Introduction
Oral cancer is a major public health problem, with
>500,000 new cases per year worldwide (1). Squamous cell carcinoma from the oral
cavity and surrounding anatomical areas is the most common
malignancy from the head and neck area, accounting for 90% of all
tumors (2). Overall survival
rates remain low at 50% after 5 years from the initial diagnosis
and have not improved significantly in the last three decades
(3,4). Oral squamous cell carcinoma (OSCC)
has a multifactorial origin with several risk factors that act
individually or in combination (5). The predominant etiological factors
for OSCC involve tobacco and alcohol consumption, with an estimated
5-fold increase risk for cancer development in heavy smokers
(2,6,7).
Emerging evidence indicates that periodontitis may
be an associated risk factor for OSCC (2,5,8,9).
In fact, the presence of periodontal disease has been associated
with an elevated risk of lung and colorectal cancer in a population
study of >7,000 participants (10). Periodontitis represents an
infection-induced chronic inflammatory disease that leads to
irreversible loss of the supporting structures of the teeth. The
heavy burden of periodontitis is directly associated with its high
prevalence. Severe periodontitis is estimated to represent the 6th
most prevalent disease, affecting around 743 million people
worldwide (11). The biological
mechanism by which periodontitis could act as a predisposing factor
to oral cancer initiation and development is hypothesized in two
theories: i) The direct involvement of the periodontopathogens; and
ii) indirectly by inducing a persistent chronic inflammatory state
(8,9). The constant polymicrobial infection
present in periodontitis and the associated release of
pro-inflammatory mediators are likely to play an important role in
the initiation and/or progression of oral cancer, or at least
increase its susceptibility (2).
Epidemiological studies present several confounding
factors that can affect the establishment of an accurate
correlation between periodontitis and oral cancer, such as smoking
and alcohol consumption. The majority of experimental studies
trying to establish a direct link between periodontal disease and
OSCC were performed with cell lines and focused on the carcinogenic
potential of Porphyromonas gingivalis (P.
gingivalis), a major pathogen of periodontal disease (12). in vivo, experimental
studies are still scarce and focus on P. gingivalis
(13). The results indicate an
association between P. gingivalis infection and OSCC
development (13,14). Despite emerging evidence, to the
best of our knowledge, the in vivo evaluation of
biofilm-induced periodontitis in oral carcinogenesis and tumor
progression has not been explored. Therefore, the present study
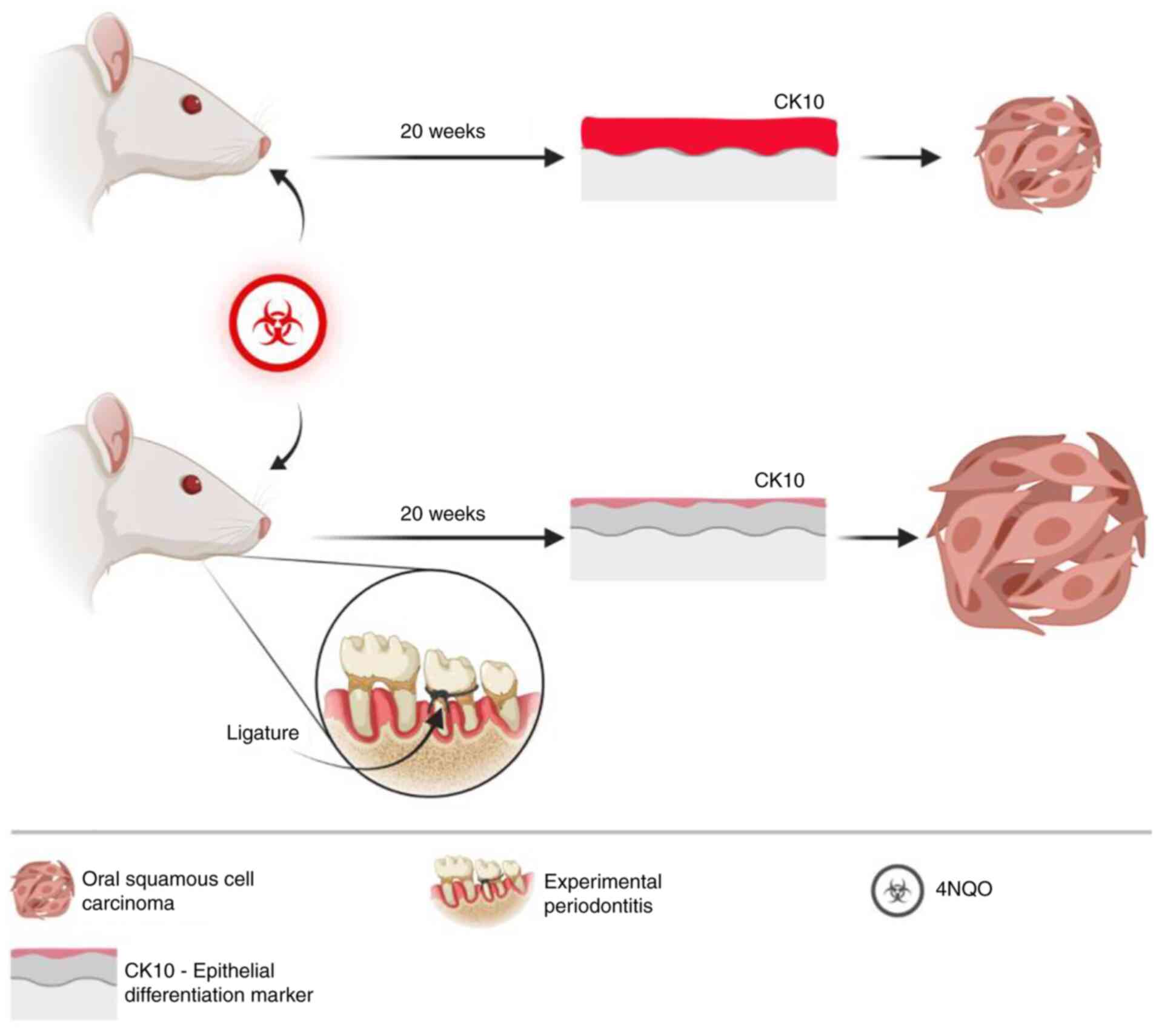
aimed to analyze the effect of ligature-induced periodontitis on
the development of 4-nitroquinoline 1-oxide (4NQO)-induced OSCC in
Wistar rats.
Materials and methods
Animals and experimental procedures
The present study was performed in accordance with
Brazilian Federal Law No. 11.794/2008 for the scientific use of
animals. The study protocol was submitted to and approved by the
Animal Use Ethics Committee, Porto Alegre General Hospital (Port
Alegre, Brazil; approval no. 150475). Each sample size calculation
for the experimental groups was performed based on previous studies
with a similar methodology. The control group and the group with
induced periodontitis (Perio) were comprised of 8 animals each,
while the groups receiving 4-nitroquinoline 1-oxide (4NQO) were
composed of 13 animals to account for the expected mortality rate
of ~30% due to 4NQO-related side effects during the 20 weeks of
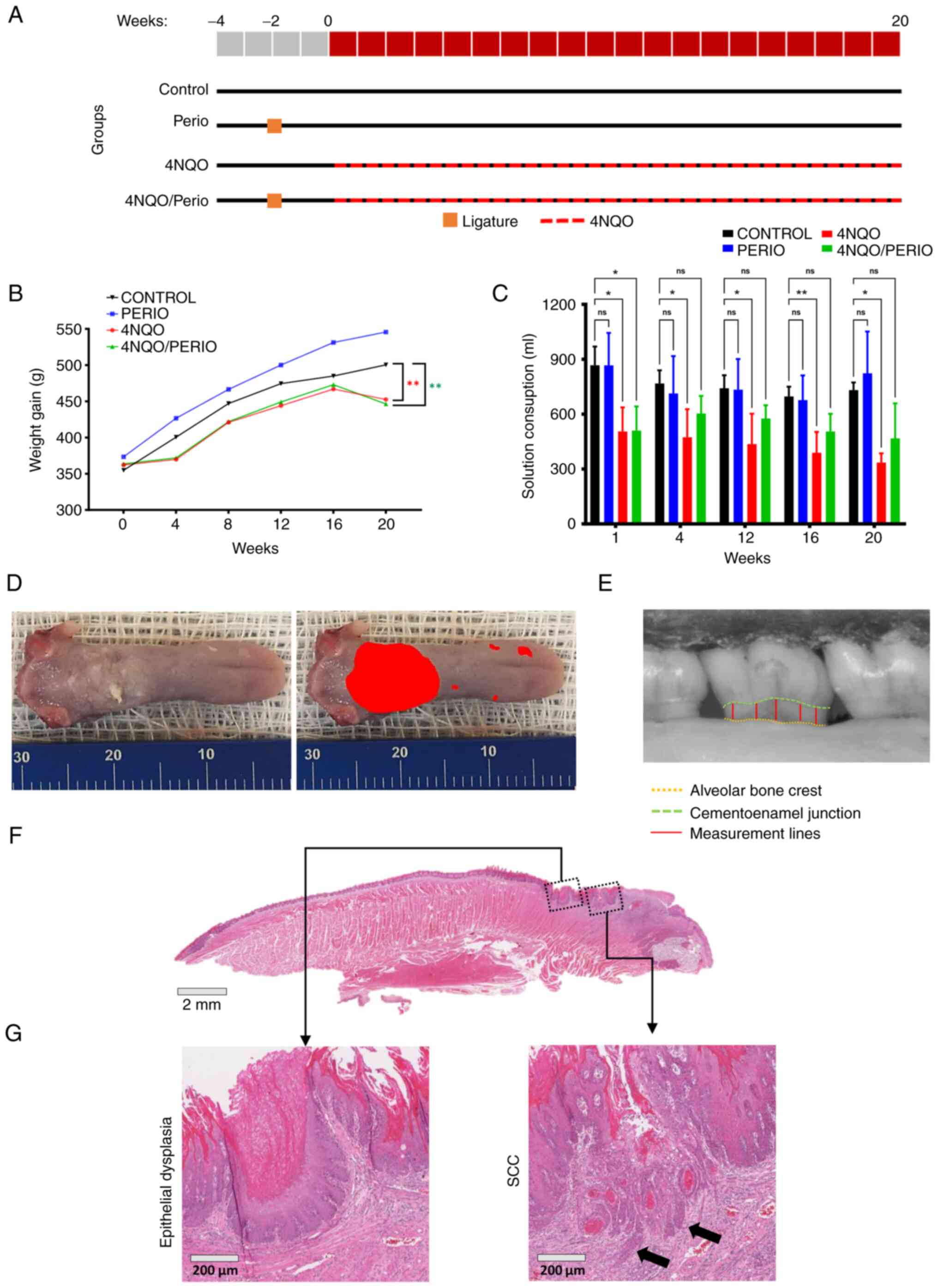
carcinogen administration (Fig.
1A) (15-17).
A total of 42 60-day-old male Wistar rats were used
in the study. Rats were shipped weighing ~250 g, and after the
acclimation period, rats gained an additional 100 g, therefore
weighing 350 g at the start date of the experiments (Fig. 1B). The rats were provided by the
Center for Reproduction and Experimentation in Laboratory Animals
(Federal University of Rio Grande do Sul, Porto Alegre, Brazil) and
acclimated for 2 weeks at the Animal Experimentation Unit of the
Porto Alegre General Hospital. Animals were kept under standard
temperature conditions at 22±2°C, using a light/dark cycle of 12 h,
with a relative humidity of 50-70% and free access to sterilized
rodent laboratory chow (Nuvilab CR1; Quimtia S.A.).
Animals were randomly distributed throughout the
four groups according to bodyweight: Control (n=8), Perio (n=8),
4NQO (n=13) and 4NQO/Perio (n=13). Periodontitis was induced by
placing a 4-0 silk ligature subgingivally (Ethicon, Inc.; Johnson
& Johnson). Ligatures were placed around the right maxilla
second molar under isoflurane anesthesia (5% of isoflurane for
induction and 2% for maintenance) 2 weeks before 4NQO
administration. Subgingival ligatures were kept in place during the
experiment (22 weeks) (18,19). 4NQO solution was used to induce
oral carcinogenesis according to a previously published protocol
(20,21). Fresh solutions were prepared every
2 days, and rats were exposed to 25 ppm of the 4NQO carcinogen
solution (Sigma-Aldrich; Merck KGaA; catalog no. N8141) diluted in
drinking water for 20 weeks (15). Cage water consumption was measured
each time the solution was replaced (Fig. 1C). Animal weight and health status
were monitored every other day by the researcher and constantly
monitored by the vivarium staff and veterinarian during the
experimental period. There was no evidence of ligature-induced
disruption of food intake. In total, 2 animals from the 4NQO group
required early euthanasia using 5% isofluorane followed by
exsanguination during the experimental period due to bodyweight
loss of 20% as an adverse effect advent from 4NQO administration.
Death was verified by the lack of vital signs, including the
absence of respiration and heartbeat. Other endpoints considered
during the experiment included oral tumor burden presenting as
disrupted eating and drinking behaviors associated with bodyweight
loss of 20%. Animals from the Control, Perio, 4NQO and 4NQO/Perio
groups were anesthetized by inhalation of isoflurane (5%), followed
by euthanasia by decapitation. After euthanasia, the tongues and
maxillae from the rats were removed and stored in 10% formalin for
24-72 h at room temperature.
Determination of lesion surface area
Tumor development was assessed on the dorsal area of
the tongues. Images were captured of each tongue specimen, and the
number and the size of the lesions were determined (Fig. 1D). Total lesion area was measured
using ImageJ 1.46r software (National Institutes of Health). The
area was calculated in pixels and converted to mm2 using
a scale reference. Briefly, the scale was set using the
straight-line tool from the ImageJ toolbar over a known scale
(i.e., ruler image next to an open image). Under Analyze>Set
Scale, the known scale was added to the 'known distance' field and
'Unit of length' was set in millimeters. Next, all images to be
analyzed were transformed into 8-bit images
(Image>Type>8-bit). Lesions were delineated using the
'Freehand selections' tool from the ImageJ toolbar. Lesion area was
calculated using the analysis tool under the menu bar
Analyze>Measure. Sequential measurements of all lesions were
recorded. Additional information on ImageJ tools can be found at
https://imagej.nih.gov/ij/docs/.
Morphometrical analysis
All maxillae were defleshed using 9% sodium
hypochlorite for 4 h at room temperature, and soft tissue was
removed carefully. Maxillae were stained for 1 min in methylene
blue (1%) to outline the cementoenamel junction (CEJ). Standardized
pictures were captured of the buccal and palatal sides of the
maxillae and measured using ImageJ 1.46r software (National
Institutes of Health) (Fig. 1E).
Five linear points were recorded, and the mean of the values
considering the distance between the CEJ and the alveolar bone
crest was considered for bone loss. To ensure that the examiner was
blinded to the groupings, pictures were randomized by a computer
program, coded and renamed by an external examiner. Approximately
10% of the maxillae were randomly selected and twice measured in
terms of alveolar bone loss, and compared using the intraclass
correlation coefficient, which showed excellent results (0.96).
Histological and immunostaining
analysis
Tongues were embedded in paraffin blocks and
longitudinally thin-sectioned (4 µm). The sections were
stained with hematoxylin for 4 min and eosin for 2 min, at room
temperature (Merck KGaA) and graded as normal, epithelial dysplasia
and carcinoma (Fig. 1F and G) by
two blinded expert oral pathologists (differences in grading were
discussed and agreement was achieved). Hematoxylin and
eosin-stained slides were scanned using an Aperion ScanScope slide
scanner (Leica Biosystems).
Immunofluorescence was performed using double
staining with anti-cytokeratin 10 (CK10) (clone Poly19054; cat. no.
905404; 1:500; BioLegend, Inc.) as the primary antibody for 1 h at
room temperature, followed by use of Alexa Fluor® 568
(cat. no. A11036; 1:200; Invitrogen; Thermo Fisher Scientific,
Inc.) as the secondary antibody for 45 min at room temperature. DNA
was stained using Hoechst 33342 (cat. no. H1399; 1:5,000;
Invitrogen; Thermo Fisher Scientific, Inc.) for 5 min at room
temperature. Bovine serum albumin (cat. no. 9048-46-8;
MilliporeSigma) diluted to 3% in PBS was used as a blocking
solution prior to the incubation of primary antibody for 45 min at
room temperature. Images of the entire tongue of different groups
were captured at ×200 magnification using a QImaging-ExiAqua
monochrome digital camera attached to an epi-fluorescence Nikon
microscope (Nikon Corporation) and visualized with QCapturePro 7
software (QImaging; Teledyne Photometrics). The Control and Perio
groups had 1 sample per group, and the 4NQO and 4NQO/Perio groups
had 6 samples per group (2 samples diagnosed with OSCC and 4
samples diagnosed with dysplasia). The 4NQO and 4NQO/Perio groups
had a total of 398 fields captured each. Images were randomized by
a computer program, and coded and renamed by an external examiner.
A blinded examiner analyzed the images by evaluating the presence
of CK10 positivity in the basal cell layer within each field. The
percentage of positive fields per experimental group was calculated
and used for statistical analysis.
Statistical analysis
The variables of this study were tested for
normality. Data were analyzed using the statistical package
GraphPad Prism 7 (GraphPad Software, Inc.). Findings were reported
as the mean ± SEM. One-way ANOVA followed by Tukey's multiple
comparison test was used on the alveolar bone analysis and weight
gain data. Solution consumption data were analyzed using one-way
ANOVA followed by Dunnett's multiple comparisons test. Unpaired
student's t-test was used for tumor size and CK10+ field
data analysis. The incidence of OSCC was compared using the
χ2 test, and the odds ratio and the 95% confidence
interval were computed using binary logistic regression. P<0.05
was used to indicate a statistically significant difference.
Results
Weight and solution consumption
analysis
Fig. 1A shows a
flowchart of the study design. All animals gained weight during the
study (Fig. 1B). Animals exposed
to 4NQO consistently gained weight during the first 16 weeks of
drug administration. The modest weight gain during the first 4
weeks of 4NQO administration is commonly observed throughout
different animal models as part of the adaptation process to 4NQO
intake. A decline in animal weight after 16 weeks of drug
administration was observed in both groups receiving 4NQO.
During the entire 20-weeks of 4NQO administration
significant weight loss was only observed in the 4NQO and
4NQO/Perio groups compared with the Perio group at week 20 (mean
difference, 93.2 and 99.4; P=0.0042 and P=0.0022, respectively).
When compared with the Control group, the 4NQO and 4NQO/Perio
groups showed no significant difference throughout the experiment.
A parallel may be drawn regarding solution consumption. The 4NQO
group consistently drank less than the control group throughout the
experiment (Fig. 1C).
Significantly lower solution consumption was observed during weeks
1, 4, 12, 16 and 20 of exposure to the carcinogen in the 4NQO group
(mean difference, 362.0, 293.6, 305.4, 307.9, and 396.3; P=0.0156,
P=0.0472, P=0.0272, P=0.00081 and P=0.0154, respectively), and in
the first week of exposure in the 4NQO/Perio group (mean
difference, 357.0; P=0.0168) compared with that in the Control
group.
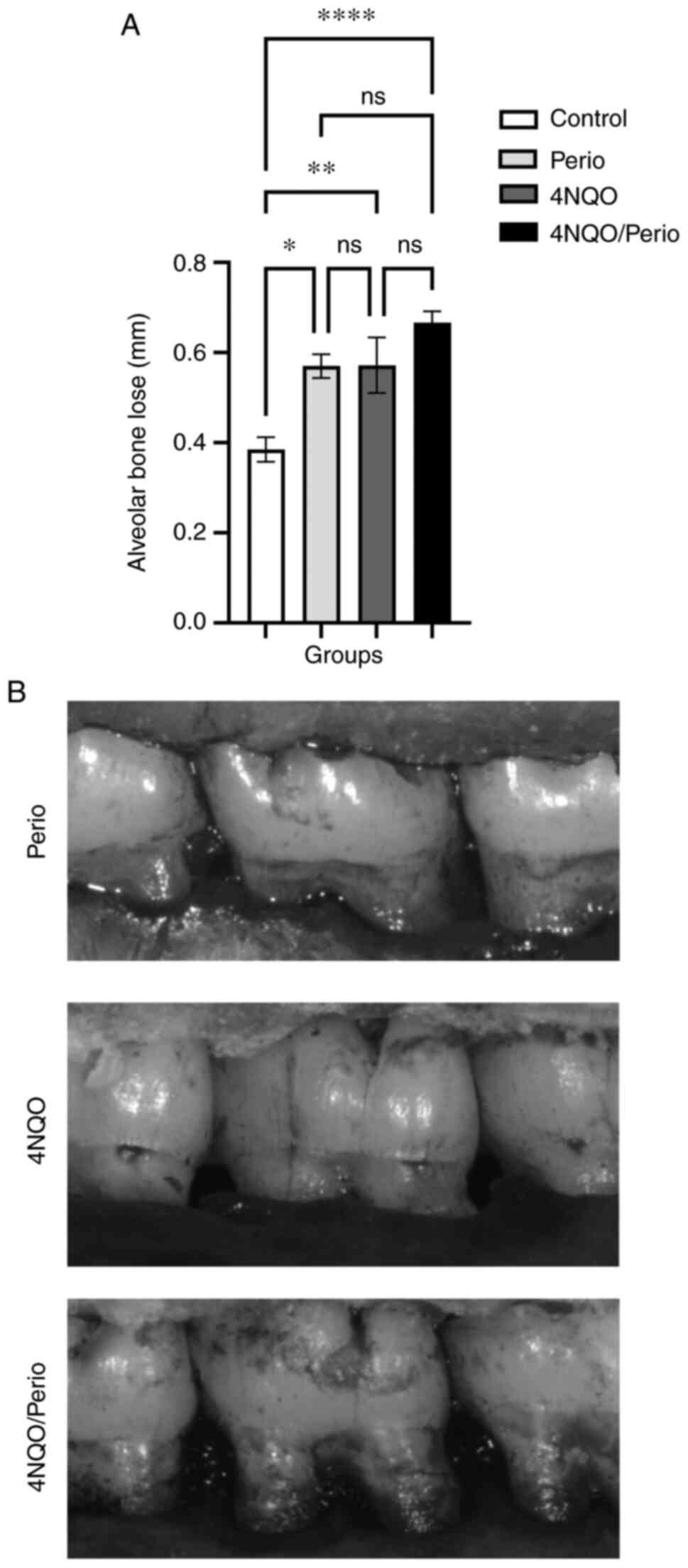
Alveolar bone loss analysis
Alveolar bone loss was measured in the second
maxillary molar on the right side, in which periodontitis was
induced using ligatures. The mean level of bone loss in the Perio,
4NQO and 4NQO/Perio groups (0.57±0.02, 0.57±0.06 and 0.66±0.02 mm,
respectively) was significantly higher compared with that in the
Control group (0.38±0.02 mm) (Fig. 2A
and B). Unexpectedly, it was observed that the administration
of 4NQO alone resulted in alveolar bone loss similar to that in the
Perio group.
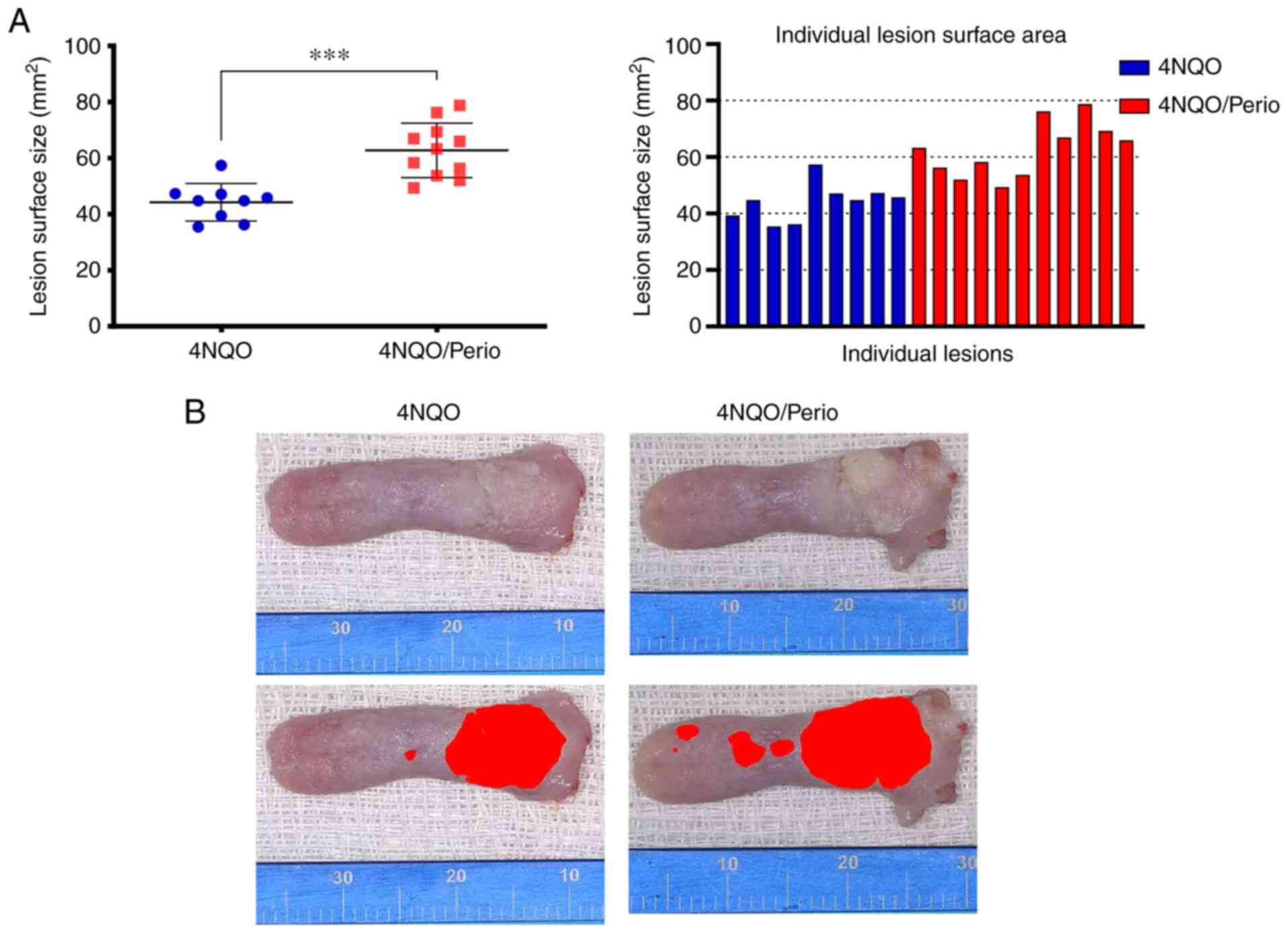
Lesion size and histopathological
analysis
All groups that received 4NQO developed lesions on
the dorsal surface of the tongue. Images of dissected tongues were
captured, and the total area of the lesions was identified by an
expert pathologist and digitally identified and marked with a red
'mask' using ImageJ to determine the lesion surface area. The mean
lesion surface area for the 4NQO/Perio group (62.75±9.72
mm2) was significantly higher than that observed for the
4NQO-treated group (44.25±6.69 mm2) (Fig. 3A and B). Histological analysis
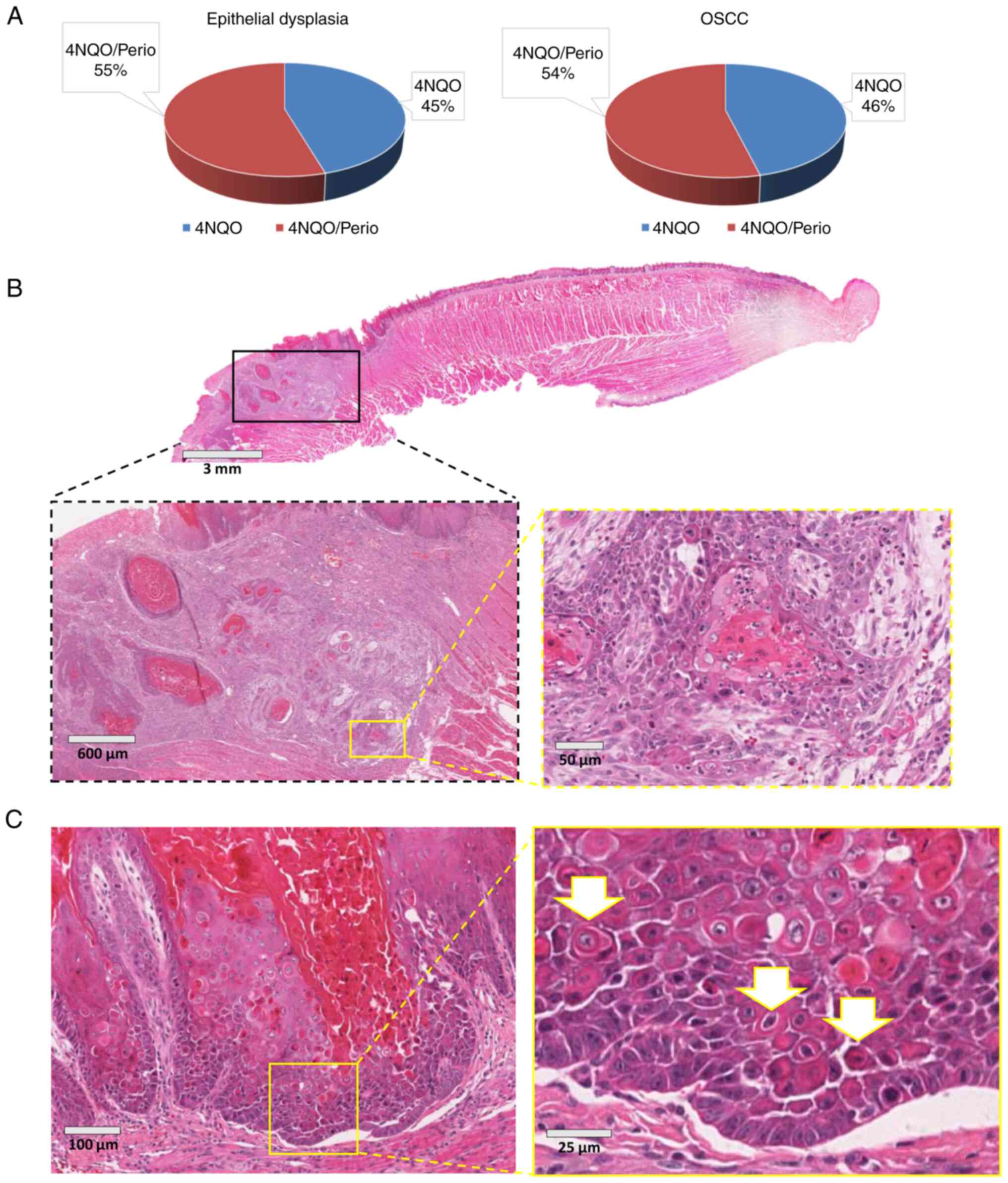
revealed that the incidence of epithelial dysplasia and invasive
squamous cell carcinoma was similar among the 4NQO and
4NQO/Perio-treated animals, with a slight increase in the number of
lesions found in 4NQO/Perio rodents (Fig. 4A). Also, both groups developed
large tumors characterized by the infiltration of the muscle tissue
of the tongue and focal poorly differentiated tumor areas (Fig. 4B).
Cell differentiation analysis
During the process of the histological analysis of
tissues from the 4NQO and 4NQO/Perio groups, abnormal morphological
changes were observed in the epithelial layer of the dorsal tongue
mucosa receiving 4NQO alone. The observed alterations were
suggestive of focal activation of cellular differentiation that
extended near to the basal cell layer of the mucosa (Fig. 4C). In order to further explore
these findings, the expression of CK10, a known marker of
epithelial cell differentiation, was assessed in the rats receiving
4NQO and 4NQO/Perio. CK10 is normally expressed in the
differentiation-committed spinous layer of the oral mucosa, while
basal cells do not express CK10 (22). Immunofluorescence staining for
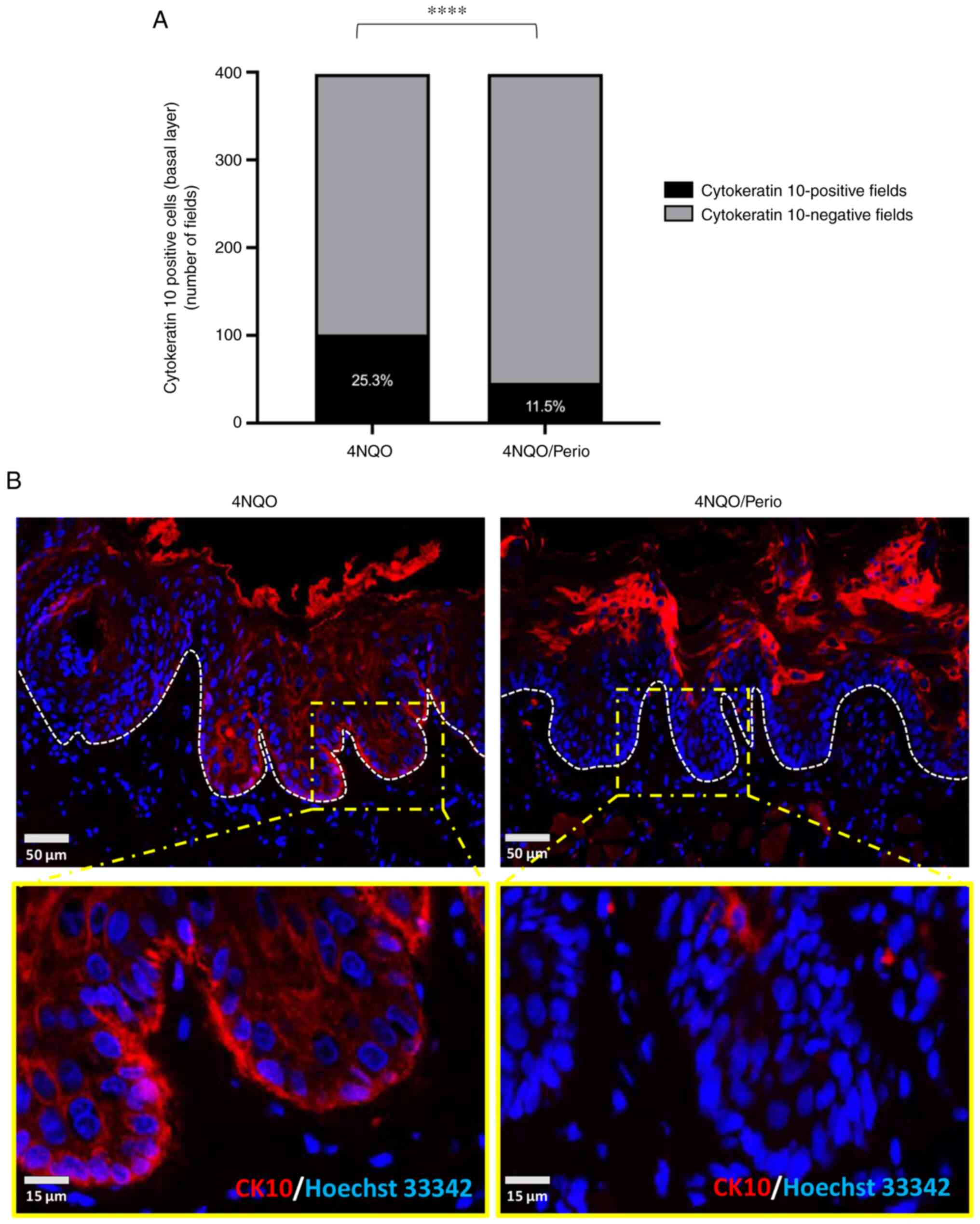
CK10 was performed, and positive basal cells were quantified. All
histological fields presenting with abnormal basal cells expressing
CK10 were computed as positive. A total of 398 fields were assessed
in which 25.3% (4NQO) and 11.5% (4NQO/Perio) of the fields shown
showed CK10 expression at the basal cells (Fig. 5A and B). These findings
demonstrated increased abnormal activation of cellular
differentiation of basal cells in 4NQO-treated animals compared
with that in the 4NQO/Perio group.
Summary
Taken together, the present results suggest that
periodontitis can influence the course of tumor development by
reducing the differentiation of epithelial cells upon exposure to
carcinogens. Differences in the rate of tumor transformation upon
the presence of periodontitis were not conclusive in the present
cohort of animals. Nonetheless, the development of periodontitis
and exposure to 4NQO resulted in the increased size of oral lesions
(Fig. 6).
Discussion
The role of periodontitis and its potential
contributions as a risk factor/indicator in the development and/or
progression of oral cancer by direct and indirect mechanisms have
been the subject of interest and investigation in recent years
(5,9). The majority of studies investigating
the biological mechanisms of the association between periodontal
disease and oral cancer mainly focus on P. gingivalis, a
major etiological agent in the development of chronic
periodontitis, yet not the only one (5,12,23-27). To the best of our knowledge, no
previous study has evaluated the impact of the dental biofilm on
oral carcinogenesis and tumor progression. The present study used a
ligature-induced periodontitis animal model, as it shares
similarities with the etiopathogenesis of periodontal biofilm
accumulation following the alveolar bone loss observed in humans
(28,29). This pioneering study indicated
that biofilm-induced periodontitis statistically contributes to the
development of larger lesions in 4NQO-induced oral carcinogenesis.
Furthermore, periodontitis associated with 4NQO administration
showed a tendency to develop more epithelial dysplasia and oral
carcinomas than animals receiving 4NQO alone (Fig. 4A and B); however, this latest
finding was not statistically significant.
In the present study, the body weight of all rodents
was assessed as a proxy for their systemic health. All animals
exhibited increased weight during the experiment compared with the
baseline. Notably, the average weight of the Perio group increased
week on week over the whole experiment; however, there was no
difference compared with the Control group, which is in accordance
with the results of other studies (18,19). Regarding the groups exposed to
4NQO, there was ~10% less weight gain from baseline to the final
week compared with that in the control groups, with a particular
slight drop in the final four weeks, which is also in line with the
results of other studies (15,30-32). Significant differences were
observed in the last weeks between the Perio group and the groups
exposed to 4NQO. A parallel may be drawn regarding water
consumption, with groups exposed to 4NQO showing significantly
lower water intake at various time points over 20 weeks compared
with the control groups. Those differences in body weight and
solution consumption observed between control and 4NQO groups are
likely to occur due to the unusual taste of the 4NQO diluted in the
drinking water during the first weeks of induced carcinogenesis, as
proposed in the literature (15).
The challenge of establishing periodontal
etiopathogenesis mediated by ligatures and producing alveolar bone
loss was successfully achieved (18,19). Exposure to 4NQO alone was
sufficient to induce alveolar bone loss to similar levels as that
found in the Perio and 4NQO/Perio groups. This result is in
accordance with our previous study in which spontaneous alveolar
bone loss was also observed in animals receiving 4NQO, and enhanced
when OSCC was present (21).
Changes in the oxidative stress balance are among the possible
mechanisms to support this association (33). 4NQO is capable of inducing
oxidative DNA damage (34). In
parallel, reactive oxygen species are capable of triggering
periodontal tissue damage (35,36). Further studies may be necessary to
elucidate whether the effect of 4NQO in periodontal tissue is a
local response associated with the direct exposure of the oral
mucosa and periodontium to 4NQO-diluted water or if this is a
systemic effect.
To investigate the direct associations between
periodontitis and oral carcinogenesis without confounding factors,
the present study used a well-defined rodent animal model for
inducible periodontitis in conjunction with the 4NQO chemical
carcinogenesis protocol (15,37,38). Administration of 4NQO results in
the formation of DNA adducts resulting in DNA damage and TP53
mutation, along with other hallmarks of tobacco-driven oral
carcinogenesis commonly observed in humans. In the present study,
all animals exposed to 25 ppm 4NQO showed alterations in the
epithelium of the tongue during the 20-week treatment period.
Periodontal disease was induced using ligatures kept in place for
the entire experimental period mimicking the biofilm-induced
periodontal disease found in humans (18,19). To the best of our knowledge, this
is the first time that a ligature-induced periodontal disease
animal model and a 4NQO-induced oral cancer model were implemented
concomitantly to explore the potential correlation between
periodontal disease and tumor progression.
We hypothesized that the presence of periodontitis
would increase the incidence of oral cancer, as suggested in
previous epidemiological and biological studies (5,9,13,39). The present study found a higher
incidence of OSCC and dysplasia lesions in rats with periodontitis
(4NQO/Perio) when compared with the 4NQO group. Although notable,
statistical significance was not demonstrated in these findings.
However, statistical significance was identified when comparing the
tumor size of rats receiving ligatures and 4NQO (overall larger
lesions) and rats receiving 4NQO alone. This finding is similar to
the recent study that showed that chronically infected mice with
P. gingivalis/F. nucleatum exposed to 4NQO in the
drinking water presented with larger tumors and more lesions than
animals without the pathogens (13,40). Also, it has been recently
demonstrated that germ-free mice exposed to 4NQO and colonized with
different oral microbiomes have an increased number and size of
lesions compared with animals that receive 4NQO alone while
remaining germ-free (41).
During the analysis of the animal data in the
present study, it became evident that tumors from the 4NQO group
presented as focal alterations on the spinous layer of the dorsal
epithelium of the tongue. Such alterations resembled the premature
activation of cellular differentiation only observed on the
uppermost layer of the mucosa, a phenomenon histologically
described as dyskeratosis. The process of cellular differentiation
is commonly found to be altered or lost during carcinogenesis,
potentially exerting an important role in tumor behavior.
Mechanistically, activation of cellular differentiation involves
the regulation of cell polarity. Emerging findings show that
several tumor suppressor genes are, in fact, controllers of
cellular polarity, and loss of function of tumor suppressor genes
directly impacts the maintenance of tissue homeostasis by
disrupting symmetrical and asymmetrical cellular division (42,43). In order to investigate the effects
of early carcinogenesis and the potential role of periodontitis in
influencing tumor formation and progression, early changes in
cellular differentiation on the dorsal mucosa of the tongue were
investigated in the present study. Immunofluorescence analysis was
performed for CK10, a major keratin and a marker of keratinocyte
differentiation and keratinization commonly present on
differentiating postmitotic keratinocytes (44,45). As this keratin is associated with
epithelial differentiation, its expression is not expected in the
proliferative basal cells of the mucosa. A recent study pointed out
that the downregulation of CK10 expression in oral dysplastic areas
can indicate malignant transformation (46). The present study found that rats
receiving 4NQO presented with patches of basal epithelial cells
with aberrant expression of CK10. CK10 was also expressed to a
significantly higher level in basal cells from the 4NQO group
(25.3% of fields) compared with that in the 4NQO/Perio (11.5% of
fields). This result indicated that the presence of periodontitis
reduces early activation of cellular differentiation, as evidenced
by the downregulation of CK10 in this group, therefore suggesting
increased transformation potential. In fact, this result aligns
with the observed increased size of lesions found in the 4NQO/Perio
group compared with the group treated with 4NQO alone. Previous
in vitro and in vivo studies suggested that CK10 may
delay and decrease tumor formation, and that its presence in the
basal layer inhibits cell proliferation and prevents skin
tumorigenesis (47-50). Combined, these findings support
the impact of biofilm-driven periodontitis as an associated risk
factor for OSCC progression, as it reduces the activation of the
terminal differentiation process upon exposure to carcinogens.
Although noteworthy, the study fell short of being able to define
the role of periodontitis in the carcinogenesis process and
increased cancer incidence. Despite efforts to recapitulate the
process of carcinogenesis in humans using the tobacco surrogate
4NQO, faster tumor development was observed in the rats, limiting
the study to 20 weeks. The fast growth of tumors in rats contrasts
with the lengthy accumulation of mutations and cellular
transformation driven by tobacco consumption in humans. Such a fast
and aggressive carcinogenesis process may have masked differences
in the tumor initiation process between the 4NQO and 4NQO/Perio
groups. Another limitation of the present study is the
quantification of the tumor volume. 4NQO-induced tumors are highly
infiltrative and difficult to properly dissect. Following
previously published 4NQO studies, in the present study, the tumor
superficial extension was measured rather than attempting to
calculate the tumor volume, in order to avoid the introduction of
bias (20,51).
Nonetheless, even with the aforementioned
limitations, a direct connection was established between the
presence of periodontal disease with larger oral lesions and a
dysfunctional cellular differentiation of surrounding mucosa. The
clinical implications of these findings suggest careful follow-up
is required for patients with periodontal disease who smoke.
Although the influences of periodontitis on cellular transformation
and cancer initiation remain unclear, the present data suggest that
periodontitis plays a role in the progression of tumors.
Nevertheless, future studies need to be performed to better
understand chronic inflammatory diseases from the oral cavity and
their role in oral cancer development and progression.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
TRS, EJG, VPW and CKR contributed to the conception
and design of the study. TRS, FN, VPW, EJG, CHS, VCC, CKR and RMC
contributed to the data acquisition, analysis and interpretation.
TRS, VPW CHS, and RMC contributed to manuscript drafting. All
authors contributed to the critical revisions of the intellectual
content and have approved the final manuscript version to be
published. All authors agree to be accountable for all aspects of
the work so that any questions relating to research integrity or
scientific accuracy in any part of the study are appropriately
investigated and resolved. TRS, VPW, VCC and RMC confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
The study protocol was submitted to and approved by
the Animal Use Ethics Committee, Porto Alegre General Hospital
(Port Alegre, Brazil; approval no. 150475).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
The authors would like to thank Ms. Flavia Rejane
Giusti (Porto Alegre General Hospital, Port Alegre, Brazil) for
providing technical support.
Funding
This study was funded by the Postgraduate Research Group of
Clinics Hospital of Porto Alegre (grant nos. 15-0475 and 16-0541).
This study was partially conducted during a visiting scholar period
at the University of Michigan, sponsored by the Capes Foundation
within the Ministry of Education, Brazil (grant no.
BEX/88881.133691/2016-01). Cassiano Kuchenbecker Rösing is a
research fellow funded by the Brazilian National Council for
Scientific and Technological Development.
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sahingur SE and Yeudall WA: Chemokine
function in periodontal disease and oral cavity cancer. Front
Immunol. 6:2142015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Leemans CR, Braakhuis BJ and Brakenhoff
RH: The molecular biology of head and neck cancer. Nat Rev Cancer.
11:9–22. 2011. View
Article : Google Scholar
|
|
4
|
Warnakulasuriya S: Global epidemiology of
oral and oropharyngeal cancer. Oral Oncol. 45:309–316. 2009.
View Article : Google Scholar
|
|
5
|
Perera M, Al-Hebshi NN, Speicher DJ,
Perera I and Johnson NW: Emerging role of bacteria in oral
carcinogenesis: A review with special reference to perio-pathogenic
bacteria. J Oral Microbiol. 8:327622016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chocolatewala N, Chaturvedi P and Desale
R: The role of bacteria in oral cancer. Indian J Med Paediatr
Oncol. 31:126–131. 2010. View Article : Google Scholar
|
|
7
|
Bravi F, Lee YA, Hashibe M, Boffetta P,
Conway DI, Ferraroni M, La Vecchia C and Edefonti V; INHANCE
Consortium investigators: Lessons learned from the INHANCE
consortium: An overview of recent results on head and neck cancer.
Oral Dis. 27:73–93. 2021. View Article : Google Scholar
|
|
8
|
Tezal M, Grossi SG and Genco RJ: Is
periodontitis associated with oral neoplasms? J Periodontol.
76:406–410. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gondivkar SM, Gondivkar RS, Gadbail AR,
Chole R, Mankar M and Yuwanati M: Chronic periodontitis and the
risk of head and neck squamous cell carcinoma: Facts and figures.
Exp Oncol. 35:163–167. 2013.PubMed/NCBI
|
|
10
|
Michaud DS, Lu J, Peacock-Villada AY,
Barber JR, Joshu CE, Prizment AE, Beck JD, Offenbacher S and Platz
EA: Periodontal disease assessed using clinical dental measurements
and cancer risk in the ARIC study. J Natl Cancer Inst. 110:843–854.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tonetti MS, Jepsen S, Jin L and
Otomo-Corgel J: Impact of the global burden of periodontal diseases
on health, nutrition and wellbeing of mankind: A call for global
action. J Clin Periodontol. 44:456–462. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nagy KN, Sonkodi I, Szöke I, Nagy E and
Newman HN: The microflora associated with human oral carcinomas.
Oral Oncol. 34:304–308. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Binder Gallimidi A, Fischman S, Revach B,
Bulvik R, Maliutina A, Rubinstein AM, Nussbaum G and Elkin M:
Periodontal pathogens Porphyromonas gingivalis and Fusobacterium
nucleatum promote tumor progression in an oral-specific chemical
carcinogenesis model. Oncotarget. 6:22613–22623. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wu JS, Zheng M, Zhang M, Pang X, Li L,
Wang SS, Yang X, Wu JB, Tang YJ, Tang YL and Liang XH:
Porphyromonas gingivalis Promotes 4-nitroquinoline-1-oxide-induced
oral carcinogenesis with an alteration of fatty acid metabolism.
Front Microbiol. 9:20812018. View Article : Google Scholar :
|
|
15
|
Ribeiro DA and Salvadori DM: Gingival
changes in wistar rats after oral treatment with 4-nitroquinoline
1-oxide. Eur J Dent. 1:152–157. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ribeiro DA, Fracalossi AC, Uatari SA,
Oshima CT and Salvadori DM: Imbalance of tumor suppression genes
expression following rat tongue carcinogenesis induced by
4-nitroquinoline 1-oxide. In Vivo. 23:937–942. 2009.PubMed/NCBI
|
|
17
|
Dayan D, Hirshberg A, Kaplan I, Rotem N
and Bodner L: Experimental tongue cancer in desalivated rats. Oral
Oncol. 33:105–109. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cavagni J, de Macedo IC, Gaio EJ, Souza A,
de Molon RS, Cirelli JA, Hoefel AL, Kucharski LC, Torres IL and
Rösing CK: Obesity and hyperlipidemia modulate alveolar bone loss
in Wistar rats. J Periodontol. 87:e9–e17. 2016. View Article : Google Scholar
|
|
19
|
Verzeletti GN, Gaio EJ, Linhares DS and
Rösing CK: Effect of obesity on alveolar bone loss in experimental
periodontitis in Wistar rats. J Appl Oral Sci. 20:218–221. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Squarize CH, Castilho RM, Abrahao AC,
Molinolo A, Lingen MW and Gutkind JS: PTEN deficiency contributes
to the development and progression of head and neck cancer.
Neoplasia. 15:461–471. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Oballe HJR, Muniz FWMG, Bueno CC, Klein
IP, Carrard VC, Rösing CK and Gaio EJ: Spontaneous alveolar bone
loss after 4NQO exposure in Wistar rats. Arch Oral Biol. 89:44–48.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dale BA, Salonen J and Jones AH: New
approaches and concepts in the study of differentiation of oral
epithelia. Crit Rev Oral Biol Med. 1:167–190. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Atanasova KR and Yilmaz O: Looking in the
Porphyromonas gingivalis cabinet of curiosities: The microbium, the
host and cancer association. Mol Oral Microbiol. 29:55–66. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ahn J, Segers S and Hayes RB: Periodontal
disease, Porphyromonas gingivalis serum antibody levels and
orodigestive cancer mortality. Carcinogenesis. 33:1055–1058. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Katz J, Onate MD, Pauley KM, Bhattacharyya
I and Cha S: Presence of Porphyromonas gingivalis in gingival
squamous cell carcinoma. Int J Oral Sci. 3:209–215. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Groeger S, Domann E, Gonzales JR,
Chakraborty T and Meyle J: B7-H1 and B7-DC receptors of oral
squamous carcinoma cells are upregulated by Porphyromonas
gingivalis. Immunobiology. 216:1302–1310. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Meyer MS, Joshipura K, Giovannucci E and
Michaud DS: A review of the relationship between tooth loss,
periodontal disease, and cancer. Cancer Causes Control. 19:895–907.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Graves DT, Fine D, Teng YT, Van Dyke TE
and Hajishengallis G: The use of rodent models to investigate
host-bacteria interactions related to periodontal diseases. J Clin
Periodontol. 35:89–105. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Duarte PM, Tezolin KR, Figueiredo LC,
Feres M and Bastos MF: Microbial profile of ligature-induced
periodontitis in rats. Arch Oral Biol. 55:142–147. 2010. View Article : Google Scholar
|
|
30
|
Al-Koshab M, Alabsi AM, Mohd Bakri M,
Ali-Saeed R and Selvi Naicker M: Antitumor activity of ficus
deltoidea extract on oral cancer: An in vivo study. J Oncol.
2020:54904682020. View Article : Google Scholar :
|
|
31
|
Yanaida Y, Kohno H, Yoshida K, Hirose Y,
Yamada Y, Mori H and Tanaka T: Dietary silymarin suppresses
4-nitroquinoline 1-oxide-induced tongue carcinogenesis in male F344
rats. Carcinogenesis. 23:787–794. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Al-Afifi N, Alabsi A, Kaid F, Bakri M and
Ramanathan A: Prevention of oral carcinogenesis in rats by Dracaena
cinnabari resin extracts. Clin Oral Investig. 23:2287–2301. 2019.
View Article : Google Scholar
|
|
33
|
Araújo AA, Pereira ASBF, Medeiros CACX,
Brito GAC, Leitão RFC, Araújo LS, Guedes PMM, Hiyari S, Pirih FQ
and Araújo Júnior RF: Effects of metformin on inflammation,
oxidative stress, and bone loss in a rat model of periodontitis.
PLoS One. 12:e01835062017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Miranda SR, Noguti J, Carvalho JG, Oshima
CT and Ribeiro DA: Oxidative DNA damage is a preliminary step
during rat tongue carcinogenesis induced by 4-nitroquinoline
1-oxide. J Mol Histol. 42:181–186. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Canakçi CF, Ciçek Y and Canakçi V:
Reactive oxygen species and human inflammatory periodontal
diseases. Biochemistry (Mosc). 70:619–628. 2005. View Article : Google Scholar
|
|
36
|
Takane M, Sugano N, Iwasaki H, Iwano Y,
Shimizu N and Ito K: New biomarker evidence of oxidative DNA damage
in whole saliva from clinically healthy and periodontally diseased
individuals. J Periodontol. 73:551–554. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kanojia D and Vaidya MM:
4-nitroquinoline-1-oxide induced experimental oral carcinogenesis.
Oral Oncol. 42:655–667. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
El-Rouby DH: Histological and
immunohistochemical evaluation of the chemopreventive role of
lycopene in tongue carcinogenesis induced by
4-nitroquinoline-1-oxide. Arch Oral Biol. 56:664–671. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yao QW, Zhou DS, Peng HJ, Ji P and Liu DS:
Association of periodontal disease with oral cancer: A
meta-analysis. Tumour Biol. 35:7073–7077. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Harrandah AM, Chukkapalli SS,
Bhattacharyya I, Progulske-Fox A and Chan EKL: Fusobacteria
modulate oral carcinogenesis and promote cancer progression. J Oral
Microbiol. 13:18494932020. View Article : Google Scholar :
|
|
41
|
Stashenko P, Yost S, Choi Y, Danciu T,
Chen T, Yoganathan S, Kressirer C, Ruiz-Tourrella M, Das B, Kokaras
A and Frias-Lopez J: The oral mouse microbiome promotes
tumorigenesis in oral squamous cell carcinoma. mSystems.
4:e00323–19. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Royer C and Lu X: Epithelial cell
polarity: A major gatekeeper against cancer? Cell Death Differ.
18:1470–1477. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Martin-Belmonte F and Perez-Moreno M:
Epithelial cell polarity, stem cells and cancer. Nat Rev Cancer.
12:23–38. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Moll R, Divo M and Langbein L: The human
keratins: Biology and pathology. Histochem Cell Biol. 129:705–733.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Paramio JM, Santos M and Jorcano JL: The
ends of a conundrum? J Cell Sci. 120:1145–1148. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Garcia NG, Oliveira DT, Lauris JR,
Domingues MA, Minicucci EM and Soares CT: Loss of cytokeratin 10
indicates malignant transformation in actinic cheilitis. Clin Oral
Investig. 20:745–752. 2016. View Article : Google Scholar
|
|
47
|
Reichelt J, Furstenberger G and Magin TM:
Loss of keratin 10 leads to mitogen-activated protein kinase (MAPK)
activation, increased keratinocyte turnover, and decreased tumor
formation in mice. J Invest Dermatol. 123:973–981. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Paramio JM, Casanova ML, Segrelles C,
Mittnacht S, Lane EB and Jorcano JL: Modulation of cell
proliferation by cytokeratins K10 and K16. Mol Cell Biol.
19:3086–3094. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Santos M, Paramio JM, Bravo A, Ramirez A
and Jorcano JL: The expression of keratin k10 in the basal layer of
the epidermis inhibits cell proliferation and prevents skin
tumorigenesis. J Biol Chem. 277:19122–19130. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Santos M, Perez P, Segrelles C, Ruiz S,
Jorcano JL and Paramio JM: Impaired NF-kappa B activation and
increased production of tumor necrosis factor alpha in transgenic
mice expressing keratin K10 in the basal layer of the epidermis. J
Biol Chem. 278:13422–13430. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Czerninski R, Amornphimoltham P, Patel V,
Molinolo AA and Gutkind JS: Targeting mammalian target of rapamycin
by rapamycin prevents tumor progression in an oral-specific
chemical carcinogenesis model. Cancer Prev Res (Phila). 2:27–36.
2009. View Article : Google Scholar
|