Introduction
Translational research in radiation oncology is
important for the detection of adverse radiation effects, cellular
responses, and radiation modifications, and may help to improve the
outcome of radiation therapy in patients with cancer (1). The simultaneous administration of
chemotherapy and radiation therapy may enhance tumor cell
destruction, and protective agents [e.g. Prussian blue, calcium
diethylenetriamene pentaacetate (DTPA) and zinc DTPA, potassium
iodide and amifostine] (2) may
reduce acute inflammatory reactions and healthy tissue
complications (3). Further studies
on the optimization of the dosage and timing of the components of
such a binary therapeutic system (radiation and radiation
modifiers) are crucial to the development of novel clinical
therapeutic strategies (4).
Therefore, there is a requirement for novel in vitro models
that better reflect in vivo conditions, which may be
reliably transferred into clinical settings. To date, limited
information is available regarding changes in cellular
proliferation and viability over time, due to the cytotoxicity of
certain therapeutic agents, or to ionizing radiation. Novel
real-time cell analysis platforms allow the continuous detection of
cell viability during a defined period following anti-cancer
treatment (5).
Clonogenic, MTS, and lactate dehydrogenase (LDH)
assays are well-known end-point methods used for the evaluation of
chemoradiation and radiosensitivity.
The clonogenic assay is commonly used to investigate
the survival and proliferative capacity of irradiated cancer cells.
The clonogenic assay is used to measure the ability of cells to
grow in an anchorage-dependent manner. If the cells are able to
proliferate, they grow in cellular aggregates, referred to as
colonies. The limiting factor for the use of this method is the
length of time taken for a colony to form, which may take days or
weeks. The colonies are then quantified following crystal violet
staining (6).
The MTS assay is used to examine the
chemosensitivity or toxicity of drugs in human tumor cell lines,
and to quantify the survival of cancer cells following radiation
(7). The MTS assay is composed of
a cell-permeable, soluble tetrazolium salt, and an electron
coupling reagent (phenazine methosul-fate) that are added to the
cellular environment following appropriate dosing periods. During
incubation with MTS, the dehydrogenase enzymes present within the
mitochondria of healthy cells reduce the MTS to a dark purple,
water-soluble formazan compound, which may then be quantified
(8,9).
Another method of detecting cytotoxicity is the
assessment of membrane integrity, which is achieved by monitoring
the passage of substances that are normally sequestered inside
cells to the outside of the cells. During the LDH assay, the
release of LDH from the cytoplasm into the supernatant indicates
cell lysis (10).
Previous studies have used cell microelectronic
sensing to assess the real-time cytoprotective effects of novel
drug candidates (11), to study
the toxicity of various compounds in brain endothelial and
epithelial cells (12), and to
dynamically monitor the integrity of experimental biological
barriers (13–15).
The xCELLigence system, which was co-developed by
Roche (Budapest, Hungary) and ACEA Biosciences, Inc., (San Diego,
CA, USA) provides real-time continuous monitoring and label-free
assessment of cellular adhesion, morphological changes,
proliferation, viability, and cytotoxicity, thereby revealing the
physiological state of cells. The system consists of four main
components: A Real-Time Cell Analyzer (RTCA), an RTCA Double Plate
station, an RTCA computer with integrated software, and microtiter
plates (E-plate 16). The core of the system is composed of
disposable microelectronic cell sensor arrays integrated into the
bottom of the E-plates, which perform cell-based assays on the RTCA
instrument. The size of the E-plate 16 corresponds to 1/6 of the
size of a 96-well microtiter plate, with 16 wells contained in two
side by side columns (16,17). The electronic impedance of the
electrode sensors is measured in order to allow the monitoring and
detection of any alterations in cellular attachment (18,19).
The impedance measured between the electrodes in an individual well
depends on electrode geometry, the ionic concentration of the well,
and whether the cells are attached to the electrodes. In the
absence of cells, electrode impedance is predominantly determined
by the ionic environment, both at the electrode/solution interface,
and in the bulk solution. In the presence of cells, the cells
attached to the electrode sensor surface act as insulators, thereby
altering the local ionic environment at the electrode/solution
interface, leading to an increase in impedance. The more cells that
have grown on the electrodes, the larger the measured electrode
impedance. The impedance measurement, which is displayed as a cell
index (CI), provides quantitative information regarding the
biological status of the cells, including cell number, viability,
and morphology. Calculation of the cell index is based on the
following formula: CI = (Zi - Z0)/15, where
Zi is the impedance at an individual time point during
the experiment, and Z0 is the impedance at the start of
the experiment. Therefore, the CI is a self-calibrated value
derived from the ratio of impedance. Impedance-based monitoring of
cell proliferation and viability correlates accurately with cell
number (20,21). The present study used a 16-well
plate system that is able to simultaneously measure three
independent 16-well plates. This allowed the simultaneous
investigation of two doses of irradiation, together with
non-irradiated control cells.
The present study presents the advantages and the
potential uses of the novel xCELLigence biosensor system for the
measurement of the effects of ionizing radiation on tumor and
primary cells, as compared with alternative cell-based assays.
Materials and methods
Cell lines and culture conditions
U251 human glioblastoma, A549 human lung carcinoma,
MCF7 human breast adenocarcinoma, HT-29 human colon adenocarcinoma
and human primary fibroblast cells (passage 4) were all purchased
from the ATCC (Wesel, Germany), and stored at-80°C in a solution
containing 90% fetal bovine serum (FBS; Gibco Life Technologies,
Budapest, Hungary) and 10% dimethyl sulf-oxide (DMSO; Molar
Chemicals Kft., Budapest, Hungary). GBM2 human glioblastoma cells
were obtained from Dr Balázs Hegedűs (National Oncology Institute,
Budapest, Hungary). The human primary fibroblast cells were
cultured in low glucose Dulbecco's modified Eagle's medium (DMEM;
Lonza, Budapest, Hungary); the A549, MCF7, HT-29, and GBM2 cells
were cultured in DMEM-F12 (Lonza); and the U251 cells were cultured
in RPMI-1640 medium (Lonza) supplemented with 10% heat-inactivated
FBS (Gibco Life Technologies), 1% L-glutamine, 100 U/ml penicillin,
and 100µg/ml streptomycin (Sigma-Aldrich, Budapest, Hungary) at
37°C in a humidified atmosphere containing 5% CO2.
Irradiation
A Teragam K-01 cobalt unit (SKODA-ÚJP, Prague, Czech
Republic) was used (average energy 1.25 MeV; source/image distance,
80 cm) to irradiate the cells seeded into 16-well E-plates (Roche),
and 96-well and 6-well normal tissue culture plates. The plates
were surrounded by water on either side and placed between two 2 cm
polymethyl methacrylate slabs to ensure adequate material build-up.
The isocentre was positioned at the geometrical centers of the
plates. Half of the radiation dose was delivered with a downward
20×20 cm beam (gantry angle, 0°), whereas the other half of the
radiation dose was delivered with an upward 20×20 cm beam (gantry
angle, 180°), in order to maximize field homogeneity. The delivered
doses were 0, 5, and 10 Gy. Due to the decay of
Cobalt60, irradiation time correction factors were
applied.
Clonogenic survival assay
For irradiation experiments, the cells were grown in
75 cm2 flasks, until they reached 75% confluence. The
cells were then harvested by trypsinization (Sigma-Aldrich Chemie
GmbH, Schnelldorf, Germany), and counted using Bürker chambers. The
cells were subsequently seeded at 1,000 cells/well into 6-well
plates, and allowed to adhere for 24 h. All clonogenic assay
experiments were performed in triplicate. During 8 days, the
irradiation medium was changed every 2 days. The medium was then
discarded and the cells were washed with phosphate-buffered saline
(PBS) prior to fixation with 4% paraformaldehyde. Colony forming
units (CFUs) were stained with a solution containing 0.5% crystal
violet (Sigma-Aldrich Chemie GmbH) for 20 min. The plates were
rinsed three times with tap water and left to dry at room
temperature. Colony counting was conducted the following day using
the Zeiss Axiovert 25 microscope (Zeiss, Oberkochen, Germany), with
the number of CFUs expressed as a percentage, as compared with the
control samples, which were considered 100%.
MTS cell viability assay
The viability of each cell line was determined using
a colorimetric MTS assay. The cells were seeded into 96-well tissue
culture plates at a density of 500, 1,000, 2,000, or 4,000
cells/well, and cultured overnight prior to irradiation. Three days
following irradiation, 20 µl MTS (Gibco Life Technologies)
was added at a working concentration of 0.3 mg/ml, and incubated
for ≥1 h at 37°C, in an atmosphere containing 5% CO2.
Following incubation, the absorbance was measured using a Wallac
1420 VICTOR2™ ELISA assay plate reader (Wallac VICTOR Plate Reader;
PerkinElmer, Inc., Waltham, MA, USA) at a wavelength of 490 nm,
with the values expressed in arbitrary units. All MTS cell
viability experiments were performed in triplicate.
Lactate dehydrogenase (LDH) assay
A Cytotoxicity Detection kit (Roche) was used to
assess cell viability following irradiation. An LDH assay measures
the activity levels of LDH in the supernatant released from damaged
cells, or the activity levels of total LDH following cell lysis.
The former correlates with the number of dead cells, whereas the
latter correlates with the number of viable cells. For the present
study, given the low cell numbers, the release assay did not
provide sufficient information, likely due to the relatively low
levels of cell apoptosis, as well as the low sensitivity of the
assay. Therefore, the total LDH activity level detection method was
used following cell lysis. The cells were seeded at four densities:
500, 1,000, 2,000, and 4,000 cells/well. Three days following
irradiation, 70 µl supernatant was removed from the wells
and plated into a 96-well plate. The remaining medium was
discarded. The cells were then washed with PBS, and 70 µl
PBS containing 1% Triton X-100 (Sigma-Aldrich Chemie GmbH) was
added to the cells to achieve total cell lysis. Following 15 min
incubation at room temperature, 70 µl LDH reagent was added
to both the lysed cells and the wells containing the supernatant,
and absorbance at 490 nm was recorded at 2 min intervals for a
duration of 20 min.
Cell growth and proliferation assay using
xCELLigence
The cells were grown in 100 mm Petri dishes until
they had reached 90% confluence; the cells were subsequently
harvested by trypsinization, and seeded into 16-well E-plates at
various densities in 100 ml medium (500, 1,000, 2,000, 4,000
cells/well). Following seeding, the cells were monitored every 10
min by the xCELLigence system (Roche) for proliferation,
attachment, and spreading. Following 24 h, during which time the
cells entered the exponential growth phase, the cells were
irradiated with 5 and 10 Gy doses, and impedance detection via the
incorporated sensor electrode arrays continued for a further 76 h.
RTCA Software 2.0 (Roche) was used to calculate the CI values. The
CI values were then converted to a percentage 72 h
post-irradiation, with the 0 Gy wells being considered 100%.
Apoptosis detection
A total of 5×104 cells were plated onto
24-well tissue culture plates in triplicate for each time point. To
detect phosphatidylserine exposure on the outer cell membrane, the
cells were irradiated with 0, 5, and 10 Gy as indicated. Following
24, 48 and 72 h the supernatant was collected and mixed with the
washed, trypsinized cells, prior to incubation with Annexin V
binding buffer containing 0.01 mM HEPES, 0.14 mM NaCl and 2.5 mM
CaCl2. Annexin V-Alexa 488 (2.5:100) (Gibco Life
Technologies) and 10 µg/ml propidium iodide (PI)
(Sigma-Aldrich Chemie GmbH) were added to the cells and incubated
for 15 min in the dark, at room temperature. Following washing, the
cells were analyzed using a FACSCalibur cytofluorimeter (BD
Biosciences, San Jose, CA, USA). Annexin V-Alexa 488 was used to
determine the percentage of positive cells (FLI), and PI was used
to determine the percentage of negative early apoptotic cells
(FL3).
Statistical analysis
Statistical differences between groups were assessed
with a paired Student's t-test, as analyzed using Windows Excel
(Microsoft Corporation, Redmond, WA, USA). P<0.05 was considered
to indicate a statistically significant difference (Table I). The data are presented as the
mean ± standard deviation.
 | Table IStatistically significant differences
measured between untreated and irradiated cells. |
Table I
Statistically significant differences
measured between untreated and irradiated cells.
| Cell density | GBM2
| MCF7
| A549
| HT29
| U251
| Human fibroblast
cells
|
|---|
| 5 Gy | 10 Gy | 5 Gy | 10 Gy | 5 Gy | 10 Gy | 5 Gy | 10 Gy | 5 Gy | 10 Gy | 5 Gy | 10 Gy |
|---|
| Clonogenic
assay | | | | | | | | | | | | |
| 1,000 | 1.64E-05 | 3.76E-06 | 1.01E-12 | 5.84E-15 | 4.02E-11 | 2.3IE-13 | 4.97E-09 | 9.47E-11 | 6.91E-13 | 1.27E-14 | | |
| xCELLigence | | | | | | | | | | | | |
| 500 | 1.58E-04 | 1.63E-06 | 4.06E-05 | 2.03E-05 | 3.95E-04 | 1.47E-04 | 3.73E-01 | 3.21E-01 | * | * | 4.02E-02 | 5.20E-04 |
| 1,000 | 2.39E-04 | 3.97E-12 | 1.27E-05 | 2.11E-05 | 3.32E-06 | 8.99E-07 | 1.89E-03 | 1.06E-05 | 2.28E-03 | 2.74E-04 | 9.04E-01 | 9.52E-03 |
| 2,000 | 5.83E-02 | 7.98E-05 | 1.17E-05 | 1.22E-05 | 7.87E-04 | 2.48E-05 | 8.00E-02 | 9.17E-03 | 2.92E-02 | 3.33E-03 | 1.70E-01 | 9.95E-02 |
| 4,000 | * | * | 1.22E-08 | 1.14E-08 | 1.66E-01 | 8.33E-03 | 4.61E-03 | 6.67E-01 | 3.44E-01 | 1.57E-04 | 3.42E-01 | 1.19E-01 |
| MTS assay | | | | | | | | | | | | |
| 500 | 3.19E-05 | 1.11E-05 | 1.80E-04 | 4.24E-06 | 1.14E-04 | 6.63E-06 | 4.69E-05 | 2.09E-06 | 1.08E-03 | 2.98E-06 | 1.21E-03 | 1.75E-02 |
| 1,000 | 2.31E-04 | 4.00E-06 | 5.28E-05 | 4.34E-05 | 4.29E-06 | 3.40E-07 | 2.29E-04 | 3.26E-05 | 2.12E-04 | 2.02E-05 | 4.88E-03 | 1.97E-02 |
| 2,000 | 5.78E-06 | 2.01E-06 | 3.94E-05 | 2.00E-05 | 1.52E-03 | 1.39E-03 | 4.34E-06 | 1.33E-07 | 3.12E-01 | 4.92E-04 | 7.26E-03 | 2.27E-02 |
| 4,000 | 7.01E-02 | 2.75E-02 | 1.13E-05 | 1.49E-05 | 2.32E-04 | 7.01E-02 | 1.31E-06 | 3.10E-06 | 3.50E-04 | 1.18E-03 | 4.24E-02 | 6.70E-01 |
| LDH assay | | | | | | | | | | | | |
| 500 | 9.41E-03 | 2.00E-04 | 2.45E-03 | 5.69E-04 | 1.55E-02 | 9.95E-05 | 3.42E-04 | 2.65E-05 | 4.45E-01 | 6.14E-03 | 5.45E-02 | 1.32E-01 |
| 1,000 | 3.21E-03 | 3.24E-05 | 4.97E-08 | 4.52E-07 | 7.44E-03 | 4.20E-03 | 2.75E-05 | 1.02E-06 | 4.12E-01 | 5.69E-06 | 6.36E-01 | 9.46E-01 |
| 2,000 | 1.74E-03 | 1.98E-05 | 3.55E-07 | 2.21E-07 | 9.16E-02 | 9.54E-04 | 1.85E-03 | 4.39E-04 | 4.08E-02 | 1.91E-02 | 8.96E-01 | 1.00E-01 |
| 4,000 | 3.15E-02 | 6.18E-03 | 2.33E-03 | 1.94E-07 | 5.30E-01 | 8.35E-04 | 3.46E-03 | 5.70E-03 | 1.11E-04 | 3.61E-05 | 6.53E-01 | 4.91E-01 |
| FACS analysis | | | | | | | | | | | | |
| 50,000 | 2.35E-03 | 1.31E-02 | 1.75E-05 | 5.39E-06 | 1.09E-02 | 4.35E-03 | 1.29E-02 | 2.66E-03 | 2.03E-01 | 1.81E-03 | 1.60E-02 | 5.04E-02 |
Results
The present study evaluated the effects of ionizing
radiation on a wide spectrum of malignant human cell lines and
human primary fibroblast cells. A systematic comparison of
end-point and real-time assays was carried out in order to reveal
the advantages of the xCELLigence system.
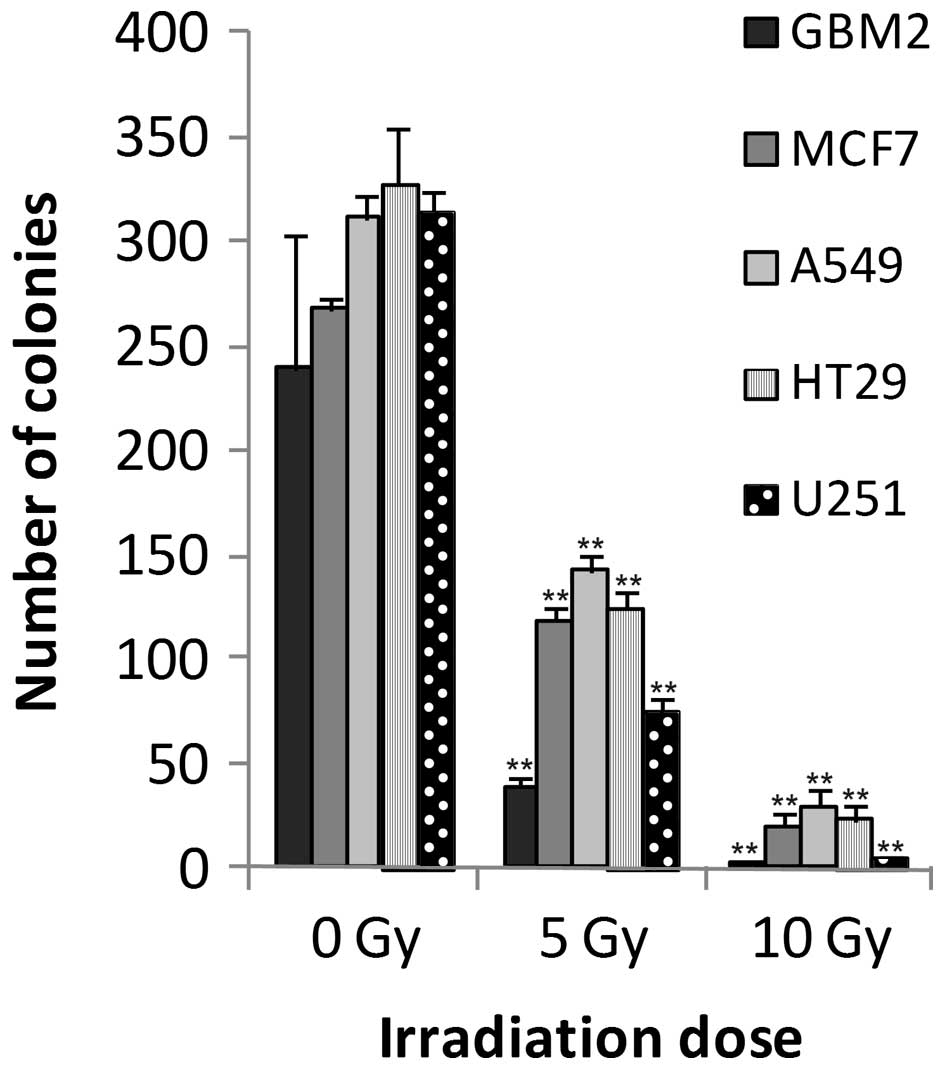
GBM2 human glioma cells
The colony forming assay served as a reference to
assess the effects of radiation on cell growth. A strong
correlation was detected between irradiation and colony formation.
Significant differences (P<0.05) were observed between the
control cells and those exposed to 5 or 10 Gy irradiation doses.
The number of colonies linearly decreased with increasing doses of
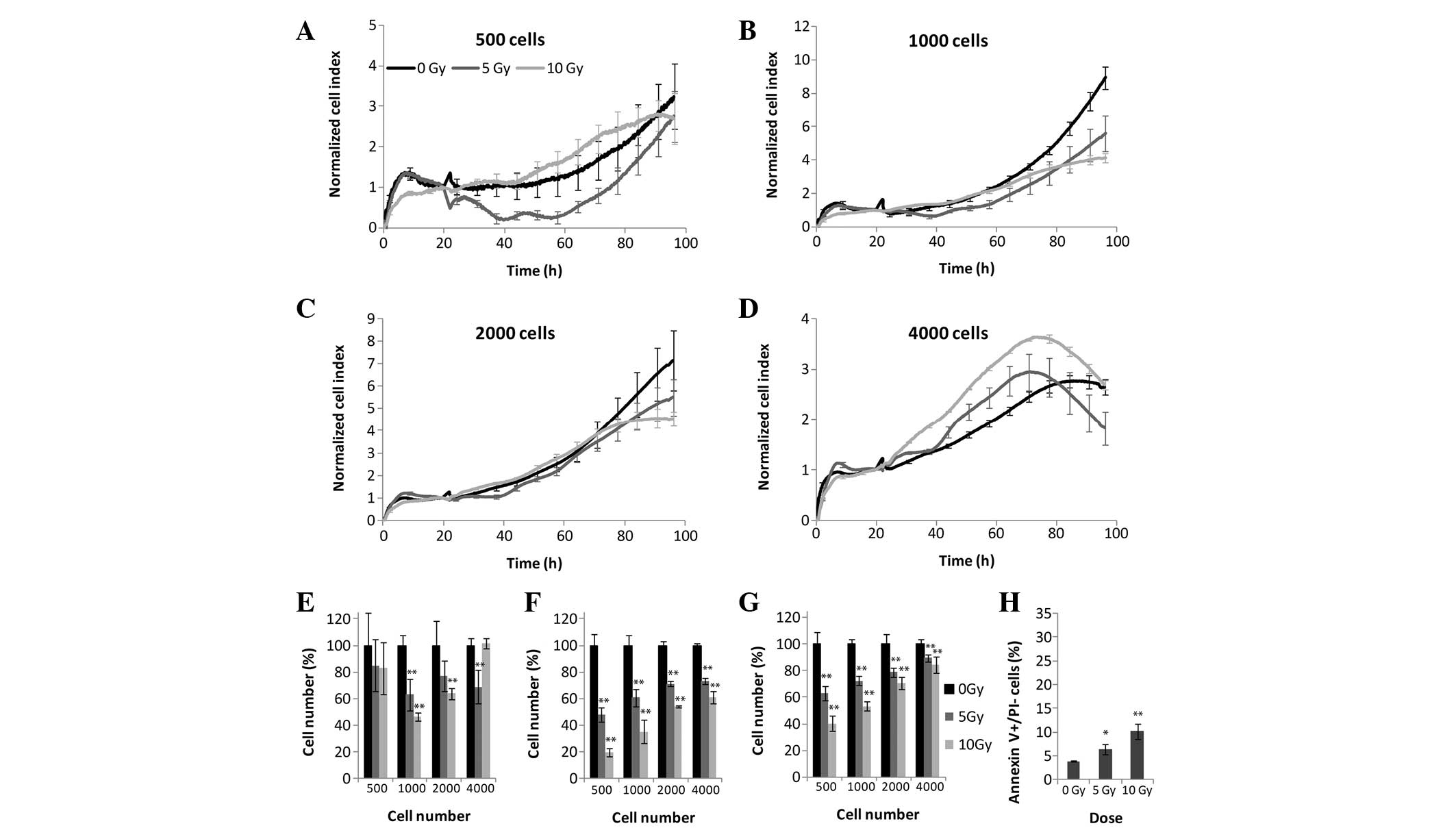
irradiation (Fig. 1). Four cell
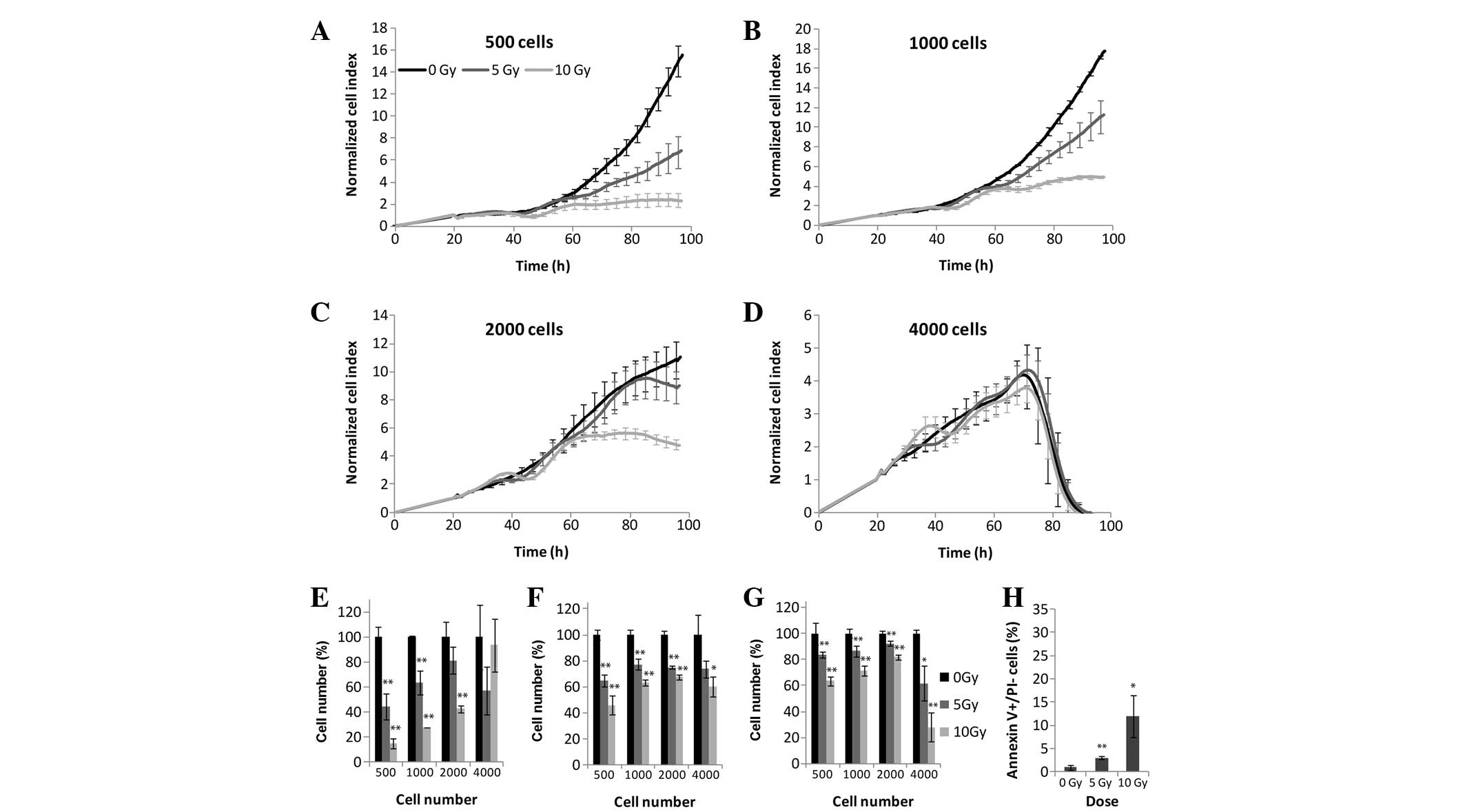
densities were tested using the RTCA (Fig. 2A–E), MTS (Fig. 2F), and LDH (Fig. 2G) assays. The effects of
irradiation were detected using the label-free method <24 h
following treatment. At the highest cell density, (4,000
cells/well), the cells were overgrown in both the untreated and
irradiated samples 50 h post-irradiation (Fig. 2D). Cellular exponential growth was
arrested due to irradiation at 24 h which was recorded as a small
plateau at 40–60 h in the cells plated at 500 and 1,000 cells/well
treated with 5 and 10 Gy irradiation. Following CI curve
normalization (the CI values were scaled so that they were
independent of initial cell densities) the strongest effects of
irradiation were observed at the two lowest cell densities
(Fig. 2A and B). Following 5 Gy
irradiation, a significant decrease in cellular proliferation was
detected, whereas at 10 Gy irradiation cellular proliferation
ceased 60 h post-irradiation. Upon comparison of the results
obtained from the xCELLigence system and the end-point assays, it
was apparent that both the LDH and MTS assays (Fig. 2F and G) detected a decrease in the
overall enzymatic activity at 500, 1,000 and 2,000 cells/well at 72
h; however, this apparent decrease in enzymatic activity was
significantly lower, as compared with the impedance measured by the
xCELLigence system (Fig. 2E). The
end-point assays revealed that the effects of irradiation decreased
with the increasing number of seeded cells, except for a density of
4,000 cells/well, as this cell density was too high for impedance
to occur. The percentage of early apoptotic cells was detected by
Annexin V binding to phosphatidylserine exposed on the outer
membrane. The percentage of apoptotic cells was correlated with the
dose of irradiation, and following 10 Gy 12% of cells exhibited
early apoptosis (Fig. 2H).
Discrepancies in the ratios of all parameters measured by each
assay are due to differences in the measured biological
targets.
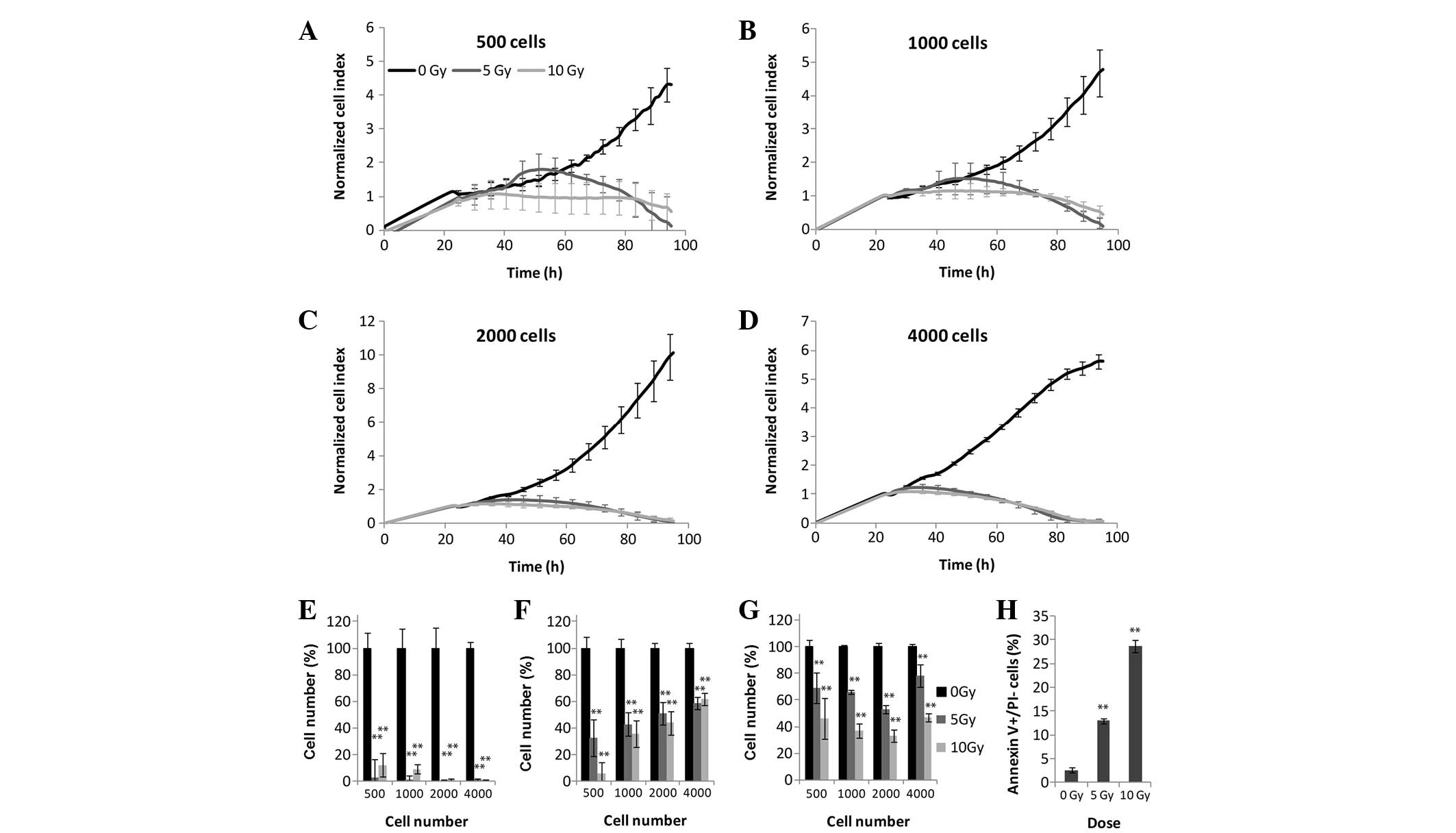
MCF7 human breast adenocarcinoma
cells
The results of the colony forming assay revealed
significant differences between the 5 and 10 Gy irradiated cells,
and the control cells (Fig. 1). In
the case of the MTS and LDH assays, all four tested cell densities
provided results that correlated with these of the xCELLigence
system (Fig. 3). The MCF7 cells
were the most radiosensitive of all the experimental cells.
Following 5 and 10 Gy irradiation, the CI values decreased, and
after 3 days all CI values returned to baseline levels suggesting
the presence of inhibited cell division and/or marked alterations
in cell attachment and shape (Fig.
3A-E). MTS (Fig. 3F) and LDH
(Fig. 3G) assays demonstrated a
marked decrease in the metabolic activity of the cells at all
densities, following 72 h irradiation. Since the MCF7 cells
exhibited high radiosensitivity, ~30% of the MCF7 cells underwent
early apoptosis following 72 h 10 Gy irradiation (Fig. 3H).
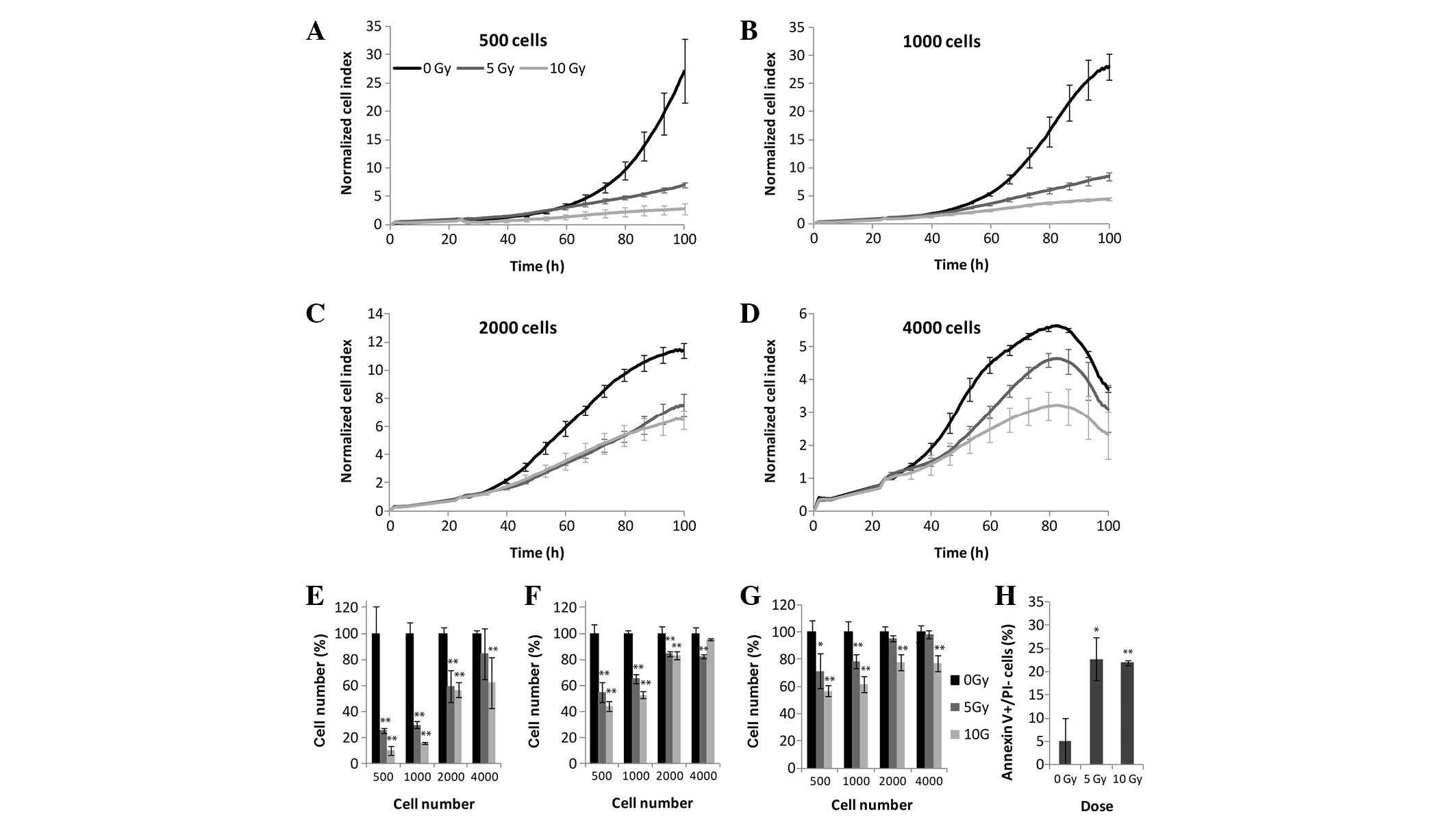
A549 human lung carcinoma cells
A strong correlation was detected between
irradiation dosage and colony formation. Significant differences
(P<0.05) were observed between the control cells and those
exposed to 5 or 10 Gy irradiation (Fig. 1). Using RTCA, MTS and LDH assays,
the effects of irradiation could be detected at all four
experimental cell densities (Fig.
4). Seeding densities of 500 or 1,000 cells/well proved to be
optimal with highly distinguishable growth curves, with the rate of
cellular proliferation markedly decreasing following irradiation,
in a dose-dependent manner (Fig. 4A, B
and E). Seeding densities of 2,000 and 4,000 cells/well
resulted in cell overgrowth over the course of 3 days (Fig. 4C, D and E). Although the most
pronounced changes were apparent with the xCELLigence system, the
effects of irradiation were also shown to decrease with increasing
cell density in the MTS (Fig. 4F)
and LDH (Fig. 4G) assays. The
curves generated following 5 and 10 Gy irradiation were
indistinguishable, exhibiting similar apoptotic rates of 22 and
22.8%, respectively (Fig. 4H).
HT-29 human colorectal adenocarcinoma
cells
In a similar manner to the other cell lines,
decreases in HT-29 human colorectal adenocarcinoma cell colony
growth were also dose-dependent, exhibiting statistically
significant differences between the control and irradiated groups
(Fig. 1). All HT-29 cell densities
were resistant to all ionizing irradiation doses in terms of cell
division and/or changes in morphology, as determined by the
xCELLigence system (Fig. 5A–E).
Lower cell densities of 1,000 and 2,000 cells/well exposed to a 10
Gy irradiation (Fig. 5A, B and E)
exhibited growth inhibition and/or detachment, results which were
concordant with the results from both the MTS and LDH assays.
However, in the MTS (Fig. 5F) and
LDH (Fig. 5G) assays, all cell
densities showed reduced enzymatic activity due to ionizing
radiation. As HT-29 cells showed low sensitivity to the 5 and 10 Gy
irradiation treatments, only 6 and 10% of the cells were Annexin
V-positive, respectively (Fig.
5H).
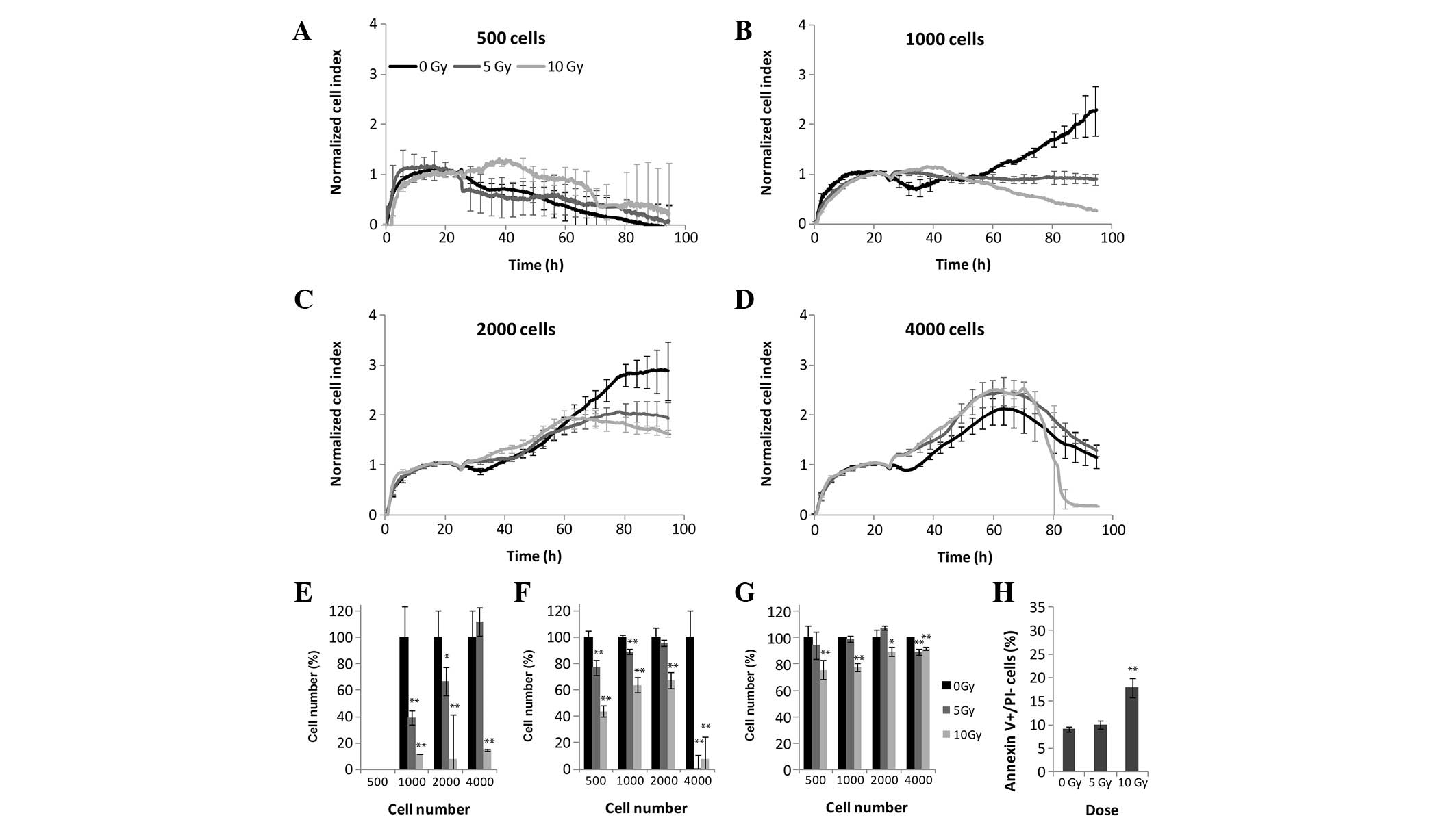
U251 human glioblastoma cells
Colony forming capacity was reduced in a
dose-dependent manner following irradiation (Fig. 1). A cell density of 500 cells/well
did not present sufficient signal throughout the experiment
(Fig. 6A), whereas at a cell
density of 4,000 cells/well, the cells became overgrown in 2 days
in both the control and irradiated samples (Fig. 6D). The results obtained from the
RTCA system were significant at cell densities of 1,000 and 2,000
cells/well. At 1,000 cells/well 5 Gy irradiation dose arrested cell
proliferation and/or influenced cell attachment, whereas
irradiation with 10 Gy led to a decrease in CI values following 48
h (Fig. 6B and E). A
dose-dependent decrease in the CI values was also detectable at a
cellular density of 2,000 cells/well, although the decrease was
less pronounced than at 1,000 cells/well (Fig. 6C). The MTS and LDH assays showed a
significant decrease in the activity levels of dehydrogenases 72 h
after irradiation, although these results were only observable in
the LDH assay at a 500 cell/well density (Fig. 6F and G). The U251 glioblastoma cell
line was moderately sensitive to γ-irradiation, with an apoptotic
cell death of 18% following 10 Gy irradiation (Fig. 6H).
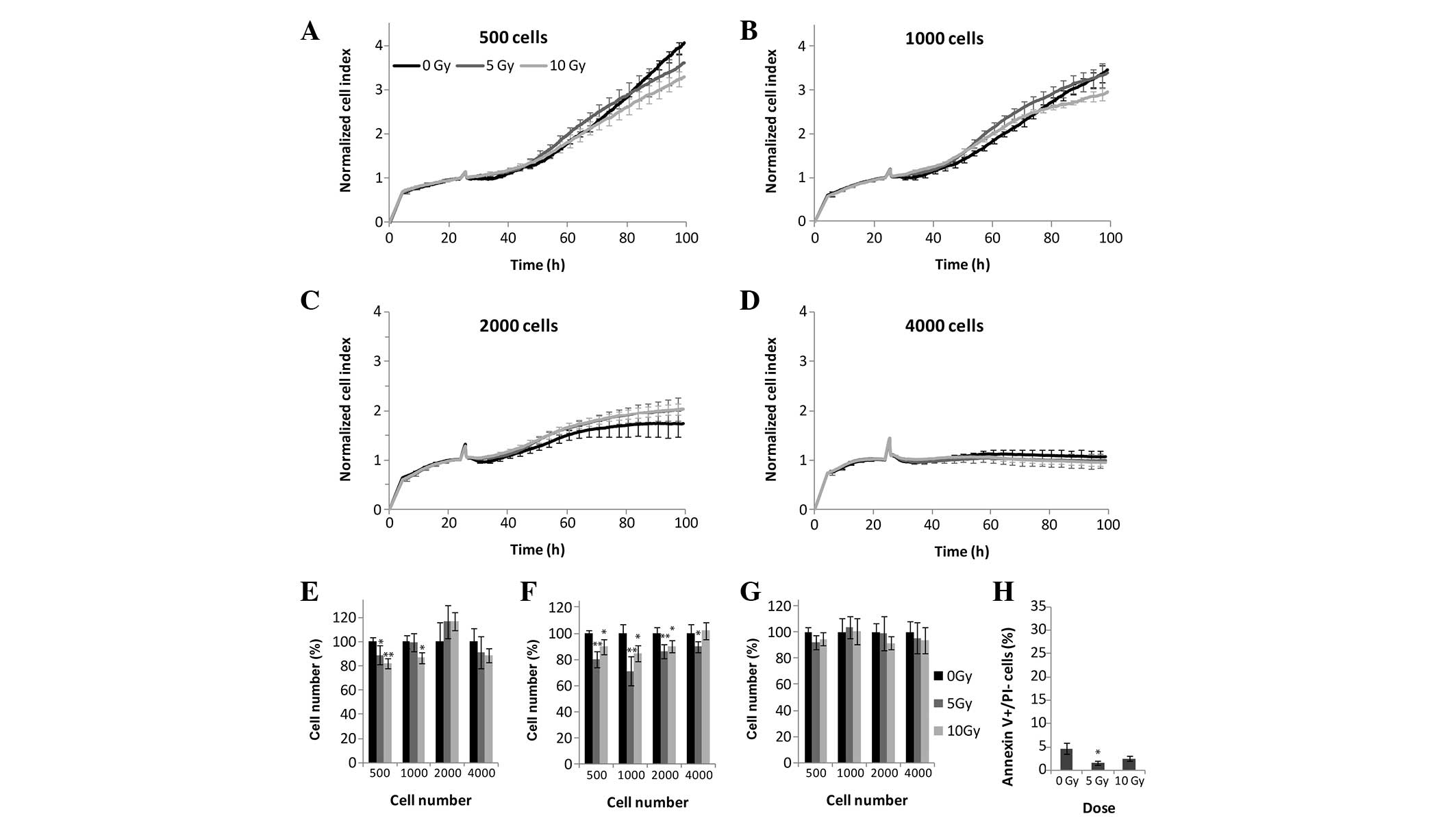
Human primary fibroblasts
Human primary fibroblasts are unable to form
colonies, and were therefore not examined in the colony forming
assay. In the RTCA, MTS, and LDH assays, the fibroblasts were more
resistant to ionizing radiation, as compared with the experimental
malignant cell lines, even following 10 Gy irradiation, following
which cellular proliferation was comparable to that of the control
cells (Fig. 7A–G). Similarly to
the malignant cell lines, xCELLigence analysis of the fibroblasts
showed sensitivity to cell density, with 500 and 1,000 cells/well
being the optimal density, whereas higher cellular densities
saturated the CI signals prior to the end of the assay (Fig. 7A–D). Concordant with the
above-mentioned methods, the human primary fibroblast slowly
dividing cells were resistant to both 5 and 10 Gy irradiation
doses, with 1.6 and 2.5% early apoptotic cells, respectively
(Fig. 7H).
Discussion
The study of radiotherapy began in 1896 with the
discovery of X-rays by Wilhelm Röntgen. Thereafter, radiation
became one of the primary strategies for the treatment of numerous
malignant tumors, through the induction of DNA damage (22). The use of radiation as a treatment
is based on reports of radiation-induced inhibition of cellular
proliferation, induction of apoptotic cell death, and inhibition of
tumor growth, due to the fact that rapidly dividing malignant cells
are more radiosensitive, as compared with normal tissue cellular
components (23). Cell survival
following radiation is usually measured using end-point assays, and
clonogenic and mitochondrial dehydrogenase activity assays, such as
MTS, are the most widely-used methods to carry out such research.
However, the limitations of end-point assays include the time taken
for colonies to form, and the inability to measure proliferation of
primary cells, which do not grow as colonies. The MTS assay
quantifies metabolically viable cells by measuring their ability to
reduce a tetrazolium dye. The present study used numerous
additional methods of cell viability detection following
irradiation in order to validate the use of the xCELLigence system.
Given the low cell densities used in the present study (500, 1,000,
2,000, and 4,000 cells/well), combined with the relatively low
rates of cell apoptosis, LDH release into the supernatant remained
under the detection limit, likely due to the low sensitivity of the
assay. Therefore, the total LDH detection method was used following
cell lysis, which showed good overall correlation with the MTS
results. The effects of irradiation were investigated using various
methods: The clonogenic method relies on CFU capacity, the
xCELLigence system detects impedance influenced by cell attachment,
the MTS and LDH assays reflect the metabolic activity levels of
viable cells, and Annexin V/PI staining determines cell apoptosis
rates.
Compared with conventional end-point cell-based
assays, dynamic monitoring of the cell response, such as cell
adhesion, spreading, proliferation, and cell death, is one of the
advantages of the xCELLigence system. Cell number optimization is
usually performed prior to experimentation with a given assay;
however, in the present study the impact of cell density on
impedance measurements was demonstrated, and 1,000 cells/well was
determined to be the optimal cell density on the E-plate 16. At
higher cell densities the length of the assay was limited either by
the lack of nutrients in the culture media, or by overgrowth of the
cells in the well. This overgrowth was represented by a plateau in
the CI values. The impedance-based data detected differences in the
radiosensitivity between the various types of human malignant and
primary cells. As the fastest dividing (data not shown) cells, the
MCF7 human breast adenocarcinoma cells were the most sensitive to
γ-rays, corresponding to the lowest metabolic activity levels and
the highest rates of apoptosis. The GBM2 human glioblastoma and
A549 human lung carcinoma cells also showed high radiosensitivity
with dose-dependent diminished proliferation rates, as determined
by the impedance measurements obtained with cell densities of 500
and 1,000 cells/well. The differences in responsiveness of the
various cells to ionizing radiation with regards to the
investigated parameters is a unique characteristic of each cell
type. Irradiated GBM2 and A549 cells exhibited high apoptotic
rates, as well as low dehydrogenase activity at low cell densities.
The U251 human glioblastoma cells were influenced only at 1,000
cells/well in the E-plate. Mitochondrial enzymatic activity
significantly diminished, whereas the levels of LDH, a cytoplasmic
enzyme, remained unaffected. As radiation induces the intrinsic
apoptotic pathway, a substantial portion (17%) of the cells were
apoptotic. HT-29 human colorectal adenocarcinoma cells showed low
radiosensitivity irrespective of the irradiation dose according to
the E-plate measurements, whereas mitochondrial and cytoplasmic
enzyme activity was greatly affected by radiation. As slowly
dividing cells, human primary fibroblasts proved to be completely
resistant to γ photons. Impedance, cellular dehydrogenases and
phosphatidylserine exposure remained unaltered. The insensitivity
to radiation of the primary fibroblasts may be relevant in clinical
settings. Tumor-associated fibroblasts are important cellular
components of the stroma of solid tumors, inducing
neovascularization, modulating the immune response, and enhancing
metastasis (24). Interactions
between malignant cells and surrounding stromal cells may have an
important role in tumor invasion and metastasis (25,26).
A previous study of human retinoblastoma delineated the
disadvantages of radiotherapy, where following radiation treatment
and tumor regression, fibrovascular proliferation on the retinal
surface was observed (27).
Radiation of the neoplastic tissues surrounded by stromal
components may undesirably affect the progression of cancer, as
irradiated fibroblasts enhance the metastatic ability of pancreatic
cancer cells (28).
To our knowledge, the present study is the first to
demonstrate the application of the xCELLigence system for the
determination of the effects of radiation on cells in culture. The
results obtained using standard end-point assays correlated with
the decline of the CI values detected following irradiation.
However, the end-point assays were only able to investigate the
reductive capacity of a cell, rather than determining alterations
in cell adherence, shape, and size, which are earlier phenotypic
changes than cell death itself.
In conclusion, the present study demonstrated that
the xCELLigence system is able to detect cell responses to ionizing
radiation, and provides real-time data on the onset of these
effects. There are numerous platforms available for the xCELLigence
system: Single plate (SP 96, 1×96 wells), double plate (DP 16, 3×16
wells). and multi plate (MP 96, 6×96 wells). The multi plate
platform offers the possibility of high-throughput real-time
screening of preclinical drugs in vitro, which influence
malignant and tumor stromal cell behavior, or examination of the
response of biopsy-derived cells to radiation with concomitant
medication ex vivo. A low cell density requirement (1,000
cells) is an advantage offered by the xCELLigence system for the
study of radiation effects, and being a non-invasive, label-free
and real-time method, subsequent analysis, such as determination of
DNA or protein expression, or enzyme activity, of the investigated
cells would be possible.
Acknowledgments
The present study was supported by a grant from
“TÁMOP-4.2.1/B-09/1/KONV-2010-0005 Creating the Centre of
Excellence at the University of Szeged”, supported by the European
Union and co-financed by the European Regional Development Fund.
The ELI-ALPS project (GOP-1.1.1-12/B-2012-0001) is supported by the
European Union and co-financed by the European Regional Development
Fund.
References
|
1
|
Bittoni A, Faloppi L, Giampieri R and
Cascinu S: Selecting the best treatment for an individual patient.
Recent Results Cancer Res. 196:307–318. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Weiss JF and Landauer MR: History and
development of radiation-protective agents. Int J Radiat Biol.
85:539–573. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Fietkau R: Concurrent radiochemotherapy
for the treatment of solid tumors. Strahlenther Onkol. 188(Suppl
3): 263–271. 2012.In German. View Article : Google Scholar
|
|
4
|
Hendry JH: Radiation biology and radiation
protection. Ann ICRP. 41:64–71. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Smout MJ, Kotze AC, McCarthy JS and Loukas
A: A novel high throughput assay for anthelmintic drug screening
and resistance diagnosis by real-time monitoring of parasite
motility. PLoS Negl Trop Dis. 16(4): e8852010. View Article : Google Scholar
|
|
6
|
Buch K, Peters T, Nawroth T, Sänger M,
Schmidberger H and Langguth P: Determination of cell survival after
irradiation via clonogenic assay versus multiple MTT Assay – a
comparative study. Radiat Oncol. 7:12012. View Article : Google Scholar
|
|
7
|
Price P and McMillan TJ: Use of
tetrazolium assay in measuring the response of human tumor cells to
ionizing radiation. Cancer Res. 50:1392–1396. 1990.PubMed/NCBI
|
|
8
|
Brubel R, Boronkai A, Reglodi D, Racz B,
Nemeth J, Kiss P, Lubics A, Toth G, Horvath G, Varga T, Szogyi D,
et al: Changes in the expression of pituitary adenylate
cyclase-activating polypeptide in the human placenta during
pregnancy and its effects on the survival of JAR choriocarcinoma
cells. J Mol Neurosci. 42:450–458. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Caltová K and Cervinka M:
Antiproliferative effects of selected chemoterapeutics in human
ovarian cancer cell line A2780. Acta Medica (Hradec Kralove).
55:116–124. 2012.
|
|
10
|
Mishra J, Mittra B and Mittra A: Effect of
whole body gamma radiation on hepatic LDH activity, lactate,
pyruvate concentration and rate of oxygen consumption in Bufo
melanostictus. Indian J Exp Biol. 40:1310–1313. 2002.
|
|
11
|
Ozsvári B, Puskás LG, Nagy LI, Kanizsai I,
Gyuris M, Madácsi R, Fehér LZ, Gerö D and Szabó C: A
cell-microelectronic sensing technique for the screening of
cytoprotective compounds. Int J Mol Med. 25:525–530.
2010.PubMed/NCBI
|
|
12
|
Kiss L, Walter FR, Bocsik A, Veszelka S,
Ozsvári B, Puskás LG, Szabó-Révész P and Deli MA: Kinetic analysis
of the toxicity of pharmaceutical excipients Cremophor EL and RH40
on endothelial and epithelial cells. J Pharm Sci. 102:1173–1181.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kürti L, Veszelka S, Bocsik A, Dung NT,
Ozsvári B, Puskás LG, Kittel A, Szabó-Révész P and Deli MA: The
effect of sucrose esters on a culture model of the nasal barrier.
Toxicol In Vitro. 26:445–454. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kürti L, Veszelka S, Bocsik A, Ozsvári B,
Puskás LG, Kittel A, Szabó-Révész P and Deli MA: Retinoic acid and
hydrocortisone strengthen the barrier function of human RPMI 2650
cells, a model for nasal epithelial permeability. Cytotechnology.
65:395–406. 2013. View Article : Google Scholar :
|
|
15
|
Atienza JM, Yu N, Kirstein SL, Xi B, Wang
X, Xu X and Abassi YA: Dynamic and label-free cell-based assays
using the real-time cell electronic sensing system. Assay Drug Dev
Technol. 4:597–607. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Urcan E, Haertel U, Styllou M, Hickel R,
Scherthan H and Reichl FX: Real-time xCELLigence impedance analysis
of the cytotoxicity of dental composite components on human
gingival fibroblasts. Dent Mater. 26:51–58. 2010. View Article : Google Scholar
|
|
17
|
Ke N, Wang X, Xu X and Abassi YA: The
xCELLigence system for real-time and label-free monitoring of cell
viability. Methods Mol Biol. 740:33–43. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Diemert S, Dolga AM, Tobaben S, Grohm J,
Pfeifer S, Oexler E and Culmsee C: Impedance measurement for
real-time detection of neuronal cell death. J Neurosci Methods.
203:69–77. 2012. View Article : Google Scholar
|
|
19
|
Erskine CL, Henle AM and Knutson KL:
Determining optimal cytotoxic activity of human Her2neu specific
CD8 T cells by comparing the Cr51 release assay to the xCELLigence
system. J Vis Exp. 66:e36832012.PubMed/NCBI
|
|
20
|
Shareef MM, Brown B, Shajahan S,
Sathishkumar S, Arnold SM, Mohiuddin M, Ahmed MM and Spring PM:
Lack of P-glycoprotein expression by low-dose fractionated
radiation results from loss of nuclear factor-kappaB and NF-Y
activation in oral carcinoma cells. Mol Canc Res. 6:89–98. 2008.
View Article : Google Scholar
|
|
21
|
Schwarz SB, Schaffer PM, Kulka U,
Ertl-Wagner B, Hell R and Schaffer M: The effect of radio-adaptive
doses on HT-29 and GM637 cells. Radiat Oncol. 3:122008. View Article : Google Scholar
|
|
22
|
Feofanova N, Geraldo JM and de Andrade LM:
Radiation oncology in vitro: Trends to improve radiotherapy through
molecular targets. Biomed Res Int. 2014:4616872014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hendry JH and West CM: Apoptosis and
mitotic cell death: Their relative contributions to normal-tissue
and tumour radiation response. Int J Radiat Biol. 71:709–719. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Togo S, Polanska UM, Horimoto Y and Orimo
A: Carcinoma-associated fibroblasts are a promising therapeutic
target. Cancers (Basel). 5:149–169. 2013. View Article : Google Scholar
|
|
25
|
Grey AM, Schor AM, Rushton G, Ellis I and
Schor SL: Purification of the migration stimulating factor produced
by fetal and breast cancer patient fibroblasts. Proc Natl Acad Sci
USA. 86:2438–2442. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Camps JL, Chang SM, Hsu TC, Freeman MR,
Hong SJ, Zhau HE, von Eschenbach AC and Chung LW:
Fibroblast-mediated acceleration of human epithelial tumor growth
in vivo. Proc Natl Acad Sci USA. 87:75–79. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Albert DM, Walton DS, Weichselbaum RR,
Cassady JR, Little JB, Leombruno D, Trantravahi R and Puliafito CA:
Fibroblast radiosensitivity and intraocular fibrovascular
proliferation following radiotherapy for bilateral retinoblastoma.
Br J Ophthalmol. 70:336–342. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ohuchida K, Mizumoto K, Murakami M, Qian
LW, Sato N, Nagai E, Matsumoto K, Nakamura T and Tanaka M:
Radiation to stromal fibroblasts increases invasiveness of
pancreatic cancer cells through tumor-stromal interactions. Cancer
Res. 64:3215–3222. 2004. View Article : Google Scholar : PubMed/NCBI
|