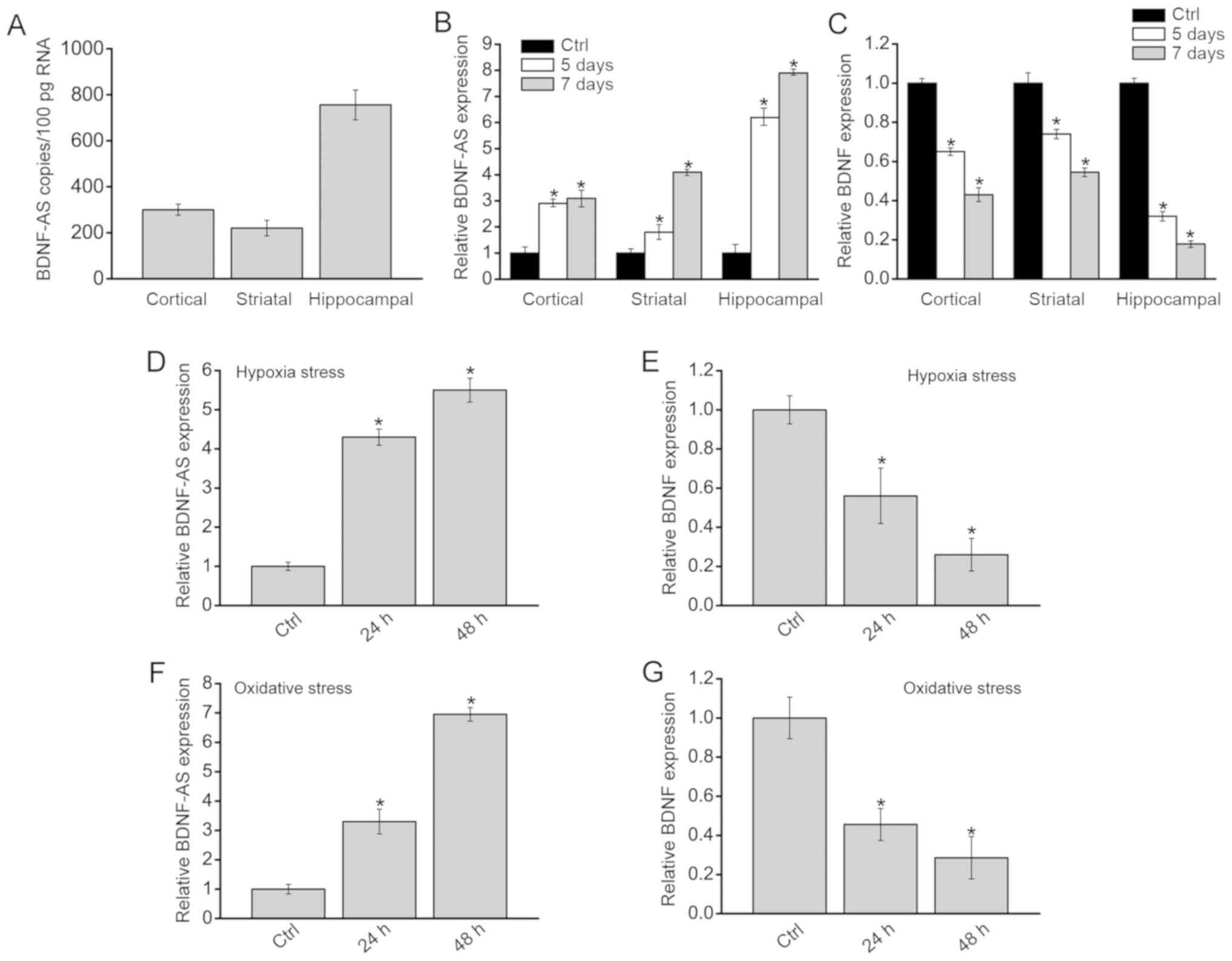
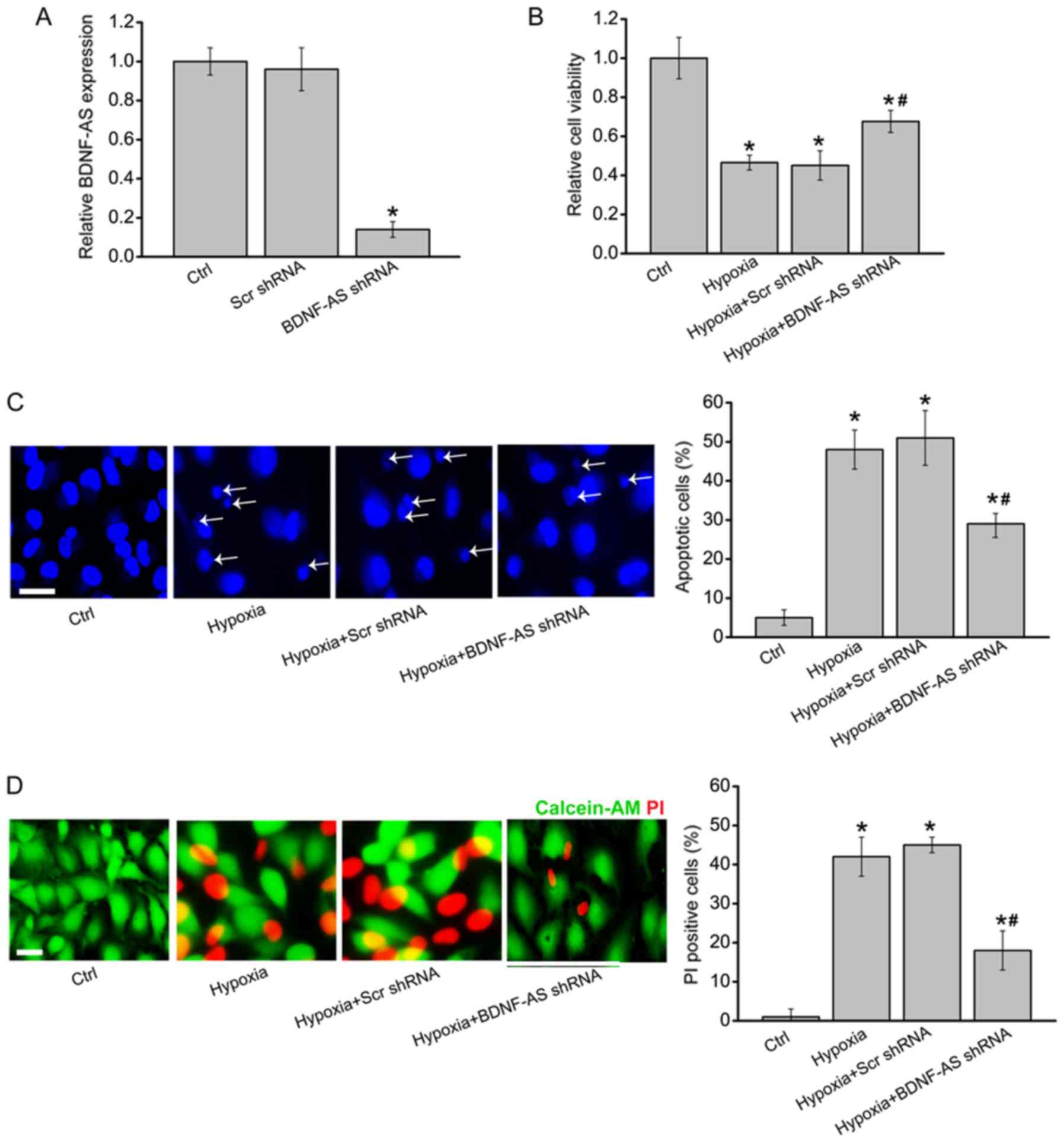
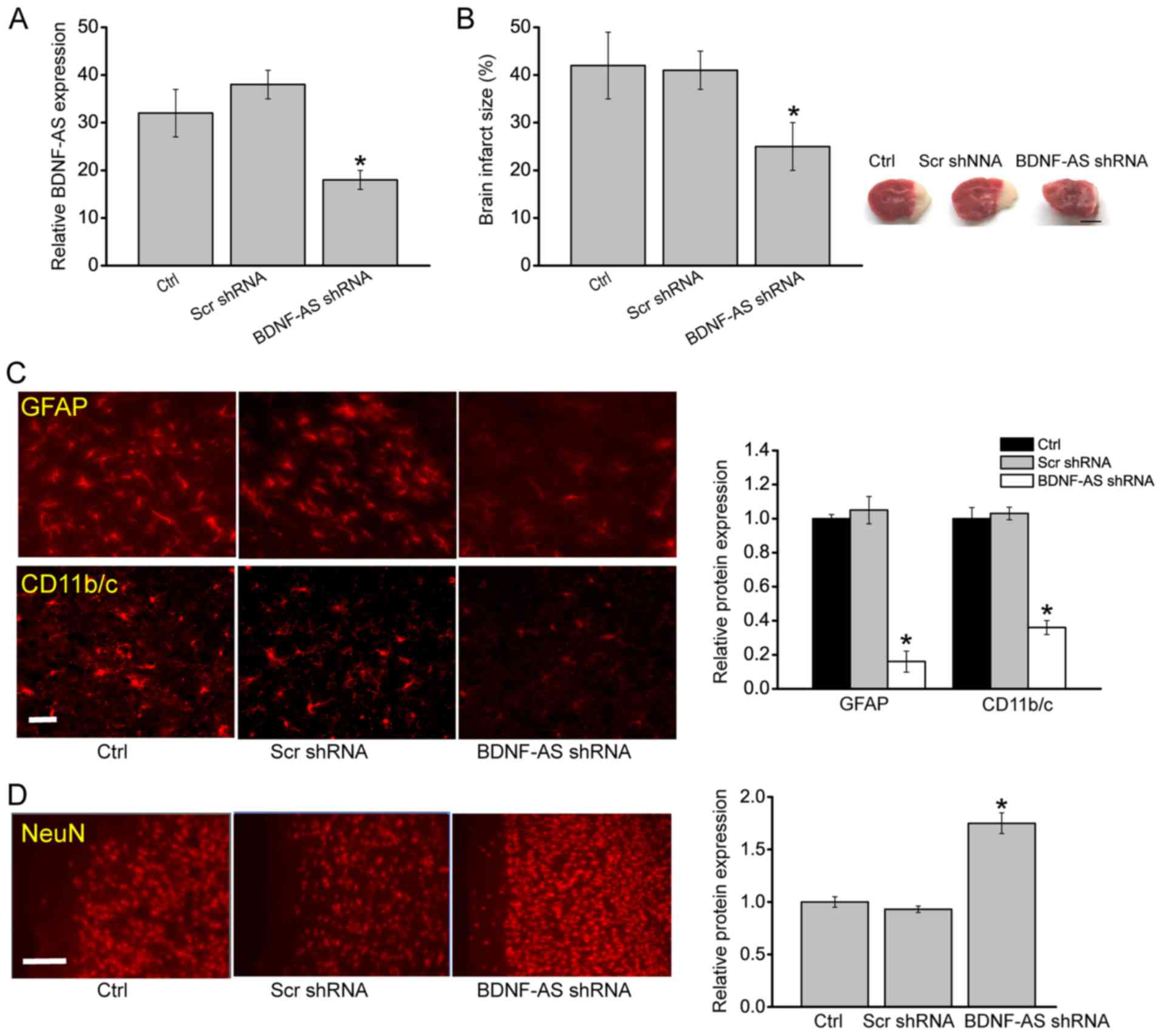
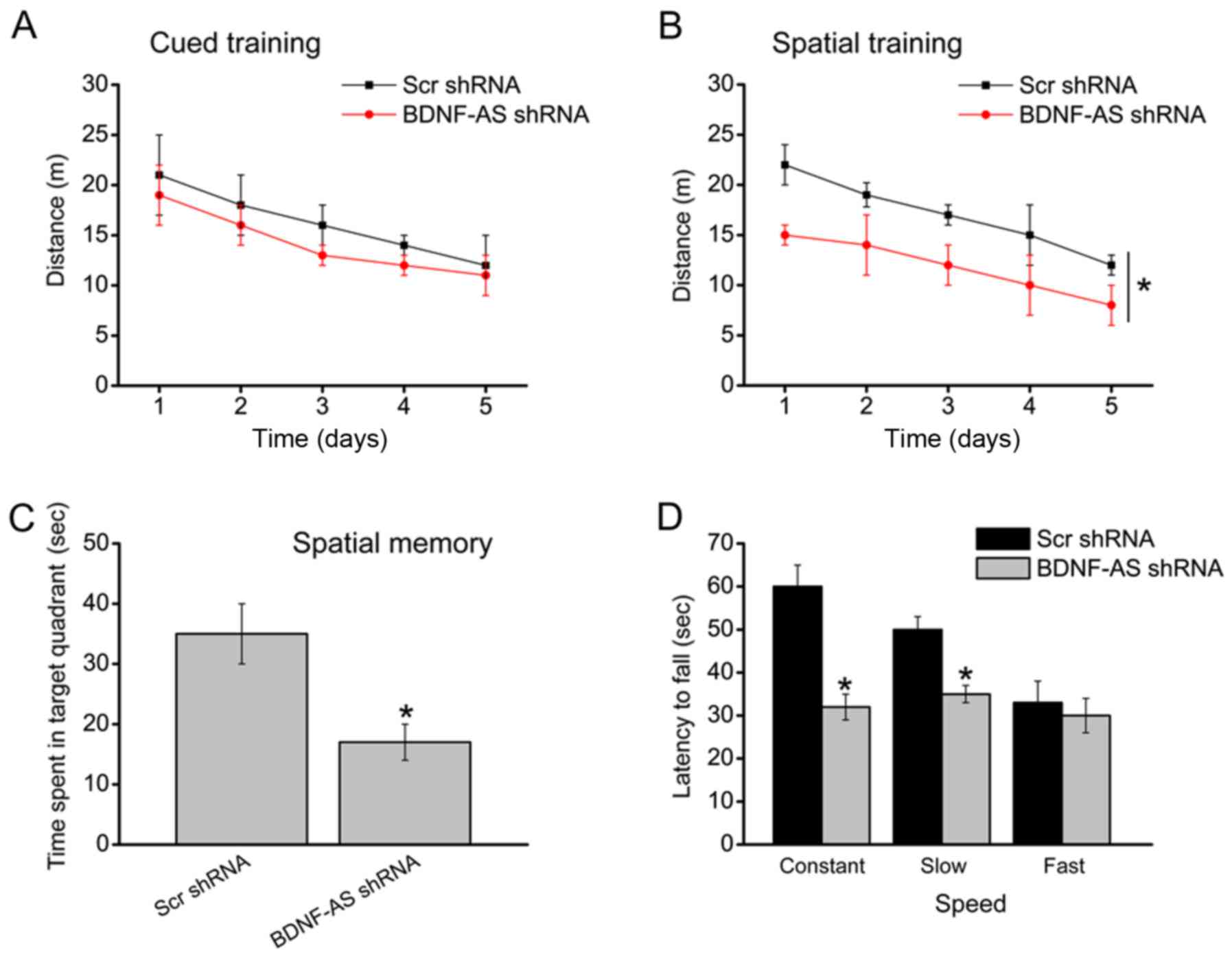
|
1
|
Zhu C, Kang W, Xu F, Cheng X, Zhang Z, Jia
L, Ji L, Guo X, Xiong H, Simbruner G, et al: Erythropoietin
improved neurologic outcomes in newborns with hypoxic-ischemic
encephalopathy. Pediatrics. 124:e218–e226. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rocha-Ferreira E and Hristova M:
Antimicrobial peptides and complement in neonatal hypoxia-ischemia
induced brain damage. Front Immunol. 6:562015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johnston MV, Trescher WH, Ishida A and
Nakajima W: Neurobiology of hypoxic-ischemic injury in the
developing brain. Pediat Res. 49:735–741. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Blackmon LR and Stark AR: American Academy
of Pediatrics Committee on Fetus and Newborn: Hypothermia: A
neuroprotective therapy for neonatal hypoxic-ischemic
encephalopathy. Pediatrics. 117:942–948. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tagin MA, Woolcott CG, Vincer MJ, Whyte RK
and Stinson DA: Hypothermia for neonatal hypoxic ischemic
encephalopathy: An updated systematic review and meta-analysis.
Arch Pediat Adol Med. 166:558–566. 2012. View Article : Google Scholar
|
|
6
|
Quinn JJ and Chang HY: Unique features of
long non-coding RNA biogenesis and function. Nat Rev Genet.
17:47–62. 2016. View Article : Google Scholar
|
|
7
|
Mercer TR, Dinger ME and Mattick JS: Long
non-coding RNAs: Insights into functions. Nat Rev Genet.
10:155–159. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fatica A and Bozzoni I: Long non-coding
RNAs: New players in cell differentiation and development. Nat Rev
Genet. 15:7–21. 2014. View
Article : Google Scholar
|
|
9
|
Zhang J, Yuan L, Zhang X, Hamblin MH, Zhu
T, Meng F, Li Y, Chen YE and Yin KJ: Altered long non-coding RNA
transcriptomic profiles in brain microvascular endothelium after
cerebral ischemia. Exp Neurol. 277:162–170. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhao F, Qu Y, Liu J, Liu H, Zhang L, Feng
Y, Wang H, Gan J, Lu R and Mu D: Microarray profiling and
co-expression network analysis of LncRNAs and mRNAs in neonatal
rats following hypoxic-ischemic brain damage. Sci Rep. 5:138502015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mehta SL, Kim T and Vemuganti R: Long
noncoding RNA FosDT promotes ischemic brain injury by interacting
with REST-associated chromatin-modifying proteins. J Neurosci.
35:16443–16449. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhao RB, Zhu LH, Shu JP, Qiao LX and Xia
ZK: GAS5 silencing protects against hypoxia/ischemia-induced
neonatal brain injury. Biochem Biophys Res Commun. 497:285–291.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang X, Tang X, Liu K, Hamblin MH and Yin
KJ: Long noncoding RNA Malat1 regulates cerebrovascular pathologies
in ischemic stroke. J Neurosci. 37:1797–1806. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Rossi C, Angelucci A, Costantin L, Braschi
C, Mazzantini M, Babbini F, Fabbri ME, Tessarollo L, Maffei L,
Berardi N and Caleo M: Brain-derived neurotrophic factor (BDNF) is
required for the enhancement of hippocampal neurogenesis following
environmental enrichment. Eur J Neurosci. 24:1850–1856. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Angelucci F, Mathe AA and Aloe L:
Neurotrophic factors and CNS disorders: Findings in rodent models
of depression and schizophrenia. Prog Brain Res. 146:151–165. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Y, Xu F, Xiao H and Han F: Long
noncoding RNA BDNF-AS inversely regulated BDNF and modulated
high-glucose induced apoptosis in human retinal pigment epithelial
cells. J Cell Biochem. 119:817–823. 2018. View Article : Google Scholar
|
|
17
|
Zheng X, Lin C, Li Y, Ye J, Zhou J and Guo
P: Long noncoding RNA BDNF-AS regulates ketamine-induced
neurotoxicity in neural stem cell derived neurons. Biomed
Pharmacother. 82:722–728. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang Y, Yan L, Cao Y, Kong G and Lin C:
Long noncoding RNA BDNF-AS protects local anesthetic induced
neurotoxicity in dorsal root ganglion neurons. Biomed Pharmacother.
80:207–212. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Guo CC, Jiao CH and Gao ZM: Silencing of
lncRNA BDNF-AS attenuates Aβ25-35-induced neurotoxicity in PC12
cells by suppressing cell apoptosis and oxidative stress. Neurol
Res. 40:795–804. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cetin A, Komai S, Eliava M, Seeburg PH and
Osten P: Stereotaxic gene delivery in the rodent brain. Nat Protoc.
1:3166–3173. 2006. View Article : Google Scholar
|
|
21
|
Klaunberg BA, O'malley J, Clark T and
Davis JA: Euthanasia of mouse fetuses and neonates. Contemp Top Lab
Anim Sci. 43:29–34. 2004.PubMed/NCBI
|
|
22
|
Artwohl J, Brown P, Corning B and Stein S:
Report of the ACLAM task force on rodent euthanasia. J Am Assoc Lab
Anim Sci. 45:98–105. 2006.PubMed/NCBI
|
|
23
|
Kramer M, Dang J, Baertling F, Denecke B,
Clarner T, Kirsch C, Beyer C and Kipp M: TTC staining of damaged
brain areas after MCA occlusion in the rat does not constrict
quantitative gene and protein analyses. J Neurosci Methods.
187:84–89. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
25
|
Merritt JR and Rhodes JS: Mouse genetic
differences in voluntary wheel running, adult hippocampal
neurogenesis and learning on the multi-strain-adapted plus water
maze. Behav Brain Res. 280:62–71. 2015. View Article : Google Scholar
|
|
26
|
Yin KJ, Hamblin M and Chen YE: Non-coding
RNAs in cerebral endothelial pathophysiology: Emerging roles in
stroke. Neurochem Int. 77:9–16. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Xin JW and Jiang YG: Long noncoding RNA
MALAT1 inhibits apoptosis induced by oxygen-glucose deprivation and
reoxygenation in human brain microvascular endothelial cells. Exp
Ther Med. 13:1225–1234. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang H, Li D, Zhang Y, Li J, Ma S, Zhang
J, Xiong Y, Wang W, Li N and Xia L: Knockdown of lncRNA BDNF-AS
suppresses neuronal cell apoptosis via downregulating miR-130b-5p
target gene PRDM5 in acute spinal cord injury. RNA Biol.
15:1071–1080. 2018.PubMed/NCBI
|
|
29
|
Hagg T: Molecular regulation of adult CNS
neurogenesis: An integrated view. Trends Neurosci. 28:589–595.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Waterhouse EG, An JJ, Orefice LL, Baydyuk
M, Liao GY, Zheng K, Lu B and Xu B: BDNF promotes differentiation
and maturation of adult-born neurons through GABAergic
transmission. J Neurosci. 32:14318–14330. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lu B, Nagappan G and Lu Y: BDNF and
synaptic plasticity, cognitive function, and dysfunction. Handb Exp
Pharmacol. 220:223–250. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yoshii A and Constantine-Paton M:
Postsynaptic BDNF-TrkB signaling in synapse maturation, plasticity,
and disease. Dev Neurobiol. 70:304–322. 2010.PubMed/NCBI
|
|
33
|
Vannucci SJ and Hagberg H:
Hypoxia-ischemia in the immature brain. J Exp Biol. 207:3149–3154.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kumral A, Gonenc S, Acikgoz O, Sonmez A,
Genc K, Yilmaz O, Gokmen N, Duman N and Ozkan H: Erythropoietin
increases glutathione peroxidase enzyme activity and decreases
lipid peroxidation levels in hypoxic-ischemic brain injury in
neonatal rats. Biol Neonate. 87:15–18. 2005. View Article : Google Scholar
|
|
35
|
Vexler ZS and Ferriero DM: Molecular and
biochemical mechanisms of perinatal brain injury. Semin Neonatol.
6:99–108. 2001. View Article : Google Scholar : PubMed/NCBI
|