Introduction
Osteoarthritis (OA) is one of the most common joint
disorders that seriously affect human health. The main pathological
characteristic involves a progressive loss of articular cartilage
degeneration and secondary osteophyte hyperplasia (1,2). As
chondrocytes serve a crucial role in the function of the normal
activity of articular cartilage (3), with deficient ability for
self-repair, stimulating the functions of the chondrocytes by
promotion of their proliferation may be a potential method for the
treatment of OA.
Polysaccharides are a high molecular weight compound
that widely exist in the cytomembrane of organisms. As demonstrated
in previous studies, not only do polysaccharides enhance
immunologic function and have a specific influence on tumors and
viruses, but they also possess the characteristic of low toxicity
(4). In recent years, numerous
works on polysaccharides have been conducted and many of those have
an encouraging future (5,6). However, the effect of Cibotium
barometz polysaccharides (CBPS) on proliferation of
chondrocytes cultured in vitro has not been investigated to
date. Therefore, in the present study, an initial attempt is made
in order to offer some experimental evidence for the treatment of
OA using CBPS to a certain degree.
Cell proliferation is one of the most important
cellular processes. The eukaryotic cell cycle is composed of 4
phases: G1, S, G2 and M. Among these stages, there are two key
restriction points. The G1/S restriction point, in which DNA
synthesis is prepared, is more important than the latter. The
second is G2/M, in which mitosis begins. It is the G1 phase that
determines whether the cell is able to continue through the cycle
or withdraw. Therefore, it directly influences whether the cell
continues to proliferate (7,8).
Progression through each phase of the cell cycle is regulated by
various cyclin-dependent kinases (CDKs) and their regulatory
subunits, the cyclins (9,10). To control the cell cycle
progression during the G1 phase, CDK4 and cyclin D1 bind to produce
a cyclin D1-CDK4 complex that phosphorylates the retinoblastoma
protein to produce phosphorylated retinoblastoma protein (pRB). pRB
binds and inhibits the activity of E2F transcription factors and
causes release of E2F from pRB-E2F complexes, thus the cell cycle
arrest in G1 phase controlled by pRB is be removed, so E2F
transcription factors are free to bind DNA and initiate
transcription required for cell cycle progression and cell
proliferation (11). Therefore,
Cyclin D1-CDK4-pRB complexes are able to promote cell proliferation
via the promotion of cell cycle transition from the G1 to the S
phase.
Cibotium barometz (CB), a type of herb used
in traditional Chinese medicine, has been extensively used in
Chinese medicinal formulations for the clinical treatment of
orthopedic diseases such as OA (12,13).
Based on previous studies, CB is composed of polysaccharides, amino
acids, flavones and phenolic acids (14). In the present study, the CB
polysaccharides (CBPS) were isolated. In addition, the content of
polysaccharides extracted from CB is higher than in other plants
from the Fujian province of China (15). The primary aim of this study was to
determine the effects of CBPS on cultured chondrocytes in
vitro via phenol-vitriol colorimetry, and the underlying
mechanisms involved. It was observed that CBPS may promote
chondrocyte proliferation via increasing the level of cyclin D1,
CDK4 and pRB.
Materials and methods
Reagents
Fetal bovine serum (FBS) was purchased from Gibco;
Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Anhydrous
glucose (cat. no. SG752802), sulfuric acid (cat. no. 10021618), and
phenol (cat. no. 100153008) were purchased from Sinopharm Chemical
Reagent Co., Ltd. (Shanghai, China). MTT was purchased from
Biosharp Inc. (Hefei, China), Dulbecco's Modified Eagle's medium
(DMEM), trypsin-EDTA, and penicillin-streptomycin were purchased
from HyClone; GE Healthcare Life Sciences (Logan, UT, USA). The
type II collagenase was purchased from Sigma-Aldrich; Merck KGaA
(Darmstadt, Germany) and the cell cycle detection kit was from
Nanjing KeyGen Biotech Co., Ltd. (Nanjing, China), The reverse
transcription system was purchased from TransGen Biotech Co., Ltd.
(Beijing, China). The DNA primers were synthesized by Generay
Biotech Co., Ltd. (Shanghai, China). The rabbit anti-rat cyclin D1,
CDK4, and pRB antibodies were purchased from Santa Cruz
Biotechnology, Inc. (cat. nos. SC-753, SC-260 and SC-32824,
respectively; Dallas, TX, USA), and the anti-rabbit IgG,
horseradish peroxidase-linked secondary antibody was purchased from
Cell Signaling Technology, Inc. (cat. no. 7074s; Danvers, MA, USA).
The study was approved by the Ethics Committee of Fujian University
of Traditional Chinese Medicine (Fuzhou, China).
Extract preparation of CBPS from
CB
The chosen CB was grown in Youxi, China. The dried
and sliced CB sections were refluxed twice with 80% alcohol for 1
h. The solvent was dried with evaporation subsequently, and then
the residue was refluxed in distilled water (100 g/l), boiled three
times for 2 h and subsequently extracted. All extractions were
concentrated to 100 ml under hypopiesia conditions. To conduct
purification of the condensed solution, the anhydrous alcohol was
used in the decoction for precipitation and incubated overnight.
The precipitates isolated by centrifugation were then lyophilized.
The crude polysaccharides were soaked and dissolved into distilled
water (200 g/l), then the protein was removed by Sevag's method
(16). The polysaccharides formed
a white power, and were dissolved in DMEM containing 10% FBS at
concentration of 10 mg/ml and subsequently the solution was
filtered through a 0.22 µm filter and stored at 4°C.
Phenol-vitriol colorimetry
To prepare the glucose standard solutions, 0.1, 0.3,
0.5, 0.6, 0.7, 0.6, 0.7 ml from a stock solution (distilled water:
Anhydrous glucose, 25 ml: 0.01 g) were added into 7 test tubes
respectively. Each tube was supplemented with distilled water to a
final volume of 1 ml, and subsequently 1 ml 5% phenol and 5 ml of
98% sulfuric acid were added for 30 min. Absorbance of distilled
water, and the glucose standards was measured at 490 nm wavelength,
and a standard curve of glucose concentrations based on the
absorbance values was drawn. Then absorbance of CBPS was measured
at 490 nm wavelength, and the concentration of CBPS was calculated
based on the glucose standard curve equation.
Chondrocytes culture and
identification
Male, 4-week-old Sprague-Dawley (SD) specific
pathogen-free (SPF) rats (total 30) were purchased and handled in
the Institutional Animal Centre affiliated to Fujian University of
Traditional Chinese Medicine which was approved by the ethics
committee (License no. SCXK2012-0001). Chondrocytes were isolated
from the knees of rats, and were stripped and soaked with 75%
ethanol for 15 min as previously described (3). The articular cartilages were cut and
precisely constrained in the surface layer of cartilages ensure the
purified cells were purified from the opening of the joint.
Subsequently, these were transferred into PBS-containing penicillin
and streptomycin and then washed three times. The cartilages were
cut into a total of three 1 mm sections and digested with 0.2% type
II collagenase in 50 ml culture flasks. Following this, the flasks
were shaken at 37°C. The isolated cells were collected every 2 h
and the chondrocytes were cultured in 50 ml culture flasks in 4 ml
DMEM containing 10% FBS at 37°C and 5% CO2. The culture
media were changed every 2 days and the cells were subcultured at
90% confluency (17). Collagen
Type II Immunohistochemical Staining was performed to confirm the
identity of the chondrocytes (data not shown), as previously
described from our team (3,17).
MTT assay for evaluation of cell
viability
In this study, chondrocytes at passage 2 were seeded
into 96-well plates at a density of 5×104/ml and
cultured for 24 h. They were treated with varying concentrations of
CBPS (0, 100, 200, 400 and 800 µg/ml) for 24, 48 and 72 h. After
stimulation, 100 µl MTT (1 mg/ml in PBS) was added to each well and
incubated at 37°C for 4 h. Subsequently, the supernatant was
collected and 150 µl dimethyl sulfoxide was added to dissolve the
formazan product and extracts were sonicated for 10 min. Finally,
the cell viability was detected by an ELISA reader at 490 nm wave
length.
Detection of cell cycle by flow
cytometry
Passage 2 chondrocytes were seeded into cell culture
flasks at a density of 5×104/ml and cultured for 24 h.
They were treated with varying concentrations of CBPS (0, 100, 200
and 400 µg/ml) for 48 h. Morphological changes of chondrocytes were
observed and images were captured using an inverted phase contrast
microscope (magnification, ×200). The same batch of passage 2
chondrocytes seeded in 6-well plates at a density of
1×104/ml were subsequently subjected to DMEM without FBS
for 24 h to synchronize the cell cycle stage. Various
concentrations of CBPS (0, 100, 200 and 400 µg/ml) were added into
the plates for 48 h, then the cells were collected and the cell
density was adjusted to 1×105/ml. To evaluate the phase
of chondrocyte proliferation, three solutions (A, B and C) were
incubated using the cell cycle detection kit (Nanjing KeyGen
Biotech Co., Ltd.) according to the manufacturer's instructions.
The results were analyzed by ModFit software version 3.0 (Verity
Software House, Inc., Topsham, ME, USA). Therefore, cells in each
phases of the cell cycle (G0/G1, S and G2/M) could be detected
using a flow cytometer.
RNA extraction and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
analysis
The chondrocytes were treated with the different
concentrations of CBPS (0, 100, 200 and 400 µg/ml) for 48 h. The
total RNA was extracted from which in accordance with the standard
instructions of the TRIZOL reagent (Thermo Fisher Scientific,
Inc.). For RNA samples with 260/280 absorbance between 1.8 and 2.0,
1 µg RNA was reverse transcribed into cDNA based on the
instructions provided by TransGen Biotech Co., Ltd. (cat. no.
H41220K060; Beijing, China). The PCR reactions were prepared,
heated to 90°C for 5 min and subjected to 35 cycles of denaturation
at 94°C for 30 sec, annealing at the specific Tm for 30 sec, and
extension at 72°C for 45 sec to complete amplification. The
expressions of cyclin D1, CDK4, RB and β-actin mRNA were analyzed
by PCR. β-actin was used as an internal control. The primer
sequences provided by the manufacturer were as follows: Cyclin D1
(product size, 399 bp), sense 5′-AGCAGAAGTGCGAAGAGGAGGTC-3′ and
antisense 5′-GGAAAGAAAGTGCGTTGTGCGGTAG-3′; CDK4 (product size, 494
bp), sense 5′-CCTACGGACATACCTGGACAA-3′ and antisense
5′-GAGGCAATGAGATCAA-3′; RB (product size, 255 bp), sense
5′-CTTTATTGGCCTGTGCTCTTG-3′ and antisense
5′-ATTCCATGATTCGATGCTCAC-3′; β-actin (product size, 385 bp), sense
5′-GGGAAGTGCTGGATAG-3′ and β-actin, antisense
5′-GTGATGTTTCGGATGG-3′. The DNA bands were detected by gel
electrophoresis (1.5% agarose) and analyzed by the Gel
Documentation system (Gel Doc 2000; Bio-Rad Laboratories, Inc.,
Hercules, CA, USA). The experiment was performed 3 independent
times.
Western blot analysis
Following treatment of chondrocytes with different
concentrations of CBPS (0, 100, 200 and 400 µg/ml) for 48 h, the
total protein was extracted from the cells by
radioimmunoprecipitation assay (RIPA) lysis buffer supplemented
with 1 mM phenylmethanesulfonyl fluoride (PMSF), according to the
manufacturer's instructions (Beyotime Institute of Biotechnology,
Haimen, China). Subsequently the protein concentrations were
determined by a bicinchoninic acid assay kit and a standard curve
was produced. Total proteins (25 µg/lane) were separated by
electrophoresis on 12% SDS-PAGE gels and transferred onto
polyvinylidene membranes correspondingly. Then the membranes were
blocked for 2 h in 5% skimmed milk at room temperature. Following
blocking, the membranes were washed in TBS-Tween for 5 min and then
incubated with the primary antibody for cyclin D1, CDK4, pRB and
β-actin (1:1,000) at 4°C overnight. Following a second wash using
TBS-Tween, the membranes were incubated with the secondary antibody
solution (1:5,000) for 1 h at room temperature. Three final 10 min
washes were performed using TBS-Tween. Finally, the antibody-bound
protein bands were examined with enhanced chemiluminescence
(Beyotime Institute of Biotechnology) and the results of images
were captured from3 independent experiments using a Kodak image
station 400R (Kodak, Rochester, NY, USA).
Statistical analysis
The results are expressed as the mean ± standard
deviation. Statistical analysis was performed by Student's t-test
or analysis of variance followed by Bonferroni post hoc test, using
SPSS software (version 18.0; SPSS Inc., Chicago, IL, USA).
P<0.05 was considered to indicate a statistically significant
difference.
Results
The concentration of CBPS detected by
phenol-vitriol colorimetry
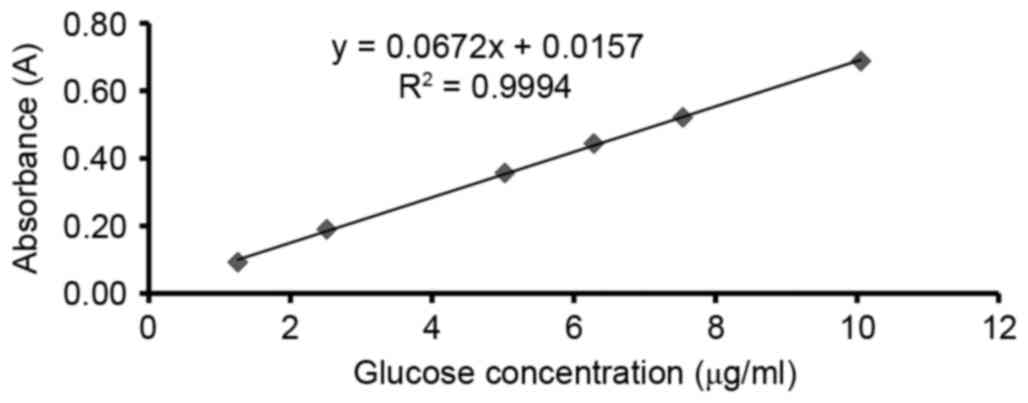
There was a favorable linearity relationship between
the absorption and content determined in the range of 1–11 µg/ml,
and the regression equation y=0.0672x+0.0157, R2=0.9994
(Fig. 1). The content of
polysaccharides was ~76.91% with good precision of relative
standard deviation (RSD) at 1.8,605% and well stability of RSD at
1.9,883%, the result of the average recovery test was 95.4% and RSD
of 1.83%. These results identified that the CBPS was at an
acceptable level.
Morphological characteristics for
chondrocytes
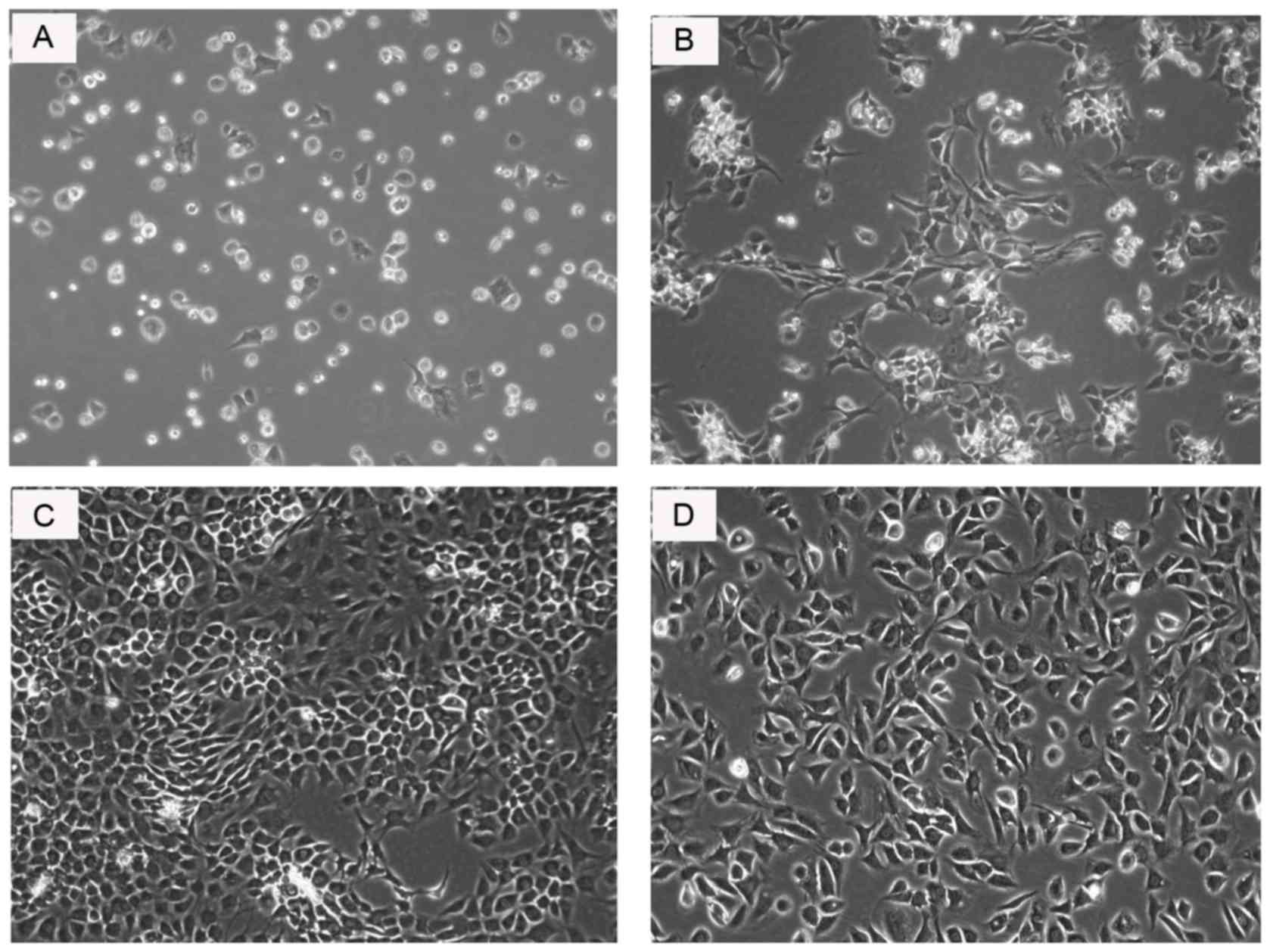
The primary chondrocytes cultured in vitro
were observed by phase microscopy (x200 magnification). From the
morphological images (Fig. 2), the
different changes of the chondrocytes over time in culture were
observed and the further descriptions have been presented in a
previous study (18). Following
observation, passage 2 chondrocytes were regarded as optimal for
their abundant extracellular matrix and convenient cultured
conditions in the present study. The passage 2 chondrocytes had a
characteristic cobblestone-like morphology and moderate counts,
with clear boundaries and distinct nuclei.
Chondrocyte proliferation was promoted
via CBPS
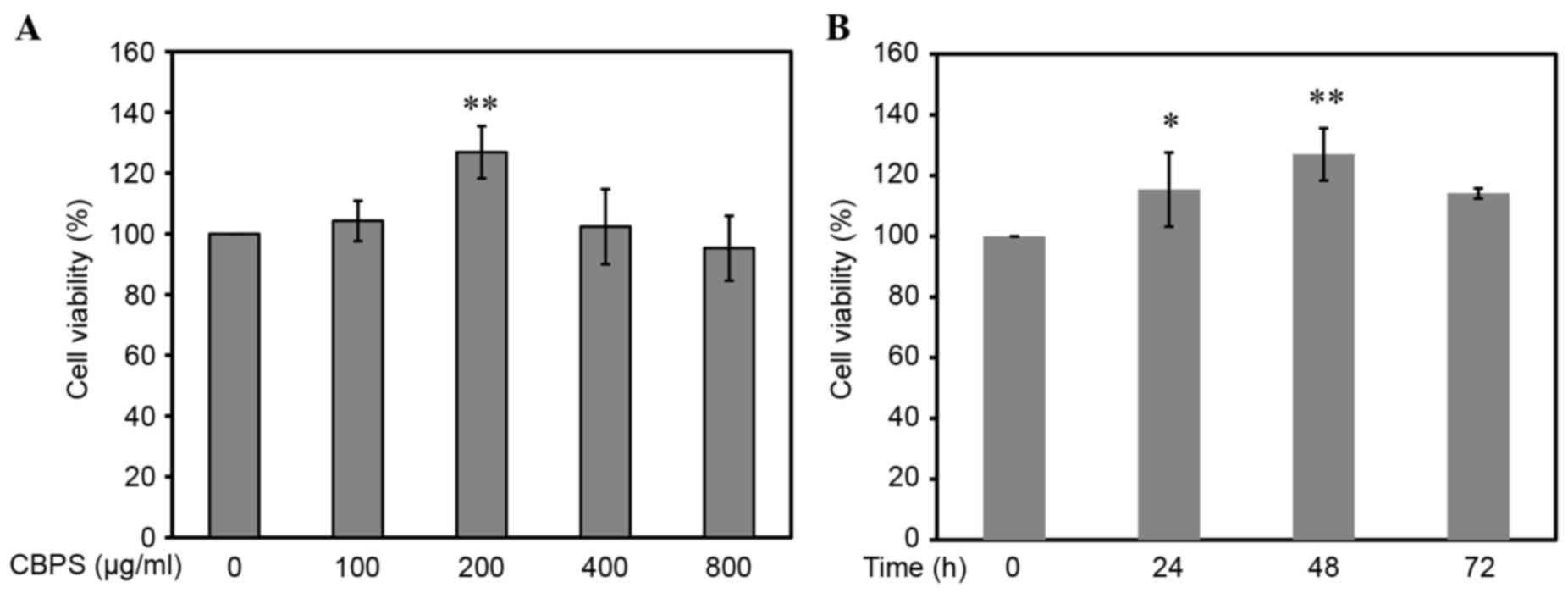
An MTT assay was used to detect the cell
proliferation following culture with different concentrations of
CBPS (0, 100, 200, 400 and 800 µg/ml) at 24, 48 and 72 h. As
presented in Fig. 3A, at 48 h, the
optical density (OD) of the 200 µg/ml CBPS group was significantly
higher compared with the 0 µg/ml group (P<0.01). When comparing
the 100, 400 and 800 µg/ml groups, there were no significant
differences in the viability of the chondrocytes compared with the
0 µg/ml group (P>0.05). Fig. 3B
illustrates the viability of chondrocytes treated with 200 µg/ml
CBPS for 24, 48 and 72 h. Chondrocyte viability significantly
increased at 24 h of treatment, and was further increased at 48 h,
compared with the 0 h group (P<0.05 and P<0.01 respectively;
Fig. 3B). These findings suggest
that chondrocyte proliferation is likely promoted by CBPS treatment
in a time-dependent manner. The optimal time and concentration of
CBPS treatment was 48 h and 200 µg/ml, respectively (Fig. 3).
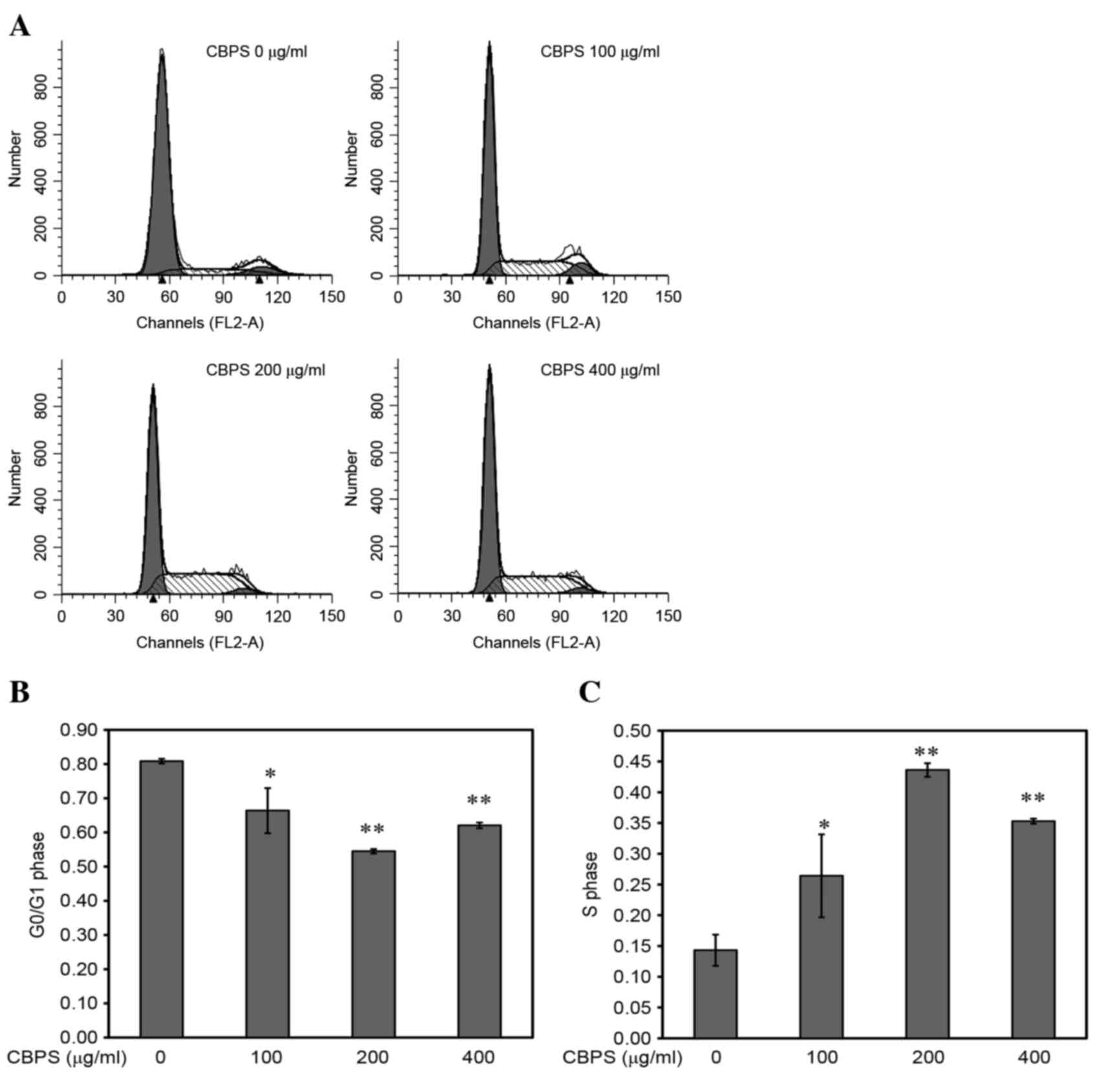
Effects of CBPS on the chondrocyte
cell cycle
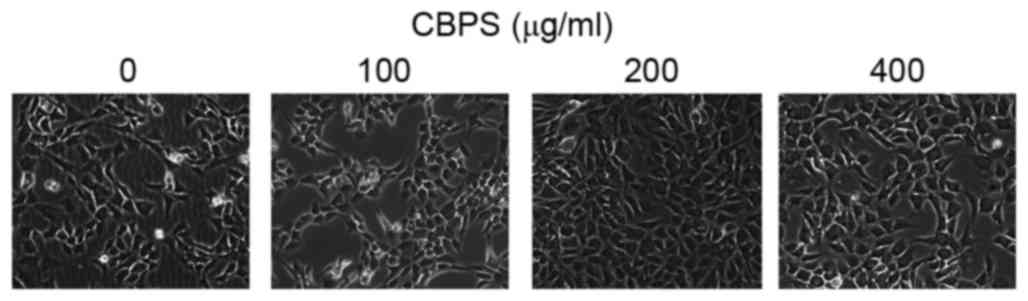
Based on the aforementioned results, the range of
intervening concentrations was reduced to 0, 100, 200 and 400
µg/ml. Following treatment of chondrocyte with these concentrations
for 48 h the morphology of chondrocytes was observed (Fig. 4). Flow cytometry analysis
identified the effects of CBPS (0, 100, 200 and 400 µg/ml) on the
chondrocyte cell cycle (Fig. 5A).
The proportion of cells in the G0/G1 phase (Fig. 5B) was lowest in the 200 µg/ml
CBPS-treated group (0 µg/ml, 0.8088±0.0069; 100 µg/ml,
0.664±0.0659; 200 µg/ml, 0.5451±0.0065; and 400 µg/ml,
0.6209±0.0083). Compared with the 0 µg/ml group, CBPS treatment
caused significant decreases in proportion of G0/G1 cells (100
µg/ml, P<0.05; 200 µg/ml, P<0.01; and 400 µg/ml, P<0.01).
The percentage of cells in S phase (Fig. 5C) in the 200 µg/ml group was
significantly higher compared with all other CBPS treatment groups
(0 µg/ml, 0.1434±0.0254; 100 µg/ml, 0.2642±0.067; 200 µg/ml,
0.4362±0.0110; and 400 µg/ml, 0.3530±0.0041). When compared with
the 0 µg/ml group, there were significant increases in the
proportion of cells in S phase following CBPS treatment (100 µg/ml,
P<0.05; 200 µg/ml, P<0.01; 400 µg/ml, P<0.01). When
comparing with the 100 and 400 µg/ml group, there were no
significant differences in the G0/G1 and S phase proportions
(P>0.05). This suggested an opposite trend to the G0/G1 phase.
Therefore, the proliferation of chondrocytes could be promoted by
CBPS in the progression of the cell cycle in the transition from G1
to S phase.
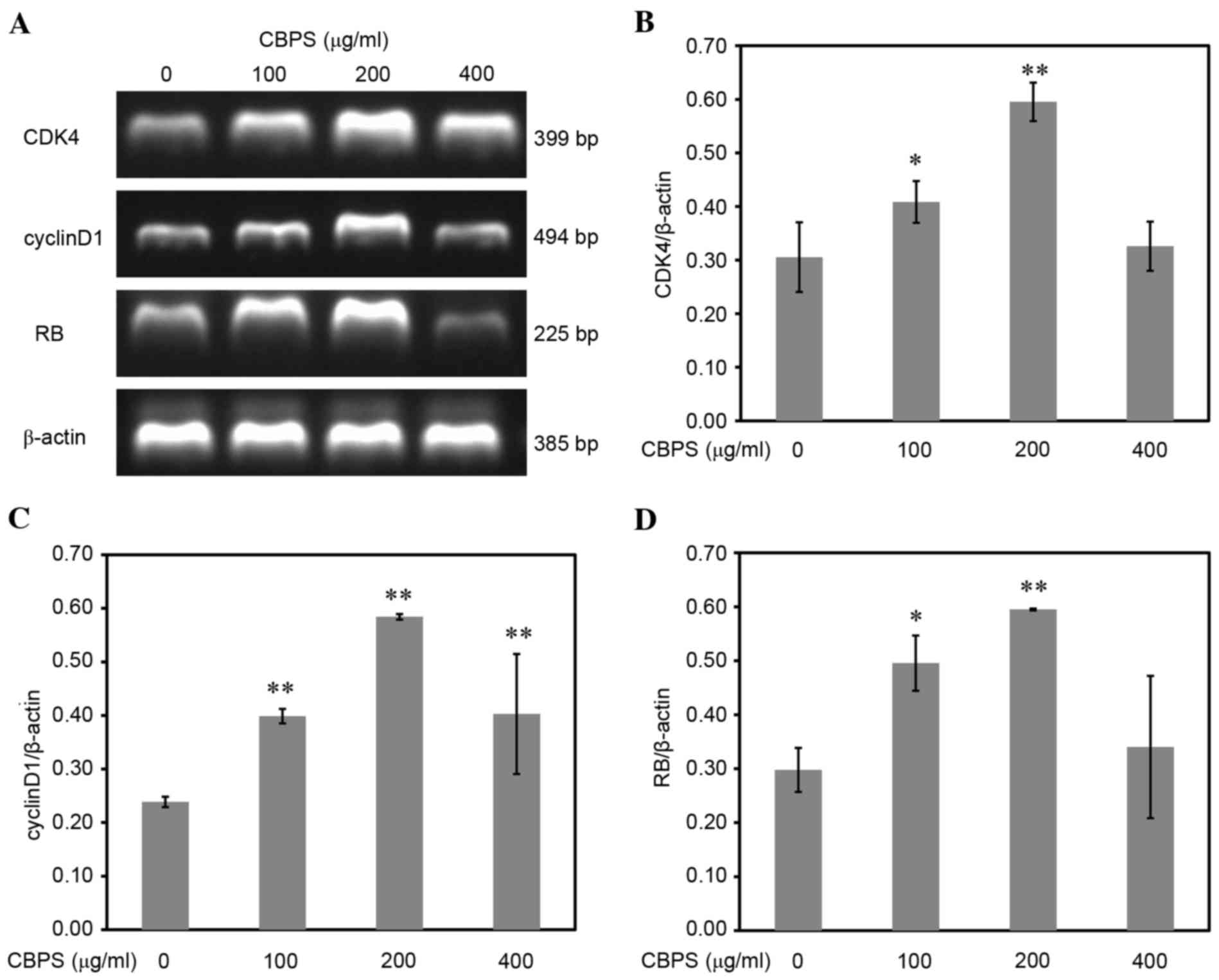
CBPS upregulates the expression of
cyclin D1, CDK4 and RB mRNA and protein
It is necessary to examine for the mechanism
underlying CBPS-induced promotion of chondrocyte proliferation.
Therefore, the mRNA and protein expression levels of cyclin D1,
CDK4 and RB were tested by RT-qPCR and western blot analysis,
respectively. As presented in Fig.
6, the mRNA expression of cyclin D1, CDK4 and RB in
CBPS-treated groups was significantly higher compared with the 0
µg/ml group, with peak mRNA expression levels in the 200 µg/ml
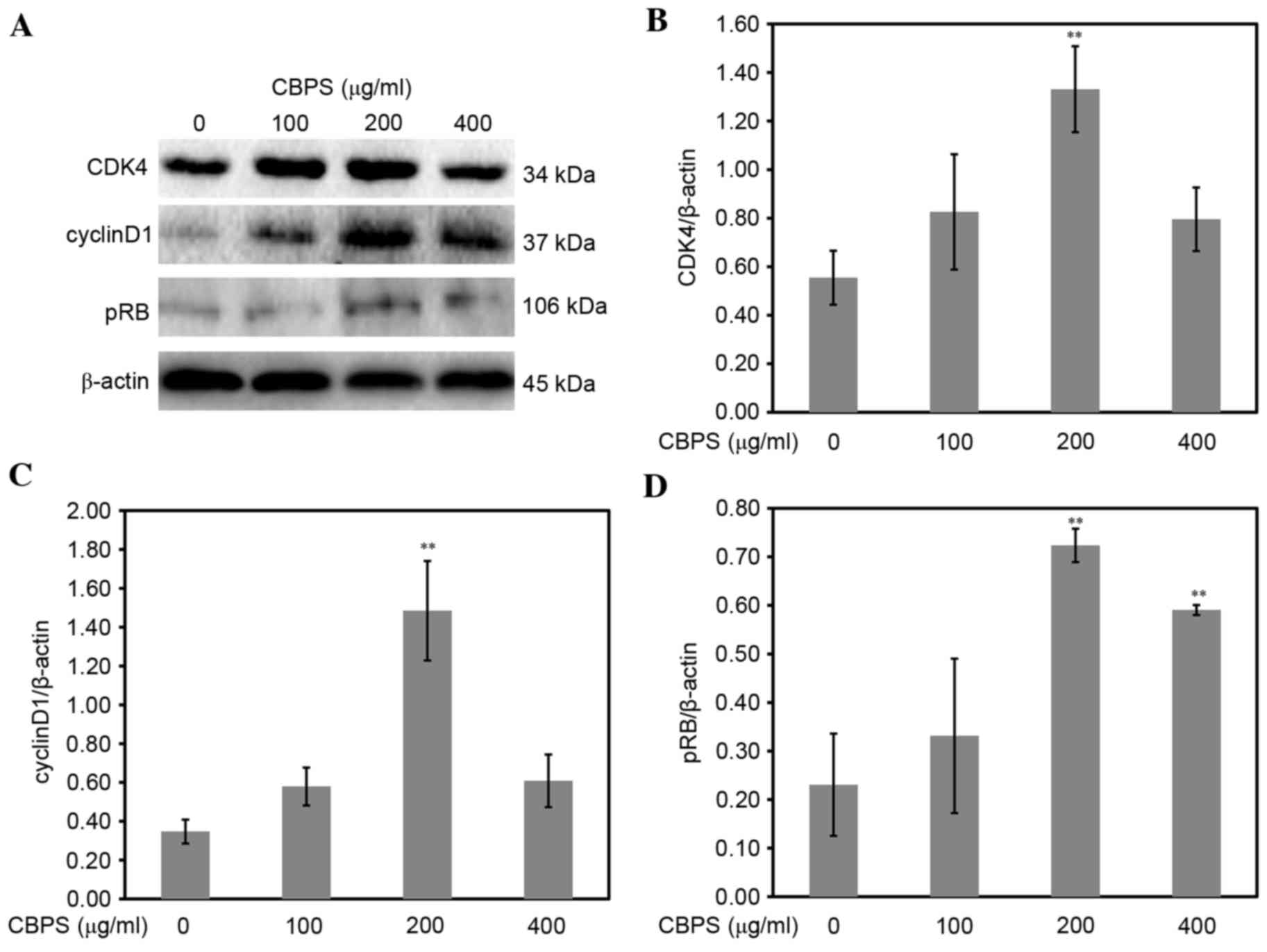
group (P<0.01). When measuring the protein expression levels by
western blotting, cyclin D1, CDK4 and pRB were again significantly
upregulated in the CBPS-treated groups compared with the 0 µg/ml
group, with peak protein expression levels in the 200 µg/ml group
(P<0.01; Fig. 7).
Discussion
The incidence of osteoarthritis (OA) still poses a
major public health challenge throughout the world, and, currently
there is no thoroughly effective therapy available. Considering
that chondrocytes are responsible for secreting extracellular
matrix, and the maintenance of cartilage homeostasis is involved in
the pathological characteristics of OA, promoting chondrocyte
proliferation may be a prospective and potential method for the
treatment of OA (19–21).
Polysaccharides are biological molecules that can
store bioenergy and support the role of structural components
(22), which are necessary for the
entire organism. In recent years, polysaccharides have also been
demonstrated to be important in protecting various physiological
functions (23,24). Polysaccharides are widely extracted
from Chinese herbs. In the present study, the CBPS were obtained
from one of the most important Chinese traditional herbs used in OA
treatment, Cibotium barometz. In order to explore the
mechanisms behind the effects of CBPS on chondrocytic functions,
chondrocytes were treated with varying concentrations of CBPS (100,
200, 400 and 800 µg/ml) and with 200 µg/ml CBPS for 24, 48 and 72
h. An MTT assay was then used to test the effect of CBPS on the
total numbers of viable chondrocytes. According to the results from
the MTT assay, the proliferation of chondrocytes is promoted by
CBPS in a time-dependent manner, and the optimum time and
concentration were 48 h and 200 µg/ml, respectively.
With regards to the transmission in the cell cycle,
cyclins and CDKs are positive regulators responsible for
proliferation; however, CDK inhibitors (CDKI) are negative
regulators that control the speed of the cell cycle. The balance of
negative and positive regulation are key factors that determine the
changes of the various phases in the cell cycle (25,26).
The cell cycle is formed of 4 stages: G1, the preparation for DNA
synthesis; S, DNA synthesis; G2, the preparation for mitosis; and
M, mitosis. Among the four stages, the S and M phases are the two
most crucial processes.
The G1/S and G2/M transitions are vital steps of
cell cycle progression. To further investigate the cell cycle in
chondrocytes, the cells were treated with 0, 100, 200 and 400 µg/ml
CBPS for 48 h. Subsequently, flow cytometry was used to detect
cells in the different stages of the cell cycle. In the present
study, the results demonstrated that the percentage of chondrocytes
in the G0/G1 phase was obviously reduced and the percentage of
chondrocytes in the S phase was evidently increased by CBPS,
demonstrating that chondrocyte proliferation may be stimulated by
CBPS treatment via promotion of the transition from G1 to S phase.
Cyclin D1-CDK4-pRB complexes are composed of cyclins, CDKs and pRB,
which are basic protein families that regulate the progression of
the cell cycle. Once cyclin D1-CDK4 complexes phosphorylate pRB,
which releases pRB from interaction with E2F transcription factors
allowing them to bind DNA and trigger transcription to directly
regulate the progression from G1 phase to S phase, consequently
promoting chondrocyte proliferation and restraining chondrocyte
apoptosis (27,28).
Based on the results in the current study, which
demonstrated that CBPS treatment may boost the mRNA and protein
expression of cyclin D1, CDK4 and pRB, indicating that CBPS
promotes the progression of chondrocytes from the G1 to the S phase
by regulating the cyclin D1-CDK4-pRB complex. In conclusion, these
data indicated that CBPS may effectively increase chondrocyte
proliferation by promoting G1/S cell cycle transition and enhance
the mRNA and protein expression of cyclin D1, CDK4 and pRB.
However, as one of the most misunderstood and
complex diseases, the exact pathological and pathogenesis
characteristics of OA still have not been clarified in full. Based
on a recent meta-analysis of the curative effect of dextrose
prolotherapy for knee osteoarthritis, dextrose prolotherapy may
improve pain and physical function in patients with OA (29). Previous research has validated the
safety of dextrose prolotherapy for clinical treatment of OA
(30,31), and this suggested that future
studies should investigate the connection between the CBPS and OA.
The mechanism underlying the effects of CBPS on OA should be
explored further. Alongside the current advances in scientific
research, the biologically active ingredient of CBPS should be
purified and synthesized, in order to determine its curative
effects in relevant clinical trials.
Acknowledgements
The present study was supported by a grant from the
Keji Chen Development Foundation for Integrated Traditional and
Western Medicine (grant no. CKJ2014001).
References
|
1
|
Zhao XY, Yang ZB, Zhang ZJ, Zhang ZQ, Kang
Y, Huang GX, Wang SW, Huang H and Liao WM: CCL3 serves as a
potential plasma biomarker in knee degeneration (osteoarthritis).
Osteoarthritis Cartilage. 23:1405–1411. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ham O, Lee CY, Kim R, Lee J, Oh S, Lee MY,
Kim J, Hwang KC, Maeng LS and Chang W: Therapeutic potential of
differentiated mesenchymal stem cells for treatment of
osteoarthritis. Int J Mol Sci. 16:14961–14978. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lin P, Weng X, Liu F, Ma Y, Chen H, Shao
X, Zheng W, Liu X, Ye H and Li X: Bushen Zhuangjin decoction
inhibits TM-induced chondrocyte apoptosis mediated by endoplasmic
reticulum stress. Int J Mol Med. 36:1519–1528. 2015.PubMed/NCBI
|
|
4
|
Shi XL, Yao CX, Lin X and Feng Y: The
applications and research progresses of polysaccharide drugs. Chin
J New Drugs. 23:1057–1062. 2014.
|
|
5
|
Loh SH, Park JY, Cho EH, Nah SY and Kang
YS: Animal lectins: Potential receptors for ginseng
polysaccharides. J Ginseng Res. 41:1–9. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li F, Wei D, Zhang J, et al: The effect of
Hedysari Radix polysaccharide on protein expression profiling of
human adenocarcinoma A549 cells in vitro. Pharmacology and Clinics
of Chinese Materia Medica. 4:54–59. 2014.
|
|
7
|
Fong YC, Yang WH, Hsu SF, Hsu HC, Tseng
KF, Hsu CJ, Lee CY and Scully SP: 2-methoxyestradiol induces
apoptosis and cell cycle arrest in human chondrosarcoma cells. J
Orthop Res. 25:1106–1114. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Woods A, Wang G and Beier F: Regulation of
chondrocyte differentiation by the actin cytoskeleton and adhesive
Interactions. J Cell Physiol. 213:1–8. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Golias CH, Charalabopoulos A and
Charalabopoulos K: Cell proliferation and cell cycle control: A
mini review. Int J Clin Pract. 58:1134–1141. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aszodi A, Hunziker EB, Brakebusch C and
Fässler R: Beta1 integrins regulate chondrocyte rotation, G1
progression, and cytkinesis. Genes Dev. 17:2465–2479. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Buxton PG, Hall B, Archer CW and
Francis-West P: Secondary chondrocyte-derived Ihh stimulates
proliferation of periosteal cells during chick development.
Development. 130:4729–4739. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Xu G, Pei QY, Ju CG, Zhang F and Jia TZ:
Detection on effect of different processed Cibotium barometz on
osteoblasts by CCK-8. Zhongguo Zhong Yao Za Zhi. 38:4319–4323.
2013.(In Chinese). PubMed/NCBI
|
|
13
|
Xu G, Sun N, Zhao MJ, Ju CG and Jia TZ:
Study on decoction's effect of different processed rhizomes of
Cibotium barometz on retinoic acid induced male rats osteoporosis.
Zhongguo Zhong Yao Za Zhi. 39:1011–1015. 2014.(In Chinese).
PubMed/NCBI
|
|
14
|
Cheng Q, Yang Z and Hu Y: Studies on the
chemical constituents of Cibotium barometz. Progress Pharmaceutical
Sci. 5:298–299. 2003.(In Chinese).
|
|
15
|
Ju C, Zhang Q, Yu H, Gao Q, Shi J and Jia
TZ: Determination of polysaccharides in Cibotium barometz and its
processed product from various cultivation locations. Chin J Exp
Trad Med Formulae. 24:46–48. 2011.(In Chinese).
|
|
16
|
Qiao DL, Zhao F, Huang HZ, Fan CC and Han
L: Ultrasonic-assisted extraction and in vitro antioxidant activity
of polysaccharides from Agaricus bisporus. Zhong Yao Cai.
35:1340–1347. 2012.PubMed/NCBI
|
|
17
|
Liu F, Liu G, Liang W, Ye H, Weng X, Lin
P, Li H, Chen J, Liu X and Li X: Duhuo Jisheng decoction treatment
inhibits the sodium nitroprussiate-induced apoptosis of
chondrocytes through the mitochondrial-dependent signaling pathway.
Int J Mol Med. 34:1573–1580. 2014.PubMed/NCBI
|
|
18
|
Yu F, Li X, Cai L, Li H, Chen J, Wong X,
Xu H, Zheng C, Liu X and Ye H: Achyranthes bidentata
polysaccharides induce chondrocyte proliferation via the promotion
of the G1/S cell cycle transition. Mol Med Rep. 7:935–940.
2013.PubMed/NCBI
|
|
19
|
Bijlsma JW, Berenbaum F and Lafeber FP:
Osteoarthritis: An update with relevance for clinical practice.
Lancet. 377:2115–2126. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Roush JK, Cross AR, Renberg WC, Dodd CE,
Sixby KA, Fritsch DA, Allen TA, Jewell DE, Richardson DC, Leventhal
PS and Hahn KA: Evaluation of the effects of dietary
supplementation with fish oil omega-3 fatty acids on weight bearing
in dogs with osteoarthritis. J Am Vet Med Assoc. 236:67–73. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ye J, Wu G, Li X, Li Z, Zheng C, Liu X and
Ye H: Millimeter wave treatment inhibits apoptosis of chondrocytes
via regulation dynamic equilibrium of intracellular free Ca(2+).
Evid Based Complement Alternat Med. 215:4641612015.
|
|
22
|
Boddohi S and Kipper MJ: Engineering
nanoassemblies of polysaccharides. Adv Mater. 22:2998–3016. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Luo YK, Cheng Y and Feng YP: Chinese
Wolfberry Polysaccharide on the immune function of diabetes II
Rats. Information Trad Chin Med. 1:13–16. 2014.
|
|
24
|
Yao H, Gu LJ and Guo JY: Study on effect
of Astragali Radix Polysaccharides in improving learning and memory
functions in aged rats and its mechanism. Zhongguo Zhong Yao Za
Zhi. 39:2071–2277. 2014.(In Chinese). PubMed/NCBI
|
|
25
|
Zhang M, Xie R, Hou W, Wang B, Shen R,
Wang X, Wang Q, Zhu T, Jonason JH and Chen D: PTHrP prevents
chondrocyte premature hypertrophy by inducing cyclin-D1-dependent
Runx2 and Runx3 phosphorylation, ubiquitylation and proteasomal
degradation. J Cell Sci. 122:1382–1389. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hossain MA, Park J, Choi SH and Kim G:
Dexamethasone induces apoptosis in proliferative canine tendon
cells and chondrocytes. Vet Comp Orthop Traumatol. 21:337–342.
2008.PubMed/NCBI
|
|
27
|
Aszodi A, Hunziker EB, Brakebusch C and
Fässler R: Beta1 integrins regulate chondrocyte rotation, G1
progression, and cytokinesis. Genes Dev. 17:2465–2479. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Beier F and LuValle P: The cyclin D1 and
cyclin A genes are targets of activated PTH/PTHrP receptors in
Jansen's metaphyseal chondrodysplasia. Mol Endocrinol.
16:2163–2173. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ye D, Chen C, Lin Z and Bai Y:
Meta-analysis on curative effect of dextrose prolotherapy for knee
osteoarthritis. J Jilin University (Medicine Edition). 41:612–618.
2015.
|
|
30
|
Reeves KD and Hassanein KM: Long-term
effects of dextrose prolotherapy for anterior cruciate ligament
laxity. Altern Ther Health Med. 9:58–62. 2003.PubMed/NCBI
|
|
31
|
Rabago D, Patterson JJ, Mundt M, Kijowski
R, Grettie J, Segal NA and Zgierska A: Dextrose prolotherapy for
knee osteoarthritis: A randomized controlled trial. Ann Fam Med.
11:229–237. 2013. View
Article : Google Scholar : PubMed/NCBI
|