Introduction
Acquired benign trachea-oesophageal fistula is a
rare pathological entity that concerns the occurrence of an
abnormal connection between the oesophagus and the trachea that
does not involve the presence of local neoplastic pathology
(1). This fistula is common in
patients that were mechanically ventilated for longer periods of
time during which, most often, the hyperinflated cuff of the
endotracheal or tracheostomy tube is in contact with a nasogastric
tube placed in the oesophagus (1).
Other causes for this type of fistula are rare, but regardless of
the aetiology, the treatment is rather the same, aimed at closing
the tracheoesophageal connection and restoring the separate
permeability of the airways and digestive tube so as to avoid
pulmonary contamination and aspiration pneumonia (2). Certain cases required a tailored
approach, e.g. patients with complex local and pathological issues,
patients regarding whom we should try to ‘think outside the box’; a
rich experience in tracheal and oesophageal surgery is always
conducive to obtaining good results. Using an oesophageal patch for
the posterior tracheal wall, staged surgery and extending the
duration of high frequency jet ventilation (HFJV) anaesthesia are
among the strategies used in order to successfully treat the case
presented below.
Case report
A 31-year old female patient was admitted to the
Department of Thoracic Surgery, ‘Marius Nasta’ National Institute
of Pneumology (Bucharest, Romania) with quasi-complete dysphagia,
severe cough exacerbated with deglutition, purulent sputum and
fever, namely symptoms that began ~1 week before and got worse in
time.
The patient's history is relevant, as she was known
to have cicatricial oesophageal stenosis after accidentally
ingesting lye ~10 years ago. At the time, she received conservative
treatment, with the patient only requiring regular oesophageal
dilatations approximately once a year in the first 6 years.
Subsequently, as the stenosis relapsed more often and extended on
longer stretches of the oesophagus, the dilatation treatment had
come to be needed 3-4 times per year, so 2 years prior to this
admission, the decision was made to insert an oesophageal
stent.
It is important to note that the surgical option
(oesophageal plasty) was also offered to the patient, who refused
such treatment. The first fully covered metallic expandable stent
inserted was kept in place for ~2 months before it was removed,
following which the oesophagus restenosis (~3 months), another
similar, but longer stent was placed in the oesophagus. This
procedure was repeated four times. The last time, during the same
digestive endoscopy, the patient was also subject to percutaneous
gastrostomy; we do not know the actual reason for this procedure,
but it suggests that the gastroenterologist performing this
procedure was clearly unsatisfied with the results. This last
manoeuvre was performed 2 weeks before the symptoms started and 3
weeks before the patient was admitted to our clinic.
When she arrived, the patient was septic (fever and
cough with mucopurulent expectoration) and severely dehydrated, and
had been unable to properly swallow for several days. The imagistic
tests showed bilateral lung lesions (aspiration pneumonia), and the
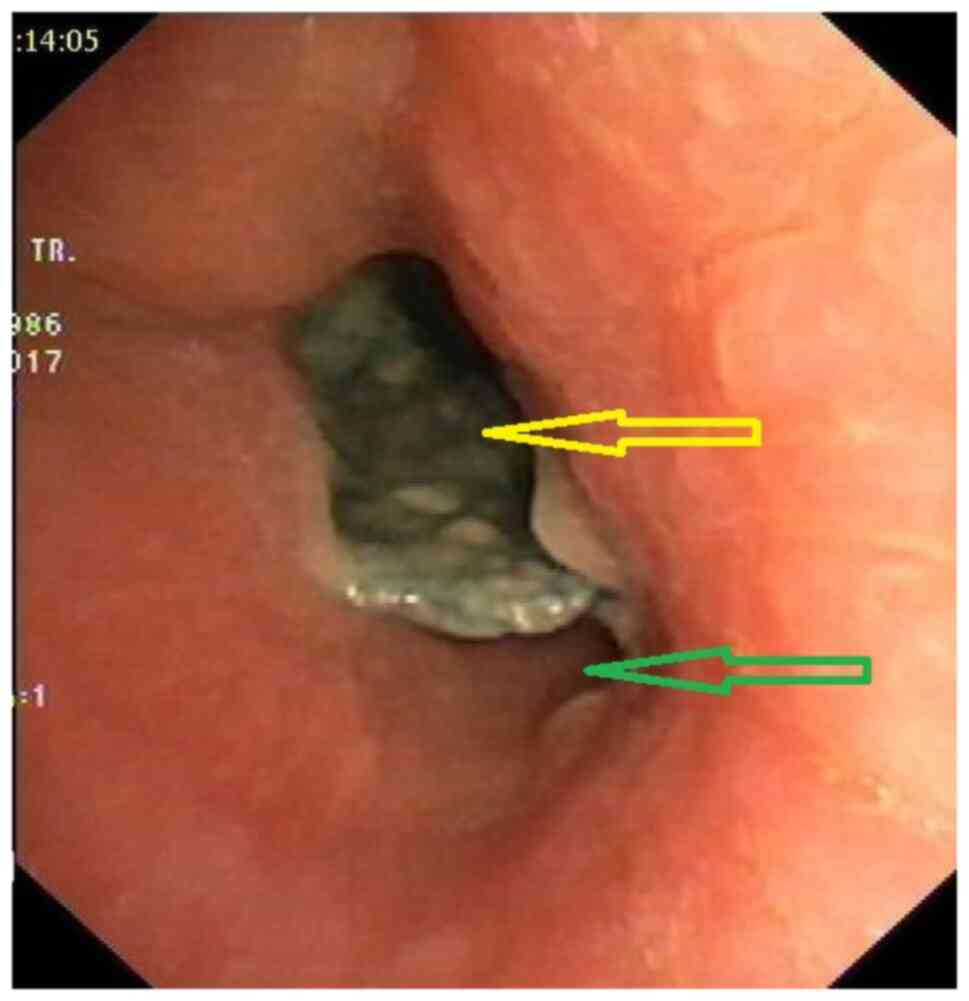
additional bronchoscopy exam indicated a tracheoesophageal fistula
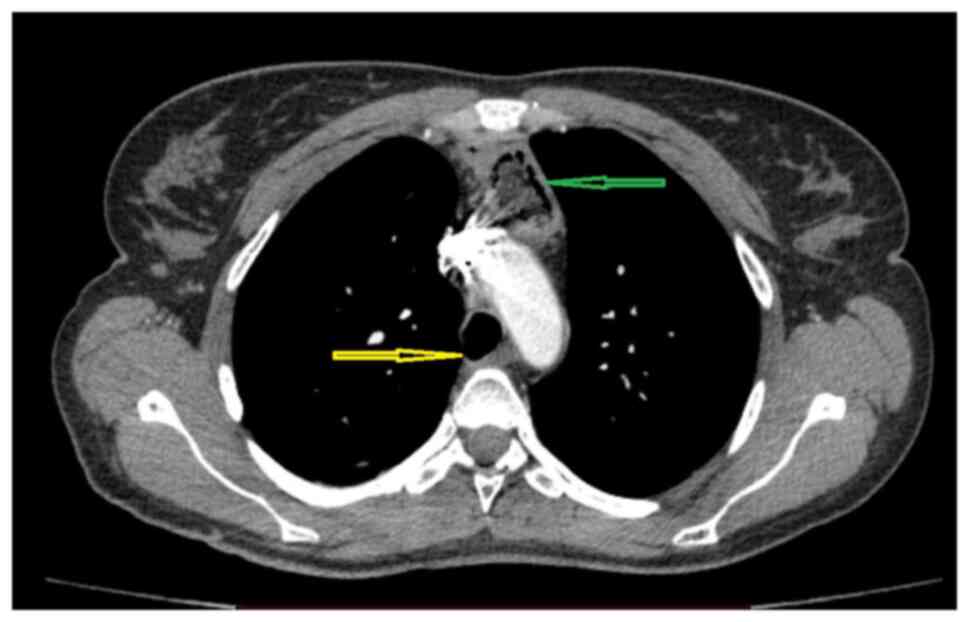
right above the carina, ~2.5 cm long, with the intratracheal
migration of the oesophageal stent (Fig. 1). The attempts to remove the stent
through the trachea or the oesophagus failed because of its size,
in the first case, and also because the proximal stenosis of the
oesophagus and, when pulled, the stent would cling to the tracheal
fistula. A rigorous cleaning of the trachea-bronchial tree was
conducted by means of bronchoaspiration.
The first steps taken consisted of completely
interrupting oral alimentation and through the gastrostomy tube-the
oesophageal reflux of the gastric content through the stent would
end up in the trachea, then in the lungs, and of starting total
parenteral nutrition. Measures were taken to correct the acid-base
and hydroelectrolyte balances, which were severely imbalanced;
broad spectrum antibiotics were administered to control the sepsis.
Practically, this first step was intended to simply improve the
health condition and reduce the effects of post-sepsis syndrome
with a view to conducting a surgery that would resolve the
connection between the airway and the digestive tube.
Following discussions about the case, it was decided
that oesophagectomy was the surgical option at the time. Also,
given the poor health status of the patient (BMI, 13.6; height, 165
cm; weight, 37 kg) who was also suffering from hypoproteinemia and
6 kg weight loss in the few weeks prior to admission (a significant
percentage) it was decided that the oesophagoplasty would be
performed at a later time, once the nutritional status of the
patient had improved.
Surgery was performed under general and epidural
anaesthesia while the patient was ventilated in a rather
unconventional manner; she was intubated using a rigid bronchoscope
and the ventilation on separate areas was performed with HFJV
associated with a bronchial blocker.
A standard posterolateral thoracotomy was used to
gain access to the right pleural cavity. First, an oesophagectomy
was performed to remove the bulky oesophageal stent that interfered
with the dissection. Prior to the surgery, the initial intent was
to conduct a total oesophagectomy and a standard lower tracheal
anastomosis resection. The peritracheal fibrosis and the very tight
existing adhesions turned the surgery into a partial
oesophagectomy, distal and proximal to the fistulous opening.
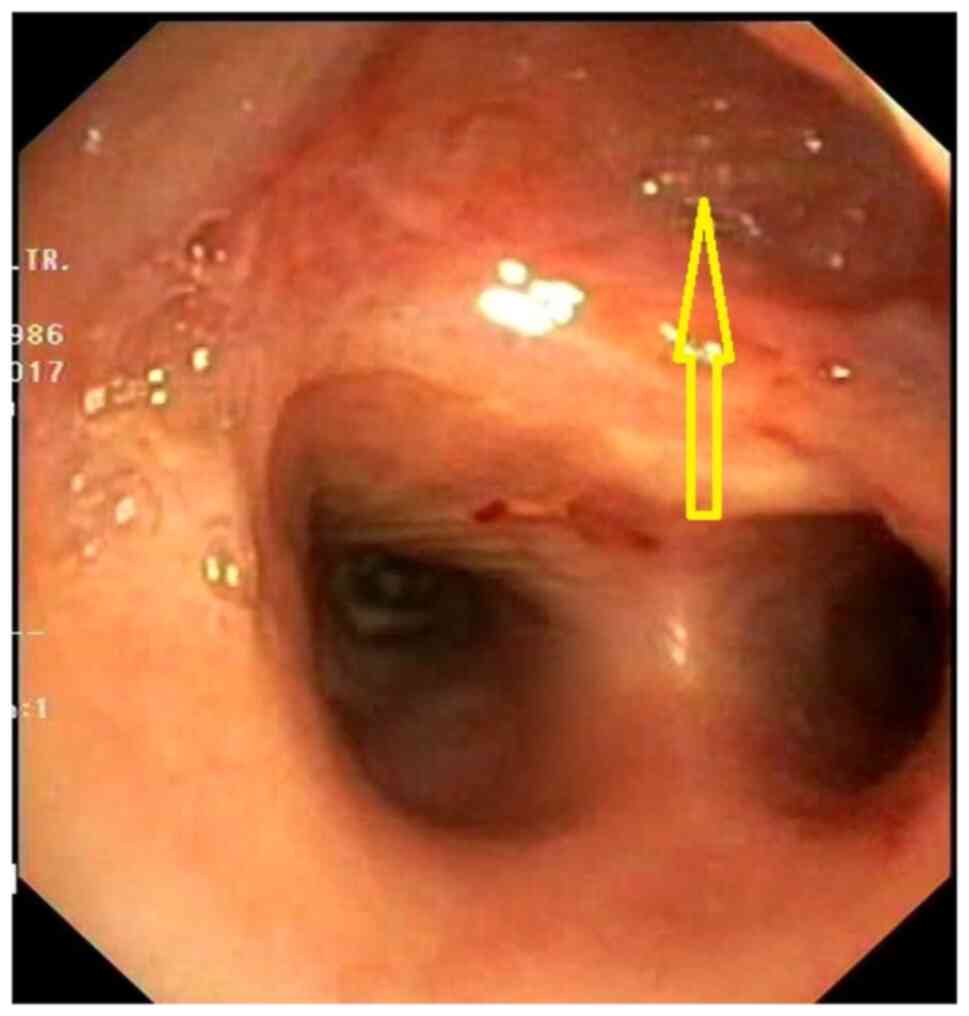
Practically, an ~4-cm long oesophagus fragment was left stuck to
the posterior trachea next to the fistulous opening, forming a sort
of tracheal diverticulum (Fig. 2).
A cervical esophagostomy completed the surgery.
The post-operative recovery was simple, the patient
was discharged on day 10 after surgery, with resolving aspiration
pneumonia and the inflammatory sepsis syndrome under control once
the lung contamination was stopped. It was jointly agreed that, for
~3 months, food should be administered through the existing
gastrostomy, while the patient was put on a nutritional and
respiratory recovery program before the oesophageal
reconstruction.
According to the agreement, after the set period,
the patient was admitted in order to restore the digestive tube
continuity. On admission, the nutritional status had improved (5 kg
weight gain), and the patient did not show hypoproteinemia; the
bilateral aspiration pneumonia was fully resolved during the
recovery period. Since the patient was young, with a benign
pathology, the oesophageal reconstruction was conducted with an
iso-peristaltic colonic loop vascularized on the middle colic
pedicle, with the graft in retrosternal position. The surgery was
successful, with standard post-operative recovery and discharge 10
days after surgery; this time, the patient was able to feed
normally.
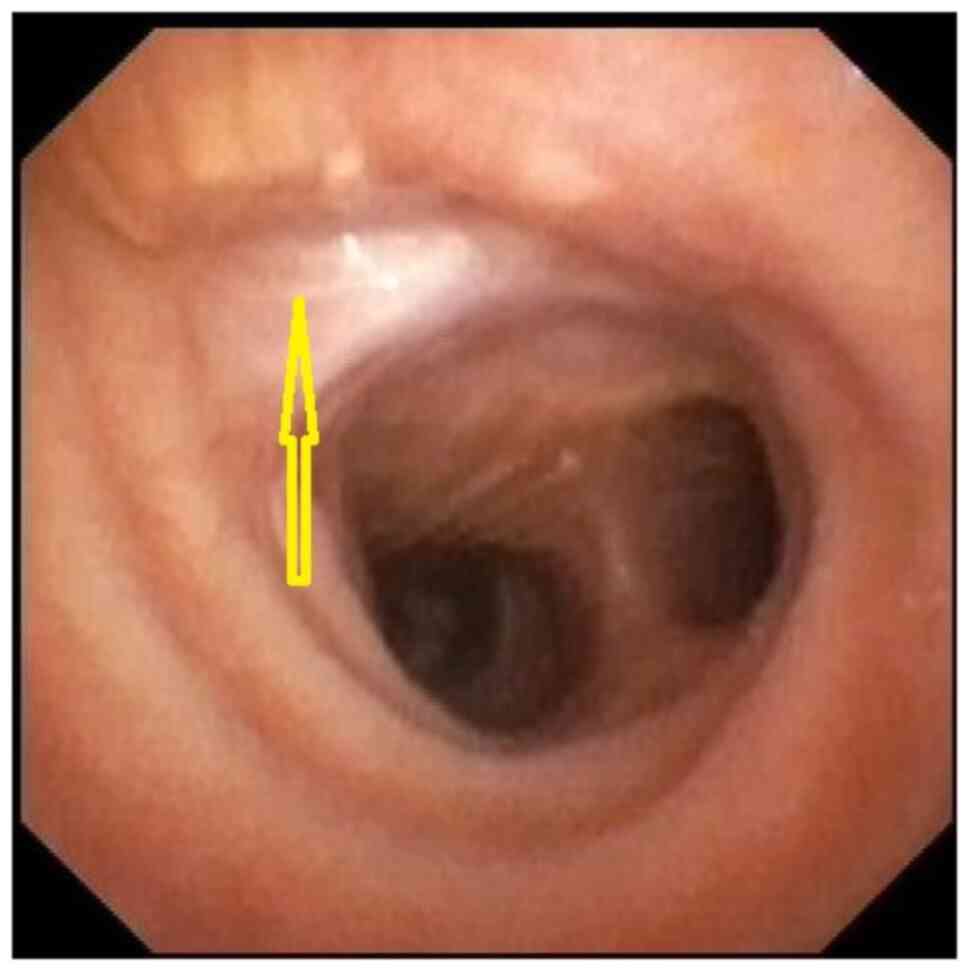
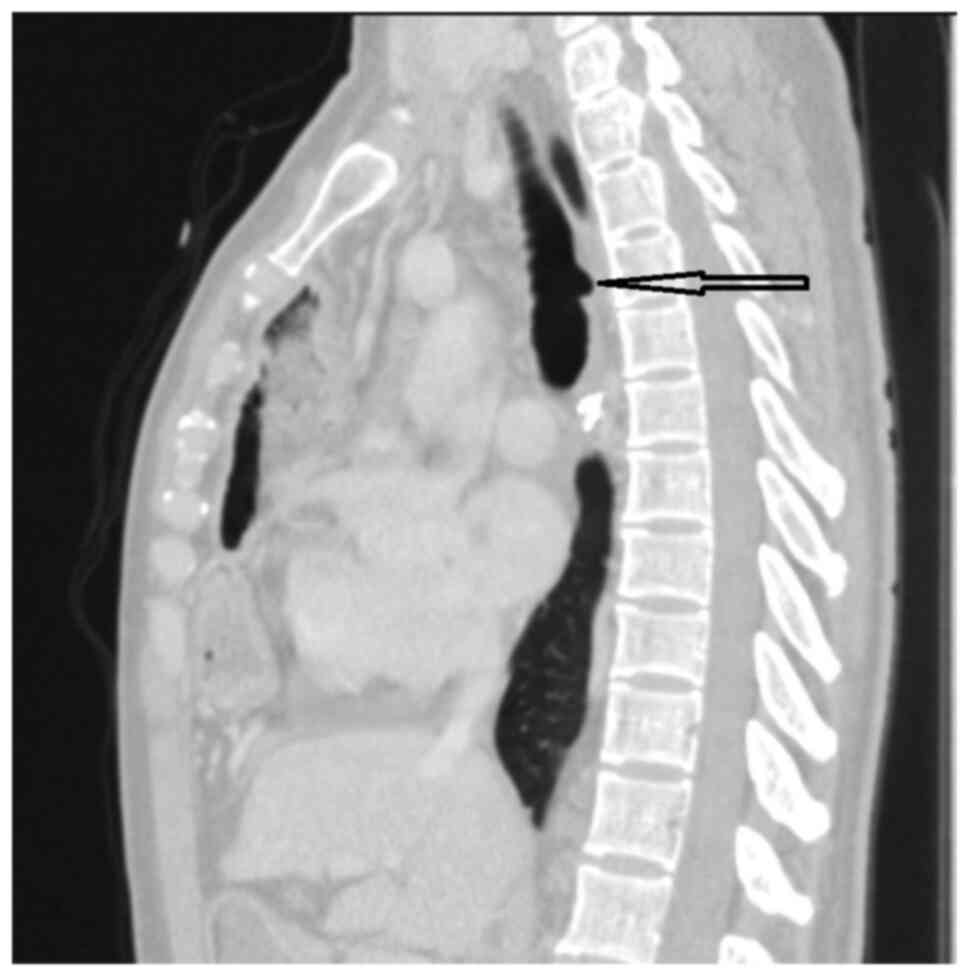
The images and bronchoscopy tests showed a reduction
to almost disappearance of the tracheal pseudo-diverticulum, with
complete scarring of the posterior side of the trachea (Fig. 3, Fig.
4 and Fig. 5). Three years
after surgery, the patient is in good health and has reintegrated
socio-professionally, with only minor feeding issues, which are
somehow normal in a patient with oesophageal plasty.
Discussion
Several aspects should be discussed in this case.
Post lye ingestion oesophageal stenoses are rather rare injuries
and it cannot be said there is an ideal or even standardized
treatment strategy. It depends on numerous factors, such as injury
size (meaning the length of the oesophagus that was affected),
organs involved (stenosis of the pharynx and stomach or laryngeal
problems may be associated), time since the initial injury (knowing
that it is an evolving condition) and type of ingested substance
(2). Thus, the management of post
lye ingestion oesophageal stenoses must be individualized to
consider all these factors, so it is more complex than it may look
at first glance.
Evidently, oesophageal resection surgery as the
first procedure is no longer an option without the acute
post-ingestion perforation; endoscopic follow-up with repetitive
dilatation is feasible as long as the need and frequency of the
procedures do not severely impair the patient's quality of life.
Inserting oesophageal stents may also be an option in certain
conditions, but keeping them in place long-term is not recommended,
as their presence may lead to complications, the most severe ones
being, just like we had in this case, the formation of fistulas,
particularly with the airway, but also with other intra-thoracic
organs (3-5).
Regarding the general anaesthesia that the patient
underwent for the first surgery, the airway could only be secured
by using a rigid bronchoscope as inserting a double-lumen tube was
impossible because the stent almost completely blocked the tracheal
lumen and could not be mobilized through flexible bronchoscopy
since it was encroached in the fistula opening. As ventilation on
separate areas is advisable in chest surgery, it was performed
using a bronchial blocker introduced through the rigid bronchoscope
in the right main bronchus.
The HFJV allowed for the ventilation of the left
lung during surgery, the total duration of the ventilation being 4
h. There is no time limit for using this type of ventilation, but
there is a risk of developing hypercapnia, a risk that increases
with the duration. Hypercapnia is caused by certain risk factors,
such as obesity and bronchospasm, which were absent in our patient,
as well as by ventilation parameters, specifically those that are
flow-related, which should be large enough to ‘wash’ CO2
(6). The patient showed no
hypercapnia at any moment during the 4 h of surgery and the blood
gases were monitored every 30 min using an arterial catheter. Our
department's extensive experience in tracheal surgery enabled us to
do all these technical methods (6-8),
the anaesthesia posed no distinct problems, even though the long
duration of the HFJV was unusual.
The partial oesophagectomy above and below the
fistula was not a technical first as we have performed this type of
surgery on other occasions too, but this was usually when the
fistula had extreme sizes that would not allow for the tracheal
resection, practically turning the oesophagus into a posterior wall
of the trachea; this procedure has also been successfully described
by other teams (1,9,10).
This was not our initial plan, but during the surgery, the fibrosis
found was extremely severe, not allowing for the safe dissection of
the oesophagus from the trachea. It is important to note that the
severity of the adhesions is linked to the repetitive dilatation
treatment and repetitive stenting rather than the lye injury itself
because oesophagectomies for lye stenoses have been performed
before in the clinic, but the peri-oesophageal adhesions have not
been comparable in severity.
There is great variability of practice worldwide
when it comes to oesophageal resection and reconstruction (11). Resections are mostly performed for
oesophageal malignancies and very rarely for benign diseases. Most
procedures performed are minimally invasive and we are aware of the
benefits brought by such a surgical approach (12). The stomach is commonly used as the
organ for oesophagoplasty and the surgery is performed in one go.
In our case, the open approach was used in order to associate a
potential lower tracheal resection, but even without it, performing
the partial oesophagectomy through a minimally invasive procedure
under the given circumstances, with extended fibrosis and very
tight adhesions, would have likely been extremely difficult.
At present, staged surgery, namely performing
resection and interval oesophageal reconstruction is very rarely
considered. The patient's poor health and the fact that we planned
to perform the oesophageal reconstructions using the colon, which
meant a more laborious procedure than the usual one where the
reconstruction is performed with the stomach, steered us towards
this decision. Using the colon as the oesophageal substitute is an
option when replacing the oesophagus for a benign disease in a
young patient, in which case it is assumed the graft will be kept
in place long-term.
To conclude, complex cases always require a tailored
approach. This case demonstrated that ventilating a patient with
HFJV for a long period of time (4 h in this case) is possible,
provided that blood gases are carefully monitored to correct
hypercapnia if required. Replacing the posterior tracheal wall with
an oesophageal patch proved once again to be a feasible option in
selected cases. Splitting the procedure into a resection stage
followed by subsequent reconstruction (staged surgery) should be
considered when the patient is not healthy enough to undergo
serious reconstructive surgery.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CB and IC were involved in the study conception and
design, data collection and analysis and the writing of the
manuscript. IB, NB and AM participated in the writing of the
manuscript, data collection and data analysis. FF and CP were
involved in the study design and data analysis. MA, GCa and FG were
involved in data analysis. AB, GCo, IB, AM and DR were involved in
study conception and design. All authors have read and approved the
final manuscript. CB, IC and NB confirm the authenticity of the raw
data.
Ethics approval and consent to
participate
The study was conducted according to the guidelines
of the Declaration of Helsinki, and approved by the Ethics
Committee of ‘Marius Nasta’ National Institute of Pneumology
(approval no. 292/12.06.2020; Bucharest, Romania). Informed consent
was obtained and signed by the patient on 11.06.2020.
Patient consent for publication
The patient provided written informed consent for
the publication of the case details and associated images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Reed MF and Mathisen DJ: Tracheoesophageal
fistula. Chest Surg Clin N Am. 13:271–289. 2003.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Shu YS, Sun C, Shi WP, Shi HC, Lu SC and
Wang K: Tubular stomach or whole stomach for esophagectomy through
cervico-thoraco-abdominal approach: A comparative clinical study on
anastomotic leakage. Ir J Med Sci. 182:477–480. 2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bakken JC, Wong Kee Song LM, de Groen PC
and Baron TH: Use of a fully covered self-expandable metal stent
for the treatment of benign esophageal diseases. Gastrointest
Endosc. 72:712–720. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Farkas ZC, Pal S, Jolly GP, Lim MMD, Malik
A and Malekan R: Esophagopericardial fistula and pneumopericardium
from caustic ingestion and esophageal stent. Ann Thorac Surg.
107:e207–e208. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Aneeshkumar S, Sundararajan L, Santosham
R, Palaniappan R and Dhus U: Erosion of esophageal stent into left
main bronchus causing airway compromise. Lung India. 34:76–78.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Cordos I, Bolca C, Paleru C, Posea R and
Stoica R: Sixty tracheal resections-single center experience.
Interact Cardiovasc Thorac Surg. 8:62–65. 2009.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Couraud L, Jougon JB and Velly JF:
Surgical treatment of nontumoral stenoses of the upper airway. Ann
Thorac Surg. 60:250–260. 1995.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Stoica RT, Cordos I and Popescu WM:
Anesthetic considerations for tracheobronchial resection in
oncologic surgery. Curr Opin Anaesthesiol. 33:55–63.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
He J, Chen M, Shao W and Wang D: Surgical
management of huge tracheo-oesophageal fistula with oesophagus
segment in situ as replacement of the posterior membranous wall of
the trachea. Eur J Cardiothorac Surg. 36:600–602. 2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
de Castro G, Iribarren M, Rivo E, Meléndez
R, Nóvoa E, Cañizares M and Gil P: Tracheoesophageal fistula in an
intubated patient. Treatment through exclusion and esophageal
patch. Cir Esp. 77:230–232. 2005.PubMed/NCBI View Article : Google Scholar : (In Spanish).
|
|
11
|
Oesophago-Gastric Anastomosis Study Group
on behalf of the West Midlands Research Collaborative.
International variation in surgical practices in units performing
oesophagectomy for oesophageal cancer: A unit survey from the
oesophago-gastric anastomosis audit (OGAA). World J Surg.
43:2874–2884. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Erus S, Öztürk AB, Albayrak Ö, İncir S,
Kapdağlı MH, Cesur EE, Yavuz Ö, Tanju S and Dilege Ş: Immune
profiling after minimally invasive lobectomy. Interact Cardiovasc
Thorac Surg. 32:291–297. 2021.PubMed/NCBI View Article : Google Scholar
|