The degeneration of the central nervous system (CNS)
is characterized by chronic progressive loss of the structure and
functions of neuronal materials, resulting in functional and mental
impairments (1). While the causes
associated with neuronal degeneration remain poorly understood, the
incidence of neurodegeneration increases with age, in mid-to-late
adult life (2). This phenomenon,
which mainly affects elder individuals (3,4),
occurs in neurodegenerative diseases such as Alzheimer's disease
(AD), multiple sclerosis (MS), Parkinson's disease (PD),
amyotrophic lateral sclerosis (ALS) following viral infections.
Viruses are able to directly injure neurons by direct killing or
induction of apoptosis (5) to
leading to neuro-degeneration (6,7).
Similarly, in MS, the pathological features involve the
permeability of the blood brain barrier (BBB), the destruction of
myelin sheath, damage of the axon, the formation of glial scar and
the presence of inflammatory cells, mostly lymphocytes infiltrated
into the CNS (8). The loss of
myelin is manifested in clinical symptoms together with neuropathic
pain, paralysis, muscle spasms and optic neuritis (9).
Neurodegeneration induced by viruses, is noteworthy
since it refers to the interaction between the CNS and
environmental and viral factors, and suggests an important role of
immune response in neurodegeneration (10). Immune activation in the CNS, always
present in viral infections, immune-mediated disorders, and
neurodegenerative diseases (11),
involves microglia and astrocytes (12) which constitute the resident immune
cells of the CNS and play an important role in the regulation of
homeostasis of the brain during development, adulthood and aging
(13). In the CNS, microglia
constantly survey the microenvironment by producing factors that
influence surrounding astrocytes and neurons (14), particularly in response to pathogen
invasion or tissue damage thereby promoting an inflammatory
response that further engages a self-limiting response through the
immune system and initiates tissue repair (15). However, inflammation in tissue
pathology that may result in the production of neurotoxic factors
amplifying the disease states, indicates the persistence of
inflammatory stimuli or failure in normal resolution mechanisms
(16,17). Accordingly, specific inducers of
inflammation associated with neurodegenerative diseases converge in
mechanisms responsible in the sensing, transduction and
amplification of the inflammatory processes that result in the
production of neurotoxic mediators, such as cytokines and
interleukins (18,19). These neurotoxic mediators are, in
general, associated with several neurodegenerative diseases
including AD, MS, PD and ALS, which are commonly linked to
intracellular mechanisms such as the degradation of protein, the
dysfunction of mitochondria, the defects of axonal transport and
apoptosis (20–22). Inflammation associated with AD, MS,
PD and ALS is not typically the initiating factor of
neurodegenerative disease. However, the emerging evidence on the
sustained inflammatory response associated with the contribution of
microglia and astrocytes in disease progression, suggest
contributory important roles of effectors of neuroinflammation in
neuronal dysfunction and death. In this review, we assessed the
role played by these inflammatory processes in neurodegenerative
diseases.
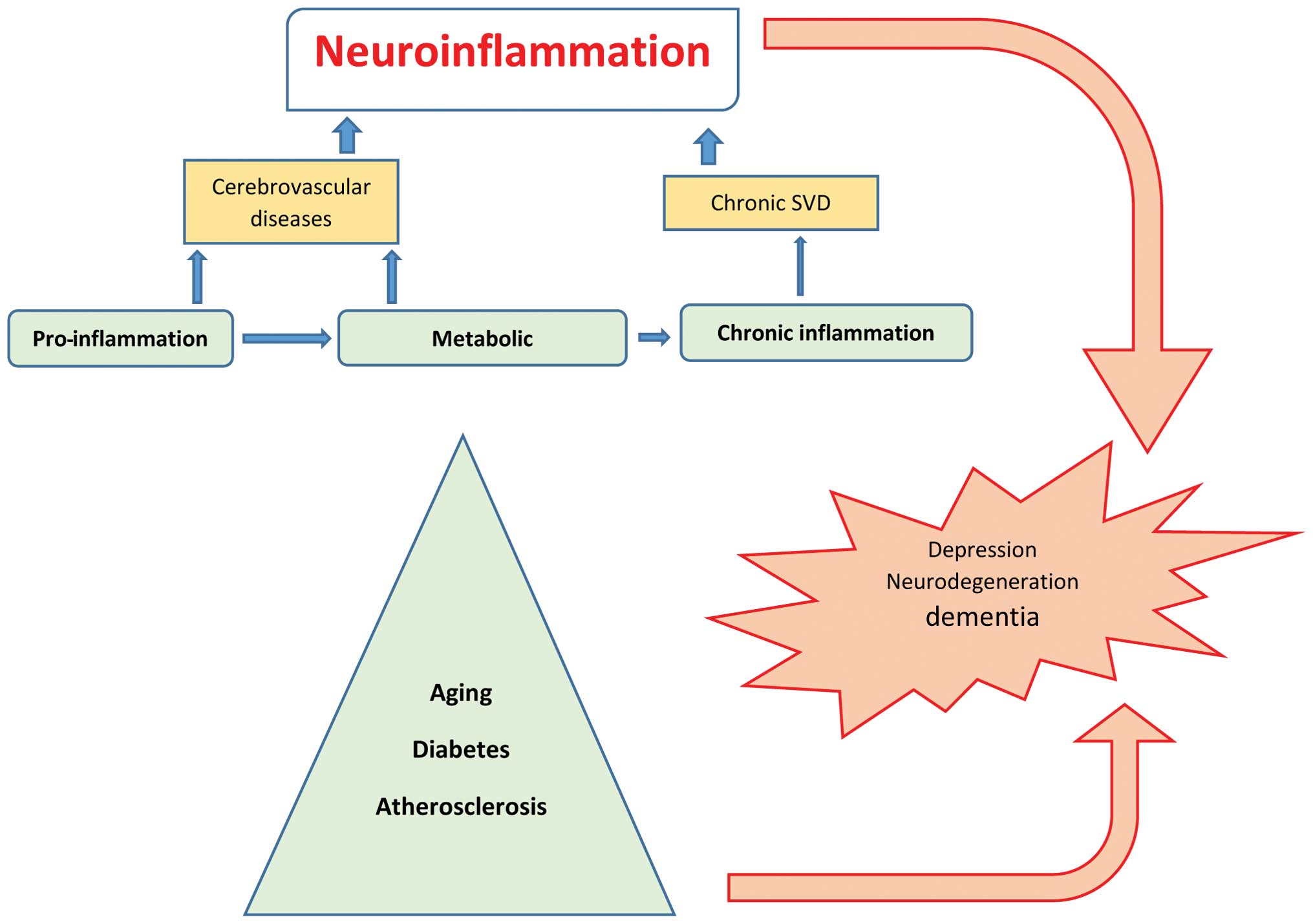
The cellular and molecular mechanisms of
neuroinflammation are likely the same in aging and metabolic
diseases such as hypertension, diabetes, depression, dementia or
after cerebral insult such as stroke (23), and are considered as silent
contributors of neuroinflammation (Fig. 1). In the elderly, inflammatory
mechanisms have been associated with the pathogenesis of dementia
and functional impairment. Systemic and local CNS inflammation
significantly contributes to cerebral small vessel disease
(SVD)-vascular dementia (24,25),
hypothesized as microvascular changes that result in a state of
chronic hypoperfusion, leading to continuous oligodendrocyte death
and the consecutive degeneration of myelinated fibers that increase
low-grade inflammation amplification of the risk of stroke
(26). Another major risk factor
for stroke and CNS tissue destruction is atherosclerosis, the
disease of arteries that is characterized by vascular inflammation
occasioned by the infiltration of monocytes into the injured
vascular wall and an increase of interleukin (IL)-6 associated with
future intracranial large artery stenosis progression after a
stroke episode (27). Additional
markers of inflammation such as C-reactive protein (CRP), which are
well established in cardiovascular disease as strong predictors of
subclinical and clinical atherosclerosis and progression of
hemorrhagic stroke, were identified in SVD (28–31).
Furthermore, adipose tissue dysfunction identified in obesity and
hypertension, contributes to chronic and low-grade inflammation,
predisposing to type 2 diabetes mellitus (DM) and cardiovascular
disease (32,33) and could determine a worse outcome
in stroke patients (34).
Mortality in DM is primarily attributed to micro- and
macro-vascular complication as well as sensory neuropathic
complications, exacerbating the consequences of vascular disease.
Sensory neuropathy promotes foot ulcers and abrogates warning
symptoms during a heart attack. However, metabolic inflammatory
disease (mataflammation) (35)
occurring in unhealthy nutritional habits, can lead to a series of
disorders and diseases such as CVD, stroke, hypertension, insulin
resistance, metabolic syndrome and DM. Lipid hormone (sphingolipids
and eicosanoids), cytokines and adipokines play an important role
in mataflammation through the induction of adverse regulatory
responses in target cells such as macrophages.
Normal aging is associated with an increase in the
expression level of systemic inflammatory factors (36) such as pro-inflammatory cytokines
(37–39). In the brain, this age-associated
inflammation manifests initially as the chronic activation of
perivascular and parenchymal macrophage/microglia expressing
pro-inflammatory cytokines together with an increased number of
astrocytes (40). Accordingly, the
chronic activation of pro-inflammatory signals in aging may
contribute to an increase in vulnerability to neuropsychiatric
disorders (41). In obese women,
the inflammation state was associated with a higher concentration
of pro-inflammatory markers including IL-6, CRP and adipokines
(42). These pro-inflammatory
markers correlated positively with symptoms of depression and
anxiety (43). Anxiety was
alleviated with the reduction of inflammation following the
surgical removal of fat tissue (44). In agreement with those findings,
metabolic diseases such as obesity, hypertension, and being elderly
are prevalent risk factors of depression, cognitive dysfunction and
dementia (45) and there is an
increase onset risk of aging-related diseases affecting the
cardiovascular, cerebrovascular, neuroendocrine, metabolic, and
immune systems in patients suffering major depression (46,47).
Although biological mechanisms of depression are
poorly understood, conventional antidepressant treatments procuring
beneficial effects were unsuccessful on one-third of depressed
patients due to the inflammation that contributed to treatment
resistance (48). The putative
mechanism linking inflammation and depression involved oxidative
stress, elevated pro-inflammatory cytokines IL-6 and IL-8 (49), endothelial nitric oxide synthase
uncoupling and hyperglutamatergia. Accordingly, indirect evidence
of neurovascular dysfunction have been found in major depressive
disorder (MDD) (50,51), a severe psychiatric illness that is
associated with increased levels of inflammatory markers in
periphery, depression and mortality from suicide (52). Therefore, inflammatory markers
identified in neurodegenerative diseases including MDD cover
chemokines, adhesion molecules, cytokines and acute phase proteins
(53).
Dynamic immune and inflammatory responses result
from several offences in the CNS, of which infection is one
(54). A virus can enter the CNS
through two distinct hypothetical mechanisms, including
hematogenous dissemination by which the virus gains access to the
brain by BBB (55), and neuronal
retrograde dissemination (56).
However, it has been suggested that a virus can replicate in
macrophage and CCR5+ T cells inside of the CNS in
relation to the development and progression of dementia (57), as is the case for HIV proteins
gp120 (58) and Tat (59) which are respectively able to induce
the apoptosis of neurons through the enhancement of CXCR4-PKC
(58), and to cause neuronal
dysfunction through the disruption of miRNA expression (59). Most importantly, as in the case of
HIV infection, other viral insults are associated with highly
secreted cytokines, cholesterol increase, elevations of
lipopolysaccharide (LPS) concentration, insulin resistance,
testosterone deficiency and APOE4 (60), which are all involved in
inflammation of the CNS.
Thus, inflammatory responses appear as the prevalent
triggering mechanism driving tissue damage that is likely
associated with different age-related diseases, as age-dependent
upregulation of the inflammatory response is a consequence of
chronic stress.
The CNS is an immune-privileged organ with the
innate and acquired immune response being closely controlled in
relation with the periphery. Evidence suggests that a strong
inflammatory response in the periphery from systemic LPS (61) or viral infections (62) results in the subsequent
infiltration of leukocytes from the periphery to the CNS with
consequent neuroinflammation and neurodegeneration. An offense is
followed by the initial activation of microglia, which induce the
release of pro-inflammatory mediators that favour the
permeabilisation of the BBB. The subsequent infiltration of
peripheral leukocytes occurs inside of the CNS, including T cells
and macrophages, which share several functional features with
microglia (61) including, the
expression of toll-like receptors (TLRs), and consequently the
ability to be activated by aggregated proteins or
pathogen-associated molecular patterns (61,63);
the expression of class II major histocompatibility complex, and
the ability to present antigens to CD4+ T cells to exert an
influence on the functional phenotype of T cells (64); as well as the ability to polarise
their functional phenotype towards inflammatory M1 and
anti-inflammatory M2 phenotypes, which can be influenced by
inflammatory T cells and the lymphocyte regulatory T cells
(65). Therefore, subsequent
permeability of BBB leads to the possibility that peripheral
macrophages can acquire a relevant role in the outcome of
neuroinflammation. Accordingly, the alteration of CD4+
and CD8+ T cells has been observed in the periphery of
neurodegenerative disease patients, suggesting a persisting
antigenic challenge and that T cells may play a role in
neurode-generative diseases. Of note, the ratio of CD8+
to CD4+ T cells or the shift to a Tc1/Th1-type immune
response may contribute to a harmful brain inflammatory reaction,
and the presence of antibodies against neuronal antigen observed in
neurodegenerative diseases (some of them being pathogenic),
solidify the involvement of the immune system in neurodegenerative
diseases (5). Consequently, an
acute neuro-inflammatory response is beneficial to the CNS,
minimizing the injury by activating the innate immune system
(66,67). By contrast, chronic inflammation is
characterized by the long-standing activation of microglia that
sustained release of inflammatory mediators, leading to an increase
of oxidative and nitrosative stress which perpetuate the
inflammatory cycle (68), further
prolonging inflammation (54,69),
which is detrimental for several neurodegenerative diseases
(70).
However, cell factors that influence microglial fate
invade the epithelial cells of the BBB while T cells infiltrate the
CNS, astrocytes and neurons (71),
the most abundant glial cell population of the CNS which also
participates in the innate immune response, triggered as a
consequence of constant insult during inflammation or infection.
Astrocytes are reservoirs of HIV-1, playing significant role in
virus-mediated neurodegeneration (17,57).
Accordingly, chronic neuroinflammation and microglia activation
play central roles in the pathophysiology of neurodegenerative
disease. For example, IL-1-positive activated microglia,
colocalized with amyloid β plaques and neurofibrillary tangles in
AD or present in degenerative motor neuron regions in patients
suffering ALS (72) lead to
abnormal phosphorylation of τ (73). Similarly, neurotropic viruses
trigger long-term neuroimmune activation to underlying mechanisms
of viral neurodegenerative diseases (74). Furthermore, neuroinflammation has
been associated with either the cause or consequence of chronic
oxidative stress, a key feature of all the neurodegenerative
diseases that causes genetic structural alteration, lipid and
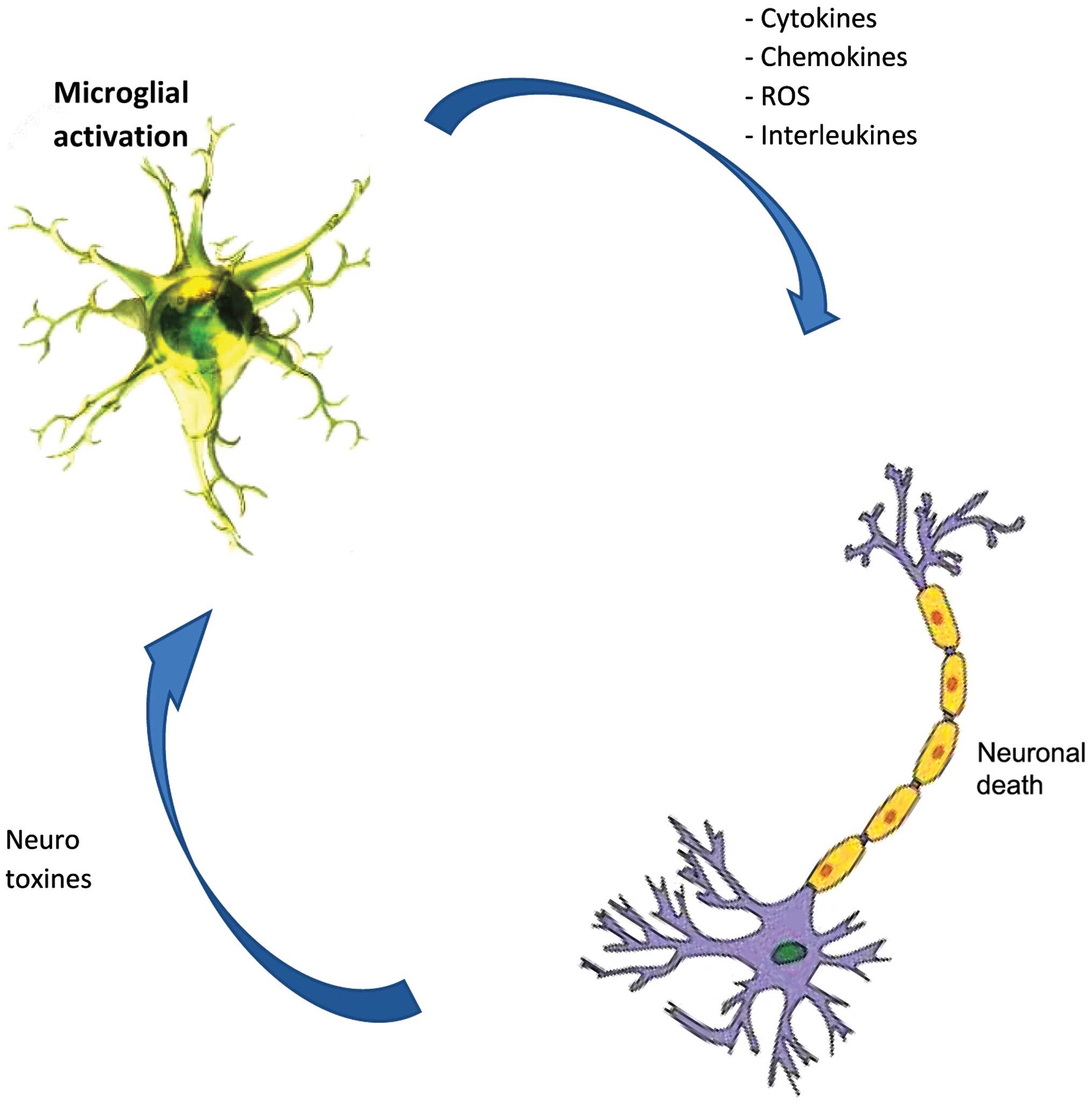
protein, resulting in neurodegeneration. Microglial cells are the
main source of reactive oxygen species and nitrogen species, tumor
necrosis-α and glutamate, all of which are neurotoxic when released
at a high dose after the activation of microglia (71,75,76)
(Fig. 2), likely due to the
stimulus from TLRs through the aggregated proteins (77,78)
as is the case of AD patients (79), (MS) (9), PD (80), and ALS (81).
Neuroinflammatory disorders are conditions involving
the immune response damage component of the nervous system. In the
CNS, inflammatory effectors derived from innate and acquired immune
systems as well as glial cells, particularly, microglia, act as
sensors for disturbed brain tissue homeostasis and accumulate
locally in response to neuronal cell injury or foreign entry in the
brain; the differential activation of microglia cells being the
central point that regulates neuroinflammation, which results in
neurotoxicity or neuroprotection. The environmental exposure is
therefore, the critical element for the fate of neurons with regard
to degeneration or protection. Additional studies must be
undertaken to benefit from the versatility of microglia, since
activated microglia can also produce anti-inflammatory mediators
and neurotrophic factors such as insulin-like growth factor-1,
glial cell-derived neurotrophic factor, brain-derived neurotrophic
factors and other factors (82–84),
and procure beneficial effects.
|
1
|
Campbell IL, Krucker T, Steffensen S, Akwa
Y, Powell HC, Lane T, Carr DJ, Gold LH, Henriksen SJ and Siggins
GR: Structural and functional neuropathology in transgenic mice
with CNS expression of IFN-α. Brain Res. 835:46–61. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hof PR and Mobbs CV: Handbook of the
neuroscience of aging. Elsevier/Academic Press; Amsterdam: pp.
1–53. 2010
|
|
3
|
Yuan J and Yankner BA: Apoptosis in the
nervous system. Nature. 407:802–809. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Przedborski S, Vila M and Jackson-Lewis V:
Neurodegeneration: What is it and where are we? J Clin Invest.
111:3–10. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Amor S, Puentes F, Baker D and van der
Valk P: Inflammation in neurodegenerative diseases. Immunology.
129:154–169. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shinya K, Shimada A, Ito T, Otsuki K,
Morita T, Tanaka H, Takada A, Kida H and Umemura T: Avian influenza
virus intranasally inoculated infects the central nervous system of
mice through the general visceral afferent nerve. Arch Virol.
145:187–195. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Reinacher M, Bonin J, Narayan O and
Scholtissek C: Pathogenesis of neurovirulent influenza A virus
infection in mice. Route of entry of virus into brain determines
infection of different populations of cells. Lab Invest.
49:686–692. 1983.PubMed/NCBI
|
|
8
|
Jadidi-Niaragh F and Mirshafiey A:
Histamine and histamine receptors in pathogenesis and treatment of
multiple sclerosis. Neuropharmacology. 59:180–189. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chastain EM, Duncan DS, Rodgers JM and
Miller SD: The role of antigen presenting cells in multiple
sclerosis. Biochim Biophys Acta. 1812:265–274. 2011. View Article : Google Scholar
|
|
10
|
Czirr E and Wyss-Coray T: The immunology
of neurodegeneration. J Clin Invest. 122:1156–1163. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ransohoff RM and Perry VH: Microglial
physiology: Unique stimuli, specialized responses. Annu Rev
Immunol. 27:119–145. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Perry VH and Teeling J: Microglia and
macrophages of the central nervous system: the contribution of
microglia priming and systemic inflammation to chronic
neurodegeneration. Sem Immunopathol. 35:601–612. 2013. View Article : Google Scholar
|
|
13
|
Schwartz M, Kipnis J, Rivest S and Prat A:
How do immune cells support and shape the brain in health, disease,
and aging? J Neurosci. 33:17587–17596. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sofroniew MV and Vinters HV: Astrocytes:
Biology and pathology. Acta Neuropathol. 119:7–35. 2010. View Article : Google Scholar
|
|
15
|
Wyss-Coray T and Mucke L: Inflammation in
neurodegenerative disease - a double-edged sword. Neuron.
35:419–432. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lull ME and Block ML: Microglial
activation and chronic neurodegeneration. Neurotherapeutics.
7:354–365. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Das Sarma J: Microglia-mediated
neuroinflammation is an amplifier of virus-induced neuropathology.
J Neurovirol. 20:122–136. 2014. View Article : Google Scholar
|
|
18
|
Glass CK, Saijo K, Winner B, Marchetto MC
and Gage FH: Mechanisms underlying inflammation in
neurodegeneration. Cell. 140:918–934. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Teeling JL and Perry VH: Systemic
infection and inflammation in acute CNS injury and chronic
neurodegeneration: Underlying mechanisms. Neuroscience.
158:1062–1073. 2009. View Article : Google Scholar
|
|
20
|
Taylor JP, Hardy J and Fischbeck KH: Toxic
proteins in neurodegenerative disease. Science. 296:1991–1995.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chevalier-Larsen E and Holzbaur EL: Axonal
transport and neurodegenerative disease. Biochim Biophys Acta.
1762:1094–1108. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen H and Chan DC: Mitochondrial dynamics
- fusion, fission, movement, and mitophagy - in neurodegenerative
diseases. Hum Mol Genet. 18(R2): R169–R176. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Allison DJ and Ditor DS: The common
inflammatory etiology of depression and cognitive impairment: A
therapeutic target. J Neuroinflammation. 11:1512014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
de Leeuw FE, de Groot JC, Oudkerk M,
Witteman JC, Hofman A, van Gijn J and Breteler MM: Hypertension and
cerebral white matter lesions in a prospective cohort study. Brain.
125:765–772. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schiffrin EL: Inflammation, immunity and
development of essential hypertension. J Hypertens. 32:228–229.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shimizu M, Ishikawa J, Yano Y, Hoshide S,
Shimada K and Kario K: The relationship between the morning blood
pressure surge and low-grade inflammation on silent cerebral
infarct and clinical stroke events. Atherosclerosis. 219:316–321.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tousoulis D, Kampoli AM, Papageorgiou N,
Androulakis E, Antoniades C, Toutouzas K and Stefanadis C:
Pathophysiology of atherosclerosis: The role of inflammation. Curr
Pharm Des. 17:4089–4110. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Di Napoli M, Godoy DA, Campi V, Masotti L,
Smith CJ, Parry Jones AR, Hopkins SJ, Slevin M, Papa F, Mogoanta L,
et al: C-reactive protein in intracerebral hemorrhage: Time course,
tissue localization, and prognosis. Neurology. 79:690–699. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Di Napoli M, Parry-Jones AR, Smith CJ,
Hopkins SJ, Slevin M, Masotti L, Campi V, Singh P, Papa F,
Popa-Wagner A, et al: C-reactive protein predicts hematoma growth
in intracerebral hemorrhage. Stroke. 45:59–65. 2014. View Article : Google Scholar
|
|
30
|
Rizzo M, Corrado E, Coppola G, Muratori I,
Mezzani A, Novo G and Novo S: The predictive role of C-reactive
protein in patients with hypertension and subclinical
atherosclerosis. Intern Med J. 39:539–545. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rizzo M, Corrado E, Coppola G, Muratori I,
Novo G and Novo S: Markers of inflammation are strong predictors of
subclinical and clinical atherosclerosis in women with
hypertension. Coron Artery Dis. 20:15–20. 2009. View Article : Google Scholar
|
|
32
|
Goossens GH: The role of adipose tissue
dysfunction in the pathogenesis of obesity-related insulin
resistance. Physiol Behav. 94:206–218. 2008. View Article : Google Scholar
|
|
33
|
Goossens GH, Bizzarri A, Venteclef N,
Essers Y, Cleutjens JP, Konings E, Jocken JW, Čajlaković M,
Ribitsch V, Clément K, et al: Increased adipose tissue oxygen
tension in obese compared with lean men is accompanied by insulin
resistance, impaired adipose tissue capillarization, and
inflammation. Circulation. 124:67–76. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Howcroft TK, Campisi J, Louis GB, Smith
MT, Wise B, Wyss-Coray T, Augustine AD, McElhaney JE, Kohanski R
and Sierra F: The role of inflammation in age-related disease.
Aging (Albany NY). 5:84–93. 2013. View Article : Google Scholar
|
|
35
|
Olefsky JM and Glass CK: Macrophages,
inflammation, and insulin resistance. Annu Rev Physiol. 72:219–246.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Bruunsgaard H, Pedersen M and Pedersen BK:
Aging and proinflammatory cytokines. Curr Opin Hematol. 8:131–136.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fagiolo U, Cossarizza A, Santacaterina S,
Ortolani C, Monti D, Paganelli R and Franceschi C: Increased
cytokine production by peripheral blood mononuclear cells from
healthy elderly people. Ann N Y Acad Sci. 663:490–493. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fagiolo U, Amadori A, Cozzi E, Bendo R,
Lama M, Douglas A and Palù G: Humoral and cellular immune response
to influenza virus vaccination in aged humans. Aging (Milano).
5:451–458. 1993.
|
|
39
|
Fagiolo U, Cossarizza A, Scala E,
Fanales-Belasio E, Ortolani C, Cozzi E, Monti D, Franceschi C and
Paganelli R: Increased cytokine production in mononuclear cells of
healthy elderly people. Eur J Immunol. 23:2375–2378. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Johnson FA, Dawson AJ and Meyer RL:
Activity-dependent refinement in the goldfish retinotectal system
is mediated by the dynamic regulation of processes withdrawal: An
in vivo imaging study. J Comp Neurol. 406:548–562. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Capuron L, Su S, Miller AH, Bremner JD,
Goldberg J, Vogt GJ, Maisano C, Jones L, Murrah NV and Vaccarino V:
Depressive symptoms and metabolic syndrome: Is inflammation the
underlying link? Biol Psychiatry. 64:896–900. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ouchi N, Parker JL, Lugus JJ and Walsh K:
Adipokines in inflammation and metabolic disease. Nat Rev Immunol.
11:85–97. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
43
|
Capuron L, Poitou C, Machaux-Tholliez D,
Frochot V, Bouillot JL, Basdevant A, Layé S and Clément K:
Relationship between adiposity, emotional status and eating
behaviour in obese women: Role of inflammation. Psychol Med.
41:1517–1528. 2011. View Article : Google Scholar
|
|
44
|
Cancello R, Henegar C, Viguerie N, Taleb
S, Poitou C, Rouault C, Coupaye M, Pelloux V, Hugol D, Bouillot JL,
et al: Reduction of macrophage infiltration and chemoattractant
gene expression changes in white adipose tissue of morbidly obese
subjects after surgery-induced weight loss. Diabetes. 54:2277–2286.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
McCrimmon RJ, Ryan CM and Frier BM:
Diabetes and cognitive dysfunction. Lancet. 379:2291–2299. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
McIntyre RS, Soczynska JK, Konarski JZ,
Woldeyohannes HO, Law CW, Miranda A, Fulgosi D and Kennedy SH:
Should depressive syndromes be reclassified as 'metabolic syndrome
type II'? Ann Clin Psychiatry. 19:257–264. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wolkowitz OM, Epel ES, Reus VI and Mellon
SH: Depression gets old fast: Do stress and depression accelerate
cell aging? Depress Anxiety. 27:327–338. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Rush AJ, Trivedi MH, Wisniewski SR,
Nierenberg AA, Stewart JW, Warden D, Niederehe G, Thase ME, Lavori
PW, Lebowitz BD, et al: Acute and longer-term outcomes in depressed
outpatients requiring one or several treatment steps: A STAR*D
report. Am J Psychiatry. 163:1905–1917. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Baune BT, Smith E, Reppermund S, Air T,
Samaras K, Lux O, Brodaty H, Sachdev P and Trollor JN: Inflammatory
biomarkers predict depressive, but not anxiety symptoms during
aging: The prospective Sydney Memory and Aging Study.
Psychoneuroendocrinology. 37:1521–1530. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Najjar S, Pearlman DM, Devinsky O, Najjar
A and Zagzag D: Neurovascular unit dysfunction with blood-brain
barrier hyper-permeability contributes to major depressive
disorder: A review of clinical and experimental evidence. J
Neuroinflammation. 10:1422013. View Article : Google Scholar
|
|
51
|
Zunszain PA, Hepgul N and Pariante CM:
Inflammation and depression. Curr Top Behav Neurosci. 14:135–151.
2013. View Article : Google Scholar
|
|
52
|
Kessler RC, Berglund P, Demler O, Jin R,
Merikangas KR and Walters EE: Lifetime prevalence and age-of-onset
distributions of DSM-IV disorders in the National Comorbidity
Survey Replication. Arch Gen Psychiatry. 62:593–602. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Papakostas GI, Shelton RC, Kinrys G, Henry
ME, Bakow BR, Lipkin SH, Pi B, Thurmond L and Bilello JA:
Assessment of a multi-assay, serum-based biological diagnostic test
for major depressive disorder: A pilot and replication study. Mol
Psychiatry. 18:332–339. 2013. View Article : Google Scholar
|
|
54
|
Rivest S: Regulation of innate immune
responses in the brain. Nat Rev Immunol. 9:429–439. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yang WX, Terasaki T, Shiroki K, Ohka S,
Aoki J, Tanabe S, Nomura T, Terada E, Sugiyama Y and Nomoto A:
Efficient delivery of circulating poliovirus to the central nervous
system independently of poliovirus receptor. Virology. 229:421–428.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Aronsson F, Robertson B, Ljunggren HG and
Kristensson K: Invasion and persistence of the neuroadapted
influenza virus A/WSN/33 in the mouse olfactory system. Viral
Immunol. 16:415–423. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Schnell G, Joseph S, Spudich S, Price RW
and Swanstrom R: HIV-1 replication in the central nervous system
occurs in two distinct cell types. PLoS Pathog. 7:e10022862011.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Chen L, Liu J, Xu C, Keblesh J, Zang W and
Xiong H: HIV-1gp120 induces neuronal apoptosis through enhancement
of 4-aminopyridine-senstive outward K+ currents. PLoS One.
6:e259942011. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Chang JR, Mukerjee R, Bagashev A, Del
Valle L, Chabrashvili T, Hawkins BJ, He JJ and Sawaya BE: HIV-1 Tat
protein promotes neuronal dysfunction through disruption of
microRNAs. J Biol Chem. 288:85642013. View Article : Google Scholar
|
|
60
|
Brew BJ, Crowe SM, Landay A, Cysique LA
and Guillemin G: Neurodegeneration and ageing in the HAART era. J
Neuroimmune Pharmacol. 4:163–174. 2009. View Article : Google Scholar
|
|
61
|
Noh H, Jeon J and Seo H: Systemic
injection of LPS induces region-specific neuroinflammation and
mitochondrial dysfunction in normal mouse brain. Neurochem Int.
69:35–40. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhou L, Miranda-Saksena M and Saksena NK:
Viruses and neurodegeneration. Virol J. 10:1722013. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ostanin DV, Bao J, Koboziev I, Gray L,
Robinson-Jackson SA, Kosloski-Davidson M, Price VH and Grisham MB:
T cell transfer model of chronic colitis: Concepts, considerations,
and tricks of the trade. Am J Physiol Gastrointest Liver Physiol.
296:G135–G146. 2009. View Article : Google Scholar :
|
|
64
|
Huber S, Schramm C, Lehr HA, Mann A,
Schmitt S, Becker C, Protschka M, Galle PR, Neurath MF and Blessing
M: Cutting edge: TGF-β signaling is required for the in vivo
expansion and immunosuppressive capacity of regulatory CD4+CD25+ T
cells. J Immunol. 173:6526–6531. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Martinez FO and Gordon S: The M1 and M2
paradigm of macrophage activation: time for reassessment.
F1000Prime Rep. 6:132014. View
Article : Google Scholar : PubMed/NCBI
|
|
66
|
Crutcher KA, Gendelman HE, Kipnis J,
Perez-Polo JR, Perry VH, Popovich PG and Weaver LC: Debate: 'is
increasing neuroinflammation beneficial for neural repair?'. J
Neuroimmune Pharmacol. 1:195–211. 2006. View Article : Google Scholar
|
|
67
|
Popovich PG and Longbrake EE: Can the
immune system be harnessed to repair the CNS? Nat Rev Neurosci.
9:481–493. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Tansey MG, McCoy MK and Frank-Cannon TC:
Neuroinflammatory mechanisms in Parkinson's disease: Potential
environmental triggers, pathways, and targets for early therapeutic
intervention. Exp Neurol. 208:1–25. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Schmid CD, Melchior B, Masek K,
Puntambekar SS, Danielson PE, Lo DD, Sutcliffe JG and Carson MJ:
Differential gene expression in LPS/IFNgamma activated microglia
and macrophages: In vitro versus in vivo. J Neurochem. 109(Suppl
1): 117–125. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Block ML and Hong JS: Microglia and
inflammation-mediated neurodegeneration: Multiple triggers with a
common mechanism. Prog Neurobiol. 76:77–98. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
González H, Elgueta D, Montoya A and
Pacheco R: Neuroimmune regulation of microglial activity involved
in neuroinflammation and neurodegenerative diseases. J
Neuroimmunol. 274:1–13. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Henkel JS, Engelhardt JI, Siklós L,
Simpson EP, Kim SH, Pan T, Goodman JC, Siddique T, Beers DR and
Appel SH: Presence of dendritic cells, MCP-1, and activated
microglia/macrophages in amyotrophic lateral sclerosis spinal cord
tissue. Ann Neurol. 55:221–235. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Mrak RE and Griffin WST: Glia and their
cytokines in progression of neurodegeneration. Neurobiol Aging.
26:349–354. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Rock RB, Gekker G, Hu S, Sheng WS, Cheeran
M, Lokensgard JR and Peterson PK: Role of microglia in central
nervous system infections. Clin Microbiol Rev. 17:942–964. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Qian L, Tan KS, Wei SJ, Wu HM, Xu Z,
Wilson B, Lu RB, Hong JS and Flood PM: Microglia-mediated
neurotoxicity is inhibited by morphine through an opioid
receptor-independent reduction of NADPH oxidase activity. J
Immunol. 179:1198–1209. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Gordon R, Anantharam V, Kanthasamy AG and
Kanthasamy A: Proteolytic activation of proapoptotic kinase protein
kinase Cδ by tumor necrosis factor α death receptor signaling in
dopaminergic neurons during neuroinflammation. J Neuroinflammation.
9:822012. View Article : Google Scholar
|
|
77
|
Magro F, Fraga S, Ribeiro T and
Soares-da-Silva P: Decreased availability of intestinal dopamine in
transmural colitis may relate to inhibitory effects of interferon-γ
upon L-DOPA uptake. Acta Physiol Scand. 180:379–386. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Panaro MA, Lofrumento DD, Saponaro C, De
Nuccio F, Cianciulli A, Mitolo V and Nicolardi G: Expression of
TLR4 and CD14 in the central nervous system (CNS) in a MPTP mouse
model of Parkinson's-like disease. Immunopharmacol Immunotoxicol.
30:729–740. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Querfurth HW and LaFerla FM: Alzheimer's
disease. N Engl J Med. 362:329–344. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Braak H, Del Tredici K, Rüb U, de Vos RA,
Jansen Steur EN and Braak E: Staging of brain pathology related to
sporadic Parkinson's disease. Neurobiol Aging. 24:197–211. 2003.
View Article : Google Scholar
|
|
81
|
Saccon RA, Bunton-Stasyshyn RK, Fisher EM
and Fratta P: Is SOD1 loss of function involved in amyotrophic
lateral sclerosis? Brain. 136:2342–2358. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Lu L, Lan Q, Li Z, Zhou X, Gu J, Li Q,
Wang J, Chen M, Liu Y, Shen Y, et al: Critical role of all-trans
retinoic acid in stabilizing human natural regulatory T cells under
inflammatory conditions. Proc Natl Acad Sci USA. 111:E3432–E3440.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Appel SH: CD4+ T cells mediate
cytotoxicity in neurodegenerative diseases. J Clin Invest.
119:13–15. 2009.
|
|
84
|
Reynolds AD, Stone DK, Mosley RL and
Gendelman HE: Proteomic studies of nitrated alpha-synuclein
microglia regulation by CD4+CD25+ T cells. J Proteome Res.
8:3497–3511. 2009. View Article : Google Scholar : PubMed/NCBI
|