Introduction
Hepatocellular carcinoma (HCC) is the third most
common cause of cancer-related mortality (1). The predominant risk factors for HCC
development are infection with hepatitis B or C virus, obesity and
excess alcohol intake. The incidence and mortality of HCC is
increasing as a result of the current obesity epidemic and rise in
alcohol consumption (2). However,
only 10–20% of patients with HCC are eligible for surgical
resection, due to poorly preserved liver function, portal vein
invasion or extrahepatic spread. Furthermore, the risk of
recurrence following HCC resection is high (3,4). The
available chemotherapeutic and radiotherapeutic treatment options
for patients with advanced HCC are also extremely limited.
Therefore, it is necessary to develop effective and practical
chemotherapeutic agents with minimal cytotoxicity for use in this
disease.
A number of previous studies have shown that regular
consumption of coffee, vitamin E and fish oil may be associated
with a reduced risk of developing HCC (5–7).
Ecological studies have investigated the association between
dietary fat and certain types of cancer (8,9).
Hursting et al (8)
demonstrated that the intake of saturated or polyunsaturated fats
was associated with incidence of breast and prostate cancer. A
causal relationship was identified between cholesterol intake and
colon cancer (9). Olive oil is the
oil obtained from the fruit of the olive tree (Olea europaea
Sativa) and its consumption is associated with lower overall
mortality patterns, which are observed in Mediterranean populations
(10). The primary component of
olive leaf extract is oleuropein. Andreadou et al reported
that oleuropein is involved in cardiomyocyte metabolism through the
activation of AMP-activated protein kinase α (AMPK) and suppression
of inducible nitric oxide synthases (iNOS) (11). It has recently been reported that
oleuropein may have therapeutic potential in a number of types of
cancer, including breast cancer (12), thyroid cancer (13) and colorectal cancer (14), via inhibition of cell growth
induction of apoptosis. However, to date the effects of oleuropein
on liver cancer remain largely unknown.
In multicellular organisms, apoptosis is a
tightly-regulated multi-step pathway that is responsible for the
process of cell death (15). The
majority of chemotherapies function as tumor suppressors by
targeting this pathway (16).
Serine/threonine protein kinase B (AKT), is important in cell
proliferation, apoptosis and cell survival.
Aberrant and excessive activation of
phosphatidylinositol 3-kinase (PI3K)/AKT signaling is a hallmark of
a number of types of human cancer (17,18).
AKT mediates PI3K-dependent cell survival by phosphorylating a
variety of substrates, such as glycogen synthase kinase-3 β,
6-phosphofructo-2 kinase and apoptosis signal-regulating kinase 1
(19). Changes in the biological
activity of specific downstream substrates of AKT affect whole
signaling cascades, including the activity of transcription factors
involved in cell proliferation, apoptosis, inflammation and
metastasis (20–22).
To understand the possible role of oleuropein in
hepatocellular carcinoma, CCK8 and flow cytometry assays were used
to evaluate HepG2 cell viability and apoptosis following oleuropein
treatment. A luciferase assay was subsequently used to effectively
identify signaling pathways modulated by oleuropein in liver cancer
cells. The results of the present study may identify the
therapeutic potential of oleuropein for the treatment of liver
cancer.
Materials and methods
Cell culture and treatment
The HepG2 and Huh7 human hepatocellular carcinoma
cell lines and the RKO human colorectal cancer cell line were
obtained from the American Type Culture Collection (ATCC, Manassas,
VA, USA), and maintained in McCoy’s 5A medium (Applichem,
Darmstadt, Germany) supplemented with 10% fetal bovine serum (Gibco
Life Technologies, Carlsbad, CA, USA) and 1%
penicillin-streptomycin (Invitrogen Life Technologies, Carlsbad,
CA, USA). Cells were incubated in an atmosphere of 5%
CO2 at 37°C. Cells were treated in serum-free medium
with the indicated concentrations of oleuropein (Sigma-Aldrich, St.
Louis, MO, USA; 12247), dimethyl sulfoxide (Sigma-Aldrich; D2650)
and glutathione (GSH; Sigma-Aldrich; V900456).
Cell viability assay
The effects of oleuropein on cell viability were
determined using a Dojindo Cell Counting kit-8 (CCK-8; Dojindo
Molecular Technologies, Kumamoto, Japan). HepG2, Huh7 and RKO cells
were cultured in 96-well plates at approximately 104
cells per well for 24 h at 37°C. Cells were treated with (0, 20,
40, 60, 80 or 100 μM) oleuropein for 24 h. Subsequently, 10 μl of
CCK-8 solution was added to each well and incubated for 1 h at
37°C. The absorbance at 450 nm was measured using a
spectrophotometer (Shimadzu UV-1800; Shimadzu Scientific
Instruments, Columbia, MD, USA). Independent experiments were
performed three times in triplicate.
Colony formation assay
HepG2 cells (2000 cells/well) were seeded into
six-well plates (in triplicate). Cells were permitted to adhere for
24 h at 37°C, following which the media was changed for media mixed
with (0, 20, 40, 60, 80 or 100 μM) oleuropein. After 48 h, cells
were cultured in normal media for 10 d. In order to visualize
colonies, cells were fixed with 4% paraformaldehyde (Sigma-Aldrich)
for 15 min and stained with crystal violet staining solution (BD
Biosciences, San Jose, CA, USA) for 30 min. Colonies were counted
and presented as the mean number of colonies formed from three
independent experiments.
DNA fragmentation
HepG2 cells were harvested, washed with
phosphate-buffered saline (PBS) and resuspended in 40 μl lysis
buffer, which contained 100 mM Tris (pH 8.0; Amresco LLC, Solon,
OH, USA), 20 mM EDTA (Sinopharm, Shanghai, China), 0.8% sodium
dodecyl sulfate (SDS; W/V; Sinopharm) and 0.5 mg/ml proteinase K
(Merck Millipore, Darmstadt, Germany), at 65°C for 3 h. RNase A
(0.5 mg/ml; Sigma-Aldrich) was added to the cell lysates and
incubated for a further 3 h at 55°C. Genomic DNA was extracted
using phenol-chloroformisoamyl alcohol (25:24:1), separated by 1%
agarose gel electrophoresis and scanned with an Image Analyzer
(Tanon-2500; Tanon Science and Technology Co., Ltd., Shanghai,
China).
Detection of cell apoptosis by flow
cytometry
Detection of apoptosis induced by oleuropein was
performed using Annexin V and Propidium Iodide (PI) staining,
according to the manufacturer’s instructions (BD, Pharmingen San
Diego, CA, USA). Cells (1×105) were suspended in 100 μl
binding buffer and stained with Annexin V-PE and PI (1 μg/ml).
Following incubation at room temperature, a further 400 μl binding
buffer was added and the apoptotic cells were quantification using
flow cytometry (Becton Dickinson FACsCalibur™, BD Biosciences,
Franklin Lakes, NJ, USA).
ROS measurement
HepG2 cells in a 24-well plate were harvested
following treatment for 24 h. Cells were washed twice with PBS,
labeled with 10 μM 2,7-dichlorodihydrofluorescein diacetate
(Sigma-Aldrich) at 37°C for 30 min in darkness, and then subjected
to flow cytometry for the measurement of reactive oxygen species
(ROS).
Luciferase assay
Signaling pathway arrays were conducted using a
luciferase assay. Reporters, NF-κB-luc, PI3K/AKT-luc and Notch-luc,
were purchased from Qiagen (Qiagen-SABiosciences, Valencia, CA,
USA). HepG2 cells were cotransfected with reporter plasmids and a
control reporter in 24-well plates. After 48 h, the luciferase
activity of firefly and Renilla cells was measured using a dual
Luciferase Reporter Assay system (Promega Corporation, Madison, WI,
USA).
Quantitative polymerase chain reaction
(qPCR)
Total RNA from HepG2 cells, with or without
oleuropein treatment, was isolated using RNAiso Plus (Takara Bio,
Inc., Otsu, Japan) according to the manufacturer’s instructions.
cDNA was synthesized using a PrimeScript™ RT-PCR kit (Takara Bio,
Inc.). qPCR was performed using LightCycler (Roche, Basel,
Switzerland) according to the manufacturer’s recommendations. The
following primer sequences (Sagon Biotech, Shanghai, China) were
used: Forward: 5′-GAGGATGATTGCCGCCGTGGACA-3′ and reverse:
5′-GGTGGGGGAGGAGGCTTGAGG-3′ for BAX, forward:
5′-ATGTGTGTGGAGAGCGTCAACC-3′ and reverse:
5′-TGAGCAGAGTCTTCAGAGACAGCC-3′ for Bcl-2 and forward:
5′-TGCACCACCAACTGCTTAGC-3′ and reverse: 5′-GCATGGACTGTGGTCATGAG-3′
for GAPDH. All samples were read in triplicate, and values were
normalized to the expression of GAPDH.
Western blot analysis
HepG2 cells, with or without oleuropein treatment,
were lysed in ice-cold radioimmunoprecipitation assay buffer
containing protease inhibitors [20 mmol/l Tris, 150 mmol/l NaCl
(Sinopharm), 1% NP40 (Sigma-Aldrich), 0.5% sodium deoxycholate
(Sigma-Aldrich), 1 mmol/l EDTA, 1 mmol/l phenylmethylsulfonyl
fluoride (Sigma-Aldrich), 0.15 units/ml aprotinin and 10 mmol/l
leupeptin (Roche Diagnostics GmBH, Mannheim, Germany)]. Cells were
lysed for 30 min on ice and lysates were then centrifuged at 11,340
× g for 10 min at 4°C. The protein concentration was determined by
a bicinchoninic acid protein assay kit (Pierce, 23227; Pierce
Biotechnology, Inc., Rockford, IL, USA) according to the
manufacturer’s instructions. Lysates were dissolved in 4X loading
buffer and boiled for 8 min. Samples were separated by 12% SDS-PAGE
and transferred onto a polyvinylidene fluoride membrane (EMD
Millipore, Bedford, MA, USA). Samples were blocked with 5% non-fat
milk in 50 mM Tris-buffered saline (pH 7.4) with 0.1% Tween-20).
Membranes were probed with primary antibodies for 3 h, followed by
a horseradish peroxidase-conjugated IgG (Thermo Fisher Scientific,
Rockford, IL, USA). Bands were revealed using an enhanced
chemiluminescence detection system (Immobilon Western
Chemiluminescent HRP Substrate; EMD Millipore). The following
primary antibodies were used: Rabbit monoclonal anti-poly ADP
ribose polymerase (PARP) antibody (1:1,000; #9532; Cell Signaling
Technology, Inc., Danvers, MA, USA); rabbit polyclonal
anti-caspase-9 antibody (1:1,000; #9504; Cell Signaling Technology,
Inc.); rabbit polyclonal anti-caspase-8 antibody (1:1,000; #4927;
Cell Signaling Technology, Inc.); mouse polyclonal anti-caspase-3
antibody (1:1,000; #9665; Cell Signaling Technology, Inc.); rabbit
polyclonal anti-BAX antibody (1:1,000; #2772; Cell Signaling
Technology, Inc.); rabbit polyclonal anti-Bcl-2 antibody (1:1,000;
#2872; Cell Signaling Technology, Inc.); mouse monoclonal anti-flag
antibody (1:2,000; #3165; Sigma-Aldrich) and rabbit monoclonal
anti-GAPDH antibody (1:5,000; #3683; Cell Signaling Technology,
Inc.).
Statistical analysis
Analysis of variance using SPSS 19.0 (IBM SPSS,
Armonk, NY, USA) or Student’s t-test were employed to
analyze the data, P<0.05 were considered to indicate a
statistically significant difference.
Results
Oleuropein inhibits HepG2 cell growth in
a dose-dependent manner
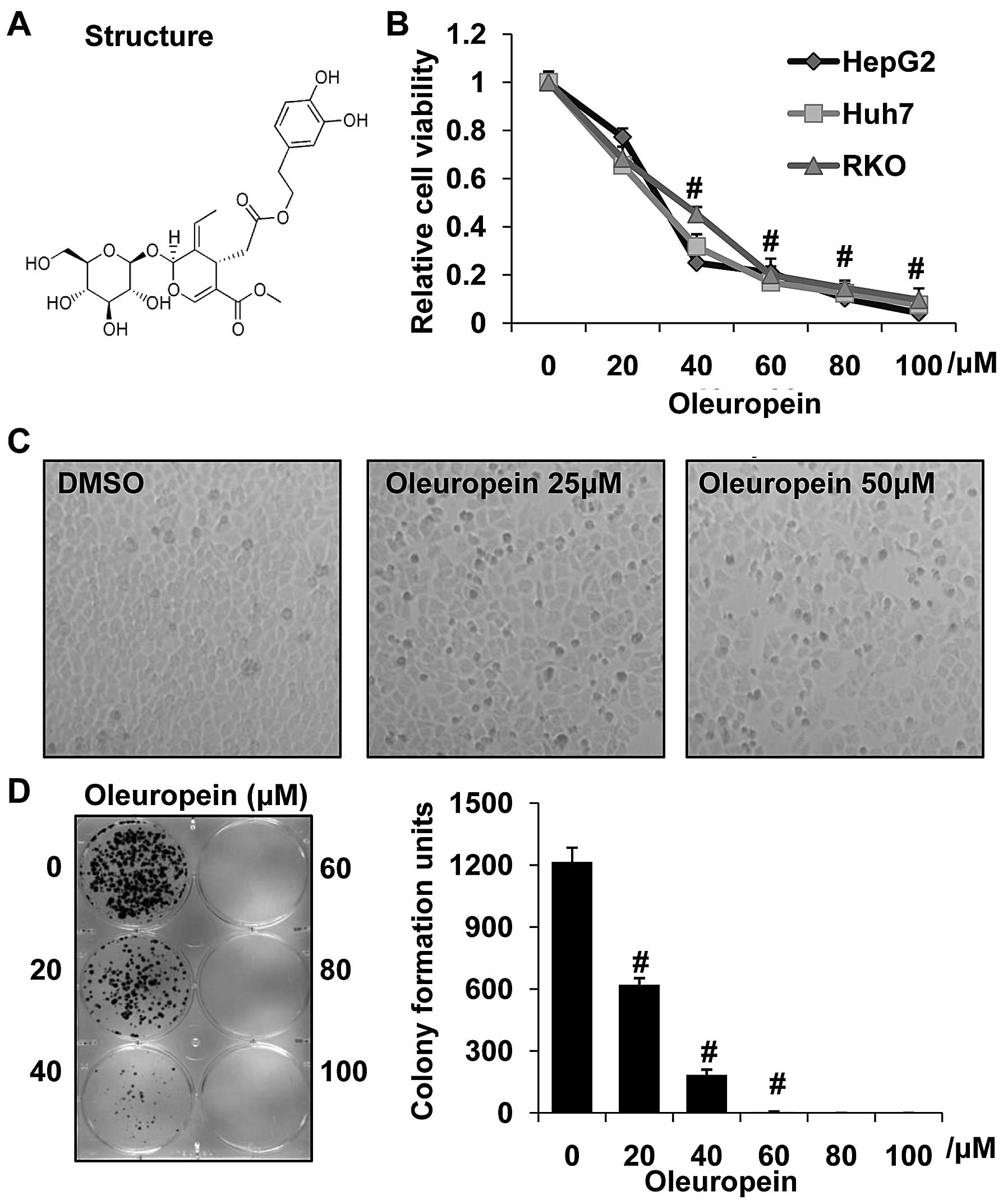
Oleuropein, whose structure is exhibited in Fig. 1A, was the bioactive polyphenol in
olive oil. In order to determine whether oleuropein affects the
tumorigenic properties of human HCC cells, HepG2 and Huh7 human
hepatoma cells were treated with various concentrations of
oleuropein. The human RKO colorectal cancer cell line was also
treated with oleuropein under the same conditions. Using a cell
viability assay, a significant dose-dependent inhibition of cell
growth was observed when the concentration of oleuropein was in the
20–80 μM range (Fig. 1B). In
addition, alterations in the morphology of HepG2 tumor cells,
including cell shrinkage and increased cell death, were observed
following treatment with oleuropein (Fig. 1C). Furthermore, the colony
formation assay demonstrated a marked reduction in the number of
colonies formed in the oleuropein-treated cells, when compared with
the control cells (Fig. 1D). These
findings suggest that oleuropein is involved in the growth of liver
cancer cells.
Oleuropein induces cell apoptosis through
activation of caspases
In order to examine the mechanisms involved in
growth inhibition following treatment with oleuropein, the effect
of oleuropein on the cascade of caspases was investigated. Caspases
are essential initiators or effectors in cell apoptosis. The
results of the present study showed that oleuropein induces
cleavage of caspase-8, caspase-9, caspase-3 and PARP, which is a
nuclear enzyme with a wide range of functions, including the
regulation of DNA repair, cell differentiation and gene expression
(Fig. 2A). To further confirm the
involvement of apoptotic pathways in cell death due to oleuropein,
a DNA fragmentation assay was conducted. A higher level of DNA
fragmentation was observed in oleuropein-treated cells compared
with control cells (Fig. 2B).
Furthermore, cell apoptosis was assessed by Annexin V-FITC/PI
staining in HepG2 cells treated with 30 μM oleuropein. Following 48
h of treatment, a significant percentage of HepG2 cells were
observed to be undergoing apoptosis (Fig. 2C). T he Bcl-2 family of
mitochondria proteins are involved in the regulation of apoptosis
(23). In order to determine
whether Bcl-2 family members are involved in oleuropein-induced
apoptosis, the expression of BAX and Bcl-2 was measured. The
results indicated that oleuropein treatment leads to the
upregulation of the expression of BAX mRNA and protein and the
downregulation of that of Bcl-2 (Fig.
2D and E). This suggests that oleuropein induces HCC cell
apoptosis and that this progress is mediated via the activation of
caspases, in addition to the regulation of the expression of
mitochondrial proteins.
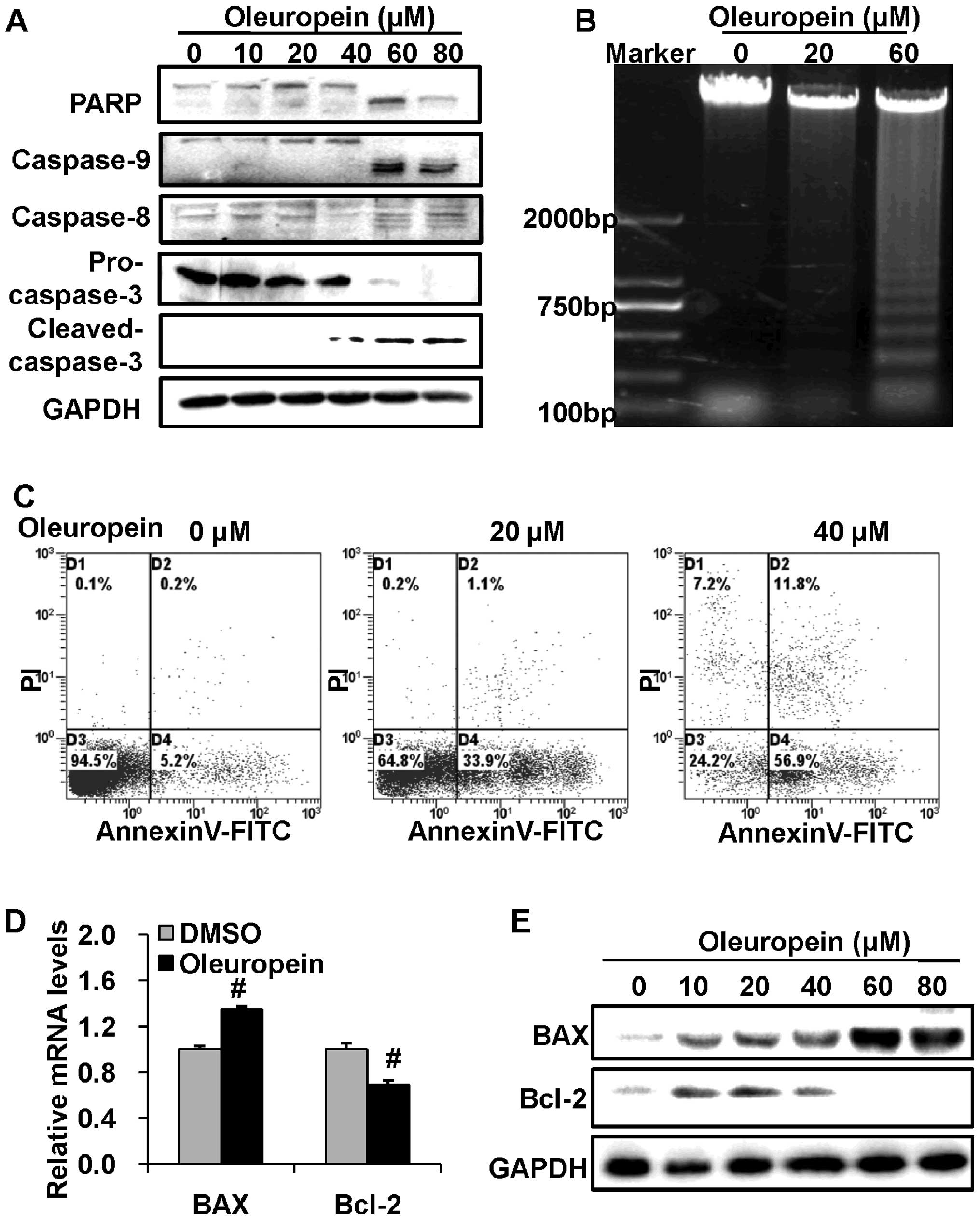 | Figure 2Cell apoptosis was induced by
oleuropein. (A) Equal amounts of cell lysates was subjected to
western blot analysis in order to detect PARP, Caspase-9, Caspase-8
and Caspase-3. (B) DNA fragmentation assay and (C) flow cytometry
analysis confirmed that apoptosis was induced by oleuropein. (D)
Oleuropein promoted the expression of BAX and reduced the
expression of Bcl-2, as shown by quantitative polymerase chain
reaction. #P<0.05 compared with the control group
(DMSO treatment). (E) Western blot analysis of the expression of
BAX and Bcl-2 proteins in HepG2 cells at 0, 10, 20, 40, 60 and 80
μM oleuropein. GAPDH served as an internal control. Data are
presented as the mean ± standard deviation. PARP, poly ADP ribose
polymerase; DMSO, dimethyl sulfoxide; FITC, fluorescein
isothiocyanate. |
Oleuropein induces apoptosis via
suppression of the PI3K/AKT signaling pathway
In order to explore the primary regulators involved
in oleuropein-induced apoptosis, a pathway array was performed
using a luciferase assay. A significant reduction in the activity
of (PI3K/AKT; Fig. 3A) was
observed. AKT is involved in cell proliferation, apoptosis and cell
survival. The activation of AKT by phosphorylation is associated
with the protection of cells from apoptosis (24). The level of the phosphorylated AKT
(S473) and total AKT proteins were analyzed. In accordance with the
results from the luciferase assay, a high concentration of
oleuropein (60 μM) reduced the level of phosphorylated
active AKT, while the level of total AKT remained unchanged with
the various concentrations used (Fig.
3B).
 | Figure 3PI3K/AKT signaling pathway was
involved in oleuropein-induced apoptosis. (A) Luciferase assay of
the activity of AKT, Notch and nfκb, with or without 50 μM
oleuropein treatment. #P<0.05 compared with the
control group (DMSO treatment) (B) Western blot analysis of the
levels of pAKT and AKT at 0, 10, 20, 40, 60 and 80 μM
oleuropein. (C) Western blot analysis confirmed overexpression of
flag-AKT in HepG2 cells. HepG2 cells were transfected with 2
μg pcDNA5-flag-AKT or pcDNA5 empty vector. (D) AKT
overexpression decreased the cleavage of PARP and caspase-3, as
shown by western blot analysis. Cells were transfected with 2
μg pcDNA5-flag-AKT or pcDNA5 empty vector for 24 h and
treated with 50 μM oleuropein. (E) Western blot analysis of
the levels of the pAKT, AKT, PARP and caspase-3 proteins in HepG2
cells pretreated with 50 μM LY294002 for 2 h and followed by
50 μM oleuropein. GAPDH served as an internal control. Data
are presented as the mean ± standard deviation. PI3K,
phosphatidylinositol 3-kinase; AKT, protein kinase B; nfκb, nuclear factor-KB; pAKT,
phosphorylated AKT; PARP, poly ADP ribose polymerase. |
To examine the role of AKT in oleuropein-induced
apoptosis, a rescue experiment was conducted, in which HepG2 cells
were transfected with pcDNA5-flag-AKT or pcDNA5-control constructs
prior to treatment with oleuropein. The results indicated that AKT
overexpression (Fig. 3C) causes
less cleavage of PARP and caspase-3 compared with intrinsic AKT
(Fig. 3D). LY294002, a specific
PI3K inhibitor (25), led to
increased cleavage of PARP and caspase-3 when used together with
oleuropein (Fig. 3E). These
results suggest that the PI3K/AKT signaling pathway contributes to
oleuropein-induced apoptosis in HCC cells.
Oleuropein promotes the generation of
ROS
It has been reported that AKT activation induces the
production of ROS. Elevated levels of intracellular ROS contribute
to tumorigenesis by activation of signaling pathways and by
increasing mutation rates (26,27).
In order to determine whether the dose-dependent inhibition of AKT
activation decreases the accumulation of intracellular ROS, HepG2
cells were treated with 50 μM oleuropein for 48 h and the
level of ROS was measured by FACS analysis. ROS accumulation was
observed following 24 h of oleuropein treatment in HepG2 cells, as
shown in Fig. 4A. We hypothesized
that ROS generation resulted in PI3K/AKT inactivation. ROS blockers
were then used to investigate this hypothesis. HepG2 cells were
pretreated with 20 mM GSH for 2 h, followed by 50 μM
oleuropein treatment for an additional 24 h. A cell viability assay
demonstrated that GSH pretreatment promoted cell survival (Fig. 4B). The results of the western blot
analysis showed that the expression of phosphorylated AKT was
increased following the removal of ROS by pretreatment with GSH
(Fig. 4C). Pretreatment with GSH
consistently inhibited the oleuropein-induced cleavage of PARP and
caspase-3 (Fig. 4D). These results
indicate that oleuropein-induced ROS generation leads to PI3K/AKT
inactivation and is thus has an important function in the apoptotic
pathway.
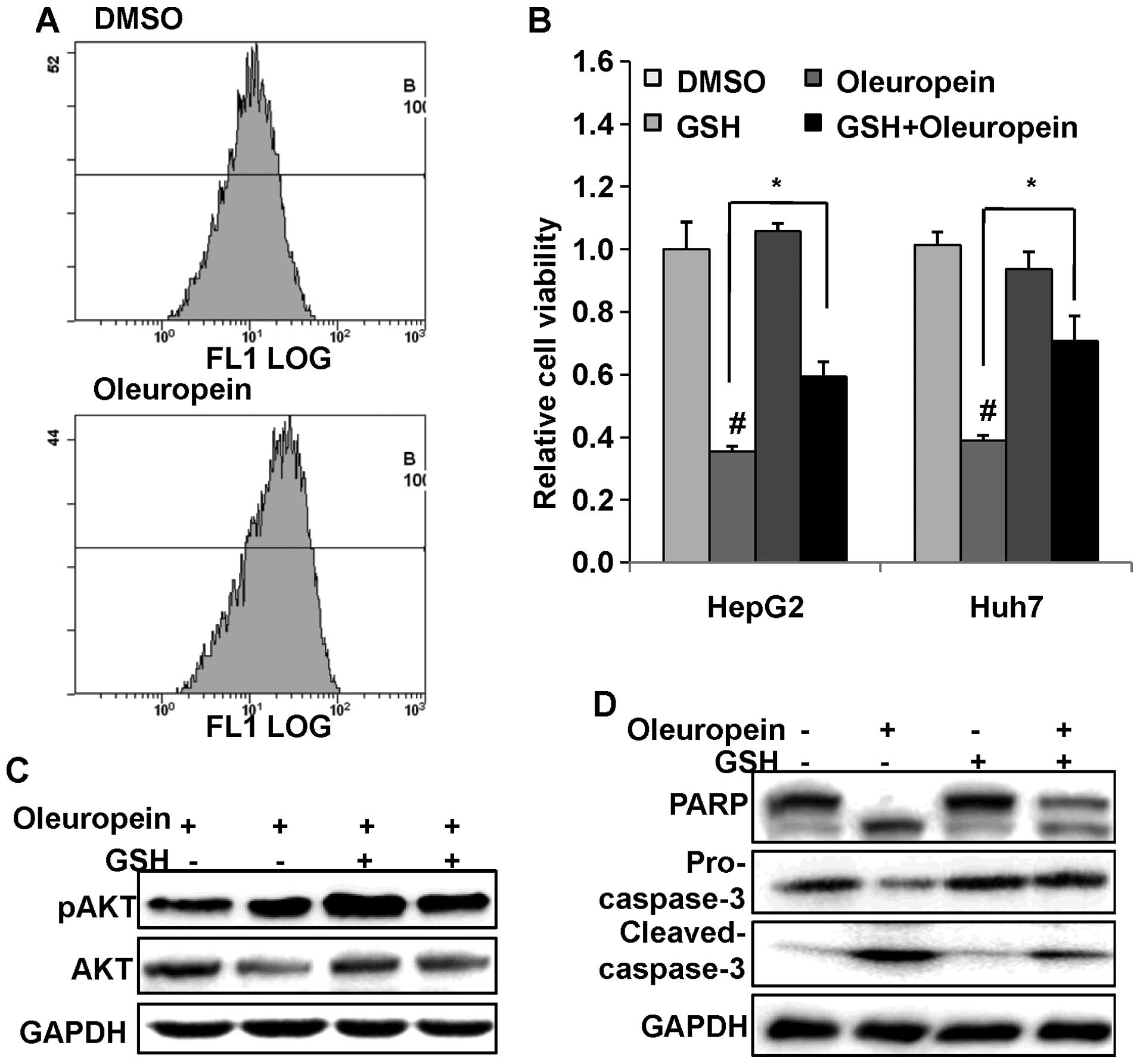 | Figure 4ROS generation was induced by
oleuropein in HepG2 cells. (A) HepG2 cells treated with 50
μM oleuropein or DMSO were subjected to flow cytometry in
order to measure ROS production. (B) Cell viability assay of HepG2
cell treated with 50 μM oleuropein or pretreated with 20 mM
GSH for 2 h, and followed by 50 μM oleuropein.
#P<0.05 compared with the control group (DMSO
treatment). *P<0.05, GST pretreatment followed by
oleuropein treatment compared with oleuropein treatment alone. (C)
Western blot analysis of the expression of pAKT and AKT, and (D)
apoptosis-related proteins, PARP and caspase-3, in HepG2 cells
pretreated with 20 mM GSH for 2 h followed by 50 μM
oleuropein. GAPDH served as an internal control. Data are presented
as the mean ± standard deviation. ROS, reactive oxygen species;
DMSO, dimethyl sulfoxide; GSH, glutathione; AKT, protein kinase B;
pAKT, phosphorylated AKT; PARP, poly ADP ribose polymerase. |
Discussion
A number of previous epidemiological studies have
indicated that there is an association between the consumption of
olive oil, and lower overall mortality patterns and risk of
developing cancer (28–30). Oleuropein is one of the primary
components of olive oil. The present study reports that oleuropein
inhibited proliferation and induced apoptosis in HCC.
Mechanistically, oleuropein-induced apoptosis appears to be
mediated by suppression of the PI3K/AKT pathway and the production
of ROS. It is suggested that oleuropein may be a promising
candidate for HCC treatment.
Recent studies have reported that oleuropein
inhibits cell proliferation and induces the apoptosis of cancer
cell lines through diverse mechanisms. Cardeno et al
(14) reported that oleuropein
limited cell growth and induced apoptosis in HT29 colorectal cancer
cells through a p53-dependent pathway (14). A similar mechanism was also
observed in breast cancer cells (12). Additionally, a recent study on
cardiomyopathy demonstrated that oleuropein has a protective effect
in doxorubicin-induced cardiomyopathy through the activation of
AMPK and suppression of iNOS (11). In the current study, evidence is
presented for a role of oleuropein as an inhibitor of cell growth
and an inducer of apoptosis, which suggests that oleuropein
possesses antitumor activity in HCC.
Apoptosis is a common mechanism by which
chemotherapies induces tumor cell death (31,32).
In mammals, two mechanisms are known to directly initiate apoptosis
(33,34). The extrinsic apoptotic pathway is
activated by ligand binding at the cell surface, while the
intrinsic cell death pathway is triggered by UV-irradiation, growth
factor deprivation or the presence of stress stimuli, for example
oxidative stress, DNA damage or endoplasmic reticulum stress, and
results in the induction of the proapoptotic Bcl-2 family members.
Western blotting experiments in the present study indicated that
the oleuropein-induced apoptosis of HepG2 cells is
caspase-dependent. Furthermore, oleuropein markedly increased BAX
gene expression, while decreasing that of the Bcl-2 gene. The
alteration in the ratio of BAX/Bcl-2 is correlated with apoptosis
through a mitochondrial pathway (35). These results are, in part,
consistent with the mechanism of apoptosis identified in MCF-7
cells treated with oleuropein (12). The two studies characterized an
apoptotic induction function of oleuropein, and the involvement of
BAX and Bcl-2 in the oleuropein-induced cell apoptotic pathway.
Notably, in MCF-7 breast cancer cells, oleuropein regulated the
expression of p53. Further study is required to elucidate the role
of p53 in liver cancer following oleuropein treatment, which may
provide a clinical basis for treatment via a combination of drug
delivery and gene therapy.
Oleuropein induces apoptosis in cancer cell lines
via diverse pathways. In the present study, key mediators that may
be involved in the apoptotic pathway were screened. It was found
that the PI3K/AKT signaling pathway was markedly suppressed in
HepG2 cells treated with oleuropein. Furthermore, oleuropein
altered the activity of the NF-κB and Notch pathways to a certain
extent, which indicated the involvement of these pathways in this
process. The PI3K/AKT pathway is known to be an important survival
pathway that is activated in cancer (36,37).
Deregulation of the PI3K/AKT/mTOR signaling pathways has been
observed in HCC (38). Activation
of the PI3K/AKT pathway is associated with the promotion of
invasion and migration of HCC (39,40).
Based on the results of the current study, oleuropein may repress
the phosphorylation of AKT. Indeed, the rescue experiments using
transfection with a plasmid containing AKT, confirmed the
involvement of AKT in apoptosis in these cells. In addition,
LY294002, a specific PI3K inhibitor, increased cell death when used
together with oleuropein. It may therefore be amenable for use in
HCC prevention. ROS are considered to be downstream of the
activation of AKT (41,42). The present study showed that
oleuropein treatment augmented intracellular ROS levels, and that
removal of ROS increased the level of phosphorylated AKT and
decreased the activation of apoptosis. This suggests that PI3K/AKT
is a critical mediator in the oleuropein-induced apoptosis of HepG2
cells and that ROS are upstream of the AKT pathway. However,
additional mechanisms are likely to be involved and require further
investigation.
In conclusion, the current study demonstrated that
oleuropein induced apoptosis in human hepatocellular carcinoma
cells via suppression of PI3K/AKT, and that ROS accumulation is
involved in the apoptotic pathway. Therefore, oleuropein may have
potential as a novel agent in the prevention and treatment of liver
cancer.
Abbreviations:
|
HCC
|
hepatocellular carcinoma
|
|
ROS
|
reactive oxygen species
|
|
BAX
|
B cell lymphoma 2-associated X
protein
|
|
Bcl-2
|
B cell lymphoma 2
|
|
mRNA
|
messenger RNA
|
|
NF-κB
|
Nuclear Factor-κB
|
|
PI3K
|
phosphatidylinositol-4,5-bisphosphate
3-kinase
|
|
AKT
|
serine/threonine protein kinase B
|
|
mTOR
|
mechanistic target of rapamycin
|
|
iNOS
|
inducible nitric oxide synthases
|
References
|
1
|
Llovet JM, Burroughs A and Bruix J:
Hepatocellular carcinoma. Lancet. 362:1907–1917. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Singh S, Singh PP, Roberts LR and Sanchez
W: Chemopreventive strategies in hepatocellular carcinoma. Nat Rev
Gastroenterol Hepatol. 11:45–54. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cheng JW and Lv Y: New progress of
non-surgical treatments for hepatocellular carcinoma. Med Oncol.
30:3812013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bruix J and Sherman M; American
Association for the Study of Liver Diseases: Management of
hepatocellular carcinoma: an update. Hepatology. 53:1020–1022.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bravi F, Bosetti C, Tavani A, Gallus S and
La Vecchia C: Coffee reduces risk for hepatocellular carcinoma: an
updated meta-analysis. Clin Gastroenterol Hepatol. 11:1143–1421.
2013. View Article : Google Scholar
|
|
6
|
Calvisi DF, Ladu S, Hironaka K, Factor VM
and Thorgeirsson SS: Vitamin E down-modulates iNOS and NADPH
oxidase in c-Myc/TGF-alpha transgenic mouse model of liver cancer.
J Hepatol. 41:815–822. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sawada N, Inoue M, Iwasaki M, et al:
Consumption of n-3 fatty acids and fish reduces risk of
hepatocellular carcinoma. Gastroenterology. 142:1468–1475. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hursting SD, Thornquist M and Henderson
MM: Types of dietary fat and the incidence of cancer at five sites.
Prev Med. 19:242–253. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu K, Stamler J, Moss D, Garside D,
Persky V and Soltero I: Dietary cholesterol, fat, and fibre, and
colon-cancer mortality. An analysis of international data. Lancet.
2:782–785. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ferro-Luzzi A and Sette S: The
Mediterranean Diet: an attempt to define its present and past
composition. Eur J Clin Nutr. 43(Suppl 2): 13–29. 1989.PubMed/NCBI
|
|
11
|
Andreadou I, Mikros E, Ioannidis K, et al:
Oleuropein prevents doxorubicin-induced cardiomyopathy interfering
with signaling molecules and cardiomyocyte metabolism. J Mol Cell
Cardiol. 69:4–16. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hassan ZK, Elamin MH, Omer SA, et al:
Oleuropein induces apoptosis via the p53 pathway in breast cancer
cells. Asian Pac J Cancer Prev. 14:6739–6742. 2013. View Article : Google Scholar
|
|
13
|
Bulotta S, Corradino R, Celano M, et al:
Antioxidant and antigrowth action of peracetylated oleuropein in
thyroid cancer cells. J Mol Endocrinol. 51:181–189. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cardeno A, Sánchez-Hidalgo M, Rosillo MA
and Alarcón de la Lastra C: Oleuropein, a secoiridoid derived from
olive tree, inhibits the proliferation of human colorectal cancer
cell through downregulation of HIF-1α. Nutr Cancer. 65:147–156.
2013. View Article : Google Scholar
|
|
15
|
Cotter TG: Apoptosis and cancer: the
genesis of a research field. Nat Rev Cancer. 9:501–507. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Johnstone RW, Ruefli AA and Lowe SW:
Apoptosis: a link between cancer genetics and chemotherapy. Cell.
108:153–164. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kumar A, Rajendran V, Sethumadhavan R and
Purohit R: AKT kinase pathway: a leading target in cancer research.
Scientific World Journal. 2013:7561342013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pavlidou A and Vlahos NF: Molecular
alterations of PI3K/Akt/mTOR pathway: a therapeutic target in
endometrial cancer. Scientific World Journal. 2014:7097362014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Franke TF, Hornik CP, Segev L, Shostak GA
and Sugimoto C: PI3K/Akt and apoptosis: size matters. Oncogene.
22:8983–8998. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Datta SR, Dudek H, Tao X, et al: Akt
phosphorylation of BAD couples survival signals to the
cell-intrinsic death machinery. Cell. 91:231–241. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hahn-Windgassen A, Nogueira V, Chen CC,
Skeen JE, Sonenberg N and Hay N: Akt activates the mammalian target
of rapamycin by regulating cellular ATP level and AMPK activity. J
Biol Chem. 280:32081–32089. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rokutanda S, Fujita T, Kanatani N, et al:
Akt regulates skeletal development through GSK3, mTOR, and FoxOs.
Dev Biol. 328:78–93. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Adams JM and Cory S: Life-or-death
decisions by the Bcl-2 protein family. Trends Biochem Sci.
26:61–66. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Nicholson KM and Anderson NG: The protein
kinase B/Akt signalling pathway in human malignancy. Cell Signal.
14:381–395. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vlahos CJ, Matter WF, Hui KY and Brown RF:
A specific inhibitor of phosphatidylinositol 3-kinase,
2-(4-morpholinyl)-8-phenyl-4H-1-benzopyran-4-one(LY294002). J Biol
Chem. 269:5241–5248. 1994.PubMed/NCBI
|
|
26
|
Nogueira V, Park Y, Chen CC, et al: Akt
determines replicative senescence and oxidative or oncogenic
premature senescence and sensitizes cells to oxidative apoptosis.
Cancer Cell. 14:458–470. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Robey RB and Hay N: Is Akt the ‘Warburg
kinase’? - Akt-energy metabolism interactions and oncogenesis.
Semin Cancer Biol. 19:25–31. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fini L, Hotchkiss E, Fogliano V, et al:
Chemopreventive properties of pinoresinol-rich olive oil involve a
selective activation of the ATM-p53 cascade in colon cancer cell
lines. Carcinogenesis. 29:139–146. 2008. View Article : Google Scholar
|
|
29
|
La Vecchia C: Association between
Mediterranean dietary patterns and cancer risk. Nutr Rev. 67(Suppl
1): S126–S129. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Menendez JA, Papadimitropoulou A, Vellon L
and Lupu R: A genomic explanation connecting ‘Mediterranean diet’,
olive oil and cancer: oleic acid, the main monounsaturated fatty
acid of olive oil, induces formation of inhibitory ‘PEA3
transcription factor-PEA3 DNA binding site’ complexes at the
Her-2/neu (erbB-2) oncogene promoter in breast, ovarian and stomach
cancer cells. Eur J Cancer. 42:2425–2432. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kaufmann SH and Earnshaw WC: Induction of
apoptosis by cancer chemotherapy. Exp Cell Res. 256:42–49. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wiita AP, Ziv E, Wiita PJ, et al: Global
cellular response to chemotherapy-induced apoptosis. Elife.
29:e012362013.
|
|
33
|
Czerski L and Nunez G: Apoptosome
formation and caspase activation: is it different in the heart? J
Mol Cell Cardiol. 37:643–652. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fulda S and Debatin KM: Apoptosis
signaling in tumor therapy. Ann NY Acad Sci. 1028:150–156. 2004.
View Article : Google Scholar
|
|
35
|
Zhang L, Yu J, Park BH, Kinzler KW and
Vogelstein B: Role of BAX in the apoptotic response to anticancer
agents. Science. 290:989–992. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fresno Vara JA, Casado E, de Castro J,
Cejas P, Belda-Iniesta C and González-Barón M: PI3K/Akt signalling
pathway and cancer. Cancer Treat Rev. 30:193–204. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hennessy BT, Smith DL, Ram PT, Lu Y and
Mills GB: Exploiting the PI3K/AKT pathway for cancer drug
discovery. Nat Rev Drug Discov. 4:988–1004. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Minguez B, Tovar V, Chiang D, Villanueva A
and Llovet JM: Pathogenesis of hepatocellular carcinoma and
molecular therapies. Curr Opin Gastroenterol. 25:186–194. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Saxena NK, Sharma D, Ding X, et al:
Concomitant activation of the JAK/STAT, PI3K/AKT, and ERK signaling
is involved in leptin-mediated promotion of invasion and migration
of hepatocellular carcinoma cells. Cancer Res. 67:2497–2507. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang GL, Iakova P, Wilde M, Awad S and
Timchenko NA: Liver tumors escape negative control of proliferation
via PI3K/Akt-mediated block of C/EBP alpha growth inhibitory
activity. Genes Dev. 18:912–925. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Nogueira V, Park Y, Chen CC, et al: Akt
determines replicative senescence and oxidative or oncogenic
premature senescence and sensitizes cells to oxidative apoptosis.
Cancer Cell. 14:458–470. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu J, Zhou J and Xing D:
Phosphatidylinositol 3-kinase plays a vital role in regulation of
rice seed vigor via altering NADPH oxidase activity. PLoS One.
7:e338172012. View Article : Google Scholar : PubMed/NCBI
|