Introduction
Acute lymphoblastic leukemia (ALL) occurs most
frequently in children, with incidence peaks between the ages of 2
and 5 years, and is one of the most common childhood malignancies
worldwide (1,2). T-cell ALL (T-ALL) is an aggressive
type of blood malignancy, deriving from T-cell progenitors in the
thymus, and comprises 10–15% of pediatric and 25% of adult primary
ALL cases (3). The age of the
patient at diagnosis, leukocyte count, ethnicity, gender and
immunophenotype are clinical prognostic parameters, which are
utilized to classify the patients into different risk groups
(1). T-ALL is classified as a
high-risk group in ALL (2). With
the currently used intensive multi-agent chemotherapeutic
procedures, the 5-year event-free survival (EFS) of children with
T-ALL is 70–75%, whereas the EFS is 30–40% for adults <60 years
of age, and 10% in those >60 years old (4). However, these types of therapy are
highly toxic, and cases of relapse often develop resistance to
chemotherapy, with a poor prognosis (2). Targeting leukemia-specific molecular
abnormalities may further improve outcomes and reduce the adverse
side effects of therapy.
Forkhead box protein M1 (FoxM1), characterized by a
100 amino acid winged-helix DNA binding domain, is a newly unified
family member of the Forkhead transcription factors (5). The protein expression of FoxM1 is
usually restricted to proliferating cells and is absent in
quiescent or terminally differentiated cells, however, elevated
expression levels of FoxM1 have been detected in a wide range of
different types of human tumor and has been implicated in cellular
transformation, tumor initiation and progression (6). By regulating the G1/S and G2/M
transitions and M phase progression, antagonizing cellular
senescence, stimulating stem cell-like characteristics (including
self-renewal), promoting multiple steps of cancer progression by
inducing mitogenic and survival signals, and promoting tumor
invasion, migration, and angiogenesis, FoxM1 is important in
tumorigenesis and cancer progression (6). Accumulating evidence has demonstrated
that increased expression levels of FoxM1 are associated with a
poor prognosis in various types of cancer (7–11),
including acute myeloid leukemia (AML) (12). In addition, FoxM1 deregulation has
also been associated with the development of cancer drug resistance
(13). A previous study confirmed
that suppression of FoxM1 enhanced the chemosen-sitivity of various
types of cancer cell to the DNA-damaging reagent, doxorubicin
(14). Although a number of
investigations have examined the role of FoxM1 in AML (12,15–17),
the role of FoxM1 in ALL, particularly pediatric ALL, remains to be
elucidated.
Thiostrepton, a natural product originally isolated
from Streptomyces azureus, has received significant
attention due to its potent anticancer activity as a FoxM1
inhibitor (18,19). Thiostrepton can downregulate the
mRNA and protein expression levels of FoxM1 (20). The mechanism by which thiostrepton
affects FoxM1 has been confirmed. Thiostrepton hinders the
proteasomal degradation of a negative regulator of FoxM1 (NRFM),
which either directly or indirectly inhibits the activity of FoxM1
as a transcription factor (21).
Notably, thiostrepton appears to exert minimal toxicity against
non-malignant cells (22).
Thiostrepton has exhibited anticancer activity in rodent xenograft
models, without observable toxicity (23,24).
Collectively, these data suggest that thiostrepton is a suitable
treatment for T-ALL, particularly in children. In the present
study, the antileukemia effects of thiostrepton were examined in
human T-ALL Jurkat cells. Crucially, the ability of thiostrepton to
sensitize Jurkat cells to doxorubicin, which is commonly used for
the treatment of ALL, was assessed.
Materials and methods
Chemicals and reagents
Thiostrepton (off-white powder, 1 g), purchased from
Merck Millipore (Darmstadt, Germany), was freshly dissolved in
dimethyl sulfoxide (DMSO; Sigma-Aldrich, St. Louis, MO, USA) to
produce a 20 mmol/l stock solution. Doxorubicin (Haizheng Medicine
Co. Ltd., Zhengjiang, China) was dissolved in a stock concentration
of 10 mmol/l with ddH2O and divided into five aliquots.
Rabbit monoclonal antibody against FoxM1 (cat. no. 5436) was
obtained from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Rabbit polyclonal antibodies against glutathione S-transferase pi
(GSTpi; cat. no. PB0144), survivin (cat. no. BA4055–2), caspase-3
(cat. no. BA2885–2) and β-actin (cat. no. BA2305) were obtained
from Boster Biological Technology, Ltd. (Wuhan, China).
Cell lines and cell culture
The human T-ALL Jurkat cells (Key Laboratory of
Tumour Molecular Biology of Binzhou Medical University (Binzhou,
China) were cultured in RPMI-1640 medium (HyClone Laboratories,
Inc., Logan, UT, USA), supplemented with 10% heat-inactivated fetal
bovine serum (FBS) (GE Healthcare Bio-Sciences, Logan, UT, USA) at
37°C in 5% CO2.
Cell viability assay
Cell viability was assessed using a Cell Counting
Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc., Shanghai,
China), according to the manufacturer’s instructions. The Jurkat
cells were seeded into 96-well flat plates (1.5×104
cells/well) and then treated with or without various concentrations
of thiostrepton (0, 1, 2 or 3 μm/l) for 24, 48 or 72 h at
37°C. The cells in the control group were treated with the
equivalent quantity of dimethyl sulfoxide, at a final concentration
of 0.02%. The CCK-8 kit was then used, according to the
manufacturer’s instructions. The optical density (OD) was measured
at 450 nm using a fluorescence spectrofluorometer (F-7000; Hitachi
High-Technologies Corporation, Tokyo, Japan), and the reference OD
was subtracted. RPMI-1640 containing 10% FBS with 0.02% DMSO served
as the reference group. Cell viability was then calculated as
follows: Cell viability = (ODsample -
ODreference) / (ODcontrol -
ODreference) × 100%. The half maximal inhibitory
concentration of cells (IC50) was obtained using a
probit regression analysis method (25). Each experiment was performed in
triplicate and repeated three times.
Cell cycle analysis
The Jurkat cells were seeded into 6-well plates in
RPMI-1640, containing 10% fetal bovine serum (FBS), with or without
the agents, for 24 h at 37°C. The cells were harvested and fixed
with 500 μl 70% cold (−20°C) ethanol (Shenzhen Kingstone
Experimental Equipment Co., Ltd., Shenzhen, China) at 4°C
overnight, and then incubated with RNase A (KeyGen Biotech Co.,
Ltd., Nanjing, China) for 30 min at 37°C, followed by incubation
with 100 μg/ml propidium iodide (PI; KeyGen Biotech Co.,
Ltd.) at room temperature for 30 min. The cell cycle distribution
was detected using a flow cytometer (FACS FC500; Beckman Coulter,
Brea, CA, USA).
Detection of apoptosis
The Jurkat cells (2.5×105 cells/ml) were
seeded into 6-well plates in RPMI-1640 containing 10% FBS, with or
without the agents, for 24 h. Tells were collected and washed twice
with phosphate-buffered saline (PBS), suspended in 500 μl
binding buffer (KeyGen Biotech Co., Ltd.), 5 μl annexin
V-fluorescein isothiocyanate and 5 μl PI (KeyGen Biotech
Co., Ltd.) for 15 min in the dark at room temperature. The
apoptotic cells were analyzed using a flow cytometer.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was isolated from the cells using TRIzol
reagent (Invitrogen Life Technologies, Carlsbad, CA, USA). RT into
complementary DNA (cDNA) was performed using a PrimeScript™ RT
reagent kit with gDNA eraser (Takara Bio, Inc., Otsu, Japan). qPCR
was performed using a SYBR® Premix Ex Taq™ kit (Takara
Bio, Inc.) on an ABI PRISM 7500 real-time PCR system (Applied
Biosystems, Foster City, CA, USA). The cycling parameters were as
follows: 95°C for 30 sec, then 40 cycles of 95°C for 5 sec, 60°C
for 34 sec. The isolation of RNA, RT and qPCR were performed,
according to the manufacturer’s instructions. The primers used
(Table I) were designed using
Primer 5 version 5.6.0 software and synthesized by Sangon Biotech
Co, Ltd. (Shanghai, China). GAPDH was used as an internal control.
qPCR for each gene of each cDNA sample was assayed in triplicate.
The results were calculated using the 2−∆∆Ct method. The
following equations were used: ΔCt = Ct(target gene) -
Ct(actin); ΔΔCt = ΔCt(thiostrepton-treated
cells) - ΔCt(untreated control).
 | Table IPrimers used in reverse
transcription-quantitative polymerase chain reaction. |
Table I
Primers used in reverse
transcription-quantitative polymerase chain reaction.
| Gene | Primer
sequence | Product length
(bp) |
|---|
| FoxM1 | F:
5′-AGGAAGTGGCAGAGTCCAAC-3′
R: 5′-TGCTGTGATGATGCTGTGAA-3′ | 128 |
| GSTpi | F:
5′-CACTCAAAGCCTCCTGCCTA-3′
R: 5′-TGCTGGTCCTTCCCATAGAG-3′ | 127 |
| GAPDH | F:
5′-TGGTATCGTGGAAGGACTCA-3′
R: 5′-CAGTAGAGGCAGGGATGATG-3′ | 131 |
Western blot analysis
The ells were lysed in 100 μl
radioimmunoprecipitation assay buffer (Beyotime Biotechnology,
Shanghai, China)., supplemented with 1 μl
phenylmeth-ylsulfonyl fluoride (Beyotime Biotechnology), and the
protein concentration of the lysate was determined using a
Bicinchoninic Acid Protein Assay kit (Beyotime Biotechnology). The
proteins (50 μg/lane) were separated by 8–12% SDS-PAGE
(Beyotime Biotechnology) and transferred to polyvinylidine
difluoride membranes (EMD Millipore, Bedford, MA, USA). The
membranes were blocked with 5% skimmed milk for 2 h, and then
incubated overnight at 4°C with specific antibodies. The primary
antibodies used were rabbit monoclonal antibody against FoxM1
(1:1,000), and rabbit polyclonal antibodies against GSTpi (1:200),
survivin (1:200), caspase-3 (1:200) and β-actin (1:1,000). The
following day, the membrane was incubated in horesradish
peroxidase-labeled goat anti-rabbit immunoglobulin G (1:5,000; cat.
no. ZB-5301; Beijing ZhongShan-Golden Bridge Technology Co., Ltd.,
Beijing, China) for 1 h at room temperature. Finally, images were
captured using a FluorChem FC2 gel imaging system (Alpha Innotech,
San Leandro, CA, USA).
Flow cytometric analysis of intracellular
doxorubicin accumulation
The intracellular accumulation of doxorubicin was
measured using a standard fluorescent method (26). The Jurkat cells were cultured in
RPMI-1640 containing 10 FBS, with increasing concentrations of
thiostrepton (0, 1, 2 or 3 μM) for 24 h at 37°C. The medium
was then replaced with RPMI-1640 containing 10% FBS and doxorubicin
(0.2 μM), in which the cells were incubated for 1 h at 37°C.
The cells were then harvested and washed twice in ice-cold PBS. The
cell-associated mean fluorescence intensity (MFI) of intracellular
doxorubicin was determined using the FACS FC500 flow cytometer with
excitation and emission wavelengths of 485 and 585 nm,
respectively.
Statistical analysis
Statistical analyses were performed using SPSS 19.0
software (IBM SPSS, Armonk, NY, USA). Data are expressed as the
mean ± standard deviation. The significance of differences between
groups were determined using Student’s t-test or analysis of
variance. P<0.05 was considered to indicate a statistically
significant difference. The synergistic or antagonistic effects of
the two drugs were evaluated, according to the formula [Q = Ea + b
/ (Ea + Eb - EaxEb)]. In this equation, Ea and Eb represent the
individual inhibitory rates of drugs A and B, respectively, and Ea
+ b represents the inhibitory rate of the combined drug therapy on
tumor cell proliferation. Q-values ranging between 0.85 and 1.15
indicated that the effects of the two drugs were simply additive,
whereas Q-values >1.15 indicated a synergistic effect, and
Q-values <0.85 indicated an antagonistic effect for the combined
drug therapy.
Results
Thiostrepton suppresses the viability of
Jurkat cells
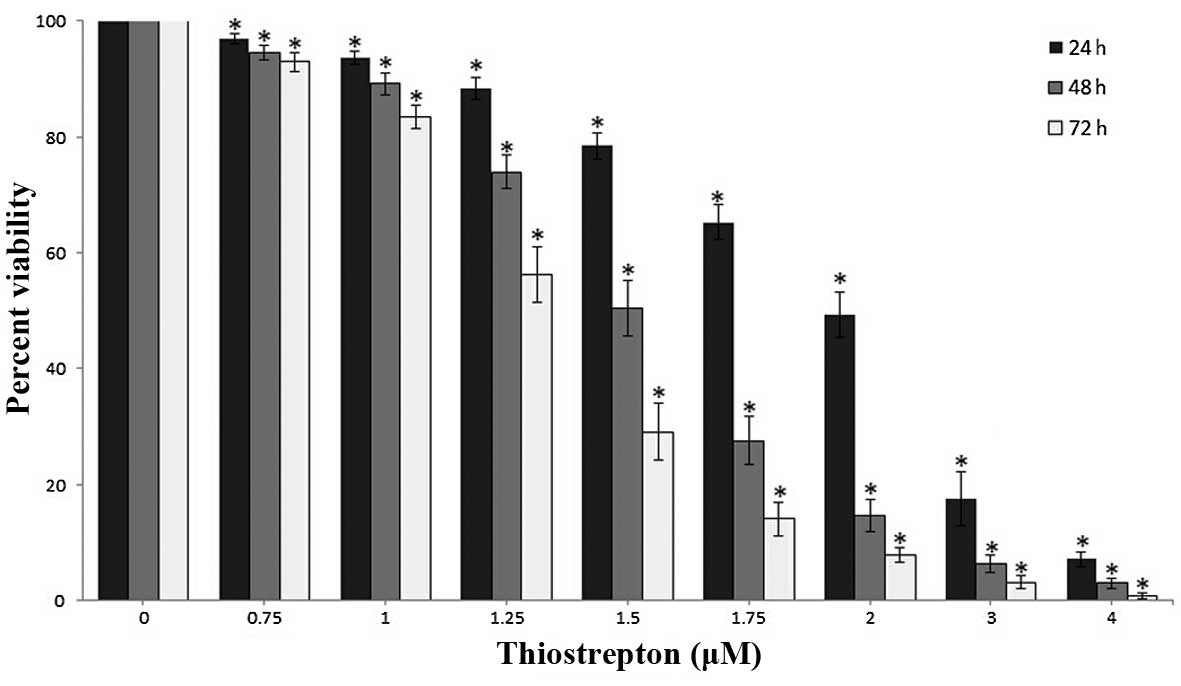
The Jurkat cells were treated with various
concentrations of thiostrepton (0–4 μM) for 24, 48 or 72 h,
and the effect of thiostrepton on cell viability was evaluated
using a CCK-8 assay. The results demonstrated that the Jurkat cells
were sensitive to thiostrepton, and that thiostrepton inhibited
cell growth in a time- and dose-dependent manner (P<0.05;
Fig. 1). The IC50
values for thiostrepton in the Jurkat cells at 24, 48 and 72 h were
2.03, 1.52 and 1.29 μM, respectively.
Thiostrepton-mediated inhibition of FoxM1
induces cell cycle arrest at the G2/M phase and leads to apoptosis
of Jurkat cells
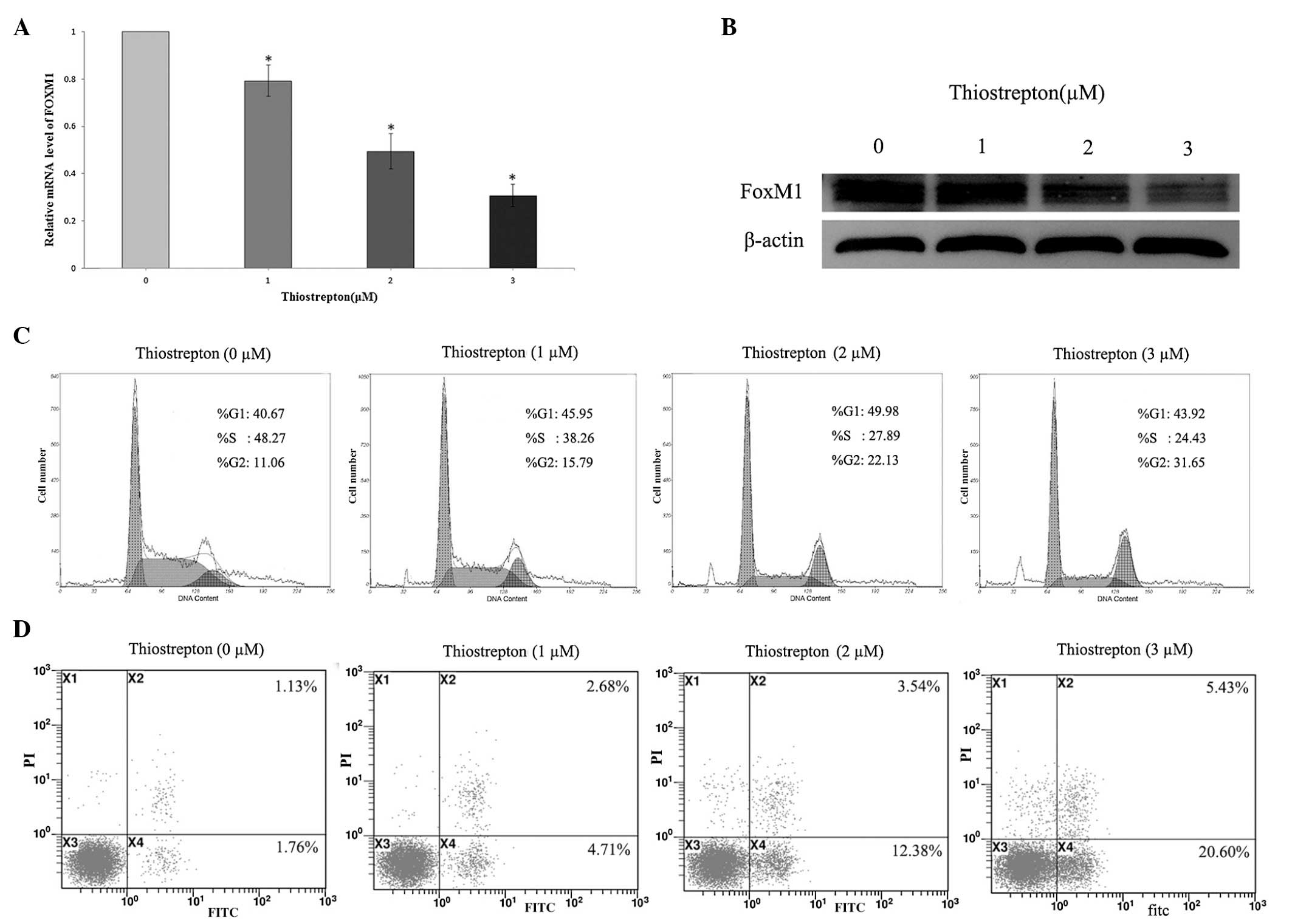
To further characterize the mechanism underlying the
antileukemia activity of thiostrepton on the Jurkat cells, the mRNA
and protein expression levels of FoxM1 were determined following
treatment with various concentrations of thiostrepton (0, 1, 2 or 3
μM) for 24 h, to examine whether thiostrepton affected cell
cycle distribution or apoptosis. The results revealed that
thiostrepton inhibited the expression of FoxM1 in a dose-dependent
manner, and induced gradual dose-dependent G2/M arrest and
apoptosis in the Jurkat cells (Fig.
2).
Inhibition of FoxM1 by thiostrepton
enhances the antiproliferative effects of doxorubicin on Jurkat
cells
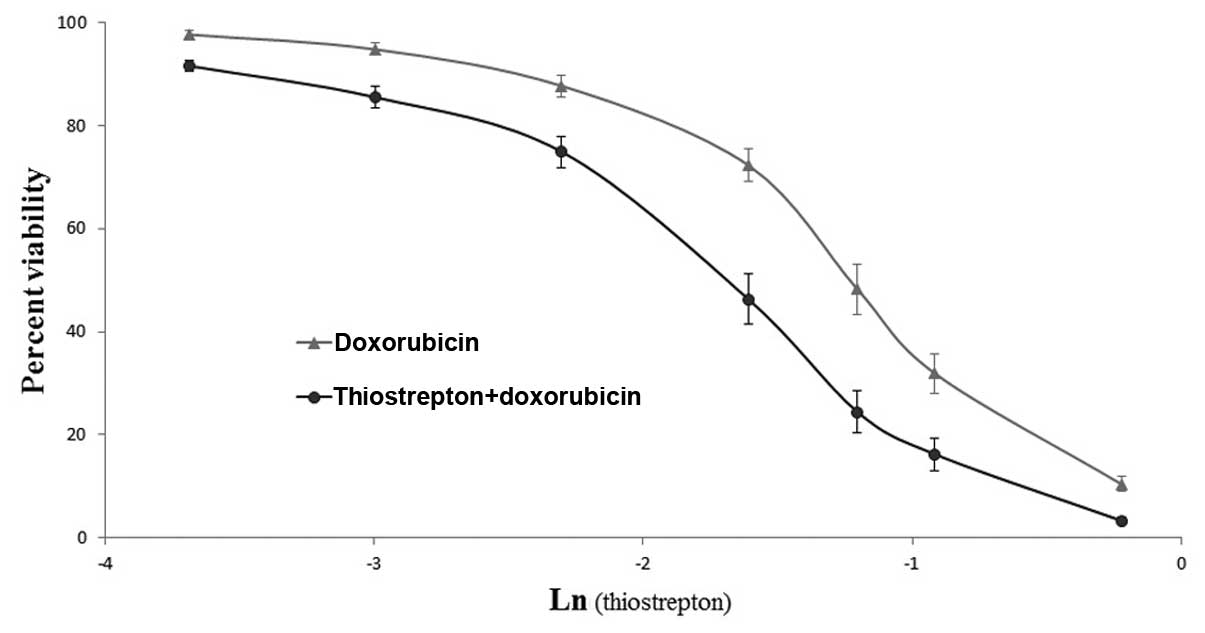
Based on the aforementioned findings, the present
study subsequently investigated whether the Jurkat cells with
reduced expression of FoxM1 were more sensitive to doxorubicin, a
common antileukemic drug used in the treatment of ALL. As shown in
Fig. 1, treatment with 1 μM
thiostrepton for 24 h resulted in a 7.70±0.95% reduction of viable
cells (P<0.05) compared with the control group, therefore, the
concentration of 1 μM was considered to be a non-cytotoxic
dose. The Jurkat cells were incubated with various concentrations
of doxorubicin (0–0.8 μm/l), with or without thiostrepton (1
μm/l), for 24 h. Cell proliferation was measured by
performing CCK-8 assays. The results demonstrated that the combined
treatment reduced the IC50 value of doxorubicin in the
Jurkat cells between 0.295 and 0.198 μmol/l (Fig. 3). Thiostrepton and doxorubicin were
identified to synergistically inhibit cell growth, with Q values
often >1.15 (Table II). It was
suggested that the inhibition of FoxM1 by thiostrepton increased
the chemosensitivity of Jurkat cells to doxorubicin.
 | Table IIInteraction between doxorubicin and
thiostrepton in combined treatment with different doses of
doxorubicin. |
Table II
Interaction between doxorubicin and
thiostrepton in combined treatment with different doses of
doxorubicin.
| Doxorubicin
(μM)a | Q-value |
|---|
| 0.025 | 0.86 |
| 0.05 | 1.17 |
| 0.1 | 1.32 |
| 0.2 | 1.61 |
| 0.3 | 1.43 |
| 0.4 | 1.25 |
| 0.8 | 1.07 |
Thiostrepton enhances doxorubicin-induced
caspase-mediated apoptosis in Jurkat cells
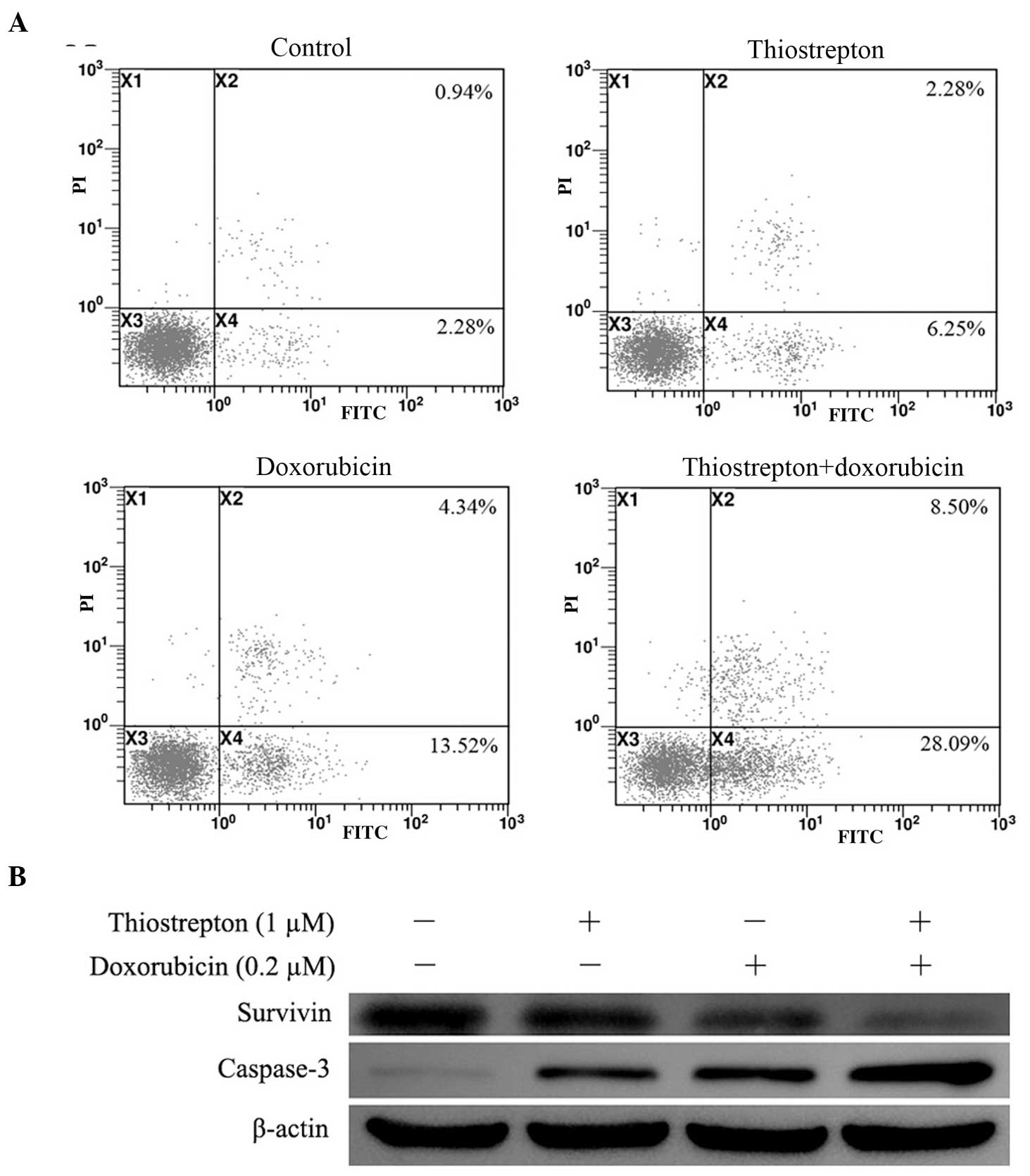
The present study then examined whether thiostrepton
enhances doxorubicin-induced cell apoptosis. The Jurkat cells were
treated with a low concentration of either thiostrepton (1
μm/l) or doxorubicin (0.2 μm/l), or treated with the
two drugs in combination for 24 h. Flow cytometric analysis
revealed that thiostrepton increased doxorubicin-induced apoptosis
between 16.98±2.87 and 36.07±4.18% (P<0.05; Fig. 4A). To determine the mechanism
leading to the cell apoptosis, the protein expression levels of
survivin and caspase-3 were examined. As shown in Fig. 4B, decreases in the expression of
survivin were significantly greater in the cells treated with
thiostrepton and doxorubicin in combination, compared with those in
the single treatment groups. In addition, combination treatment
resulted in greater increases in the protein expression of
caspase-3 compared with either drug alone (Fig. 4B).
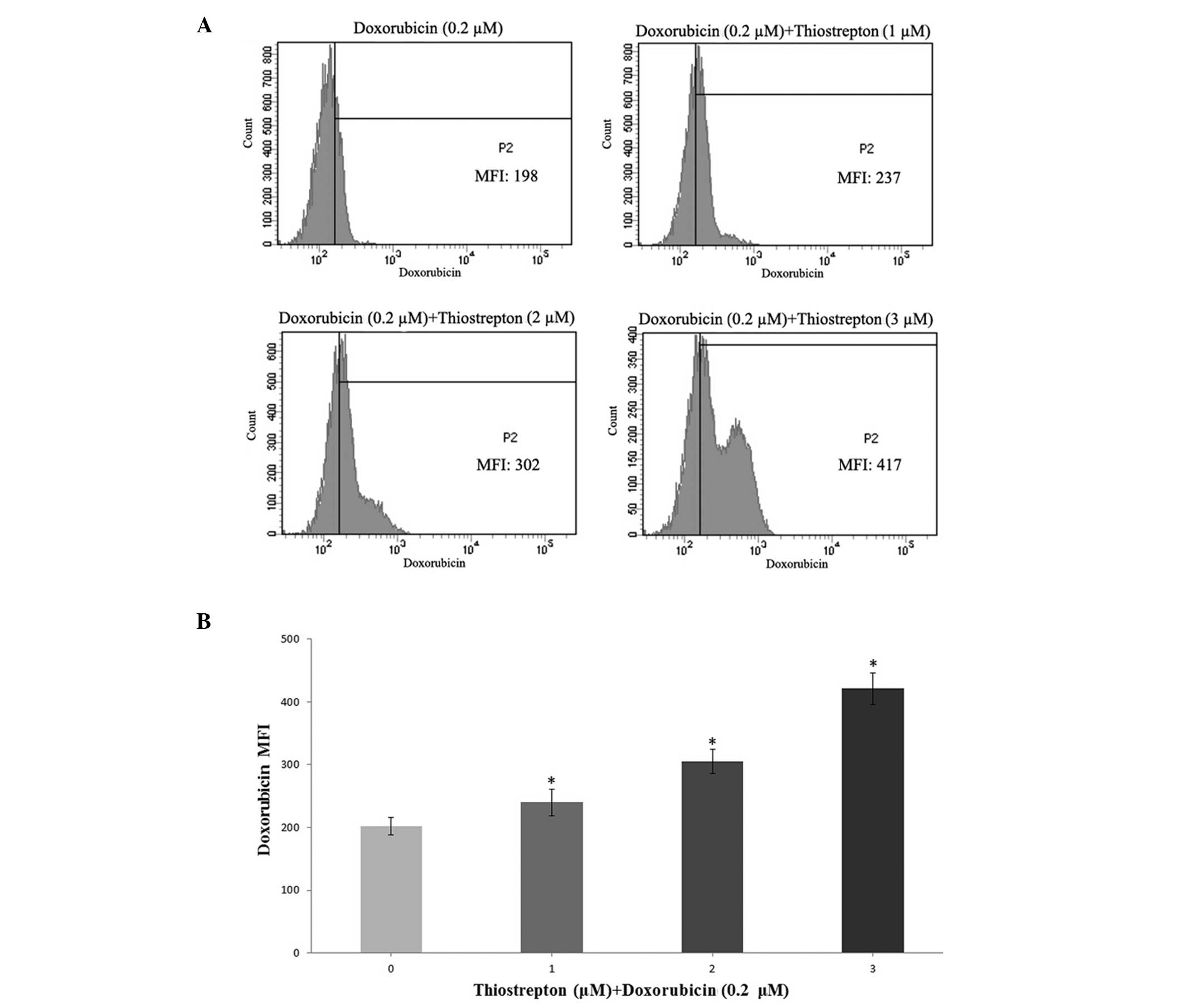
Thiostrepton increases the intracellular
accumulation of doxorubicin in Jurkat cells
The capability of thiostrepton to promote
doxorubicin accumulation within Jurkat cells was observed (Fig. 5). The doxorubicin-associated MFI
was significantly increased in Jurkat cells, which were treated
with various concentrations of thiostrepton (1, 2 or 3 μM)
for 24 h, which explained the enhanced effects of thiostrepton on
doxorubicin cytotoxicity in the Jurkat cells. In addition,
thiostrepton was observed to elevate the concentration of
doxorubicin in the Jurkat cells in a dose-dependent manner
(P<0.05).
Inhibition of FoxM1 by thiostrepton
decreases the gene and protein expression levels of GSTpi within
the Jurkat cells
To better understand the mechanism by which
thiostrepton functionally elevated drug accumulation within the
Jurkat cells, the mRNA and protein expression levels of GSTpi were
quantitatively measured. The Jurkat cells were treated with various
concentrations of thiostrepton (0, 1, 2 or 3 μM) for 24 h.
The gene and protein expression levels of GSTpi in the Jurkat cells
decreased significantly when the cells were treated with
thiostrepton (P<0.05; Fig. 6).
The results indicated that the inhibition of FoxM1 by thiostrepton
downregulated the expression of GSTpi in a dose-dependent manner in
the Jurkat cells (P<0.05).
Discussion
T-ALL is a genetically heterogeneous type of cancer,
with 20% of childhood patients and the majority of adult patients
succumbing to mortality from disease resistance or relapse
(27). Current standard therapies
have unsatisfactory effects on survival rates, and
therapy-associated side-effects have led to a focus on identifying
novel therapeutic approaches for T-ALL. The present study was the
first, to the best of our knowledge, to demonstrate
thiostrepton-induced inhibition of FoxM1 in human T-ALL Jurkat
cells. The results revealed that thiostrepton inhibited Jurkat cell
proliferation and induced apoptosis in a dose-dependent manner. It
was also demonstrated that thiostrepton enhanced the
chemo-sensitivity of the Jurkat cells to doxorubicin by enhancing
doxorubicin-induced apoptosis and increasing the intracellular
accumulation of doxorubicin.
The present study investigated the antileukemic
effects of thiostrepton on Jurkat cell proliferation. Thiostrepton
significantly inhibited the proliferation of Jurkat cells, and the
IC50 value of thiostrepton in the Jurkat cells following
48 h treatment was 1.52 μmol/l, suggesting the sensitivity
of the Jurkat cells to thiostrepton was comparable to that of other
types of cancer cell (28,29). Furthermore, the results of the
present study indicated that thiostrepton induced G2/M cell cycle
phase arrest and increased apoptosis in the Jurkat cells. These
results demonstrated the efficient antileukemic activity of
thiostrepton on Jurkat cells, and suggested that the effects of
thiostrepton may depend on the downregulation of FoxM1 target
genes, which have been found to be involved in the regulation of
cell growth, apoptosis, cell cycle and progression (30).
Previous studies have suggested that silencing FoxM1
enhances sensitivity to chemotherapeutic agents in various types of
cancer cells (14,29), therefore, the present study
investigated whether the thiostrepton-mediated inhibition of FoxM1
sensitized the Jurkat cells to the effects of chemotherapy. the
IC50 value of doxorubicin in the Jurkat cells decreased
between 0.295 and 0.198 μmol/l when thiostrepton (1
μmol/l) was used in combination. These results demonstrated
that thiostrepton enhanced the doxorubicin cytotoxicity effect
within the Jurkat cells through a synergistic effect of
thiostrepton and doxorubicin. Therefore, a combination therapy
approach using doxorubicin and thiostrepton can improve the effect
of doxorubicin and also reduce the adverse side-effects of
doxorubicin, by allowing it to be used at lower doses in treating
human T-ALL.
The ability to induce leukemic cell death is
critical for successful treatment with chemotherapeutic drugs. In
the present study, flow cytometric analysis revealed a more marked
increase in apoptosis in cells treated with a combination of
thiostrepton and doxorubicin, compared with those treated with
either drug alone. To further examine the mechanism underlying the
increased efficacy of combination treatment, proteins, which are
known to regulate apoptosis, were examined. Survivin is known for
its role in preventing apoptosis and conferring resistance to
chemotherapeutic agents in various cell lines (31). Specifically, its anti-apoptotic
function appears to be associated with the ability to directly or
indirectly inhibit caspases (31).
In the present study, the cells treated with thiostrepton and
doxorubicin combined exhibited a greater decrease in the protein
expression of survivin compared with the cells treatedwith
doxorubicin only, and thiostrepton also enhanced
doxorubicin-induced increases in the protein expression of
caspase-3. These results suggested that the inhibition of FoxM1 by
thiostrepton sensitized the Jurkat cells to doxorubicin, partly
through enhanced apoptosis, possibly via a caspase-3-dependent
pathway.
In the present study, thiostrepton significantly
elevated the concentration of doxorubicin in Jurkat cells in a
dose-dependent manner, while promoting doxorubicin accumulation
within the Jurkat cells. To determine the mechanism by which
thiostrepton functionally elevated drug accumulation within the
Jurkat cells, the expression of GSTpi was measured. Previous
studies have demonstrated that the mRNA and protein expression
levels of GSTpi are high in Jurkat cells (32). GSTpi is a cytosolic isoform of
glutathione s-transferases (GSTs) and its overexpression is
associated with resistance to chemotherapy in various types of
human cell (33). GSTs belong to a
large family of functional enzymes, which catalyze the
S-conjugation of glutathione (GSH) with a wide variety of
electrophilic compounds (34). In
addition to increasing GST enzyme activity, alteration of cellular
GSH levels has also been observed to contribute the resistance of
cells to doxorubicin (35). The
results of the present study further revealed that the mRNA and
protein expression levels of the GSTpi gene were significantly
inhibited by thiostrepton. These results indicated that
thiostrepton elevated the sensitivity of Jurkat cells to
doxorubicin, partly by increasing the accumulation of intracellular
doxorubicin, possibly via downregulating the expression of
GSTpi.
In conclusion, the results of the present study
demonstrated that thiostrepton significantly inhibited the
proliferation, and induced the apoptosis of Jurkat cells. The
inhibition of FoxM1 by thiostrepton also chemosensitized the Jurkat
cells to doxorubicin by enhancing doxorubicin-induced apoptosis and
elevating the accumulation of intracellular doxorubicin. These
results suggested that the inhibition of FoxM1 by thiostrepton have
wide therapeutic and/or adjuvant applications for T-ALL
chemotherapy. Thiostrepton may be particularly useful in young
children with resistant or relapsed T-ALL, who currently require
more aggressive chemotherapeutic agents.
Acknowledgments
This study was supported by the NCET-10-0919,
‘Taishan Scholar’ position, National Natural Science Foundation
(nos. 31371321 and 81200601), the Shandong Science and Technology
Committee (no. 2010GSF10264), the Foundation of Shandong
Educational Committee (nos. J10LC60 and J11LC01) and the Binzhou
Science and Technology Committee of China (no. 2011ZC0905).
References
|
1
|
Inaba H, Greaves M and Mullighan CG: Acute
lymphoblastic leukaemia. Lancet. 381:1943–1955. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bhojwani D and Pui CH: Relapsed childhood
acute lymphoblastic leukaemia. Lancet Oncol. 14:e205–217. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Van Vlierberghe P and Ferrando A: The
molecular basis of T cell acute lymphoblastic leukemia. J Clin
Invest. 122:3398–3406. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Martelli AM, Lonetti A, Buontempo F, et
al: Targeting signaling pathways in T-cell acute lymphoblastic
leukemia initiating cells. Adv Biol Regul. 56:6–21. Apr
30–2014.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wierstra I and Alves J: FOXM1, a typical
proliferation-associated transcription factor. Biol Chem.
388:1257–1274. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Koo CY, Muir KW and Lam EW: FOXM1: From
cancer initiation to progression and treatment. Biochim Biophys
Acta. 1819:28–37. 2012. View Article : Google Scholar
|
|
7
|
Priller M, Pöschl J, Abrão L, et al:
Expression of FoxM1 is required for the proliferation of
medulloblastoma cells and indicates worse survival of patients.
Clin Cancer Res. 17:6791–6801. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xue YJ, Xiao RH, Long DZ, et al:
Overexpression of FoxM1 is associated with tumor progression in
patients with clear cell renal cell carcinoma. J Transl Med.
10:2002012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xia JT, Wang H, Liang LJ, et al:
Overexpression of FOXM1 is associated with poor prognosis and
clinicopathologic stage of pancreatic ductal adenocarcinoma.
Pancreas. 41:629–635. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xu N, Jia D, Chen W, et al: FoxM1 is
associated with poor prognosis of non-small cell lung cancer
patients through promoting tumor metastasis. PLoS One.
8:e594122013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li X, Qiu W, Liu B, et al: Forkhead box
transcription factor 1 expression in gastric cancer: FOXM1 is a
poor prognostic factor and mediates resistance to docetaxel. J
Transl Med. 11:2042013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu LL, Zhang DH, Mao X, Zhang XH and
Zhang B: Over-expression of FoxM1 is associated with adverse
prognosis and FLT3-ITD in acute myeloid leukemia. Biochem Biophys
Res Commun. 446:280–285. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Myatt SS and Lam EW: The emerging roles of
forkhead box (Fox) proteins in cancer. Nat Rev Cancer. 7:847–859.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Halasi M and Gartel AL: Suppression of
FOXM1 sensitizes human cancer cells to cell death induced by
DNA-damage. PLoS One. 7:e317612012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nakamura S, Hirano I, Okinaka K, et al:
The FOXM1 transcriptional factor promotes the proliferation of
leukemia cells through modulation of cell cycle progression in
acute myeloid leukemia. Carcinogenesis. 31:2012–2021. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang X, Zeng J, Zhou M, et al: The tumor
suppressive role of miRNA-370 by targeting FoxM1 in acute myeloid
leukemia. Mol Cancer. 11:562012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mencalha AL, Binato R, Ferreira GM, Du
Rocher B and Abdelhay E: Forkhead box M1 (FoxM1) gene is a new
STAT3 transcriptional factor target and is essential for
proliferation, survival and DNA repair of K562 cell line. PloS one.
7:e481602012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bhat UG, Halasi M and Gartel AL: FoxM1 is
a general target for proteasome inhibitors. PLoS One. 4:e65932009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hegde NS, Sanders DA, Rodriguez R and
Balasubramanian S: The transcription factor FOXM1 is a cellular
target of the natural product thiostrepton. Nat Chem. 3:725–731.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hedge NS, Sanders DA, Rodriguez R and
Balasubramanian S: The transcription factor FOXM1 is a cellular
target of the natural product thiostrepton. Nat Chem. 3:725–731.
2011. View Article : Google Scholar
|
|
21
|
Gartel AL: Thiostrepton, proteasome
inhibitors and FOXM1. Cell Cycle. 10:4341–4342. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Halasi M, Schraufnagel DP and Gartel AL:
Wild-type p53 protects normal cells against apoptosis induced by
thiostrepton. Cell Cycle. 8:2850–2851. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ahmed M, Uddin S, Hussain AR, et al: FoxM1
and its association with matrix metalloproteinases (MMP) signaling
pathway in papillary thyroid carcinoma. J Clin Endocrinol Metab.
97:E1–13. 2012. View Article : Google Scholar
|
|
24
|
Wang M and Gartel AL: Micelle-encapsulated
thiostrepton as an effective nanomedicine for inhibiting tumor
growth and for suppressing FOXM1 in human xenografts. Mol Cancer
Ther. 10:2287–2297. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu X, Li X, Wang L, et al: Realgar
induces apoptosis in the chronic lymphocytic leukemia cell line
MEC1. Mol Med Rep. 8:1866–1870. 2013.PubMed/NCBI
|
|
26
|
Ji BS, He L and Liu GQ: Reversal of
p-glycoprotein-mediated multidrug resistance by CJX1, an amlodipine
derivative, in doxorubicin-resistant human myelogenous leukemia
(K562/DOX) cells. Life Sci. 77:2221–2232. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Van Vlierberghe P, A mbesi-I mpiombato A,
De Keersmaecker K, et al: Prognostic relevance of integrated
genetic profiling in adult T-cell acute lymphoblastic leukemia.
Blood. 122:74–82. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bhat UG, Halasi M and Gartel AL: Thiazole
antibiotics target FoxM1 and induce apoptosis in human cancer
cells. PLoS One. 4:e55922009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lin J, Zheng Y, Chen K, Huang Z, Wu X and
Zhang N: Inhibition of FOXM1 by thiostrepton sensitizes
medulloblastoma to the effects of chemotherapy. Oncol Rep.
30:1739–1744. 2013.PubMed/NCBI
|
|
30
|
Halasi M and Gartel AL: FOX(M1) news - it
is cancer. Mol Cancer Ther. 12:245–254. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zaffaroni N, Pennati M and Daidone MG:
Survivin as a target for new anticancer interventions. J Cell Mol
Med. 9:360–372. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou L, Jing Y, Styblo M, Chen Z and
Waxman S: Glutathione-S-transferase pi inhibits As2O3-induced
apoptosis in lymphoma cells: involvement of hydrogen peroxide
catabolism. Blood. 105:1198–1203. 2005. View Article : Google Scholar
|
|
33
|
Lourenco GJ, Lorand-Metze I, Delamain MT,
et al: Polymorphisms of glutathione S-transferase mu 1, theta 1 and
pi 1 genes and prognosis in Hodgkin lymphoma. Leuk Lymphoma.
51:2215–2221. 2010. View Article : Google Scholar
|
|
34
|
Ballerini S, Bellincampi L, Bernardini S,
Iori R, Cortese C and Federici G: Analysis of GSTP1–1 polymorphism
using real-time polymerase chain reaction. Clin Chim Acta.
329:127–132. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Song YN, Guo XL, Zheng BB, et al:
Ligustrazine derivate DLJ14 reduces multidrug resistance of
K562/A02 cells by modulating GSTπ activity. Toxicol In Vitro.
25:937–943. 2011. View Article : Google Scholar : PubMed/NCBI
|