Introduction
Calprotectin is a calcium-binding protein with a
molecular weight of 36 kDa, which primarily originates from
neutrophils and has a regulatory role in inflammatory processes.
Calprotectin is stable and resistant to bacterial degradation in
faeces (1,2). Upon inflammatory damage in the
intestinal mucosa, calprotectin protein levels increase and
calprotectin is released into the intestinal lumen. Therefore,
calprotectin may have potential as a noninvasive biomarker of
intestinal inflammation. Moreover, faecal calprotectin may
represent a beneficial marker for the diagnosis and follow-up of
inflammatory bowel disease (IBD) (3,4).
Previous studies have shown that the concentration of faecal
calprotectin in patients with IBD, including ulcerative colitis
(UC) and Crohn’s disease (CD), is significantly higher than that in
patients with irritable bowel syndrome (IBS) (3,4).
Faecal calprotectin levels have been correlated with histological
disease activity in colonic biopsies from patients with UC and CD.
Therefore, calprotectin is considered to be a marker of intestinal
inflammation (2,5,6).
Normalisation of faecal calprotectin levels has been reported to be
a predictive marker of mucosal healing in patients with IBD;
therefore, faecal calprotectin is an important measure when
treating IBD. Although endoscopy is the gold standard for
diagnosing intestinal inflammation, the procedure is invasive and
unsuitable for frequent use. Faecal calprotectin concentration
represents an increase in neutrophils and correlates strongly with
disease activity (7,8). The present study investigated whether
faecal calprotectin may be used to differentiate between patients
with IBD and those with IBS when monitoring disease activity and
performing follow-ups in patients with IBD.
Patients and methods
Patient samples
In the present study, stool samples were obtained
from 104 patients, including 20 healthy participants, 26 patients
with IBS and 58 patients with IBD, of which 22 were diagnosed with
UC and 36 with CD. Written informed consent was obtained from the
patients or their famililes. Faecal samples (5 g) were collected
from all of the patients over 12 months. Diagnoses of CD and UC
were based on standard criteria (9,10).
In accordance with their medical history, the healthy participants
did not have any type of bowel disease and were required to undergo
endoscopy and routine laboratory blood tests to exclude IBS.
Following the exclusion of organic pathology by performing routine
blood tests and thyroid function tests, stool examination for
bacteria and parasites, ultrasound examination, colonoscopy and
intestinal radiology, IBS was diagnosed according to the Rome III
Diagnostic Criteria (11). Patient
stool samples were used for measuring the calprotectin levels and
blood samples were used for measuring the levels of C-reactive
protein (CRP) as well as the erythrocyte sedimentation rate (ESR).
The study was approved by the Ethics Committee of the Department of
Internal Medicine, China Medical University Hospital, Taichung,
Taiwan (DMR98-013-IRB-251).
Inclusion criteria
All of the patients with IBD were diagnosed based on
standard endoscopic, radiological and histological criteria, which
included; disease duration of less than three months, complete
ileocolonoscopy including biopsies, an age range between 20 and 70
years, and the delivery of faecal samples between one and three
days prior to bowel preparation.
Exclusion criteria
The exclusion criteria for the present study were as
follows: Patients with gastric cancer, incomplete ileocolonoscopy
(ileum not intubated), infectious enterocolitis or acute infection
diseases, pregnant females, individuals with a history of extensive
bowel resection, alcohol abuse, symptoms associated with perianal
penetration, and individuals who regularly consumed aspirin,
antibiotics, cytotoxic drugs and non-steroidal anti-inflammatory
drugs (more than two tablets per week).
Faecal calprotectin
Stool samples were collected and placed in clean
tubes within 72 h and stored at 2–8°C. Faecal calprotectin was
measured using a commercial qualitative point-of-care testing assay
and a Bühlmann Quantum® Blue kit (Bühlmann Laboratories
AG, Schönenbuch, Switzerland). In brief, faecal samples were placed
in an extraction tube and diluted to 1:16 (w/v) with an extraction
buffer (lower range Quantum Blue® LF-CAL) or 1:150 (w/v)
with chase buffer (high range Quantum Blue® LF-CHR) to
provide quantitative results from 30 to 300 μg/g or from 100 to
1,800 μg/g faecal calprotectin, respectively. For quantitative
measurements, unknown samples reading >300 μg/g can be re-tested
in the Bühlmann Quantum Blue® High Range calprotectin
assay (LF-CHR25). The mixture was vortexed for 1 min and
centrifuged at 3000 × g for 5 min. Following a predetermined
dilution, large particles were allowed to settle and the
supernatant was assayed for 12 min. The high faecal calprotectin
concentration was assayed for 15 min using the calibrated Bühlmann
Quantum Blue Reader® (Bühlmann Laboratories AG). The
cut-off level of faecal calprotectin was 50 μg/g.
Statistical analysis
Statistical analyses were performed using the SPSS
software for Windows 16.0 (SPSS, Inc., Chicago, IL, USA).
Biochemical parametric data were presented as the mean ± standard
deviation. Calprotectin values were presented as medians, ranges
and 95% confidence intervals (CIs). Mann-Whitney U tests were
performed to compare faecal calprotectin and CRP levels, and the
ESR between the CD and UC, healthy control, IBS and IBD groups. The
cut-off values for the ESR (normal range, >9 mm/h) and CRP
(upper limit of normal, <0.4 mg/dl) were determined as routine
laboratory values. For the clinical section of the present study,
the cut-off point was 50 μg/g, which was recommended as positive
for gastrointestinal inflammation. Kruskal-Wallis tests were used
to analyse faecal calprotectin levels within the groups, whereas
Dunn’s multiple comparison tests were used to compare differences
in faecal calprotectin levels between the groups. Receiver
operating characteristic (ROC) curves were used to assess faecal
calprotectin as previously described by Henderson (12). All significant values were
two-sided and P<0.05 was considered to indicate a statistically
significant difference.
Results
CRP and faecal calprotectin levels, and
ESR in patients with CD and UC
Table I shows
clinical and biochemical data of the patients with CD (n=36) and UC
(n=22) and the healthy control subjects (n=20). The mean CRP
(1.045±1.333mg/dl; range, 0.017–4.900), ESR (21.94±25.32 mm/h;
range, 4–104) and faecal calprotectin (815.4±720.7 μg/g; range,
30–1,800) values were observed to be significantly higher in the
patients with CD compared with the healthy control subjects (all
P<0.0001). Similar to the patients with CD, the mean CRP
(0.536±0.882 mg/dl; range, 0.014–3.330), ESR (11.9±9.05 mm/h;
range, 2–42) and faecal calprotectin (497.4±584.8 μg/g; range,
35–1810) values in the patients with UC were found to be
significantly higher compared with the healthy control subjects
(all P<0.0001).
 | Table IClinical and biochemical data of
patients with CD and UC, and the control subjects. |
Table I
Clinical and biochemical data of
patients with CD and UC, and the control subjects.
| CD (n=36) | UC (n=22) | Control (n=20) |
|---|
|
|
|
|
|---|
| Parameter | Mean ± SD | Range | Mean ± SD | Range | Mean ± SD | Range |
|---|
| CRP (mg/dl) | 1.045±1.333a | 0.017–4.900 | 0.536±0.882a | 0.014–3.330 | 0.064±0.062 | 0.007–0.191 |
| ESR (mm/h) | 21.94±25.32a | 4–104 | 11.9±9.05a | 2.0–42.0 | 4.45±2.16 | 1–8 |
| Faecal calprotectin
(μg/g) | 815.4±720.7a | 30–1,800 | 497.4±584.8a | 35–1,810 | 36.85±6.80 | 30–51 |
CRP, faecal calprotectin and ESR in
patients with IBD and IBS
Table II shows
clinical and biochemical data of the patients with IBD (n=58) and
IBS (n=26). The mean CRP (0.851±1.200 mg/dl; range, 0.014–4.900)
and faecal calprotectin (694.8±685.0 μg/g; range, 30–622) values
were observed to be significantly higher in the patients with IBD
compared with the patients with IBS (CRP: 0.162±0.229 mg/dl; range,
0.010–1.040; faecal calprotectin, 85.77±136.1 μg/g; range, 30–622;
P<0.0001). The difference in the mean ESR between the patients
with IBD (18.14±21.16 mm/h; range, 2–104) and those with IBS
(9.11±4.02 mm/h; range, 5–22) was not identified to be significant
(P=0.220).
 | Table IIClinical and biochemical data of the
IBD and IBS patients. |
Table II
Clinical and biochemical data of the
IBD and IBS patients.
| IBD (n=58) | IBS (n=26) | |
|---|
|
|
| |
|---|
| Parameter | Mean ± SD | Range | Mean ± SD | Range | P-value |
|---|
| CRP (mg/dl) | 0.851±1.200 | 0.014–4.900 | 0.16±0.23 | 0.010–1.040 | <0.0001 |
| ESR (mm/h) | 18.14±21.16 | 2–104 | 9.11±4.02 | 5–22 | 0.220 |
| Faecal calprotectin
(μg/g) | 694.8±685.0 | 30–1810 | 85.8±136.1 | 30–622 | <0.0001 |
ROC curve analysis of faecal
calprotectin, CRP and ESR in patients with IBD
The area under the ROC curve (AUC) of faecal
calprotectin (AUC, 0.931±0.029; 95% CI, 0.874–0.987) was
significantly higher than that of CRP (AUC, 0.865±0.041; 95% CI,
0.785–0.946) and the ESR (AUC, 0.869±0.042; 95% CI, 0.786–0.952) in
patients with IBD (Fig. 1). The
ROC curve indicated that a faecal calprotectin level of 48.5 μg/g
was the optimal cut-off value in the group of patients with IBD,
with a sensitivity of 90%, a specificity of 95%, a positive (P)
predictive value (PV) of 94% and a negative (N) PV of 89%. In the
patients with IBD, the sensitivity, specificity, PPV, and NPV were
62, 95, 92 and 71% for CRP, and 86, 75, 77 and 84% for ESR,
respectively.
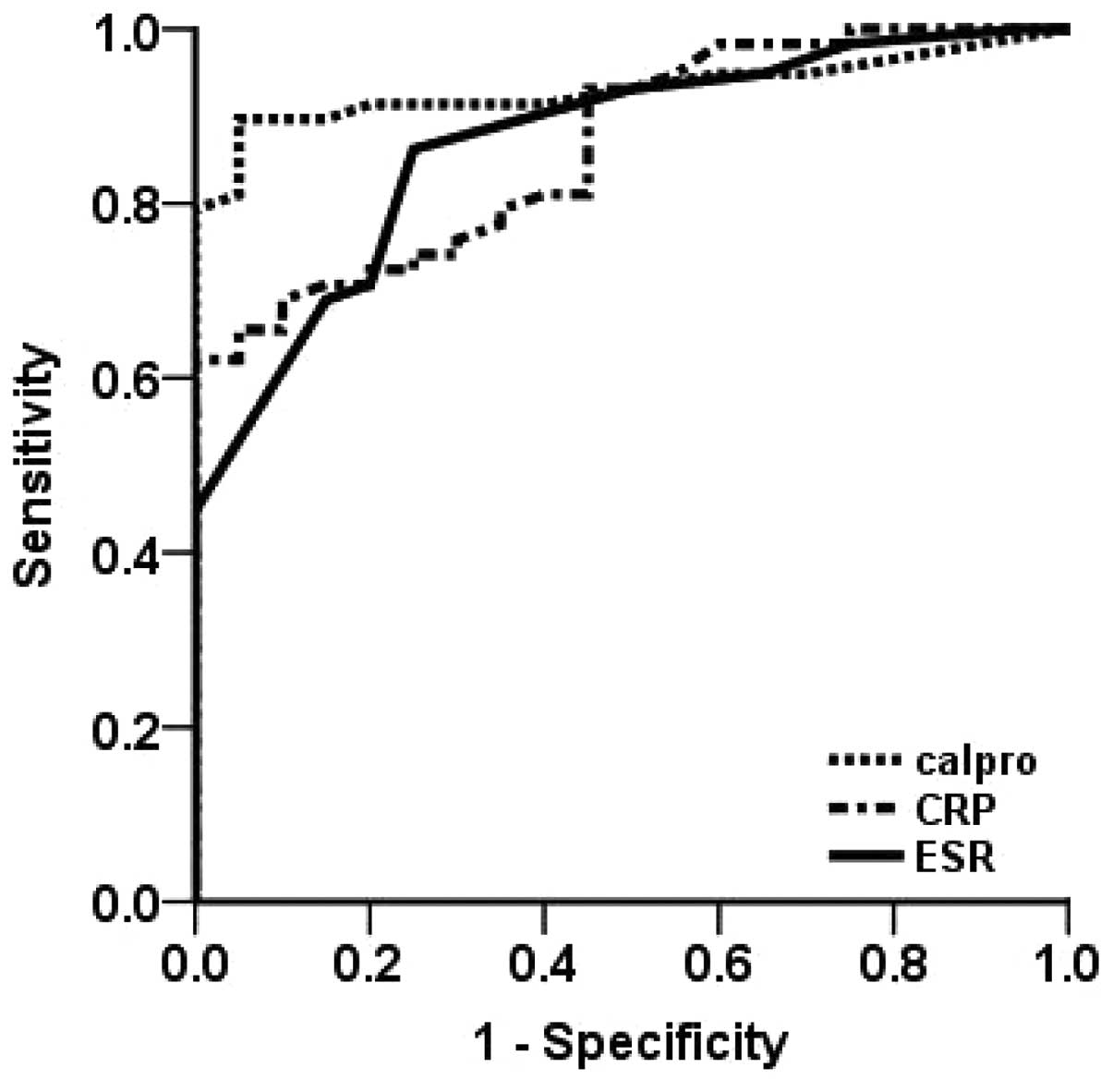 | Figure 1ROC curves of faecal calprotectin, CRP
and the ESR in all patients with IBD, including those with Crohn’s
disease and ulcerative colitis. The ROC curve indicates that a
faecal calprotectin level of 48.5 μg/g is the optimal cut-off
value. The AUC of faecal calprotectin in the patients with IBD
(AUC, 0.931±0.029; 95% CI, 0.874–0.987) was significantly higher
than that of CRP (AUC, 0.865±0.041; 95% CI, 0.785–0.946) and the
ESR (AUC, 0.869±0.042; 95% CI, 0.786–0.952). ROC, receiver operator
characteristic; CRP, C-reactive protein; ESR, erythrocyte
sedimentation rate; calpro, calpronectin; IBD, inflammatory bowel
disease; AUC, area under the ROC curve. |
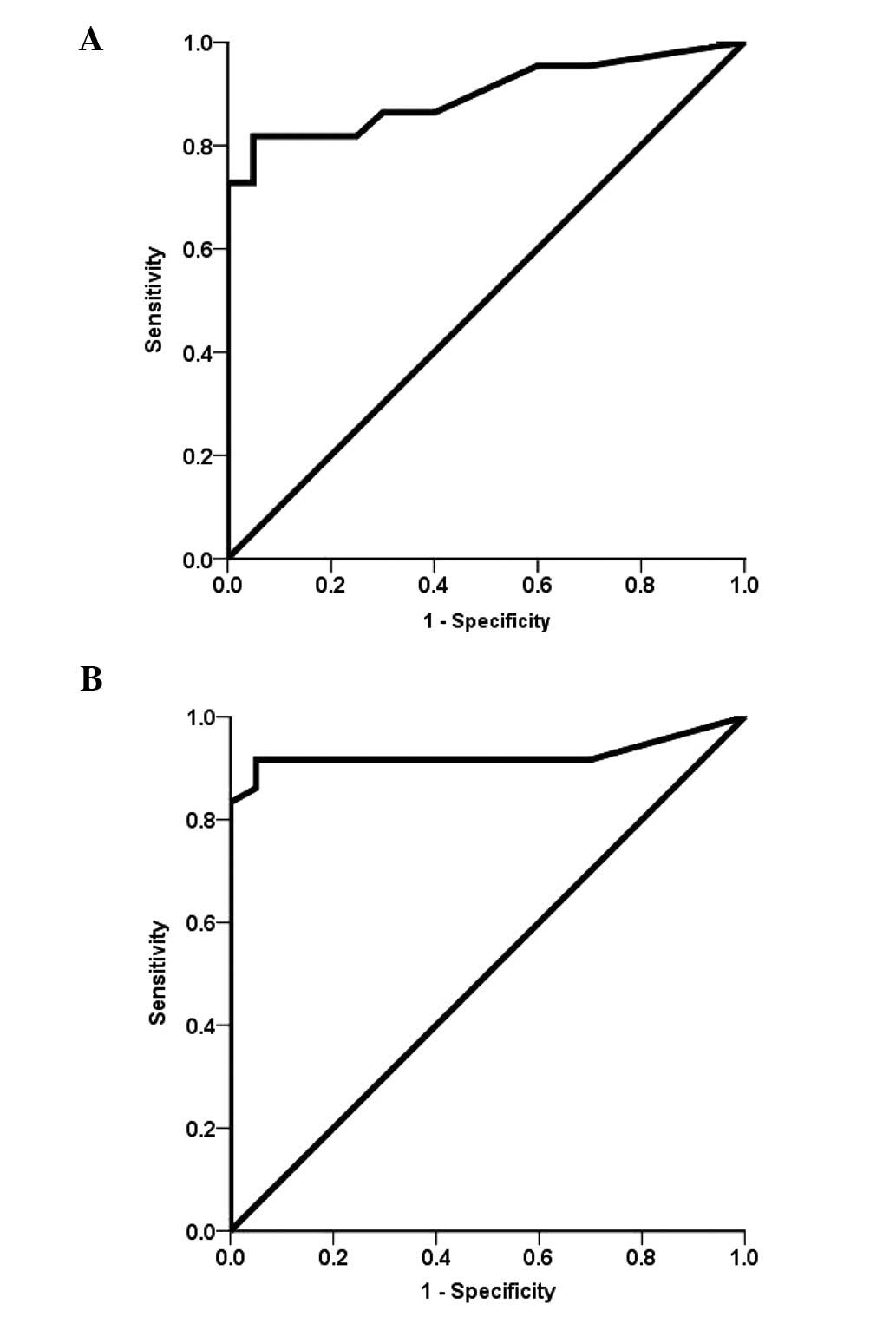
ROC curve analysis of faecal calprotectin
in patients with CD and UC
The median calprotectin concentration was 595 μg/g
(95% CI, 571–1059; range, 30–1,800 μg/g) in the patients with CD
and 219 μg/g (95% CI, 238–756; range, 35–1810 μg/g) in the patients
with UC. The AUC was significantly higher in the patients with UC
(0.939; 95% CI, 0.869–1.01; Fig.
2A) than that in the patients with CD (0.925; 95% CI,
0.84–1.00; Fig. 2B). The
sensitivity, specificity, PPV and NPV were 86, 95, 94 and 87% in
the patients with UC, and 91, 95, 94 and 91% in those with CD,
respectively.
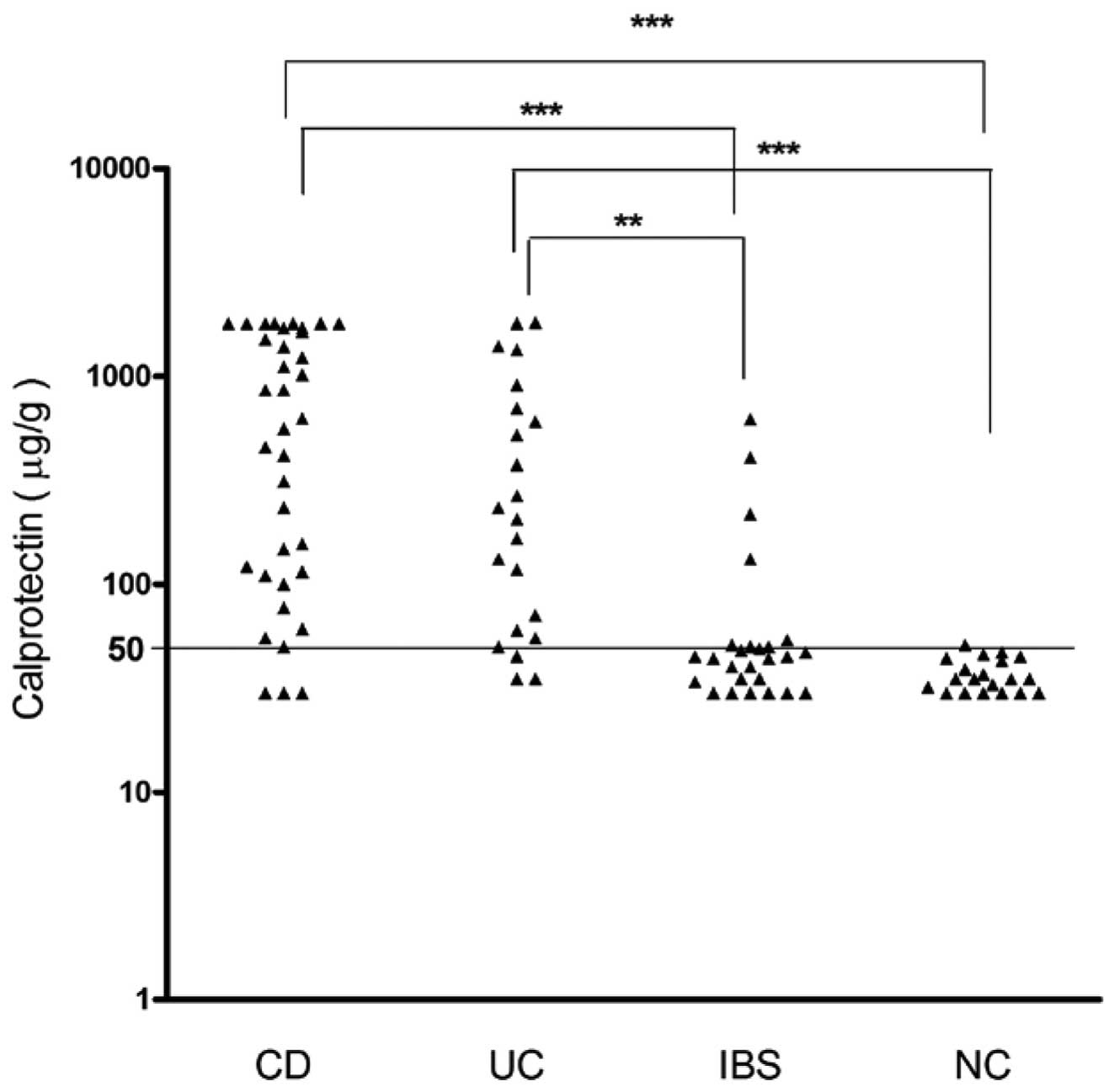
Faecal calprotectin concentration in
patients with CD, UC and IBS and the healthy control subjects
Fig. 3 displays the
faecal calprotectin levels in the examined groups. The median
calprotectin levels were observed to be significantly increased in
the patients with CD (595 μg/g; 95% CI, 571–1,059 μg/g) and UC (219
μg/g; 95% CI, 238–756 μg/g) compared with the healthy control
subjects (35 μg/g; 95% CI, 33–40 μg/g; all P<0.0001).
Furthermore, significant variations were observed among the IBS, CD
and UC groups (P<0.0001 and P<0.001). Normal calprotectin
levels were found in the patients with IBS (44.50 μg/g; 95% CI,
32.6–141.9 μg/g), in addition, no statistical difference was
identified between the IBS patients and the healthy control
subjects. These findings demonstrate that the group of patients
with CD and UC exhibited higher faecal calprotectin levels than
those in the group of patients with IBS. These data indicate that
faecal calprotectin levels may be used to differentiate between
patients with IBD and IBS.
Discussion
In the present study, CRP and faecal calprotectin
levels, and the ESR were found to be significantly higher in
patients with CD and UC compared with those of the healthy control
subjects. Furthermore, it was identified that faecal calprotectin
may be a potential marker of intestinal inflammation to enable
differentiation between IBD and IBS in patients. A significant
difference was observed in CRP and faecal calprotectin levels
between patients with IBS and those with IBD; however, no
significant difference was identified in the ESR between these
patients. In the patients with IBD, the AUC of faecal calprotectin
was significantly higher than that of CRP and ESR; however, the
sensitivity for CRP and ESR was low. It was identified that faecal
calprotectin levels may be used to distinguish between IBD and IBS
according to its high sensitivity, specificity, PPV and high NPV
enabling exclusion of IBD in undiagnosed patients with abdominal
pain or diarrhoea. Furthermore, it was observed that the AUC of
faecal calprotectin concentration was significantly higher in
patients with UC than those with CD. It has previously been
reported that a high concentration of faecal calprotectin is
associated with a two-fold relapse risk in patients with CD and a
14-fold relapse risk in patients with UC, indicating that a high
concentration of faecal calprotectin may be a more accurate
predictive marker of relapse in UC than in CD. Therefore, detecting
faecal calprotectin may facilitate the identification of patients
with UC and CD who possess a high risk of clinical disease relapse
(13,14). Furthermore, a previous study
demonstrated that faecal calprotectin may be used to distinguish
between active and inactive UC (6), and faecal calprotectin and CRP have
also been shown to be more adequate than ESR in detecting
leukocytosis in patients with active UC (15). Faecal calprotectin levels have been
found to be beneficial in predicting clinical relapse in patients
with IBD, including those with UC and CD, in a large long-term
follow-up study and faecal calprotectin determination may be useful
in predicting impending clinical relapse, particularly during the
subsequent three months (16). The
effectiveness of faecal calprotectin measurements in screening for
spontaneous bacterial peritonitis and hepatic encephalopathy (HE)
in cirrhosis has also been reported to facilitate the grading of
HE-severity (17). In addition, a
previous study reported that measuring faecal calprotectin levels
was a useful screening tool for identifying patients who were
likely to require an endoscopy for suspected IBD. The sensitivity
and specificity of faecal calprotectin testing was found to be
greater in adults than in children (sensitivity, 0.93 vs. 0.92 and
specificity, 0.96 vs. 0.76). Thus, measuring faecal calprotectin
levels may provide the capacity to distinguish between IBD and IBS
(18) and faecal calprotectin has
been reported to be an accurate marker of IBD in children and
adults (19).
In conclusion, faecal calprotectin was observed to
be a more direct and useful biomarker than CRP or ESR for
diagnosing intestinal inflammation in patients with IBD. Faecal
calprotectin may facilitate the diagnosis of patients with UC and
CD who are at a high risk of clinical relapse. Furthermore, faecal
calprotectin may potentially be used to distinguish between IBD and
IBS.
References
|
1
|
Walkiewicz D, Werlin SL, Fish D, et al:
Fecal calprotectin is useful in predicting disease relapse in
pediatric inflammatory bowel disease. Inflamm Bowel Dis.
14:669–673. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Paduchova Z and Durackova Z: Fecal
calprotectin as a promising marker of inflammatory diseases.
Bratisl Lek Listy. 110:598–602. 2009.PubMed/NCBI
|
|
3
|
Jahnsen J, Røseth AG and Aadland E:
Measurement of calprotectin in faeces. Tidsskr Nor Laegeforen.
129:743–745. 2009.(In Norwegian).
|
|
4
|
Costa F, Mumolo MG, Bellini M, et al: Role
of faecal calprotectin as non-invasive marker of intestinal
inflammation. Dig Liver Dis. 35:642–647. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tibble JA, Sigthorsson G, Bridger S, et
al: Surrogate markers of intestinal inflammation are predictive of
relapse in patients with inflammatory bowel disease.
Gastroenterology. 119:15–22. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Önal IK, Beyazit Y, Şener B, et al: The
value of fecal calprotectin as a marker of intestinal inflammation
in patients with ulcerative colitis. Turk J Gastroenterol.
23:509–514. 2012.PubMed/NCBI
|
|
7
|
Røseth AG, Aadland E and Grzyb K:
Normalization of faecal calprotectin: a predictor of mucosal
healing in patients with inflammatory bowel disease. Scand J
Gastroenterol. 39:1017–1020. 2004.PubMed/NCBI
|
|
8
|
Sherwood RA: Faecal markers of
gastrointestinal inflammation. J Clin Pathol. 65:981–985. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Malchow H, Ewe K, Brandes JW, et al:
European Cooperative Crohn’s Disease Study (ECCDS): results of drug
treatment. Gastroenterology. 86:249–266. 1984.
|
|
10
|
Bitton A, Peppercorn MA, Antonioli DA, et
al: Clinical, biological, and histologic parameters as predictors
of relapse in ulcerative colitis. Gastroenterology. 120:13–20.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Drossman DA: The functional
gastrointestinal disorders and the Rome III process.
Gastroenterology. 130:1377–1390. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Henderson AR: Assessing test accuracy and
its clinical consequences: a primer for receiver operating
characteristic curve analysis. Ann Clin Biochem. 30:521–539. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Costa F, Mumolo MG, Ceccarelli L, et al:
Calprotectin is a stronger predictive marker of relapse in
ulcerative colitis than in Crohn’s disease. Gut. 54:364–368.
2005.PubMed/NCBI
|
|
14
|
D’Incà R, Dal Pont E, Di Leo V, et al: Can
calprotectin predict relapse risk in inflammatory bowel disease? Am
J Gastroenterol. 103:2007–2014. 2008.PubMed/NCBI
|
|
15
|
Hanai H, Takeuchi K, Iida T, et al:
Relationship between fecal calprotectin, intestinal inflammation,
and peripheral blood neutrophils in patients with active ulcerative
colitis. Dig Dis Sci. 49:1438–1443. 2004. View Article : Google Scholar
|
|
16
|
Gisbert JP, Bermejo F, Pérez-Calle JL, et
al: Fecal calprotectin and lactoferrin for the prediction of
inflammatory bowel disease relapse. Inflamm Bowel Dis.
15:1190–1198. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gundling F, Schmidtler F, Hapfelmeier A,
et al: Fecal calprotectin is a useful screening parameter for
hepatic encephalopathy and spontaneous bacterial peritonitis in
cirrhosis. Liver Int. 31:1406–1415. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
van Rheenen PF, Van de Vijver E and Fidler
V: Faecal calprotectin for screening of patients with suspected
inflammatory bowel disease: diagnostic meta-analysis. BMJ.
341:c33692010.PubMed/NCBI
|
|
19
|
Carroccio A, Iacono G, Cottone M, et al:
Diagnostic accuracy of fecal calprotectin assay in distinguishing
organic causes of chronic diarrhea from irritable bowel syndrome: a
prospective study in adults and children. Clin Chem. 49:861–867.
2003. View
Article : Google Scholar
|