Introduction
Nitric oxide (NO) is generated by endothelial nitric
oxide synthase (eNOS) and has diverse biological functions,
including anti-inflammatory effects, inhibition of platelet
accumulation and hypertension, anti-atherosclerosis effects and
attenuation of diabetes and insulin resistance. Therefore, NO is
critical for maintaining cardiovascular homeostasis. Endothelial NO
release is affected by various factors, such as hormones, the
inflammatory state, cytokines and mechanical stress. In addition to
the regulation of eNOS at a substrate level, through competition
between L-arginine and the competitive inhibitor endogenous
asymmetric dimethylarginine, the activity and function of eNOS is
regulated by its cofactor, tetrahydrobiopterin (BH4). Biopterin has
two isoforms, BH4 and its enzyme-recycling oxidized form,
7,8-dihydrobiopterin (BH2). Upon BH4 deficiency or the addition of
BH2, endothelial cells metabolize L-arginine to produce reactive
oxygen species (ROS) through eNOS uncoupling. BH4 supplementation
recovers eNOS activity and increases NO production. Thus, the
concentration of BH4 and the relative concentrations of BH4 and BH2
regulate eNOS bioactivity and endothelial function.
BH4 is derived from synthesis and redox recycling
in vivo. The former occurs through a biosynthetic pathway
involving guanosine triphosphate cyclohydrolase I, which catalyzes
the formation of dihydroneopterin triphosphate from guanosine
triphosphate (GTP), and two further steps catalyzed by
6-pyruvoyltetrahydropterin synthase and sepiapterin reductase. In
addition, BH2, a by-product of the synthesis pathway, can be
converted to BH4 by dihydrofolate reductase (DHFR). The generation
of BH4 through redox recycling is catalyzed by dihydropteridine
reductase (DHPR). Therefore, the generation of BH4 through
synthesis and redox recycling involves the reductases DHFR and
DHPR, respectively, whose activities are dependent on
5-methyltetrahydrofolate (1).
Folate enhances the binding of BH4 to NOS through a
pteridine-binding domain and then 5-methyltetrahydrofolate
facilitates the electron transfer by BH4 from the NOS reductase
domain to the heme. Therefore, folic acid (FA) enhances the
chemical synthesis and stabilization of BH4 (2).
Hyperhomocysteine damages endothelial cells in the
vasculature through increased formation of ROS and inflammation,
thereby increasing the likelihood of cardiac diseases and stroke.
Hyperhomocysteinemia also promotes low-density lipoprotein (LDL)
oxidation and internalization by macrophages, which is the initial
step in atherosclerosis (3–5).
Folate has been identified to reduce the level of homocysteine
(Hcy), but the clinical application of folate is controversial,
with certain studies demonstrating that folate does not affect the
process of atherosclerosis directly (6–7). In
the present study, the protective effect of FA on endothelial cells
was investigated.
Materials and methods
Chemicals
Hcy, BH4 and neopterin standard substances were
purchased from Sigma-Aldrich (St. Louis, MO, USA), as were the cell
culture media. All other chemicals were obtained from the Tianjin
Chemical Raw Materials Company (Tianjin, China). Human umbilical
vein endothelial cells (HUVECs) were purchased from American Type
Culture Collection (Manassas, VA, USA).
Determination of total Hcy (tHcy)
tHcy levels were measured using a fluorescence
polarization immunoassay adapted to the IMx® analyzer
(Abbott Diagnostics, Abbott Park, IL, USA).
Measurement of biopterins and
neopterin
Measurement of BH4 and BH2 was performed using
high-performance liquid chromatography (HPLC) analysis, following
iodine oxidation in acidic or alkaline conditions, as previously
described (8). Briefly, cell
pellets were lysed in cold extract buffer [50 mM Tris-HCl (pH 7.4),
1 mM dithiothreitol and 1 mM EDTA, containing 0.1 mM neopterin as
an internal recovery standard]. Protein concentration was measured
using the Bio-Rad protein assay (Bio-Rad, Hercules, CA, USA).
Proteins were extracted with the addition of 10 ml 1:1 mixture of
1.5 M HClO4 and 2 M H3PO4 to 90-μl
extracts, followed by centrifugation. Subsequent to deproteination,
paired samples were subjected to oxidation with 1% iodine/2%
potassium iodide under acidic and basic conditions. Biopterin
concentrations were determined using HPLC in 5% methanol/95% water
with a Spherisorb® ODS-1 column (Waters Ltd., Elstree,
UK) and fluorescence detection (350 nm excitation and 450 nm
emission). For each assay, a standard curve of fresh BH4 (range,
10−7–10−9 M) was included using the same
extract buffer. BH4 concentration was calculated by subtracting BH2
plus oxidized biopterin from the total biopterins. Neopterin
concentration was measured in the same manner, using a neopterin
standard curve.
Determination of NO production
NO is rapidly converted to nitrite
(NO2−) and nitrate
(NO3−); therefore, the total concentration of
NO2− and NO3− is used
as a quantitative measure of NO production. An NO assay kit was
purchased from Biyuntian Biotech Co., Ltd. (Shanghai, China) and NO
production was calculated in accordance with the manufacturer’s
instructions.
Cell culture
Human umbilical vein endothelial cells (HUVECs) were
cultured in M199 media containing 10% fetal bovine serum (FBS) in
75-cm2 flasks, and were maintained in an incubator at
37°C with 5% CO2. Upon reaching 90% confluence, HUVECs
were passaged using 0.25% trypsin and were synchronized. In brief,
following growth to 70–80% confluence, HUVECs were placed in
reduced serum (0.5% FBS) media for 12 h and, subsequent to
attachment, the media were changed to media without serum.
Following synchronization, HUVECs were randomly divided into normal
(not stimulated by Hcy) and Hcy-stimulated groups, and treated with
various concentrations of FA (0.1, 1, 5 and 10 mol/l). HUVECs were
then lysed on ice and the culture medium, including the lysate, was
collected. HPLC was performed in order to measure the effect of
exogenous FA on HUVEC activity.
Statistical analysis
Experiments were performed at least in triplicate.
Statistical analyses were performed using the SPSS 17.0 statistical
software package (SPSS, Inc., Chicago, IL, USA) for Windows.
Normally distributed data are presented as the mean ± standard
error of the mean, whereas non-normally distributed data are
presented as the median. Baseline comparisons between groups were
performed using one-way analysis of variance for multiple
comparisons, followed by Bonferroni correction. The effect of FA on
NO production and biopterin and tHcy levels was assessed using
Mann-Whitney U, Wilcoxon signed rank and t-tests for unpaired or
paired data, as appropriate. A two-tailed value of P<0.05 was
considered to indicate statistical significance.
Results
Effect of FA on intracellular Hcy
concentration
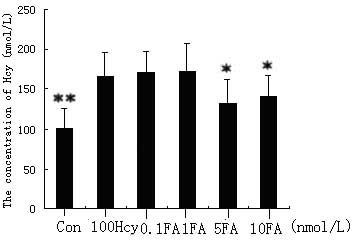
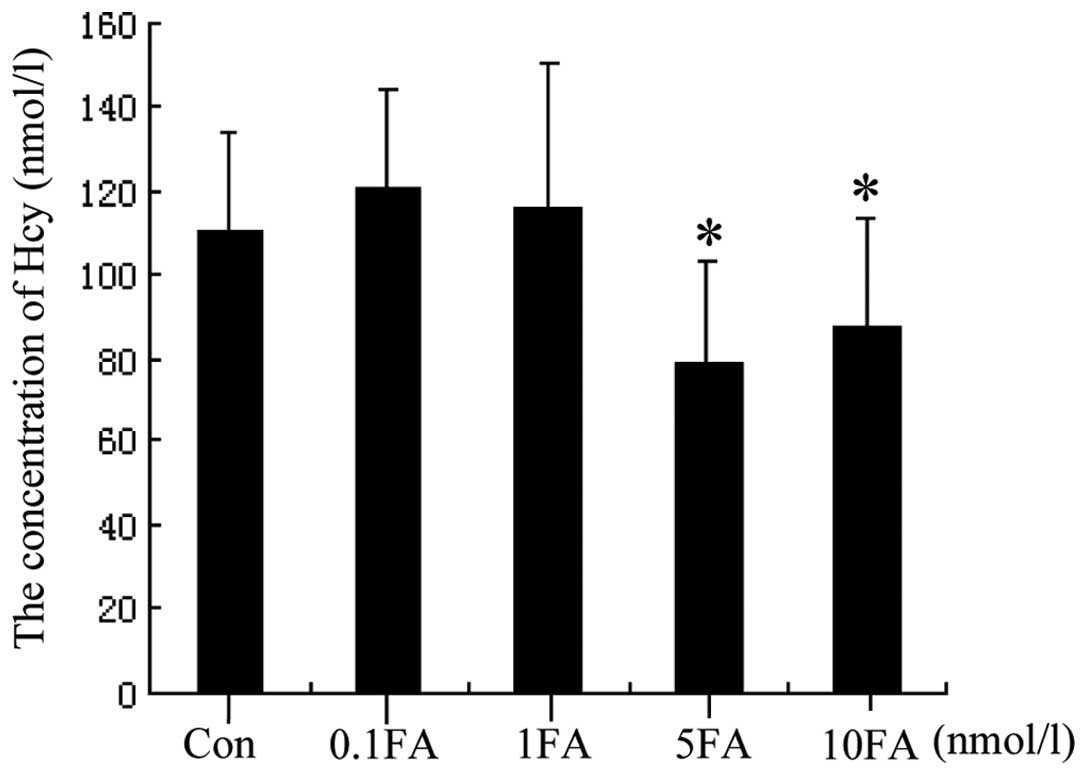
The concentration of Hcy in HUVECs treated with
various doses of FA was investigated in normal and Hcy-stimulated
cells. Cell lysates and culture medium were harvested after 24 h
and were analyzed using HPLC to determine the cellular Hcy
concentration. The levels of Hcy were found to be reduced in the
high-dose FA groups in the cells not stimulated by Hcy; by
contrast, the levels of Hcy in the Hcy-stimulated cells were
increased (P<0.05 Hcy group vs. high-dose FA groups). However,
no significant differences were observed in the 0.1 and 1 nmol/l FA
groups compared with the Hcy group, suggesting that the effect of
FA on Hcy concentration is dose-dependent, with FA only capable of
reducing Hcy concentration at high doses (Figs. 1 and 2).
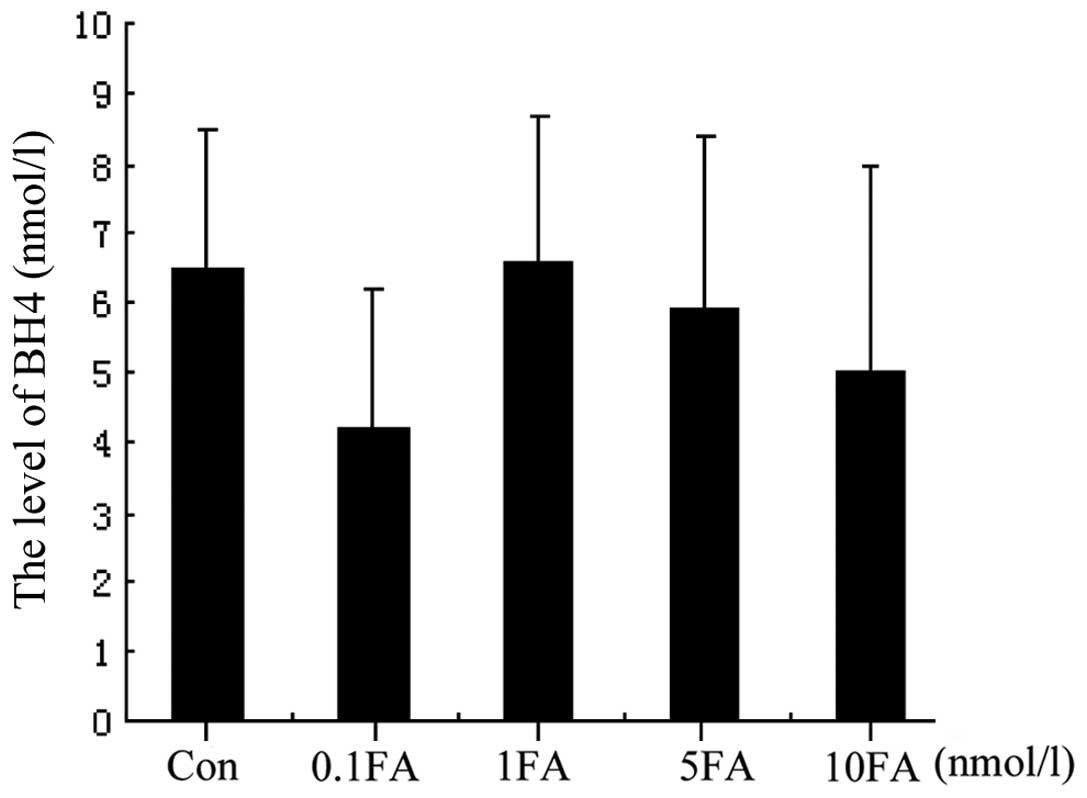
Effect of FA on the intracellular level
of BH4 and neopterin
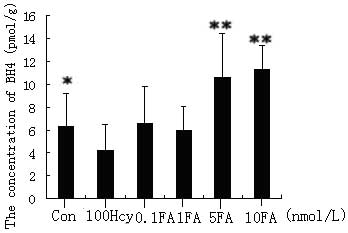
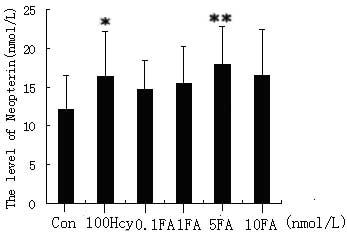
To investigate whether FA could directly protect
vascular endothelial cells, the effects of FA on BH4 and neopterin
levels were investigated in Hcy-stimulated cells. BH4 is one of the
most important cofactors of eNOS and is capable of reducing or
reversing endothelial dysfunction by restoring the production of
NO. HUVEC cultivation with FA for 24 h was observed to increase the
level of BH4 (Fig. 3) and the
BH4:neopterin ratio, particularly in the 5 and 10 nmol/l FA
treatment groups. However, FA was not found to reduce the level of
intracellular neopterin (Fig. 4).
Neopterin is a novel marker of inflammation in humans. Therefore,
the data indicate that although FA appears to increase the activity
of eNOS through increasing levels of the eNOS cofactor BH4, it is
unable to reduce the level of inflammation in HUVECs.
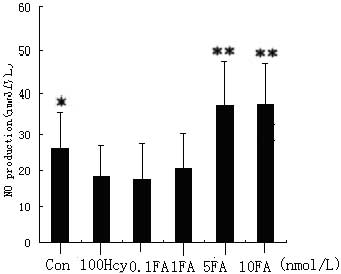
Effect of FA on NO availability in
endothelial cells
To further investigate the enhancement of eNOS
coupling induced by FA, the intracellular concentration of NO was
detected in Hcy-stimulated cells. FA was found to significantly
increase NO levels, particularly in the 5 and 10 nmol/l FA groups
(37.86±12.34 and 38.451±11.23 nmol/l) (Fig. 5), compared with the paired Hcy
group (26.21±9.24) (P<0.001). These observations indicate that
FA may enhance NO-mediated endothelial function through increasing
e-NOS coupling (via increasing levels of the cofactor BH4) and NO
production in endothelial cells.
The effect of FA on normal, unstimulated endothelial
cells was also assessed. The results were inconsistent with those
mentioned above. High doses of FA were found to reduce the levels
of Hcy in normal endothelial cells; however, the levels of BH4 and
NO production were not found to increase in the cells (Fig. 6).
Discussion
Numerous studies have reported that
hyperhomocysteine causes vascular endothelial injury (9–11).
Hyperhomocysteinemia is an independent risk factor for cerebral and
cardiovascular diseases, due to its association with increased
risks of thrombotic and atherosclerotic vascular diseases (12–15).
Folate is a key cofactor for enzymes that metabolize Hcy, and has
been used to reduce Hcy levels in humans (16). Although treatment with high doses
of FA may be beneficial specifically in patients with high tHcy
levels, benefits in other patients have been inconsistent (7). In the present study, high, but not
low, FA doses were found to reduce the levels of Hcy and increase
the concentrations of BH4 and NO, thus exhibiting a beneficial
effect. However, FA was not found to protect normal endothelial
cells through increases in BH4 and NO.
Hcy is a non-protein-forming amino acid that is a
product of S-adenosyl methionine-dependent transmethylation
reactions following the action of S-adenosyl-L-homocysteine
hydrolase. In order to prevent Hcy accumulation and the development
of hyperhomocysteinemia, Hcy is either remethylated back to
methionine or irreversibly catabolized through the
trans-sulfuration pathway to cysteine. It is well established that
hyperhomocysteinemia is an independent risk factor for
atherosclerosis, since it can promote LDL oxidation and
internalization by macrophages, leading to the oxidation of
numerous substances in the body, including BH4, eNOS and NO. Hcy is
capable of directly accelerating NO degradation. Endothelial
dysfunction is a major marker of cardiovascular risk and is
characterized by a reduced production/availability of NO and/or an
imbalance between endothelium-derived relaxation and contracting
factors and oxidants (9,17). In the present study, FA was found
to significantly reduce the levels of Hcy in normal cells and in
cells treated with Hcy.
Neopterin, a by-product of the GTP pathway, is
produced by activated macrophages upon stimulation with
interferon-γ released from T lymphocytes, and is an activation
marker for monocytes/macrophages (18–19).
Inflammatory processes have an important role in the pathogenesis
of atherosclerotic plaque and its complications, and
monocyte/macrophage activation contributes to the processes
associated with atherosclerosis and plaque vulnerability. Increased
levels of inflammatory biomarkers have an important role in the
initiation and progression of atherosclerotic vascular disease
(20). Therefore, neopterin is a
key factor in numerous diseases. However, in the present study, FA
was not observed to reduce the concentration of neopterin in
endothelial cells, suggesting that FA treatment did not change the
inflammatory reaction or reduce the development of atherosclerosis.
Inflammation is an important factor in the development of
atherosclerosis. Without improving the inflammation status in the
body, the diseases are likely to continue. This may explain why FA
does not have long-term effects for cardiovascular diseases and may
restrict its application in the clinic.
The role of BH4 in the control of NOS activity was
initially identified almost 20 years ago. It is now well
established that BH4 is a key factor required for the enzymatic
activity of all three of the NOS isoforms; however, eNOS, which is
important for endothelial cells, has attracted the most attention.
Numerous studies have demonstrated that the chemical inactivation
of BH4 by oxidative stress causes uncoupling of eNOS, which
catalyzes the formation of the superoxide anion and oxidizes NO to
NO2−. By contrast, if BH4 levels increase,
eNOS becomes coupled, which catalyzes the formation of NO (21–22).
In summary, NO bioavailability is a result of NO production and
degradation. NO production is catalyzed by eNOS, which requires BH4
as an essential cofactor. In the present study, BH4 production was
investigated in normal and stimulated endothelial cells. High doses
of FA were found to increase the levels of BH4 in the stimulated
cells; however, the concentration of BH4 remained unchanged in the
normal cells. Therefore, high-dose FA treatment may be beneficial
in patients with high tHcy levels, but benefits for other
individuals are uncertain.
Folate is involved in numerous reactions in the body
and folate deficiency has been implicated in various diseases,
including atherosclerosis, neural tube defects and cancer. Folate
is used widely in the clinic, for example in neuropsychiatric and
neurological disorders, anemia and cardiovascular diseases. An
increasing number of studies have focused the application of folate
in cardiovascular diseases. However, the conclusions are not
consistent. Therefore, the potential for the application of folate
in cardiovascular diseases is yet to be elucidated. In the present
study, high doses of FA were found to protect endothelial cells by
increasing the levels of BH4 and the production of NO. However, FA
was not observed to reduce inflammation, which may explain why
folate is not capable of providing long-term effects for
cardiovascular diseases. Similar results, with regard to BH4 and
NO, were not found in unstimulated endothelial cells.
In conclusion, in the present study, high doses of
FA were observed to be important for injured endothelial cells, but
not for normal, unstimulated endothelial cells. Although FA was
found to have a beneficial effect on injured cells, the underlying
mechanism is yet to be elucidated. Furthermore, the reason why FA
was not found to be beneficial for normal cells remains unclear.
Further investigations are required to elucidate these mechanisms
and provide evidence for the use of folate in the clinic.
References
|
1
|
Hamon CG, Blair JA and Barford PA: The
effect of tetrahydrofolate on tetrahydrobiopterin metabolism. J
Ment Defic Res. 30:179–183. 1986.PubMed/NCBI
|
|
2
|
Moens AL and Kass DA: Tetrahydrobiopterin
and cardiovascular disease. Arterioscler Thromb Vasc Biol.
26:2439–2444. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Coppola A, Davi G, De Stefano V, et al:
Homocysteine, coagulation, platelet function, and thrombosis. Semin
Thromb Hemost. 26:243–254. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bunout D, Garrido A, Suazo M, et al:
Effects of supplementation with folic acid and antioxidant vitamins
on homocysteine levels and LDL oxidation in coronary patients.
Nutrition. 16:107–110. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Voutilainen S, Morrow JD, Roberts LJ II,
et al: Enhanced in vivo lipid peroxidation at elevated plasma total
homocysteine levels. Arterioscler Thromb Vasc Biol. 19:1263–1266.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Woo KS, Chook P, Lolin YI, et al: Folic
acid improves arterial endothelial function in adults with
hyperhomocystinemia. J Am Coll Cardiol. 34:2002–2006. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Antoniades C, Shirodaria C, Warrick N, et
al: 5-methyltetrahydrofolate rapidly improves endothelial function
and decreases superoxide production in human vessels: effects on
vascular tetrahydrobiopterin availability and endothelial nitric
oxide synthase coupling. Circulation. 114:1193–1201. 2006.
View Article : Google Scholar
|
|
8
|
Alp NJ, Mussa S, Khoo J, et al:
Tetrahydrobiopterin-dependent preservation of nitric oxide-mediated
endothelial function in diabetes by targeted transgenic
GTP-cyclohydrolase I overexpression. J Clin Invest. 112:725–735.
2003. View
Article : Google Scholar
|
|
9
|
de Andrade CR, Leite PF, Montezano AC, et
al: Increased endothelin-1 reactivity and endothelial dysfunction
in carotid arteries from rats with hyperhomocysteinemia. Br J
Pharmacol. 157:568–580. 2009.PubMed/NCBI
|
|
10
|
Mahadeo KM, Dhall G, Panigrahy A, et al:
Subacute methotrexate neurotoxicity and cerebral venous sinus
thrombosis in a 12-year-old with acute lymphoblastic leukemia and
methylenetetrahydrofolate reductase (MTHFR) C677T polymorphism:
homocysteine-mediated methotrexate neurotoxicity via direct
endothelial injury. Pediatr Hematol Oncol. 27:46–52. 2010.
|
|
11
|
Papatheodorou L and Weiss N: Vascular
oxidant stress and inflammation in hyperhomocysteinemia. Antioxid
Redox Signal. 9:1941–1958. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Vizzardi E, Bonadei I, Zanini G, et al:
Homocysteine and heart failure: an overview. Recent Pat Cardiovasc
Drug Discov. 4:15–21. 2009. View Article : Google Scholar
|
|
13
|
Guilland JC, Favier A, Potier de Courcy G,
et al: Hyperhomocysteinemia: an independent risk factor or a simple
marker of vascular disease?. 1. Basic data. Pathol Biol (Paris).
51:101–110. 2003.(In French).
|
|
14
|
Cattaneo M: Hyperhomocysteinemia and
thrombosis. Lipids. 36(Suppl): S13–S26. 2001. View Article : Google Scholar
|
|
15
|
Herrmann W: The importance of
hyperhomocysteinemia as a risk factor for diseases: an overview.
Clin Chem Lab Med. 39:666–674. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Krebs HA, Hems R and Tyler B: The
regulation of folate and methionine metabolism. Biochem J.
158:341–353. 1976.PubMed/NCBI
|
|
17
|
Zhang JG, Wang LZ, Han XQ, et al: The
pathogenic mechanism of homocysteine -induced endothelial nitric
oxide synthase dysfunction and the antagonistic effects by folic
acid. Fen Zi Xi Bao Sheng Wu Xue Bao. 40:17–23. 2007.(In
Chinese).
|
|
18
|
Aisaki K, Tsuboi I, Harada T, et al:
Neopterin, inflammation-associated product, prolongs erythropoiesis
suppression in aged SAMP1 mice due to senescent stromal-cell
impairment. Exp Biol Med (Maywood). 237:279–286. 2012. View Article : Google Scholar
|
|
19
|
Yadav AK, Sharma V and Jha V: Association
between serum neopterin and inflammatory activation in chronic
kidney disease. Mediators Inflamm. 2012:4769792012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zamani P, Ganz P, Libby P, et al:
Relationship of antihypertensive treatment to plasma markers of
vascular inflammation and remodeling in the Comparison of
Amlodipine versus Enalapril to Limit Occurrences of Thrombosis
study. Am Heart J. 163:735–740. 2012. View Article : Google Scholar
|
|
21
|
Hyndman ME, Verma S, Rosenfeld RJ, et al:
Interaction of 5-methyltetrahydrofolate and tetrahydrobiopterin on
endothelial function. Am J Physiol Heart Circ Physiol.
282:H2167–H2172. 2002.PubMed/NCBI
|
|
22
|
Ronco AM, Llanos M, Tamayo D and Hirsch S:
5-methyltetrahydrofolic acid stimulates endothelin-1 production in
low density lipoprotein-treated human endothelial cells. Nutr Metab
Cardiovasc Dis. 17:188–194. 2007. View Article : Google Scholar : PubMed/NCBI
|