Introduction
Neighbouring normal cells are recruited by cancer
cells to perform tumour processes. Thus, the study of cancer
requires not only the focus on tumour cell activity, but to also
evaluate the activity of the neighbouring normal cells (1). Macrophages are the most abundant
non-tumour cell in cancers (2).
Plasticity and flexibility are key features of macrophages because
their cytokine production responds to specific environmental cues
(3,4). According to their functional
properties, previous studies proposed the concept of macrophage
polarization (1,5). There are two phenotypes of polarized
macrophages: M1 (classical) and M2 (alternative) macrophages. M1
macrophages, also termed classically-activated macrophages, which
exhibit a strong proinflammatory and pathogen-killing effect in
tissues. They are characterized by the production of destructive
free radicals and inflammatory cytokines (6). M2 macrophages, which are also
alternatively called activated macrophages, serve a role in
immunoregulation, tissue remodelling and angiogenesis. It is now
generally accepted that tumour-associated macrophages (TAMs) have
an M2 phenotype and have the ability to promote tumour growth,
migration, invasion and metastasis in various cancers (7–9).
Therefore, the examination of TAMs may be a valuable addition to
standard of care therapies.
Without vascular proliferation, tumour growth is
restricted. Therefore, the process of angiogenesis is important for
the growth and metastasis of tumors (10). Endothelial cell proliferation,
migration and extracellular matrix decomposition are the three main
stages of new blood vessel formation. This process is regulated by
angiogenic factors, which are released from host cells and tumour
cells, and is controlled by positive and negative regulatory
factors. These vascular factors interact with receptors in the
vicinity of blood vessels and induce endothelial cell activation,
proliferation and migration towards the tumour. Inhibiting
angiogenesis is an effective method for treating cancer.
The tumour microenvironment is a special environment
that is formed by the interaction of tumour cells, stromal cells
and extracellular matrix. During the progression of numerous
malignant tumours, hypoxia is one of the most common
microenvironmental conditions and it may stimulate the expression
of vascular endothelial growth factor (VEGF) (11). Hypoxia-inducible factor-1α (HIF-1α)
is a key protein upregulated by hypoxia. HIF-1α regulates the
expression of VEGF at the gene level and influences tumour
development. Signal transducer and activator of transcription 3
(STAT3) is an important component of the signal transducer and
activator of transcription family of proteins. STAT3 was initially
identified as a component of the interleukin (IL)-6/Janus kinase
signalling pathway and was confirmed to participate in numerous
physiological processes, including cell proliferation, apoptosis
and differentiation (12). Its
role in the development of cancer should not be overlooked.
Numerous medicinal plants or chemical substances
extracted from plants are potential anti-vascular drugs. Flavonoids
have been reported to affect the initiation and development of
tumours. Luteolin, a common dietary flavonoid, is found in fruits,
vegetables and herbs. This compound has a variety of functions that
induce anti-tumour effects, including the inhibition of tumour cell
proliferation and the promotion of tumour cell apoptosis (13). However, the mechanisms by which
luteolin exerts its effects on angiogenesis and on M2-like TAMs,
under hypoxic conditions, are unclear. In the present study, a
chemical hypoxic model was established using CoCl2
(14) to investigate whether
hypoxia promotes the expression of HIF-1α and VEGF in M2-like TAMs.
Furthermore, by culturing cells in the presence of CoCl2
and luteolin, under hypoxic conditions, the present study
investigated whether luteolin has an anti-cancer role via M2-like
TAMs.
Materials and methods
Reagents
Luteolin (Sichuan Chengdu Mansite Biotechnology Co.
Ltd., Sichuan China) was stored at −4°C and dissolved in dimethyl
sulfoxide prior to use. Cobalt chloride (CoCl2) was
obtained from Sigma-Aldrich (Merck-KGaA, Darmstadt, Germany) and
IL-4 was from PeproTech EC Ltd (London, UK). Foetal bovine serum
(FBS) was purchased from Sigma-Aldrich (Merck KGaA, Darmstadt,
Germany). Trypsin and Dulbecco's modified Eagle's medium (DMEM)
were purchased from Gibco (Thermo Fisher Scientific, Inc., Waltham,
MA, USA). Anti-HIF-1α (cat. no. ab113642), anti-VEGF (cat. no.
ab69479), anti-MMP9 (cat. no. ab38898) and anti-tissue inhibitor of
metalloproteinase 1 (TIMP1) antibodies (cat. no. ab38978) were
purchased from Abcam (Cambridge, MA, USA). The anti-β-actin
antibody (cat. no. ap0731) was purchased from Bioworld Technology
Inc. (St. Louis Park, MN, USA). Antibodies against STAT3 (cat. no.
12640) and phosphorylated (p)-STAT3 (cat. no. 9145) were obtained
from Cell Signaling Technology, Inc. (Danvers, MA, USA).
Cell culture
The mouse macrophage cell line RAW264.7 was obtained
from The Cell Bank of Type Culture Collection of the Chinese
Academy of Sciences (KCB200603YJ, Shanghai, China). Cells were
cultured in DMEM with 10% FBS, 100 U/ml penicillin and 100 µg/ml
streptomycin in a 5% CO2 incubator at 37°C. When the
cells were ~80% confluent, trypsin containing 0.25% EDTA was used
to detach the cells from the plates to proceed with the specific
experimental treatments.
Induced polarization
To induce the polarization of macrophages into
M2-like TAMs, the cells were treated with IL-4 (10 ng/ml) for 2 h
at 37°C. Macrophages and M2-like TAMs were used for the following
experiments.
Cell viability assay
A Cell Counting Kit-8 (CCK8, Dojindo Molecular
Technologies, Inc., Kumamoto, Japan) was used to detect whether
varying concentrations of CoCl2 (25, 50, 100, 150, 200,
250, 300 or 600 µM) and luteolin (5, 10, 20, 30, 40, 60, 80 or 160
µM) affected the survival rate of mouse macrophages. Cells
(1×104) were plated in 96-well plates, and at ~70%
confluence, the medium was replaced with serum-free DMEM for 12 h.
A total of 10 µl of CCK8 solution was added to each well following
incubation with various concentrations of CoCl2/luteolin
for 24 h at 37°C. After 2 h, a microplate reader was used to
measure the absorbance of each well at 450 nm.
Cell invasion and migration
assays
First, to polarize macrophages to M2-like TAMs, when
cells were ~80–90% confluent, the cells were treated with IL-4 for
2 h at 37°C. Then, the macrophages and TAMs were used for the
following experiments. To test cell invasion and migration,
Transwell filter chambers (Corning, Inc., Corning, NY, USA) were
used with an 8-µm pore size, which were coated with or without
Matrigel basement membrane matrix (Corning, Inc.). In the invasion
assays, 100 µl serum-free DMEM with 5×104 cells was
added to the upper chamber, and 600 µl DMEM with 10% FBS was added
to the lower Transwell chamber. Conversely, for the migration
assays, 100 µl serum-free DMEM with 5×103 cells was
added to the upper chamber, and 600 µl DMEM with 10% FBS was placed
in the lower chamber. Following the addition of CoCl2
(100 µM) or luteolin (20 µM), the cells were placed in in a 5%
CO2 incubator for 24 h at 37°C. Subsequently, the cells
were washed three times with PBS, and the chambers were soaked in
4% paraformaldehyde for 30 min at room temperature. Following three
washes with PBS, the cells were stained with haematoxylin for 20
min at room temperature. Finally, an inverted microscope at
magnification of ×200 was used to observe and count the stained
cells by randomly selecting 10 fields of view.
Reverse transcription-quantitative
polymerase chain reaction (qPCR)
First, cells were treated with IL-4 for 2 h at 37°C
to polarize macrophages into M2-like TAMs. When cells were 80–90%
confluent, the cells were then cultured with CoCl2 (100
µM) for 2 h and with luteolin (20 µM) for 24 h. TRIzol reagent
(Thermo Fisher Scientific, Inc.) was used to extract total RNA
according to the manufacturer's protocols. The quantitative
analysis of β-actin, MMP9, VEGF and TIMP1 expression was performed
using a Fast Start Universal SYBR Green Master kit (ROX; Roche
Diagnostics, Basel, Switzerland) on a 7500 Real-Time PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.). The reactions
were then subjected to the following temperature protocol: Stage 1,
95°C for 3 min; stage 2, 95°C for 15 sec and 60°C for 1 min for 40
cycles, and stage 3, 95°C for 15 sec, 60°C for 1 min, 95°C for 15
sec and 60°C for 15 sec. Finally, the 2−ΔΔCq method
(15) was to calculate the
relative expression of the target gene for the analysis. The
details: ΔΔCq=(Cqexperimental group target
gene-Cqexperimental group β-actin
gene)-(Cqcontrol group target gene-Cqcontrol
group β-actin gene). The qPCR primers were
purchased from Synbio-Tech (Jiangsu, China), and their sequences
are listed in Table I.
 | Table I.Primers used in reverse
transcription-quantitative polymerase chain reaction. |
Table I.
Primers used in reverse
transcription-quantitative polymerase chain reaction.
| Gene | Sense (5′-3′) | Antisense
(5′-3′) |
|---|
| VEGF |
ACTTTCTGCTCTCTTGGGT |
GACTTCTGCTCTCCTTCTGT |
| MMP9 |
TACGATAAGGACGGCAAA |
CAAAGATGAACGGGAACA |
| TIMP1 |
TCTGGCATCCTCTTGTTG |
GGTGGTCTCGTTGATTTCT |
| β-actin |
GGGAAATCGTGCGTGAC |
AGGCTGGAAAAGAGCCT |
Western blot analysis
Following the aforementioned treatments, at 80–90%
confluence, total proteins were extracted from the cells.
Radioimmunoprecipitation assay lysate buffer (Beyotime Institute of
Biotechnology, Shanghai, China) with 10% phosphatase inhibitors
(Roche Diagnostics GmbH, Mannheim, Germany) and 1% PMSF (Beyotime
Institute of Biotechnology). A Bicinchoninic Acid kit (Beyotime
Institute of Biotechnology) was used to determine the protein
concentration. Then, 50 µg protein was dissolved in loading buffer
containing SDS and was heated for 5 min at 100°C. Following
denaturation, 10% SDS-PAGE was used to isolate the proteins, which
were transferred onto polyvinylidene fluoride (PVDF) membranes via
wet blotting transfer. The PVDF membranes were incubated with
tris-buffered saline and Tween-20 (TBST) with 5% skim milk for 1.5
h at room temperature. Various antibodies at different dilutions
(HIF-1α, VEGF, MMP9, TIMP1, STAT3 and p-STAT3 all 1:1,000 and
β-actin 1:10,000) were incubated with the membranes overnight at
4°C. Following three washes with TBST, the membranes were incubated
with horseradish peroxidase-conjugated secondary antibodies (goat
anti-rabbit immunoglobulinG (H+L)-horseradish peroxidase; 1:10,000;
cat. no. BS13278, Bioworld Technology, Inc.) for 1.5 h at room
temperature. Enhanced chemiluminescence was used to detect and
image the bands with a Bio-Rad gel imaging system (version 5.1,
Bio-Rad Laboratories, Inc., Hercules, CA, USA) or with X-ray
film.
Immunofluorescence staining
Following the aforementioned treatments, cells were
washed with PBS and fixed in 4% paraformaldehyde for 15 min at 4°C
Then, the cells were permeabilized with 0.5% Triton X-100 for 10
min and blocked with PBS containing 10% goat serum (Beyotime
Institute of Biotechnology) for 1 h at room temperature.
Subsequently, the cells were incubated with anti-VEGF antibody
(1:200) at 4°C overnight. Following three washes with PBS, the
cells were incubated with a secondary antibody conjugated with
Alexa Fluor 488 (1:200; cat. no. A-11034; Thermo Fisher Scientific,
Inc.) at room temperature for 1 h. Following three washes with PBS,
DAPI was used to stain the cell nuclei for 5 min at room
temperature. Finally, the glass slides were photographed using an
automated upright microscope system with anti-fluorescence
quenching (magnification, ×400).
Statistical analysis
All analyses were performed using SPSS version 19.0
software (IBM Corp., Armonk, NY, USA). The results are expressed as
the means ± standard deviation of multiple experiments or
representative images. One-way analysis of variance followed by
Tukey's Multiple Comparison test) was used to assess significance
when evaluating the statistical correlation of data between groups.
P<0.05 was considered to indicate a statistically significant
difference; all experiments were repeated three times.
Results
Effect of luteolin or CoCl2
on cell survival rate and the impact of CoCl2 on the
expression of HIF-1α and VEGF
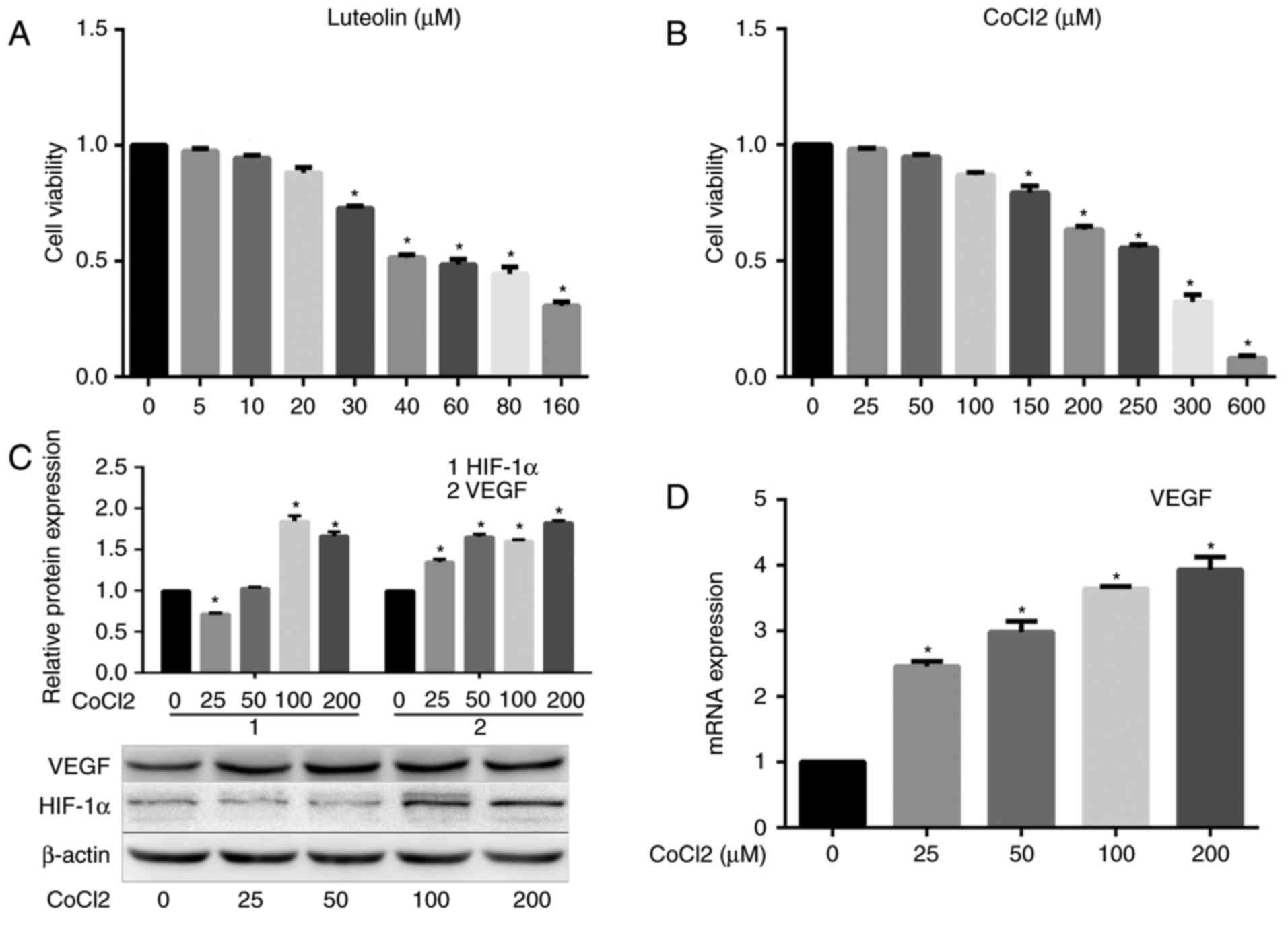
A CCK8 assay was used to investigate the effect of
various concentrations of luteolin and CoCl2 on cell
survival rate. In the CCK8 experimental results with luteolin
treatment only, the cell survival rate was not significantly
reduced following treatment with 5,10 or 20 µM of luteolin
(Fig. 1A). However, upon treatment
with >20 µM luteolin, the cell survival rate was significantly
decreased. The CCK8 experimental results with CoCl2
treatment confirmed that 100 µM CoCl2 did not
significantly decrease the cell survival rate (Fig. 1B). However, concentrations of
CoCl2 ≥150 µM caused a significant decline in the cell
survival rate. At these concentrations, CoCl2 led to a
substantial increase in macrophage death. Therefore, the expression
of VEGF and HIF-1α was used to evaluate the efficacy of the hypoxic
environment induced by CoCl2. The western blotting
results demonstrated that HIF-1α expression was low in the normal
group. However, when the concentration of CoCl2 was
increased, the expression of HIF-1α also increased; the same result
was observed for the protein and mRNA expression of VEGF (Fig. 1C and D). Treatment with 200 µM
CoCl2 did not markedly increase the expression of these
proteins. These results indicated that CoCl2 (100 µM)
effectively induced a hypoxic environment and provided appropriate
conditions for the following experiments. A total of 20 µM luteolin
and 100 µM CoCl2 were used for subsequent
experiments.
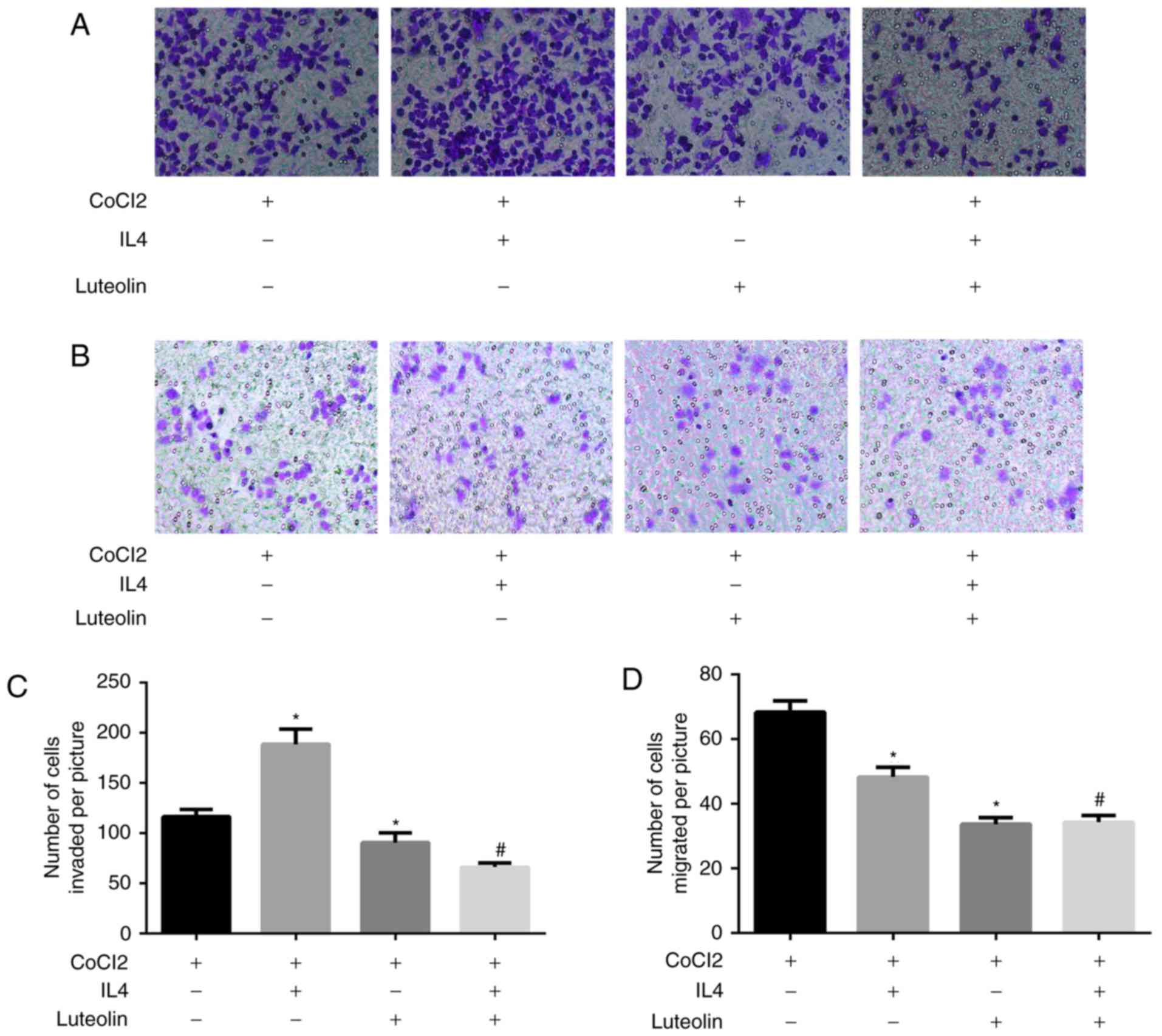
Effects of luteolin on invasion and
migration
Transwell invasion and migration assays were used to
detect the effect of luteolin on cell invasion and migration.
Following the addition of IL-4, the invasive ability of the cells
was increased. Under the hypoxic environment, the invasion ability
of M2-like TAMs was greater than that of the macrophages, which was
consistent with the MMP9 expression results from western blotting
described below. However, following treatment with luteolin, the
invasive capability of these two cell types was significantly
weakened, particularly that of the M2-like TAMs (Fig. 2A and C). In addition, in the
migration experiment, the number of M2-like TAMs was significantly
lower than the number of macrophages untreated with IL-4. The
migration ability of the cells was also significantly inhibited
following treatment with luteolin, particularly in the M2-like TAMs
treated with IL-4 (Fig. 2B and D).
Therefore, the results of the present study indicated that luteolin
may inhibit M2-like TAM and macrophage invasion and migration,
particularly in the M2-like TAMs.
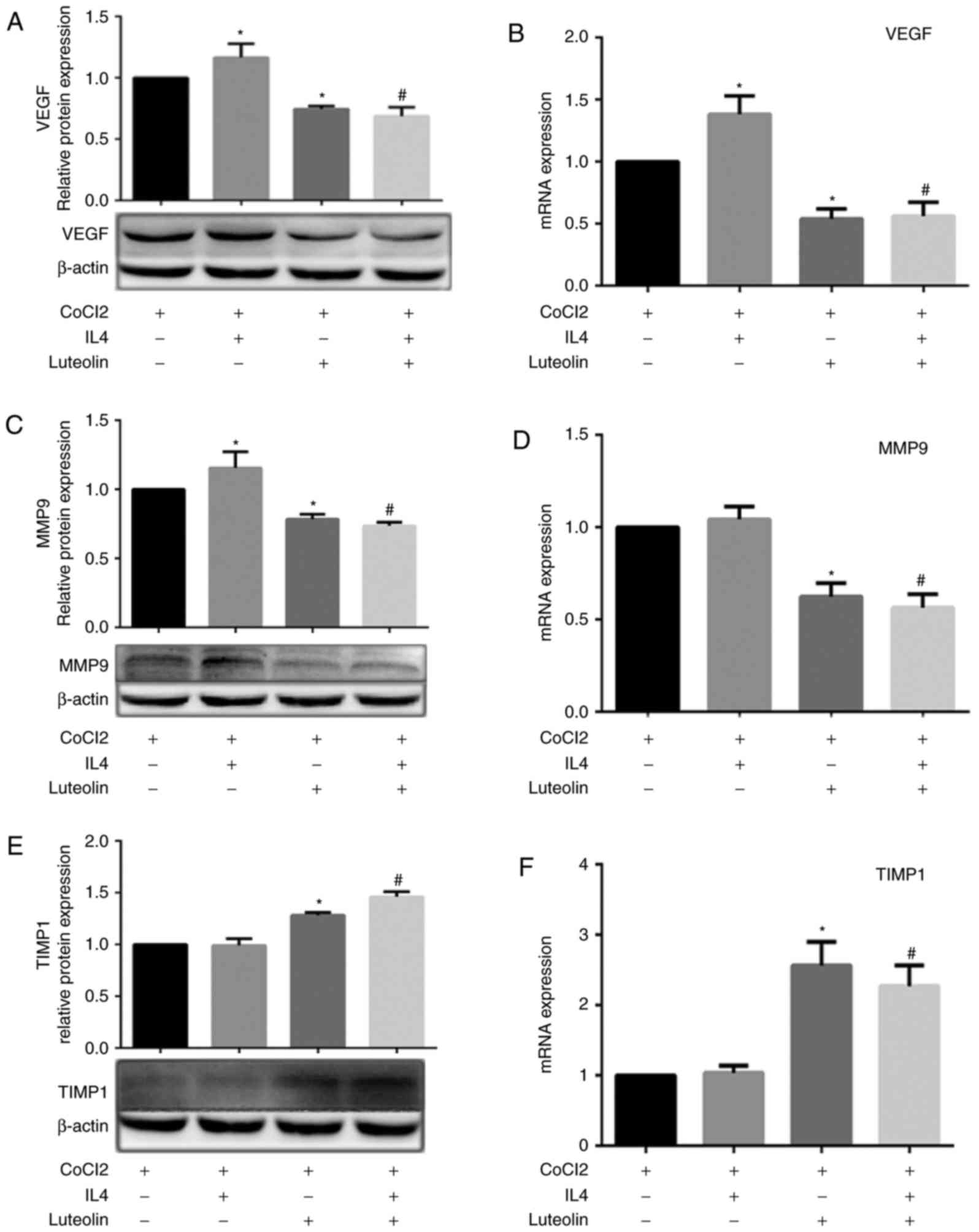
Under hypoxic conditions, luteolin
regulates angiogenesis
As hypoxic environments promote angiogenesis, the
ability of M2-like TAMs to promote angiogenesis was increased
compared with that of macrophages in a hypoxic environment. VEGF
and MMP9 are the main factors that promote angiogenesis (16–18).
The expression levels of VEGF and MMP9 were higher within M2-like
TAMs than in macrophages, as presented by western blotting and PCR
experiments (Fig. 3A-D). However,
following treatment with luteolin (20 µM), the expression levels of
VEGF and MMP9 were significantly decreased. In addition, the
expression of TIMP1, a biological inhibitor of MMP9, was increased
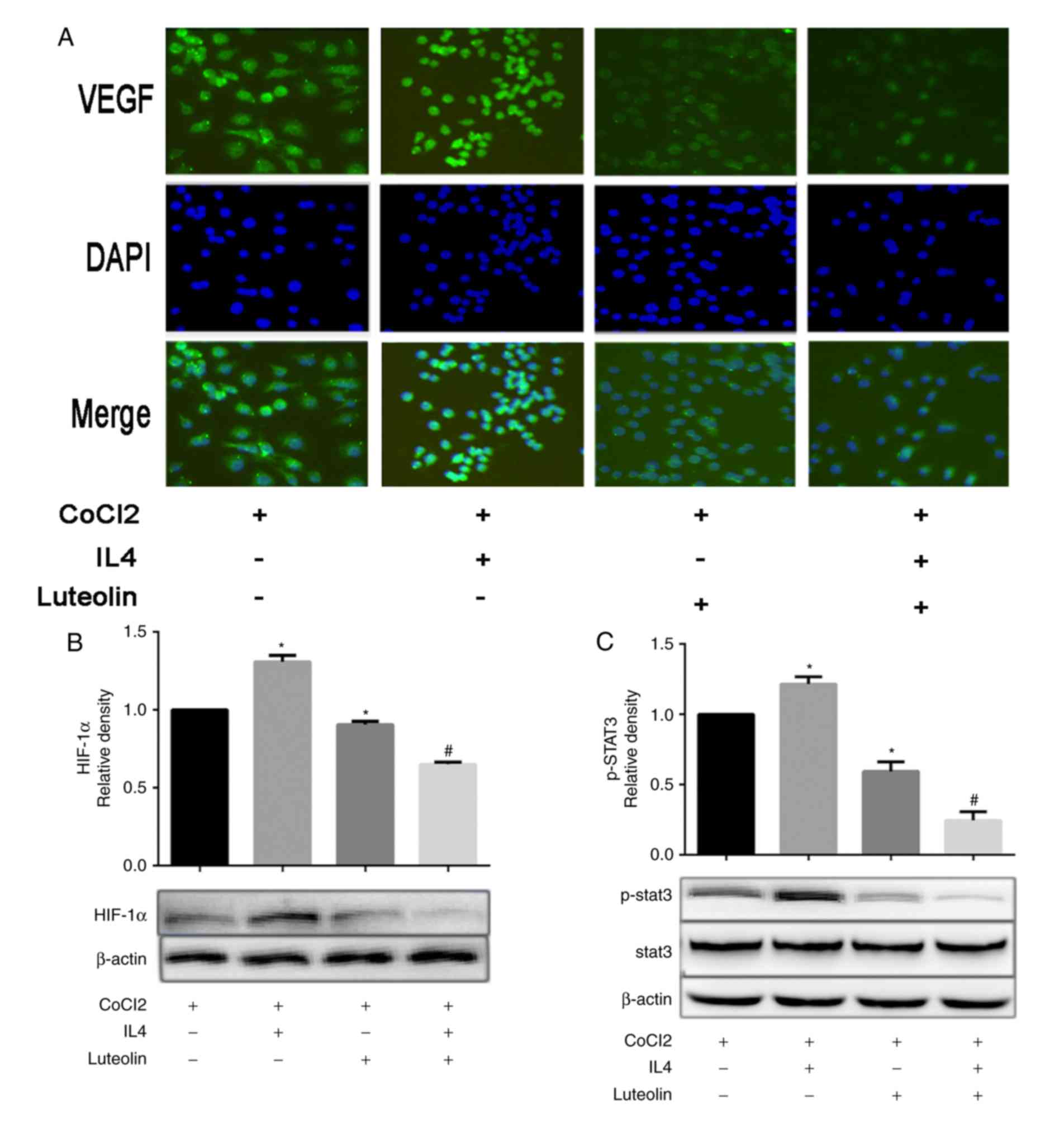
when cells were treated with luteolin (20 µM; Fig. 3E and F). Immunofluorescence also
revealed that the expression of VEGF was lower in macrophages than
in M2-like TAMs (Fig. 4A). The
expression of VEGF was inhibited following the addition of
luteolin, which was consistent with the PCR and western blotting
results. Therefore, luteolin may inhibit the ability of macrophages
and M2-like TAMs in particular, to promote angiogenesis.
Under hypoxia, luteolin regulates the
HIF-1α and STAT3 signalling pathways
There is a close association between angiogenesis
and the STAT3 and HIF-1α signalling pathways: Both pathways have
been previously reported to contribute to the formation of blood
vessels (16,19). In the hypoxic environment of the
present study, the ability of M2-like TAMs to promote angiogenesis
was confirmed by the activation of the HIF-1α and p-STAT3
signalling pathways (Fig. 4B and
C). The present study demonstrated via western blotting that
STAT3 expression was not significantly different among the groups.
This indicated that within M2-like TAMs, the STAT3 signalling
pathway may have served an angiogenic role via the phosphorylation
of STAT3 rather than by an increase in STAT3 expression. Following
treatment with luteolin (20 µM), the expression of HIF-1α and
p-STAT3 was significantly reduced, possibly inhibiting the
expression of angiogenic factors, such as VEGF and MMP9. These data
indicated that under hypoxic environments, luteolin may mainly have
an anti-angiogenic role via the inhibition of the HIF-1α and
p-STAT3 signalling pathways, particularly within the M2-like
TAMs.
Discussion
Angiogenesis occurs due to the proliferation of
tumour cells, which causes an increased demand for nutrients and
oxygen (20). Angiogenesis has an
essential role in tumour processes, including migration, metastasis
and growth and is regulated by pro-angiogenic and anti-angiogenic
factors (21). The process of
angiogenesis includes the activation, proliferation and migration
of endothelial cells. In most malignant tumours, the factors that
induce angiogenesis are increased; among these, VEGF is the most
important (22). VEGF is a highly
specific vascular endothelial cell mitogen that has an important
role in physiological and pathological processes (22). VEGF exerts its biological effects
by binding to the tyrosine kinase receptors expressed on
endothelial cells. The VEGF receptor belongs to the tyrosine kinase
family, and based on structure and function, this receptor is
divided into three types: VEGFR1 (Flt21), VEGFR2 (KDR/Flk21) and
VEGFR3 (Flk24) (23). VEGFR2 and
VEGFR1 are mainly expressed on vascular endothelial cells and have
an important role in the formation of blood vessels (24). Conversely, VEGFR3 is mainly
expressed within lymphatic endothelial cells. VEGF achieves its
effect by primarily interacting with VEGFR2 (25). VEGF binding to its receptor
directly promotes endothelial cell mitosis, induces plasminogen
degradation and leads to increased vascular permeability, inducing
the exudation of plasma proteins, and thereby promoting the
proliferation and migration of tumour vascular endothelial cells
(22). A previous study
demonstrated that higher levels of VEGF were associated with a
greater probability of micrometastasis in liver cancer (26). VEGF activation leads to the
activation of a series of signal transduction proteins, including
extracellular signal-regulated kinase, protein kinase B and
mechanistic target of rapamycin (mTOR) (27–29).
These signal transduction proteins may promote the growth,
migration, differentiation and proliferation of vascular
endothelial cells. Previous studies have suggested that TAMs are
involved in processes of a variety of different types of cancers
(30). Thus, cancer progression
may be suppressed by inhibiting M2-like TAMs (31). Previous epidemiological studies
have indicated an association between increased macrophage
infiltration and poor prognoses of ovarian, cervical, thyroid, lung
and hepatocellular cancers (32–34).
In addition, an increasing evidence suggests that M2-like TAMs have
an important role in cancer progression and metastasis,
establishing TAMs, M2-like TAMs in particular, as an appealing
target for therapeutic interventions. Therefore, specifically
affecting M2-like TAMs and reducing macrophage infiltration may
effectively interfere with tumour progression. A previous study
demonstrated that luteolin promotes apoptosis and inhibits cancer
cell metastasis, thereby interfering with the progression of cancer
(35). In the present study, mouse
macrophages were used to investigate the potential anticancer
effect of luteolin in hypoxic environments.
Due to the lack of hypoxia incubators,
CoCl2 was used to mimic hypoxic conditions in the
experiment design. A low-oxygen environment was simulated with
CoCl2 treatment and the results revealed that in the
presence of CoCl2, the expression levels of VEGF and
HIF-1α were increased. The results of the present study indicated
that the treatment with CoCl2 successfully simulated low
oxygen conditions. VEGF may promote the migration and invasion of
tumour cells and the formation of tumour blood vessels, resulting
in an increase in the degree of tumour malignancy (36). The results of western blotting,
RT-qPCR and immunofluorescence experiments revealed that the
expression of VEGF was increased in M2-like TAMs; however, the
expression of VEGF was significantly inhibited following treatment
with luteolin, particularly within the M2-like TAMs. VEGF has an
important role in tumour angiogenesis, vascular permeability,
tumour stem cell function and the occurrence and development of
tumours (18). The finding that
luteolin reduces the expression of VEGF of the present study,
indicated that luteolin may have an anti-angiogenic role and
inhibits tumour progression under hypoxic conditions.
In the formation of blood vessels, the first step is
to degrade the extracellular matrix and basement membrane,
conducted by MMPs, which belong to a family of endopeptidases
(37). Degradation of the vascular
basement membrane is an indispensable step for the invasion of
endothelial cells. An increase in MMPs is associated with tumour
invasion, metastasis and angiogenesis (38). Among the proteolytic enzymes, MMP9
has been reported both in vivo and in vitro, to have
an important role in angiogenesis. In the present study, western
blotting and RT-qPCR confirmed the increased expression of MMP9
within M2-like TAMs, which is conducive to tumour angiogenesis.
However, following treatment with luteolin, the expression of MMP9
was significantly decreased; the expression of its inhibitor,
TIMP1, was increased. This result suggested that the
anti-angiogenic effect of luteolin may be achieved via the
inhibition of VEGF and MMP9 expression.
The hypoxic environment simulated by
CoCl2 significantly promoted the expression of HIF-1α,
which was confirmed by western blotting. The protein expression
level of HIF-1α was increased in hypoxic conditions, mimicked by
CoCl2, but no alterations in its mRNA expression level
was observed (39). A previous
study reported that luteolin significantly inhibited the expression
of ubiquitin E2S ligase, which regulates the expression of HIF-1α
(40). Another study demonstrated
that luteolin may inhibit the activation of HIF-1, which may
contribute to the inhibition of the mitogen-activated protein
kinase pathway (41). In addition,
the other major regulator in hypoxia is mTOR, which promotes HIF-1α
protein activation when hyperactivated (42,43).
A previous study indicated that luteolin may inhibit the activation
of Mtor (44). Therefore, the
negative effect of luteolin and HIF-1α may contribute to the
hyperactivation of mTOR. HIF-1α, a key regulator of hypoxia,
initiates the gene expression of associated factors that contribute
to angiogenesis, cell survival, invasion and migration (45). Research on effective HIF-1α
inhibitors has gained attention. It has been reported that HIF-1α
inhibitors may have potential novel anticancer drugs (46,47).
HIF-1α may regulate the expression of VEGF and MMP9 and thereby
contribute to the occurrence and development of tumours (48). However, the
CoCl2-induced increase in HIF-1α expression was
inhibited by luteolin in the present study. The ability of luteolin
to inhibit VEGF and MMP9 expression may be achieved via the
regulation of HIF-1α. Therefore, luteolin may exert its
anti-angiogenic effects via the HIF-1α-VEGF/MMP9 signalling
pathway.
The STAT3 protein in the cytoplasm of normal cells
has an important role in regulating cell growth and
differentiation. STAT3 appears to be a nexus for numerous oncogenic
signalling pathways. The STAT3 gene, located at 17q21.2, is
considered a proto-oncogene, and p-STAT3 enters the nucleus and
directly binds to DNA, thereby inducing downstream gene expression.
Recently, many studies have shown that activated STAT3 upregulates
the expression of VEGF and induces tumour angiogenesis (26,36).
Furthermore, previous studies have described STAT3 as a potential
modulator of HIF-1α-induced VEGF signalling in cancer cells
(39,49). P-STAT3 is additional promoter of
angiogenesis (50,51). The activation of p-STAT3 was
significantly inhibited by luteolin in M2-like TAMs. The STAT3
signalling pathway may be another mechanism luteolin uses to
contribute to angiogenesis. The expression of HIF-1α and VEGF was
reduced concomitantly with the expression of p-STAT3 in the present
study. This suggested that the STAT3 signalling pathway may also be
involved with the expression of HIF-1α, VEGF and MMP9 under hypoxic
conditions. In addition, the reduced expression of VEGF and MMP9
due to luteolin may be the result of the combined action of HIF-1α
and MMP9. Under hypoxic conditions, luteolin may have an
anti-angiogenic role, which may be achieved via the HIF-1α and
STAT3 signalling pathways; however, the association between STAT3
and HIF-1α requires further investigation (39).
In summary, luteolin may inhibit the abilities of
M2-like TAMs, which are induced by IL-4, to induce angiogenesis,
thereby inhibiting tumour growth. In combination with previous
research, luteolin may have a potent anticancer role, either under
normoxic or hypoxic conditions. The use of luteolin may be a novel
therapeutic strategy for targeting tumour invasion, migration,
apoptosis and vessel generation. In conclusion, the development of
novel luteolin-based drugs may be a new direction for anticancer
research.
Acknowledgements
Not applicable.
Funding
The present study was sponsored by the National
Health and Family Planning Commission of Zhejiang Province (grant
nos. 2013ZDA014, 2014ZB073), National Natural Science of China
(grant no. 81572087), Natural Science Foundation of Zhejiang
Province (grant nos. LY2H05004, LY16H030013), and the Wenzhou
Municipal Science and Bureau (grant nos. Y20140672, Y20150062).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
BF, XC, MW, HK, GC, ZZ and CZ performed the
experiments. BF and XC wrote the manuscript. BC, BF and XC designed
the experiments and analysed all data.
Ethics approval and consent to
participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing
interests.
References
|
1
|
Hanahan D and Coussens LM: Accessories to
the crime: Functions of cells recruited to the tumor
microenvironment. Cancer Cell. 21:309–322. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dannenmann SR, Thielicke J, Stöckli M,
Matter C, von Boehmer L, Cecconi V, Hermanns T, Hefermehl L,
Schraml P, Moch H, et al: Tumor-associated macrophages subvert
T-cell function and correlate with reduced survival in clear cell
renal cell carcinoma. Oncoimmunology. 2:e235622013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Murray PJ and Wynn TA: Obstacles and
opportunities for understanding macrophage polarization. J Leukoc
Biol. 89:557–563. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wynn TA, Chawla A and Pollard JW:
Macrophage biology in development, homeostasis and disease. Nature.
496:445–455. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mantovani A, Romero P, Palucka AK and
Marincola FM: Tumour immunity: Effector response to tumour and role
of the microenvironment. Lancet. 371:771–783. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Murray PJ, Llen JE, Biswas SK, Fisher EA,
Gilroy DW, Goerdt S, Gordon S, Hamilton JA, Ivashkiv LB, Lawrence
T, et al: Macrophage activation and polarization: Nomenclature and
experimental guidelines. Immunity. 41:14–20. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pickert G, Lim HY, Weigert A, Häussler A,
Myrczek T, Waldner M, Labocha S, Ferreirós N, Geisslinger G, Lötsch
J, et al: Inhibition of GTP cyclohydrolase attenuates tumor growth
by reducing angiogenesis and M2-like polarization of tumor
associated macrophages. Int J Cancer. 132:591–604. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cardoso AP, Pinto ML, Pinto AT, Oliveira
MI, Pinto MT, Gonçalves R, Relvas JB, Figueiredo C, Seruca R,
Mantovani A, et al: Macrophages stimulate gastric and colorectal
cancer invasion through EGFR Y(1086), c-Src, Erk1/2 and Akt
phosphorylation and smallGTPase activity. Oncogene. 33:2123–2133.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen W, Ma T, Shen XN, Xia XF, Xu GD, Bai
XL and Liang TB: Macrophage-induced tumor angiogenesis is regulated
by the TSC2-mTOR pathway. Cancer Res. 72:1363–1372. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bellon G, Martiny L and Robinet A: Matrix
metalloproteinases and matrikines in angiogenesis. Crit Rev Oncol
Hematol. 49:203–220. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li SH, Shin DH, Chun YS, Lee MK, Kim MS
and Park JW: A novel mode of action of YC-1 in HIF inhibition:
Stimulation of FIH-dependent p300 dissociation from HIF-1{alpha}.
Mol Cancer Ther. 7:3729–3738. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hasanain M, Bhattacharjee A, Pandey P,
Ashraf R, Singh N, Sharma S, Vishwakarma AL, Datta D, Mitra K and
Sarkar J: α-Solanine induces ROS-mediated autophagy through
activation of endoplasmic reticulum stress and inhibition of
Akt/mTOR pathway. Cell Death Dis. 6:e18602015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Huang X, Dai S, Dai J, Xiao Y, Bai Y, Chen
B and Zhou M: Luteolin decreases invasiveness, deactivates STAT3
signaling, and reverses interleukin-6 induced
epithelial-mesenchymal transition and matrix metalloproteinase
secretion of pancreatic cancer cells. OncoTargets Ther.
8:2989–3001. 2015. View Article : Google Scholar
|
|
14
|
Schulz K and Dabels J: Glucose-6-phosphate
dehydrogenase of blood platelets in diseases of the thrombopoietic
system. Folia haematol Int Mag Clin Morphol Blutforsch. 94:223–229.
1970.(In German).
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xiao B, Lin D and Zhang X, Zhang M and
Zhang X: TTF1, in the form of nanoparticles, inhibits angiogenesis,
cell migration and cell invasion in vitro and in vivo in human
hepatoma through STAT3 regulation. Molecules. 21:E15072016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Song H, Pan D, Sun W, Gu C, Zhang Y, Zhao
P, Qi Z and Zhao S: SiRNA directed against annexin II receptor
inhibits angiogenesis via suppressing MMP2 and MMP9 expression.
Cell Physiol Biochem. 35:875–884. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mahecha AM and Wang H: The influence of
vascular endothelial growth factor-A and matrix metalloproteinase-2
and −9 in angiogenesis, metastasis, and prognosis of endometrial
cancer. OncoTargets Ther. 10:4617–4624. 2017. View Article : Google Scholar
|
|
19
|
Yao ZG, Li WH, Hua F, Cheng HX, Zhao MQ,
Sun XC, Qin YJ and Li JM: LBH589 inhibits glioblastoma growth and
angiogenesis through suppression of HIF-1α expression. J
Neuropathol Exp Neurol. 76:1000–1007. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
John A and Tuszynski G: The role of matrix
metalloproteinases in tumor angiogenesis and tumor metastasis.
Pathol Oncol Res. 7:14–23. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hegde PS, Wallin JJ and Mancao C:
Predictive markers of anti-VEGF and emerging role of angiogenesis
inhibitors as immunotherapeutics. Semin Cancer Biol. Dec
8–2017.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Olsen JJ, Pohl SÖ, Deshmukh A, Visweswaran
M, Ward NC, Arfuso F, Agostino M and Dharmarajan A: The Role of wnt
signalling in angiogenesis. Clin Biochem Rev. 38:131–142.
2017.PubMed/NCBI
|
|
23
|
Yang JG, Wang LL and Ma DC: Effects of
vascular endothelial growth factors and their receptors on
megakaryocytes and platelets and related diseases. Br J Haematol.
180:321–334. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kong DH, Kim MR, Jang JH, Na HJ and Lee S:
A review of anti-angiogenic targets for monoclonal antibody cancer
therapy. Int J Mol Sci. 18:pii: E1786. 2017.
|
|
25
|
Dahlof B, Hansson L, Lindholm L, Rastam L,
Schersten B and Wester PO: STOP-hypertension: Swedish trial in old
patients with hypertension. J Hypertens. 4:511–513. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lee M, Choi JY, Lim JS, Park MS, Kim MJ
and Kim H: Lack of anti-tumor activity by anti-VEGF treatments in
hepatic hemangiomas. Angiogenesis. 19:147–153. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zachary I and Gliki G: Signaling
transduction mechanisms mediating biological actions of the
vascular endothelial growth factor family. Cardiovasc Res.
49:568–581. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cho CH, Lee CS, Chang M, Jang IH, Kim SJ,
Hwang I, Ryu SH, Lee CO and Koh GY: Localization of VEGFR-2 and
PLD2 in endothelial caveolae is involved in VEGF-induced
phosphorylation of MEK and ERK. Am J Physiol Heart Circ Physiol.
286:H1881–1888. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kim BW, Choi M, Kim YS, Park H, Lee HR,
Yun CO, Kim EJ, Choi JS, Kim S, Rhim H, et al: Vascular endothelial
growth factor (VEGF) signaling regulates hippocampal neurons by
elevation of intracellular calcium and activation of
calcium/calmodulin protein kinase II and mammalian target of
rapamycin. Cell Signal. 20:714–725. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qian BZ and Pollard JW: Macrophage
diversity enhances tumor progression and metastasis. Cell.
141:39–51. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Attri KS, Mehla K and Singh PK: Evaluation
of macrophage polarization in pancreatic cancer microenvironment
under hypoxia. Methods Mol Biol. 1742:265–276. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ryder M, Ghossein RA, Ricarte-Filho JC,
Knauf JA and Fagin JA: Increased density of tumor-associated
macrophages is associated with decreased survival in advanced
thyroid cancer. Endocr Relat Cancer. 15:1069–1074. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gazzaniga S, Bravo AI, Guglielmotti A, van
Rooijen N, Maschi F, Vecchi A, Mantovani A, Mordoh J and Wainstok
R: Targeting tumor-associated macrophages and inhibition of MCP-1
reduce angiogenesis and tumor growth in a human melanoma xenograft.
J Invest Dermatol. 127:2031–2041. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jia JB, Wang WQ, Sun HC, Zhu XD, Liu L,
Zhuang PY, Zhang JB, Zhang W, Xu HX, Kong LQ, et al: High
expression of macrophage colony-stimulating factor-1 receptor in
peritumoral liver tissue is associated with poor outcome in
hepatocellular carcinoma after curative resection. Oncologist.
15:732–743. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lin Y, Shi R, Wang X and Shen HM:
Luteolin, a flavonoid with potential for cancer prevention and
therapy. Curr Cancer Drug Targets. 8:634–646. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shih T and Lindley C: Bevacizumab: An
angiogenesis inhibitor for the treatment of solid malignancies.
Clin Ther. 28:1779–1802. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Browne S and Pandit A: Engineered systems
for therapeutic angiogenesis. Curr Opin Pharmacol. 36:34–43. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Padwal M, Siddique I, Wu L, Tang K, Boivin
F, Liu L, Robertson J, Bridgewater D, West-Mays J, Gangji A, et al:
Matrix metalloproteinase 9 is associated with peritoneal membrane
solute transport and induces angiogenesis through β-catenin
signaling. Nephrol Dial Transplant. 32:50–61. 2017.PubMed/NCBI
|
|
39
|
Wen Z, Huang C, Xu Y, Xiao Y, Tang L, Dai
J, Sun H, Chen B and Zhou M: α-Solanine inhibits vascular
endothelial growth factor expression by down-regulating the
ERK1/2-HIF-1α and STAT3 signaling pathways. Eur J Pharmacol.
771:93–98. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lin TH, Hsu WH, Tsai PH, Huang YT, Lin CW,
Chen KC, Tsai IH, Kandaswami CC, Huang CJ, Chang GD, et al: Dietary
flavonoids, luteolin and quercetin, inhibit invasion of cervical
cancer by reduction of UBE2S through epithelial-mesenchymal
transition signaling. Food Funct. 8:1558–1568. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Triantafyllou A, Mylonis I, Simos G,
Bonanou S and Tsakalof A: Flavonoids induce HIF-1alpha but impair
its nuclear accumulation and activity. Free Radic Biol Med.
44:657–670. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Nguyen A, Moussallieh FM, Mackay A, Cicek
AE, Coca A, Chenard MP, Weingertner N, Lhermitte B, Letouzé E,
Guérin E, et al: Characterization of the transcriptional and
metabolic responses of pediatric high grade gliomas to mTOR-HIF-1α
axis inhibition. Oncotarget. 8:71597–71617. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang Q, Wang H, Jia Y, Ding H, Zhang L and
Pan H: Luteolin reduces migration of human glioblastoma cell lines
via inhibition of the p-IGF-1R/PI3K/AKT/mTOR signaling pathway.
Oncol Lett. 14:3545–3551. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu Y, Huang J, Zheng X, Yang X, Ding Y,
Fang T, Zhang Y, Wang S, Zhang X, Luo X, et al: Luteolin, a natural
flavonoid, inhibits methylglyoxal induced apoptosis via the
mTOR/4E-BP1 signaling pathway. Sci Rep. 7:78772017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wouters BG and Koritzinsky M: Hypoxia
signalling through mTOR and the unfolded protein response in
cancer. Nat Rev Cancer. 8:851–864. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Masoud GN and Li W: HIF-1α pathway: Role,
regulation and intervention for cancer therapy. Acta Pharm Sin B.
5:378–389. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Hsu CW, Huang R, Khuc T, Shou D, Bullock
J, Grooby S, Griffin S, Zou C, Little A, Astley H and Xia M:
Identification of approved and investigational drugs that inhibit
hypoxia-inducible factor-1 signaling. Oncotarget. 7:8172–8183.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhang Z, Amorosa LF, Coyle SM, Macor MA,
Lubitz SE, Carson JL, Birnbaum MJ, Lee LY and Haimovich B:
Proteolytic cleavage of AMPKα and intracellular MMP9 expression are
both required for TLR4-mediated mTORC1 activation and HIF-1α
expression in leukocytes. J Immunol. 195:2452–2460. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang K, Han ES, Dellinger TH, Lu J, Nam
S, Anderson RA, Yim JH and Wen W: Cinnamon extract reduces VEGF
expression via suppressing HIF-1α gene expression and inhibits
tumor growth in mice. Mol Carcinog. 56:436–446. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Reiter P: A field trial of expanded
polystyrene balls for the control of Culex mosquitoes breeding in
pit latrines. J Am Mosq Control Assoc. 1:519–521. 1985.PubMed/NCBI
|
|
51
|
Chen YC, Chien LH, Huang BM, Chia YC and
Chiu HF: Aqueous extracts of toona sinensis leaves inhibit renal
carcinoma cell growth and migration through JAK2/stat3, Akt,
MEK/ERK, and mTOR/HIF-2α pathways. Nutr Cancer. 68:654–666. 2016.
View Article : Google Scholar : PubMed/NCBI
|