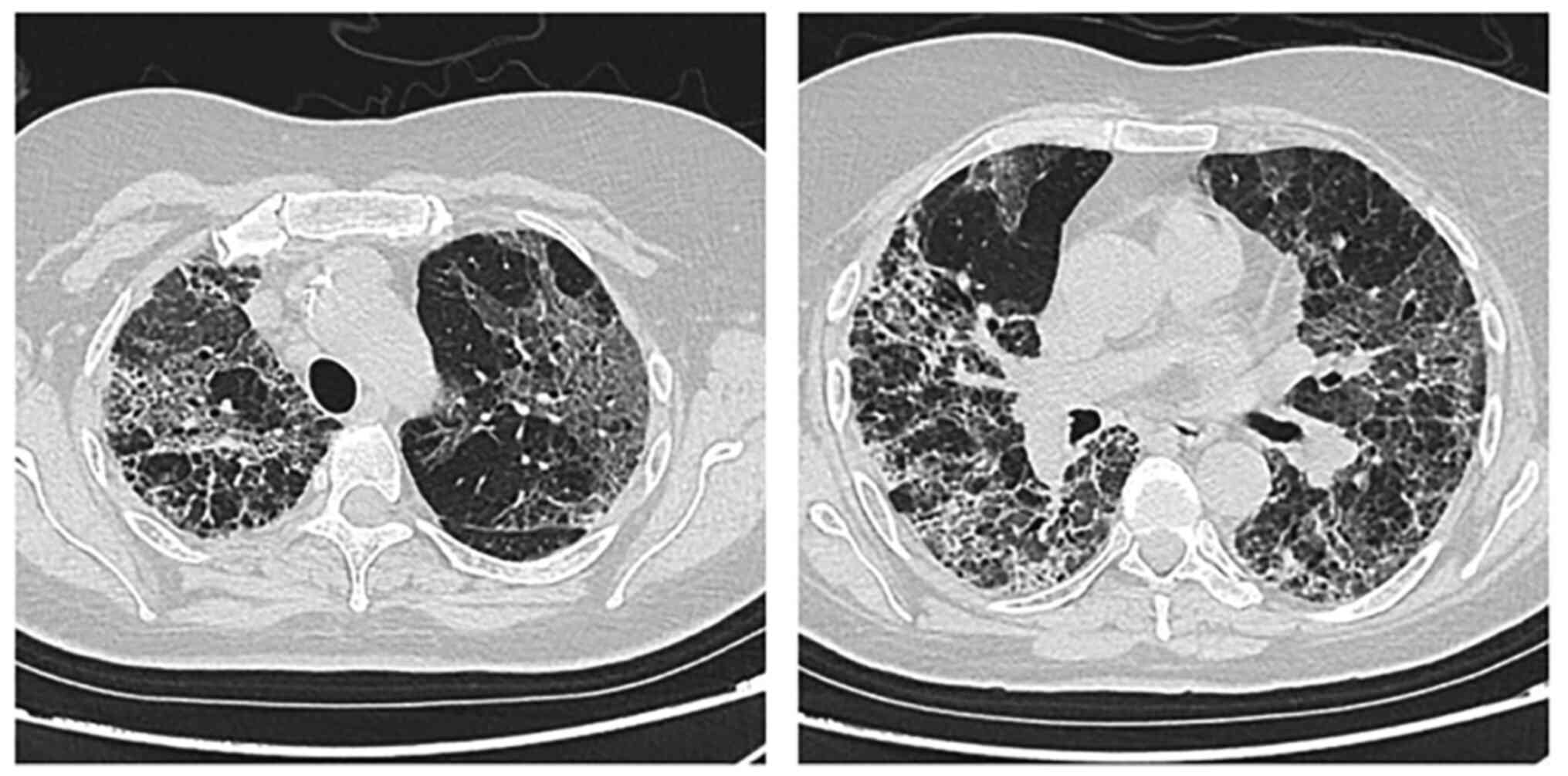
computed tomography scan was therefore performed, which ruled out a
pulmonary embolism and showed diffuse ground-glass opacities,
peripheral consolidation and bronchiectasis (Fig. 2). Blood cultures were positive for
Enterococcus faecium sensible to vancomycin, linezolid,
teicoplanin and tigecycline.
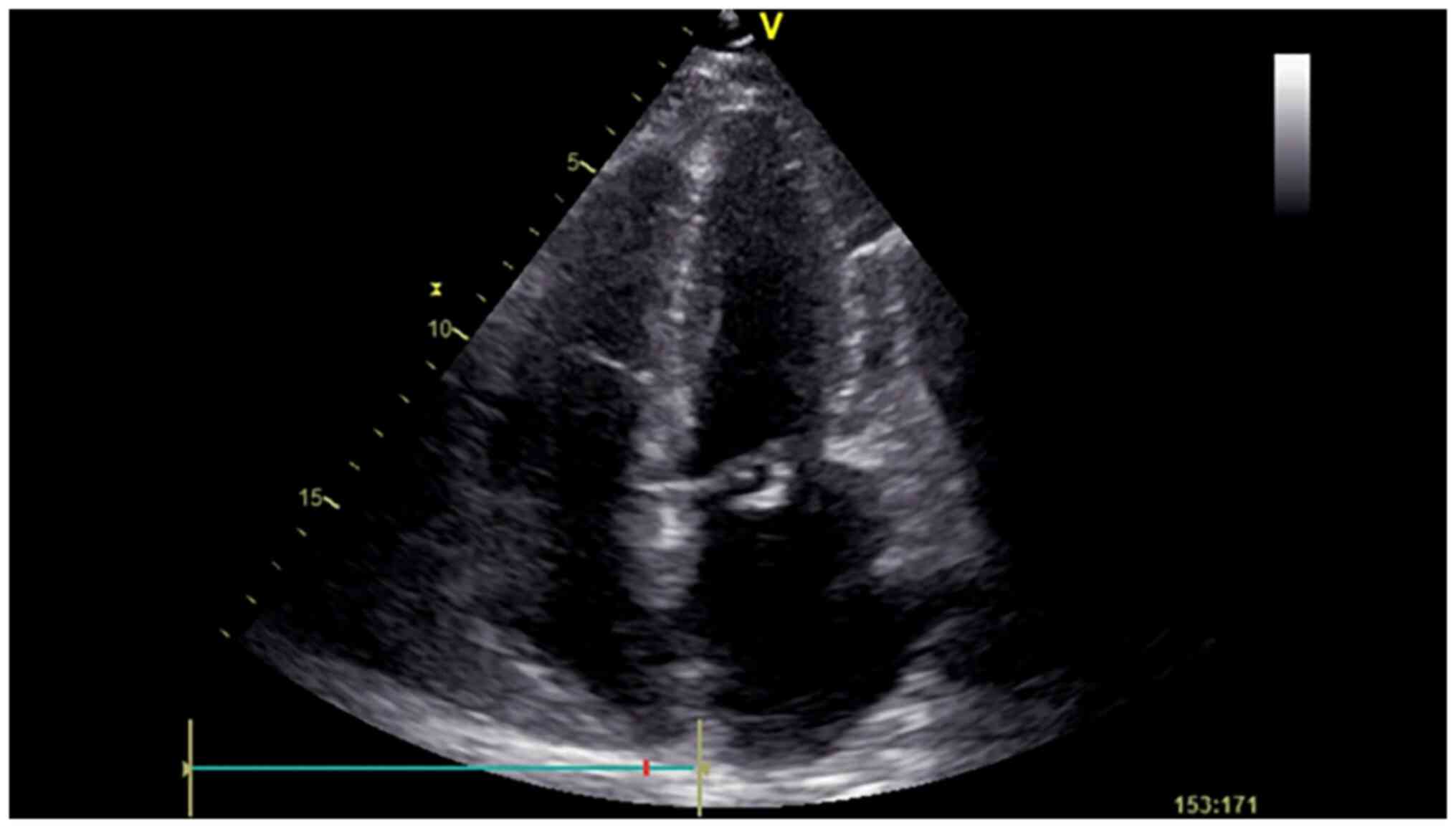
A transthoracic echocardiography was performed and
revealed vegetation on the posterior mitral valve leaflet measuring
14x5 mm, leading to moderate mitral regurgitation (Fig. 3). Infective endocarditis was defined
by the modified Duke criteria (4).
A positive diagnosis of infective endocarditis manifesting >48 h
after hospital admission was based on a positive Enterococcus
faecium blood culture and posterior mitral valve vegetation
revealed by transthoracic echocardiography.
Antibiotic therapy with 1,000 mg meropenem (3 times
daily) and 600 mg linezolid (2 times daily) was administered.
Initially the patient had a favorable evolution, with improvements
in symptomatology and inflammatory syndrome, but after 20 days of
treatment, thrombocytopenia (platelet count, 58,000/mm3)
developed. Thus, linezolid was switched to teicoplanin (12 mg/kg
body weight twice daily as the loading dose, followed by 12 mg/kg
body weight daily for the maintenance dose).
Despite antibiotic treatment, the patient developed
multiple organ system failure and died 25 days after admission.
Discussion
Bacterial endocarditis is a less frequent disease
among patients infected with SARS-CoV-2 compared to other cardiac
comorbidities in these patients. However, a study published by
Ramos-Martínez et al (5)
showed a higher incidence of hospital-acquired infective
endocarditis in a single hospital during the first 2 months of the
COVID-19 pandemic. Compared with the pre-pandemic period, when the
incidence was 0.3 patients/month, during March and April 2020, the
incidence was 2 patients/month (P=0.033). The etiology of infective
endocarditis was Enterococcus faecalis (2 cases),
Staphylococcus aureus (1 case) and Candida albicans
(1 case), with an in-hospital mortality rate of 50%.
It has also been observed that the etiology of
endocarditis associated with SARS-COV-2 infection is different from
the etiology of endocarditis in patients without SARS-COV-2
infection. Erdem et al (6)
performed a recent study on 867 patients diagnosed with bacterial
endocarditis and the most frequently involved germ was found to be
Staphylococcus aureus, followed by Streptococcus
viridans and Enterococci (untyped, E. Faecium and
E. Faecalis). Endocarditis can be overlooked in patients
with COVID-19, as SARS-CoV-2, acute respiratory distress syndrome
and inflammatory syndrome can mask the symptoms. Still, the
reported endocarditis cases revealed gram-negative germs,
especially Klebsiella pneumoniae and Enterococcus
faecium (7,8).
Echocardiography is the most useful imaging
technique to diagnose endocarditis due to its large availability,
accuracy and safety, allowing a comprehensive evaluation of cardiac
valves (9).
In the present study, the patient exhibited the risk
factors of advanced age, pre-existing cardiac comorbidities, type 2
diabetes mellitus, obesity, immunosuppressive therapy and central
venous catheterization upon admission. Similar to patients with
HIV, patients infected with COVID-19 can have multiple
comorbidities, which cause higher mortality rates compared with
that in the general population, in spite of therapeutic care
advances (10).
The number and severity of respiratory infections is
continuously increasing due to environmental changes and the lack
of vaccination (11). The mRNA
vaccine against SARS-COV-2, based on a new genetic method, turns
the human body into living laboratory (12). Although vaccination represents the
main weapon against the virus, prevention strategies such as early
detection of infected individuals, updating new discoveries and
social distancing still play a major role (13).
In conclusion, SARS-COV2-infected patients have a
higher risk of developing cardiovascular complications. This viral
infection is able to mask bacterial endocarditis. The management of
patients with severe of SARS-COV-2 infection who also exhibit
associated infectious endocarditis is difficult, especially in
mono-specialty hospitals (such as infectious diseases hospitals)
where access to cardiological investigations is limited. A complete
routine examination, including echocardiography, in patients with
severe SARS-COV-2 pneumonia admitted to intensive care units may be
required for the early diagnosis and treatment of infective
endocarditis. Bacterial endocarditis remains a major challenge in
patients infected with SARS-COV-2, with a poor prognosis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
MB is the corresponding author and participated in
the design of the case report. MD, NDV and EM were involved in
acquisition of data and described the patient evolution and
management. IMD, RCC and SR were involved in the analysis and
interpretation of data, and revising the final manuscript. All
authors confirm the authenticity of all the raw data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
Ethics approval was obtained from the Medical Ethics
Commission for Clinical Studies in the Constanta Clinical Hospital
of Infectious Diseases (approval no. 6/24.05.2021).
Patient consent for publication
Written informed consent was obtained from the
patient prior to publication at the time of admission.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Momtazmanesh S, Shobeiri P, Hanaei S,
Mahmoud-Elsayed H, Dalvi B and Malakan EM: Cardiovascular disease
in COVID-19: A systematic review and meta-analysis of 10,898
patients and proposal of a triage risk stratification tool. Egypt
Heart J. 72(41)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hessami A, Shamshirian A, Heydari K,
Pourali F, Alizadeh-Navaei R, Moosazadeh M, Abrotan S, Shojaie L,
Sedighi S, Shamshirian D and Rezaei N: Cardiovascular diseases
burden in COVID-19: Systematic review and meta-analysis. Am J Emerg
Med. 46:382–391. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Linschoten M, Peters S, van Smeden M,
Jewbali LS, Schaap J, Siebelink HM, Smits PC, Tieleman RG, van der
Harst P, van Gilst WH, et al: Cardiac complications in patients
hospitalised with COVID-19. Eur Heart J Acute Cardiovasc Care.
9:817–823. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Li JS, Sexton DJ, Mick N, Nettles R,
Fowler VG Jr, Ryan T, Bashore T and Corey GR: Proposed
modifications to the duke criteria for the diagnosis of infective
endocarditis. Clin Infect Dis. 30:633–638. 2000.PubMed/NCBI View
Article : Google Scholar
|
|
5
|
Ramos-Martínez A, Fernández-Cruz A,
Domínguez F, Forteza A, Cobo M, Sánchez-Romero I and Asensio A:
Hospital-acquired infective endocarditis during Covid-19 pandemic.
Infect Prev Pract. 2(100080)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Erdem H, Puca E, Ruch Y, Santos L,
Ghanem-Zoubi N, Argemi X, Hansmann Y, Guner R, Tonziello G,
Mazzucotelli JP, et al: Portraying infective endocarditis: Results
of multinational ID-IRI study. Eur J Clin Microbiol Infect Dis.
38:1753–1763. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hayes DE, Rhee DW, Hisamoto K, Smith D, Ro
R, Vainrib AF, Bamira D, Zhou F and Saric M: Two cases of acute
endocarditis misdiagnosed as COVID-19 infection. Echocardiography.
38:798–804. 2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Amir M, Djaharuddin I, Sudharsono A and
Ramadanyd S: COVID-19 concomitant with infective endocarditis: A
case report and review of management. Int J Infect Dis. 98:109–112.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Gurghean AL, Savulescu-Fiedler I and
Mihailescu A: Multiple cardiac complications after adjuvant therapy
for breast cancer: The importance of echocardiography. A case
report and review of the literature. Med Ultrason. 19:117–120.
2017.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Streinu-Cercel A, Săndulescu O, Poiană C,
Dorobanţu M, Mircescu G, Lăzureanu VE, Dumitru IM, Chirilă O and
Streinu-Cercel A: Extended Consensus Group. Consensus statement on
the assessment of comorbidities in people living with HIV in
Romania. Germs. 9:198–210. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dumitru IM, Lilios G and Arbune M:
Respiratory infections and air pollution, retrospective study over
the past 10 years. J Environ Prot Ecol. 19:1445–1451. 2018.
|
|
12
|
Calina D, Docea AO, Petrakis D, Egorov AM,
Ishmukhametov AA, Gabibov AG, Shtilman MI, Kostoff R, Carvalho F,
Vinceti M, et al: Towards effective COVID-19 vaccines: Updates,
perspectives and challenges (Review). Int J Mol Med. 46:3–16.
2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Docea AO, Tsatsakis A, Albulescu D,
Cristea O, Zlatian O, Vinceti M, Moschos SA, Tsoukalas D, Goumenou
M, Drakoulis N, et al: A new threat from an old enemy: Re-emergence
of coronavirus (Review). Int J Mol Med. 45:1631–1643.
2020.PubMed/NCBI View Article : Google Scholar
|