Introduction
Breast cancer is the most prevalent cancer in women
worldwide (1); triple-negative
breast cancer (TNBC), accounts for ~15% of all breast cancer cases,
which has a dismal prognosis (2).
TNBC is characterized by the absence of expression of estrogen
receptor (ER), progesterone receptor (PR), and epidermal growth
factor receptor 2 (ERBB2 or HER2) and exhibits significant
intratumoral heterogeneity (3).
Despite the improved survival rate of patients with ER+,
PR+, or HER2+ breast cancer with the
development of endocrine and targeted therapies (4), patients with TNBC encounter
difficulties as there are no particular drug targets available
(3). Patients with TNBC are
primarily treated with chemotherapy.
Guanylate-binding protein 2 (GBP2), a member of the
interferon-gamma (IFN-γ)-inducible GTPase superfamily, was
identified for its protective effects against microorganisms
(5). In studies on tumors, GBP2
was differentially expressed in various cancer cells (6-10).
Although some studies have provided evidence of GBP2 involvement in
breast cancer progression (11-13), the specific mechanisms underlying
its role require further investigation.
Paclitaxel (PTX), a mitotoxic agent (14), is extensively used in TNBC
chemotherapy due to its cytotoxic effects (15). However, drug resistance often
leads to treatment failure. Multiple mechanisms contribute to the
resistance of PTX in breast cancer, including drug efflux proteins
of the ABC superfamily (16), MAP
proteins, SACs (17), and
epigenetic regulation by miRNAs (18). Among these mechanisms, PTX-induced
protective autophagy is quite critical (19).
Autophagy is a dynamic process essential for
maintaining cellular homeostasis; it can promote tumor cell
survival through protective autophagy or trigger type II
(autophagic) cell death (20).
Autophagy and drug resistance have a complicated interaction, and
different autophagy inhibitors and inducers have been identified to
restore sensitivity to PTX in tumor cells, with efficacy influenced
by tumor characteristics and progression stages (21). Existing studies have confirmed
that autophagy activated by inhibition of the Ras-Raf,
PI3K/AKT/mTOR signaling pathway results in increased PTX
sensitivity, which in turn suppresses tumor cell growth (22,23).
The development of multiple tumors is strongly
linked to autophagy, with autophagosome formation being a crucial
step in this process (24).
Autophagy-related protein 2 (ATG2) is responsible for the
transmembrane transport of phospholipids and is essential for
autophagosome formation (25).
Recently, several proteins have been reported to regulate ATG2
transcription (26-28); however, the roles of GBP2 and ATG2
in autophagy have not yet been investigated, therefore exploring
the relationship between the roles of GBP2 and ATG2 in autophagy
may reveal new mechanistic insights to improve TNBC therapy.
Before studying the role of GBP2 and autophagy,
results demonstrated that the concentration of PTX significantly
affected the expression of GBP2; therefore, it was hypothesized
that GBP2 may be related to PTX sensitivity through certain
mechanism. Based on the knowledge that autophagy has a significant
impact on drug-resistance in TNBC (29), it was reasonable to conduct a
study on GBP2 and autophagy in order to further explore whether
there was a tandem relationship between them and PTX
sensitivity.
The present study verified the low level of GBP2
expression in TNBC and its inhibitory effects in vivo and
in vitro. The function of GBP2 in regulating autophagy in
TNBC cells was investigated and the mechanism by which GBP2
facilitates autophagy via inhibiting the PI3K/AKT/mTOR signaling
pathway and interacting with ATG2 was elucidated. Furthermore, it
was demonstrated that GBP2-activated autophagy reversed PTX
resistance in TNBC, providing a promising strategy for overcoming
resistance.
Materials and methods
Plotting of Kaplan-Meier (K-M)
curves
In the present study, the expression data of GBP2 in
breast cancer was queried from The Cancer Genome Atlas dataset
(https://www.cancer.gov/ccg/research/genome-sequencing/tcga).
The role of GBP2 in patient survival was assessed with the KM
Plotter online database (http://kmplot.com/analysis/) using the log-rank test.
'Gene Symbol=GBP2' was entered, 'Jetset Best Probe Set' was
selected, and 'Median' was used to classify patients. Then
'StGallen's classification' was selected, and survival analysis was
performed for each subtype (including basal/luminal A/luminal
B/HER2+).
Cell lines and cultures
MDA-MB-231 (cat. no. HTB-26) and MDA-MB-436 (cat.
no. HTB-130) were selected to represent TNBC cell lines and MCF10A
(CRL-10317) to represent a normal (non-carcinogenic) breast
epithelial cell line. All cell lines were provided by The American
Type Culture Collection. At 37°C and in 5%
CO2-humidified air, all the cell lines were cultured in
DMEM media (Gibco; Thermo Fisher Scientific, Inc.) supplemented
with 10% fetal bovine serum, 100 g/ml penicillin and 100 U/ml
streptomycin.
PTX-resistant MDA-MB-231/PR and MDA-MB-436/PR cells
were developed by exposure to low doses of PTX (Selleck Chemicals)
over an extended period. The dosage was gradually increased until
reaching a concentration of 50 nM, which is the maximum recommended
concentration for clinical use, to ensure the cells could survive
consistently.
RNA transfection
The overexpressing (OE)-DNA sequence targeting GBP2
was cloned into the lentiviral vector (Shanghai GenePharma Co.,
Ltd.) to generate a stable cell line overexpressing GBP2. Empty
load virus (provided by the same supplier) as negative control for
transfection. The RNA sequences were as follows: GBP2-OE-DNA
forward, 5′-GCGATCGCATGGCTCCAGAGATCAACT-3′
and reverse, 5′-ACGCGTGAGTATGTTACATATTGGCT-3′
(Underlined are Restriction Enzyme cutting site). A total of 1.5
μg of pBI-GBP2-EGFP vector (cat. no. 6154-1; BD
Biosciences), which expresses GBP2 with the C-terminal EGFP tag,
and 2 μg of pIRESpuro plasmid (cat. no. KL101-994; Shanghai
kanglang biological technology Co., Ltd.) were co-transfected into
293T cells (Shanghai GenePharma Co., Ltd.) to generate the
lentivirus. In addition, the two vectors were also cotransfected
with 1 μg of psPAX2 (cat. no. P0261) and pMD2.G (cat. no.
P0262; both from miaolingbio) viral packaging plasmids during
transfection. After incubation in a 37°C cell culture incubator for
about 48 h, filtered viral supernatant could be collected and
stored at -20°C. Based on the screening of positive resistant cell
lines, the encapsidated lentivirus generated in the supernatant of
their culture was collected to transfect TNBC cells to obtain
stable GBP2 overexpressing cell lines. Briefly, seeding
~0.5-1×106 TNBC cells each in 10-cm2 tissue
culture plates until the confluent prior reached to 40-50%, then
polybrene medium (target cell medium) mix were prepared (8
μg/ml). The medium in the target cells was aspirated using
Pasteur pipette and 1 ml polybrene medium mix was added and then
incubated for ~1-2 min. Next, lentivirus was picked out and thawed
in a 37°C water bath before adding it to the TNBC cells. A total of
9 ml of lentiviral mix were added. When puromycin was present at a
concentration of 0.5 mg/ml, puromycin-resistant clones were chosen
after incubation for 48 h. Utilizing reverse transcription
quantitative PCR (RT-qPCR) and western blotting (WB), the
inducibility of the GBP2-EGFP fusion protein in the chosen clones
was evaluated.
RT-qPCR
The relative GBP2 mRNA expression was calculated by
RT-qPCR, using the 2−ΔΔCq method for quantification
(30). The cells were treated
according to the manufacturer's instructions to extract total RNA
using a TRIzol reagent (cat. no. 15596018; Thermo Fisher
Scientific, Inc.). According to the manufacturer's instructions,
extracted RNA was then reverse-transcribed into cDNA using a cDNA
synthesis kit from Fermentas; Thermo Fisher Scientific, Inc. The
SYBR Premix Ex Taq Master Mix (cat. no. PR820A; Takara Bio, Inc.)
was used to conduct qPCR. Thermocycling conditions were as follows:
95°C for 30 sec; 95°C for 5 sec; 55°C for 30 sec; and 72°C for 30
sec, 40 cycles in total. The chosen endogenous control was β-actin.
The primer pairs used for RT-qPCR were as follows: β-actin forward,
5′-TCA CCA ACT GGG ACG ACA-3′ and reverse, 5′-ACA GCC TGG ATA GCA
ACG-3′; and GBP2 forward, 5′-CTA TCT GCA ATT ACG CAG CCT-3′ and
reverse, 5′-TGT TCT GGC TTC TTG GGA TGA-3′.
WB
Total protein within cells was extracted with
precooled RIPA lysate (cat. no. P0013B; Beyotime Institute of
Biotechnology) and quantified using a BCA kit (cat. no. 23227;
Thermo Fisher Scientific, Inc.). Protein was denatured first with
incubation at 100°C for 5 min, and then 20 μl was added to
10% SDS-PAGE gel for electrophoresis at 80 and 120 V for 30 and 90
min. The PVDF membranes were blocked with 5% skimmed milk before
incubating with primary antibodies (1:1,000) overnight at 4°C, then
the secondary antibodies (1:5,000; cat. no. ab150077; Abcam) were
incubated for 1 h at room temperature. Under the application of
increased chemiluminescence (cat. no. 34002; Thermo Fisher
Scientific, Inc.), the membranes were visualized under a gel
imaging system (Tanon Co., Ltd.) and gray value analysis was based
on ImageJ software (version 2.0.0; National Institutes of Health).
The primary antibodies listed below were employed: anti-β-actin
(cat. no. A5441), anti-p62 (cat. no. SAB5700845), anti-LC3I/II
(cat. no. L8918; all from Sigma-Aldrich; Merck KGaA), anti-GBP2
(cat. no. 11854-1-AP), anti-ATG2 (cat. no. 23226-1-AP), anti-ATG5
(cat. no. 10181-2-AP; all from Proteintech Group, Inc.), anti-PI3K
(cat. no. 4292), anti-Phospho-PI3K (cat. no. 4228), anti-Akt (cat.
no. 9272), anti-Phospho-Akt (cat. no. 9271), anti-AMPK (cat. no.
2532), anti-Phospho-AMPK (cat. no. 2531), anti-mTOR (cat. no.
2972), anti-Phospho-mTOR (cat. no. 2971), anti-ULK1 (cat. no. 8054)
and anti-Phospho-ULK1 (cat. no. 5869; all from Cell Signalling
Technology, Inc.).
Colony formation assay
A total of 1,000 cells/well were inoculated in
6-well culture plates. The medium was renewed every 3 days and the
culture was continued until 14 days or until the number of cells in
the majority of individual clones was >50. After fixation by
adding 1 ml of 4% paraformaldehyde to each well for 30 min at 20°C,
the cells were observed and images were captured under a Nikon
microscope (Nikon Corporation).
Cell Counting Kit-8 (CCK-8) assay
96-well plates containing transfected cells were
planted with 1×104 cells per well. Then, each well
received 100 ml of diluted CCK-8 solution (cat. no. CK04; Dojindo
Laboratories, Inc.) and was incubated at 37°C for 4 h. The optical
density (OD) was measured at 450 nm. For assessing cell viability,
the OD value of each group was recorded at fixed time points,
calculating relative cell viability as follows: Relative cell
viability=(ODt-OD0)/OD0, where ODt represents the optical density
value of each well at the given time point, and OD0 is the optical
density value of the cell culture solution at the start of
incubation (0 h). IC50 values were calculated after the
relative viability curves plotted after 24 h of treatment with
equal gradient concentrations of PTX were completed.
5-ethynyl-2′-deoxyuridine (EDU)
assay
Following the directions from the manufacturer,
tumor pieces were found using an EDU immunofluorescence labeling
test (cat. no. 15176-29-1; Sigma-Aldrich, Merck KGaA). The cells
were exposed to 50 mM EDU for a duration of 6 h before fixing,
permeabilizing and staining with EDU in accordance with the
manufacturer's instructions. The cell nuclei were dyed for 20 min
with 1 mg/ml of DAPI (Sigma-Aldrich; Merck KGaA). Fluorescence
microscope (Nikon 80i; Nikon Corporation) was used to observe the
cells containing EDU.
Matrigel invasion assay
The upper chamber of the Transwell plate (cat. no.
3422; Corning, Inc.) was filled with 25 μl of diluted
Matrigel (cat. no. YZ-354234; Becton, Dickinson and Company),
covered with a polycarbonic membrane (cat. no. 3428; Corning,
Inc.), and subsequently polymerized into a gel by incubating at
37°C for 30 min. Next, 4×104 cells starved for 24 h were
planted in medium without serum in the inserts. Cell invasion
caused by cell proliferation was less likely to happen when the
cells were cultured in a medium without serum. The inserts were
stored in 24-well plates containing DMEM and 10% FBS at 37°C for 24
h. The invasive cells were located at the bottom side of the
membrane in the inserts. These cells were fixed with ice-cold
methanol for 10 min at 20°C and then stained for 20 min with a
solution of 0.2% crystal violet at room temperature. A Nikon
digital camera was used to count the number of marked cells on the
membrane.
Gap closure test
Cell migration was assessed using the gap closure
assay. Culture insert wells were injected with cells that were in
the logarithmic growth phase. In an incubator, cells were cultured
for 24 h. Using sterile forceps, the insert was carefully removed,
and 1 ml of serum-free media was introduced to each well. At 0 and
24 h after gap closure, the experimental field of view was observed
under a Nikon microscope (Nikon Corporation).
Transmission electron microscopy
The specimens underwent a treatment using 2.5%
glutaraldehyde in a solution of 0.1 M sodium cacodylate and were
subsequently kept at a temperature of 4°C before being embedded.
Following this, they were subjected to a post-fixation process
utilizing 1% osmium tetroxide and underwent a gradual dehydration
procedure utilizing ethanol and propylene oxide. Following
embedding, an ultramicrotome (LKB-I) was used to cut ultrathin
(55-60 nm) sections from the samples. Samples were viewed utilizing
the electron microscope (JEM-1200EX) after staining.
Immunofluorescence staining and confocal
fluorescence microscopy
Cells were fixed for 20 min at 20°C with 100%
ice-cold methanol. The slides were then blocked with closure buffer
(1 PBS/5% normal serum/0.3% Triton X-100) for 1 h at 4°C. Then,
cells were treated with specific primary antibody (1:200), and kept
at the temperature of 4°C for an overnight period, followed by 1 h
at 25°C with an Alexa Fluor 488-conjugated goat anti-rabbit IgG
secondary antibody (1:1,000). The 4′,6-diamidino-2-phenylindole dye
was used to stain the nuclei for 5 min at room temperature before
blocking them with Antifade Reagent. Finally, a confocal
fluorescence microscope (STELLARIS 5; Leica Microsystems, Inc.) was
used to gather and evaluate images. The tumors were stained with
immunofluorescence in accordance with the manufacturer's
instructions (Wuhan Servicebio Technology Co., Ltd.).
The mRFP-GFP-LC3 lentivirus (Hanbio Biotechnology
Co., Ltd.) transfection was utilized to identify and track LC3 in
order to track the autophagic flux. According to the manufacturer's
recommendations, cells were infected for 48 h with a tandem
fluorescent mRFP-GFP-tagged adenovirus. Confocal fluorescence
microscopy was used to capture the images. Red (RFP signal alone)
and yellow (merge of GFP and RFP signal) puncta depict early and
late autolysosomes, respectively. The color shift of GFP/mRFP was
used to assess autophagic flow.
Xenotransplantation studies
A total of 48 SPF grade female BALB/c nude mice,
aged 4 weeks and weighing between 16-20 g, were used in the present
study. The mice were randomly assigned to either the experimental
group or the control group. In the experimental group, a
100-μl cell suspension prepared in PBS (1×107
cells) was injected, while in the control group, an equivalent dose
of physiological saline was administered. Then, each group of mice
was subjected to identical culture conditions, with a 12/12-h
light-dark cycle and controlled temperature maintained at 22-25°C
for optimal growth, while maintaining a relative humidity range of
40~60%. Calipers were used to measure tumor growth, and the tumor
volume was calculated as follows: Tumor volume
(mm3)=[width (mm)]2 × length (mm) ×0.5.
Immunofluorescence, EDU labeling, and electron microscopy were all
applied to the tumor sections. On the 35th day following the
placement of the tumor, intraperitoneal injections of mice with 150
mg/kg sodium pentobarbital were performed following the euthanasia
guidelines of the Group Standard of the Chinese Society of
Laboratory Animals (T/CALAS 31-2017). Animal studies were approved
(approval no. 2022-0016) by The Animal Experimentation Ethics
Committee of Chongqing Medical University approved (Chongqing,
China).
Patient studies
The specimens used for immunohistochemistry in the
study were obtained from patients with confirmed TNBC, with an age
range of 45-68 years old, recruited at the First Hospital of
Chongqing Medical University (Chongqing, China), and the
recruitment period was from January 2020 to January 2022, totaling
two years. Approval for the utilization of clinical samples
(approval no. 2020-279) was granted by The Review Board of the
First Affiliated Hospital of Chongqing Medical University
(Chongqing, China). Written informed consent for participation in
the present study was provided by all patients.
Specialty solvents
In some experiments, DMSO (cat. no. D2650;
Sigma-Aldrich; Merck KGaA) was used to configure Compound C (CC;
cat. no. HY-13418A; MedChemExpress); Chloroquine (CQ; cat. no.
C6628; Sigma-Aldrich; Merck KGaA) reagent was used to treat
MDA-MB-231 cells for 24 h. The concentration of CC was 7.5
μmol/ml, and the concentration of CQ was 50
μmol/l.
Statistical analysis
The SPSS Statistics 25 (IBM Corp.) was used to
conduct statistical analysis. The immunofluorescence data were
analyzed using ImageJ software. Pearson correlation coefficients
were subsequently calculated to quantify the degree of
co-localization between the two proteins as previously described
(31). The Cancer Genome Atlas
dataset (https://www.ncbi.nlm.nih.gov/projects/gap/cgi-bin/study.cgi?study_id=phs001709.v1.p1)
was analyzed to compare the expression levels of GBP2 in breast
cancer tissues vs. normal breast tissues (https://portal.gdc.cancer.gov/projects/CMI-MBC).
The results of at least three independent experiments are reported
as the mean ± standard deviation. Using the Student's t-test,
differences across groups were examined. P<0.05 was considered
to indicate a statistically significant difference.
Results
GBP2 expression is downregulated in
patients with TNBC and is associated with poor prognosis
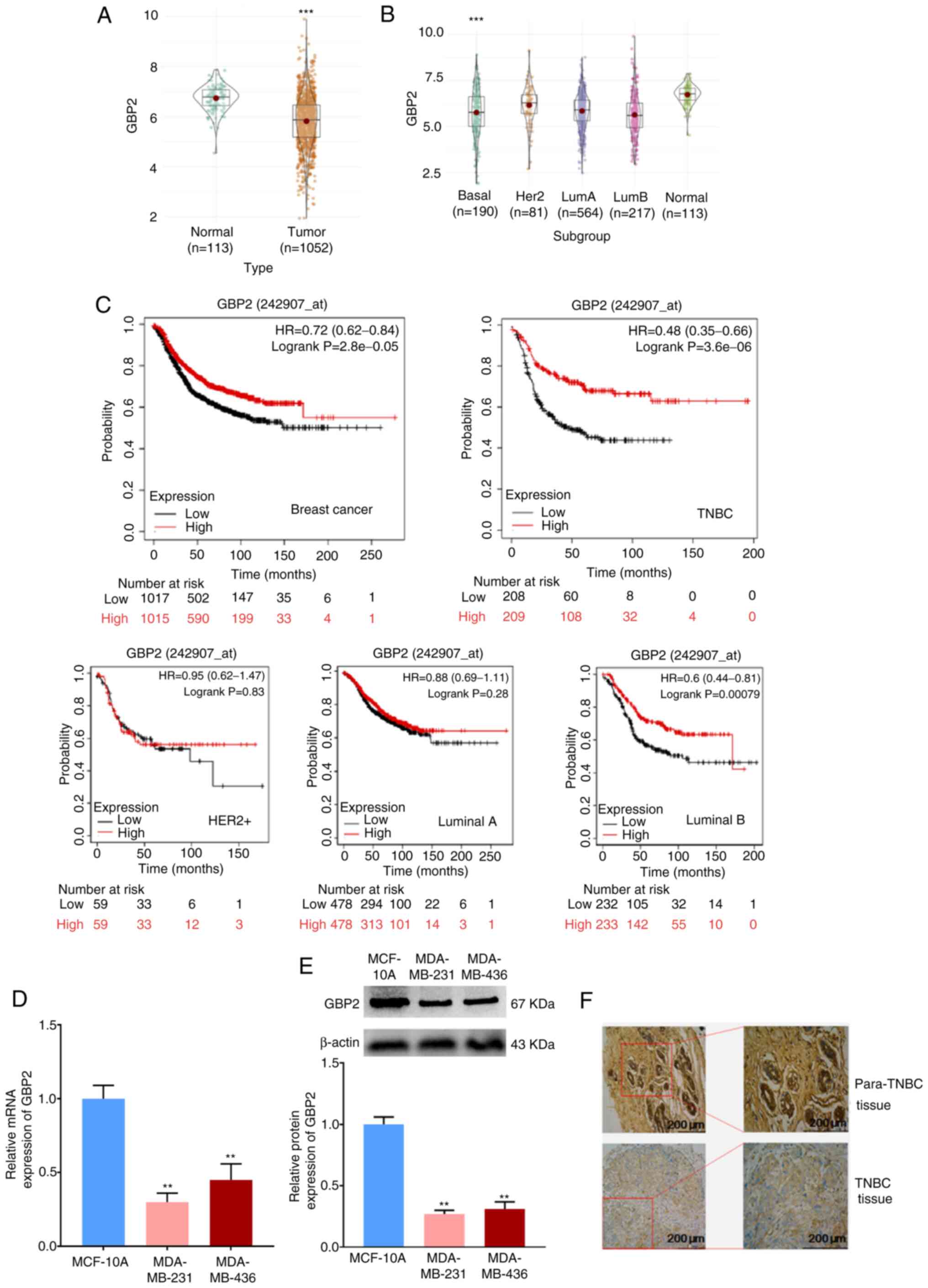
First, it was confirmed that the expression level of
GBP2 was significantly lower in breast cancer tissues compared with
normal tissues (P<0.001). (Fig.
1A). Comparison of the expression levels of GBP2 in various
subtypes of breast cancer with those in normal breast tissue
revealed that GBP2 expression was low and statistically different
in TNBC (basal) (Fig. 1B). The
role of GBP2 in patient survival was assessed using the KM Mapper
database (http://kmplot.com/analysis/),which
demonstrated that patients with low expression of GBP2 had a
significantly lower relapse-free survival rate
(P=2.8×10−5), and not all patients with lower GBP2
expression had a significantly decreased recurrence-free survival
rate in each subtype, but the subtype basal (log-rank
P=3.6×10−6) was statistically different (Fig. 1C), suggesting that that low GBP2
level is predictive of a poor prognosis in TNBC (basal)
subtype.
MDA-MB-231 and MDA-MB-436 cells were chosen as
experimental cell lines, whereas MCF10A cells were designated as
the control cell line. The use of RT-qPCR assay to detect the mRNA
level of GBP2 transcripts (Fig.
1D) and WB assay to detect GBP2 protein (Fig. 1E) demonstrated the significantly
low expression of GBP2. Finally, immunohistochemical experiments
(Fig. 1F) using paraffin
specimens of cancer and paracancerous tissues from patients with
TNBC identified that TNBC cells expressed GBP2 at a considerably
lower level.
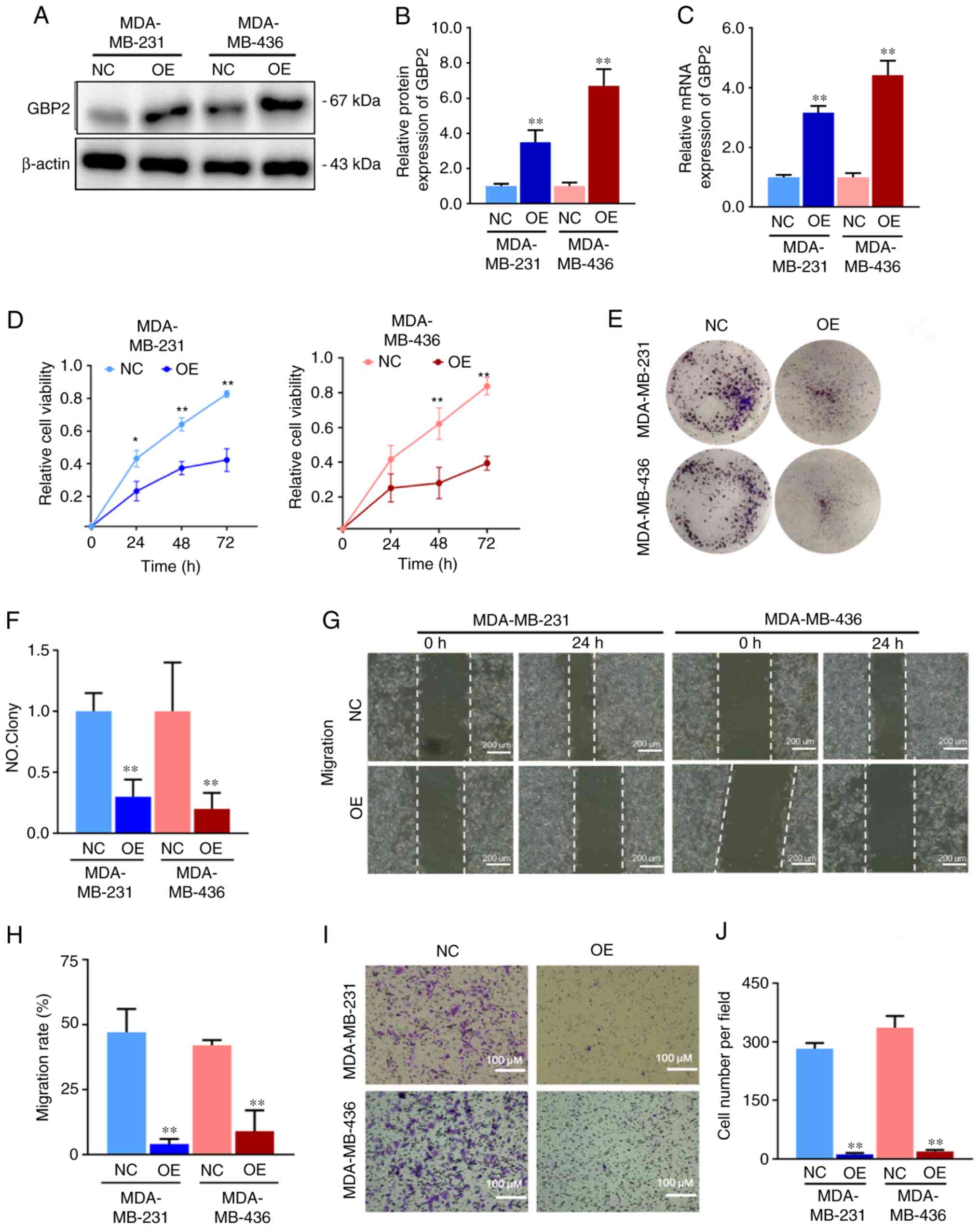
GBP2 overexpression suppresses the
proliferation, invasion and metastasis of TNBC cells
For further comprehension of GBP2's function in
TNBC, MDA-MB-231 and MDA-MB-436 cells were stably transfected with
GBP2-OE lentivirus. The transfection efficiency of lentivirus
overexpressing GBP2 was evaluated in advance and the influence of
other factors on transfection was eliminated (Fig. S1A). After screening as specified,
the mRNA and protein expression levels of GBP2 were significantly
higher (Fig. 2A-C), suggesting
that GBP2 was successfully stably transfected. MDA-MB-231 and
MDA-MB-436 cells overexpressing GBP2 are termed OE group, and GBP2
wild-type cells are termed NC group.
The effect of GBP2 on cell viability and
proliferation was evaluated using the CCK-8 (Fig. 2D) and colony formation assays
(Fig. 2E and F). Based on the
outcomes, the proliferation of MDA-MB-231 cells was significantly
decreased 24 h after inoculation, and the proliferation of
MDA-MB-436 cells significantly decreased 48 h after inoculation
following the overexpression of GBP2. In addition, colony formation
experiments were performed to further examine the proliferation of
MDA-MB-231 and MDA-MB-436 cells after GBP2 overexpression,
especially in terms of clone-forming ability, which is closely
related to the ability of the cells to generate metastasis.
MDA-MB-231-OE and MDA-MB-436-OE cells generated significantly fewer
colonies compared with NC, suggesting a significant role of GBP2 in
inhibiting the proliferation and clone-forming ability of TNBC
cells.
Migration and invasion are the key cellular events
involved in tumor progression. The migration of TNBC cells after
GBP2 overexpression was first examined using the gap closure assay
(Fig. 2G and H). The results
indicated that the migration rate of MDA-MB-231 or MDA-MB-436 cells
overexpressing GBP2 at 24 h was significantly lower than that of
the control cells, revealing that GBP2 conducted a significant
function in the inhibition of TNBC cell migration. Cancer cells
cross the basement membrane when they invade the lymphatic or
vascular bed to spread. To investigate whether GBP2 is crucial in
the invasion of TNBC cells, the number of invasive cells migrating
through the precoated Matrigel on the upper side of the pore
filters was measured using invasion chambers in Transwell
experiments. Similar to what the cell migration test revealed in
findings, the invasion rates of MDA-MB-231 and MDA-MB-436 cells
overexpressing GBP2 were significantly lower than those of their
respective control tumor cells (Fig.
2I and J), suggesting that GBP2 can significantly reduce the
invasive potential of TNBC cells and inhibit their progression of
tumor cells.
Analysis of the malignant behavior of MDA-MB-231 and
MDA-MB-436 TNBC cells identified that the proliferation, migration
and invasion of TNBC cells in vitro were significantly
reduced after GBP2 overexpression, indicating that GBP2 serves a
crucial role in preventing the progression of TNBC cancer.
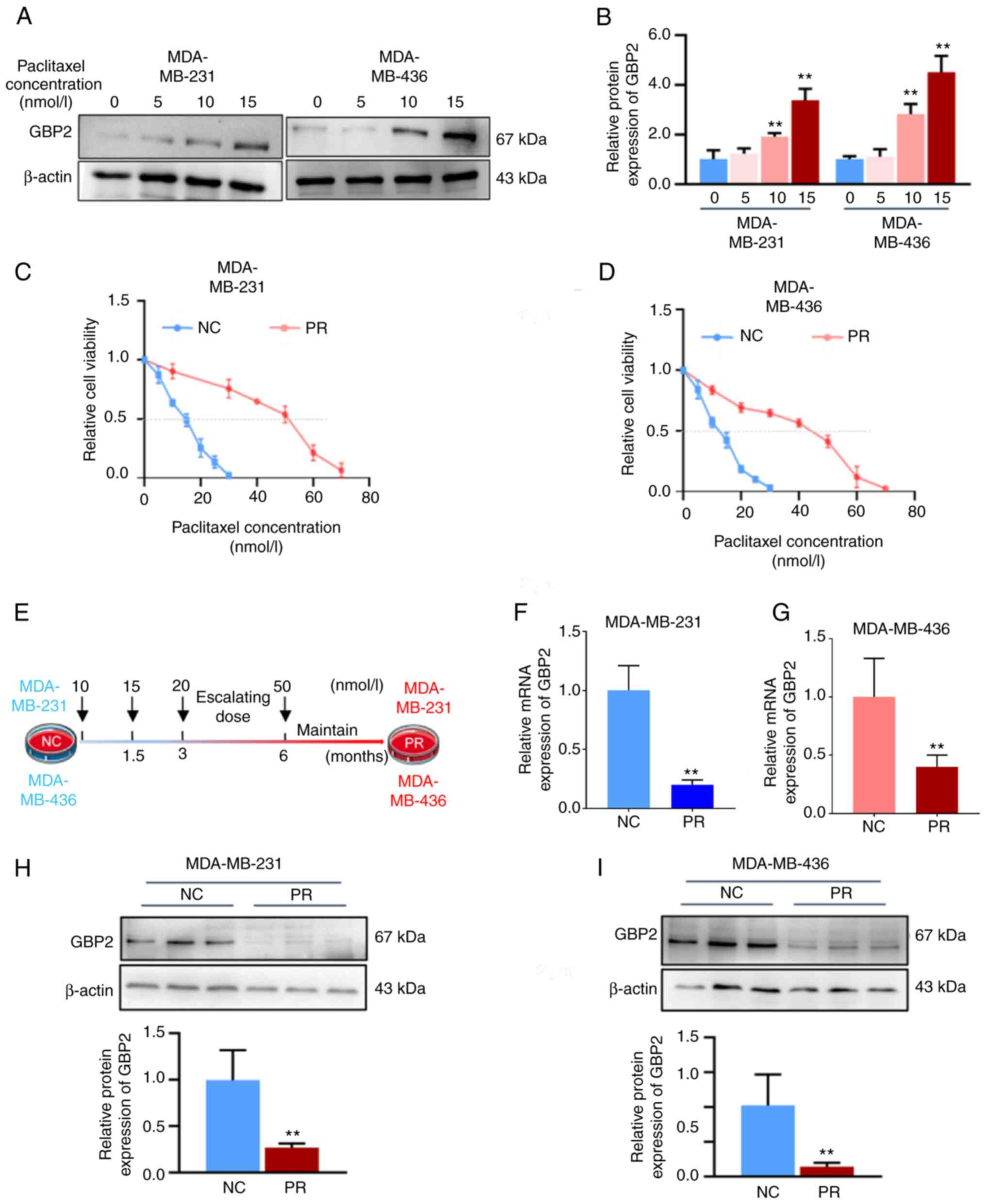
Establishing and validated PTX-resistant
TNBC cell lines
PTX is the most commonly employed chemotherapeutic
agent in the clinical management of TNBC. To explore whether GBP2
affects the sensitivity of PTX, MDA-MB-231 and MDA-MB-436 cells
were initially treated with various PTX concentrations (5, 10 and
15 nM) for 24 h, and the expression levels of GBP2 protein were
detected by WB. In MDA-MB-231 and MDA-MB-436 cells, GBP2 protein
levels increased with increasing PTX concentrations, with
significant differences starting at 10 nM (Fig. 3A and B). This revealed that GBP2
protein levels were positively associated with PTX drug
concentration, suggesting that GBP2 may play a synergistic role
with PTX in exerting cytotoxicity, and indicated that GBP2 likely
affects PTX drug sensitivity.
Subsequently, the PTX-resistant MDA-MB-231 and
MDA-MB-436 cell lines were established. Various PTX concentrations
were applied to the cell lines for 24 h; CCK-8 assay was used to
assess the IC50 values of PTX in MDA-MB-231-NC and
MDA-MB-436-NC, and the results were 14.9 and 12.5 nM, respectively
(Fig. 3C and D). Subsequently,
the concentration of PTX was started at 10 nM, and the MDA-MB-231
and MDA-MB-436 cells were provided with PTX treatments at higher
concentrations once the cells were acclimated for 6 months, after
which the concentration of PTX was maintained at 50 nM, and the
cells proliferated stably (Fig.
3E). The cell viability assay data suggested that the
IC50 values of the domesticated treated cells (referred
to as PR group) became significantly higher (51.5 nM in
MDA-MB-231-PR, 42.4 nM in MDA-MB-436-PR) (Fig. 3D and E), proving that
PTX-resistant cell lines were successfully established in TNBC.
Overexpression of GBP2 reverses PTX
resistance in TNBC
First, RT-qPCR and WB assays were utilized to detect
mRNA and protein levels of GBP2 in wild-type (NC group) and
PTX-resistant (PR group) MDA-MB-231 and MDA-MB-436 cells,
respectively, and it was revealed that both PTX-resistant strains
had significantly lower GBP2 expression (Fig. 3F-I).
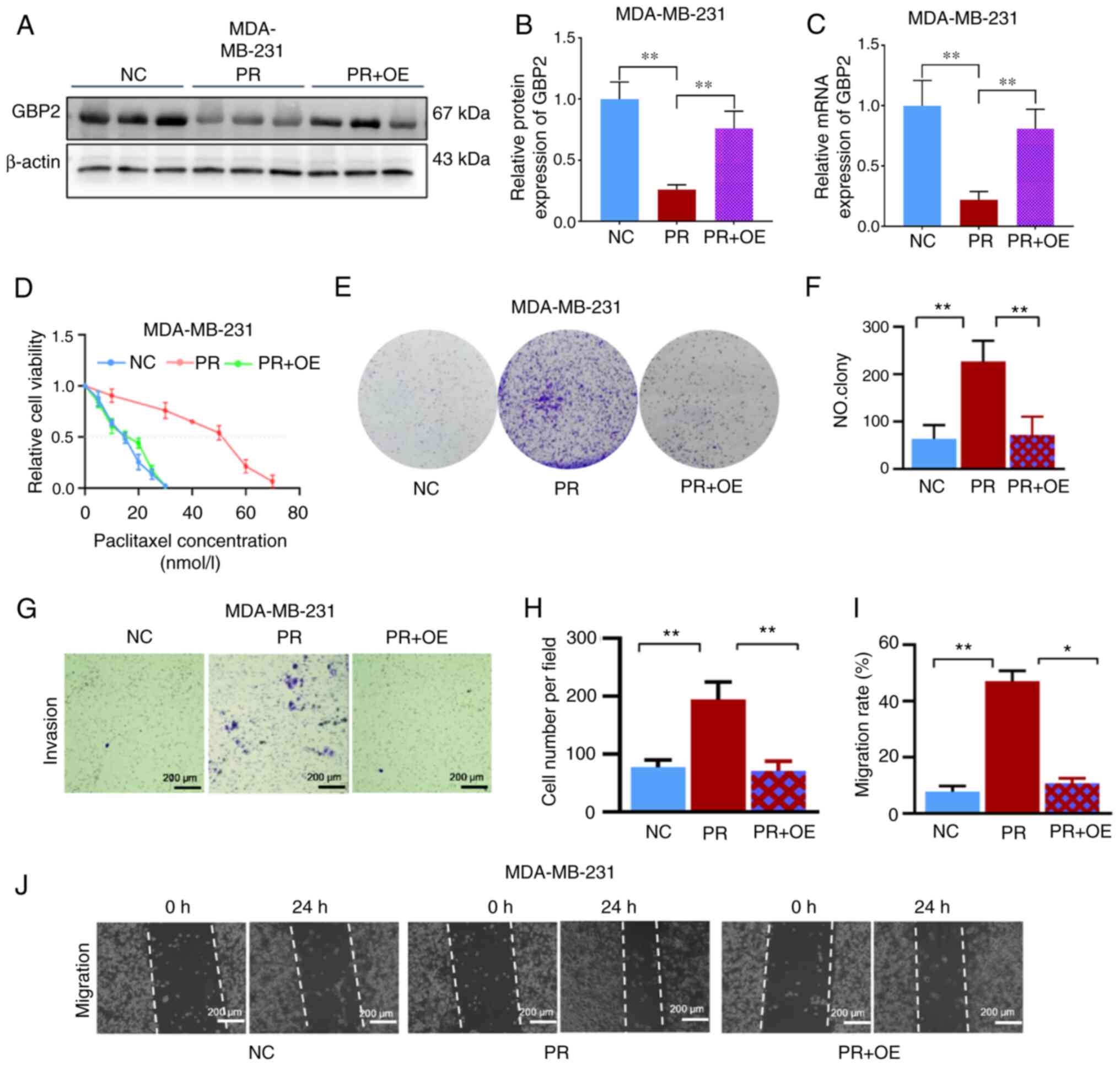
Subsequently, the primary experimental target
consisted of PTX-resistant MDA-MB-231 cells and transfected with
overexpression of GBP2 adenovirus. The efficiency of viral
transfection in resistant cells was examined to ensure the accuracy
of subsequent experiments (Fig.
S1B). WB assays (Fig. 4A and
B) and RT-qPCR experiments (Fig.
4C) were performed on MDA-MB-231 parental cells (NC),
PTX-resistant cells (PR), and resistant + transfected cells
overexpressing GBP2 (PR + OE); the mRNA level of GBP2 in PR was
found to be substantially lower than that of NC, and after
adenoviral transfection, the mRNA level of GBP2 rebounded
significantly; the situation of GBP2 protein expression level was
similar to that of mRNA, confirming the successful overexpression
of GBP2 in PTX-resistant MDA-MB-231 cells.
According to findings from the CCK-8 experimental
cell viability assay, the PR + OE group's IC50 value
(15.0 nM) was significantly lower than the PR group's value (51.5
nM) (Fig. 4D), indicating that
GBP2 overexpression significantly enhanced the PTX sensitivity of
MDA-MB-231 cells. To further illustrate this, colony formation
experiments were performed, which revealed that PR cells produced
significantly more colonies than the NC group (P<0.01); whereas,
after GBP2 overexpression (PR + OE), cells had significantly fewer
colonies compared with PR (P<0.01) (Fig. 4E and F). The Transwell assay was
used to measure the number of migratory cells in each of the three
groups; the PR group had a significantly higher number of cells per
unit area compared with the NC group (P<0.01), and substantially
lower in the PR + OE group than in the PR group (P<0.01)
(Fig. 4G and H). Finally, the
migration of cells in the three groups was examined using the gap
closure assay; considering the images and the calculated migration
rates, it was determined that cells in the PR group had a markedly
greater capacity for invasion than those in the NC group, while the
invasive ability of the drug-resistant cell line with GBP2
overexpression was substantially reduced in the PR + OE group
compared with the PR group (P<0.05) (Fig. 4I and J).
In conclusion, it was demonstrated that
PTX-resistant TNBC cells had considerably lower levels of GBP2
expression, and the resistance to PTX was reversed after the
overexpression of GBP2 by adenoviral transfection. Additionally,
the outcomes revealed that PTX-resistant cells' viability was
considerably decreased when GBP2 was overexpressed, which renders
PTX more toxic to drug-resistant TNBC cells and is crucial in
reversing PTX resistance in TNBC.
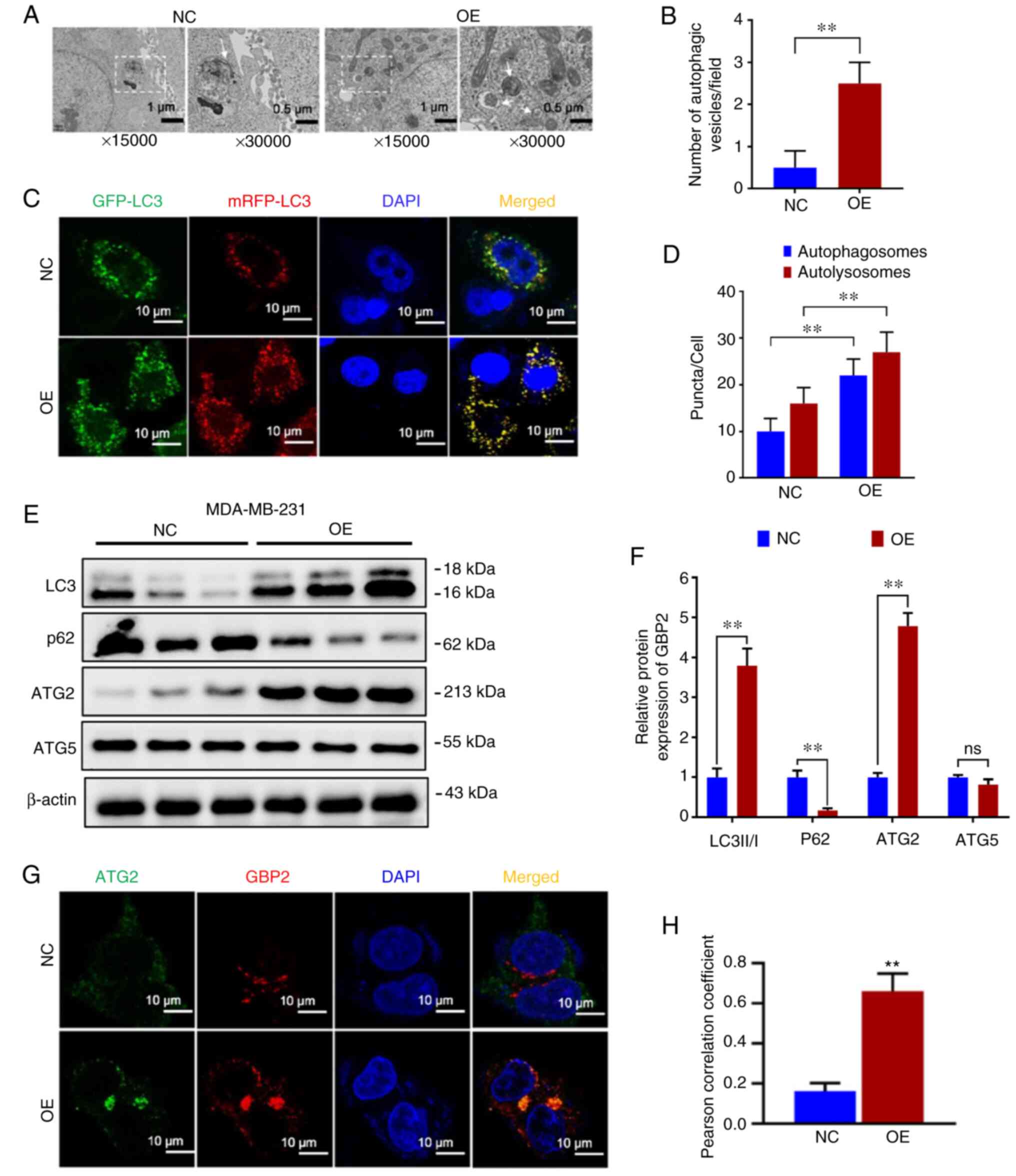
Overexpression of GBP2 activates
autophagy in TNBC
Because drug resistance is often closely related to
autophagy, the difference in autophagy between parental cells (NC)
and GBP2 overexpressing (OE) MDA-MB-231 cells was examined. First,
cell crawls were made, and autophagic vesicles were observed under
electron microscopy. Compared with the NC group, the OE group was
demonstrated to have more autophagic vesicles per unit field of
view (Fig. 5A and B), suggesting
enhanced autophagy in the OE group. To further confirm this, the
expression of LC3 was racked by infecting with GFP-mRFP-LC3
double-labeled virus, and it was observed that the level of red
fluorescence showed a significant increase in the OE group compared
with the NC group, and yellow fluorescence was significantly
increased following merging (Fig.
5C), suggesting that autophagy lysosomes and autophagosomes
were increased, which were quantified as demonstrated in the
histogram (P<0.01) (Fig. 5D).
Based on the results, it was observed that the autophagic flux was
significantly higher in the OE group compared with the NC group,
indicating that GBP2 overexpression promoted autophagy in
MDA-MB-231 cells. Finally, WB was used to compare the
autophagy-related proteins of the two groups of cells. It was
revealed that LC3 II/I ratio of the OE group was significantly
higher than the NC group, while P62 was significantly lower
(Fig. 5E and F), indicating that
GBP2 overexpression enhanced cellular autophagy.
GBP2 enhances autophagy by inhibiting the
PI3K/AKT/mTOR pathway and acting in combination with ATG2
To explore the mechanism underlying autophagy
activation by GBP2 overexpression, an examination of
autophagy-related proteins was conducted. Among the ATG family
proteins closely related to autophagy, ATG5 was not significantly
altered; however, ATG2 was significantly elevated in the OE group
(Fig. 5E and F), suggesting that
there may be a specific synergistic effect of GBP2 and ATG2 in
autophagy. Subsequently, GBP2 and ATG2 were fluorescently labeled
by immunofluorescence co-localization and it was observed that both
demonstrated significantly increased expression in the OE group,
with highly consistent localization (Fig. 5G). The Pearson's correlation
coefficient was calculated using ImageJ with the collection of
fluorescent spots expression and localization data; the OE group
demonstrated a significant increase (P<0.01) (Fig. 5H), suggesting that GBP2 and ATG2
interacted and jointly enhanced autophagy.
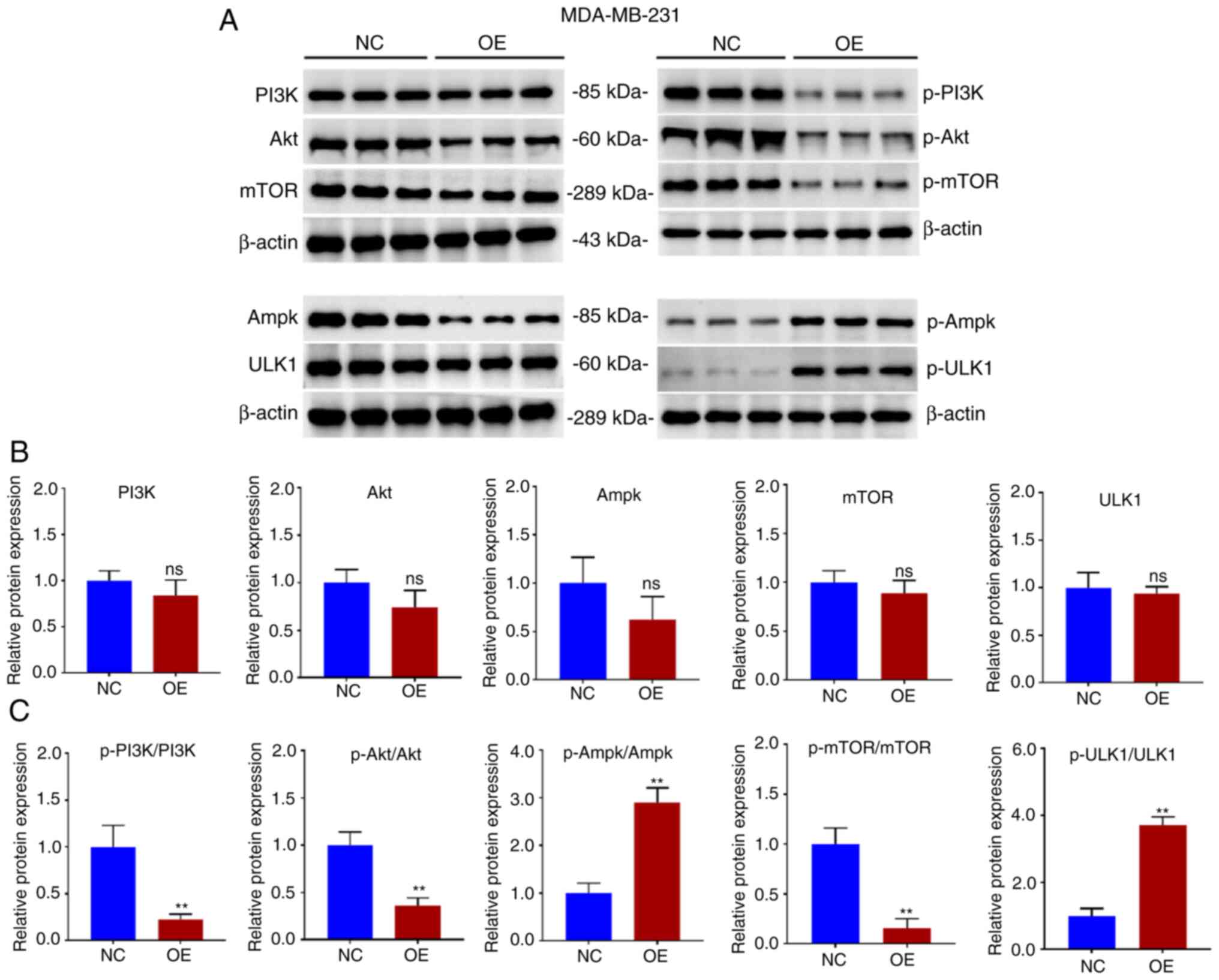
PI3K/AKT/mTOR is a primary pathway affecting
autophagy, and whether the overexpression of GBP2 affected
autophagy was further explored. Proteins were extracted from
MDA-MB-231 parental cells (NC) and GBP2 overexpression stable
transgenic cells (OE) and WB experiments were performed (Fig. 6A). It was revealed that the
overall protein levels of PI3K/AKT/mTOR did not exhibit significant
differences between the group OE and NC (Fig. 6B), whereas the group that revealed
overexpression (OE) exhibited a notable decrease in the levels of
phosphorylated (p-) PI3K, AKT and mTOR (P<0.01) (Fig. 6C), suggesting that GBP2
overexpression significantly suppressed the PI3K/AKT/mTOR pathway's
activity. Ampk, an upstream protein of mTOR, inhibits mTOR, and
ULK1, a downstream target of mTOR, is repressed by mTOR. The
results of WB identified that there was an increase in the
phosphorylation proportions of p-Ampk/Ampk and p-ULK1/ULK1,
consistent with previous results. CC is an Ampk inhibitor that
activates downstream mTOR signaling. The treatment of CC in
MDA-MB-231 cells with GBP2-OE decreased LC3-II and p-Ampk, while
increased p62 and p-mTOR levels (Fig.
7A-F), suggesting that reactivation of PI3K/AKT/mTOR after GBP2
overexpression leads to the inhibition of autophagy. Based on the
results, it can be inferred that GBP2 plays a crucial role in
regulating autophagy by suppressing PI3K/AKT/mTOR signaling.
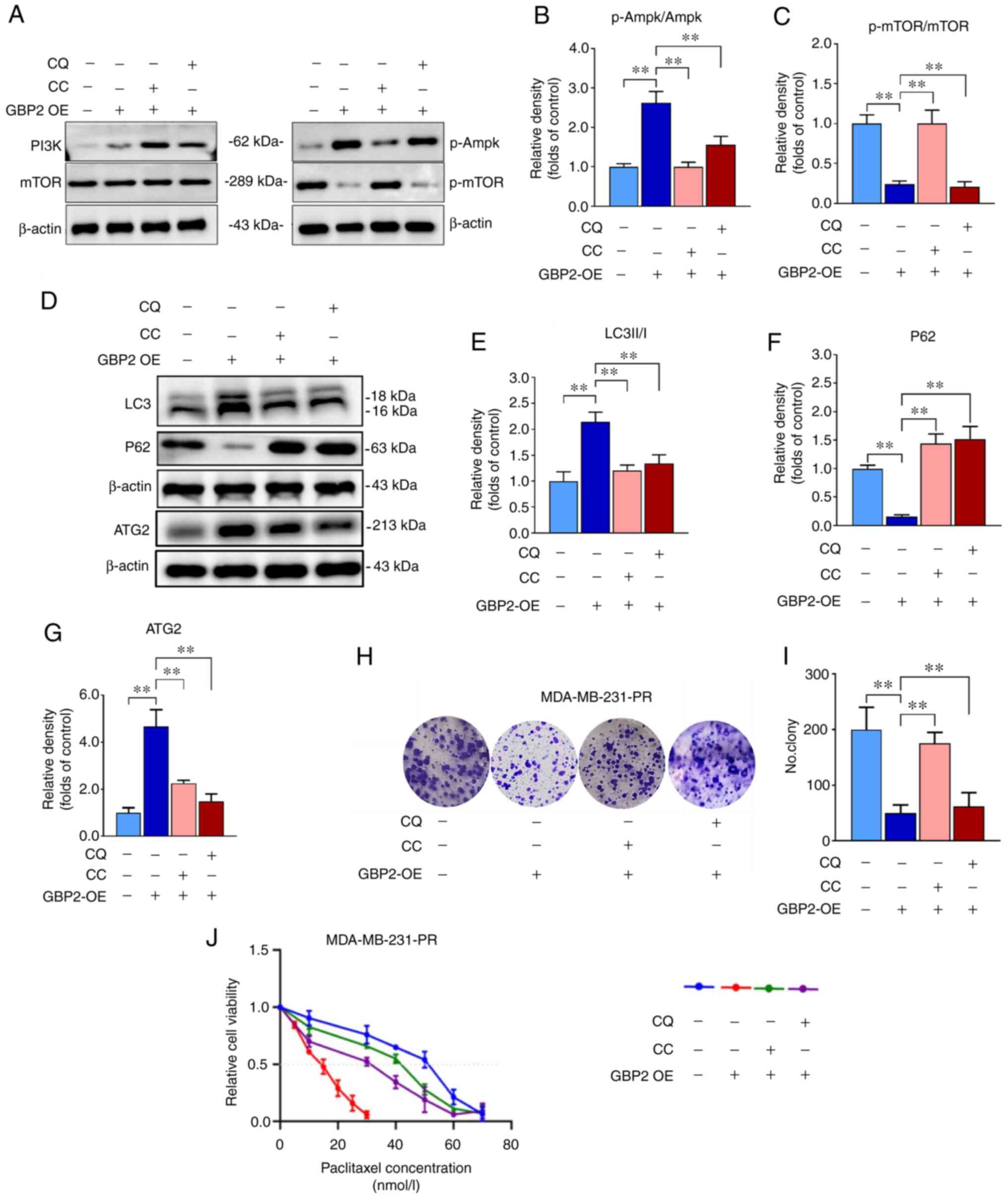 | Figure 7Enhanced autophagy by GBP2 leads to
the reversal of paclitaxel resistance in triple-negative breast
cancer cells. GBP2 overexpression or empty adenovirus was
transfected into MB231 paclitaxel-resistant cells, and cells were
treated with CQ (50 μmol/l) or CC (7.5 μmol/ml) or
none for 24 h. (A) Ampk and mTOR protein and phosphorylation levels
were detected using WB. (B and C) Relative density represents
relative phosphorylation protein expression of AMPK and mTOR. (D)
Autophagy-related proteins LC3 II/I, P62 and ATG2 were detected
using WB. (E-G) The relative protein expression levels were
quantified using bar graphs. (H and I) The colony formation assay
was utilized to ascertain proliferation of cells in every treatment
group. (J) Cell vitality was determined utilizing the Cell Counting
Kit-8 assay after treating four groups of cells with increasing
concentrations of paclitaxel for 24 h. The IC50 value
was calculated using SSPS, and results from low to high are as
follows: 18.0, 31.5, 40.5 and 52.5 nM. **P<0.01.
GBP2, guanylate-binding protein 2; CQ, chloroquine; CC, Compound C;
ATG, autophagy-related gene; WB, western blotting; OE,
GBP2-overexpression group; p-, phosphorylated. |
Enhanced autophagy by GBP2 leads to
reversal of PTX resistance in TNBC cells
CQ, a classic autophagy inhibitor, prevents
autophagosomal fusion and degradation; this effect of CQ was
applied in MDA-MB-231-PR cells overexpressing GBP2, and the results
indicated a decline in the LC3II/I ratio and an increase in P62,
confirming that autophagy was inhibited (Fig. 7D-F). The expression level of ATG2
in each group was also examined and it was reconfirmed that GBP2
enhances autophagy synergistically with ATG2 (Fig. 7D and G). Additionally, colony
formation experiments revealed that overexpression of GBP2 in
MDA-MB-231-PR cells substantially augmented inhibition of cell
viability and that CQ could mitigate this effect (Fig. 7H and I), suggesting the inhibition
of autophagy while restoring cell growth viability; similar
outcomes were obtained utilizing CC (Fig. 7H and I). Notably, the decrease in
cell viability caused by GBP2 expression is crucial in reversing
PTX resistance. These results suggested that autophagy may be
related to the chemosensitivity of PTX to MDA-MB-231-PR cells.
It was investigated whether GBP2-mediated autophagy
activation contributes to the reverse of drug resistance in TNBC
cells. The IC50 was determined to verify the effect of
various factors on the sensitivity of MDA-MB-231-PR cells to PTX
using the CCK-8 assay. The results revealed that GBP2
overexpression significantly reduced the IC50 value,
suggesting that the PTX sensitivity of drug-resistant cells was
significantly increased, and the addition of CQ or CC caused the
IC50 value to rise significantly, leading to increased
resistance to PTX (Fig. 7J).
These results suggested that CQ and CC inhibit autophagy activated
by GBP2 overexpression, consequently increasing cellular resistance
to PTX and demonstrating that autophagy activated by GBP2 leads to
the reversal of PTX resistance in TNBC cells.
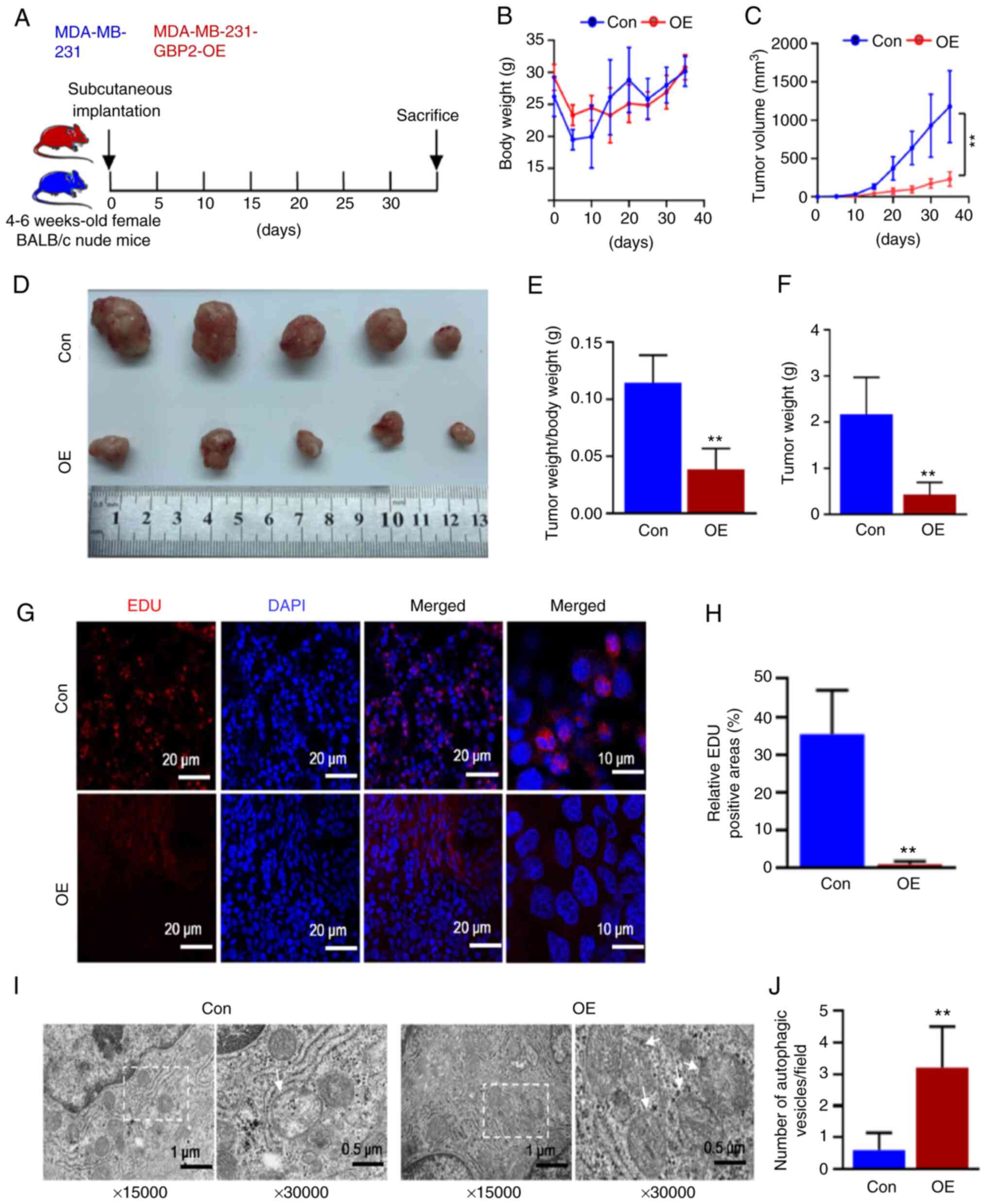
GBP2 overexpression suppresses the growth
of TNBC tumors in vivo by inhibiting proliferation and enhancing
autophagy
To confirm the oncogenic and autophagy-enhancing
effects of GBP2 overexpression on TNBC in vivo, female
BALB/c nude mice aged between 4-6 weeks were selected and randomly
divided into control (Con) and GBP2-overexpression (OE) groups for
the subcutaneous injection of MDA-MB-231 parental cells and GBP2
overexpression cells for subcutaneous tumor formation experiments
(Fig. 8A). Every five days, the
volume of tumors and body weight of the mice were recorded
(Fig. 8B and C). There was not a
substantial disparity in weight regarding the two groups, according
to the results, whereas the OE group revealed significantly reduced
tumor volume and growth rate. The tumor size was observed after 35
days of feeding (Fig. 8D), and
the tumor weight and tumor weight/body weight ratio of the OE group
were calculated to be lower (Fig. 8E
and F). These results confirmed that GBP2 overexpression
significantly inhibited the growth of TNBC tumors.
Subsequently, the tumors were sectioned and BrdU
staining experiments were performed. Confocal microscopy revealed
that the EDU red fluorescence in the OE group was considerably
weaker (Fig. 8G). The
fluorescence intensity was quantified (Fig. 8H), demonstrating that the growth
of tumor cells was significantly reduced in the OE group. According
to electron microscopy findings (Fig.
8I and J), the OE group had significantly more autophagic
vesicles per unit of area, indicating enhanced autophagy.
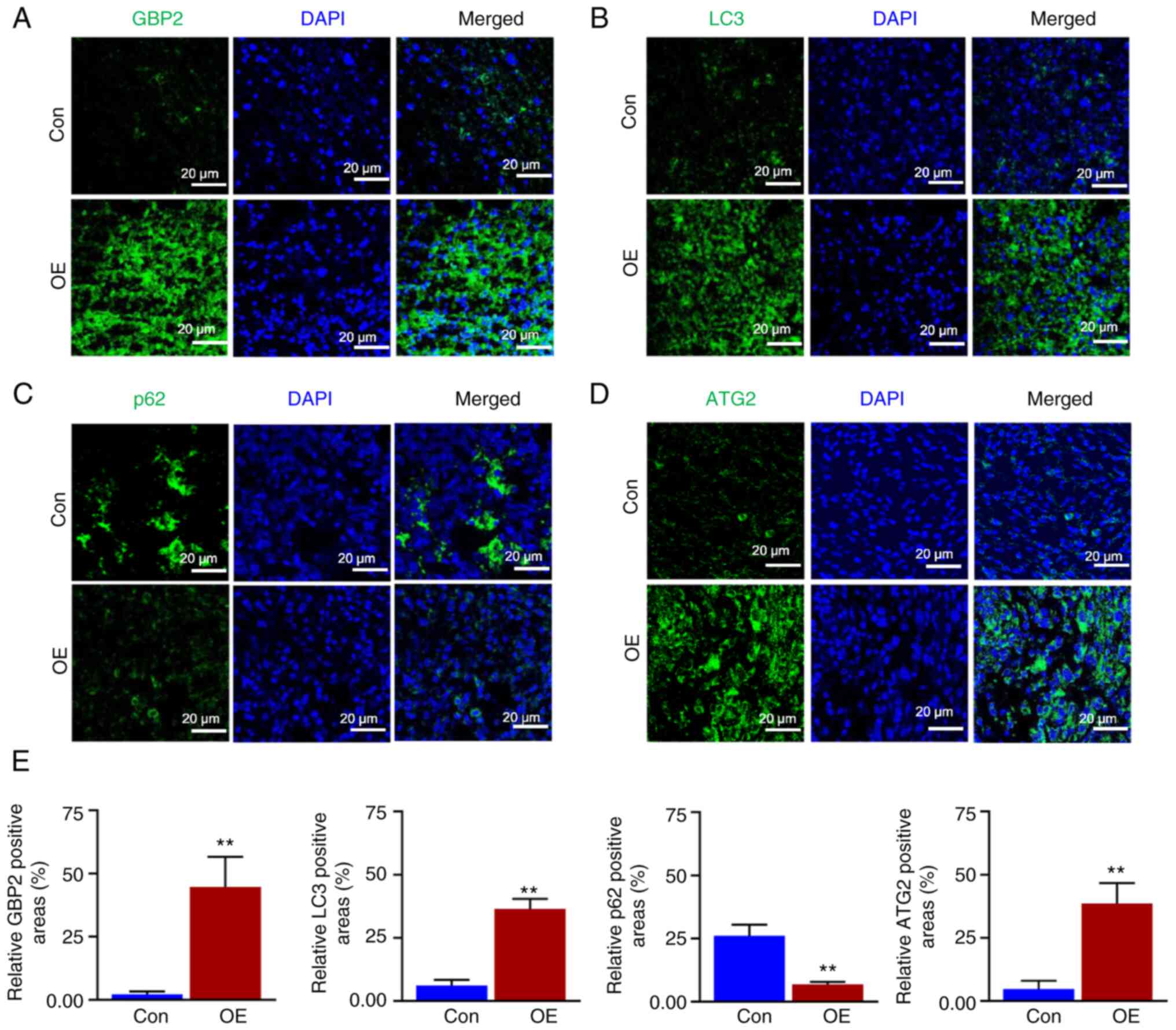
Fluorescence staining experiments were performed (Fig. 9A-D), and the relative fluorescence
positivity was quantified using histograms (Fig. 9E). The level of GBP2 expression
was significantly greater in the OE group than in the control
group, thus confirming that GBP2 was overexpressed in the OE group
(Fig. 9A), and the expression
level of the LC3 protein related to autophagy was significantly
higher in the OE group than in the control group (Fig. 9B); the autophagic substrate p62
was significantly lower (Fig.
9C), confirming that the overexpression of GBP2 enhanced
autophagy in TNBC in vivo. Additionally, the outcomes
demonstrated a parallel upward trend in ATG2 and GBP2 expression
levels (Fig. 9A and D), which
confirmed that GBP2 and ATG2 expression levels were positively
associated and jointly enhanced autophagy in vivo.
Discussion
The present study was conducted to investigate the
function of GBP2 in TNBC, particularly PTX-resistant TNBC, by
utilizing numerous in vivo and in vitro experiments.
The results indicated that forced expression of GBP2 in TNBC cells
led to decreased cell proliferation, invasion and migration.
Notably, it was revealed that that the overexpression of GBP2
significantly activated cellular autophagy. The mechanism
underlying this effect was further elucidated, revealing that GBP2
enhanced autophagy by inhibiting the PI3K/AKT/mTOR pathway and
interacting with ATG2. Consequently, activation of autophagy
increases TNBC cell sensitivity to PTX. These results highlighted
the anticancer role of GBP2 in TNBC and demonstrated its ability to
promote autophagic cell death, which improves PTX resistance.
GBP2 is expressed in numerous tumor cells at
different levels and plays distinct roles. The current results
indicated the detection of GBP2 in the nucleoplasm and cytoplasm of
TNBC, which is consistent with the findings of The Human Protein
Atlas database (https://www.proteinatlas.org/ENSG00000162645-GBP2/subcellular),
but a more precise localization deserves to be verified by further
experiments. GBP2 is upregulated in esophageal squamous carcinoma,
glioblastoma, and renal clear cell carcinoma and plays a
significant pro-cancer role (6-8).
The expression level of GBP2 was noticeably reduced in both
colorectal cancer cells and cutaneous melanoma, which exhibited
anti-cancer properties (9,10).
However, further study is required to determine the role of GBP2 in
TNBC. Notably, the present study confirmed low GBP2 expression in
TNBC cells, verified by cellular RT-qPCR, WB and pathological
tissue IHC experiments. Previous bioinformatics analysis by the
authors revealed that GBP2 expression levels in TNBC were lower
compared with those in normal breast tissues (Zhang et al,
unpublished data). Notably, in previous immunohistochemical
staining experiments by the authors, no instances of high GBP2
expression in TNBC samples were observed. However, the limited
sample size prevented the authors from identifying individual cases
of IHC staining indicating high GBP2 expression in TNBC. It is
possible that further investigation with a larger sample set may
uncover instances of high GBP2 expression. Previous studies and the
current analysis using online resources have identified that
patients with TNBC who express low levels of GBP2 have a poorer
prognosis, suggesting that GBP2 may conduct an essential function
in the progress of TNBC. In vitro experiments illustrated
that overexpressing GBP2 significantly reduced the proliferation,
migration and invasion of TNBC cells, confirming that hypothesis.
In vivo, tumors in the GBP2 overexpression group revealed
significant growth inhibition in a subcutaneous transplantation
model of TNBC mice. EDU staining suggested that GBP2 overexpression
significantly reduced tumor proliferation; GBP2 overexpression
significantly enhanced the autophagy level of tumor cells by
immunofluorescence detection. In brief, the present study revealed
for the first time, to the best of the authors' knowledge, that
GBP2 inhibits TNBC cell progression both in vitro and in
vivo. Since GBP2 has been demonstrated to inhibit colon cancer
proliferation through the Wnt pathway (9), and to promote glioma invasion
through the Stat3-fibronectin pathway (7), together with the present current
experimental results, it is reasonable to assume that GBP2 also
modulates the malignant behavior of TNBC by affecting some
independent signaling pathway, but this needs to be explored in
further experiments. Meanwhile, outcomes suggested that
overexpression of GBP2 activates autophagy, which may be the key
reason for stronger cell death. Therefore, the present study
focused on autophagy activation by GBP2.
Autophagy is essential for the development and
advancement of breast cancer and influences breast cancer cell
proliferation, invasion, migration and drug resistance (32). As research progresses, the key
role of autophagy in TNBC has emerged, providing new therapeutic
paradigms (29). However,
autophagy performs a complex function in tumor progression, and it
exhibits functions that inhibit or promote tumor progression in
different tumor types and experimental models (33); several studies have revealed that
autophagy protects TNBC tumor cells and the tumor microenvironment
from cell proliferation, invasion and migration (34-36). Conversely, higher levels of ATG7
expression induce apoptosis in TNBC cells (37), and anti-CD73 monoclonal
antibody-induced autophagy impedes TNBC cells from migrating and
encroaching (38). This finding
demonstrates that autophagy acts as a tumor suppressor in TNBC. The
findings of the present study indicated that in vitro
experiments, including examination of autophagic vesicles by
electron microscopy, detection of autophagy-associated proteins by
WB, observation of immunofluorescence under confocal microscopy and
in vivo experiments of fluorescence staining in tumor
sections, suggested that the autophagic flux of TNBC cells was
significantly enhanced after overexpression of GBP2. To determine
whether activated autophagy acts as a mechanism of cell death or
protection (39), cells were
treated with reagents to suppress autophagy, and the results
suggested that cell growth was restored after autophagy inhibition,
demonstrating that pro-death autophagy activated by GBP2 is crucial
in inhibiting TNBC progression.
Further experiments were performed to explore the
specific mechanisms of GBP2 autophagy activation. Previous studies
have identified several autophagic pathways (40) including the Ampk (41,42), MAPK/JNK (43) and Wnt/β-catenin pathways (44). Notably, the PI3K/AKT/mTOR pathway
has a significant effect on autophagy (45), and mTOR signaling is highly
correlated with autophagy regulation (46). The present study indicated that
the overexpression of GBP2 in MDA-MB-231 cells decreased the
phosphorylation of PI3K/AKT/mTOR pathway proteins significantly;
however, the application of CC, an Ampk inhibitor upstream of mTOR,
activated the mTOR expression and restored the vitality. This
demonstrated that GBP2 overexpression activated pro-death autophagy
in MDA-MB-231 cells by inhibiting the PI3K/AKT/mTOR pathway.
Simultaneously, it was explored whether GBP2 could
link proteins closely related to autophagy in eliciting its effect.
In vitro, the WB assay of MDA-MB-231 cells overexpressing
GBP2 indicated a noticeable rise in ATG2 compared with parental
cells, while no significant difference was observed with ATG5.
In vivo, tumor tissue immunofluorescence staining revealed
that the GBP2 overexpression had elevated ATG2 expression.
Subsequently, the results of immunofluorescence co-localization
suggested that the protein regions of GBP2 and ATG2 significantly
overlapped. These outcomes indicated that GBP2 and ATG2 exert
synergistic effects on autophagy. Recent research has centered on
the identification of a protein or factor that interacts with ATG2
to preserve efficient lipid transference during autophagy and
provides energy for unidirectional transport (47). Based on the results of the current
study and GBP2 being a member of the GTPase superfamily, which is
closely related to energy metabolism, it can be inferred (for the
first time) that GBP2 is a protein that closely synergizes with
ATG2 to enhance autophagy. However, the specific mechanisms of the
interaction between GBP2 and ATG2 require further experimental
exploration.
PTX is typically the primary chemotherapy drug used
to treat TNBC. However, the emergence of drug resistance has
resulted in a poor patient prognosis. Hence there is an urgent need
to address this issue. Wang et al (9) revealed that GBP2 could improve the
chemosensitivity of colorectal cancer cells. Notably, the
expression level of GBP5 (a cognate family of GBP2) was correlated
with PTX resistance (48); GBP5
expression level was associated with PTX resistance. In a recent
study by the authors (49), a
biomarker model with a strong predictive ability for chemotherapy
response in breast cancer was constructed, with GBP2 being a
crucial member. The aforementioned study confirmed that GBP2
overexpression can significantly affect the autophagy of TNBC
cells, and autophagy is crucial in PTX resistance (21). Therefore, it is reasonable to
assume that GBP2-activated autophagy is related to PTX sensitivity
in TNBC. A PTX-resistant strain of MDA-MB-231 cells was firstly
established and GBP2 was subsequently overexpressed in the
resistant strain by adenoviral transfection; the IC50
values of PTX were significantly reduced after treatment relative
to the control group. These results suggested that the increased
sensitivity of TNBC resistant cells to PTX was observed after the
overexpression of GBP2. Collectively, it has been already confirmed
that GBP2 overexpression can significantly enhance autophagy in
TNBC cells, therefore it could be hypothesized that GBP2-activated
autophagy is the cause of the reversal of drug resistance in TNBC.
To prove this, a rescue experiment in which the autophagy inhibitor
CQ was added on the basis of GBP2 overexpression was performed, and
the results showed that the sensitivity of the cells to PTX
decreased again, suggesting that the activation of autophagy
contributed to the enhanced sensitivity to PTX. Since the present
study has confirmed that one of the mechanisms by which GBP2
activates autophagy is through inhibition of PI3K/AKT/mTOR, mTOR
activator CC was added to GBP2 overexpression and decreased PTX
sensitivity was also observed. This further proved that autophagy
activated by GBP2 overexpression causes the reversal of TNBC drug
resistance. However, in addition to its use as an autophagy
inhibitor, CQ is now widely used as an antimalarial and
rheumatologic drug with apoptotic and necrotic and
immunomodulatory/anti-inflammatory properties (50), thus there is still the possibility
of potential effects on cells other than autophagy. CC also has
potential effects on kinases other than AMPK (51). These effects were therefore
considered by the authors and their evaluation in future
experiments is considered.
Several studies have highlighted autophagy-induced
enhancement of PTX sensitivity. For example, treatment of
PTX-resistant thyroid cancer cells with a dual mTOR inhibitor,
vizusertib, led to a substantial decrease in the IC50 of
PTX; notably, cells treated with the combination of the two
compounds accumulated more autophagic vesicles (52). In addition, natural compounds
(53) and recombinant
proteins/antibodies (54,55) enhance the cytocidal effects of PTX
by inducing autophagy. Although there appears to be a dearth of
ongoing clinical trials that concentrate on utilizing cytotoxic
autophagy as a viable treatment option for TNBC, autophagy
induction combined with PTX has great therapeutic potential and
deserves further extensive investigations, which emphasizes the
clinical significance of the present study.
The present study still has certain limitations. In
the CCK-8 assay to assess cell viability, efforts were made to
minimize error by consistently adding the same number of cells to
each well at the experiment's outset, uniformly measuring the OD
value at a wavelength of 450 nm, and averaging the results from
three wells. However, it was challenging to predict whether the OD
value would fall within the range most consistent with the
linearity rule, which is between 0.75-1.25. Therefore, the
limitation is acknowledged and more precise experimental design
will be pursued in future endeavors. In the follow-up experiments
involving the autophagy activation mechanism and drug sensitivity,
only MDA-MB-231 cells were selected but not MDA-MB-436 cells, which
was based on the fact that the expression of GBP2 was relatively
lower in both parental and PTX-resistant MDA-MB-231 cells.
Therefore, under the time constraints, it was subsequently chosen
to perform the follow-up experiments with only MDA-MB-231 cells.
There are certain related studies in which investigators made
similar decisions (56-58). However, results should be also
validated in MDA-MB-436 cells in future experiments to increase the
rigor of the results.
In summary, the present study demonstrated that GBP2
is a prognostically relevant marker for TNBC, and it exerts
tumor-suppressive activities such as inhibition of proliferation,
migration and invasion. Notably, GBP2 enhances PTX sensitivity in
TNBC by promoting autophagy. Mechanistically, the positive
regulation of autophagy by GBP2 involves its inhibition in
PI3K/AKT/mTOR signaling pathway and engagement with ATG2. These
findings enable an understanding of the mechanisms underlying the
GBP2-mediated reversal of chemotherapy resistance in TNBC cells and
highlight GBP2 as a potential therapeutic target for overcoming
drug resistance in TNBC.
Supplementary Data
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
WZ and SL conceived the study and designed the
experiments. WZ conducted the experiments and wrote the manuscript.
XT and YP carried out bioinformatics and data analysis. YX, LL, WZ
and SL confirm the authenticity of all the raw data, collaborated
on the figures, edited the manuscript, and contributed to data
interpretation. All authors thoroughly reviewed the final
manuscript. All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
All animal experiments were approved (approval no.
2022-0016) by The Animal Experimentation Ethics Committee of
Chongqing Medical University (Chongqing, China). Approval for the
utilization of clinical samples (approval no. 2020-279) was granted
by The Review Board of the First Affiliated Hospital of Chongqing
Medical University (Chongqing, China). Written informed consent was
obtained from all patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
Funding
The present study was supported by the Key Research and
Development Project of Chongqing's Technology Innovation and
Application Development Special Big Health Field (grant no.
CSTC2021jscx-gksb-N0027), Chongqing Natural Science Foundation
(grant no. CSTB2022NSCQ-MSX0940) and the Chongqing Tongliang
Science and Technology Bureau Foundation (grant no. TL2021-04).
References
|
1
|
International Agency for Research on
Cancer (IARC): Global Cancer Observatory. IARC; Lyon: 2020
|
|
2
|
Smolarz B, Nowak AZ and Romanowicz H:
Breast cancer-epidemiology, classification, pathogenesis and
treatment (review of literature). Cancers (Basel). 14:25692022.
|
|
3
|
da Silva JL, Cardoso Nunes NC, Izetti P,
de Mesquita GG and de Melo AC: Triple negative breast cancer: A
thorough review of biomarkers. Crit Rev Oncol Hematol.
145:1028552020.
|
|
4
|
M Braden A, V Stankowski R, M Engel J and
A Onitilo A: Breast cancer biomarkers: risk assessment, diagnosis,
prognosis, prediction of treatment efficacy and toxicity, and
recurrence. Curr Pharm Des. 20:4879–4898. 2014.
|
|
5
|
Vestal DJ and Jeyaratnam JA: The
guanylate-binding proteins: emerging insights into the biochemical
properties and functions of this family of large interferon-induced
guanosine triphosphatase. J Interferon Cytokine Res. 31:89–97.
2011.
|
|
6
|
Guimarães DP, Oliveira IM, de Moraes E,
Paiva GR, Souza DM, Barnas C, Olmedo DB, Pinto CE, Faria PA, De
Moura Gallo CV, et al: Interferon-inducible guanylate binding
protein (GBP)-2: A novel p53-regulated tumor marker in esophageal
squamous cell carcinomas. Int J Cancer. 124:272–279. 2009.
|
|
7
|
Yu S, Yu X, Sun L, Zheng Y, Chen L, Xu H,
Jin J, Lan Q, Chen CC and Li M: GBP2 enhances glioblastoma invasion
through Stat3/fibronectin pathway. Oncogene. 39:5042–5055.
2020.
|
|
8
|
Ye S, Li S, Qin L, Zheng W, Liu B, Li X,
Ren Z, Zhao H, Hu X, Ye N and Li G: GBP2 promotes clear cell renal
cell carcinoma progression through immune infiltration and
regulation of PD-L1 expression via STAT1 signaling. Oncol Rep.
49:492023.
|
|
9
|
Wang J, Min H, Hu B, Xue X and Liu Y:
Guanylate-binding protein-2 inhibits colorectal cancer cell growth
and increases the sensitivity to paclitaxel of paclitaxel-resistant
colorectal cancer cells by interfering Wnt signaling. J Cell
Biochem. 121:1250–1259. 2020.
|
|
10
|
Zhang S, Chen K, Zhao Z, Zhang X, Xu L,
Liu T and Yu S: Lower expression of GBP2 associated with less
immune cell infiltration and poor prognosis in skin cutaneous
melanoma (SKCM). J Immunother. 45:274–283. 2022.
|
|
11
|
Godoy P, Cadenas C, Hellwig B, Marchan R,
Stewart J, Reif R, Lohr M, Gehrmann M, Rahnenführer J, Schmidt M
and Hengstler JG: Interferon-inducible guanylate binding protein
(GBP2) is associated with better prognosis in breast cancer and
indicates an efficient T cell response. Breast Cancer. 21:491–499.
2014.
|
|
12
|
Nyabuto GO, Wilson JP, Heilman SA, Kalb
RC, Kopacz JP, Abnave AV and Vestal DJ: The large GTPase, GBP-2,
regulates Rho family GTPases to inhibit migration and invadosome
formation in breast cancer cells. Cancers (Basel). 13:56322021.
|
|
13
|
Zhang J, Zhang Y, Wu W, Wang F, Liu X,
Shui G and Nie C: Guanylate-binding protein 2 regulates
Drp1-mediated mitochondrial fission to suppress breast cancer cell
invasion. Cell Death Dis. 8:e31512017.
|
|
14
|
Škubník J, Jurášek M, Ruml T and Rimpelová
S: Mitotic poisons in research and medicine. Molecules.
25:46322020.
|
|
15
|
Alqahtani FY, Aleanizy FS, El Tahir E,
Alkahtani HM and AlQuadeib BT: Paclitaxel. Profiles Drug Subst
Excip Relat Methodol. 44:205–238. 2019.
|
|
16
|
Wang H, Vo T, Hajar A, Li S, Chen X,
Parissenti AM, Brindley DN and Wang Z: Multiple mechanisms
underlying acquired resistance to taxanes in selected
docetaxel-resistant MCF-7 breast cancer cells. BMC Cancer.
14:372014.
|
|
17
|
McGrogan BT, Gilmartin B, Carney DN and
McCann A: Taxanes, microtubules and chemoresistant breast cancer.
Biochim Biophys Acta. 1785:96–132. 2008.
|
|
18
|
Wang J, Yang M, Li Y and Han B: The role
of MicroRNAs in the chemoresistance of breast cancer. Drug Dev Res.
76:368–374. 2015.
|
|
19
|
Ho CJ and Gorski SM: Molecular mechanisms
underlying autophagy-mediated treatment resistance in cancer.
Cancers (Basel). 11:17752019.
|
|
20
|
Hashemi M, Paskeh MDA, Orouei S, Abbasi P,
Khorrami R, Dehghanpour A, Esmaeili N, Ghahremanzade A, Zandieh MA,
Peymani M, et al: Towards dual function of autophagy in breast
cancer: A potent regulator of tumor progression and therapy
response. Biomed Pharmacother. 161:1145462023.
|
|
21
|
Škubník J, Svobodová Pavlíčková V, Ruml T
and Rimpelová S: Autophagy in cancer resistance to paclitaxel:
Development of combination strategies. Biomed Pharmacother.
161:1144582023.
|
|
22
|
Dunn LA, Fury MG, Xiao H, Baxi SS, Sherman
EJ, Korte S, Pfister C, Haque S, Katabi N, Ho AL and Pfister DG: A
phase II study of temsirolimus added to low-dose weekly carboplatin
and paclitaxel for patients with recurrent and/or metastatic (R/M)
head and neck squamous cell carcinoma (HNSCC). Ann Oncol.
28:2533–2538. 2017.
|
|
23
|
Li S and De Souza P: Ras isoprenylation
and pAkt inhibition by zoledronic acid and fluvastatin enhances
paclitaxel activity in T24 bladder cancer cells. Cancers (Basel).
3:662–674. 2011.
|
|
24
|
Hama Y, Ogasawara Y and Noda NN: Autophagy
and cancer: Basic mechanisms and inhibitor development. Cancer Sci.
114:2699–2708. 2023.
|
|
25
|
Osawa T, Kotani T, Kawaoka T, Hirata E,
Suzuki K, Nakatogawa H, Ohsumi Y and Noda NN: Atg2 mediates direct
lipid transfer between membranes for autophagosome formation. Nat
Struct Mol Biol. 26:281–288. 2019.
|
|
26
|
Xiao W, Wang J, Wang X, Cai S, Guo Y, Ye
L, Li D, Hu A, Jin S, Yuan B, et al: Therapeutic targeting of the
USP2-E2F4 axis inhibits autophagic machinery essential for zinc
homeostasis in cancer progression. Autophagy. 18:2615–2635.
2022.
|
|
27
|
Liu N, Luo T, Zhang J, Han LN, Duan WQ, Lu
WX, Qiu H, Lin Y, Wu YM, Zhang H, et al: YF343, a novel histone
deacetylase inhibitor, combined with CQ to inhibit-autophagy,
contributes to increased apoptosis in triple-negative breast
cancer. Curr Med Chem. 30:4605–4621. 2023.
|
|
28
|
Li Q, Ni Y, Zhang L, Jiang R, Xu J, Yang
H, Hu Y, Qiu J, Pu L, Tang J and Wang X: HIF-1α-induced expression
of m6A reader YTHDF1 drives hypoxia-induced autophagy and
malignancy of hepatocellular carcinoma by promoting ATG2A and ATG14
translation. Signal Transduct Target Ther. 6:762021.
|
|
29
|
Niklaus NJ, Tokarchuk I, Zbinden M,
Schläfli AM, Maycotte P and Tschan MP: The multifaceted functions
of autophagy in breast cancer development and treatment. Cells.
10:14472021.
|
|
30
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
|
|
31
|
Dunn KW, Kamocka MM and McDonald JH: A
practical guide to evaluating colocalization in biological
microscopy. Am J Physiol Cell Physiol. 300:C723–C742. 2011.
|
|
32
|
Abd El-Aziz YS, Gillson J, Jansson PJ and
Sahni S: Autophagy: A promising target for triple negative breast
cancers. Pharmacol Res. 175:1060062022.
|
|
33
|
Folkerts H, Hilgendorf S, Vellenga E,
Bremer E and Wiersma VR: The multifaceted role of autophagy in
cancer and the microenvironment. Med Res Rev. 39:517–560. 2019.
|
|
34
|
Chen W, Bai Y, Patel C and Geng F:
Autophagy promotes triple negative breast cancer metastasis via YAP
nuclear localization. Biochem Biophys Res Commun. 520:263–268.
2019.
|
|
35
|
Wang J, Dang MN and Day ES: Inhibition of
Wnt signaling by Frizzled7 antibody-coated nanoshells sensitizes
triple-negative breast cancer cells to the autophagy regulator
chloroquine. Nano Res. 13:1693–1703. 2020.
|
|
36
|
Wang M, Zhang J, Huang Y, Ji S, Shao G,
Feng S, Chen D, Zhao K, Wang Z and Wu A: Cancer-associated
fibroblasts autophagy enhances progression of triple-negative
breast cancer cells. Med Sci Monit. 23:3904–3912. 2017.
|
|
37
|
Li M, Liu J, Li S, Feng Y, Yi F, Wang L,
Wei S and Cao L: Autophagy-related 7 modulates tumor progression in
triple-negative breast cancer. Lab Invest. 99:1266–1274. 2019.
|
|
38
|
Qiao Z, Li X, Kang N, Yang Y, Chen C, Wu
T, Zhao M, Liu Y and Ji X: A novel specific anti-CD73 antibody
inhibits triple-negative breast cancer cell motility by regulating
autophagy. Int J Mol Sci. 20:10572019.
|
|
39
|
Chen Y, Azad MB and Gibson SB: Methods for
detecting autophagy and determining autophagy-induced cell death.
Can J Physiol Pharmacol. 88:285–295. 2010.
|
|
40
|
Chen C, Gao H and Su X: Autophagy-related
signaling pathways are involved in cancer (review). Exp Ther Med.
22:7102021.
|
|
41
|
Ciccarese F, Zulato E and Indraccolo S:
LKB1/AMPK pathway and drug response in cancer: A therapeutic
perspective. Oxid Med Cell Longev. 2019:87308162019.
|
|
42
|
Lu C, Wang W, Jia Y, Liu X, Tong Z and Li
B: Inhibition of AMPK/autophagy potentiates parthenolide-induced
apoptosis in human breast cancer cells. J Cell Biochem.
115:1458–1466. 2014.
|
|
43
|
Zhou YY, Li Y, Jiang WQ and Zhou LF:
MAPK/JNK signaling: A potential autophagy regulation pathway.
Biosci Rep. 35:e001992015.
|
|
44
|
Lorzadeh S, Kohan L, Ghavami S and
Azarpira N: Autophagy and the Wnt signaling pathway: A focus on
Wnt/β-catenin signaling. Biochim Biophys Acta Mol Cell Res.
1868:1189262021.
|
|
45
|
Ravikumar B, Sarkar S, Davies JE, Futter
M, Garcia-Arencibia M, Green-Thompson ZW, Jimenez-Sanchez M,
Korolchuk VI, Lichtenberg M, Luo S, et al: Regulation of mammalian
autophagy in physiology and pathophysiology. Physiol Rev.
90:1383–1435. 2010.
|
|
46
|
Wang Y and Zhang H: Regulation of
autophagy by mTOR signaling pathway. Adv Exp Med Biol. 1206:67–83.
2019.
|
|
47
|
Li L, Tong M, Fu Y, Chen F, Zhang S, Chen
H, Ma X, Li D, Liu X and Zhong Q: Lipids and membrane-associated
proteins in autophagy. Protein Cell. 12:520–544. 2021.
|
|
48
|
Cheng SW, Chen PC, Ger TR, Chiu HW and Lin
YF: GBP5 serves as a potential marker to predict a favorable
response in triple-negative breast cancer patients receiving a
taxane-based chemotherapy. J Pers Med. 11:1972021.
|
|
49
|
Peng Y, Yu H, Jin Y, Qu F, Ren H, Tang Z,
Zhang Y, Qu C, Zong B and Liu S: Construction and validation of an
immune infiltration-related gene signature for the prediction of
prognosis and therapeutic response in breast cancer. Front Immunol.
12:6661372021.
|
|
50
|
Muller R: Systemic toxicity of chloroquine
and hydroxychloroquine: Prevalence, mechanisms, risk factors,
prognostic and screening possibilities. Rheumatol Int.
41:1189–1202. 2021.
|
|
51
|
Wu Y, Yan B, Xu W, Guo L, Wang Z, Li G,
Hou N, Zhang J and Ling R: Compound C enhances the anticancer
effect of aspirin in HER-2-positive breast cancer by regulating
lipid metabolism in an AMPK-independent pathway. Int J Biol Sci.
16:583–597. 2020.
|
|
52
|
Milošević Z, Banković J, Dinić J,
Tsimplouli C, Sereti E, Dragoj M, Paunović V, Milovanović Z,
Stepanović M, Tanić N, et al: Potential of the dual mTOR kinase
inhibitor AZD2014 to overcome paclitaxel resistance in anaplastic
thyroid carcinoma. Cell Oncol (Dordr). 41:409–426. 2018.
|
|
53
|
Park GB, Jeong JY and Kim D: Gliotoxin
enhances autophagic cell death via the DAPK1-TAp63 signaling
pathway in paclitaxel-resistant ovarian cancer cells. Mar Drugs.
17:4122019.
|
|
54
|
Guo Y, Yuan J, Yin S, Wang X, Shuai R and
Kang J: MAP2K6-FP enhances the sensitiveness of paclitaxel for
ovarian cancer via inducing autophagy. Int J Gynecol Cancer.
27:1082–1087. 2017.
|
|
55
|
Song H, Pan B, Yi J and Chen L: Featured
article: Autophagic activation with nimotuzumab enhanced
chemosensitivity and radiosensitivity of esophageal squamous cell
carcinoma. Exp Biol Med (Maywood). 239:529–541. 2014.
|
|
56
|
Lian B, Pei YC, Jiang YZ, Xue MZ, Li DQ,
Li XG, Zheng YZ, Liu XY, Qiao F, Sun WL, et al: Truncated HDAC9
identified by integrated genome-wide screen as the key modulator
for paclitaxel resistance in triple-negative breast cancer.
Theranostics. 10:11092–11109. 2020.
|
|
57
|
Oudin MJ, Barbier L, Schäfer C, Kosciuk T,
Miller MA, Han S, Jonas O, Lauffenburger DA and Gertler FB: MENA
confers resistance to paclitaxel in triple-negative breast cancer.
Mol Cancer Ther. 16:143–155. 2017.
|
|
58
|
Wang Y, Sui Y and Tao Y: Gambogic acid
increases the sensitivity to paclitaxel in drug-resistant
triple-negative breast cancer via the SHH signaling pathway. Mol
Med Rep. 20:4515–4522. 2019.
|