Introduction
Estrogen receptors (ERα and ERβ) are members of the
super-family of nuclear steroid hormone receptors (1). After the binding of hormone to these
receptors, the hormone-receptor complexes directly bind to specific
sequences on the DNA, named estrogen response elements (EREs), or
interact with other transcription factors (2–4). ERα
and ERβ are expressed both in normal and cancerous breast
epithelium (5). It has been
reported that a high ERα:ERβ ratio was responsible for enhanced
cell proliferation (6). Consistent
with the critical role of ERα in mediating the proliferative effect
of estrogen, mice that selectively lacked ERα showed a severely
under-developed mammary epithelium with only rudimentary ductal
structure (7,8). In 2003, Shaaban et al
demonstrated that the malignant progression of breast lesion was
correlated with elevated ERα expression (9). Therefore, selective disruption of ERα
signaling in breast cancer cells may be a potent therapy to prevent
the development of breast cancers.
Current selective estrogen receptor modulators
(SERMs) exhibit their effects through three different ways
(10): i) interfering with
estrogen receptor α (ERα) binding, such as tamoxifen and faslodex;
ii) inhibiting endogenous estrogen production, such as
oophorectomy, anastrozole and letrozole; and iii) down-regulating
ERα protein level, such as fulvestrant. SERMs may exert estrogen
agonist action in some target tissues while acting as estrogen
agonist action in others (11).
Therapeutical SERMs are considered to be able to inhibit the
proliferation of breast cancer without the influence on bone
density, cholesterol level or uterine endometrium (12,13).
Natural products have long been considered as a rich
source of novel bioactive chemicals providing potential effective
new drugs. Epidemiological and physiological studies have shown
that phytochemicals from vegetables and fruits possess potent
anticancer activities with minimal to no side-effect (14). Huaier extract is a kind of
officinal fungi and has been used in China for nearly 1,600 years.
In the past few years, its anticancer activities are attracting
increasing interest. Our previous studies showed that the antitumor
effect of Huaier extract on breast cancer may be associated with
induction of apoptosis and inhibition of angiogenesis (15,16).
Similar effects were seen in human liver cancer (17–19).
In view of these findings, it was of great interest to determine
whether Huaier extract could affect the ER signaling pathway in
human breast cancer cells.
In this study, we used three ERα-positive breast
cancer cell lines (MCF-7, T47D and ZR-75-1). Our findings
demonstrated that Huaier extract markedly decreased expression of
both ERα and its downstream genes, inhibited the
estrogen-stimulated proliferation and reversed the estrogen-induced
activation of the NF-κB pathway. These effects provided a further
rationale for the use of Huaier extract in the prevention and
treatment of ERα-dependent human breast cancers.
Materials and methods
Cell cultures and reagents
Human breast cancer cell lines, MCF-7, T47D and
ZR-75-1, were purchased from ATCC (Manassas, VA, USA). MCF-7 cells
were routinely maintained in DMEM medium (Gibco-BRL, Rockville, IN,
USA) supplemented with 10% fetal bovine serum (Haoyang Biological
Manufacture Co. Ltd., Tianjin, China), 100 U/ml penicillin and 100
μg/ml streptomycin. T47D cells were cultured in RPMI-1640
medium (Gibco-BRL) with 10% fetal bovine serum and 10 μg/ml
bovine insulin (Sigma-Aldrich, St. Louis, MO, USA). ZR-75-1 cells
were cultured in the above medium in the absence of bovine insulin.
For estrogen-free experiments, medium was changed to phenol
red-free DMEM or RPMI-1640 with 0.5% dextran-coated charcoal (DCC)
stripped FBS (Haoyang Biological Manufacture) for 72 h. The medium
was then changed to the indicated serum and phenol red-free medium
for 24 h before various treatments as described in the figure
legends (20,21). Electuary ointment of Huaier extract
was kindly provided by Gaitianli Medicine Co. Ltd. (Jiangsu,
China). The stock solutions were generated by dissolving electuary
ointment in indicated medium (15,16).
MG 132 was purchased from Sigma-Aldrich. β-estradiol (E2) was
obtained from Wako and was dissolved in ethanol with a final
concentration of ethanol in medium of less than 0.1%. Anti-ERα
antibody was from Dako. All other antibodies were purchased from
Cell Signaling Technology.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
assay
Cells were plated into 96-well plates and grown
overnight at 37°C in 5% CO2 for 24 h. After treatment
with the appropriate concentration of Huaier extract for indicated
time, 20 μl of MTT solution (5 mg/ml) was added to each well
and incubated for 4–6 h at 37°C. The formazan crystal was dissolved
in DMSO for 10 min. Optical density (OD) was measured by the
Microplate Reader (Bio-Rad, Hercules, CA, USA).
For E2-stimulated proliferation, cells were seeded
in 96-well plates in phenol red-free medium in the presence of 10%
charcoal-stripped fetal bovine serum. The next day, the medium was
changed to fresh serum and phenol red-free medium for another 24 h.
Then 10 nM E2 in the presence or absence of different
concentrations of Huaier extract was added for 48 h.
Western blot analysis
For western blot analysis, treated cells were washed
with ice-cold PBS and lysed in a modified RIPA buffer (1X PBS, 1%
NP-40, 0.1% sodium dodecyl sulfate, 5 mM EDTA, 0.5% sodium
deoxycholate and 1 mM sodium orthovanadate) with protease
inhibitors for 20 min at 4°C. After centrifugation at 12,000 rpm
for 15 min at 4°C, the supernatant was collected. Subsequently, 50
μg of total cellular protein from each sample were separated
by 10% SDS-PAGE and electrotransferred onto a polyvinylidene
fluoride (PVDF) membranes by a semi-dry blotting apparatus
(Bio-Rad). Then the membranes were immunoblotted with primary
antibodies followed by horseradish peroxidase-conjugated secondary
antibodies (1:7,000). Labeled protein spots were visualized by ECL
(PerkinElmer) according to the manufacturer’s instruction. β-actin
was used as the loading control.
Quantitative real-time reverse
transcriptase (RT)-PCR
Total RNA was isolated from cells using the TRIzol
reagent (Invitrogen, Carlsbad, CA, USA) and methods described
previously (22). Briefly, total
RNA from treated cells were isolated with TRIzol reagent according
to the manufacturer’s protocol. Total RNA was used to synthesize
cDNA using PrimerScript RT Reagent kit (Takara). The cDNA reaction
product was amplified with following primers: ERα forward,
5′-CGGCATTCTACAGGCCAAATT-3′ and ERα reverse,
5′-AGCGAGTCTCCTTGGCAGATT-3′; PR forward, 5′-AGCC
CACAATACAGCTTCGAG-3′ and PR reverse, 5′-TTTCGACCTCCAAGGACCAT-3′;
pS2 forward, 5′-TGACTCGGGGTCGCCTTTGGAG-3′ and pS2 reverse,
5′-GTGAGCCGAGGCACAGCTGCAG-3′; cathepsin D forward,
5′-TGAGGCCATTGTGGACAAGGCAC-3′ and cathepsin D reverse,
5′-GTCACGGTCAAACACAGTGTAGTAG-3′; GAPDH forward,
5′-AGAAGGCTGGGGCTCATTTG-3′ and GAPDH reverse,
5′-AGGGGCCATCCACAGTCTTC-3′. GAPDH was used for equal RNA loading.
The experiments were repeated for at least three times.
Statistical analysis
Data were expressed as means ± standard deviations
(SD) for three replicate experiments and considered significant at
P<0.05. The statistical analysis was carried out by using SSPS
edition 19.0.
Results
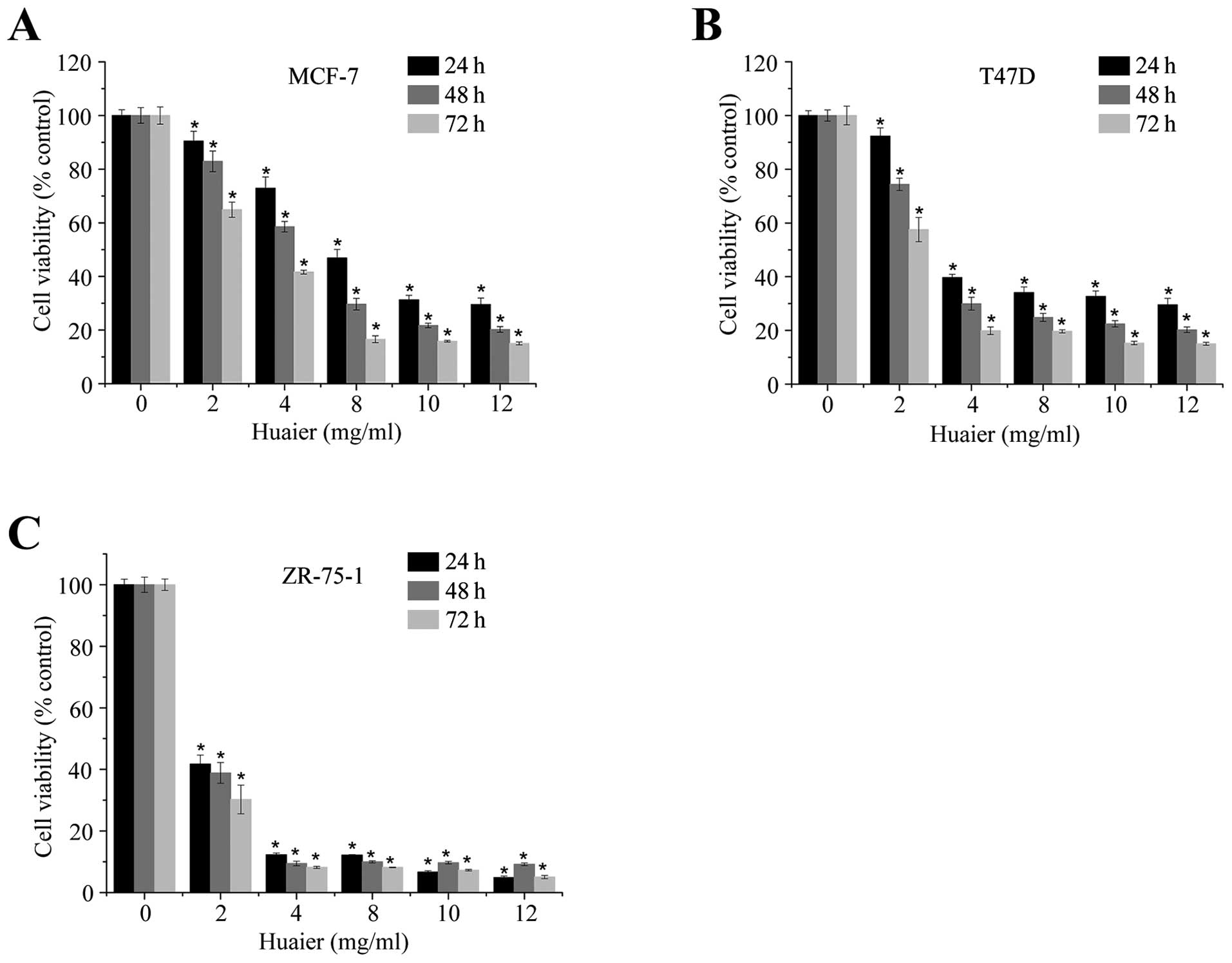
Huaier extract effectively inhibited the
proliferation of ERα-positive breast cancer cell lines
We first examined the effect of Huaier extract on
the growth of three ERα-positive human breast cancer cell lines,
MCF-7, T47D and ZR-75-1. As shown in Fig. 1, Huaier extract significantly
inhibited the proliferation of these three cell lines in a dose-
and time-dependent manner (P<0.05). The results from MCF-7 were
similar to our previous reports (15). In addition, at 24 h, the respective
IC50 values for MCF-7 (Fig.
1A), T47D (Fig. 1B) and
ZR-75-1 (Fig. 1C) were 7.27±0.86,
5.12±0.71 and 1.30±0.11 mg/ml. Thus the ZR-75-1 cell line was more
sensitive to the growth inhibition caused by Huaier extract than
the other two.
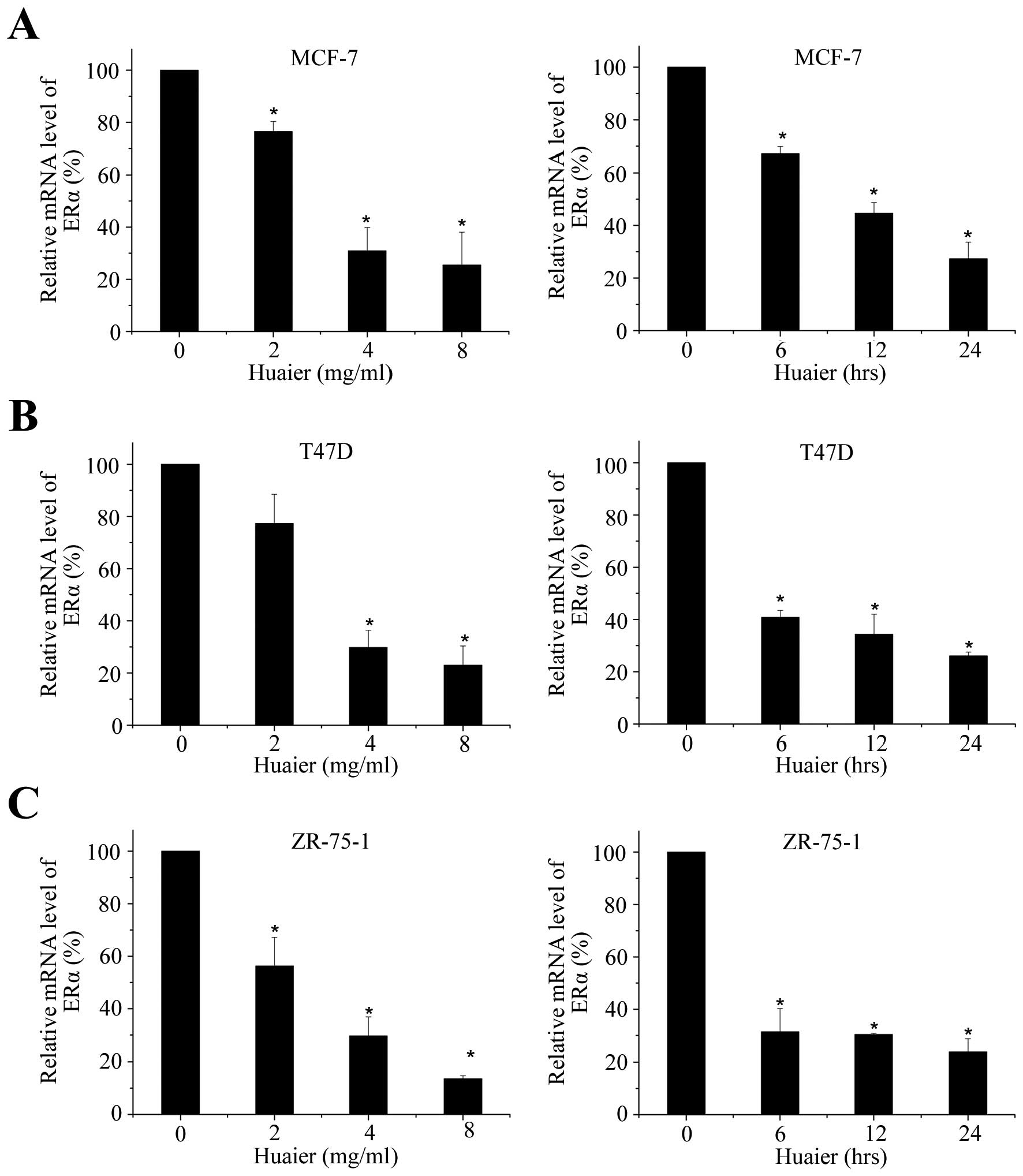
Huaier extract effectively decreased the
mRNA and protein levels of ERα in three ERα-positive human breast
cancer cell lines
ERα is generally associated with cell cycle
progression and has been reported to promote both
anchorage-dependent and anchorage-independent growth of breast
cancer cells (23–25). To determine the effect of Huaier
extract on the level of ERα, cell lines were exposed to various
concentrations of Huaier extract for 24, 48 or 72 h. We then
examined the effect of Huaier extract on ERα mRNA levels. As shown
in Fig. 2A, Huaier extract could
decreased the mRNA level of ERα dose- and time-dependently in MCF-7
cell line. After various times of 4 mg/ml Huaier treatment, the
transcriptional levels were significantly inhibited by 32.9±2.7,
55.4±4.1 and 72.7±6.3% (P<0.05), respectively. Reduction of ERα
mRNA levels was also observed in T47D and ZR-75-1 cells (Fig. 2B and C).
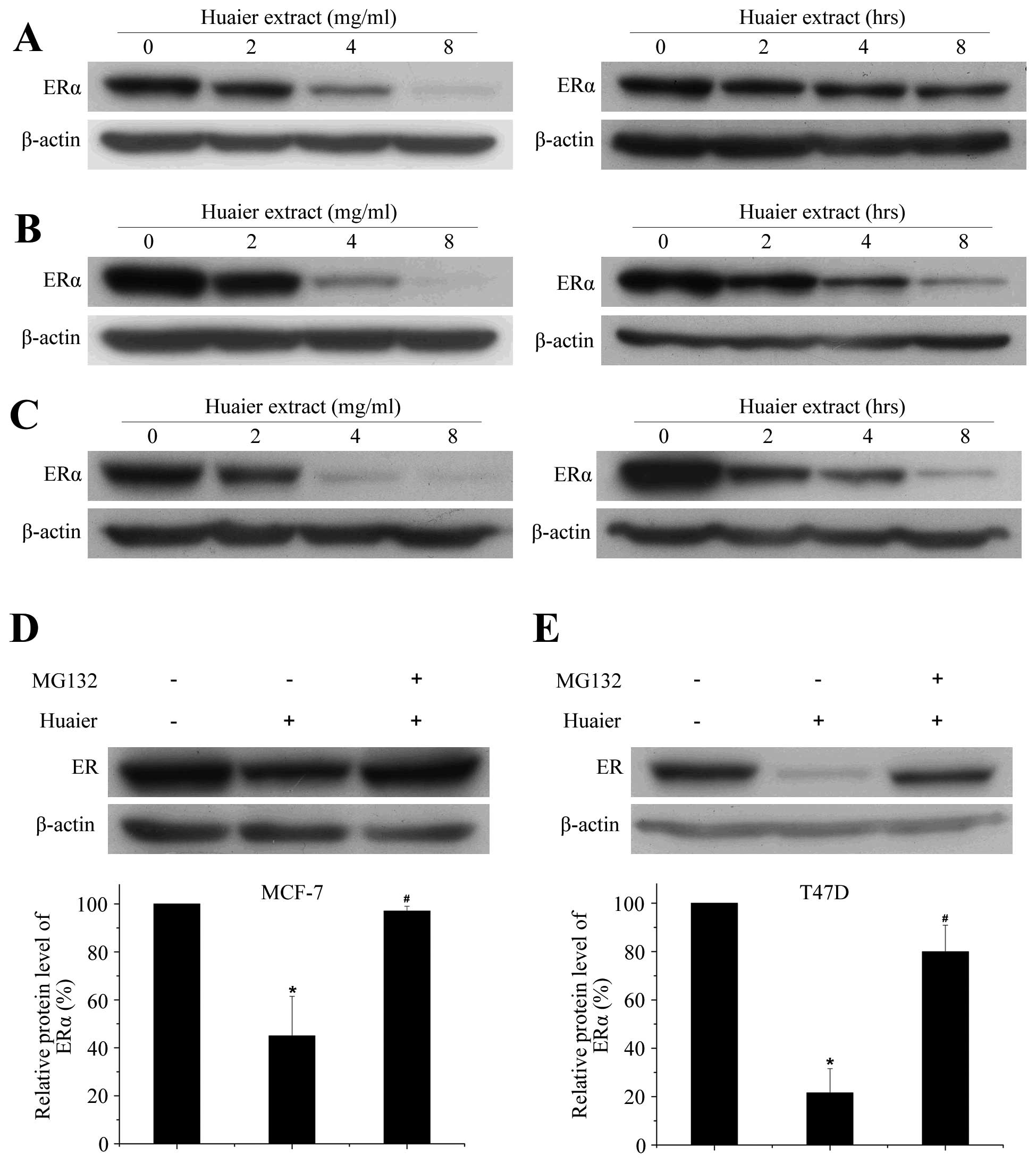
Data from Fig. 3
demonstrated that Huaier extract markedly downregulated the ERα
protein levels in a time- and dose-dependent manner (P<0.05).
After 24 h incubation of 4 mg/ml Huaier extract, the levels of ERα
were reduced by 67.2±9.8, 86.1±7.0 and 85.3±4.0% in MCF-7 (Fig. 3A), T47D (Fig. 3B) and ZR-75-1 (Fig. 3C) cell lines, respectively.
Reduction of ERα protein levels was observed in all three
ERα-positive breast cancer cell lines, suggesting that the impact
of Huaier extract on this protein is common to ERα-positive breast
cancer cell lines.
Huaier-induced downregulation of ERα
protein was associated with activated proteasome
Previous studies have revealed that regulation of
ERα protein is associated with protein degradation via proteasomes
(26). To explore the involvement
of proteasome in the Huaier-induced ERα degradation, the proteasome
inhibitor, MG132, was used. Treatment of MCF-7 cells with Huaier
extract led to a potent and persistent decrease in ERα protein
levels. Addition of MG132 before Huaier treatment markedly rescued
the downregulation of ERα (P<0.05, Fig. 3D). In addition, MG132 was also able
to abolish the inhibitory effect of Huaier on ERα protein in T47D
cell line (Fig. 3E). These
findings demonstrated that inhibition of both gene transcription
and proteasome-mediated degradation was required to reduce
endogenous levels of ERα in response to Huaier extract
treatment.
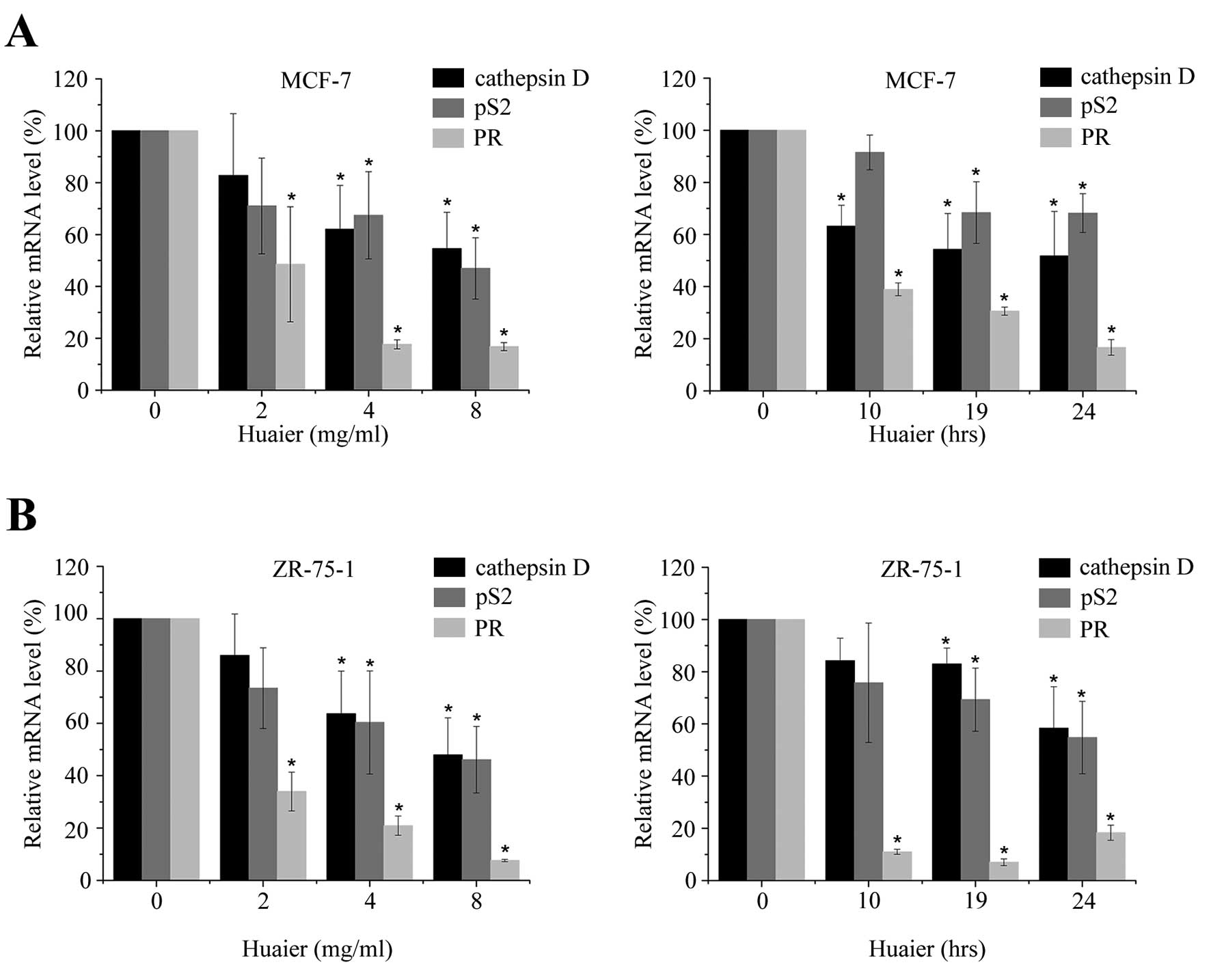
Huaier extract significantly
downregulates the transcription of estrogen responsive genes
It has been reported that some activators of ERα,
such as 17-β-estradiol, could also decrease ERα level (27), suggesting that a reduced level of
ERα was not closely associated with a reduction of ERα activity.
Therefore, we next determined the transcriptional activity of ERα
by detecting its downstream target genes after Huaier extract
treatment (28,29).
The expressions of cathepsin D, pS2 and progestogen
receptor (PR) were reported to be regulated by estrogen (30). To further determine the
transcriptional activity of ERα, we examined their mRNA levels
after Huaier extract treatment within 24 h. The levels of
transcription of each gene were normalized to the level of GAPDH.
As shown in Fig. 4A, MCF-7 cells
were cultured in the absence or presence of Huaier extract (2–8
mg/ml) for 10, 19 or 24 h. Significant reduction of mRNA could be
observed dose- and time-dependently (P<0.05). Similar results
were obtained in ZR-75-1 cells (Fig.
4B). However, as PR expression in T47D cells was demonstrated
to be independent of estrogen (31), we did not detect the mRNA level of
PR in T47D cells. In addition, Huaier extract failed to cause
significant reduction on gene expression of pS2 and cathepsin D in
T47D cells (data not shown).
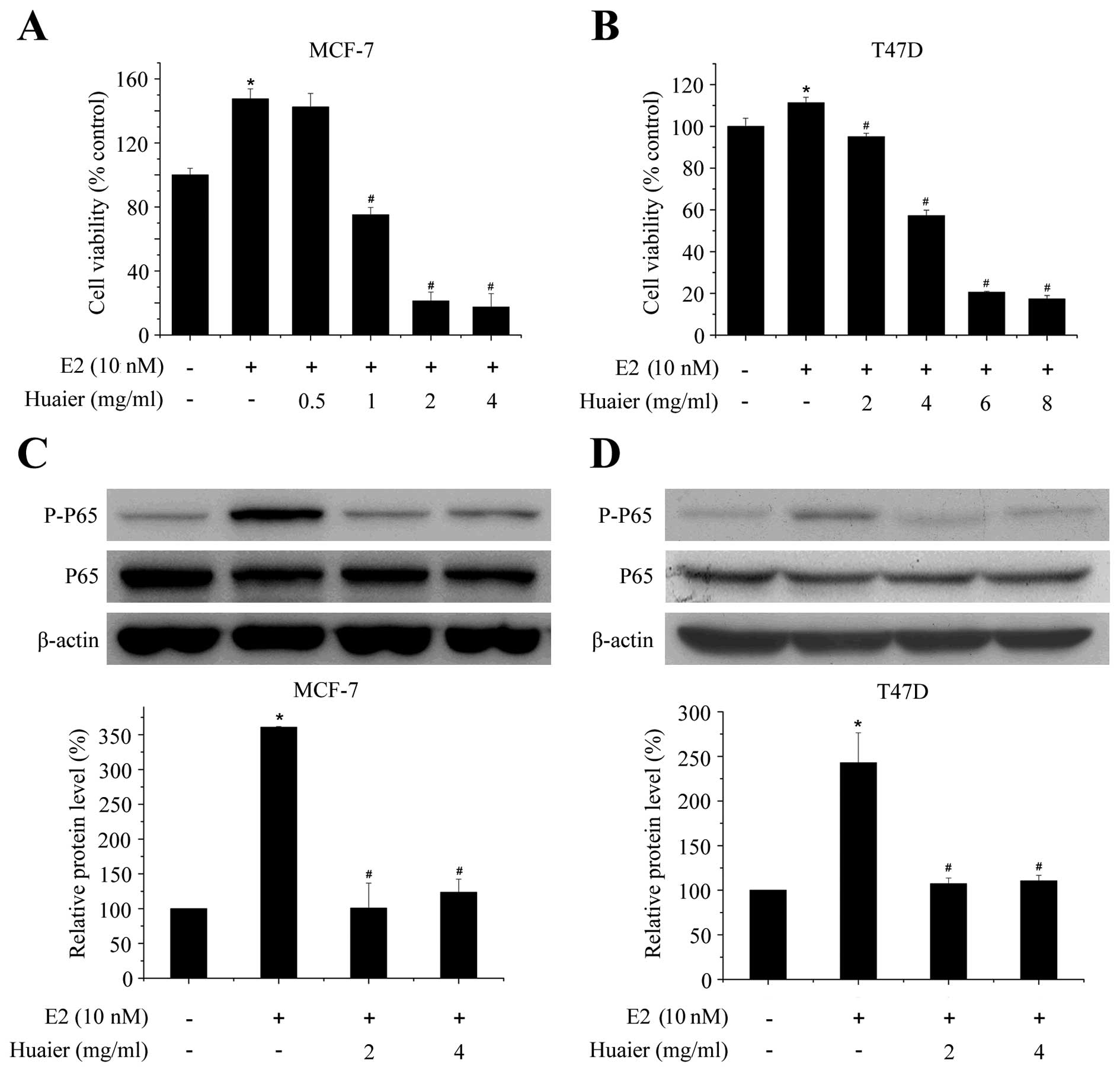
Huaier extract inhibits E2-induced
proliferation and NF-κB activation
The proliferation of ERα-positive human breast
cancer cell lines is strongly stimulated by estrogens (25). To specifically determine the
inhibitory effect of Huaier extract on E2-induced effect, MTT and
western blot assays were used. As shown in Fig. 5A, Huaier extract significantly
inhibited E2-induced cell proliferation in a dose-dependent manner.
The survival of MCF-7 cells was significantly reduced by 49.1±4.6%
after 48-h treatment with 1 mg/ml Huaier extract as compared with
E2 treatment alone. The inhibition was observed in both MCF-7 and
T47D cell lines (Fig. 5A and
B).
It has been reported that activation of NF-κB
contributed to the estrogen-induced proliferation in breast cancer
cells (32). Thus, we detected the
effect of Huaier extract on NF-κB pathway. The activation of NF-κB
was assessed at 1 h in MCF-7 and T47D cells treated with control,
estrogen alone or in combination with Huaier extract (2–4 mg/ml).
As shown in Fig. 5C and D, 10 nM
estrogen significantly phosphorylated P65 without influencing the
total protein level of P65. After addition of Huaier extract, the
phosphorylated levels of P65 were reduced almost to the basal level
without estrogen. These results demonstrated that Huaier extract
completely abolished the effect of estrogen on the activation of
NF-κB and thus suppressed the proliferation of breast cancer cells
induced by estrogen.
Discussion
ERα has become an important target in the treatment
of hormone-responsive breast cancers. It has been reported that
treatment with tamoxifen has enhanced patient survival (33). Unfortunately, most patients
initially responding to anti-estrogen therapies, such as tamoxifen,
will eventually become resistant (34), along with a possible association
with endometrial carcinomas (35,36).
Although the mechanisms of endocrine resistance are not fully
identified, cross-talk between ERα and growth factor signaling
pathways may be involved (37,38).
Therefore, downregulating the levels of ERα may be a potent
therapeutic therapy for both primary estrogen-dependent breast
cancer and hormone-refractory breast cancer.
Fulvestrant (ICI 182780) is a selective estrogen
receptor downregulator (SERD) with no agonist effects and has been
demonstrated to decrease the level of ERα protein to block both
ligand-dependent and -independent receptor activation (39). However, recent studies have
reported that fulvestrant could cause gastro-intestinal disturbance
and hot flashes (40). Thus,
alternative interventions such as compounds from natural products
are needed to replace or to supplement current therapies.
Recently, Huaier extract was shown to exhibit
antitumor and anti-angiogenesis activities both in vitro and
in vivo (15,16). Studies in our laboratory have
demonstrated that Huaier extract inhibited breast cancer growth via
a direct pro-apoptotic effect on tumor cells, as well as through an
indirect effect on endothelial cells (15,16).
However, little is known about the effect of Huaier extract on the
estrogen receptor signaling pathway in breast cancers.
In the present study, we explored the mechanisms of
action of Huaier extract on breast cancer cell growth by focusing
on ERα. Following the treatment with various concentrations of
Huaier extract, the mRNA and protein levels of ERα were
significantly decreased in MCF-7, ZR-75-1 and T47D cell lines in a
time- and dose-dependent manner (Figs.
2 and 3), suggesting that this
effect was general in ERα-positive breast cancer cells. In search
for detail mechanisms causing reduced ERα level, we observed that
MG 132, a proteasome inhibitor, could effectively suppress
Huaier-induced ERα degradation (Fig.
3D and E). Thus, our data demonstrated that Huaier extract
reduced the mRNA levels of ERα and decreased its protein through
promotion of the proteasome pathway. However, fulvestrant reduce
ERα levels only through increasing protein turnover without
affecting its mRNA levels (39).
In this view, Huaier extract may be useful in the treatment of
breast cancer that are resistant to the pure antiestrogen.
After binding to ERα, the estrogen-ERα complex will
trans-locate to the target DNA binding site, the estrogen
responsive element, in the promoter region of the target gene for
gene transcription activation (41). Therefore, we next examined the
expression level of downstream genes regulated by estrogen, such as
cathepsin D, pS2 and PR (30).
After treatment by Huaier extract, the mRNA level of these genes
were significantly reduced in a time- and dose-dependent manner
(Fig. 4), suggesting that the
Huaier extract suppressed the transcription activity of ERα along
with its reduced levels.
To further examine the effect of Huaier extract on
the 17β-estradiol-stimulated cell growth and its potential
mechanisms, we analyzed several pathways regulated by estrogen and
discovered NF-κB pathway. Although some studies showed that
estrogen inhibited the tumor necrosis factor-α-induced activation
of NF-κB (42), Rubio et al
demonstrated no antagonism between ER and NF-κB in T47D and HC11
cells (32). In addition, in their
study, activation of NF-κB was needed for estrogen-induced
proliferation and expression of cyclin D1. In our study, data from
Fig. 5 shows that 17β-estradiol
significantly enhanced the phosphorylation of P65, the NF-κB
component, in a phenol red and serum-free condition. After addition
of Huaier extract, the level of P-P65 was reduced to the basal
level without estrogen. Therefore, we suggest that Huaier extract
inhibited the E2-induced proliferation of breast cancer through
activation of the NF-κB pathway.
In summary, we found that Huaier extract potently
inhibited the proliferation of ERα-positive breast cancer cells,
and identified ERα as a possible target for Huaier extract
treatment. Therefore, it is of great importance to develop this
natural product to treat or prevent ERα-positive breast
cancers.
Acknowledgements
This study was supported by National
Natural Science Foundation of China (nos. 81072150, 81172529 and
81272903) and Shandong Science and Technology Development Plan (no.
2012GZC22115). We thank Shi Yan and Cunzhong Yuan for technical
support with experiments. We also thank Weiting Gu and Min Gao for
critical discussions and Dong Lun for substantial help.
References
|
1.
|
Hedden A, Müller V and Jensen EV: A new
interpretation of antiestrogen action. Ann NY Acad Sci.
761:109–120. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Klinge CM: Estrogen receptor interaction
with estrogen response elements. Nucleic Acids Res. 29:2905–2919.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Kushner PJ, Agard DA, Greene GL, et al:
Estrogen receptor pathways to AP-1. J Steroid Biochem Mol Biol.
74:311–317. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Safe S: Transcriptional activation of
genes by 17β-estradiol through estrogen receptor-Sp1 interactions.
Vitam Horm. 62:231–252. 2001.
|
|
5.
|
Dotzlaq H, Leygue E, Watson PH and Murphy
LC: Expression of estrogen receptor-β in human breast tumors. J
Clin Endocrinol Metab. 82:2371–2374. 1997.
|
|
6.
|
Ali S and Coombes RC: Estrogen receptor
alpha in human breast cancer: occurrence and significance. J
Mammary Gland Biol Neoplasia. 5:271–281. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Korach KS, Couse JF, Curtis SW, et al:
Estrogen receptor gene disruption: molecular characterization and
experimental and clinical phenotypes. Recent Progr Horm Res.
51:1591996.PubMed/NCBI
|
|
8.
|
Bocchinfuso WP and Korach KS: Mammary
gland development and tumorigenesis in estrogen receptor knockout
mice. J Mammary Gland Biol Neoplasia. 2:323–334. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Shaaban AM, O’Neill PA, Davies MPA, et al:
Declining estrogen receptor-[beta] expression defines malignant
progression of human breast neoplasia. American J Surg Pathol.
27:1502–1512. 2003.
|
|
10.
|
Gralow JR: Optimizing the treatment of
metastatic breast cancer. Breast Cancer Res Treat. 89:9–15. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Shang Y and Brown M: Molecular
determinants for the tissue specificity of SERMs. Science.
295:2465–2468. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Osborne CK, Zhao HH and Fuqua SAW:
Selective estrogen receptor modulators: structure, function, and
clinical use. J Clin Oncol. 18:3172–3186. 2000.PubMed/NCBI
|
|
13.
|
McCarthy TL, Clough ME, Gundberg CM and
Centrella M: Expression of an estrogen receptor agonist in
differentiating osteoblast cultures. Proc Natl Acad Sci USA.
105:7022–7027. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Cassady JM, Baird WM and Chang CJ: Natural
products as a source of potential cancer chemotherapeutic and
chemopreventive agents. J Nat Prod. 53:23–41. 1990.PubMed/NCBI
|
|
15.
|
Zhang N, Kong X, Yan S, Yuan C and Yang Q:
Huaier aqueous extract inhibits proliferation of breast cancer
cells by inducing apoptosis. Cancer Sci. 101:2375–2383. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Wang X, Zhang N, Huo Q and Yang Q:
Anti-angiogenic and antitumor activities of Huaier aqueous extract.
Oncol Rep. 28:1167–1175. 2012.PubMed/NCBI
|
|
17.
|
Ren J, Zheng C, Feng G, et al: Inhibitory
effect of extract of fungi of Huaier on hepatocellular carcinoma
cells. J Huazhong Univ Sci Technolog Med Sci. 29:198–201. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Xu X, Wei Q, Wang K, et al: Anticancer
effects of Huaier are associated with down-regulation of P53. Asian
Pac J Cancer Prev. 12:2251–2254. 2011.PubMed/NCBI
|
|
19.
|
Sun Y, Sun T, Wang F, et al: A
polysaccharide from the fungi of Huaier exhibits anti-tumor
potential and immunomodulatory effects. Carbohydr Polym.
92:577–582. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Lippman M, Bolan G and Huff K: The effects
of estrogens and antiestrogens on hormone-responsive human breast
cancer in long-term tissue culture. Cancer Res. 36:4595–4601.
1976.PubMed/NCBI
|
|
21.
|
Dupont J, Karas M and LeRoith D: The
potentiation of estrogen on insulin-like growth factor I action in
MCF-7 human breast cancer cells includes cell cycle components. J
Biol Chem. 275:35893–35901. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
22.
|
Zhu J, Li X, Kong X, et al: Testin is a
tumor suppressor and prognostic marker in breast cancer. Cancer
Sci. 103:2092–2101. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Altucci L, Addeo R, Cicatiello L, et al:
17beta-estradiol induces cyclin D1 gene transcription,
p36D1-p34cdk4 complex activation and p105Rb phosphorylation during
mitogenic stimulation of G (1)-arrested human breast cancer cells.
Oncogene. 12:23151996.
|
|
24.
|
Kodama F, Greene GL and Salmon SE:
Relation of estrogen receptor expression to clonal growth and
antiestrogen effects on human breast cancer cells. Cancer Res.
45:2720–2724. 1985.PubMed/NCBI
|
|
25.
|
Lu J, Pierron A and Ravid K: An adenosine
analogue, IB-MECA, down-regulates estrogen receptor α and
suppresses human breast cancer cell proliferation. Cancer Res.
63:6413–6423. 2003.PubMed/NCBI
|
|
26.
|
Wormke M, Stoner M, Saville B, et al: The
aryl hydrocarbon receptor mediates degradation of estrogen receptor
α through activation of proteasomes. Mol Cell Biol. 23:1843–1855.
2003.
|
|
27.
|
Wijayaratne AL and McDonnell DP: The human
estrogen receptor-α is a ubiquitinated protein whose stability is
affected differentially by agonists, antagonists, and selective
estrogen receptor modulators. J Biol Chem. 276:35684–35692.
2001.
|
|
28.
|
Horwitz KB, Koseki Y and McGuire WL:
Estrogen control of progesterone receptor in human breast cancer:
role of estradiol and antiestrogen. Endocrinology. 103:1742–1751.
1978. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Dauvois S, White R and Parker MG: The
antiestrogen ICI 182780 disrupts estrogen receptor
nucleocytoplasmic shuttling. J Cell Sci. 106:1377–1388.
1993.PubMed/NCBI
|
|
30.
|
Baker P, Wilton J, Jones C, Stenzel D,
Watson N and Smith G: Bile acids influence the growth, oestrogen
receptor and oestrogen-regulated proteins of MCF-7 human breast
cancer cells. Br J Cancer. 65:5661992. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Horwitz KB, Mockus MB and Lessey BA:
Variant T47D human breast cancer cells with high
progesterone-receptor levels despite estrogen and antiestrogen
resistance. Cell. 28:633–642. 1982. View Article : Google Scholar
|
|
32.
|
Rubio M, Werbajh S, Cafferata E, et al:
TNF-alpha enhances estrogen-induced cell proliferation of
estrogen-dependent breast tumor cells through a complex containing
nuclear factor-kappa B. Oncogene. 25:1367–1377. 2005. View Article : Google Scholar
|
|
33.
|
Fisher B, Costantino JP, Wickerham DL, et
al: Tamoxifen for prevention of breast cancer: report of the
National Surgical Adjuvant Breast and Bowel Project P-1 Study. J
Natl Cancer Inst. 90:1371–1388. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
Ali S and Coombes RC: Endocrine-responsive
breast cancer and strategies for combating resistance. Nat Rev
Cancer. 2:101–112. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Yao K and Jordan VC: Questions about
tamoxifen and the future use of antiestrogens. Oncologist.
3:104–110. 1998.PubMed/NCBI
|
|
36.
|
Cortesi L, De Matteis E, Rashid I, et al:
Distribution of second primary malignancies suggests a
bidirectional effect between breast and endometrial cancer: a
population-based study. Int J Gynecol Cancer. 19:13582009.
View Article : Google Scholar
|
|
37.
|
Gutierrez MC, Detre S, Johnston S, et al:
Molecular changes in tamoxifen-resistant breast cancer:
relationship between estrogen receptor, HER-2, and p38
mitogen-activated protein kinase. J Clin Oncol. 23:2469–2476. 2005.
View Article : Google Scholar
|
|
38.
|
Levin ER: Bidirectional signaling between
the estrogen receptor and the epidermal growth factor receptor. Mol
Endocrinol. 17:309–317. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
39.
|
Osborne C, Wakeling A and Nicholson R:
Fulvestrant: an oestrogen receptor antagonist with a novel
mechanism of action. Br J Cancer. 90:S2–S6. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40.
|
Buijs C, de Vries EG, Mourits MJ and
Willemse PH: The influence of endocrine treatments for breast
cancer on health-related quality of life. Cancer Treat Rev.
34:640–655. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41.
|
Klinge CM: Estrogen receptor interaction
with co-activators and co-repressors. Steroids. 65:227–251. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
42.
|
Hsu SM, Chen YC and Jiang MC:
17β-estradiol inhibits tumor necrosis factor-α-induced nuclear
factor-κB activation by increasing nuclear factor-κB p105 level in
MCF-7 breast cancer cells. Biochem Biophys Res Commun. 279:47–52.
2000.
|