Introduction
Gonadotropin-releasing hormone (GnRH) is a
hypothalamic hormone, which regulates reproduction. Pulsatile
secretion of GnRH stimulates the secretion of follicular
stimulating hormone (FSH) and luteinizing hormone (LH) from the
pituitary gland. These pituitary hormones stimulate the synthesis
of gonadal steroid hormones and gametogenesis in the gonads
(1). It has been established that
continuous stimulation by GnRH agonists downregulates GnRH
receptors (GnRH-R) in the pituitary, resulting in a decrease in the
secretions of FSH and LH, ultimately resulting in chemical
castration (2).
GnRH binds to GnRH-R, resulting in effects on
cellular function (3). Upon
binding to its ligand, GnRH-R activates various intracellular
mechanisms mediated by G-protein coupled receptor (GPCR)
signalling, which leads to the inhibition of mitogenic signalling
and results in a reduction in cell proliferation and arrest of the
cell cycle in the G0/G1 phase (1).
The presence of GnRH receptors in gonadal
steroid-dependent organs and tissues, including breast, ovary,
endometrium and prostate tissue, and in cancer in the respective
organs has been reported. The expression of GnRH-R has been
observed in 80% of human ovarian and endometrial cancer cases and
50% of breast cancer cases (4). In
addition, GnRH and/or its agonists have been demonstrated to have
direct inhibitory effects on cell proliferation or invasiveness in
breast (5), prostate (6), endometrial (7) and ovarian (8) cancer.
The effects of GnRH on cancer in peripheral organs
other than the gonadal steroid-dependent organs remain to be fully
elucidated. GnRH-R has been reported to be expressed in colorectal
carcinoma cells and GnRH analogues exert direct inhibitory effects
on the cells (9). GnRH has been
reported to suppress the motility and invasiveness of melanoma
cells (10). By contrast, while
GnRH and GnRH-R are expressed in bladder cancer, treatment with
GnRH does not have any effects on the proliferation of the cells
(11).
GnRH analogues, including leuprolide and
triptorelin, which have been developed to treat gonadal
steroid-dependent cancer through suppression of the pituitary
gonadal axis, have also been observed to have direct effects on
GnRH-R-expressing cancer cells (12). In addition, the presence of GnRH
receptors in cancer cells has been suggested for use in molecular
targeted therapies. Anticancer drug treatments have been conjugated
to GnRH analogues to target cancer cells expressing GnRH-R
(5). For example,
cytotoxic-radical-conjugated GnRH analogues have been observed to
have anticancer effects in colorectal carcinoma (9).
Nasopharyngeal carcinoma (NPC) is a major type of
cancer in southern China, North Africa and Southeast Asian
countries, including Malaysia (13). Patients with NPC often present in
the late stages of the disease at the time of diagnosis (14). Radiotherapy is the primary modality
of treatment for this type of cancer, however this type of therapy
risks damage to the hypothalamic-pituitary axis due to the location
of the nasopharynx (15). If GnRH
has effects on NPC cells, alteration of secretion of GnRH may
affect the progression of NPC. To investigate this, the present
study aimed to examine the expression of GnRH-R in NPC tumours, and
the effects of GnRH on the NPC cells. To address this, the present
study examined the expression of GnRH-R in NPC tumors, and
investigated the effects of GnRH on the viability, motility and
signaling of NPC cells
Materials and methods
GnRH and its analogues
Human GnRH and the GnRH analogues, leuprolide and
triptorelin, were purchased from Sigma-Aldrich (St. Louis, MO,
USA).
Snap-frozen biopsies, cell lines and
culture conditions
Snap-frozen biopsies were obtained from the
Department of Otorhinolaryngology, Hospital Tengku Ampuan Rahimah
(Klang, Malaysia). Written informed consent was obtained from all
patients prior to the biopsy procedures. The tissues were
immediately snap-frozen and preserved in liquid nitrogen prior to
RNA extraction. The diagnosis of NPC was confirmed from the
hospital histopathology reports. All procedures for obtaining and
investigating human tissues were approved by the Medical Research
and Ethics Committee (Ministry of Health, Malaysia). The HK1 cells,
which were provided by Professor GSW Tsao, (Hong Kong University,
Hong Kong, China), is an Epstein-Barr virus (EBV)-negative NPC cell
line, previously derived from a patient with recurrent NPC from
Hong Kong (16) was used in the
present study. The HK1 cells were maintained at 37°C in a
humidified atmosphere of 5% CO2 in RPMI 1640 medium
(Gibco Life Technologies, Carlsbad, CA, USA), supplemented with 10%
fetal bovine serum (FBS; Gibco Life technologies), 50 U/ml
penicillin (Invitrogen Life Technologies, Carlsbad, CA, USA) and 50
μg/ml streptomycin (Invitrogen Life Technologies). The
identity of the HK1 cells was validated by DNA fingerprinting using
the AmpFiSTR Identifiler® PCR amplification kit (Applied
Biosystems Life Technologies, Foster City, CA, USA) and confirmed
mycoplasma free by regular assessment using a VenorGeM®
mycoplasma detection kit (Minerva Biolabs GmbH, Berlin, Germany).
The C-666-1 cells, provided by Dr KW Lo (The Chinese University of
Hong Kong, Hong Kong, China) is an EBV-positive NPC cell line
(17), were maintained in the same
conditions as the HK1 cells, but with 15% FBS. NP69 (18) and NP460hTERT (19) are immortalised nasopharyngeal
epithelial cell lines. The NP69 cells were maintained in
keratinocyte-serum free medium (KSFM) supplemented with 25
μg/ml bovine pituitary extract and 0.16 ng/ml recombinant
epidermal growth factor (Gibco Life Technologies). The NP460 cells
were maintained in a 1:1 ratio of defined KSFM supplemented with
growth factor (Gibco Life Technologies) and EpiLife medium
supplemented with EpiLife Defined Growth Supplement (Cascade
Biologics, Portland, OR, USA). HK1 cells were used throughout the
present study, whereas the C-666-1, NP69 and NP460hTERT cells were
only used in the reverse transcription-quantitative polymerase
chain reaction (RT-qPCR) experiments.
Gene expression omnibus (GEO) database
data mining
A microarray dataset for NPC (GSE12452) was
retrieved from the GEO database (http://www.ncbi.nlm.nih.gov/sites/GDSbrowser). The raw
data were re-analysed using GeneChip® Operating Software
version 5.0 (Affymetrix, Inc., Santa Clara, CA, USA) and the number
of specimens expressing the GnRH and GnRH-R transcripts were
determined.
RT-qPCR of GnRH-R mRNA
Total RNA was isolated from the homogenised
snap-frozen nasopharyngeal cancer tissue specimens and cultured
nasopharyngeal cancer cells using an RNeasy mini kit (Qiagen GmbH,
Hilden, Germany), according to the manufacturer's instructions.
First strand complementary DNA (cDNA) was synthesised from 1
μg total RNA using a High-Capacity cDNA Reverse
Transcription kit (Applied Biosystems Life Technologies). The
primer sequences for Type-1 GnRH-R were as follows: Sense 5′-TCT
GGA AAG ATC CGA GTGAC-3′ and antisense 5′-TCA GCC ATC AAC AAC AGC
ATCCC-3′; and the primer sequences for Type 1 GnRH (GnRH-I) were as
follows: Sense 5′-TTT CCA AGA GAT AGT CAA AGA GGTT-3′ and antisense
5′-TCA GAC TTT CCA GAG CTC CTT TCAG-3′. RT-qPCR was performed on a
Thermal cycler (C1000 Thermal Cycler, Bio-Rad Laboratories, Inc.,
Hercules, CA, USA) using the cDNA product (1:25) as a template, 0.2
μM of each primer and 1X i-PCR master mix (i-DNA
Biotechnology Pte. Ltd., Singapore). The cycling conditions were as
follows: Initial denaturation step at 94°C for 5 min, denaturation
step at 94°C for 30 sec, annealing step at 58°C for 30 sec and an
extension step at 72°C for 30 sec, and a final extension for 10 min
at 72°C following the final cycle. The reactions were subjected to
35 cycles in total. As an internal reference, β-actin mRNA was
amplified from the same samples. The identity of the representative
amplified qPCR products were confirmed by sequencing (First Base
Laboratories, Seri Kembangan, Malaysia).
Immunohistochemistry
The protein expression of GnRH-R in the NPC tissues
was investigated using immunostaining with mouse anti-human GnRH-R
monoclonal antibody (Clone A9E4; Leica Biosystems, Newcastle Upon
Tyne, UK). Sections (4 μM) from the formalin-fixed,
paraffin-embedded archival tissues were cut using a microtome
(HM340E; Thermo Fisher Scientific, Walldorf, Germany). These
sections were then dewaxed, and antigen retrieval was performed
using citrate buffer (Dako Target Retrieval Solution, Glostrup,
Denmark). The endogenous peroxidase activity was inhibited using
3.3% hydrogen peroxide. The tissue sections were rinsed with 1X
Tris-buffered saline (TBS) buffer (Dako Wash Buffer; Dako Life
Technologies)and incubated with anti-GnRH-R antibody (1:50
dilution) at room temperature for 2 h. Following rinsing with 1X
TBS, the sections were incubated for 30 min at room temperature
with anti-rabbit/mouse secondary antibody (1:5; K5007; Dako Life
Technologies). The antigen-antibody complex was detected using
avidin-biotin peroxidase complex solution containing
3,3′-diaminobenzidine stain and counter-stained with hematoxylin
(Dako Life Technologies, Glostrup, Denmark). Negative controls were
performed by omitting the primary antibody. Tissue sections of
breast cancer were used as a positive control.
Assessment of proliferative activity,
determined using a
3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-
(4-sulfophenyl)-2H-tetrazolium (MTS) colorimetric assay
An MTS assay was performed using a CellTiter
96® AQueous Non-Radioactive Cell Proliferation assay kit
(Promega Corporation, Madison, WI, USA). The HK1 cells were
rendered quiescent by incubation under serum-free conditions for 16
h at 37°C, following which the cells were cultured at an
approximate density of 4.0×103 cells/well in 96-well
flat-bottom cell culture plates (Orange Scientific, Braine-l'
Alleud, Belgium) containing 10% FBS supplemented medium with and
without graded concentrations of GnRH or its analogues
(10-12-10-9 M) for 1-6 days at 37°C. The
culture medium and GnRH or analogues were refreshed each day.
The viability of the cells were determined each day
of treatment with GnRH using the MTS assay. Each day, 20 μl
MTS solution was added to each well and the plates were incubated
at 37°C for 4 h. The absorbance was measured on an MRX 96-well
plate reader (Dynatec Laboratories, Houston, TX, USA) at a
wavelength of 490 nm and a reference wavelength of 630 nm.
Statistical calculations were performed using SPSS Advanced
Statistical Software SPSS Inc, Chicago, IL, USA). Student's t-test
was used to determine the significance of the comparison. P<0.05
was considered to indicate a statistically significant
difference.
Calcium imaging
The HK1 cells were seeded at a density of
2.5×105 cells/plate in poly-L-lysine
(Sigma-Aldrich)-coated 30 mm WillCo-dish® glass bottom
dishes (WillCo Wells, Amsterdam, The Netherlands) in the presence
of 10% FBS RPMI and cultured overnight at 37°C. The cells were then
stained with 5 μM Fluo3-Am (Molecular Probes Life
Technologies, Carlsbad, CA, USA) and 0.1% pluronic acid
(Sigma-Aldrich) in Hank's balanced salt solution-bovine serum
albumin-probenecid (HBSS-BSA-probenecid; AfCS Solution Protocol ID
PS00000574; pH 7.45; Gibco Life Technologies) and incubated in the
dark for 30 min at 37°C. The cells were washed in
HBSS-BSA-probenecid (Sigma-Aldrich) at least twice, and incubated
in fresh RPMI with 10% FBS. The cells were subjected to time-lapse
live cell imaging. The images were captured under a Nikon Eclipse
Ti-E automated inverted fluorescence microscope (Nikon, Tokyo,
Japan; magnification, ×20) at a speed of four frames/sec for 2 min
and, 2 sec after the start of image capture, the cells were treated
with ionomycin (10-5 M, positive control; Calbiochem,
Darmstadt, Germany), water (control) or GnRH (10-10 M),
respectively. The intracellular intensity changes of 10 cells from
each sample were measured and compared using NIS Elements software
version 4.0 (Nikon). Student's t-test was performed using SPSS.
P<0.05 was considered to indicate a statistically significant
difference.
Cell-cycle analysis
The HK1 cells were seeded into 100-mm culture plates
at a density of 4.0×105 cells/dish in 6 ml culture
medium. The cells were treated with GnRH at a concentration of
10-9 M for 48 h. Untreated cells were used as a control.
The cells were prepared for cell-cycle analysis using a CycleTEST
PLUS DNA Reagent kit (BD BioSciences, San Jose, CA, USA). A pellet
containing 5×105 cells was gently resuspended in 250
μl solution A, containing trypsin (BD Biosciences), followed
by 200 μl solution B, containing trypsin inhibitor and RNase
A (BD Biosciences), and incubated at room temperature for 10 min
each. A total of 200 μl propidium iodide (PI) was added and
the cell suspensions were incubated at 4°C in the dark for 10 min.
Flow-cytometric analysis of the cellular DNA content was performed
using Cell Quest Pro software version 6.0 (BD Biosciences) on a
FACS Calibur flow cytometer (BD BioSciences) and the results were
analysed using ModFit LT™ software version 4.0 (Verity
Software House, Inc., Topsham, ME, USA).
Time-lapse microscopy
The HK1 cells were starved overnight with 0.5% BSA
(Sigma-Aldrich, MO, USA) in RPMI medium. A total of
1.5×105 cells were then seeded in glass-bottom culture
plates with 1% FBS in RPMI medium. Following attachment, the cells
were treated with 10-10 M GnRH, while untreated cells
served as a control. Cell migration and changes in morphology were
examined under a Nikon Eclipse Ti-E inverted microscope (Nikon)
with a 10× differential interference contrast objective lens for 16
h, with a time-lapse frame interval of 10 min. During image
capture, the cells were cultured in 5% CO2 at 37°C
within a humidified stage chamber. Image-Pro Plus version 6.3
(Media Cybernetics, Inc., Rockville, MD, USA) was used for
analysis. The maximum speed of all cells within the frames were
measured after 10 h treatment, and the mean maximum speed of the
samples were compared using Student's t-test on GraphPad Prism 5
(GraphPad Software, Inc., La Jolla, CA, USA).
Results
GnRH-R is expressed in NPC
The microarray dataset deposited by Sengupta et
al (20) on the public
database, GEO, revealed that GnRH-R was expressed in 22 of the 31
NPC specimens (71%), while the hormone, GnRH, was expressed in 25
of the 31 specimens (81%). To verify this finding, several
snap-frozen NPC biopsies were examined for their mRNA expression
levels of GnRH-R and GnRH. As shown in Fig. 1A, amplified products of GnRH-R and
GnRH, with predicted sizes of 209 bp and 116 bp, respectively, were
observed from at least four samples, with a faint β-actin (internal
control) band observed in sample 3. The PCR products were confirmed
by sequencing.
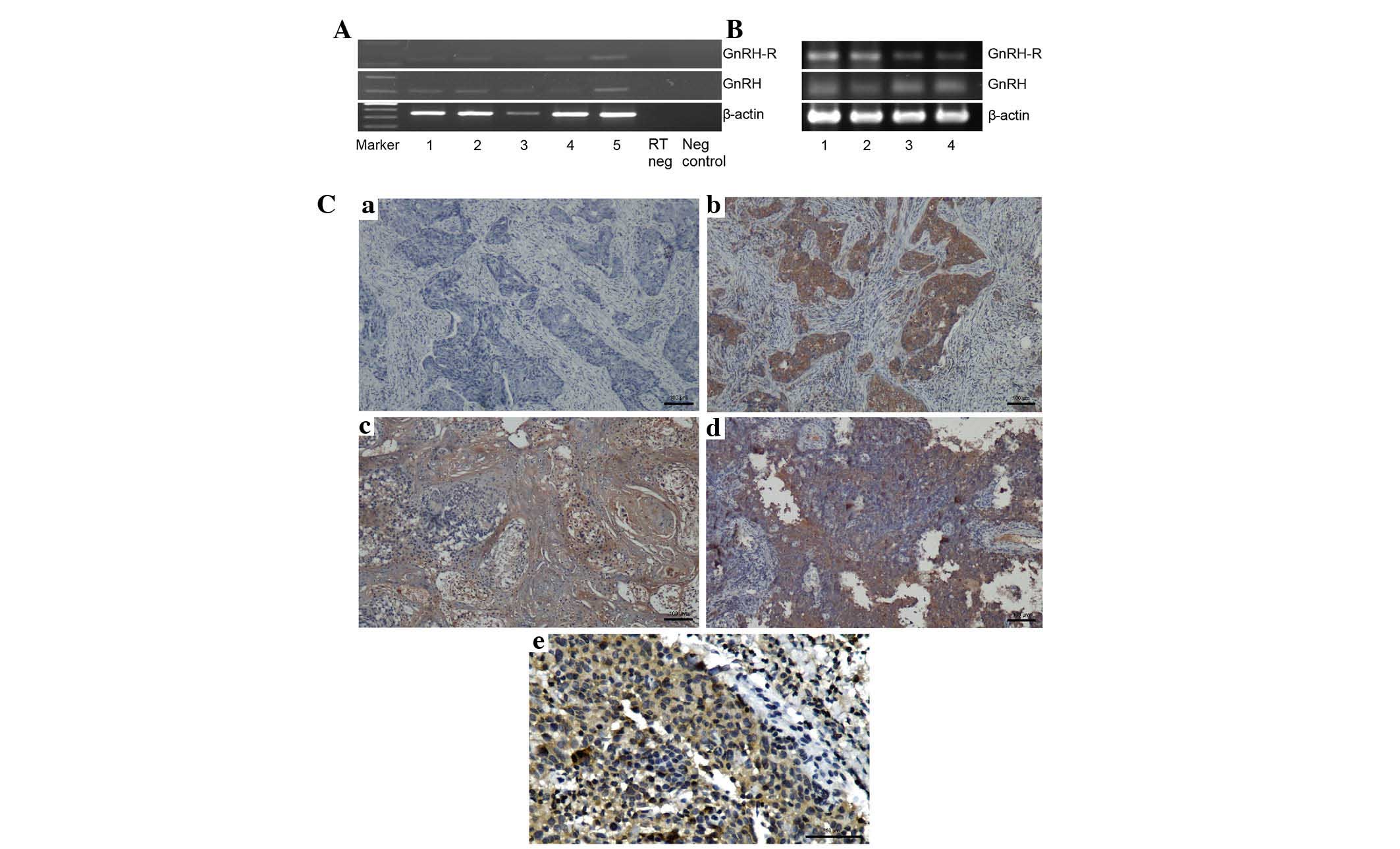 | Figure 1Expression levels of GnRH-R and GnRH
in NPC. (A) mRNA expression levels of GnRH-R and GnRH in biopsies
from patients with NPC, determined using reverse
transcription-quantitative polymerase chain reaction. The GnRH-R
and GnRH transcripts were detected in the majority of specimens
(Lane 1, 2, 4 and 5). β-actin mRNA was amplified as a control. (B)
mRNA expression levels of GnRH-R and GnRH in the NPC cell lines
(lane 1, HK1; lane 2, C666-1) and nasopharyngeal epithelial cells
(lanes 3 and 4, NP69 and NP460). (C) Immunohistochemistry for the
expression of GnRH-R in NPC xenografts and specimens, visualized
using a Nikon ECLIPSE Ti microscope (Nikon Corporation, Tokyo,
Japan). Breast cancer tissue was stained as a positive control (b),
while the primary antibody was omitted as a negative control (a).
GnRH-R was detected in HK1 NPC xenograft (c) and NPC biopsy (d) and
(e) at a high magnification. NPC, nasopharyngeal carcinoma; GnRH,
gonadotropin releasing hormone; GnRH-R, GnRH-receptor. |
The GnRH-R and GnRH transcripts were expressed in
the NPC and nasopharyngeal epithelial cell lines (Fig. 1B). In addition, GnRH-R was detected
by immunohistochemically in at least 25% (2/8) of NPC specimens
(Fig. 1C). The HK1 cells used were
confirmed via DNA fingerprinting (data not shown) to be comparable
to the cells used in other investigations (21).
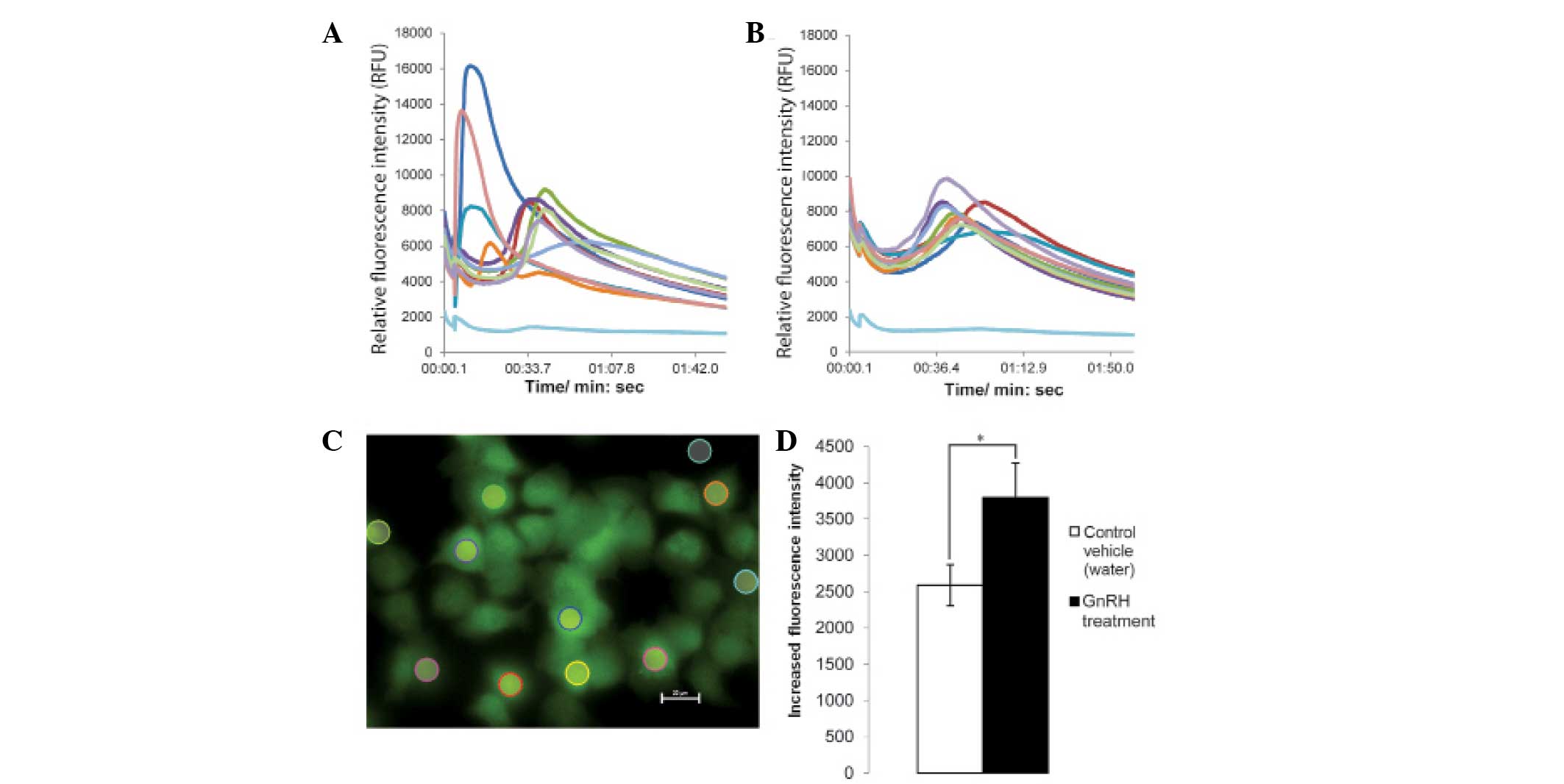
GnRH induces an increase in ionised
calcium concentration in NPC cells
Following the addition of GnRH, the fluorescence
intensity of the HK1 cells increased rapidly (Fig. 2A). The fluorescence intensities of
the untreated cells and vehicle control-treated cells (Fig. 2B) were determined for comparison.
The cells, which were treated with GnRH exhibited a significantly
higher increase in fluorescence intensity, compared with the cells
in the vehicle control group (P<0.05; Fig. 2C and D). These data suggested that
transient elevation of ionized calcium concentration occurred when
the HK1 cells were treated with GnRH, thus suggesting that the
hormone induced calcium signalling in the NPC cells.
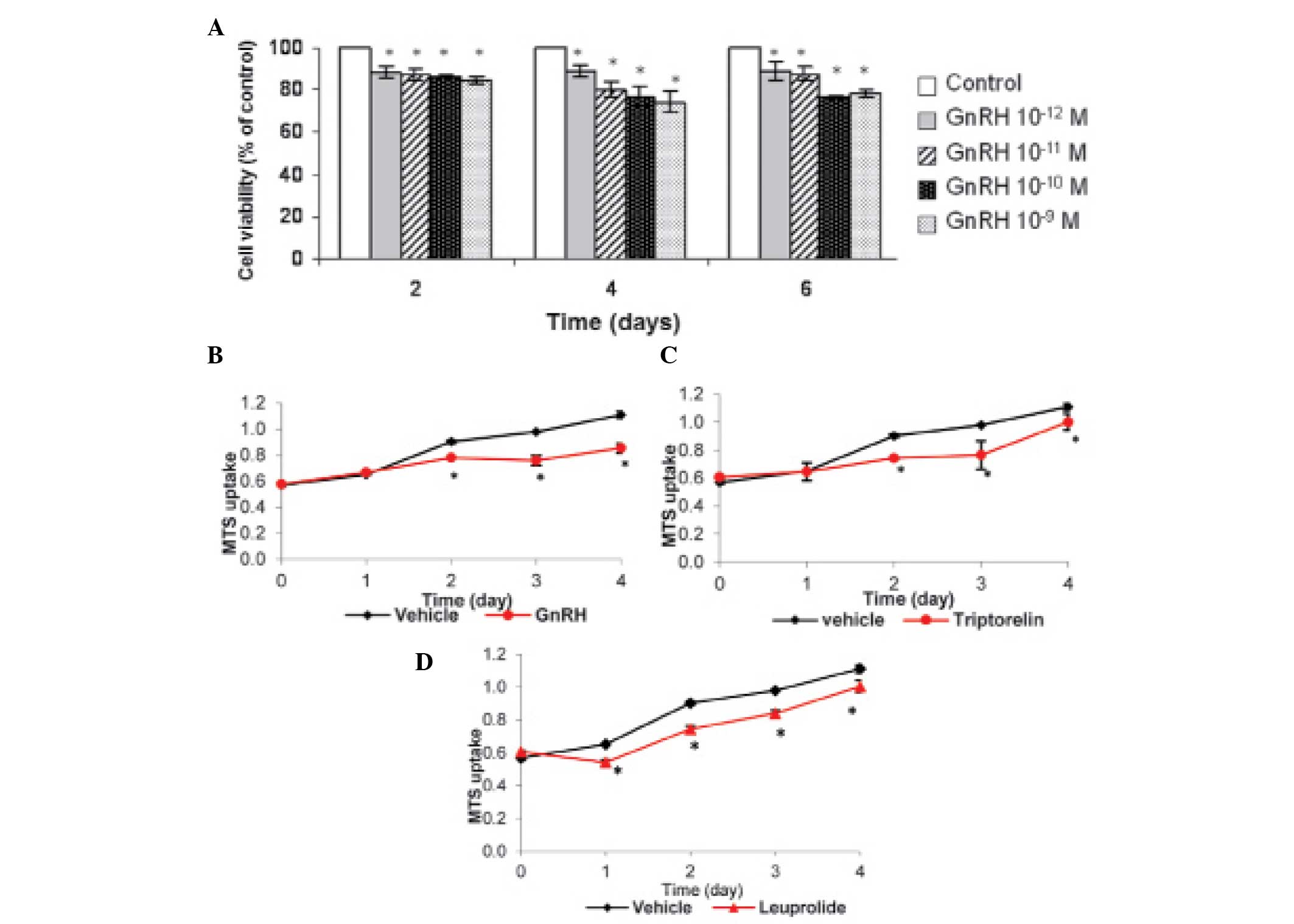
GnRH causes a reduction in cell
viability, but does not cause cell cycle arrest
An MTS assay was used to investigate the effect of
GnRH on the viability of the NPC/HK1 cells. The cells were cultured
and treated with GnRH, leuprolide or triptorelin at graded
concentrations (10−12–10−9 M) and assessed
with MTS for up to 6 days. GnRH, at nanomolar concentrations
(10−9–10−12 M) significantly inhibited the
growth of the cultured cells following 2, 4 and 6 days of treatment
(Fig. 3A and B). The maximum
growth inhibition was reached with 10−9 M and
10−10 M GnRH. Similar results were obtained with GnRH
analogues (Fig. 3C and D).
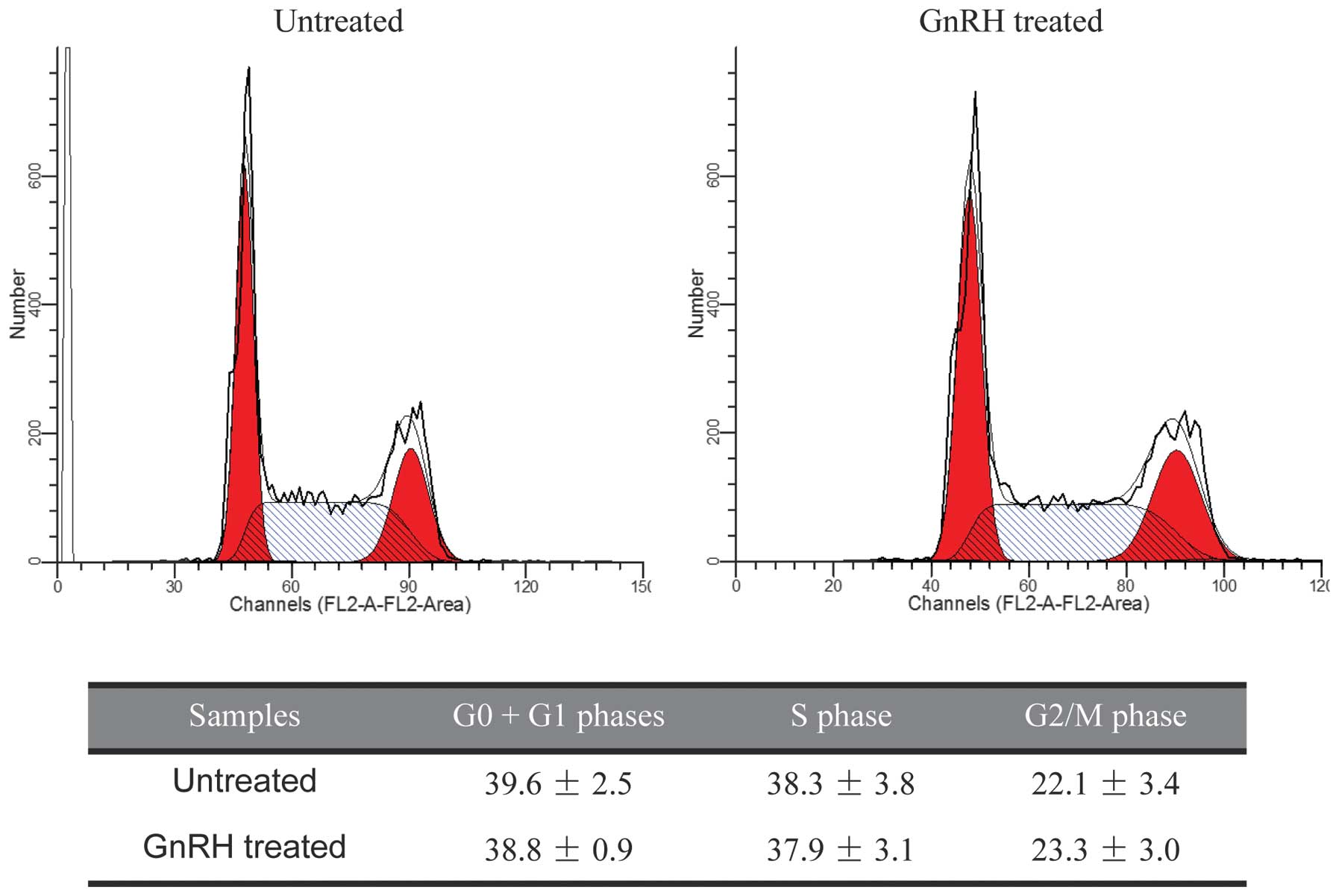
DNA contents of GnRH treated or control HK1 cells
were quantitated for cell cycle analysis. The distribution of cells
in the different phases of the cell cycle did not significantly
change following GnRH exposure (Fig.
4), suggesting that GnRH did not cause cell cycle arrest under
the conditions described.
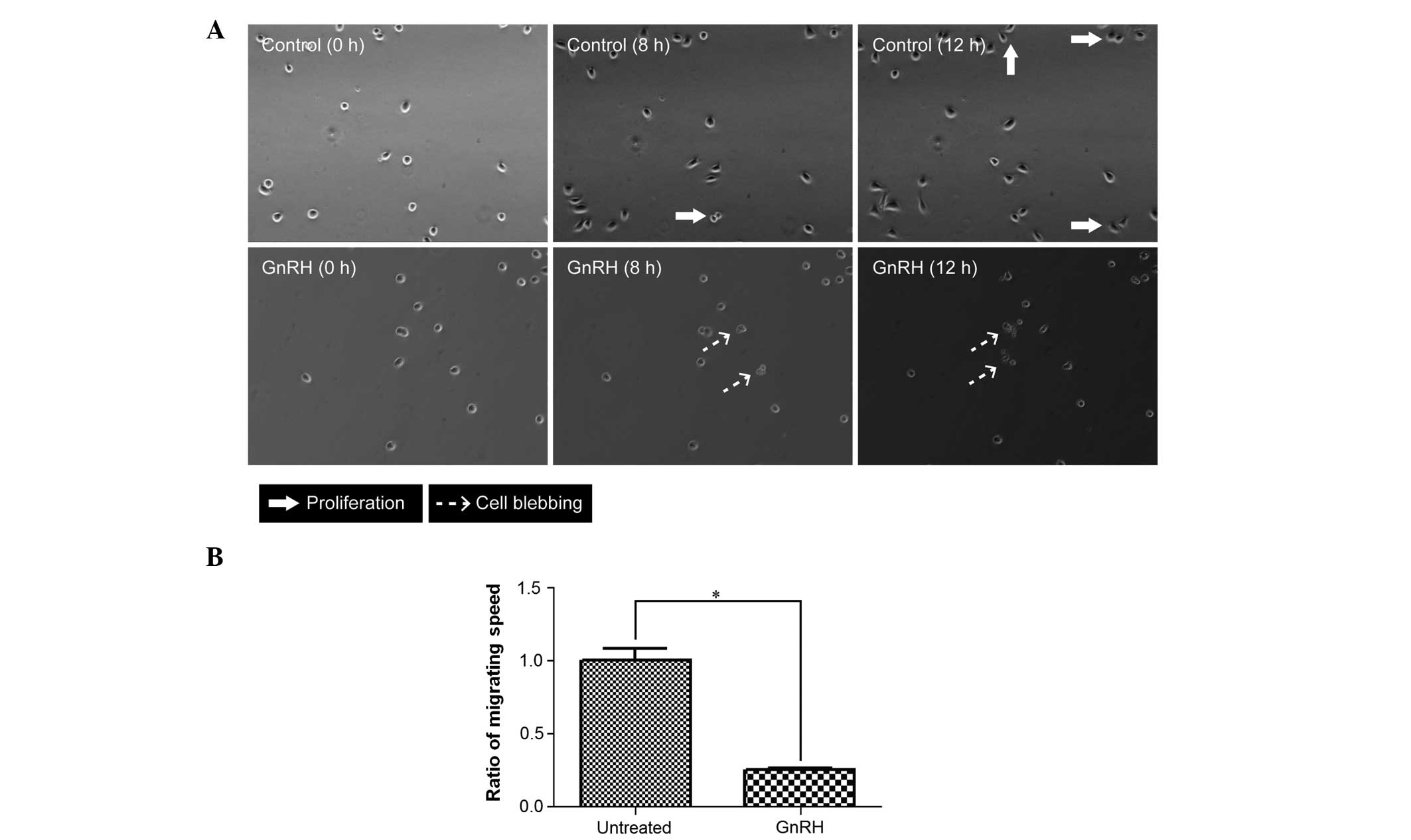
Effects of GnRH on cell morphology and
migration
The HK1 cells were treated with 10−10 M
GnRH in low serum medium prior to time-lapse-live cell microscopic
imaging. The morphology and migration of the HK1 cells were
observed for 16 h with an interval of 10 min. The control
(untreated) cells were found to proliferate, while the treated
cells were found to undergo apoptosis (Fig. 5A). In general, cell motility was
decreased by GnRH, which was four times slower (Fig. 5B), compared with the untreated
cells.
Discussion
The expression of GnRH-R in tumour cells, including
breast cancer cells, has been reported to determine the sensitivity
of the cells to GnRH (22,23). In the present study, the expression
of GnRH-R was observed in NPC at the transcriptional level,
determined in frozen biopsy samples and cell lines, and protein
level, determined in paraffin sections of xenograft and NPC
biopsies (Fig. 1), which indicated
that NPC cells potentially respond to GnRH. The data (Fig. 1B) suggested that the expression of
GnRH-R was higher in the NPC cells, compared with their non-NPC
counterpart, although further investigations are required to
confirm this. The exploitation of GnRH-R to target cancer may be
possible if the levels of GnRH-R are higher in the cancer cells
than in normal cells. The protein expression of GnRH-R in NPC is
variable. However, the detection of GnRH-R protein in a fraction
(25%) of NPC specimens suggested the potential sensitivity of a
subset of NPC tumours to GnRH.
The induction of calcium signalling in NPC cells by
GnRH suggest that GnRH-Rs are functional. Treatment of the NPC
cells with GnRH resulted in modest anti-proliferative effects on
the cells. In addition, treatment with GnRH resulted in the
appearance of cells undergoing apoptosis as well as a reduction in
motility, although modest in certain cases.
The effects of GnRH appeared to be variable.
Experiments involving the treatment of cells with GnRH are
complicated by several factors, including errors in measurement,
which can be significant due to the low concentration of the
hormone required for treatment. In addition GnRH activity may be
affected by the stability of the hormone in solution. Goodwin et
al (24) reported that the
half life of GnRH in treatment of Caco-2 cells was 12.2 mins, but
it is known that treatment of certain tissues with GnRH can lead to
rapid desensitization of the cells to the hormone (25), leading to a reduction in the effect
of GnRH. GnRH has been reported to induce opposite effects on
different cell lines of similar tissue origin, including ovarian
and prostate cancer (26–28). Furthermore, GnRH is reported to
have biphasic effects in the cell motility and invasiveness of
ovarian cancer cells (29). In
addition, the GnRH-R signalling pathway may be affected by cross
talk with other pathways and these pathways may vary with different
cells types (30). It is also
possible that differences in culture conditions can affect the
activation state of these other pathways, which affect the
sensitivity of the cells towards GnRH.
The GnRH-R is a GPCR, which can trigger activation
of the GPCR signalling pathway, leading to cellular responses
(31). During signal transduction,
calcium uptake was elevated in the present study. This transient
increase in ionized calcium concentration in the NPC cells upon
GnRH treatment suggested the possibility that GnRH exerted effects
on the cells through GPCR signalling.
In addition, endogenous GnRH may also affect
responsiveness to a treatment. Endogenous GnRH has been revealed to
promote cell proliferation in ovarian cancer (32). The levels of endogenous GnRH in the
cells may be controlled by a regulatory feedback mechanism, which
in turn could be affected by the presence of exogenous GnRH.
Therefore, treatment with exogenous GnRH may not only directly
affect the cells, but it may affect the overall response indirectly
by altering the levels of endogenous GnRH and, thus complicating
the observation.
The anti-proliferative effects of GnRH on NPC cells,
although modest, suggested that GnRH was involved, at least in
part, in controlling the growth of such cancer cells. Factors,
which alter the levels of GnRH, including damage to the
hypothalamic-pituitary axis, may have potential effects on the
growth of residual NPC cells in the body.
In conclusion, GnRH or its analogues exerted a
modest inhibitory effect on cell proliferation and motility in the
NPC cells. The presence of GnRH-Rs, in at least a subset of the NPC
specimens, suggested that the levels of GnRH may have effects on
NPC cells.
Acknowledgments
The authors would like to thank the Director General
of Health Malaysia for permission to publish this study and the
Director of the Institute for Medical Research for her support. The
authors would also like to thank Dr Katia Manova of the Molecular
Cytology Core Facility, Memorial Sloan Kettering Cancer Centre (New
York, USA) and Dr Stephen Cody of Monash Micro Imaging, Monash
University (Australia) for their advice on imaging experiments, and
Professor George Tsao of The Hong Kong University (Hong Kong,
China) and staff at the Institute for Medical Research and Monash
University Sunway Campus for their support. This study was funded
by the Ministry of Health of Malaysia [MRG-IMR-2007-NPC (06-059)
and 07-034].
References
|
1
|
Harrison GS, Wierman ME, Nett TM and Glode
LM: Gonadotropin-releasing hormone and its receptor in normal and
malignant cells. Endocr Relat Cancer. 11:725–748. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rothman MS and Wierman ME: The role of
gonadotropin releasing hormone in normal and pathologic endocrine
processes. Curr Opin Endocrinol Diabetes Obes. 14:306–310. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Everest HM, Hislop JN, Harding T, Uney JB,
Flynn A, Millar RP and McArdle CA: Signaling and antiproliferative
effects mediated by GnRH receptors after expression in breast
cancer cells using recombinant adenovirus. Endocrinology.
142:4663–4672. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nagy A and Schally AV: Targeting of
cytotoxic luteinizing hormone-releasing hormone analogs to breast,
ovarian, endometrial and prostate cancers. Biol Reprod. 73:851–859.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Schally AV and Nagy A: Chemotherapy
targeted to cancers through tumoral hormone receptors. Trends
Endocrinol Metab. 15:300–310. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Labrie F: Hormonal therapy of prostate
cancer. Prog Brain Res. 182:321–341. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Park DW, Choi KC, MacCalman CD and Leung
PC: Gonadotropin-releasing hormone (GnRH)-I and GnRH-II induce cell
growth inhibition in human endometrial cancer cells: Involvement of
integrin beta3 and focal adhesion kinase. Reprod Biol Endocrinol.
7:812009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Grundker C and Emons G: Role of
gonadotropin-releasing hormone (GnRH) in ovarian cancer. Reprod
Biol Endocrinol. 1:652003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Szepeshazi K, Schally AV and Halmos G:
LH-RH receptors in human colorectal cancers: Unexpected molecular
targets for experimental therapy. Int J Oncol. 30:1485–1492.
2007.PubMed/NCBI
|
|
10
|
Moretti RM, Montagnani Marelli M, Mai S
and Limonta P: Gonadotropin-releasing hormone agonists suppress
melanoma cell motility and invasiveness through the inhibition of
alpha3 integrin and MMP-2 expression and activity. Int J Oncol.
33:405–413. 2008.PubMed/NCBI
|
|
11
|
Bahk JY, Kim MO, Park MS, Lee HY, Lee JH,
Chung BC and Min SK: Gonadotropin-releasing hormone (GnRH) and GnRH
receptor in bladder cancer epithelia and GnRH effect on bladder
cancer cell proliferation. Urol Int. 80:431–438. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Montagnani Marelli M, Moretti RM,
Januszkiewicz-Caulier J, Motta M and Limonta P:
Gonadotropin-releasing hormone (GnRH) receptors in tumors: a new
rationale for the therapeutical application of GnRH analogs in
cancer patients? Curr Cancer Drug Targets. 6:257–269. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chang ET and Adami HO: The enigmatic
epidemiology of nasopharyngeal carcinoma. Cancer Epidemiol
Biomarkers Prev. 15:1765–1777. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pua KC, Khoo AS, Yap YY, Subramaniam SK,
Ong CA, Gopala Krishnan G and Shahid H; Malaysian Nasopharyngeal
Carcinoma Study Group: Nasopharyngeal carcinoma database. Med J
Malaysia. 63(Suppl C): 59–62. 2008.
|
|
15
|
Darzy K: Endocrine complications following
radiotherapy and chemotherapy for nasopharyngeal carcinoma.
Carcinogenesis, Diagnosis, and Molecular Targeted Treatment for
Nasopharyngeal Carcinoma. Chen Shih-Shun: InTech China; Shanghai,
China: pp. 133–154. 2012
|
|
16
|
Huang DP, Ho JH, Poon YF, Chew EC, Saw D,
Lui M, Li CL, Mak LS, Lai SH and Lau WH: Establishment of a cell
line (NPC/HK1) from a differentiated squamous carcinoma of the
nasopharynx. Int J Cancer. 26:127–132. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cheung ST, Huang DP, Hui AB, Lo KW, Ko CW,
Tsang YS, Wong N, Whitney BM and Lee JC: Nasopharyngeal carcinoma
cell line (C666-1) consistently harbouring Epstein-Barr virus. Int
J Cancer. 83:121–126. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tsao SW, Wang X, Liu Y, Cheung YC, Feng H,
Zheng Z, Wong N, Yuen PW, Lo AK, Wong YC, et al: Establishment of
two immortalized nasopharyngeal epithelial cell lines using SV40
large T and HPV16E6/E7 viral oncogenes. Biochim Biophys Acta.
1590:150–158. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li HM, Man C, Jin Y, Deng W, Yip YL, Feng
HC, Cheung YC, Lo KW, Meltzer PS, Wu ZG, et al: Molecular and
cytogenetic changes involved in the immortalization of
nasopharyngeal epithelial cells by telomerase. Int J Cancer.
119:1567–1576. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sengupta S, den Boon JA, Chen IH, Newton
MA, Dahl DB, Chen M, Cheng YJ, Westra WH, Chen CJ, Hildesheim A, et
al: Genome-wide expression profiling reveals EBV-associated
inhibition of MHC class I expression in nasopharyngeal carcinoma.
Cancer Res. 66:7999–8006. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chan SY, Choy KW, Tsao SW, Tao Q, Tang T,
Chung GT and Lo KW: Authentication of nasopharyngeal carcinoma
tumor lines. Int J Cancer. 122:2169–2171. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Imai A and Tamaya T: GnRH receptor and
apoptotic signaling. Vitam Horm. 59:1–33. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Morgan K, Stewart AJ, Miller N, Mullen P,
Muir M, Dodds M, Medda F, Harrison D, Langdon S and Millar RP:
Gonadotropin-releasing hormone receptor levels and cell context
affect tumor cell responses to agonist in vitro and in vivo. Cancer
Res. 68:6331–6340. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Goodwin D, Varamini P, Simerska P and Toth
I: Stability, permeability and growth-inhibitory properties of
gonadotropin-releasing hormone liposaccharides. Pharm Res.
32:1570–1584. 2015. View Article : Google Scholar
|
|
25
|
Conn PM, McArdle CA, Andrews WV and Huckle
WR: The molecular basis of gonadotropin-releasing hormone (GnRH)
action in the pituitary gonadotrope. Biol Reprod. 36:17–35. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cheung LW and Wong AS:
Gonadotropin-releasing hormone: GnRH receptor signaling in
extrapituitary tissues. Febs J. 275:5479–5495. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen CL, Cheung LW, Lau MT, Choi JH,
Auersperg N, Wang HS, Wong AS and Leung PC: Differential role of
gonadotropin-releasing hormone on human ovarian epithelial cancer
cell invasion. Endocrine. 31:311–320. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Enomoto M, Utsumi M and Park MK:
Gonadotropin-releasing hormone induces actin cytoskeleton
remodeling and affects cell migration in a cell-type-specific
manner in TSU-Pr1 and DU145 cells. Endocrinology. 147:530–542.
2006. View Article : Google Scholar
|
|
29
|
Cheung LW, Leung PC and Wong AS:
Gonadotropin-releasing hormone promotes ovarian cancer cell
invasiveness through c-Jun NH2-terminal kinase-mediated activation
of matrix metal-loproteinase (MMP)-2 and MMP-9. Cancer Res.
66:10902–10910. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Aguilar-Rojas A and Huerta-Reyes M: Human
gonadotropin-releasing hormone receptor-activated cellular
functions and signaling pathways in extra-pituitary tissues and
cancer cells (Review). Oncol Rep. 22:981–990. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kakar SS, Malik MT, Winters SJ and
Mazhawidza W: Gonadotropin-releasing hormone receptors: Structure,
expression and signaling transduction. Vitam Horm. 69:151–207.
2004. View Article : Google Scholar
|
|
32
|
Arencibia JM and Schally AV: Luteinizing
hormone-releasing hormone as an autocrine growth factor in ES-2
ovarian cancer cell line. Int J Oncol. 16:1009–1013.
2000.PubMed/NCBI
|