Introduction
Asthma is a complicated systemic disease of the
airways, which is characterized by variable and recurring symptoms,
including bronchial hyper-responsiveness, inflammation and airflow
obstruction. When an asthma exacerbation occurs, certain symptoms,
such as shortness of breath, wheezing, coughing, and chest
tightness are uncontrolled (1).
Together, the symptoms eventually impair lung function by
decreasing the flow of gases to and from the alveoli in the distal
lung (2). The prevalence of asthma
has increased 2–3-fold over recent decades in developed countries
(3–5). It is estimated that asthma affects
~300 million people in the world and causes a quarter of a million
mortalities (6). Globally, asthma
accounts for approximately one in every 250 mortalities (7), which is a significant cause of
morbidity and mortality in developed countries (8). In America, 9–20% of children and 1–3%
of adults suffer from asthma (8,9), and
the financial burden of asthma is estimated to be >$10 billion
(10). Furthermore, there is a
high prevalence of ~5% in China (11). Numerous factors, including improved
epidemiological reporting, medical care and increased environmental
allergens contribute to the increased prevalence. However, the
molecular mechanism of asthma remains poorly understood.
Asthma is a chronic airway inflammatory disease,
where persistent inflammation in the airway results in structural
changes termed airway remodeling (12). Airway smooth muscle cells (ASMCs)
confer an abnormally exaggerated bronchoconstriction in asthma, and
the phenomenon is commonly referred to as airway
hyper-responsiveness (AHR), which is a hallmark characteristic of
asthma (13). Airway remodeling
encompasses subepithelial fibrosis, deposition of extracellular
matrix (ECM) proteins, increased smooth muscle mass and mucus gland
hyperplasia. Generation of growth factors from patients with asthma
drive mesenchymal cell proliferation and differentiation toward
increased matrix deposition and smooth muscle production (14–16).
Airway remodeling is particularly observed in patients with
refractory asthma and progressive decline in lung function
(17).
Studies demonstrate that airway remodeling is
closely connected with the progression of AHR (18).
It has previously been demonstrated that asthma
markedly increases Th2-mediated responses and decreases
Th1-mediated responses (19–21).
Clinically, the majority of asthma patients show a significant
increase in ASM bundles, primarily due to increases in cell number,
collectively contributing to airway remodeling (12). Furthermore, airway remodeling is
progressive and the degree of structural changes correlates with
disease severity (18). In this
sense, ASMC hyperplasia has been postulated as the predominant
mechanism of ASM thickening (22).
Simultaneously, increased proliferation results in decreased
pulmonary function in asthmatic patients (23). It is critical to further understand
the mechanism of airway remodeling in patients with asthma;
however, the molecular mechanisms remain unclear. Thus novel
targets must be identified to prevent airway remodeling and thus,
treat asthma (24).
In the current study, proteomic technology was
applied to analyze the difference between normal lung and asthmatic
lung tissue samples. In addition, the molecular mechanisms of
asthma were detected.
Materials and methods
Animals and materials
A total of 20 female BALB/c mice (specific
pathogen-free; aged 5–6 weeks; weight, 16–20 g) were purchased from
the Animal Center of Guangdong Province (Guangzhou, China) and fed
in a specific pathogen-free grade breeding room (separated into two
groups of ten, bred separately and conventionally in cages, at a
temperature of 18–22°C, 50–60% humidity and 10–14 h lighting with
an air flow of 10–25 cm/min).
The present study was performed in strict accordance
with the recommendations of the Guide for the Care and Use of
Laboratory Animals of the National Institutes of Health. The
protocol was approved by the Committee on the Ethics of Animal
Experiments of Shenzhen University (Shenzhen, China). Reagents and
chemicals were analytical grade (Sigma-Aldrich, St. Louis, MO, USA)
and solvents were HPLC grade (Mallinckrodt Australia Pty Ltd., NSW,
Australia). Sodium dodecyl sulphate-polyacrylamide gel
electrophoresis (SDS-PAGE) and western blotting were performed
using a Mini Protean II apparatus (Bio-Rad Laboratories, Inc.,
Hercules, California, USA) with 12% gels and a Tris/Tricine buffer
system. Two-dimensional PAGE was performed using Immobiline IPG
strips (13 cm) with a pH range of 3–10 (GE Healthcare Life
Sciences, Shanghai, China). Al(OH)3 and methacholine
were purchased from Sigma-Aldrich. Biotin-labeled goat anti-mouse
immunoglobulin IgE was purchased from Novus Biologicals, (Colorado,
USA), horseradish peroxidase (HRP)-labeled goat anti-mouse IgG and
IgG2a were obtained from eBioscience, Inc. (San Diego, CA, USA).
Mouse interleukin [IL; IL-4 (catalog no. 431104) and IL-10 (catalog
no. 431414)], and interferon (INF)-γ enzyme-linked immunosorbent
assay (ELISA) kits (catalog no. 430801) were obtained from
BioLegend, Inc. (San Diego, CA, USA). The vector of
pET-28b/Dermatophagoides farinaeI (Derf I) was constructed
in the State Key Laboratory of Respiratory Disease for Allergy,
Shenzhen University (Shenzhen, China).
Expression and purification of
recombinant protein Derf I
Expression of recombinant protein, Derf I was
induced using 1 mM isopropyl β-D-1-thiogalactopyranoside at mid-log
phase at 37°C and the samples were collected 4 h post-induction.
Harvested induced cells were lysed with 10 mg/ml lysozyme in Tris
buffer (100 mM NaH2PO4 and 10 mM Tris-Cl) and
homogenized by sonication. Inclusion bodies were collected by
centrifugation at 12,000 × g for 20 min at 4°C and then washed
three times with Tris buffer containing 0.5% (v/v) Triton X-100.
Inclusion bodies, solubilized in 6 M GuHCl, were purified by Ni-NTA
at 4°C. The recombinant protein was electrophoresed (110 V) on 12%
(w/v) SDS polyacrylamide gels and its protein concentration was
determined using the bicinchoninic acid assay (AppliChem GmbH,
Darmstadt, Germany) according to the manufacturer's protocol.
Sensitization and asthma
challenge
The animal sensitization model was designed for the
following experiment. Briefly, the mice were randomly divided into
two groups (20 mice per group). The model groups were sensitized
intraperitoneally (I.P.) with 50 µg Derf I with 4 mg Al
(OH)3 on days 1, 3 and 7, while the normal group was
sensitized, challenged and treated with phosphate-buffered saline
(PBS) and 4 mg Al(OH)3. The model groups were induced
with 50 µg/ml Derf I every day for one week, which was substituted
with 50 µg/ml PBS in the normal group.
Assessment of AHR to methacholine
challenge
Twenty-four h after the final challenge, AHR was
measured using unrestrained whole-body plethysmography with a
four-chamber system (Buxco Research Systems, Wilmington, NC, USA)
as previously described (25).
PAGE and mass analysis
The recombinant protein Derf I was diluted with an
identical volume of lysis buffer (0.5L) as follows: 9 mol/l Urea,
0.8% IPG buffer (pH, 3–10), 1% dithiothreitol (DTT) and 2%
3-[(3-cholamidopropyl) dimethylammonio] propane-1-sulphonic acid.
Subsequently, PAGE and mass analysis were performed as described by
Cheng et al (26) and Li
et al (27).
Determination of allergen-specific
IgE, IgGand IgG2a antibodies in serum
Blood samples (1 ml) were obtained from the eyes of
the mice, and centrifuged at 4°C and 324 × g for 10 min. The
supernatant serum was collected and the IgE, IgG, and IgG2a were
subsequently analyzed using an indirect ELISA protocol according to
the manufacturer's protocol.
Bronchoalveolar lavage fluid (BALF)
and differential cell counts
BALF was collected as previously described (28) with slight modifications. Briefly,
mice were sacrificed by euthanasia, and BALF was obtained using 1
ml PBS containing protease inhibitor cocktail (Roche Applied
Science, Mannheim, Germany). Cytospin preparations were prepared
and counted using Liu's stain under an optical microscope. Cells
were classified as eosinophils according to morphologic and
histologic criteria (29).
Measurement of cytokines in BALF and
the splenocyte culture medium
The splenocyte cells from the mice were cultured in
RPMI-1640 medium with 10% fetal bovine serum (Thermo Fisher
Scientific, Inc., Waltham, MA, USA) in 24-well plates at a density
of 5×104/well, stimulated with the recombinant Derf I
protein (200 µg/well), and incubated at 37°C for 72 h. The IL-4,
IL-10 and INF-γ levels in the splenocyte culture medium and BALF
were detected using an ELISA kit (catalog no. 421701, Biolegend,
Inc.).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
RT-qPCR was performed to detect the mRNA expression
of ATP5b on an Applied Biosystem 7000 Sequence Detection System
(Thermo Fisher Scientific, Inc., Waltham, MA, USA), which was
performed as described by Ding et al (28) and Zuo et al (30). Five micrograms of total RNA from
each sample was reverse transcribed to cDNA using an A3500 RT
system (Promega Corporation, Madison, WI, USA). The following
forward and reverse primers were used: Forward, AGT TGC TGA GGT CTT
CAC GG and reverse, CTT TGC CAC GGC TTC TTC for ATP5b; and forward,
TGG CAG AGA TGC GTG GAG A and reverse, GGC AAG TCT TCC GAG TAG TTT
T for GAPDH. The PCR conditions were as follows: 5-mindenaturation
at 95°C followed by 40 cycles at 95°C for 30 sec, 55°C for 30 sec,
and 72°C for 30 sec. Amplification of the target gene was monitored
as a function of increased SYBR-Green. I (Qiagen GmbH, Hilden,
Germany) fluorescence. An analysis threshold was set, and the cycle
threshold (Cq) was computed for each sample (31). The comparative difference in gene
expression was then determined.
RNA interference
siRNA targeting human ATP5b was delivered into ASMCs
using the Lipofectamine® 2000 reagent (Invitrogen;
Thermo Fisher Scientific, Inc.) according to the manufacturer's
instructions. In addition, a non-targeting siRNA pool (Dharmacon,
Inc., Lafayette, CO, USA) was used at the same concentration as a
control for the RNA interference assays. After 48 h of
transfection, cells were subjected to MTT assays or RNA were
collected and analyzed by RT-qPCR.
MTT assay
ASMCs were seeded into96-well culture plates at
adensity of 500 cells/well with scramble siRNA-ATP5b and
siRNA-ATP5b. After 48 h, the cell culture was removed and 20 µl of
MTT was added to each well. The cells were further cultured at 37°C
for 4 h, and the cell culture was removed and 150 µl DMSO was added
to dissolve crystals completely by pipetting up and down. A
microplate reader (SynergyH1; Bio-Tek Instruments, Inc., Winooski,
VT, USA) measured the corresponding absorption value at a
wavelength of 562 nm indicated cell proliferation as the number of
live cells was proportional to the optical density value.
Statistical analysis
The data are expressed as the mean ± standard
deviation. Student's t-test was used for the statistical analysis
of interval data and P<0.05 was considered to indicate a
statistically significant difference.
Results
Identification of the murine model of
allergic asthma induced by DerfI
In the current study, DerfI as an important antigen
of asthma was applied to construct the asthma model. Purified
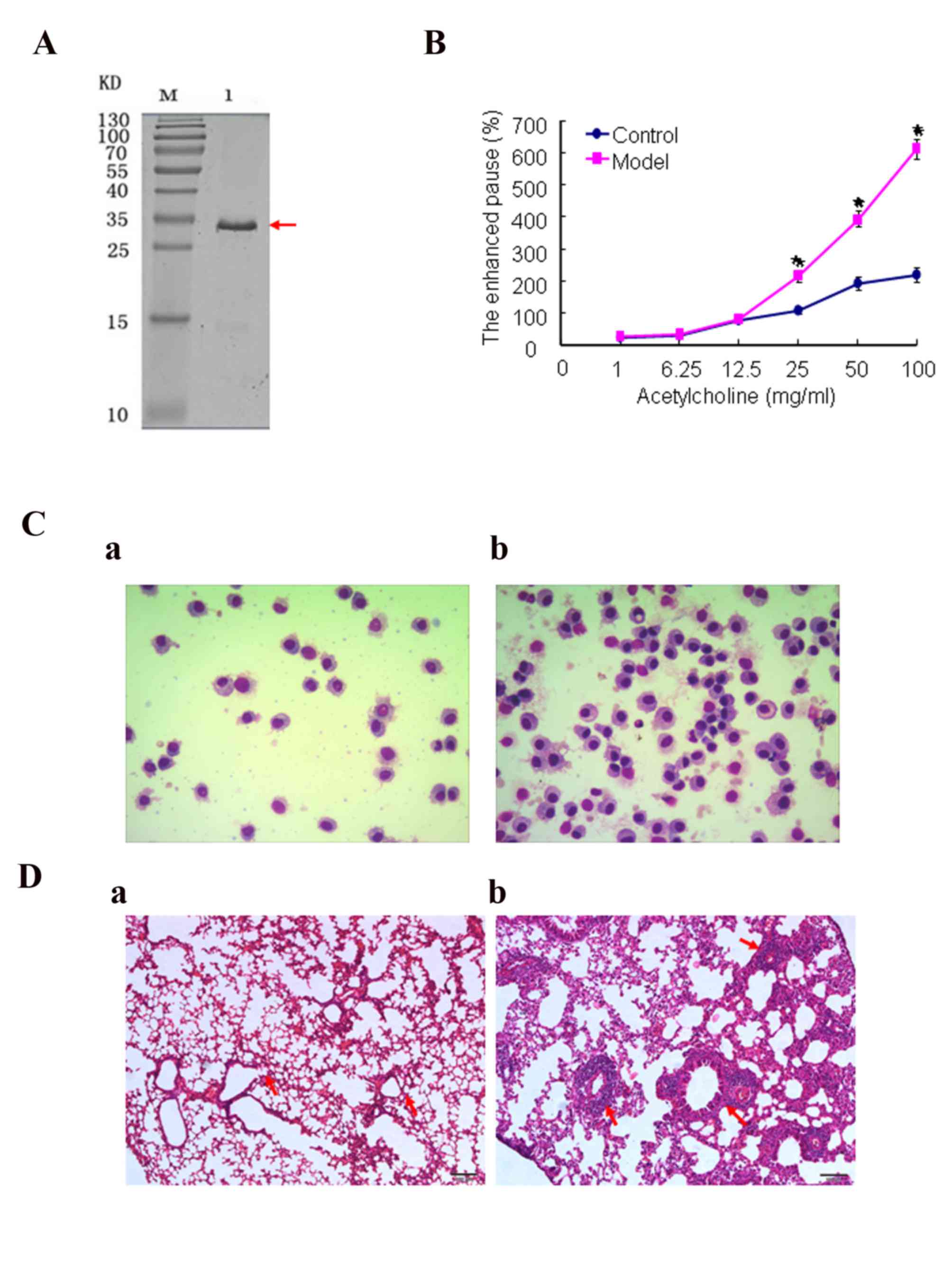
DerfIprotein was obtained (Fig.
1A). AHR changes were assessed by methacholine challenge 24 h
after the final allergen challenge. The results (Fig. 1B) demonstrated that AHR in the
model groups was significantly greater (P<0.05) than in the
normal control group. In addition, the AHR of the mice increased
with the increasing concentration of methacholine. Fig. 1C and Table I showed that the total counts of
cells and inflammatory cells in the BALF sampled from the model
groups were significantly greater than those in the normal group
(P<0.05), particularly the eosinophils. As shown in Fig. 1D, lung tissue samples from the
model group were damaged by internal hemorrhage and edema. The
bronchial and vascular walls became thickened and infiltrated by a
marked number of inflammatory cells. In addition, the trachea in
the models was filled with markedly more mucus than the normal
control group. However, lung tissue structures in the normal group
were well defined without discernible damage or edema, and the
trachea and blood vessels were not infiltrated peripherally by
inflammatory cells. Thus, the asthma model had been successfully
induced.
 | Table I.Total cell number and number of
eosinophils in bronchoalveolar lavage fluid. |
Table I.
Total cell number and number of
eosinophils in bronchoalveolar lavage fluid.
| Cell
(×104/ml) | Control group | Model group |
|---|
| Total cells | 20.30±3.35 |
64.50±5.08a |
| Eosinophils | 0 |
15.32±3.04a |
Allergen-specific T helper (Th)2
immune responses induced by the DerfI protein
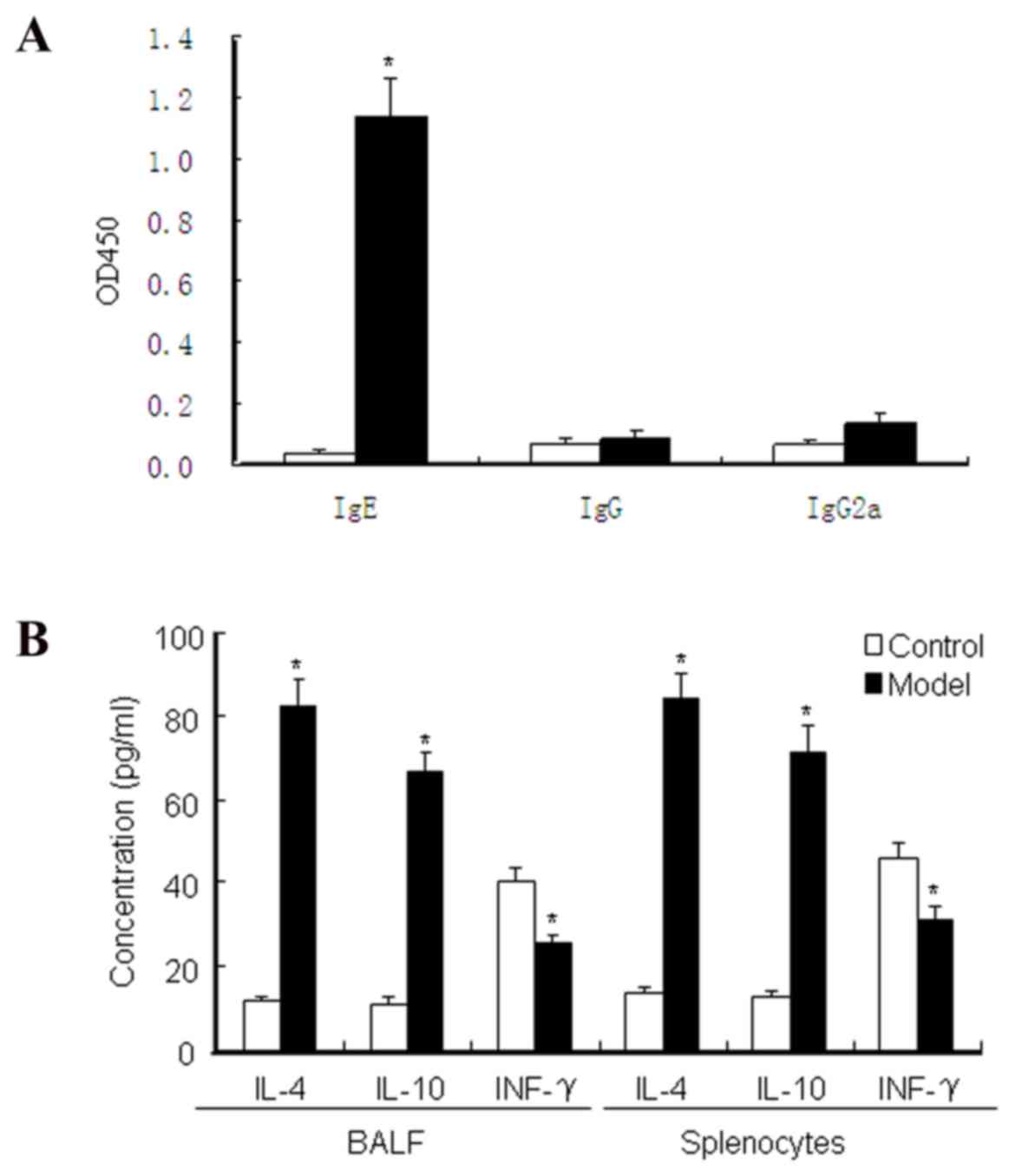
Serum IgE, IgG and IgG2a levels were measured by
ELISA to evaluate the asthma model. The results (Fig. 2A) showed that the levels of serum
IgEin the asthma model group were significantly higher than those
in the normal control group (P<0.05). Furthermore, the
concentrations of IL-4, IL-10 and IFN-γ in the splenocyte culture
medium supernatant and BALF were detected by ELISA. The asthma
model groups (Fig. 2B) exhibited
allergen-specific Th2 immune responses, where by IL-4 and IL-10
levels were evidently upregulated compared with the normal control
group (P<0.05). However, the IFN-γ level in the asthma model
group was significantly lower than that in the normal control group
(P<0.05), which enhances inflammatory and immunosuppressive
function. These data demonstrated that Derf I protein disturbed the
Th1/Th2 balance, which promoted the Th2 immune response and
suppressed the Th1 response.
Mass spectral analysis and
identification of ATP5b
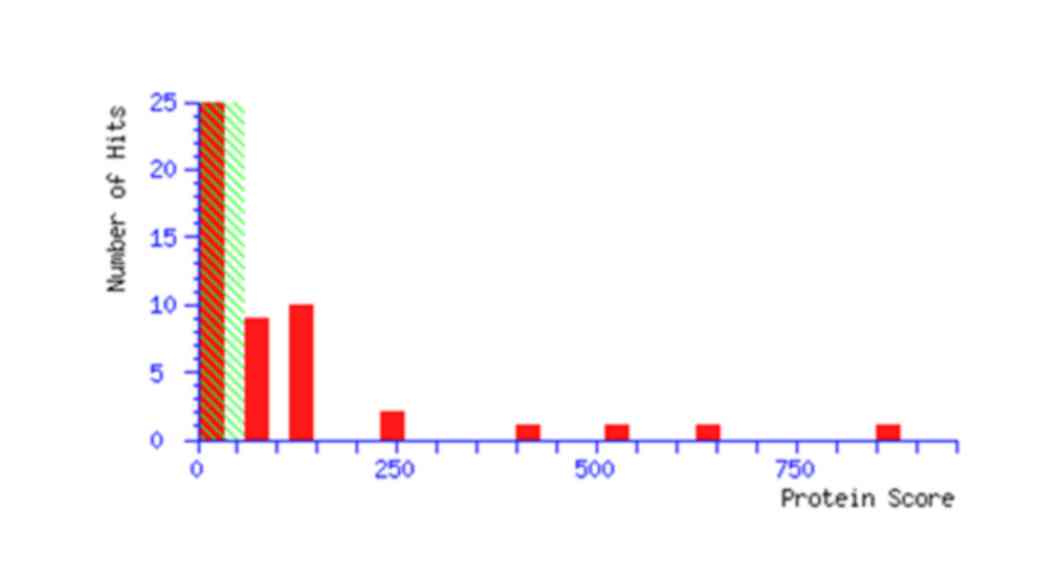
A total of 23 monoisotopic peaks were input into the
Mascot search engine to search the Swiss-Prot database (http://www.gpmaw.com/html/swiss-prot.html). The
annotation of all the identified proteins is summarized in Table II. The query result showed that
protein spot 2606 is the ATP5b protein. In addition, the database
query result and mascot score of a representative ATP5b protein are
demonstrated in Table III and
Fig. 3.
 | Table II.Analysis of mass spectral data. |
Table II.
Analysis of mass spectral data.
| Spots | Master no. | Score | NCBInr no. | Protein name | Expression |
|---|
| 1 | 701 | 382 | gi|7331218 | TPA exp: keratin
Kb40 | − |
| 2 | 1103 | 240 | gi|13775198 | SH3 domain-binding
glutamic acid-rich-like protein 3 | + |
| 3 | 2304 | 269 | gi|55291 | Alpha-tubulin
isotype M-alpha-2 | + |
| 4 | 2602 | 612 | gi|26344461 | Unnamed protein
product | + |
| 5 | 2606 | 1106 | gi|23272966 | Atp5b protein | + |
| 6 | 3302 | 500 | gi|6678281 | Indolethylamine
N-methyltransferase | − |
| 7 | 3401 | 391 | gi|2437840 | Annexin III | − |
| 8 | 3604 | 646 | gi|22164798 | Selenium-binding
liver protein | + |
| 9 | 3701 | 306 | gi|191765 | Alpha-fetoprotein,
partial | − |
| 10 | 3702 | 711 | gi|26340966 | Alpha-fetoprotein,
partial | + |
| 11 | 3803 | 94 | gi|26080429 | Aldehyde
dehydrogenase family 16 member A1 | − |
| 12 | 4605 | 459 | gi|468546 | CCT (chaperonin
containing TCP-1) beta subunit | − |
| 13 | 4805 | 643 | gi|199765 | Moesin | − |
| 14 | 5307 | 135 | gi|7242156 | Acyl-protein
thioesterase 2 | + |
| 15 | 6304 | 163 | gi|54855 | Triosephosphate
isomerase | − |
| 16 | 6307 | 167 | gi|6680121 | Glutathione
S-transferase Mu 2 | + |
| 17 | 6405 | 274 | gi|293317 | Adenylyl
cyclase-associated protein | + |
| 18 | 6702 | 590 | gi|62653546 |
Glyceraldehyde-3-phosphate
dehydrogenase-like | + |
| 19 | 6703 | 859 | gi|6678359 | Transketolase | + |
| 20 | 8303 | 125 | gi|424143 | Heat shock protein
HSP27 | − |
| 21 | 8403 | 569 | gi|62653546 |
Glyceraldehyde-3-phosphate
Dehydrogenase | + |
| 22 | 8408 | 186 | gi|1125026 | 3-hydroxyacyl CoA
dehydrogenase | + |
| 23 | 9301 | 507 | gi|6671688 | Carbonyl reductase
[NADPH] 2 | + |
 | Table III.Mascot scores of representative ATP5b
protein matched peptides. |
Table III.
Mascot scores of representative ATP5b
protein matched peptides.
|
1 | MLSLVGRVAS | ASASGALRGL | SPSAALPQAQ | LLLRAAPAGV | HPARDYAAQA |
| 51 | SAAPKAGTAT | GRIVAVIGAV | VDVQFDEGLP | PILNALEVQG |
RDSRLVLEVA |
| 101 |
QHLGESTVRT | IAMDGTEGLV |
RGQKVLDSGA |
PIKIPVGPET |
LGRIMNVIGE |
| 151 |
PIDERGPIKT | KQFAPIHAEA | PEFIEMSVEQ | EILVTGIKVV | DLLAPYAKGG |
| 201 | KIGLFGGAGV | GKTVLIMELI |
NNVAKAHGGY |
SVFAGVGERT |
REGNDLYHEM |
| 251 | IESGVINLKD |
ATSKVALVYG |
QMNEPPGARA |
RVALTGLTVA |
EYFRDQEGQD |
| 301 | VLLFIDNIFR | FTQAGSEVSA |
LLGRIPSAVG |
YQPTLATDMG |
TMQERITTTK |
| 351 | KGSITSVQAI | YVPADDLTDP | APATTFAHLD |
ATTVLSRAIA |
ELGIYPAVDP |
| 401 |
LDSTSRIMDP |
NIVGNEHYDV |
ARGVQKILQD | YKSLQDIIAI | LGMDELSEED |
| 451 | KLTVSRARKI | QRFLSQPFQV | AEVFTGHMGK | LVPLKETIKG | FQQILAGEYD |
| 501 | HLPEQAFYMV | GPIEEAVAKA | DKLAEEHGS |
|
|
ATP5b was identified as an
overexpressed protein and is involved in cell proliferation
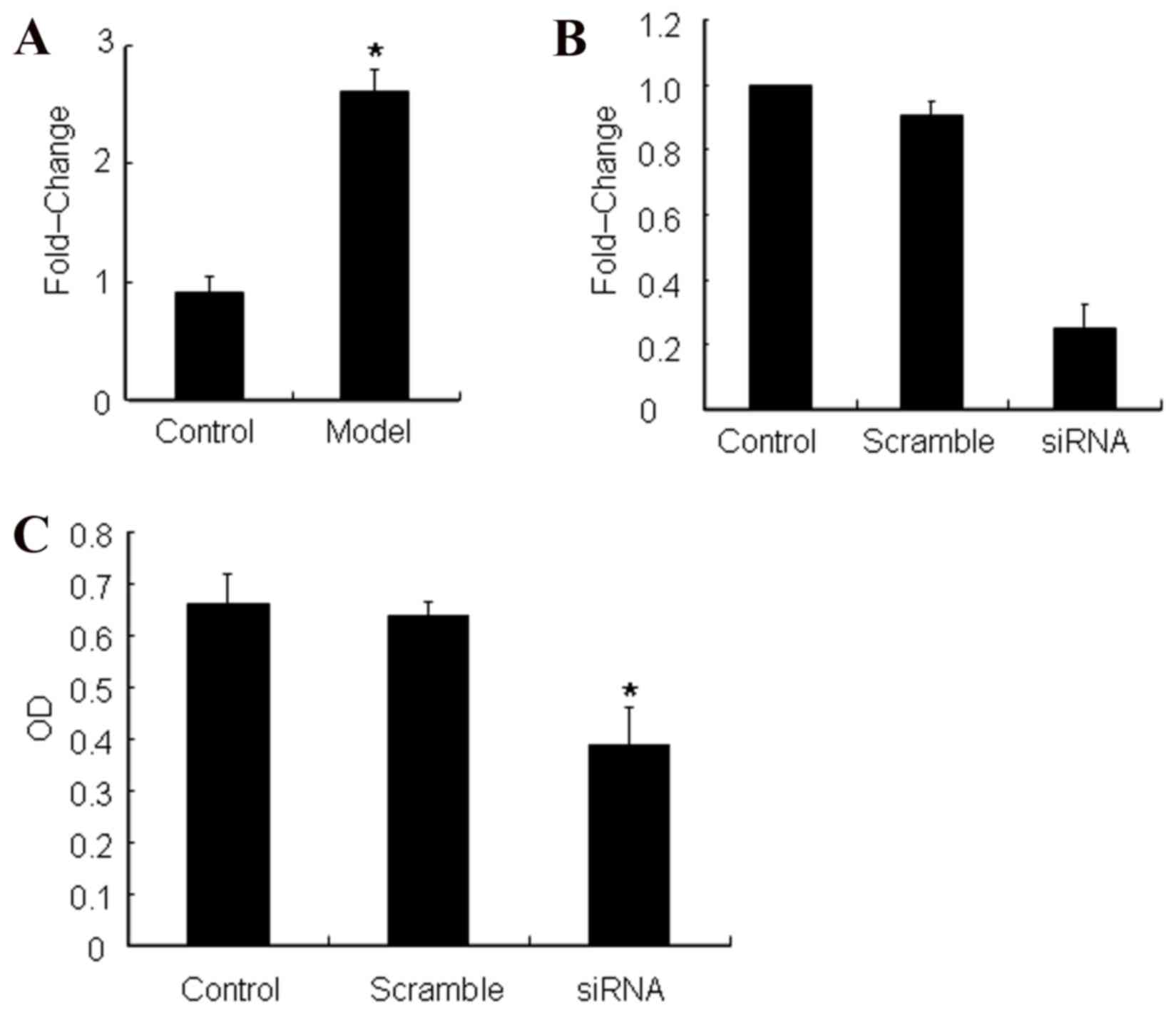
It was further verified by RT-qPCR that ATP5b was
expressed in the asthma model mice to a significantly greater
extent when compared with the normal control mice (Fig. 4A). In order to confirm the role and
function of ATP5b, the experiments were designed to silence ATP5b
expressionin ASMCs and detect its effect on cell proliferation
(Fig. 4B). The data indicated that
silencing of ATP5b inhibited ASMC growth by MTT (Fig. 4C). Thus, overexpression of ATP5b
enhances ASMC proliferation.
Discussion
Asthma is an inflammatory disease of the airways.
Sensitivity to asthma is determined by many factors, such as
multigenic predisposition, aberrant immune response and
environmental factors (32).
More than 50% of asthma cases are caused by
allergies to environmental allergens (33). Clinicians may effectively intervene
in these factors to prevent the development of asthma (34). One of the most frequent allergens
associated with asthma is dust mite, as 10% of individuals with
asthma are allergic to it (35).
Therefore, dust mite is often used to induce experimental asthma in
mice models that closely resemble human asthma (36,37).
Therefore, in the current study, Derf I was selected as the
important dust mite antigen to construct the asthma model.
In the present study, the recombinant protein, Derf
I was used to create the asthma model. The results demonstrate that
construction of the animal model was successful, as the typical
symptoms, including persistent inflammation, hyper-reactivity and
remodeling of the airways (involving smooth muscle thickening,
mucus overproduction and sub epithelial thickening) were observed.
There are certain characteristics of airway inflammation in asthma
resulting from bronchial wall infiltration due to eosinophils, T
lymphocytes and activated mast cells. The current data show that
the model groups had specific Th2 immune responses, where IL-4 and
IL-10 levels were evidently upregulated compared with the normal
control group (P<0.05). However, the IFN-γ level in the model
mice was significantly lower than that of the control mice
(P<0.05). The above data indicated that the DerfI protein
promoted the Th2 immune response and suppressed the Th1 response.
Our results are consistent with those of Mihaltan reports that
cytokines from Th2 cells are key in mediating airway inflammation
(38).
In the current study, ATP5b was observed to be
overexpressed in the asthma model. Recent studies show that ATP5b
is a subunit of a mitochondrial ATP synthase complex (39). ATP5b, as a receptor for various
ligands, is involved in biological processes, such as metabolism of
lipid formation, regulation of proliferation, differentiation and
recognition of immune responses of tumor cells (39,40).
The present study indicated that ATP5b overexpression leads to ASMC
proliferation, since rapidly proliferating cells have a high demand
for energy and glycolytic intermediates for anabolic processes.
Furthermore, certain glycolytic enzymes directly participate in
pathways of proliferation as ATP5b increases ATP extracellularly.
However, when the ATP5b was knocked down in ASMCs, decreased cell
growth was observed. The present results are consistent with the
study by Huang et al (40),
which reports that targeting breast cancer cell lines with ATP5b
inhibitor, aurovertin B decreases proliferation (40,41).
In the present study. The results of proteomics and
RT-qPCR demonstrated that, compared with normal control lung tissue
samples, the lung tissue samples of the asthma mouse models
overexpressed ATP5b, leading to ASMC proliferation and ultimately
to ASM thickening. Thus, to the best of our knowledge, this is the
first study to report that ATP5b overexpression promoted the
proliferation of ASMCs and contributed to airway remodeling.
Acknowledgements
The present study was supported by the National
Nature Science Foundation of China (grant no. 81272960), the Key
Research Program from the Science and Technology Department of
Hunan Province, China (grant no. 2013WK2010), the Key Research
Program from Ministry of Human Resources and Social Security of the
People's Republic of China (grant no. 2016-176), Hunan Province Key
Laboratory of Tumor Cellular and Molecular Pathology (grant no.
2016TP1015) and the Construction Program of the Key Discipline in
Hunan Province, China (Basic Medicine Sciences in University of
South China).
References
|
1
|
Kokotajlo S, Degnan L, Meyers R, Siu A and
Robinson C: Use of intravenous magnesium sulfate for the treatment
of an acute asthma exacerbation in pediatric patients. J Pediatr
Pharmacol Ther. 19:91–97. 2014.PubMed/NCBI
|
|
2
|
Huang F, Zhang H, Wu M, Yang H, Kudo M,
Peters CJ, Woodruff PG, Solberg OD, Donne ML, Huang X, et al:
Calcium-activated chloride channel TMEM16A modulates mucin
secretion and airway smooth muscle contraction. Proc Natl Acad Sci
USA. 109:16354–16359. 2012; View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kanchongkittiphon W, Gaffin JM and
Phipatanakul W: The indoor environment and inner-city childhood
asthma. Asian Pac J Allergy Immunol. 32:103–110. 2014.PubMed/NCBI
|
|
4
|
Hon KL, Leung TF and Leung AK: Clinical
effectiveness and safety of montelukast in asthma. What are the
conclusions from clinical trials and meta-analyses? Drug Des Devel
Ther. 8:839–850. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cannon E: Aligning patient care and asthma
treatment guidelines. Am J Manag Care. 11 14 Suppl:S416–S421; quiz
S427-S433. 2005.PubMed/NCBI
|
|
6
|
Strina A, Barreto ML, Cooper PJ and
Rodrigues LC: Risk factors for non-atopic asthma/wheeze in children
and adolescents: A systematic review. Emerg Themes Epidemiol.
11:52014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Masoli M, Fabian D, Holt S and Beasley R:
Global Initiative for Asthma (GINA) Program: The global burden of
asthma: Executive summary of the GINA Dissemination Committee
report. Allergy. 59:469–478. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Simpson EL: Atopic dermatitis: A review of
topical treatment options. Curr Med Res Opin. 26:633–640. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shaw TE, Currie GP, Koudelka CW and
Simpson EL: Eczema prevalence in the United States: Data from the
2003 National Survey of Children's Health. J Invest Dermatol.
131:67–73. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Van Der Velden J, Sum G, Barker D,
Koumoundouros E, Barcham G, Wulff H, Castle N, Bradding P and
Snibson K: K(Ca)3.1 channel-blockade attenuates airway
pathophysiology in a sheep model of chronic asthma. PLoS One.
8:e668862013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li F, Zhou Y, Li S, Jiang F, Jin X, Yan C,
Tian Y, Zhang Y, Tong S and Shen X: Prevalence and risk factors of
childhood allergic diseases in eight metropolitan cities in China:
A multicenter study. BMC Public Health. 11:4372011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Redhu NS, Shan L, Al-Subait D, Ashdown HL,
Movassagh H, Lamkhioued B and Gounni AS: IgE induces proliferation
in human airway smooth muscle cells: Role of MAPK and STAT3
pathways. Allergy Asthma Clin Immunol. 9:412013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Brannan JD and Lougheed MD: Airway
hyperresponsiveness in asthma: Mechanisms, clinical significance,
and treatment. Front Physiol. 3:4602012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Holgate ST: The airway epithelium is
central to the pathogenesis of asthma. Allergol Int. 57:1–10. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Knight DA and Holgate ST: The airway
epithelium: Structural and functional properties in health and
disease. Respirology. 8:432–446. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gregory LG, Mathie SA, Walker SA, Pegorier
S, Jones CP and Lloyd CM: Overexpression of Smad2 drives house dust
mite-mediated airway remodeling and airway hyper-responsiveness via
activin and IL-25. Am J Respir Crit Care Med. 182:143–154. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yamagata S, Tomita K, Sato R, Niwa A,
Higashino H and Tohda Y: Interleukin-18-deficient mice exhibit
diminished chronic inflammation and airway remodelling in
ovalbumin-induced asthma model. Clin Exp Immunol. 154:295–304.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gabehart KE, Royce SG, Maselli DJ,
Miyasato SK, Davis EC, Tang ML and Le Saux CJ: Airway
hyperresponsiveness is associated with airway remodeling but not
inflammation in aging Cav1-/- mice. Respir Res. 14:1102013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zuo J, Wen M, Lei M, Xiao X and Liu Z:
PLGA-Der p1 vaccine inhibited tumor growth in a murine model of
lung cancer. Arch Med Res. Dec 16–2015.(Epub ahead of print).
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Duramad P, Tager IB, Leikauf J, Eskenazi B
and Holland NT: Experession of Th1/Th2 cytokines in human blood
after in vitro treatment with chlorpyrifos, and its metabolites, in
combination with endotoxin lps and allergen derp1. J Appl Toxicol.
26:458–465. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Rachmiel M, Bloch O, Bistrizer T, Weintrob
N, Ofan R, Koren-Morag N and Rapoport MJ: Th1/Th2 cytokine balance
in patients with both type 1 diabetes mellitus and asthma.
Cytokine. 34:170–176. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Placeres-Uray FA, Febres-Aldana CA,
Fernandez-Ruiz R, de Alfonzo Gonzalez R, de Becemberg IA Lippo and
Alfonzo MJ: M2 Muscarinic acetylcholine receptor modulates rat
airway smooth muscle cell proliferation. World Allergy Organ J.
6:222013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yan L, Xiao-Ling S, Zheng-Yan C, Guo-Ping
L, Sen Z and Zhuang C: HSP70/CD80 DNA vaccine inhibits airway
remodeling by regulating the transcription factors T-bet and GATA-3
in a murine model of chronic asthma. Arch Med Sci. 9:906–915. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wei Y, Xu YD, Yin LM, Wang Y, Ran J, Liu
Q, Ma ZF, Liu YY and Yang YQ: Recombinant rat CC10 protein inhibits
PDGF-induced airway smooth muscle cells proliferation and
migration. Biomed Res Int. 2013:6909372013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xiao X, Zeng X, Zhang X, Ma L, Liu X, Yu
H, Mei L and Liu Z: Effects of Caryota mitis profilin-loaded PLGA
nanoparticles in a murine model of allergic asthma. Int J
Nanomedicine. 8:4553–4562. 2013.PubMed/NCBI
|
|
26
|
Cheng AL, Huang WG, Chen ZC, Peng F, Zhang
PF, Li MY, Li F, Li JL, Li C, Yi H, et al: Identification of novel
nasopharyngeal carcinoma biomarkers by laser capture
microdissection and proteomic analysis. Clin Cancer Res.
14:435–445. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li S, Li J, Hu T, Zhang C, Lv X, He S, Yan
H, Tan Y, Wen M, Lei M and Zuo J: Bcl-2 overexpression contributes
to laryngeal carcinoma cell survival by forming a complex with
Hsp90β. Oncol Rep. 37:849–856. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ding D, Enriquez-Algeciras M, Dave KR,
Perez-Pinzon M and Bhattacharya SK: The role of deimination in
ATP5b mRNA transport in a transgenic mouse model of multiple
sclerosis. EMBO Rep. 13:230–236. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Metso T, Venge P, Haahtela T, Peterson CG
and Sevéus L: Cell Specific markers for eosinophils and neutrophils
in sputum and bronchoalveolar lavage fluid of patients with
respiratory conditions and healthysubjects. Thorax. 57:449–451.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zuo JH, Zhu W, Li MY, Li XH, Yi H, Zeng
GQ, Wan XX, He QY, Li JH, Qu JQ, et al: Activation of EGFR promotes
squamous carcinoma SCC10A cell migration and invasion via inducing
EMT-like phenotype change and MMP-9-mediated degradation of
E-cadherin. J Cell Biochem. 112:2508–2517. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu MC, Hubbard WC, Proud D, Stealey BA,
Galli SJ, Kagey-Sobotka A, Bleecker ER and Lichtenstein LM:
Immediate and late inflammatory responses to ragweed antigen
challenge of the peripheral airways in allergic casthmatics.
Cellular, mediator, and permeability changes. Am Rev Respir Dis.
144:51–58. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhao G, Lin X, Zhou M and Zhao J:
Association between CC10 +38A/G polymorphism and asthma risk: A
meta-analysis. Pak J Med Sci. 29:1439–1443. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Inoue Y and Shimojo N: Epidemiology of
virus-induced wheezing/asthma in children. Front Microbiol.
4:3912013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bessot JC and Pauli G: Mite allergens: An
overview. Eur Ann Allergy Clin Immunol. 43:141–156. 2011.PubMed/NCBI
|
|
36
|
Yao X, Dai C, Fredriksson K, Dagur PK,
McCoy JP, Qu X, Yu ZX, Keeran KJ, Zywicke GJ, Amar MJ, et al: 5A,
an apolipoprotein A-I mimetic peptide, attenuates the induction of
house dust mite-induced asthma. J Immunol. 186:576–583. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Yao X, Fredriksson K, Yu ZX, Xu X,
Raghavachari N, Keeran KJ, Zywicke GJ, Kwak M, Amar MJ, Remaley AT
and Levine SJ: Apolipoprotein E negatively regulates house dust
mite-induced asthma via a low-density lipoprotein receptor-mediated
pathway. Am J Respir Crit Care Med. 182:1228–1238. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Mihălţan F and Ulmeanu R: Tiotropium in
asthma-a new opportunity? Pneumologia. 63:200–202. 2014.(In
Romanian). PubMed/NCBI
|
|
39
|
García J: The calcium channel α2/δ1suunit
interacts with ATP5b in the plasma membrane of developing muscle
cells. Am J Physiol Cell Physiol. 301:C44–C52. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Huang TC, Chang HY, Hsu CH, Kuo WH, Chang
KJ and Juan HF: Targeting therapy for breast carcinoma by ATP
synthase inhibitor aurovertin B. J Proteome Res. 7:1433–1444. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hjerpe E, Brage S Egyhazi, Carlson J,
Stolt M Frostvik, Schedvins K, Johansson H, Shoshan M and
Avall-Lundqvist E: Metabolic markers GAPDH, PKM2, ATP5B and
BEC-index in advanced serous ovarian cancer. BMC Clin Pathol.
13:302013. View Article : Google Scholar : PubMed/NCBI
|