Introduction
Numerous previous studies have demonstrated that
cancer stem cells (CSCs) are a key obstacle in providing effective
treatment and improving survival rate in patients with cancer
(1–3). CSCs are a small population within the
heterogeneous tumor that escape conventional therapies and are
responsible for minimal residual disease (4). Previous studies involving solid
tumors have reported that CSCs are potentiators of differentiation
and drug resistance, and are highly self-renewal due to the
expression of stem cell surface proteins, including CD133, CD44,
octamer-binding transcription factor 4 (Oct-4) and Nestin, which
contribute to metastasis and tumor invasion (5–8).
Therefore, improved characterization of CSCs and the elucidation of
additional signaling factors involved in CSC-mediated therapy
failure and tumor recurrence are crucial for providing effective
cancer treatment.
Hepatocellular carcinoma (HCC) is one of the most
common types of cancer, and is associated with a high mortality
rate. HCC is frequently diagnosed at the stage of metastases to the
lungs, adrenal and lymph nodes, and therefore the overall survival
rate of patients is poor following treatment (1,9,10).
Previous studies in liver cancer reported that the upregulation of
stemness and anti-apoptotic genes, the enhanced expression of
dysadherin and the overproduction of chemokines o serve crucial
roles in therapy failure, cancer metastases and invasion (2,3,11,12).
In addition, point mutations in the p53 gene and the inactivation
of Bcl-2 leads to the upregulation of anti-apoptotic pathways in
CSCs (13). Furthermore, previous
studies in breast, liver and colon cancer (5,6,14)
have suggested that the Wnt/β-catenin pathway is involved in the
regulation of CSC survival maintenance. Therefore, the current
study investigated the Wnt/β-catenin pathway and Wnt/β-catenin
targeted gene expression in CSCs from liver cancer samples. In
addition, the Wnt/β-catenin pathway was investigated in cancer
stem-like side-population (SP) liver cancer cells for its
involvement in proliferation, tumorigenicity and invasiveness.
Materials and methods
Sample collection and cell culture
HCC samples were obtained from patients during
surgery according to the ethical principle approved by the
Department of Hepatopancreatobiliary Surgery, Second Affiliated
Hospital, School of Medicine, Zhejiang University (Hangzhou,
China). In total, samples were obtained from 30 patients (15 male
and 15 female; age, 49–57) undergoing surgery at the Department of
Hepatopancreatobiliary Surgery. The collected tumor samples were
minced to fine fragments and cultured in 1 ml fetal bovine serum
(FBS; Gibco, Thermo Fisher Scientific, Inc., Waltham, MA, USA). For
the subsequent culturing, Dulbecco's modified Eagle's medium (DMEM;
Sigma-Aldrich, St. Louis, MO, USA) with 10% FBS, supplemented with
100 U/ml penicillin, and 100 μg/ml streptomycin (Invitrogen;
Thermo Fisher Scientific, Inc.) was used. Cultures were maintained
in T-75 flasks at 37°C in a humidified 5% CO2 and 95%
air atmosphere. Once cells were 90% confluent, they were removed
from the culture flask using Trypsin-ethylenediaminetetraacetic
acid (EDTA EDTA, Sigma-Aldrich) [0.25% 53 mM (EDTA)]. Cells were
then washed and suspended in DMEM with 10% FBS and centrifuged at
4,000 × g for 6 min. Cells were resuspended in DMEM with 10% FBS,
and cells were counted using a Bulker-Turk hemocytometer (Sunlead
Glass Corp., Saitama, Japan).
Fluorescence-activated cell sorting
(FACS) analysis
Cells were cultured in DMEM with 10% FBS,
supplemented with antibiotics and maintained in T-75 flasks at 37°C
in a humidified 5% CO2 and 95% air atmosphere. Once
cells were 90% confluent, they were removed from the culture flask
using Trypsin-EDTA (0.25% 53 mM EDTA), washed and suspended in DMEM
with 10% FBS. A cell count was taken using hemocytometer. The study
groups were as follows: Control, cells labeled with Hoechst 33342
dye (Sigma-Aldrich) alone (n=9); and drug-treated cells, treated
with verapamil (Sigma-Aldrich) and Hoechst 33342 dye (n=9). Cells
were counted using a hemocytometer, and ~106 cells/ml in
DMEM with 10% FBS were labeled with Hoechst 33342-bis-benzimide (5
μl/ml) either with dye alone or in combination with drug
[ABC transporter inhibitor verapamil (50 μmol/l)]. Further,
cells were counter stained with 2 μg/ml propidium iodide
(Sigma-Aldrich). The cells were sorted using a FACS Aria II flow
cytometer (BD Biosciences, Franklin Lakes, CA, USA) and the sorted
cells were cultured and maintained in DMEM/F-12 supplemented with
10% FBS. The Hoechst 33342 emission was first split by using a 610
nm dichroic short-pass filter, and the red and the blue emissions
were collected through 670/30 and 450/65 nm band pass filters,
respectively.
Cell resistance assay
In total ~1×103 cells/well were cultured
in 96-well plates and treated with the chemotherapeutic drugs at
the following concentrations: 10 μ/ml 5-fluorouracil (5-FU),
250 mM gemcitabine, 100 mM oxaliplatin, 30 ng/ml paclitaxel, 5
mg/ml cisplatin, 10 mg/ml etoposide and 2 μg/ml oxaliplatin
(all from Sigma-Aldrich). The mean value of the optical density
(OD) 450 obtained was represented as a graph. Cell resistance in
each group was calculated using the following formula: Cell
resistance rate (%) = (experimental group OD450 value / control
group OD450 value) × 100. The values presented in the graph are the
average of three independent experiments. OD values were determined
using a spectrophotometer (Multiskan FC Microplate Photometer,
Thermo Scientific, Inc.)
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted, and 2 μg RNA was
reverse transcribed into cDNA using a Reverse Transcriptase kit
(Fermentas; Thermo Fisher Scientific, Inc.). RT-qPCR analysis was
subsequently performed on an iCycler IQ Real-Time PCR Detection
system (Bio-Rad Laboratories, Inc., Hercules, CA, USA), using IQ
Supermix with SYBR-Green (Bio-Rad Laboratories, Inc.). The
sequences of the human specific primers used were as follows
(5′-3′): CD133, forward TCT TGA CCG ACT GAG AC and reverse ACT TGA
TGG ATG CAC CAA GCA C); cyclin D1 (CCND1), forward TGA TGC TGG GCA
CTT CAT CTG and reverse TCC AAT CAT CCC GAA TGA GAG TC); Oct-4,
forward ATC CTG GGG GTT CTA TTT GG and reverse CTC CAG GTT GCC TCT
CAC TC); ATP-binding cassette (ABC) subfamily B member 5 (ABCB5),
forward CAC AAG TTG GAC TGA AAG GA and reverse ACC ACT AGG CAT GTC
CTT CC); glyceraldehyde-3-phosphate dehydrogenase (GAPDH), forward
GCA CCG TCA AGG CTG AGA AC and reverse TGG TGA AGA CGC CAG TGG A);
ABC subfamily G member 2 (ABCG2), forward TCA ATC AAA GTG CTT CTT
TTT TATG and reverse TTG TGG AAG AAT CAC GTG GC); multidrug
resistance protein 1 (MDR1), forward ACA GGA AGA GAT TGT GAG GG and
reverse TAT CCA GAG CTG ACG TGG CT); axis inhibition protein 2
(AXIN2), forward CTG GCT TTG GTG AAC TGT TG and reverse AGT TGC TCA
CAG CCA AGA CA); and dickkopf Wnt signaling pathway inhibitor 1
(DKK1), forward AGC ACC TTG GAT GGG TAT TC and reverse CAC AAT CCT
GAG GCA CAG TC (15,16). The PCR cycling conditions were as
follows: 58°C for 1 min and 96°C for 5 min, followed by 40 cycles
of denaturation at 95°C for 15 sec, annealing at 58°C for 30 sec
and extension at 72°C for 60 sec. A final extension step was
conducted at 72°C for 10 min. GAPDH was used as a housekeeping gene
(primers: Forward AATCCCATCACCATCTTCCA and reverse
TGGACTCCACGACGTACTCA). The values presented in the quantification
graph are the average values of three independent experiments. The
2−ΔΔCq method was used for the quantification of the
RT-qPCR results (17).
Luciferase assay
A total of ~106 cells were seeded into
12-well plates and were transfected with 100 ng TOPflash or
FOPflash (Upstate Cell Signaling Solutions, Billerica, MA, USA)
using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific,
Inc.), A total of 20 ng p-cytomegalovirus-Renilla luciferase
reporter Promega Corporation (Madison, WI, USA) was cotransfected
as an internal control. Cell lysates were collected at 24 h
post-transfection and the luciferase activity was measured using
the Dual-Luciferase Reporter Assay system (Promega Corporation)
according to the manufacturer's instructions.
RNA interference
A small interfering RNA (siRNA) sequence specific to
β-catenin (GeneBank accession no. CTNNB1, NM001904), was purchased
from Dharmacon (GE Life Sciences, Lafayette, CO, USA). siRNA
transfection with a final concentration of 200 nm was conducted
using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific,
Inc.) according to the manufacturer's instructions. A scramble
siRNA sequence (5′-TTCTCCGAACGTGTCACGT-3′) was used as a control
(Gima Biol Engineering Inc., Shanghai, China). The transfected
siRNA cells were analyzed following 48 h of transfection.
Invasion assay
The cellular invasiveness of SP and non-SP cells was
investigated using 6-well Matrigel invasion chambers (BD
Biosciences). Cells were seeded in DMEM at a density of
2×105/insert. Outer wells were filled with DMEM
containing 5% FBS as a chemoattractant and incubated at 37°C for 48
h. Subsequently, the non-invading cells were washed by swabbing the
top layer of the Matrigel with a Q-tip. The membrane containing the
invading cells was stained with hematoxylin for 3 min, washed and
mounted on slides. The entire membrane containing the invading
cells was counted using a CX31 light microscope (Olympus, Tokyo,
Japan) at 40× objective. The values presented in the graph are the
mean value of three independent experiments.
Western blotting
Cell extracts were harvested from the SP and non-SP
cells using RIPA buffer (Sigma-Aldrich) containing protease
inhibitor cocktail (Roche Diagnostics Deutschland GmbH, Mannheim,
Germany) and protein concentration was determined using a Bradford
assay (Sigma-Aldrich) (18).
Protein lysates (40 μg) from each sample were subjected to
10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis
(Sigma-Aldrich). Separated proteins were transferred to a
polyvinylidene difluoride membrane (Sigma-Aldrich). The membranes
were treated with the primary antibodies against β-catenin
(1:2,400; cat. no. ab6302; Abcam, Shanghai, China) and GAPDH (0.7
μg/ml; cat. no. ab37168, Abcam). Subsequently, the membranes
were incubated in the secondary antibodies horseradish
peroxidase-conjugated secondary antibody (goat anti-rabbit IgG with
alkaline phosphatase markers; cat. no. ab97048, Abcam). The protein
was detected using chemiluminescence reagents (Amersham
Biosciences). Blots were scanned using a Bio-Rad GS-710
densitometer (Bio-Rad Laboratories, Inc.).
In vitro proliferation activity
The sorted SP and non-SP cells were seeded in a
96-well plate at 2×106 cells/well and then cultured in a
CO2 incubator. Each group was analyzed in triplicate.
Cell proliferation was measured daily for 7 days. Each well was
supplemented with Cell Counting kit-8 solution (10 μl;
Dojindo Laboratories, Kumamoto, Japan) and incubated in a
CO2 incubator for 2–3 h. The OD was determined at 450
nm. These data were used to calculate cell growth graphs based on
the mean value of OD450 and the standard deviation values for each
well.
Differentiation assay
Following 16–18 days of cell sorting, cells were
cultured in normal RPMI-1640 (Gibco; Thermo Fisher Scientific,
Inc.). Subsequently, differentiation ability of two subpopulations
was determined using an Olympus CX31 microscope.
Immunofluorescence staining
The sorted SP and non-SP cells were fixed onto glass
slides in ice-cold 4% formaldehyde (4°C, 10 min), and blocked with
blocked with 1% bovine serum albumin (Sigma-Aldrich) for 30 min.
Slides were incubated with mouse monoclonal anti-CD133 (1:200; cat.
no. ab5558; Abcam), anti-Oct-4 (1:100; cat. no. ab59545; Abcam) and
anti-ABCG2 (1:100; cat. no. ab3380; Abcam) for 1 h. Following
washing the slides with phosphate-buffered saline, slides were
incubated with fluorescein isothiocyante-conjugated chicken
anti-mouse IgG (1:200; cat. no. ab112455; Abcam) overnight in dark
room. Nuclei were counterstained with 4,6-diamidino-2-phenylindole
and viewed using a DMI 4000 B fluorescence microscope (Leica,
Wetzlar, Germany). All images were processed using Adobe Photoshop,
version CS6 (Adobe System, Inc., San Jose, CA, USA).
Sphere formation assay
The sorted SP cells and non-SP cells were plated at
a density of 1,000 cells/ml and suspended in tumor sphere medium
consisting of serum-free 1:1 mixture of Ham's F-12/DMEM, N2
supplement, 10 ng/ml human recombinant basic fibroblast growth
factor (Sigma-Aldrich) and 10 ng/ml epidermal growth
factor(Sigma-Aldrich), and subsequently cultured in ultra-low
attachment plates for ~2 weeks. Sorted SP and non-SP cells were
seeded at a low density of 20 cells/l and the number of generated
spheres (>100 μM) was counted following 7 days of
culture. The values presented in the graph are the mean values of
three independent experiments.
Tumor cell implantation
NOD SCID mice (n=20; age, 6 weeks) were purchased
from the Hubei Provincial Center of Disease Control and Prevention
(Wuhan, China). They were kept in individually ventilated cages
with access to food and water ad libitum. Mice were kept in
a 14/10 h light/dark cycle at 25°C and 55–60%.humidity as approved
by Institutional Ethical Committee of the School of Medicine
(Zhejiang University, Hangzhou, China). FACS sorted SP and non-SP
cells (4×105 cells) were mixed with Matrigel and
administered to NOD/SCID mice by subcutaneous injection. The
subsequent steps were conducted as described previously (19). At 21 days following implantation,
mice were sacrificed by cervical dislocation and tumors were
harvested and observed.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism 5 software (GraphPad Software, Inc., La Jolla, CA, USA). Data
are represented as the mean ± standard deviation. Statistical
differences between the experimental groups were analyzed by
Student's t-test, one-way analysis of variance and with post-hoc
test where needed. P<0.05 was considered to indicate a
statistically significant difference.
Results
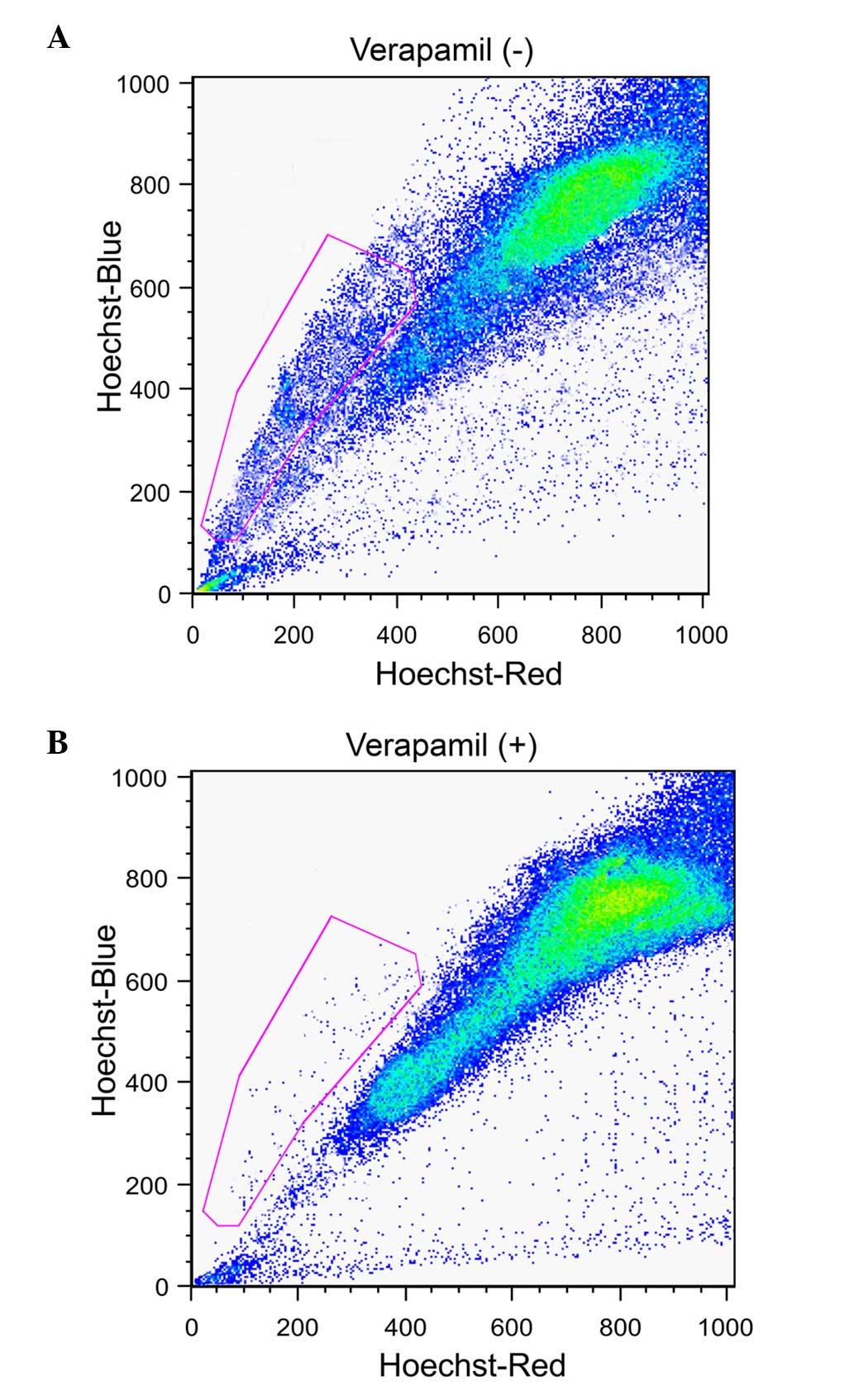
Existence of CSCs in liver cancer
The liver cancer samples were assessed for the
presence of cancer stem-like SP cells using a FACS-based Hoeschst
33342 dye exclusion method. This identified a small population of
SP cells (3.6%; Fig. 1A, gated
region) whose presence was significantly reduced to 0.6% (Fig. 1B, gated region) following treatment
with verapamil. The function of ABC transporter proteins may be
efficiently blocked by verapamil and therefore the SP cell
population was significantly reduced.
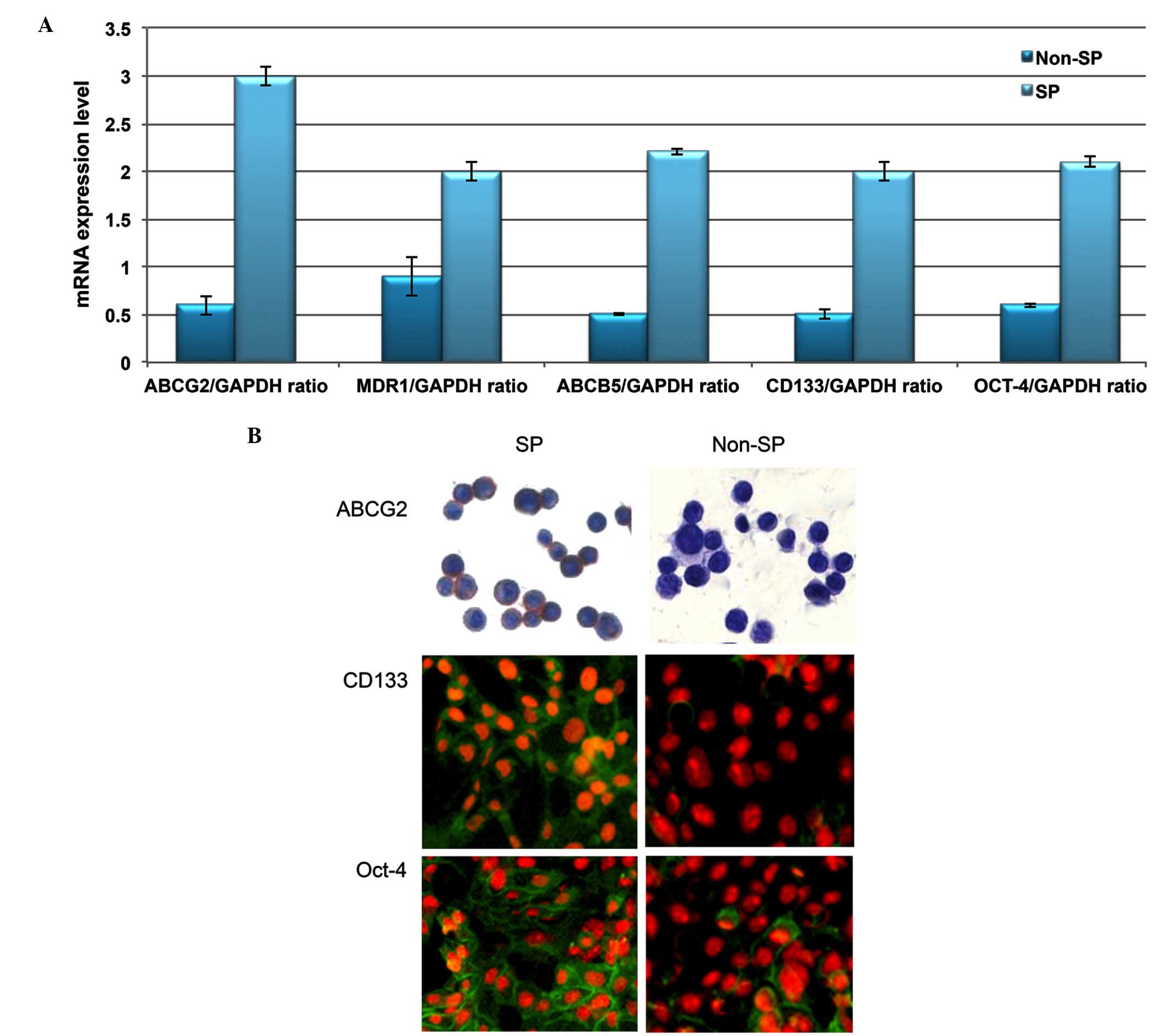
Phenotypic characterization of liver
cancer SP cells
Using RT-qPCR analysis, the transcriptional
regulation of drug efflux genes (ABCG2, MDR1 and ABCB5) and stem
cell surface genes (CD133 and Oct-4) was investigated. Increased
levels of these genes were observed in SP cells compared with
non-SP cells (Fig. 2A). In
addition, the SP cells exhibited enhanced staining intensity for
ABCG2, CD133 and Oct-4 (Fig. 2B).
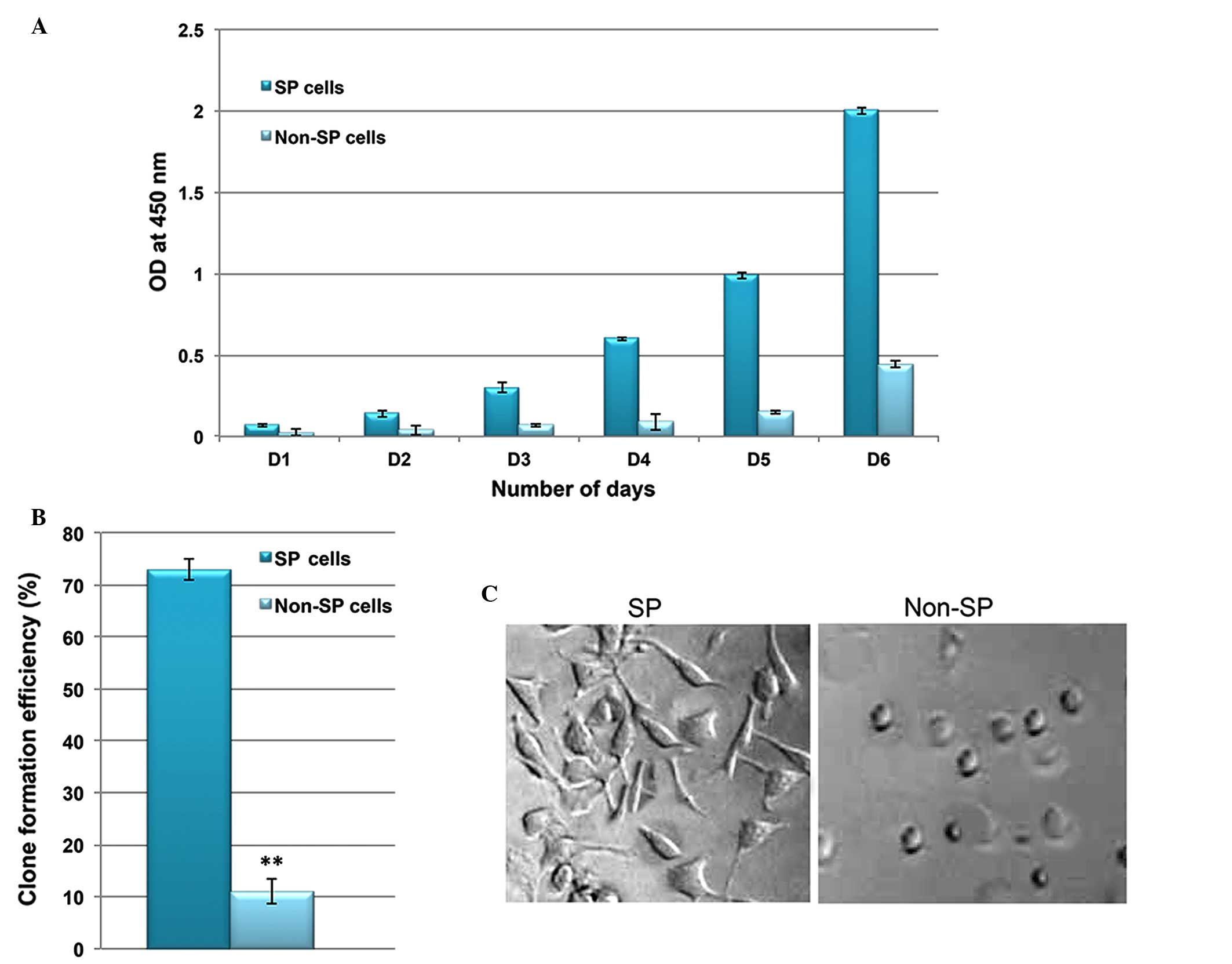
Furthermore, the in vitro proliferation and clone formation
efficiency assays indicated that liver cancer SP cells exhibit an
enhanced rate of proliferation, with a high potential for
generating tumor spheres compared with non-SP cells (Fig. 3A and B). Additionally, SP cells
were observed to lose their normal morphological appearance
following 5–7 days in culture, SP cells began to form filamentous
structures, whereas the non-SP cells did not form these structures
(Fig. 3C). The SP cells are able
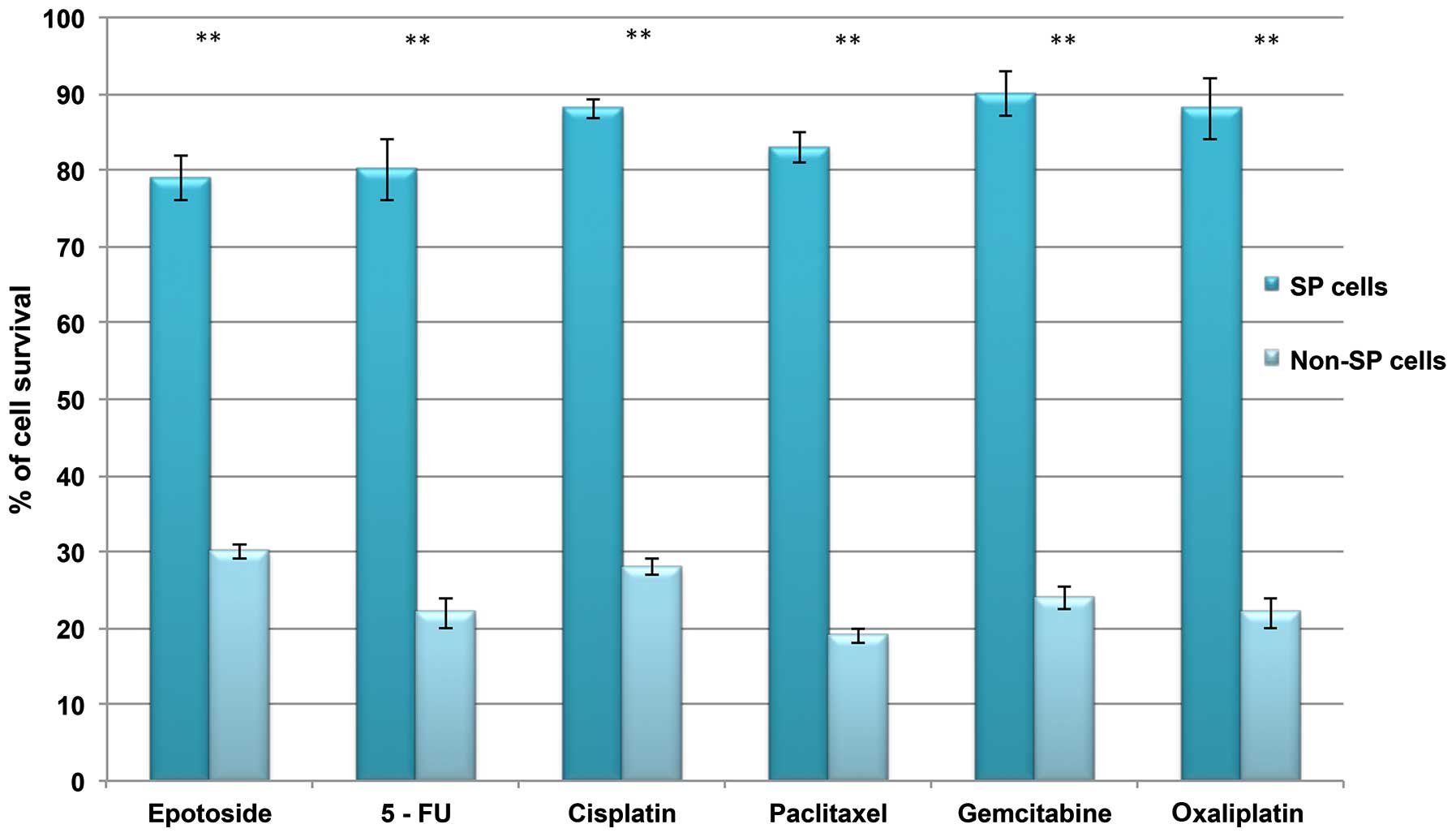
to resist DNA targeting drugs, including 5-FU, gemcitabine,
oxaliplatin, paclitaxel, cisplatin, etoposide and oxaliplatin, as
indicated by the increased cell survival rate in SP cells (Fig. 4). Together, these data suggest that
the presence of a small proportion of SP cells in liver cancer
which possess stem cell features may be responsible for
chemotherapeutic failure and tumor recurrence.
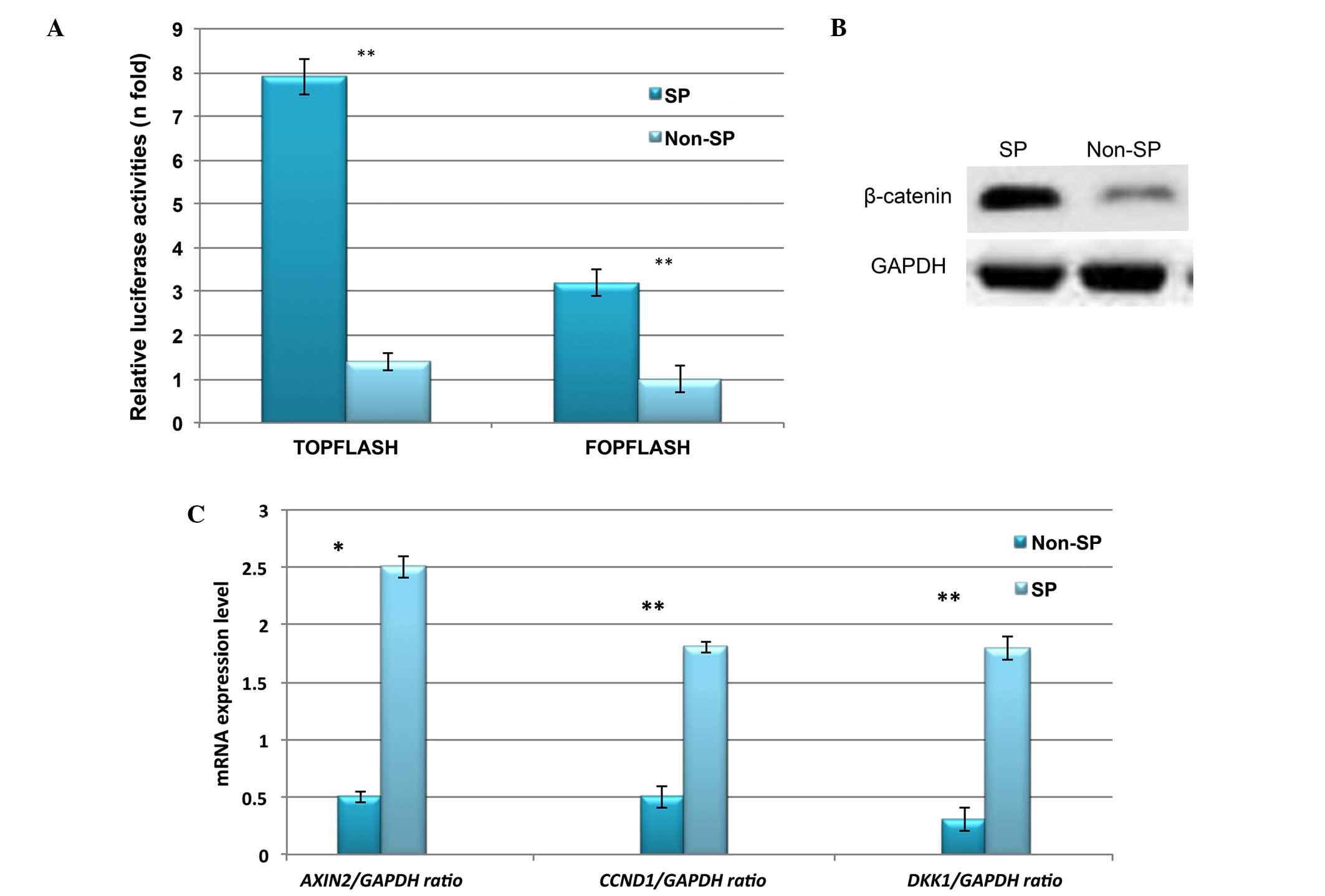
Constitutive expression of Wnt/β-catenin
signaling in liver cancer SP cells
Abnormal activation of β-catenin and its downstream
signaling targets, such as cyclin D1, have been demonstrated to be
involved in the enhanced proliferation and self-renewal of CSCs
(15,16). Therefore, the expression profile of
Wnt/β-catenin in liver cancer SP cells was investigated. Using
TOPflash and FOPflash luciferase reporter assays, the
transcriptional regulation of Wnt/β-catenin was observed to be
highly upregulated in liver cancer SP cells (Fig. 5A). In addition, the protein
expression of β-catenin was increased in SP cells (Fig. 5B). Furthermore, RT-qPCR indicated
significantly increased relative mRNA expression of Wnt/β-catenin
target genes including CCND1, DKK1 and AXIN2 (Fig. 5C). Together, these results suggest
that the abnormal activation of Wnt/β-catenin signaling in SP cells
may serve a role in the SP cell phenotype.
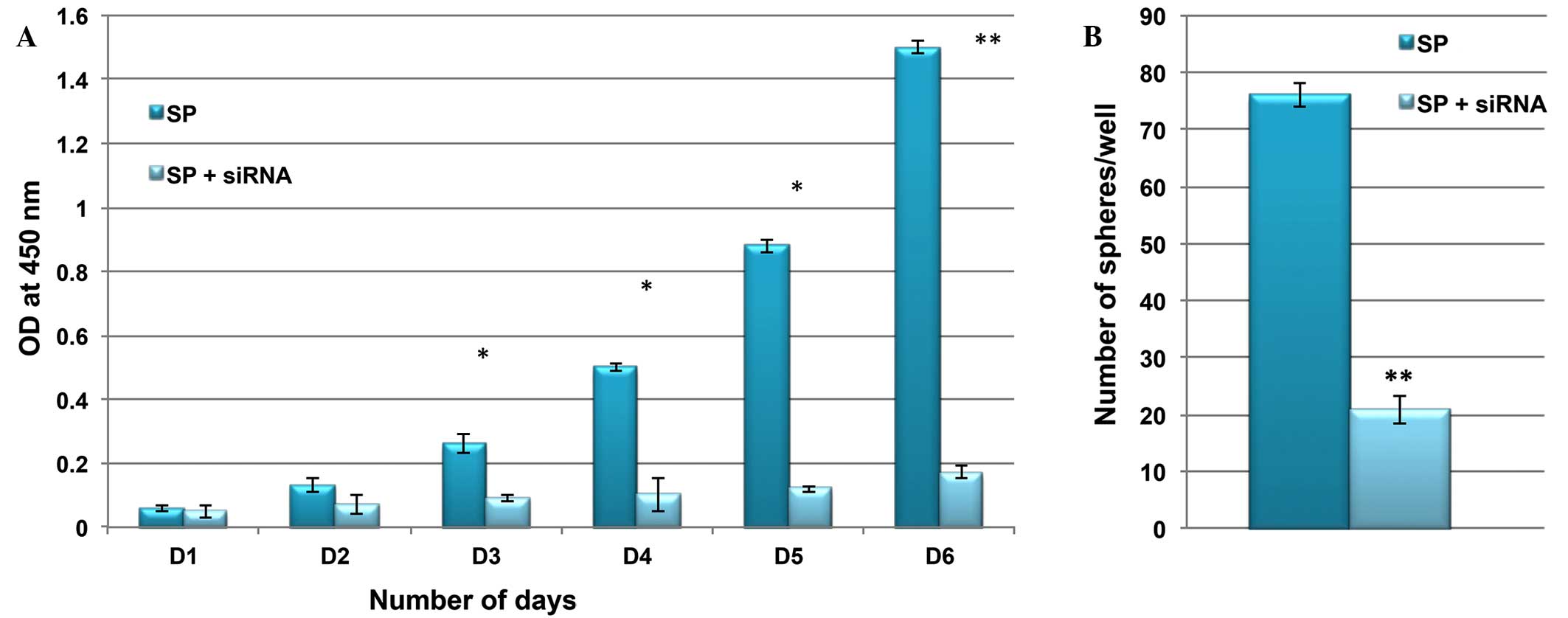
Knockdown of β-catenin suppresses the SP
cell phenotype
It was then investigated whether the inactivation of
Wnt/β-catenin signaling was able to suppress the rapid
proliferation and self-renewal of SP cells. Using siRNA technology,
β-catenin was inactivated in SP cells, and the rate of cell
proliferation and ability to form tumor spheres was compared
between control and siRNA SP cells. As presented in Fig. 6, the cell proliferation rate and
sphere generation capacity of SP cells were markedly reduced
following knockdown of the Wnt/β-catenin signaling pathway. These
results indicated that Wnt/β-catenin signaling and its target genes
are involved in the tumorigenic properties of CSCs.
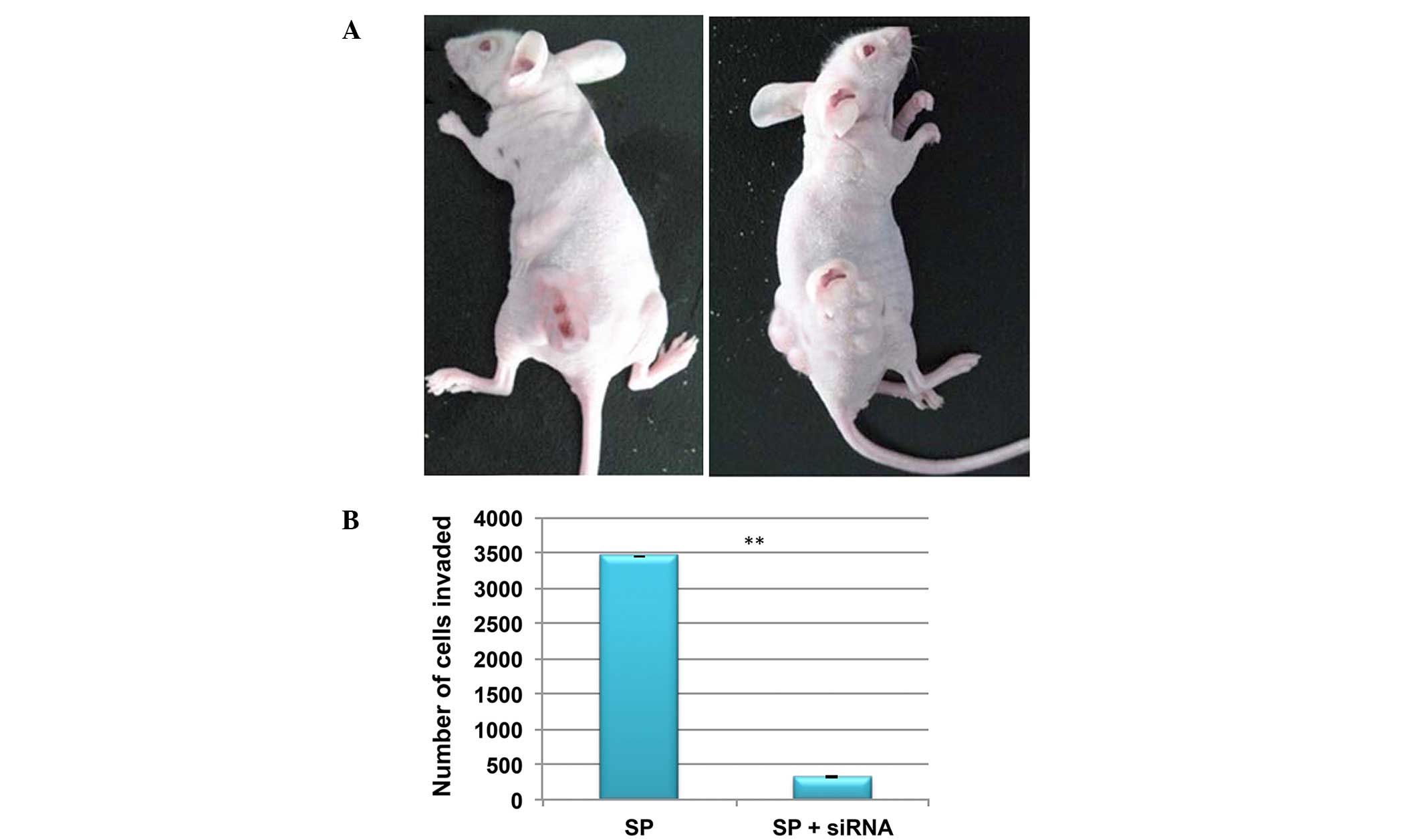
Knockdown of Wnt/β-catenin in SP cells
reduces invasion
In order to investigate tumorgenicity, a low density
(4×105 cells) of SP and non-SP cells were injected into
NOD/SCID mice. At this cell concentration, SP cells were able to
induce tumor growth in NOD/SCID mice, whereas the non-SP cells were
not (Fig. 7A). In addition, the
in vitro invasion assay demonstrated that the number of SP
cells invading the Matrigel was significantly reduced following the
knockdown of β-catenin (Fig. 7B).
Therefore, this suggests that Wnt/β-catenin signaling may
additionally contribute to CSC-mediated tumor invasion and
metastasis.
Discussion
To date, clinical and experimental studies in
several solid tumors have revealed that the presence of CSCs is a
major obstacle for treatment and the complete eradication of
refractory cancer (5,6,20–22).
Studies in HCC cell lines have additionally demonstrated the
existence of a small subset of cancer stem-like SP cells that are
resistant to chemotherapeutic drugs and are highly tumorigenic
(20–22). A noteworthy characteristic feature
of SP cells is the elevated expression of drug efflux transporter
proteins, such as ABCG1 and MDR1, which are actively involved in
expelling drugs out of the cell and thus result in therapy failure
and tumor recurrence (23–25). Therefore, it is high time to
improve the treatment strategies that may efficiently target the
tumor cell of origin.
In the current study, liver cancer samples were
observed to contain a small population of tumor initiating SP cells
which shared the features of CSCs. The liver cancer SP cells were
observed to induce the rapid formation of tumor spheres, due to the
increased expression of stem cell surface proteins. In addition,
increased transcriptional regulation of the levels of drug efflux
genes (ABCG2, MDR1 and ABCB5) were observed in the present study.
Previously, a crucial role of ABC transports in resistance to
chemotherapeutic drugs has been reported (7,8,23).
The current study demonstrated that liver cancer SP cells were
resistant to numerous DNA targeting drugs including 5-FU,
gemcitabine, oxaliplatin, paclitaxel, cisplatin, etoposide and
oxaliplatin. This drug resistance property of SP cells is
hypothesized to be partly due to the presence of ABC transporters.
However, there may be additional signaling factors involved in the
tumorgenicity of CSCs.
The Wnt/β-catenin pathway is an important signal
transduction pathway, and serves a key role in the tumorigenesis,
invasion and metastasis of CSCs (26–28).
Increased activation of Wnt/β-catenin signaling has been reported
in CSCs, and as a consequence the CSCs possess unlimited cell
proliferation and therefore, the Wnt/β-catenin pathway is crucial
for the maintenance of self-renewal (15). Notably, the current study suggested
that the elevated expression of β-catenin in liver cancer SP cells
leads to increased activation Wnt/β-catenin targeted genes
including CCND1, DKK1 and AXIN2. This drives the SP cells to
proliferate at a higher rate, rapidly form tumor spheres and become
highly invasive, as demonstrated by reduction in these phenotypes
following the siRNA-mediated knockdown of β-catenin SP cells.
Further studies to elucidate the different processes and factors
involved in the cross talk between Wnt/β-catenin signaling and
CSC-induced tumor relapse may aid in the innovation of novel cancer
therapies.
In conclusion, increased Wnt/β-catenin signaling
contributes to the ability of SP cells to proliferate, rapidly
generate tumor spheres and become highly invasive. Therefore,
further investigation of novel anticancer drugs which target and
suppress Wnt/β-catenin signaling may aid in the improvement of
cancer treatment strategies.
Acknowledgments
The current study was supported by the National
Natural Science Foundation of China (grant nos. 81000959 and
81201781), the Guangdong Natural Science Foundation (grant no.
S2013010016023), the Science and Technology Planning Project of
Guangdong Province, China (grant no. 2009B030801007), the
Fundamental Research Funds for the Central Universities (grant no.
12ykpy47) and the National 12th Five-Year Science and Technology
Plan Major Projects of China (grant nos. 2012ZX10002017-005,
2012ZX10002016-023 and 2012ZX10002010-001-007).
The authors would like to thank Dr Wanshan Li
(Department of Oral and Maxillofacial Surgery, Children's Hospital,
Chongqing Medical University), Dr Wei-Dong Han (Department of
Oncology, Zhejiang University) and Dr Nan Jiang (Department of
Hepatic Surgery, The Third Affiliated Hospital of Sun Yat-sen
University) for their collaboration, sharing of reagents and
protocols, and their contribution.
References
|
1
|
Al-Hajj M, Wicha MS, Benito-Hernandez A,
Morrison SJ and Clarke MF: Prospective identification of
tumorigenic breast cancer cells. Proc Natl Acad Sci USA.
100:3983–3988. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Patrawala L, Calhoun-Davis T,
Schneider-Broussard R and Tang DG: Hierarchical organization of
prostate cancer cells in xenograft tumors: The CD44+alpha2beta1+
cell population is enriched in tumor-initiating cells. Cancer Res.
67:6796–6805. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dalerba P, Dylla SJ, Park IK, Liu R, Wang
X, Cho RW, Hoey T, Gurney A, Huang EH, Simeone DM, et al:
Phenotypic characterization of human colorectal cancer stem cells.
Proc Natl Acad Sci USA. 104:10158–10163. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gupta PB, Chaffer CL and Weinberg RA:
Cancer stem cells: Mirage or reality? Nat Med. 15:1010–1012. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hirschmann-Jax C, Foster AE, Wulf GG,
Nuchtern JG, Jax TW, Gobel U, Goodell MA and Brenner MK: A distinct
'side population' of cells with high drug efflux capacity in human
tumor cells. Proc Natl Acad Sci USA. 101:14228–14233. 2004.
View Article : Google Scholar
|
|
6
|
Kondo T, Setoguchi T and Taga T:
Persistence of a small subpopulation of cancer stem-like cells in
the C6 glioma cell line. Proc Natl Acad Sci USA. 101:781–786. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Feuring-Buske M and Hogge DE: Hoechst
33342 efflux identifies a subpopulation of cytogenetically normal
CD34(+)CD38(−) progenitor cells from patients with acute myeloid
leukemia. Blood. 97:3882–3889. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wulf GG, Wang RY, Kuehnle I, Weidner D,
Marini F, Brenner MK, Andreeff M and Goodell MA: A leukemic stem
cell with intrinsic drug efflux capacity in acute myeloid leukemia.
Blood. 98:1166–1173. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Farazi PA and DePinho RA: Hepatocellular
carcinoma pathogenesis: From genes to environment. Nat Rev Cancer.
6:674–687. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lee TK, Castilho A, Ma S and Ng IO: Liver
cancer stem cells: Implications for a new therapeutic target. Liver
Int. 29:955–965. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ino Y, Gotoh M, Sakamoto M, Tsukagoshi K
and Hirohashi S: Dysadherin, a cancer-associated cell membrane
glycoprotein, down-regulates E-cadherin and promotes metastasis.
Proc Natl Acad Sci USA. 99:365–370. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nam JS, Hirohashi S and Wakefield LM:
Dysadherin: A new player in cancer progression. Cancer Lett.
255:161–169. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Royds JA and Iacopetta B: p53 and disease:
When the guardian angel fails. Cell Death Differ. 13:1017–1026.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim CF, Jackson EL, Woolfenden AE,
Lawrence S, Babar I, Vogel S, Crowley D, Bronson RT and Jacks T:
Identification of bronchioalveolar stem cells in normal lung and
lung cancer. Cell. 121:823–835. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Teng Y, Wang X, Wang Y and Ma D:
Wnt/β-catenin signaling regulates cancer stem cells in lung cancer
A549 cells. Biochem Biophys Res Commun. 392:373–379. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Song J, Chang I, Chen Z, Kang M and Wang
CY: Characterization of side populations in HNSCC: Highly invasive,
chemoresistant and abnormal Wnt signaling. PLoS One. 5:e114562010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bradford MM: Rapid and sensitive method
for the quantitation of microgram quantities of protein utilizing
the principle of protein-dye binding 72. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Shi Y, Fu X, Hua Y, Han Y, Lu Y and Wang
J: The side population in human lung cancer cell line NCI-H460 is
enriched in stem-like cancer cells. PLoS One. 7:e333582012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Szotek PP, Pieretti-Vanmarcke R, Masiakos
PT, Dinulescu DM, Connolly D, Foster R, Dombkowski D, Preffer F,
Maclaughlin DT and Donahoe PK: Ovarian cancer side population
defines cells with stem cell-like characteristics and Mullerian
Inhibiting Substance responsiveness. Proc Natl Acad Sci USA.
103:11154–11159. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Haraguchi N, Utsunomiya T, Inoue H, Tanaka
F, Mimori K, Barnard GF and Mori M: Characterization of a side
population of cancer cells from human gastrointestinal system. Stem
Cells. 24:506–513. 2006. View Article : Google Scholar
|
|
22
|
Patrawala L, Calhoun T,
Schneider-Broussard R, Zhou J, Claypool K and Tang DG: Side
population is enriched in tumorigenic, stem-like cancer cells,
whereas ABCG2+ and ABCG2- cancer cells are similarly tumorigenic.
Cancer Res. 65:6207–6219. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bunting KD, Zhou S, Lu T and Sorrentino
BP: Enforced P-glycoprotein pump function in murine bone marrow
cells results in expansion of side population stem cells in vitro
and repopulating cells in vivo. Blood. 96:902–909. 2000.PubMed/NCBI
|
|
24
|
Visvader JE and Lindeman GJ: Cancer stem
cells in solid tumours: Accumulating evidence and unresolved
questions. Nat Rev Cancer. 8:755–768. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Norwood K, Wang RY, Hirschmann-Jax C,
Andreeff M, Brenner MK, Goodell MA and Wulf GG: An in vivo
propagated human acute myeloid leukemia expressing ABCA3. Leuk Res.
28:295–299. 2004. View Article : Google Scholar
|
|
26
|
Kawaguchi-Ihara N, Murohashi I, Nara N and
Tohda S: Promotion of the self-renewal capacity of human acute
leukemia cells by Wnt3A. Anticancer Res. 28(5A): 2701–2704.
2008.PubMed/NCBI
|
|
27
|
Khan NI, Bradstock KF and Bendall LJ:
Activation of Wnt/beta-catenin pathway mediates growth and survival
in B-cell progenitor acute lymphoblastic leukaemia. Br J Haematol.
138:338–348. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ysebaert L, Chicanne G, Demur C, De Toni
F, Prade-Houdellier N, Ruidavets JB, Mansat-De Mas V, Rigal-Huguet
F, Laurent G, Payrastre B, et al: Expression of beta-catenin by
acute myeloid leukemia cells predicts enhanced clonogenic
capacities and poor prognosis. Leukemia. 20:1211–1216. 2006.
View Article : Google Scholar : PubMed/NCBI
|