Introduction
Diabetes and hypertension are common metabolic
diseases. Previous epidemiological studies have indicated that the
two diseases often occur one after the other and cause a condition
termed diabetic hypertension (DHT). According to a World Health
Organization study, ~60% of patients with type 2 diabetes also
suffer from hypertension (1). When
a patient suffers the two diseases at the same time, damage to
target organs, such as the brain, is more significant.
Brain damage is a serious chronic complication of
DHT. Previous studies have demonstrated that following cerebral
infarction, cerebral white matter lesions and other medical imaging
changes are observed (2,3). These changes may be examined using
conventional tools, including computed tomography (CT) and magnetic
resonance imaging (MRI); however, such tools can only provide the
morphological information on the late complications of brain damage
caused by DHT. For a number of diseases, it is crucial and useful
for diagnosis and treatment (4,5) to
know at an early stage the metabolic information of the organs
affected by the disease, as the metabolic changes usually occur
prior to the pathological alterations in the tissue structure.
Magnetic resonance spectroscopy (MRS) is a safe,
non-invasive means of performing biochemical analyses in
vivo. Using this technique, information regarding the metabolic
alterations in different tissue types, including brain tissues
(6) can be observed. Generally,
MRS consists of two techniques, termed single voxel spectroscopy
(SVS) and multi-voxel spectroscopy (MVS). SVS receives a signal of
a volume limited to a single voxel. The advantages of SVS include a
shorter acquisition time, more explicit spatial localization, more
homogeneous shimming and improved water suppression. However, it
has a number of crucial disadvantages. Notably, only one spectrum
may be obtained from one data acquisition and the comparison cannot
be performed simultaneously between the contralateral tissues
during one data collection. Furthermore, the accuracy of SVS is
also affected by the localization of voxel. MVS, also known as
chemical shift imaging, is a multi-voxel technique, which allows
for the measurement of larger volumes of tissue that can be divided
into smaller voxels during the processing period. It is capable of
comparing spectra from multiple brain regions simultaneously. When
the lesion is uneven in quality, the use of small voxels may reduce
the average volume effect (7,8).
Numerous studies have predominantly used SVS to
investigate the brain metabolites in patients with diabetes or
hypertension. It was identified that the N-acetylaspartate (NAA)
concentrations in the bilateral hippocampus were lower in diabetic
patients than in the normal group and the Choline (Cho)
concentrations in the bilateral hippocampus were higher in the
normal group (9). There are also
certain studies, which noted that when comparing type 2 diabetic
patients with the control group, the NAA/creatine (Cr) and Cho/Cr
values were decreased in the frontal cortex (10–12).
In addition, Catani et al (13) observed that the NAA/Cr ratio and
Cho/Cr values of the white matter were not altered for patients in
the hypertension group and those in the control group. Ben Salem
et al (14) found that the
NAA/Cr values of bilateral thalamus and the insular cortex were
lower in hypertensive patients than in the normal control group.
Previous studies (15-18) indicated that cerebral blood flow
with hypertension usually induced early changes in the white matter
of bilateral frontal cortex and the parietal lobe. In addition,
lacunar infarction often occurred in the basal ganglia, corona
radiate and centrum ovale in patients with diabetes.
Currently, there are few studies that focus on the
metabolic changes in the brain of patients with DHT. Thus, in the
present study, the technique of MVS was used and a semioval center
was selected as the region of interest for monitoring the early
brain metabolite changes in DHT.
Materials and methods
Subjects
A total of 33 patients with DHT (14 males and 19
females; mean age, 62.8±8.6 years) were enrolled in the present
study. A total of 30 age-matched volunteers (20 males, 10 females,
mean age 59.8±7.7 years) were included as the control group. All
subjects were right-handed.
All patients diagnosed with DTH had to meet the
following criteria: The patient had a fasting plasma glucose level
≥7.0 mmol/l, random blood glucose level ≥11.1 mmol/l or following
an oral glucose tolerance test (OGTT) a blood glucose level ≥11.1
mmol/l after 2 h; they were not taking antihypertensive drugs and
they had a systolic blood pressure (SBP) ≥140 mmHg and (or)
diastolic blood pressure (DPB) ≥90 mmHg. For the control group, all
selected candidates had to meet the following criteria: They were
not taking anti-hypertensive drugs; they had an SBP <140 mmHg
and a diastolic blood pressure <90 mmHg; their fasting blood
glucose level was <6.1 mmol/l and following OGTT, they had a
blood glucose level of <7.8 mmol/l after 2 h, and no history of
abnormal lipid metabolism.
Exclusion criteria for the present study included
patients who had previously suffered central nervous system damage
caused by other diseases or a similar central nervous illness,
history of drug dependence or other substance abuse, a history of
mental illness or a history of severe medical illness.
Proton magnetic resonance spectroscopy
(2D-1HMRS) imaging
The present study was conducted in the Medical
Imaging Department of The Second Affiliated Hospital of Shantou
University Medical College (Shantou, China) between December 2011
and July 2012. The study was approved by the ethics committee of
Shantou University Medical College (The Second Affiliated Hospital,
Shantou, China)The local ethics committee approved the study and
all volunteers provided informed consent.
The MR study was performed on a 1.5T GE Signa HDX
scanner (GE Healthcare, Wauwatosa, WI, USA) with an 8-channel head
coil. During the scan, the heads of subjects were fixed with a
sponge pad in order to enable the patient to remain stationary.
Routine MRI imaging, including an axial T1-weighted
image (WI), a T2WI and a sagittal or coronal
T2WI was conducted for each subject. The scanning
parameters were as follows: T1WI [repetition time/echo
time (TR/TE)=2,162 ms/20.6 ms, matrix=320×256, field of view
(FOV)=24×18 cm, slice thickness=7 mm, gap=1.5 mm]; T2WI
(TR/TE=4,420 ms/112.1 ms, matrix=384×256, FOV=24×18 cm, slice
thickness=7 mm, gap=1.5 mm). The scan range was from the parietal
to the foramen magnum. Localized proton spectra were acquired using
the point resolved selective spectroscopy sequence. The parameters
were as follows: TR=1,500 ms, TE=35 ms, matrix=512×512, phase and
frequency=18×18, FOV=16×16 cm, number of excitations=1.0. A total
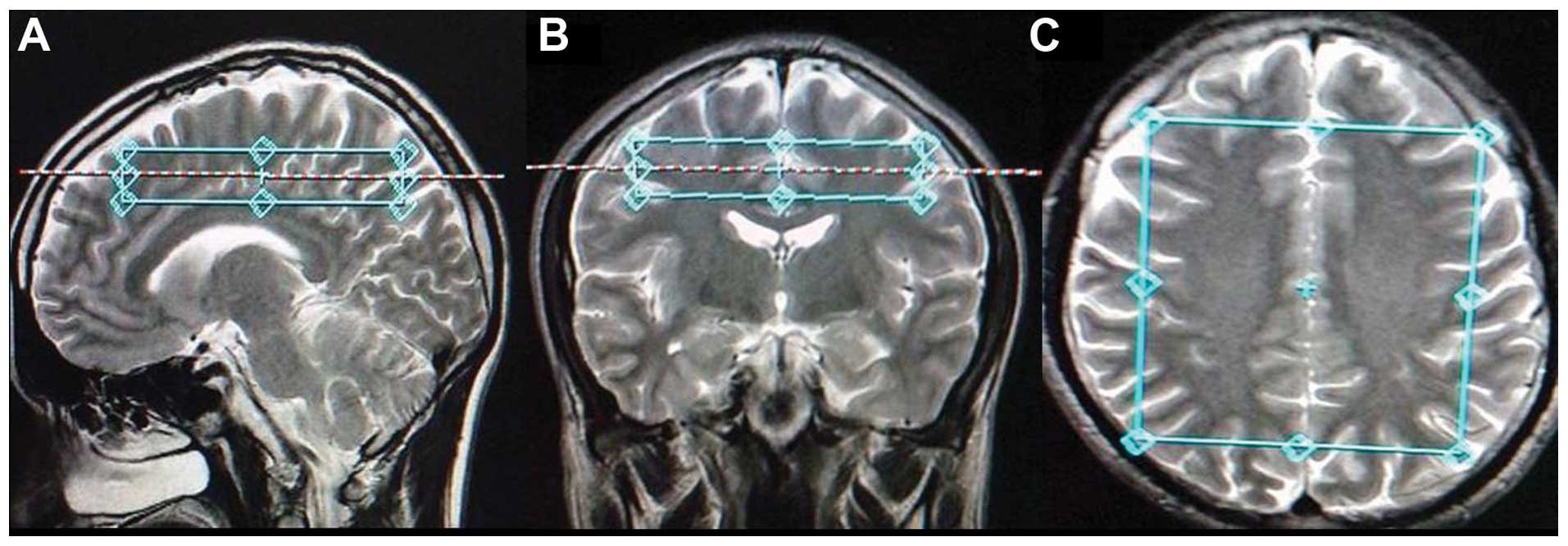
of three planes were used to determine the volumes of interest
(VOI) and the VOI was placed in the central layer of a semioval.
The volume of VOI was 7×10×2 cm and each single small voxel size
was 1.58 cm3 (Fig. 1).
In order to meet the requirements for analyzing the metabolites,
the full width at half maximum was controlled within 10 Hz and the
inhibited water level was >98%, which can be accomplished by
alternatively using the automatic and manual shimming methods. The
total acquisition time for MRS data was 8 min and 12 sec.
Data processing
Following the 2D-1HMRS scan, the data
were imported, and SAGE, version 2007.1 (GE Company, Waukesha, WI,
USA) and LCModel software, version 6.3 (http://s-provencher.com/pages/lcmodel.shtml) were
applied to signal averaging, baseline correction, phase cycling,
metabolite identification and calculating the ratio of the
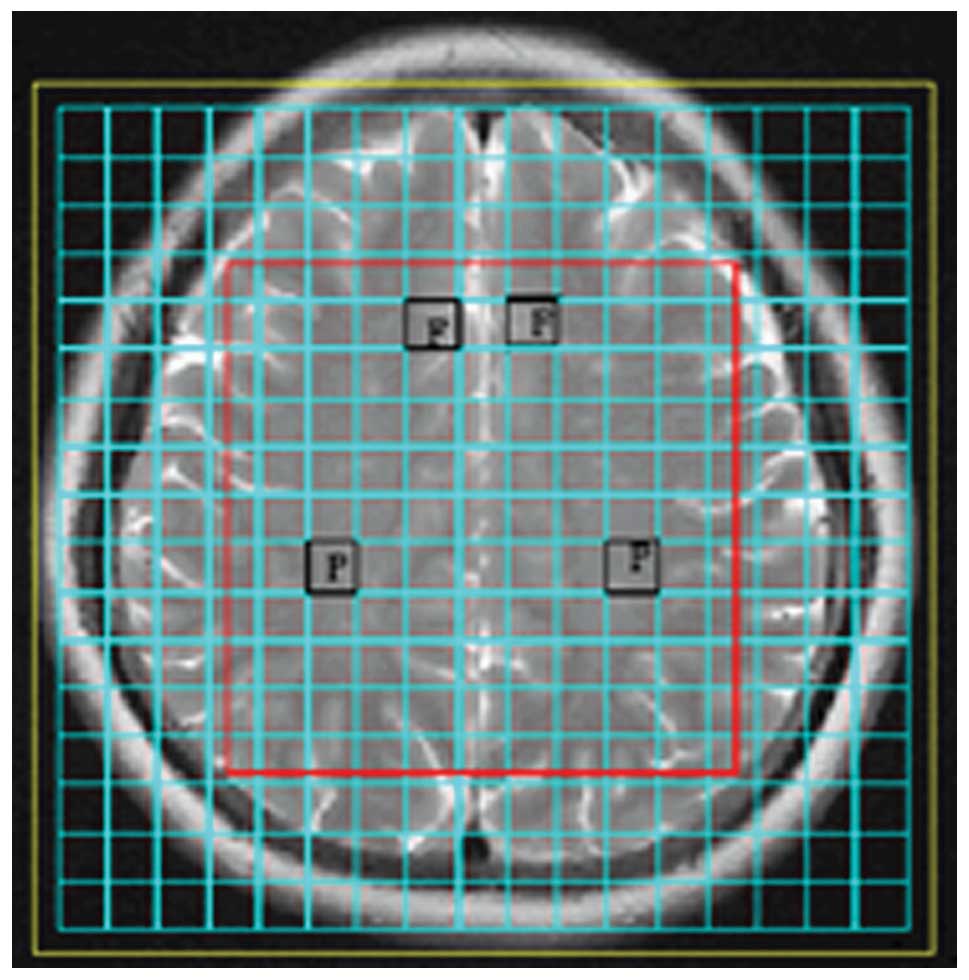
metabolites in each voxel. In the present study, the bilateral
prefrontal cortex and bilateral parietal white matter were selected
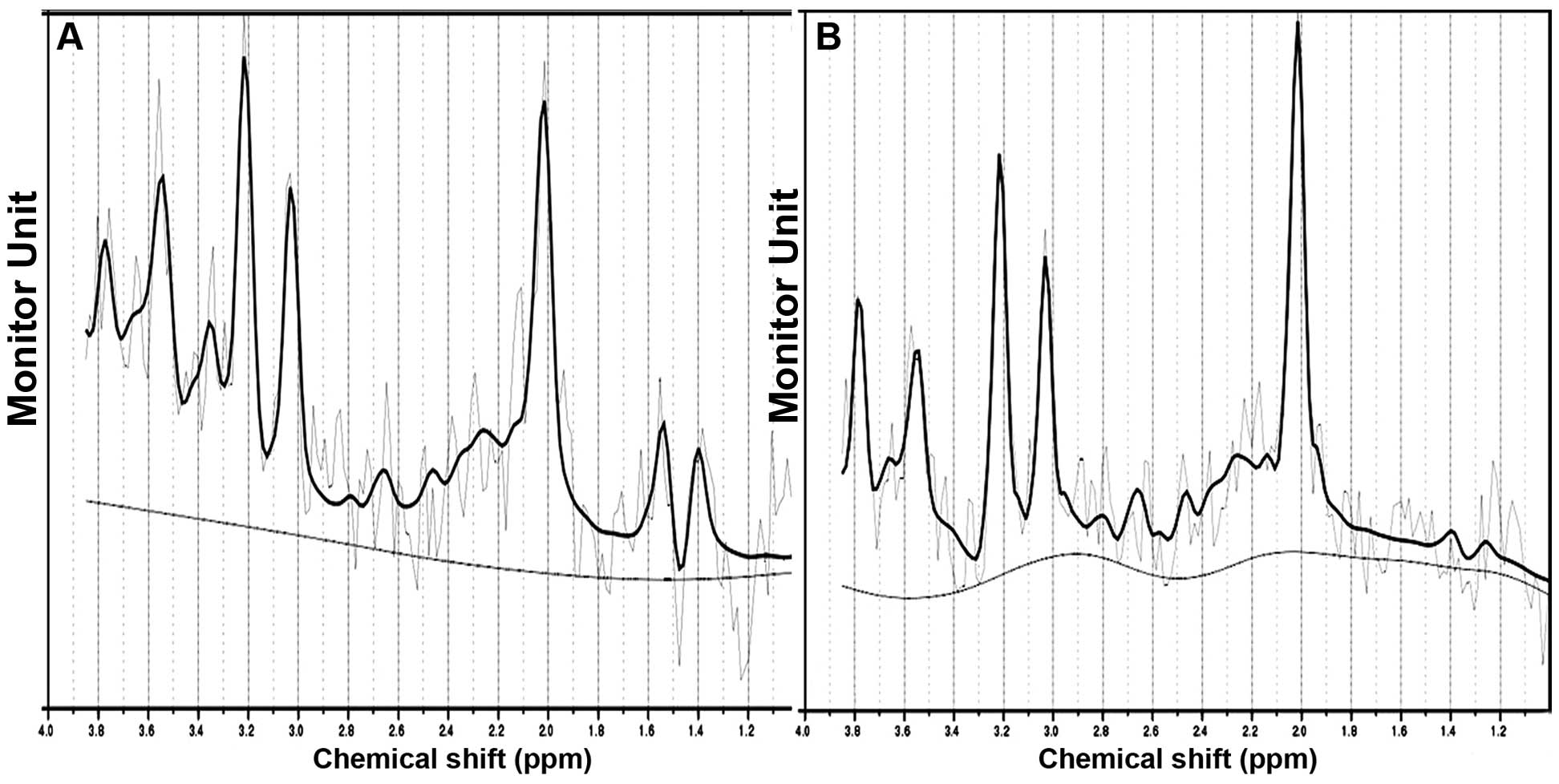
as regions of interest (Fig. 2)
and the ratios of NAA/Cr and Cho/Cr were calculated.
Statistical analysis
The data were analyzed using SPSS 17.0 (SPSS, Inc,
Chicago, IL, USA). An independent-sample t-test was used to compare
the metabolic ratios in different groups. P<0.05 was considered
to indicate a statistically significant difference. The measurement
data were expressed as the mean ± standard deviation.
Results
The age and gender differences did not reach
statistical significance between the DHT and the control group.
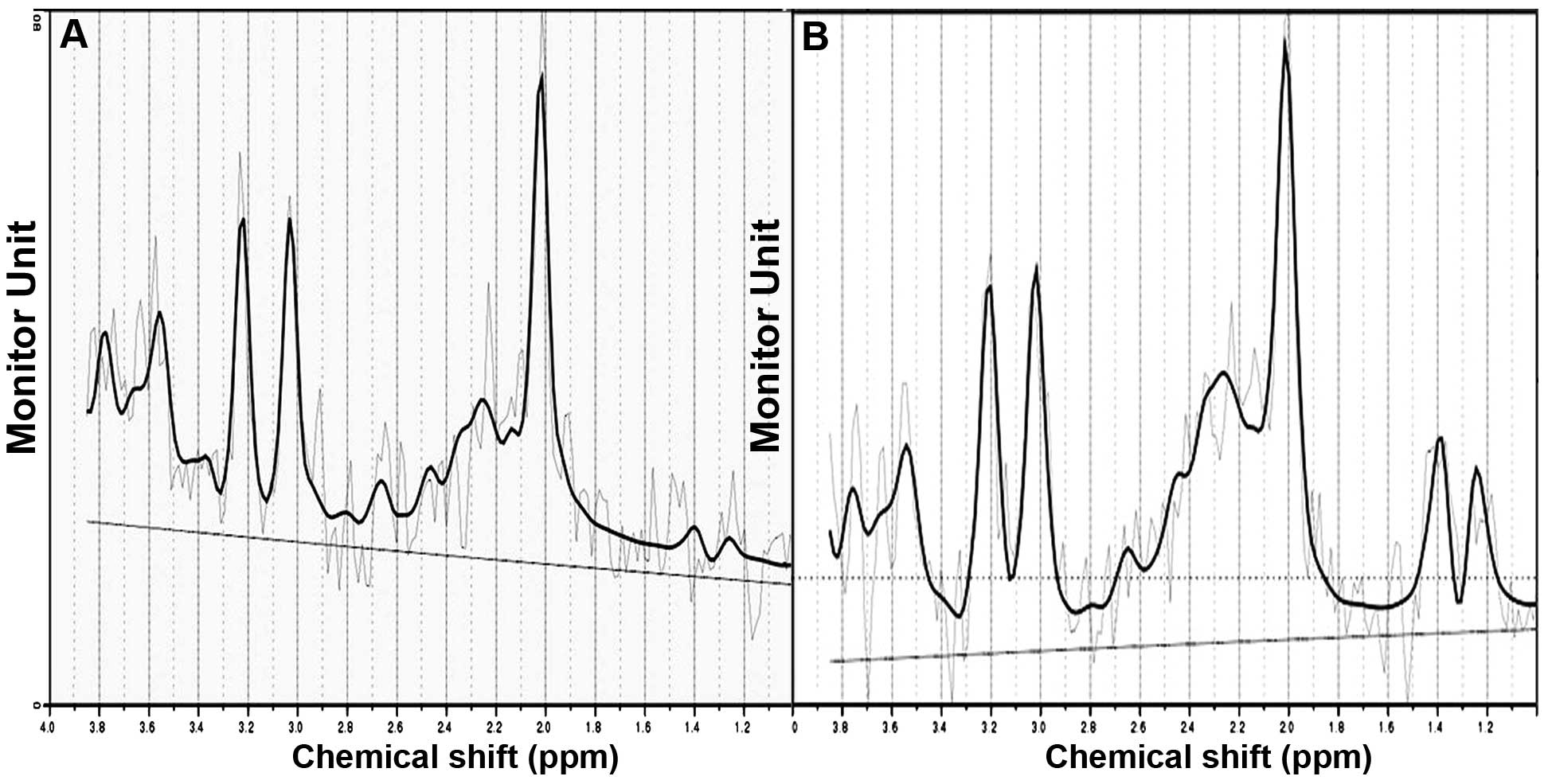
High quality 2D-1HMRS data were acquired. The metabolite
peak intensities were as follows: NAA=2.02 ppm, Cr=3.0 ppm, Cho=3.2
ppm and MI=3.56 ppm. The ratios of NAA/Cr and Cho/Cr of the
bilateral prefrontal cortex and the parietal lobe white matter in
the two groups are shown in Tables
I and II. The NAA/Cr ratios
in the bilateral prefrontal cortex of the DHT group were
significantly lower than that of the control group (left t=−7.854,
P=0.000; right t=−5.787, P=0.000), The Cho/Cr ratios in bilateral
prefrontal cortex in the DHT group were higher than that of the
control group and revealed a significant difference (left t=2.422,
P=0.024 and right t=2.920, P=0.007; Figs. 3 and 4).
 | Table IMagnetic resonance spectroscopy values
of the diabetic hypertension group and the control group in the
prefrontal cortex. |
Table I
Magnetic resonance spectroscopy values
of the diabetic hypertension group and the control group in the
prefrontal cortex.
| Variable | NAA/Cr
| Cho/Cr
|
|---|
| Left | Right | Left | Right |
|---|
| DHT | 0.707±0.257 | 1.071±0.298 | 0.396±0.086 | 0.441±0.114 |
| CON | 1.483±0.499 | 1.506±0.298 | 0.311±0.091 | 0.334±0.068 |
| t-valuea | −7.854 | −5.787 | 2.422 | 2.920 |
| P-value | 0.000 | 0.000 | 0.024 | 0.007 |
 | Table IIMagnetic resonance spectroscopy
values of the diabetic hypertension and the control group in the
parietal white matter. |
Table II
Magnetic resonance spectroscopy
values of the diabetic hypertension and the control group in the
parietal white matter.
| Variable | NAA/Cr
| Cho/Cr
|
|---|
| Left | Right | Left | Right |
|---|
| DHT | 1.020±0.512 | 1.316±0.373 | 0.349±0.112 | 0.315±0.088 |
| CON | 1.500±0.379 | 1.450±0.503 | 0.327±0.086 | 0.324±0.085 |
| t valuea | −4.199 | −1.215 | 0.874 | −0.432 |
| P value | 0.000 | 0.229 | 0.386 | 0.667 |
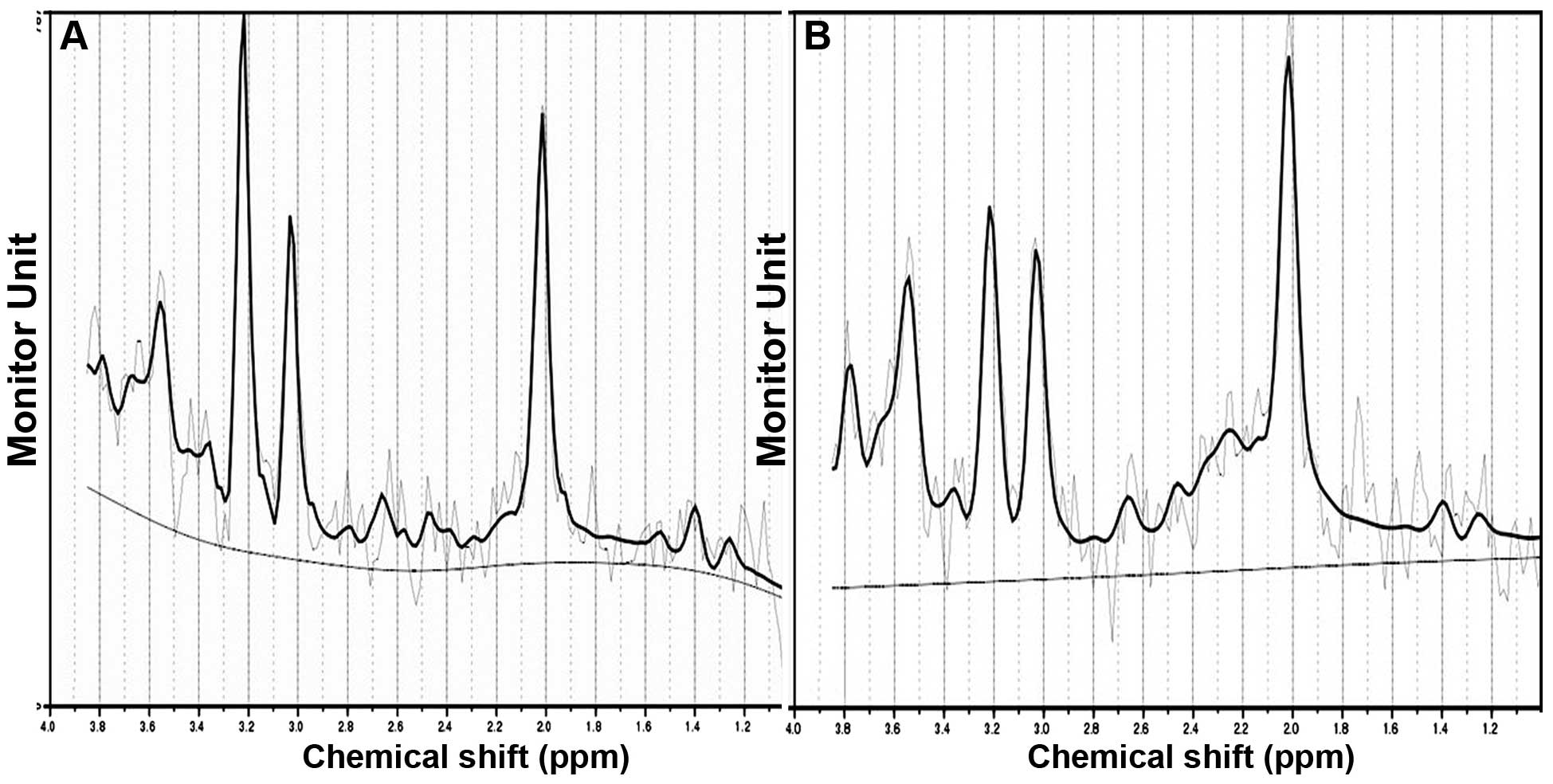
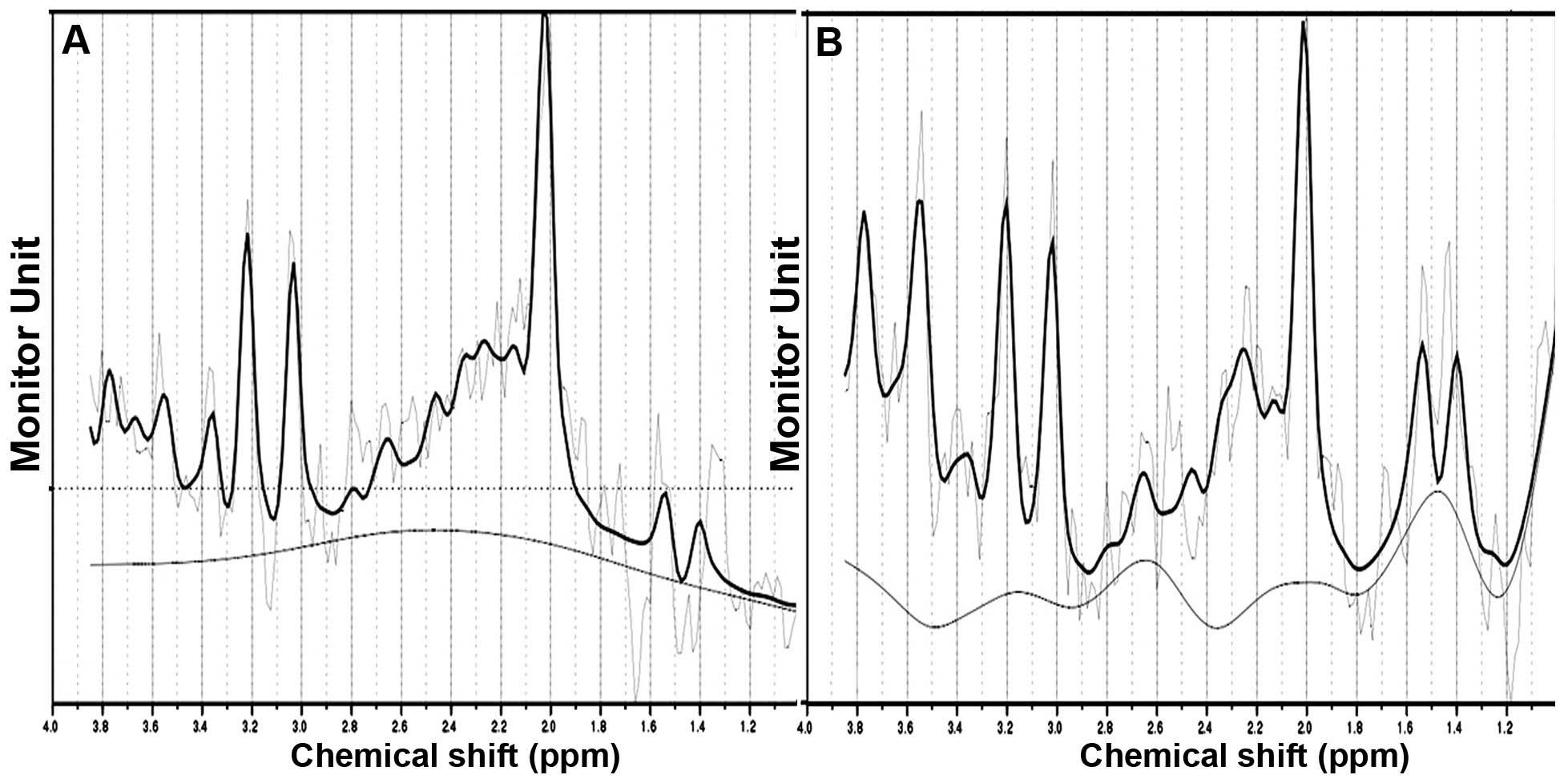
Compared with the control group, the NAA/Cr levels
in the left parietal lobe white matter in the DHT group decreased
and were significantly different (t=−4.199, P=0.000). There was a
downward trend in the NAA/Cr ratio in the right parietal white
matter in the DHT group, however it was not statistically
significant (t=−1.215, P=0.229). No statistically significant
differences were identified in the Cho/Cr ratio in the bilateral
parietal lobe white matter between the DHT group and the control
group (left t=0.874, P=0.386; right t=−0.432, P=0.667; Figs. 5 and 6).
Discussion
DHT has a high incidence and may increase the extent
of brain damage associated with diabetes and simple essential
hypertension as patients have numerous coinciding risk factors
(19). The pathogenic mechanism
remains to be elucidated; however, it has been reported to be
closely associated with cerebrovascular damage, oxidative stress
and non-enzymatic protein glycosylation. These pathophysiological
changes may be identified by observing the relevant metabolite
alterations (20,21).
MRS, which is a non-invasive imaging technique with
reproducibility is a method used to detect metabolites in the body
(22). MRS can currently detect
numerous types of metabolites, including NAA, Cho, Cr, lactate,
lipids, alanine, glutamic acid, γ-amino butyric acid, inositol and
a number of others. Cr is used as a reference standard with other
metabolites as it is not usually affected by pathological states
(23). Therefore, the ratios of
NAA/Cr and Cho/Cr to a certain extent may reflect the NAA and Cho
concentration alterations.
The levels of the amino acid NAA in the brain are
only marginally lower than that of glutamic acid, which is the most
abundant amino acid in the brain. NAA is primarily found in
neuronal cell bodies and axons, but not in glial cells, therefore
NAA is considered to be a marker for neurons. It is generally
accepted that a decrease in NAA reflects a loss of neurons, a
decline in neuronal activity, a metabolic decrease in the gray
matter and also axonal injury in the white matter (24,25).
Although Kario et al (26)
identified that NAA had an independent association with type 2
diabetes, a study has also revealed that diabetes may cause DNA
damage in hippocampal neurons, leading to the reduction in neurons
and further affecting the synaptic plasticity (27). In addition, the studies by Kreis
et al (28) and Kreis and
Ross (29) indicated that NAA
levels were significantly decreased in the parietal white matter of
patients with diabetes. In addition, a study by Shiino et al
(30) demonstrated that the NAA/Cr
ratio was significantly lower in the lesions of subcortical
arteriosclerotic encephalopathy compared with that in normal
tissue. The authors hypothesized that the NAA level was associated
with the ongoing hypertension, which led to chronic cerebral
circulatory disorders, ischemia, hypoxic changes and neuronal loss.
In the present study, The NAA/Cr levels in the bilateral prefrontal
cortex were observed to be lower in the DHT group than those in the
control group and the difference was statistically significant. The
reason for these findings may be that the prefrontal cortex is
sensitive to ischemia. It is well-established that hypertension and
diabetes can cause vascular wall thickening and luminal stenosis,
which decreases cerebral tissue perfusion and results in ischemia
and hypoxia in the brain. Under the pathological state of sustained
ischemia, the NAA level decreases due to neuronal cell and axonal
loss (31). In the present study,
it was also identified that the NAA/Cr ratios in the DHT group were
lower in the left parietal white matter than those in the control
group and the difference was statistically significant. The main
reason for these alterations is that the left hemisphere is usually
dominant in right-handed subjects, thus in these individuals it has
a lower tolerance to ischemia than the right side and is more
vulnerable to neuronal damage and metabolic disorders (32).
The Cho level is higher in glial cells than in other
types of cells, thus it is classified as a metabolic biomarker of
glial cells (33). Cho is a
constituent of acetylcholine and phosphatidylcholine, and it is
also a constituent of cell membranes (34). A rise in the Cho level indicates
intense metabolic activation of the cell or membranous
disintegration. The reduction in Cho may be relevant to the
renovation of the membrane phospholipids, inositol synthesis, cell
density changes, endocrine state and local metabolic rate changes
(35,36). In addition, the Cho level also
reflects the severity of inflammation, with a Cho increase
indicating a greater level of inflammation.
In the present study, the Cho/Cr values of patients
with DHT in bilateral prefrontal cortex were found to be increased
compared with those in the normal control group. This result may be
associated with the interaction of diabetes and hypertension. These
two diseases damage the brain blood vessels so that
ischemia/hypoxia is induced in the gray matter. Ultimately, the
nerve cells of the gray matter degenerate and undergo necrosis,
resulting in disintegration of the nerve cell membranes. It may
also be relevant that there is increased inflammation in DTH. Liu
et al (37) suggested that
type 2 diabetes is an inflammatory disease and thus the increased
inflammatory mediators may result in raised Cho/Cr levels (38,39).
To conclude, based on the present experimental
results and compared with the existing data, it was identified that
DHT may result in metabolic disorders in the frontal cortex and
parietal white matter; however, the metabolic alterations are
distinct in these regions of the brain. Therefore, the ratios of
NAA/Cr and Cho/Cr, which can be detected by 2D-1HMRS in
these regions, may be used as potential metabolite markers for the
brain damage induced by DHT.
Acknowledgments
The present study was supported by the key program
of the National Natural Science Foundation of China (grant no.
30930027), the National Natural Science Foundation of China (grant
no. 81371612) and the Natural Science Foundation of Guangdong
Province, China (grant no. S2013010013867).
References
|
1
|
Chobanian AV, Bakris GL, Black HR, et al:
The seventh report of the Joint National Committee on prevention,
detection, evaluation, and treatment of high blood pressure: the
JNC 7 report. JAMA. 289:2560–2572. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
van Elderen SG, Brandts A, van der Grond
J, Westenberg JJ, Kroft LJ, van Buchem MA, Smit JW and de Roos A:
Cerebral perfusion and aortic stiffness are independent predictors
of white matter brain atrophy in type 1 diabetic patients assessed
with magnetic resonance imaging. Diabetes Care. 34:459–463. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van Elderen SG, Westenberg JJ, Brandts A,
van der Meer RW, Romijn JA, Smit JW and de Roos A: Increased aortic
stiffness measured by MRI in patients with type 1 diabetes mellitus
and relationship to renal function. AJR Am J Roentgenol.
196:697–701. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tomiyasu M, Aida N, Endo M, Shibasaki J,
Nozawa K, Shimizu E, Tsuji H and Obata T: Neonatal brain metabolite
concentrations: An in vivo magnetic resonance spectroscopy study
with a clinical MR system at 3 Tesla. PLoS One. 8:1–7. 2013.
View Article : Google Scholar
|
|
5
|
Yildiz-Yesiloglu A and Ankerst DP:
Neurochemical alterations of the brain in bipolar disorder and
their implications for pathophysiology: a systematic review of the
in vivo proton magnetic resonance spectroscopy findings. Prog
Neuropsychopharmacol Biol Psychiatry. 30:969–995. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schaeffter T and Dahnke H: Magnetic
resonance imaging and spectroscopy. Molecular Imaging. Handb Exp
Pharmacol. 185:75–90. 2008. View Article : Google Scholar
|
|
7
|
Tate AR, Griffiths JR, Martínez-Pérez I,
Moreno A, Barba I, Cabañas ME, Watson D, Alonso J, Bartumeus F,
Isamat F, et al: Towards a method for automated classification of
1H MRS spectra from brain tumours. NMR Biomed. 11:177–191. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Vicente J, Fuster-Garcia E, Tortajada S,
García-Gómez JM, Davies N, Natarajan K, Wilson M, Grundy RG,
Wesseling P, Monleón D, et al: Accurate classification of childhood
brain tumours by in vivo ¹H MRS a multi centre study. Eur J Cancer.
49:658–667. 2013. View Article : Google Scholar
|
|
9
|
Sinha S, Ekka M, Sharma U, P R, Pandey RM
and Jagannathan NR: Assessment of changes in brain metabolites in
Indian patients with type-2 diabetes mellitus using proton magnetic
resonance spectroscopy. BMC Res Notes. 7:412014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sahin I, Alkan A, Keskin L, Cikim A,
Karakas HM, Firat AK and Sigirci A: Evaluation of in vivo cerebral
metabolism on proton magnetic resonance spectroscopy in patients
with impaired glucose tolerance and type 2 diabetes mellitus. J
Diabetes Complications. 22:254–260. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang M, Sun X, Zhang Z, Meng Q, Wang Y,
Chen J, Ma X, Geng H and Sun L: Brain metabolite changes in
patients with type 2 diabetes and cerebral infarction using proton
magnetic resonance spectroscopy. Int J Neurosci. 124:37–41. 2014.
View Article : Google Scholar
|
|
12
|
Hajek T, Calkin C, Blagdon R, Slaney C and
Alda M: Type 2 diabetes mellitus: A potentially modifiable risk
factor for neurochemical brain changes in bipolar disorders. Biol
Psychiatry. 87:295–303. 2015. View Article : Google Scholar
|
|
13
|
Catani M, Mecocci P, Tarducci R, Howard R,
Pelliccioli GP, Mariani E, Metastasio A, Benedetti C, Senin U and
Cherubini A: Proton magnetic resonance spectroscopy reveals similar
white matter biochemical changes in patients with chronic
hypertension and early Alzheimer’s disease. J Am Geriatr Soc.
50:1707–1710. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ben Salem D, Walker PM, Bejot Y, Aho SL,
Tavernier B, Rouaud O, Ricolfi F and Brunotte F:
N-acetylaspartate/creatine and choline/creatine ratios in the
thalami, insular cortex and white matter as markers of hypertension
and cognitive impairment in the elderly. Hypertens Res.
31:1851–1857. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Davis BR, Vogt T, Frost PH, Burlando A,
Cohen J, Wilson A, Brass LM, Frishman W, Price T and Stamler J:
Risk factors for stroke and type of stroke in persons with isolated
systolic hypertension. Systolic Hypertension in the Elderly Program
Cooperative Research Group. Stroke. 29:1333–1340. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Djelilovic-Vranic J, Alajbegovic A,
Zelija-Asimi V, Niksic M, Tiric-Campara M, Salcic S and Celo A:
Predilection role diabetes mellitus and dyslipidemia in the onset
of ischemic stroke. Med Arch. 67:120–123. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hägg S, Thorn LM, Putaala J, Liebkind R,
Harjutsalo V, Forsblom CM, Gordin D and Tatlisumak T; Groop PH
FinnDiane Study Group: Incidence of stroke according to presence of
diabetic nephropathy and severe diabetic retinopathy in patients
with type 1 diabetes. Diabetes Care. 36:4140–4146. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tanaka R, Ueno Y, Miyamoto N, Yamashiro K,
Tanaka Y, Shimura H, Hattori N and Urabe T: Impact of diabetes and
prediabetes on the short-term prognosis in patients with acute
ischemic stroke. J Neurol Sci. 332:45–50. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
van Bilsen M, Daniels A, Brouwers O,
Janssen BJ, Derks WJ, Brouns AE, Munts C, Schalkwijk CG, van der
Vusse GJ and van Nieuwenhoven FA: Hypertension is a conditional
factor for the development of cardiac hypertrophy in type 2
diabetic mice. PLoS One. 9:e850782014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Nakagawa T: A new mouse model resembling
human diabetic nephropathy: uncoupling of VEGF with eNOS as a novel
pathogenic mechanism. Clin Nephrol. 71:103–109. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Baldwin MD: Assessing cardiovascular risk
factors and selecting agents to successfully treat patients with
type 2 diabetes mellitus. J Am Osteopath Assoc. 111:S2–S12.
2011.PubMed/NCBI
|
|
22
|
Bruzzone MG, D’Incerti L, Farina LL,
Cuccarini V and Finocchiaro G: CT and MRI of brain tumors. Q J Nucl
Med Mol Imaging. 56:112–137. 2012.PubMed/NCBI
|
|
23
|
Law M: Advanced imaging techniques in
brain tumors. Cancer Imaging. 9:S4–S9. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Moffett JR, Ross B, Arun P, Madhavarao CN
and Namboodiri AM: N-Acetylaspartate in the CNS: from
neurodiagnostics to neurobiology. Prog Neurobiol. 81:89–131. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Szulc A: First and second generation
antipsychotics and morphological and neurochemical brain changes in
schizophrenia. Review of magnetic resonance imaging and proton
spectroscopy findings. Psychiatr Pol. 41:329–338. 2007.In Polish.
PubMed/NCBI
|
|
26
|
Kario K, Ishikawa J, Hoshide S, Matsui Y,
Morinari M, Eguchi K, Ishikawa S and Shimada K: Diabetic brain
damage in hypertension: role of renin-angiotensin system.
Hypertension. 45:887–893. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Eguchi K, Pickering TG and Kario K: Why is
blood pressure so hard to control in patients with type 2 diabetes?
J Cardiometab Syndr. 2:114–118. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kreis R, Ross BD, Farrow NA and Ackerman
Z: Metabolic disorders of the brain in chronic hepatic
encephalopathy detected with H-1 MR spectroscopy. Radiology.
182:19–27. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kreis R and Ross BD: Cerebral metabolic
disturbances in patients with subacute and chronic diabetes
mellitus: detection with proton MR spectroscopy. Radiology.
184:123–130. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shiino A, Watanabe T, Shirakashi Y, Kotani
E, Yoshimura M, Morikawa S, Inubushi T and Akiguchi I: The profile
of hippocampal metabolites differs between Alzheimer’s disease and
subcortical ischemic vascular dementia, as measured by proton
magnetic resonance spectroscopy. J Cereb Blood Flow Metab.
32:805–815. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ciobanu AO, Gherghinescu CL, Dulgheru R,
Magda S, Dragoi Galrinho R, Florescu M, Guberna S, Cinteza M and
Vinereanu D: The impact of blood pressure variability on
subclinical ventricular, renal and vascular dysfunction, in
patients with hypertension and diabetes. Maedica (Buchar).
8:129–136. 2013.
|
|
32
|
Heiss WD, Hartmann A, Rubi Fessen I,
Anglade C, Kracht L, Kessler J, Weiduschat N, Rommel T and Thiel A:
Noninvasive brain stimulation for treatment of right and left
handed poststroke aphasics. Cerebrovasc Dis. 36:363–372. 2013.
View Article : Google Scholar
|
|
33
|
Hang L, Friedman J, Ernst T, Zhong K,
Tsopelas ND and Davis K: Brain metabolite abnormalities in the
white matter of elderly schizophrenic subjects: implication for
glial dysfunction. Biol Psychiatry. 62:1396–1404. 2007. View Article : Google Scholar
|
|
34
|
Abdel Razek AA and Poptani H: MR
spectroscopy of head and neck cancer. Eur J Radiol. 82:982–989.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Takenaka S, Shinoda J, Asano Y, Aki T,
Miwa K, Ito T, Yokoyama K and Iwama T: Metabolic assessment of
monofocal acute inflammatory demyelination using MR spectroscopy
and (11)C-methionine, (11)C-choline-, and
(18)F-fluorodeoxyglucose-PET. Brain Tumor Pathol. 28:229–238. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mellergård J, Tisell A, Dahlqvist Leinhard
O, Blystad I, Landtblom AM, Blennow K, Olsson B, Dahle C, Ernerudh
J, Lundberg P and Vrethem M: Association between change in normal
appearing white matter metabolites and intrathecal inflammation in
natalizumab-treated multiple sclerosis. PLoS One. 7:1–9. 2012.
View Article : Google Scholar
|
|
37
|
Liu LL, Yan L, Chen YH, Zeng GH, Zhou Y,
Chen HP, Peng WJ, He M and Huang QR: A role for diallyl trisulfide
in mitochondrial antioxidative stress contributes to its protective
effects against vascular endothelial impairment. Eur J Pharmacol.
14:11–19. 2014.
|
|
38
|
Danyel LA, Schmerler P, Paulis L, Unger T
and Steckelings UM: Impact of AT2-receptor stimulation on vascular
biology, kidney function, and blood pressure. Integr Blood Press
Control. 6:153–161. 2013.
|
|
39
|
Fukasawa R, Hanyu H, Namioka N, Hatanaka
H, Sato T and Sakurai H: Elevated inflammatory markers in
diabetes-related dementia. Geriatr Gerontol Int. 14:229–231. 2014.
View Article : Google Scholar : PubMed/NCBI
|