Introduction
The Polycomb group (PcG) of proteins consists of
transcriptional repressors that orchestrate changes in chromatin
structure to regulate gene activity (1–2). The
BMI1 proto-oncogene (Bmi-1), a member of PcG proteins, was
previously known as a transcriptional repressor targeting the
cyclin-dependent kinase inhibitor 2A gene locus. Bmi-1 is described
as an oncogene in many tumor types and has critical roles in the
oncogenesis of cancers and cancer stem cells (CSCs) (3). CSCs are defined as a subpopulation of
cancer cells that have stem-like features, including self-renewal,
differentiation abilities, metastatic potential, and resistance to
conventional chemoradiotherapies (4–6). The
discovery of CSC-specific markers has helped identify CSC
population in many cancer types, including blood, neck, thoracic,
abdominal and genital system cancers (7–15).
CD133 (official name: prominin 1) is regarded as an important CSC
marker in hepatocellular carcinoma (HCC). Epithelial-mesenchymal
transition (EMT) is considered to occur during cancer invasion and
migration; following EMT, epithelial cells lose their epithelial
characteristics and adopt mesenchymal appearance and
characteristics (16,17). The downregulation or loss of
E-cadherin and the upregulation of Vimentin are regarded as
hallmarks of EMT (18).
HCC is a highly lethal cancer and its incidence is
increasing in the United States, particularly in the population
infected with Hepatitis C virus, based on data from the National
Comprehensive Cancer Network from the year 2016 (https://www.nccn.org/professionals/physician_gls/f_guidelines.asp).
In 2014, an estimated 33,190 people in the United States were
diagnosed and ~23,000 succumbed to liver and intrahepatic bile duct
cancer (19). HCC frequently has
an insidious onset, being highly invasive, fast-growing and with a
high fatality risk. Early detection and prevention of metastasis
are key aspects to the treatment of HCC. Since CSCs and EMT
contribute to cancer progression, the present study aimed to
explore the interaction between them and their role in the
development and progression of HCC in the hope that the findings
may offer novel targets for HCC therapy.
Materials and methods
Cell culture
The hepatocellular carcinoma G2 (Hep G2) cells
originated from the Cell Bank of Type Culture Collection of the
Chinese Academy of Sciences (Shanghai, China). The cells were
cultured in Minimum Essential Medium (MEM; Gibco; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) at 37°C
with 5% CO2.
Flow cytometry
Hep G2 cells were washed twice with PBS and
resuspended in PBS at a density of 1×107 cells/100 µl.
The resuspended cells were stained with phycoerythrin
(PE)-conjugated anti-human CD133 (130–098-826; 1:10; Miltenyi
Biotec, Inc., Cambridge, MA, USA), incubated for 20 min on ice and
then washed twice with PBS. The respective isotype control was used
at the same concentration according to the manufacturer's
instructions (12–4714; eBioscience; Thermo Fisher Scientific,
Inc.). Cells magnetically-enriched for CD133 did not require
staining again and were used as such in flow cytometry. Samples
were analyzed on a flow cytometer (FACSverse; BD Biosciences,
Franklin Lakes, NJ, USA) at the Sun Yat-sen Memorial Hospital of
the Sun Yat-sen University (Guangzhou, China).
Magnetic-activated cell sorting
(MACS)
Hep G2 cells (before MACS; CD133+ and
CD133− cells) were resuspended in PBS/2% FBS and
centrifuged at 111 × g for 10 min at room temperature to a total
volume of 1 ml at a density of 1×108/ml in a 12×75 mm
polystyrene tube to fit properly into the magnet (EasySep; Stemcell
Technologies, Inc., Vancouver, BC, Canada). Then, 100 µl anti-human
CD32 (Fcγ receptor II) blocker, 50 µl PE-conjugated anti-human
CD133, 100 µl PE-selection cocktail and 50 µl of magnetic
nanoparticles (all from one kit) were added sequentially to the
cells, according to the manufacturer's instructions (EasySep™ Human
PE Positive Selection kit; 18551; Stemcell Technologies, Inc.). The
cell suspension was adjusted to a total volume of 2.5 ml by adding
PBS/2% FBS (step A). The tube was then placed into the magnet for 5
min and the supernatant fraction was removed (step B). Steps A and
B were repeated twice at room temperature. The magnetically-labeled
CD133+Hep G2 cells remained attached to the walls of the
tube due to the magnetic field. The cells in the supernatant
fraction were CD133−Hep G2 cells. Following removal from
the magnet, the cells in the tube were used for flow cytometry or
other assays as indicated.
Cell counting kit-8 (CCK-8) viability
assay
CD133+ and CD133−Hep G2 cells
in 100 µl suspension in MEM only were plated in 96-well plates and
cultured for 4 h until the cells had completely attached to the
bottom. A variety of starting cell numbers were plated for the
assay, at 1,000, 2,000, 4,000 and 8,000 cells per well. Then 10 µl
of CCK-8 solution (Dojindo Molecular Technologies, Inc., Kumamoto,
Japan) was added to each well and incubated for 4 h. The optical
density (OD) was measured at 450 nm using a spectrophotometer
(Multiskan GO; Thermo Fisher Scientific, Inc.) and normalized to
the OD of MEM alone as a control.
Transfection with
Bmi-1/pcDNA3.1(+)
The coding sequences of Bmi-1 (accession no.,
NM_005180.8; forward, 5′-CTAGCTAGCATGCATCGAACAACGAGAATCA-3′ and
reverse 3′-CCGCTCGAGTCAACCAGAAGAAGTTGCTGATG-5′) and the
Bmi-1-expressing plasmid, Bmi-1/pcDNA3.1 (+), were custom ordered
from Geneseed Biotech Co., Ltd (Guangzhou, China) and the correct
coding sequence of Bmi-1 in the expression plasmid was confirmed by
IGE Biotechnology, Ltd. (Guangzhou, China). CD133+Hep G2
cells were transfected with 2 µg Bmi-1/pcDNA3.1(+) or empty vector
control pcDNA3.1 using Lipofectamine 2000 for 24 h (Invitrogen;
Thermo Fisher Scientific, Inc.), according to manufacturer's
protocols.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cultured cells using
TRIzol Reagent (Takara Biotechnology Co., Ltd., Dalian, China), and
1 µg of total RNA was used for cDNA synthesis with PrimeScript RT
Master Mix reagent (Takara Biotechnology Co., Ltd.). qPCR was
performed using the SYBR Premix ExTaq II (TliRNaseH Plus) kit
(Takara Biotechnology Co., Ltd.) in an ABI 7500 Fast Real-Time PCR
System (Bio-Rad Laboratories, Inc., Hercules, CA, USA) with the
following program: 95°C for 30 sec, followed by 40 cycles of 95°C
for 5 sec, 60°C for 1 min, and 95°C for 30 sec. Results were
analyzed using the 2−∆∆Cq method (20). β-actin gene expression was measured
as endogenous control. Experiments were performed in technical
triplicates and were repeated at least twice independently. Primers
were custom ordered using the following sequences: Bmi-1, forward
5′-TCTGGGAGTGACAAGG-3′ and reverse 5′-AAACAAGAAGAGGTGGA-3′; and
β-actin, forward 5′-GCCAACACAGTGCTGTCTG-3′ and reverse
5′-TACTCCTGCTTGCTGATCCA-3′.
Western blot analysis
Cells were lysed in lysis buffer (50 mM Tris pH 7.4,
150 mM NaCl, 0.1% NP-40, 0.5% sodium deoxycholate) for 5 min at
4°C. The protein concentration of the lysate was quantitated by BCA
method. Equal amounts of lysate (30 mg protein) were loaded and
separated by 10% SDS-polyacrylamide gels, and transferred onto
nitrocellulose membranes. The membranes were blocked with 5%
non-fat milk powder in TBS for 1 h at room temperature and probed
with primary antibodies against Bmi-1 (6964; 1:1,000), E-cadherin
(14472; 1:1,000), Vimentin (3932; 1:1,000) (all from Cell Signaling
Technology, Inc., Danvers, MA, USA), and GAPDH (KC-5G4; 1:8,000;
Zhejiang Kangchen Biotech Co., Ltd., Hangzhou, China) at 4°C for 16
h. Following washing with TBS/0.1% Tween 20, the membranes were
incubated with secondary horseradish peroxidase-conjugated
antibodies (A21020 and A21010; 1:6,000; Abbkine Scientific Co.,
Ltd., Wuhan, China) at 4°C for 45 min and visualized using
chemiluminescence with a ImageQuant LAS 500 (GE Healthcare Life
Sciences, Shanghai, China). Each assay was carried out in
triplicate.
Wound healing/migration assay
Cells (5×105/well) were plated in 6-well
plates and cultured until they reached confluence. A diametric
scratch was generated using a pipette tip in each well and washed
with PBS three times. Cells were cultured in MEM alone for 24 h,
and then they were photographed under a light microscope (DMi 1;
Leica Microsystems, Inc., Buffalo Grove, IL, USA) the same 3 wells
in each plate were also photographed and pre-marked at 0 h. The
scratch width was measured and average migration rates were
calculated for each group using Image-Pro Plus version 6.0 (Media
Cybernetics, Inc., Rockville, MD, USA). Each assay was carried out
in triplicate.
Transwell invasion assay
Matrigel matrix (BD Biosciences) was diluted at a
working concentration of 300 µg/ml and then 100 µl was added to the
upper surface of Transwell chambers for 1 h in the incubator to
coat them prior to the assays. Cells were resuspended in MEM at a
concentration of 1×105/ml (the concentration required by
the assay). The Matrigel-coated Transwells were inserted into
24-well plates and loaded with 100 µl of cell suspension in the
upper chamber and 500 µl of MEM/20% FBS in the lower chamber.
Following incubation for 24 h at 37°C with 5% CO2, the
Transwell filter was fixed with methanol and stained with 10%
Giemsa. The cells on the upper side of the filter were removed with
a cotton swab prior to fixation. The cells that had invaded to the
bottom surface of the Transwell filter were counted under a light
microscope (DMi 1; Leica Microsystems, Inc.). A total of 9
microscopic fields (×100 magnification) were randomly selected to
count cells. Each assay was done in triplicate.
Statistical analysis
All data were reported as means ± standard error of
the mean and SPSS version 20.0 (BM Corp., Armonk, NY, USA) was
used. Statistical significance of differences between mean values
was assessed by Student's t-test for unpaired data. Comparisons of
data between multiple groups were performed with analysis of
variance followed by LSD post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
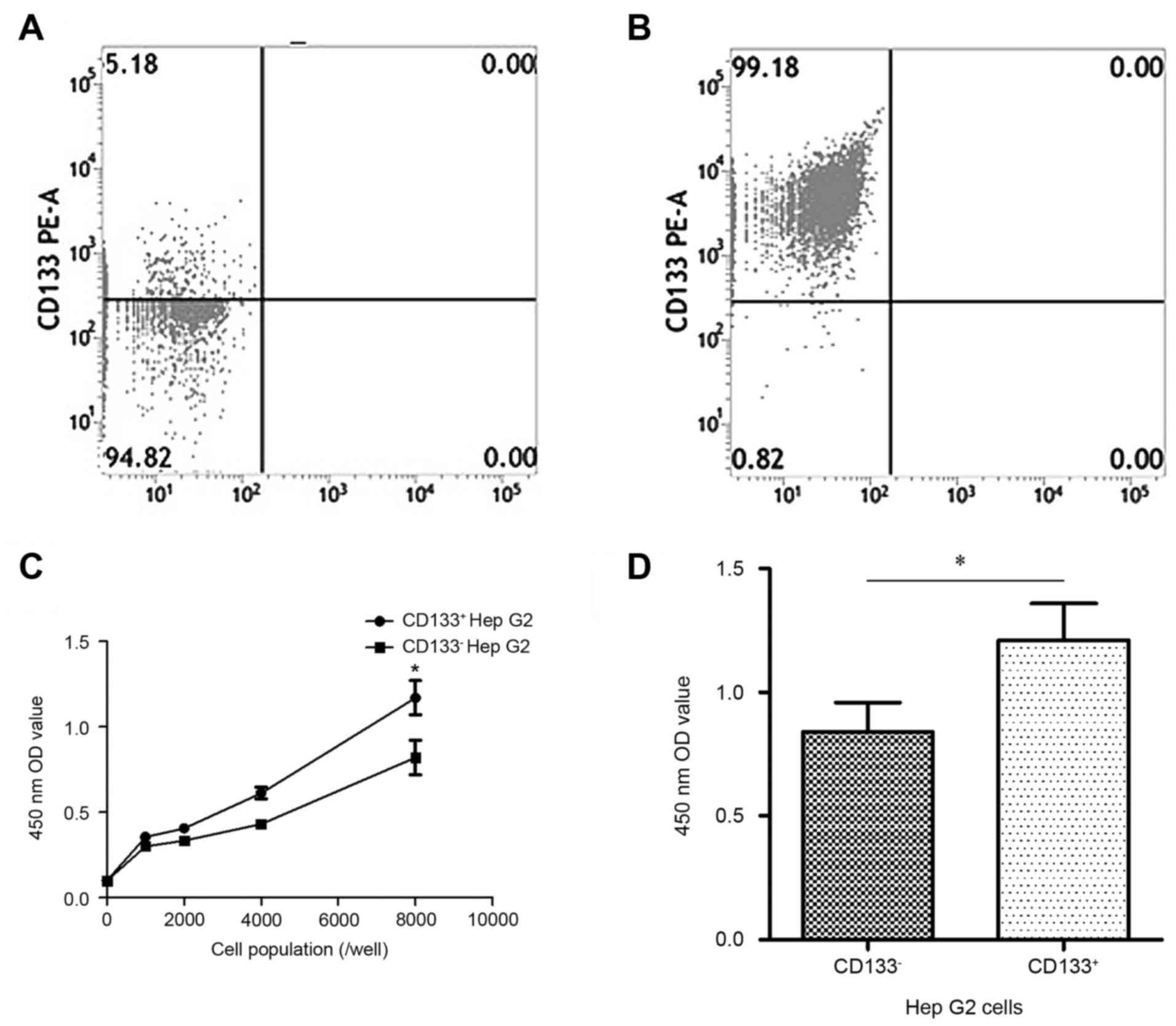
CD133+Hep G2 cells exhibit
increased viability
The surface marker CD133 was used for selection and
enrichment of CSCs in the HCC cell line Hep G2. CSCs have been
reported to exhibit self-renewal and differentiation abilities,
metastatic potential, and resistance to conventional
chemoradiotherapies. Using the MACS technique, a
CD133+-enriched subpopulation of Hep G2 cells was
obtained (Fig. 1A and B). By CCK-8
assay, CD133+Hep G2 cells exhibited increased numbers of
viable cells compared with CD133−Hep G2 cells (Fig. 1C and D). These findings suggest
that CD133+Hep G2 cells may be similar to stem-like
cells and were used in subsequent experiments.
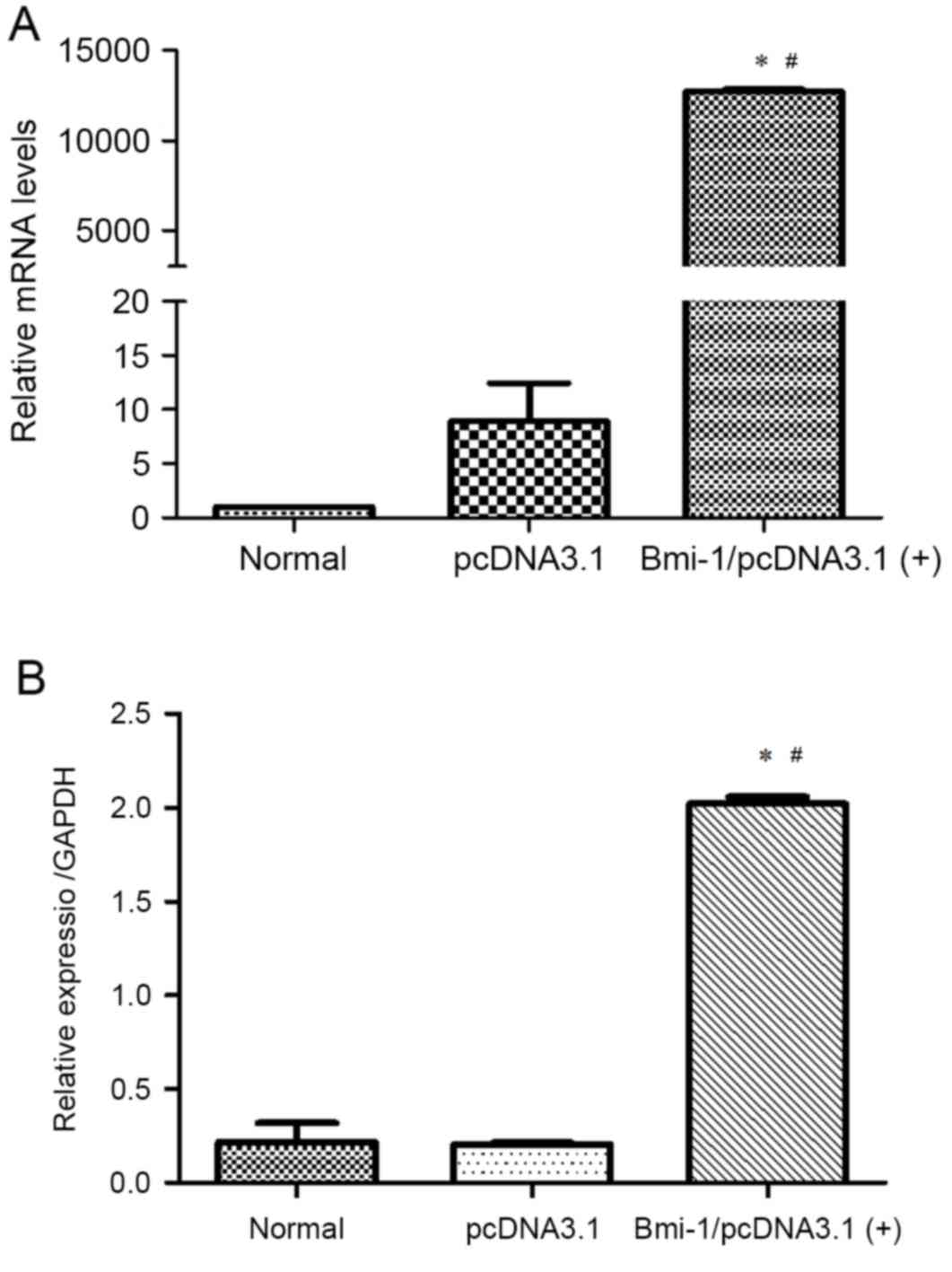
Overexpression of Bmi-1 in
CD1333+Hep G2 cells
CD133+Hep G2 cells were transfected with
either the empty vector plasmid pcDNA3.1 or the Bmi-1-expressing
plasmid Bmi-1/pcDNA3.1(+). Overexpression of Bmi-1 was confirmed at
them RNA and protein level, by RT-qPCR and western blotting,
respectively (Fig. 2).
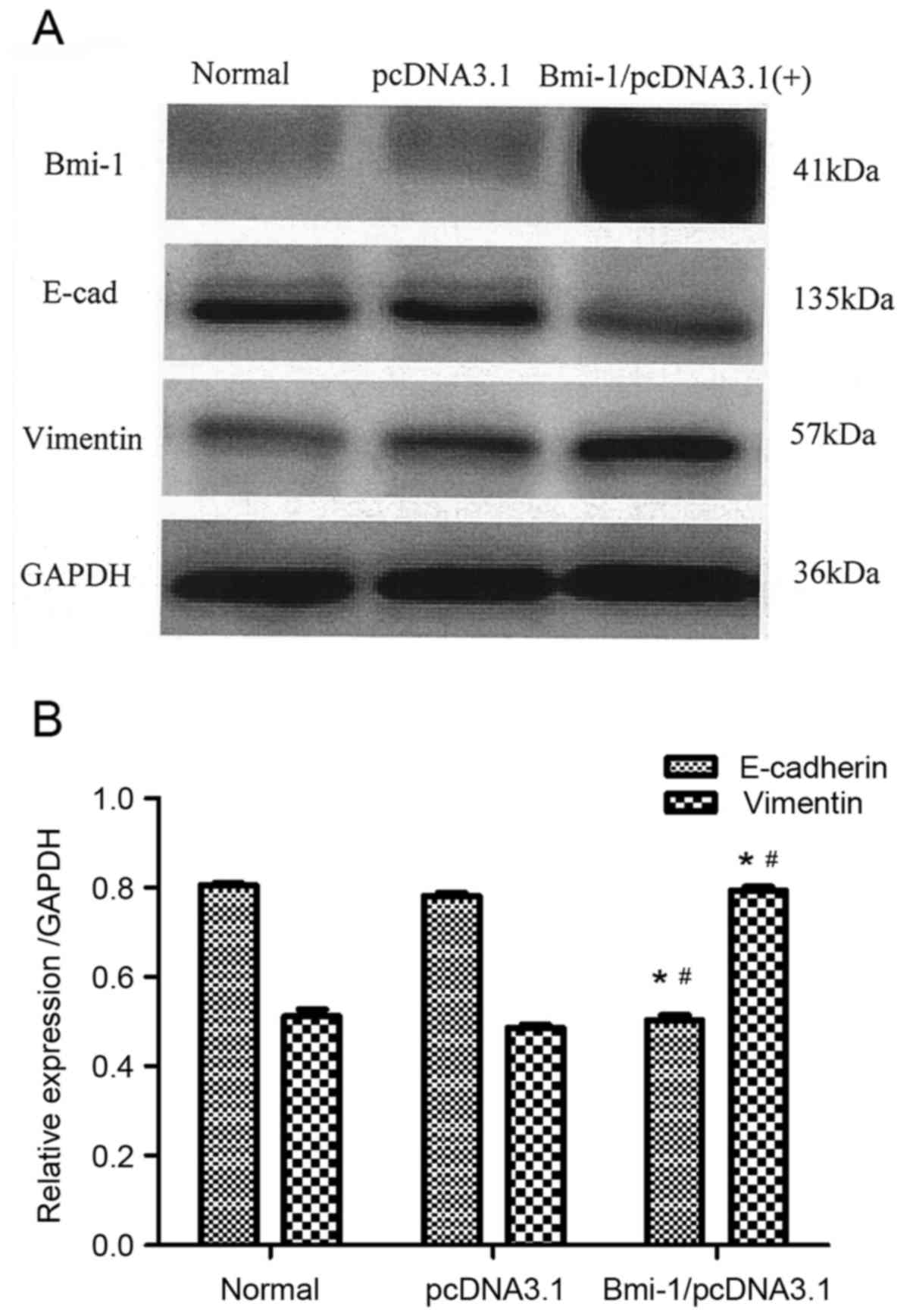
Overexpression of Bmi-1 promotes EMT
in CD133+Hep G2 cells
In order to evaluate the EMT status, the protein
expression levels of E-cadherin and Vimentin were examined by
western blotting in the CD133+Hep G2 cells following
transfection with Bmi-1/pcDNA3.1(+). The results demonstrated that
Bmi-1-overexpressing CD133+Hep G2 had decreased
E-cadherin but increased Vimentin, compared with control (Fig. 3). Downregulation of E-cadherin and
upregulation of Vimentin are regarded as hallmarks of EMT (18). Bmi-1 has been reported to possess
an important role in the maintenance of CSCs and is associated with
EMT (3). Therefore, the present
findings suggested that overexpression of Bmi-1 might promote EMT
in the CD133+Hep G2 cells.
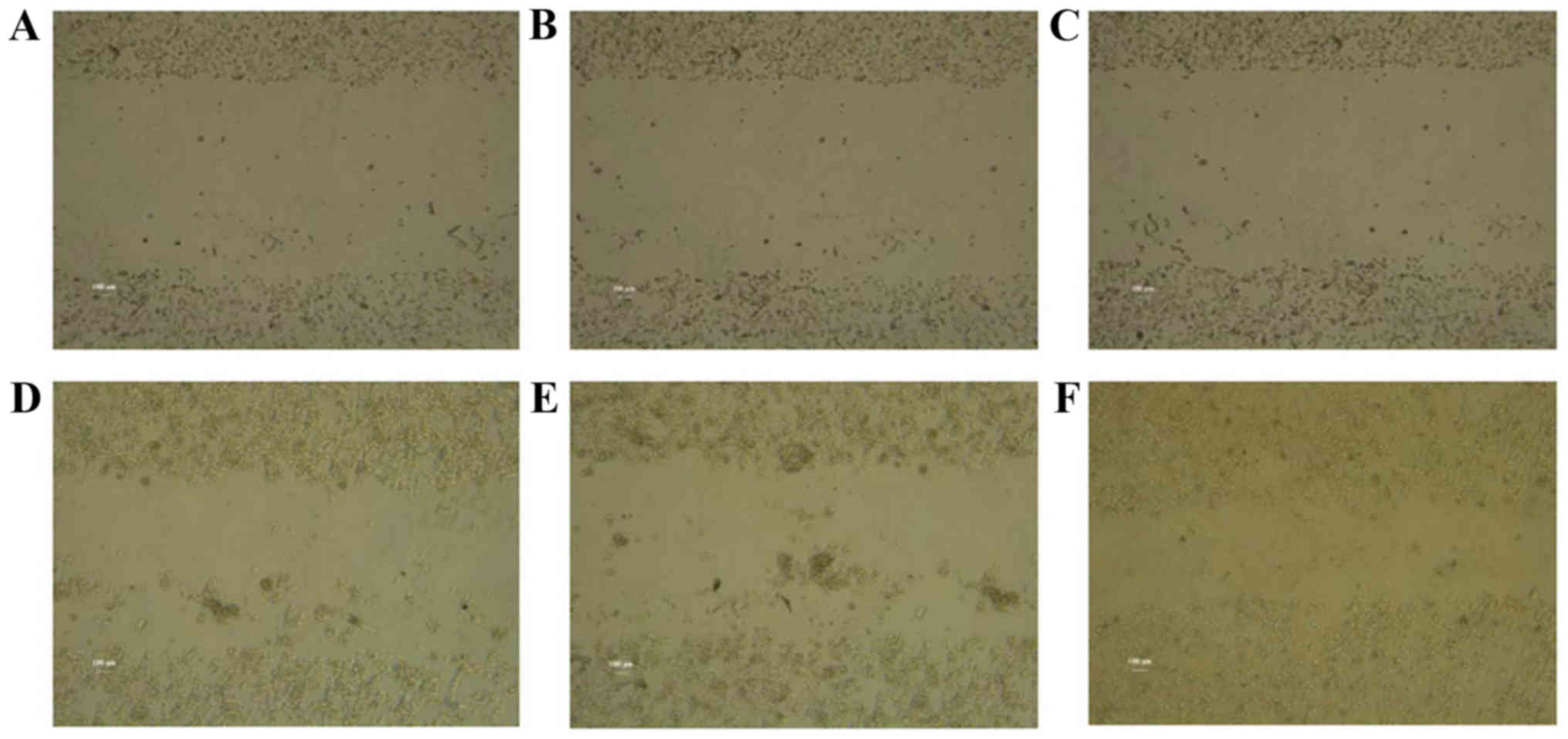
CD133+Hep G2 cells exhibit
increased invasion and migration abilities following overexpression
of Bmi-1
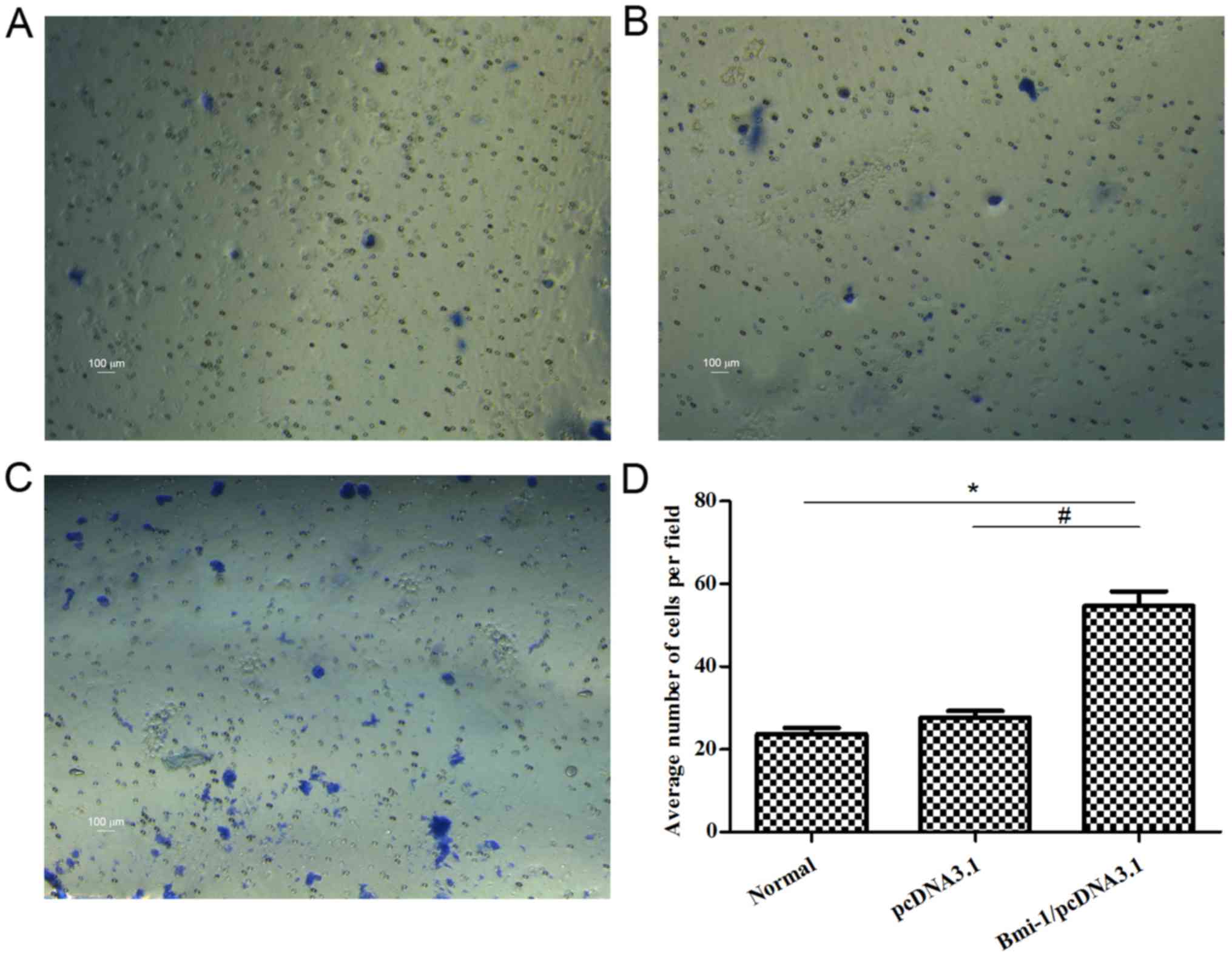
A wound healing assay and a Transwell invasion assay
were next employed in order to assess the migration and invasion
abilities, respectively, in the Bmi-1-overexpressing
CD133+Hep G2 cells. The results demonstrated that
CD133+Hep G2 cells exhibited increased migration
(Fig. 4) and invasion (Fig. 5) abilities following Bmi-1
overexpression, compared with control cells. These findings
indicate that Bmi-1 promoted invasion and migration of
CD133+Hep G2 cells, and this effect may be associated
with inducing EMT.
Discussion
HCC is one of the most common malignancies, with an
increasing incidence in recent years and a poor prognosis. Since
the manifestations of HCC are always nonspecific, patients are
often diagnosed at an advanced stage, resulting in a 5-year
survival rate of <10% (19).
Early detection and prevention of metastasis are of crucial
importance for treatment of HCC. Therefore, further studies on
preventing HCC progression are urgently needed.
Cancer stem cell markers can be used to identify CSC
populations in tumors and the MACS technique can be used to enrich
for CSCs. Since CSCs have been demonstrated to be crucial in the
development of cancer, further studies evaluating the functions and
biological characteristics of CSCs are vital for cancer therapy. In
the present study, it was demonstrated that CD133+Hep G2
cells had greater viability than CD133−Hep G2 cells, and
therefore these cells were used as CSC-like in the rest of the
present study. It is hypothesized that inhibiting the CSC functions
may aid in preventing cancer metastasis and improving overall
survival rates; however, to date, no effective way to inhibit CSCs
has been reported.
During the process of metastasis, CSCs are
hypothesized to undergo functional changes. EMT is reported to
occur during cancer development and progression, with E-cadherin
downregulated and Vimentin upregulated, two expression changes that
are regarded as hallmarks of EMT (18). Therefore, understanding these
processes is important for understanding cancer progression. The
present study speculated that CD133+ cancer cells, when
induced to undergo EMT, may contribute to increased migration and
invasion in HCC, and the related initiator or trigger of EMT was
explored.
Bmi-1 is highly expressed in hematopoietic
malignancies and several solid tumors, serving a critical role in
the maintenance of CSCs (3). The
present results demonstrated that overexpression of Bmi-1 resulted
in downregulation of E-cadherin and upregulation of Vimentin in
CD133+Hep G2 cells. Furthermore, CD133+Hep G2
cells exhibited increased invasion and migration abilities
following overexpression of Bmi-1. These findings may indicate that
Bmi-1 promoted EMT in CD133+Hep G2 cells. Bmi-1 has been
previously associated with EMT in nasopharyngeal (21), breast (22,23),
melanoma (24), endometrial
(25), prostate (26), bladder (27), and colon (28) cancers. Bmi-1 is considered to
contribute to the pathogenesis of nasopharyngeal cancer through
inducing EMT partially by targeting the tumor suppressor
phosphatase and tensin homolog (PTEN), thus activating the
phosphoinositide 3-kinase (PI3K)/Akt pathway (21). It has also been demonstrated that
Bmi-1 induces invasion through activation of the Akt pathway in
breast cancer cells (23). In
addition, overexpression of Bmi-1has been reported to contribute to
the invasion and metastasis of HCC by increasing the expression of
matrix metalloproteinase (MMP)2, MMP-9 and vascular endothelial
growth factor via the PTEN/PI3K/Akt pathway (29). Therefore, it can be hypothesized
that Bmi-1 may promote invasion and migration of HCC through
inducing EMT and the PI3K/Akt pathway. The underlying signaling
pathways and molecular mechanisms will be the focus of future
studies.
In conclusion, Bmi-1 overexpression increased the
invasion and migration abilities of CD133+HepG2 cells by
inducing EMT, which indicates that Bmi-1 may have a role in
promoting metastasis and progression of HCC. It is possible that
Bmi-1 may be a suitable target for the inhibition of EMT in CSCs
and for the development of novel strategies in the therapy of
HCC.
Acknowledgements
We sincerely thank other colleagues in our
laboratory for their active help in this study. The present study
was supported by the following grants: Natural Science Foundation
of Guangdong Province (grant no. 2014A030313543), Science and
Technology Planning Project of Guangdong Province (grant no.
2014A020212547).
References
|
1
|
Jacobs JJ and van Lohuizen M: Polycomb
repression: From cellular memory to cellular proliferation and
cancer. Biochim Biophys Acta. 1602:151–161. 2002.PubMed/NCBI
|
|
2
|
Piunti A and Pasini D: Epigenetic factors
in cancer development: Polycomb group proteins. Future Oncol.
7:57–75. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Molofsky AV, Pardal R, Iwashita T, Park
IK, Clarke MF and Morrison SJ: Bmi-1 dependence distinguishes
neural stem cell self-renewal from progenitor proliferation.
Nature. 425:962–967. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Polyak K and Hahn WC: Roots and stems:
Stem cells in cancer. Nat Med. 12:296–300. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li F, Tiede B, Massagué J and Kang Y:
Beyond tumorigenesis: Cancer stem cells in metastasis. Cell Res.
17:3–14. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kakarala M and Wicha MS: Implications of
the cancer stem-cell hypothesis for breast cancer prevention and
therapy. J Clin Oncol. 26:2813–2820. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bonnet D and Dick JE: Human acute myeloid
leukemia is organized as a hierarchy that originates from a
primitive hematopoietic cell. Nat Med. 3:730–737. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Singh SK, Clarke ID, Terasaki M, Bonn VE,
Hawkins C, Squire J and Dirks PB: Identification of a cancer stem
cell in human brain tumors. Cancer Res. 63:5821–5828.
2003.PubMed/NCBI
|
|
9
|
Ponti D, Costa A, Zaffaroni N, Pratesi G,
Petrangolini G, Coradini D, Pilotti S, Pierotti MA and Daidone MG:
Isolation and in vitro propagation of tumorigenic breast cancer
cells with stem/progenitor cell properties. Cancer Res.
65:5506–5511. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li C, Heidt DG, Dalerba P, Burant CF,
Zhang L, Adsay V, Wicha M, Clarke MF and Simeone DM: Identification
of pancreatic cancer stem cells. Cancer Res. 67:1030–1037. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
O'Brien CA, Pollett A, Gallinger S and
Dick JE: A human colon cancer cell capable of initiating tumour
growth in immunodeficient mice. Nature. 445:106–110. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ricci-Vitiani L, Lombardi DG, Pilozzi E,
Biffoni M, Todaro M, Peschle C and De Maria R: Identification and
expansion of human colon-cancer-initiating cells. Nature.
445:111–115. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chu P, Clanton DJ, Snipas TS, Lee J,
Mitchell E, Nguyen ML, Hare E and Peach RJ: Characterization of a
subpopulation of colon cancer cells with stem cell-like properties.
Int J Cancer. 124:1312–1321. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Laganà AS, Colonese F, Colonese E, Sofo V,
Salmeri FM, Granese R, Chiofalo B, Ciancimino L and Triolo O:
Cytogenetic analysis of epithelial ovarian cancer's stem cells: An
overview on new diagnostic and therapeutic perspectives. Eur J
Gynaecol Oncol. 36:495–505. 2015.PubMed/NCBI
|
|
15
|
López J, Valdez-Morales FJ,
Benítez-Bribiesca L, Cerbón M and Carrancá AG: Normal and cancer
stem cells of the human female reproductive system. Reprod Biol
Endocrinol. 11:532013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yilmaz M and Christofori G: EMT, the
cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev.
28:15–33. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Birchmeier W and Birchmeier C:
Epithelial-mesenchymal transitions in development and tumor
progression. EXS. 74:1–15. 1995.PubMed/NCBI
|
|
18
|
Onder TT, Gupta PB, Mani SA, Yang J,
Lander ES and Weinberg RA: Loss of E-cadherin promotes metastasis
via multiple downstream transcriptional pathways. Cancer Res.
68:3645–3654. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Siegel R, Ma J, Zou Z and Jemal A: Cancer
statistics, 2014. CA Cancer J Clin. 64:9–29. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Song LB, Li J, Liao WT, Feng Y, Yu CP, Hu
LJ, Kong QL, Xu LH, Zhang X, Liu WL, et al: The polycomb group
protein Bmi-1 represses the tumor suppressor PTEN and induces
epithelial-mesenchymal transition in human nasopharyngeal
epithelial cells. J Clin Invest. 119:3626–3636. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li H, Song F, Chen X, Li Y, Fan J and Wu
X: Bmi-1 regulates epithelial-to-mesenchymal transition to promote
migration and invasion of breast cancer cells. Int J Clin Exp
Pathol. 7:3057–3064. 2014.PubMed/NCBI
|
|
23
|
Guo BH, Feng Y, Zhang R, Xu LH, Li MZ,
Kung HF, Song LB and Zeng MS: Bmi-1 promotes invasion and
metastasis, and its elevated expression is correlated with an
advanced stage of breast cancer. Mol Cancer. 10:102011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu S, Tetzlaff MT, Cui R and Xu X:
miR-200c inhibits melanoma progression and drug resistance through
down-regulation of BMI-1. Am J Pathol. 181:1823–1835. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Dong P, Kaneuchi M, Watari H, Hamada J,
Sudo S, Ju J and Sakuragi N: MicroRNA-194 inhibits epithelial to
mesenchymal transition of endometrial cancer cells by targeting
oncogene BMI-1. Mol Cancer. 10:992011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nanta R, Kumar D, Meeker D, Rodova M, Van
Veldhuizen PJ, Shankar S and Srivastava RK: NVP-LDE-225
(Erismodegib) inhibits epithelial-mesenchymal transition and human
prostate cancer stem cell growth in NOD/SCID IL2Rγ null mice by
regulating Bmi-1 and microRNA-128. Oncogenesis. 2:e422013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu L, Qiu M, Tan G, Liang Z, Qin Y, Chen
L, Chen H and Liu J: miR-200c inhibits invasion, migration and
proliferation of bladder cancer cells through down-regulation of
BMI-1 and E2F3. J Transl Med. 12:3052014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang Z, Bu X, Chen H, Wang Q and Sha W:
Bmi-1 promotes the invasion and migration of colon cancer stem
cells through the downregulation of E-cadherin. Int J Mol Med.
38:1199–1207. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li X, Yang Z, Song W, Zhou L, Li Q, Tao K,
Zhou J, Wang X, Zheng Z, You N, et al: Overexpression of
Bmi-1contributes to the invasion and metastasis of hepatocellular
carcinoma by increasing the expression of matrix metalloproteinase
(MMP)-2, MMP-9 and vascular endothelial growth factor via the
PTEN/PI3K/Akt pathway. Int J Oncol. 9:793–802. 2013. View Article : Google Scholar
|