Introduction
Idiopathic pulmonary fibrosis (IPF) is a type of
interstitial lung disease (ILD) (1). This chronic and progressively fatal
disease is characterized by the formation of scar tissue deep in
the lung, causing the thickening of the pleural lining and leading
to the irreversible loss of pulmonary function (2,3). It
usually affects adults between 50 and 70 years of age, with a
higher incidence in men than in women. The incidence rate of IPF is
five times higher than that of cystic fibrosis or Lou Gehrig's
disease (also termed amyotrophic lateral sclerosis), with a poor
prognosis, a median survival time of 3 years and fatality rate of
2/3 in 5 years (4,5); however this disease remains largely
unknown to the general public. Although other ILDs may be
attributed to the exposure to asbestos or certain medications, IPF
has not been linked to any known pathogenic factors. Potential risk
factors for IPF may include smoking, and exposure to wood or metal
dust (6,7).
MicroRNAs (miRNAs/miRs) have been proposed to have
fundamental roles in the pathogenesis of several pulmonary
diseases, including interstitial lung disease, chronic obstructive
pulmonary disease and IPF (8).
miRs are small noncoding RNAs that subtly regulate gene expression
by inhibiting mRNA translation or mediating mRNA degradation.
Exosomes are present in the majority of bodily fluids, and their
composition differs depending on their cellular origins (8). Studies have demonstrated that miRs
encapsulated in exosomes in certain bodily fluids, including saliva
(9) and serum (10), can be used as biomarkers for
disease screening and prognosis purposes (11).
Potential alterations of exosome miR contents caused
by IPF have not yet been investigated. In the present study, the
differences between bronchoalveolar lavage fluid (BALF) exosomal
miRs from patients with IPF and healthy participants were
characterized. Exosomes were collected from patients with IPF or
healthy participants by bronchoalveolar lavage prior to exosomal
miR extraction. Quantitative analysis of miR expression profiles
revealed that miR-125b-5p, miR-128-3p, miR-21-5p, miR-100-5p,
miR-140-3p and miR-374b-5p were upregulated by more than 2-fold in
patients with IPF compared with healthy participants; while
let-7d-5p, miR-103-3p, miR-27b-3p and miR-30a-5p were
downregulated. The expression level of miR-30a-5p was further
examined by reverse transcription-quantitative polymerase chain
reaction (RT-qPCR). Target gene prediction analysis of miR-30a-5p
indicated that it may directly interact with TGF-β activated kinase
1/MAP3K7 binding protein 3 (TAB3) mRNA. Further examination using a
dual-luciferase reporter assay and western blotting analysis
confirmed the association between miR-30a-5p and TAB3 mRNA.
Functional assays demonstrated that miR-30a-5p overexpression
attenuated transforming growth factor-β1 (TGF-β1)-induced
upregulation of TAB3, α-SMA and fibronectin expression in 293T
cells. The decreased expression of miR-30a-5p in patients with IPF,
along with the consequential increase in TAB3 expression may be a
key factor in IPF progression.
Materials and methods
Patients and clinical specimens
The present study was approved by the institutional
review board of People's Hospital of Zhengzhou University
(Zhengzhou, China). A total of 30 patients (2016.10–2017.09)
diagnosed with IPF were selected as the IPF group based on their
positive IPF diagnosis prior to enrollment. A total of 16 healthy
participants were selected as a control group in the same facility
(People's Hospital of Zhengzhou University) based on their negative
diagnosis results in routine blood test, chest X-ray, chest and
abdominal ultrasound examination. All patients and healthy
participants were diagnosed as free from structural heart disease
or brain, liver, kidney and endocrine associated metabolic
diseases. Informed consent was obtained from each participant in
written form prior to undergoing BALF collection, following the
Chinese medical association alveolar lavage operation instructions
(12). Clinical data of patients
with IPF and healthy participants is summarized in Table I.
 | Table I.Clinical data of patients with IPF
and healthy participants. |
Table I.
Clinical data of patients with IPF
and healthy participants.
|
| IPF (n=30) | Healthy (n=16) | P-value |
|---|
| Age
(year)a |
64.38±9.88 |
66.90±7.48 | 0.18 |
| Gender
(male/female) | 27/3 | 12/4 | 0.54 |
| Smoking
(current/ever/never) | 0/21/9 | 0/13/3 | 0.19 |
| Height
(cm)a | 167.30±0.75 | 153.40±0.23 | 0.36 |
| Weight
(kg)a |
62.75±6.74 |
58.77±8.66 | 0.66 |
| BMI
(kg/m2)a |
11.95±1.61 |
19.12±1.79 | 0.45 |
| FVC (%
predicted)a |
59.80±15.30 | N/A | N/A |
| TLC (%
predicted)a |
62.40±7.90 | N/A | N/A |
| DLCO [Hb] (%
predicted)a |
44.60±21.10 | N/A | N/A |
Exosome collection and miR
extraction
Exosomes in the BALF obtained from each subject were
isolated using 100 ml BALF by ExoQuick-TC™ exosome precipitation
(System Biosciences, LLC, Palo Alto, CA, USA) following the
manufacturer's protocol. The isolated exosomes were resuspended in
lysis buffer provided in NucleoSpin miRNA extraction kit
(Macherey-Nagel GmbH & Co., KG, Düren, Germany) and stored at
−80°C for no longer than 24 h prior to miR extraction. Total miRs
from each subject's exosome sample were extracted using NucleoSpin
miRNA extraction kit (Macherey-Nagel GmbH & Co., KG) following
the manufacturer's protocol. A 100 µl exosome sample from each
group prior to and following precipitation, as well as supernatant
solution from precipitation, were reserved for cluster of
differentiation 63 (CD63) detection by western blotting.
Exosomal miR profiling and miR-30a-5p
expression level
Half of the total miRs extracted from each BALF
sample were subject to miR expression profile analysis using
miProfile™ human miRNome miRNA qPCR Array kit (Genecopoeia, Inc.,
Rockville, MD, USA) following the manufacturer's protocol. Each
96-well plate included 12 wells that contained different types of
controls for monitoring the performance of the entire experimental
process. Particularly, the wells include two negative controls, six
different housekeeping controls for normalization (snRNAs), two RT
and two PCR controls. The expression level of each detected miR was
first normalized to small nuclear (sn)RNA U6 using the
2−ΔΔCq method (13),
and the fold-change (|log2n|>1) of
normalized miR expression is calculated by comparing the expression
level of each detected miR in IPF exosome lysates to that in
exosome lysates from healthy participants. The other half of
extracted miRs from each sample was subjected to evaluation of the
miR-30a-5p expression level using a custom-built hsa-miR-30a-5p
RT-qPCR assay kit (Genecopoeia, Inc.) following the manufacturer's
protocol. miR-30a-5p expression level of each sample was first
normalized to snRNA U6 using the 2−ΔΔCq method, and the
expressional difference was revealed by calculating the fold-change
of the mean value of miR-30a expression level in exosome lysates
from healthy participants compared with that of patients with IPF.
The receiver operating characteristic curve of miR-30a-5p
expression in BALF exosomes of IPF patients was analyzed with
reference to the expression level of miR-30a-5p in healthy
participants.
Cell culture and preparation
A549 and 293T cells previously purchased from the
American Type Culture Collection, (Manassas, VA, USA) and stored in
liquid nitrogen were resuscitated and cultured in RPMI-1640 medium
supplemented with 10% fetal bovine serum (HyClone; GE Healthcare,
Life Sciences, Logan, UT, USA) and penicillin-streptomycin cocktail
at 37°C in a humidified incubator with 5% CO2. Cells
were transfected with miR-30a-5p mimic
(5′-UGUAAACAUCCUCGACUGGAAG-3′, 250 µg/ml) or non-targeting miR
mimic (mimic control, 5′-UCACAACCUCCUAGAAAGAGUAGA-3′) vector
(Genecopoeia, Inc.) using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) for
12 h according to the manufacturer's protocol prior to subsequent
assays. Recombinant human TGF-β1 cytokine (5 ng/ml for 24 h) for
cell treatment was purchased from Invitrogen (Thermo Fisher
Scientific, Inc.) and applied according to the manufacturer's
protocol.
Dual-luciferase reporter assay
For the firefly luciferase reporter assay, firefly
luciferase vector carrying wild type (WT) or mutant (MT) human TAB3
3′untranslated region (UTR) were purchased from Genecopoeia, Inc.
Briefly, the firefly luciferase reporter vectors were constructed
by inserting the amplified WT or MT TAB3 3′UTR cDNA sequence
downstream of a secreted firefly luciferase gene in pEZX-MT01
vector (Genecopoeia, Inc.). 293T cells were transfected with 2
nmol/ml either miR-30a-5p mimic vector, mimic control vector or
empty vehicle as blank control, in combination with 2.5 µg/ml of
both the reporter vector and Renilla luciferase control
vector pRL-TK (Genecopoeia, Inc.) using Lipofectamine®
2000 (Invitrogen; Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. Luciferase reporter assay was performed 24
h following transfection. Firefly and Renilla luciferase
activities were measured consecutively by GLO-MAX 20/20
fluorescence detector device (Promega Corporation) using Secrete
Pair™ Dual Luminescence assay kit (Lipofectamine® 2000)
following the manufacturers' protocol. The firefly luciferase
activity was normalized to Renilla luciferase activity.
RT-qPCR and western blotting
assay
For evaluating the miR-30a-5p expression level and
TAB3, α-SMA and fibronectin mRNA expression level in A549 cells
with different treatments, custom-built RT-qPCR kits (Genecopoeia,
Inc.) were used following manufacturer's protocol. The sequences of
miR-30a-5p primer: 5′-TTCAGTCGGATGTTTGCAGCAAA-3′. Detection was
achieved by SYBR green qPCR with the following conditions: 95°C for
10 min followed by 40 cycles of 95°C for 10 sec, 60°C for 20 sec
and 72°C for 20 sec. The miRNA expression levels were normalized to
levels of U6 snRNA (5′-AAATTCGTGAAGCGTTCC-3′) using the
2−ΔΔCq method (13).
For detecting exosomal CD63, the same volume of 30 µl each exosome
lysate sample or supernatant sample was used. For detecting
cellular TAB3 and β-actin protein expression level in A549 and 293T
cells 24 h after transfection, cells cultured in 12-well-plates
were lysed with pre-chilled NP-40 lysis buffer (200 µl per well;
Thermo Fisher Scientific, Inc.) by pipetting on ice, and a same
volume of 30 µl each cell lysate sample was used following
centrifugation at 10,000 × g for 3 min at 4°C to remove cell
debris. Protein concentrations were determined using a BCA assay
kit (Thermo Fisher Scientific, Inc.). The total protein of each
cell group (20 µl) was separated using polyacrylamide gel (15%)
electrophoresis under reducing conditions, followed by transferral
onto nitrocellulose membranes. Following blocking at 4°C overnight
with non-fat milk and preparation with PBS. The membranes were
probed with antibodies, incubated 16 h at 4°C. And then incubated
with horseradish peroxidase conjugated secondary antibodies,
incubated 1 h at room temperature, and colorized using the Pierce
ECL Plus substrate (Pierce; Thermo Fisher Scientific, Inc.). The
image of each sample was captured using
chemo-fluorescence-sensitive film and the gray value of each band
was analyzed. The quantity of target protein was evaluated by the
ratio of its gray value to the internal reference using ImageJ
software, version 1.48 (National Institutes of Health, Bethesda,
MD, USA). Antibodies against human CD63 (cat. no. ab134045;
1:1,000), TAB3 (cat. no. ab124723; 1:2,000), β-actin (cat. no.
ab8227; 1:1,000) and secondary antibody goat anti-rabbit IgG
H&L (cat. no. ab6721; 1:5,000) were purchased from Abcam
(Cambridge, UK). Antibodies against human CD63, TAB3 and β-actin
incubated 16 h at 4°C. The secondary antibody incubated 1 h at room
temperature.
Data visualization and statistical
analysis
Data were visualized and statistically analyzed
using GraphPad Prism 7 (GraphPad Software, Inc., La Jolla, CA, USA)
and presented as the mean ± standard deviation, unless otherwise
indicated. An unpaired parametric t-test was used for analysis. All
P-values are two-tailed. P<0.05 was considered to indicate a
statistically significant difference. Online tools for miRNA
analysis are miRDB (http://www.mirdb.org/) online database and the STRING
online platform (https://string-db.org/cgi/input.pl, version 10.5). The
target genes of these increased miRs were predicted using FunRich
software (http://www.funrich.org/; ver. 3).
Results
Characteristics of patients with IPF
and healthy participants
The physiological and pathological data of 30
patients with IPF and 16 healthy participants involved in the
present study are summarized for comparison in Table I. Patients were diagnosed with IPF
by high-resolution computed tomography and other causes of
interstitial lung disease in medical history, including family
environment, occupational exposure, connective tissue disease and
drug-induced pulmonary lesion were excluded (14,15).
No statistically significant (P>0.05) difference in age, height,
weight or body mass index was detected between the two groups.
Alterations in BALF exosomal miRNA
expression patterns between IPF pathological and healthy
conditions
BALF exosomes from patients with IPF and healthy
participants were obtained by precipitation as described.
High-throughput quantitative analysis of miR expression using a
microarray was performed to reveal the differentially expressed
BALF exosomal miRs between patients with IPF and healthy
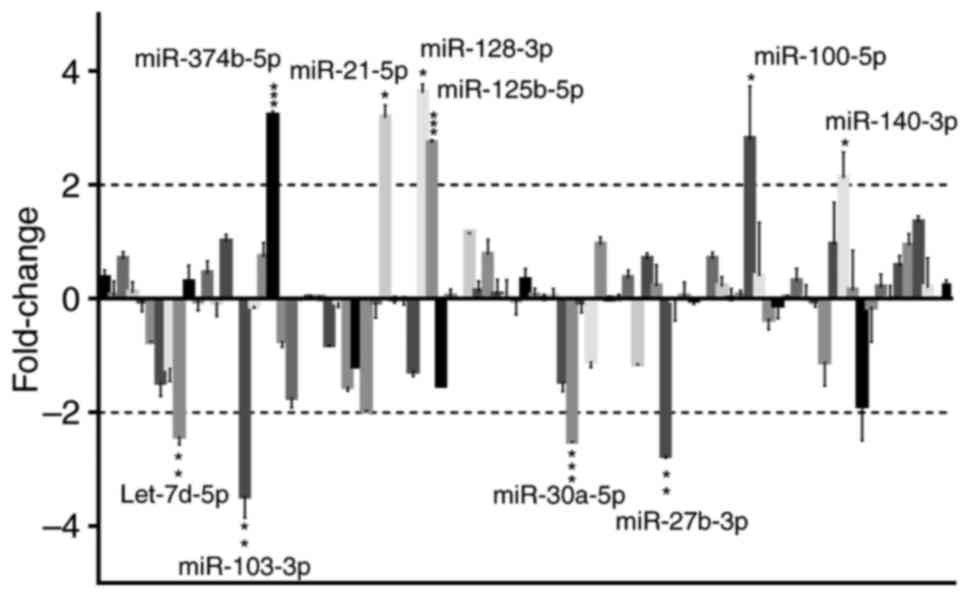
participants (Fig. 1). A total of
69 miRs exhibited significantly different expression levels in BALF
exosomes between the two groups. Of these 69 miRs there was a
greater than 2-fold increase in the expression level of
miR-125b-5p, miR-128-3p, miR-21-5p, miR-100-5p, miR-140-3p and
miR-374b-5p, and a greater than 2-fold decrease in let-7d,
miR-103-3p, miR-30a-5p and miR-27b-3p. The majority of miRs with
significant expression differences have been previously reported as
potential regulators and biomarkers in IPF progression or
IPF-associated carcinogenesis (16–23),
among which miR-30a-5p demonstrated the most significance and was
selected for further investigation in the present study
(P<0.001).
Decreases in exosomal miR-30a-5p
expression in patients with IPF compared with healthy
participants
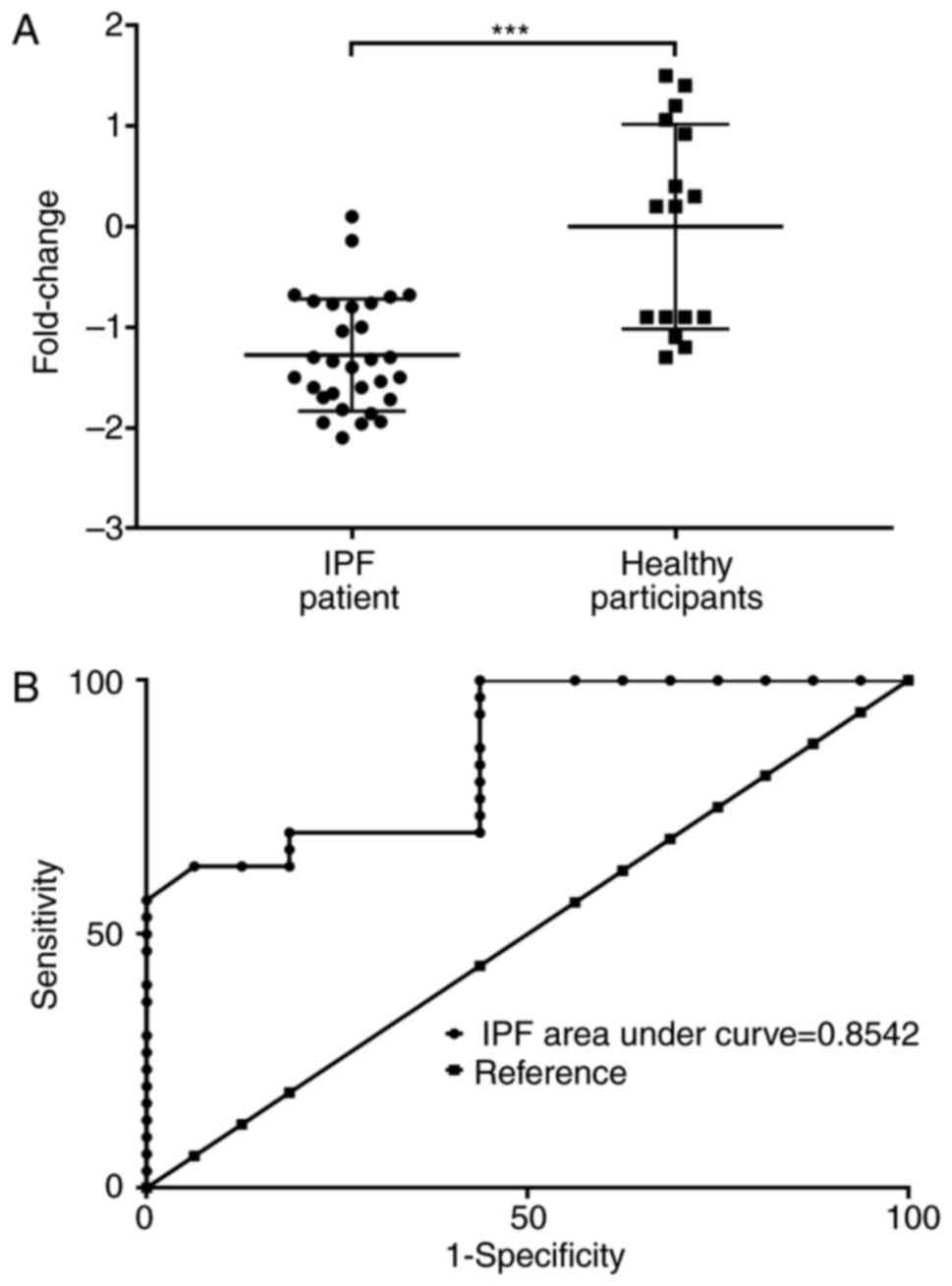
The miR-30a-5p expression level in BALF exosomes
obtained from patients with IPF and healthy participants was
validated by RT-qPCR (Fig. 2A).
miR-30a-5p expression was significantly decreased under IPF
pathological conditions, which is consistent with the result of the
miR profiling assay (P<0.001). These data suggested that
miR-30a-5p may be a potential regulator involved in IPF
pathogenesis, and the downregulation of miR-30a-5p may contribute
to IPF progression. The receiver operating characteristic curve
analysis based on miR-30a-5p expression level in two groups further
demonstrated that decreased miR-30a-5p expression may be a
biomarker for IPF (Fig. 2B).
TAB3 as direct regulatory target of
miR-30a-5p
To investigate the regulatory role of miR-30a-5p in
IPF, potential target mRNAs of miR-30a-5p were predicted using
miRDB online database. miRNA-30a-5p was predicted to interact with
842 genes, among which 111 genes were involved in intracellular
signal transduction, as revealed by functional enrichment analysis
using the STRING online platform. The potential target genes were
filtered further based on their association with TGF-β1 signaling
pathway, which serves vital role in fibrogenesis and IPF
progression via manual literature searching (24,25).
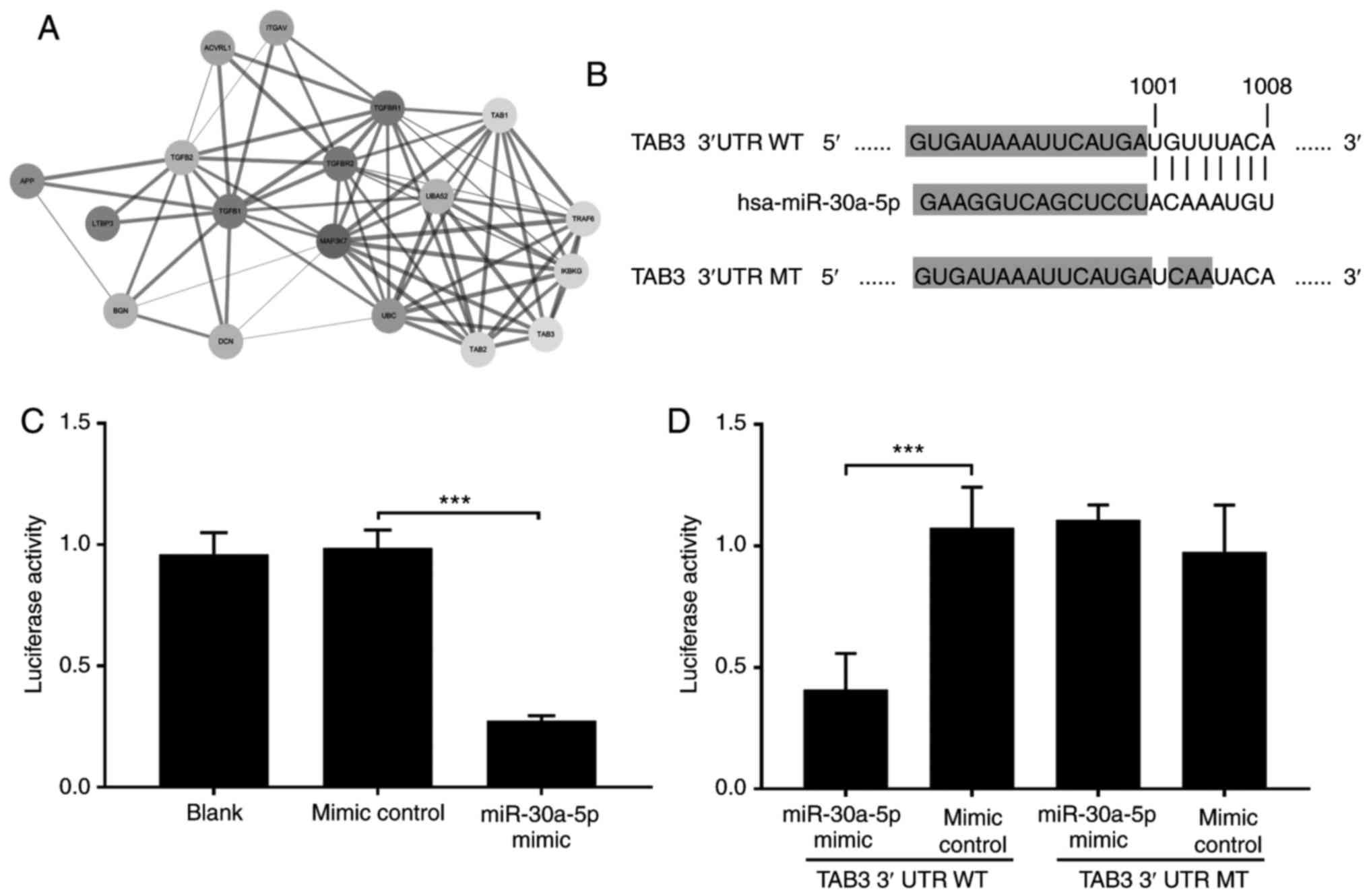
Protein interaction data mining using the STRING online platform
indicated that TAB3 may directly interact with TGF-β1 receptor and
mitogen-activated protein kinase 7 (Fig. 3A), which has been demonstrated to
serve an important role in fibrosis process in various tissues
(26,27). Sequence analysis of TAB3 mRNA and
miR-30a-5p also demonstrated that miR-30a-5p may directly interact
with the 3′UTR region in TAB3 mRNA (Fig. 3B). Based on these analysis results,
TAB3 was selected for further investigation.
A dual-luciferase reporter assay was performed to
confirm this interaction (Fig.
3C). Firefly luciferase activity in 293T cells co-transfected
with TAB3 3′UTR-flanked firefly luciferase vector and miR-30a-5p
mimic was significantly lower compared the control group
co-transfected with TAB3 3′UTR flanked firefly luciferase and
non-targeting mimic control (P<0.001), suggesting a direct
interaction between miR-30a-5p and the 3′UTR region of TAB3 mRNA.
This miR targeting mechanism was confirmed by inducing three
consecutive point mutations within the predicted miR-30a-5p binding
site on 3′UTR region of TAB3 mRNA (Fig. 3B and D). Firefly luciferase was
significantly decreased in 293T cells co-transfected with WT TAB3
3′UTR flanked firefly luciferase vector and miR-30a-5p mimic
compared with mimic control (P<0.001). Firefly luciferase was no
significant in TAB3 3′UTR MT miR-30a-5p mimic compared with mimic
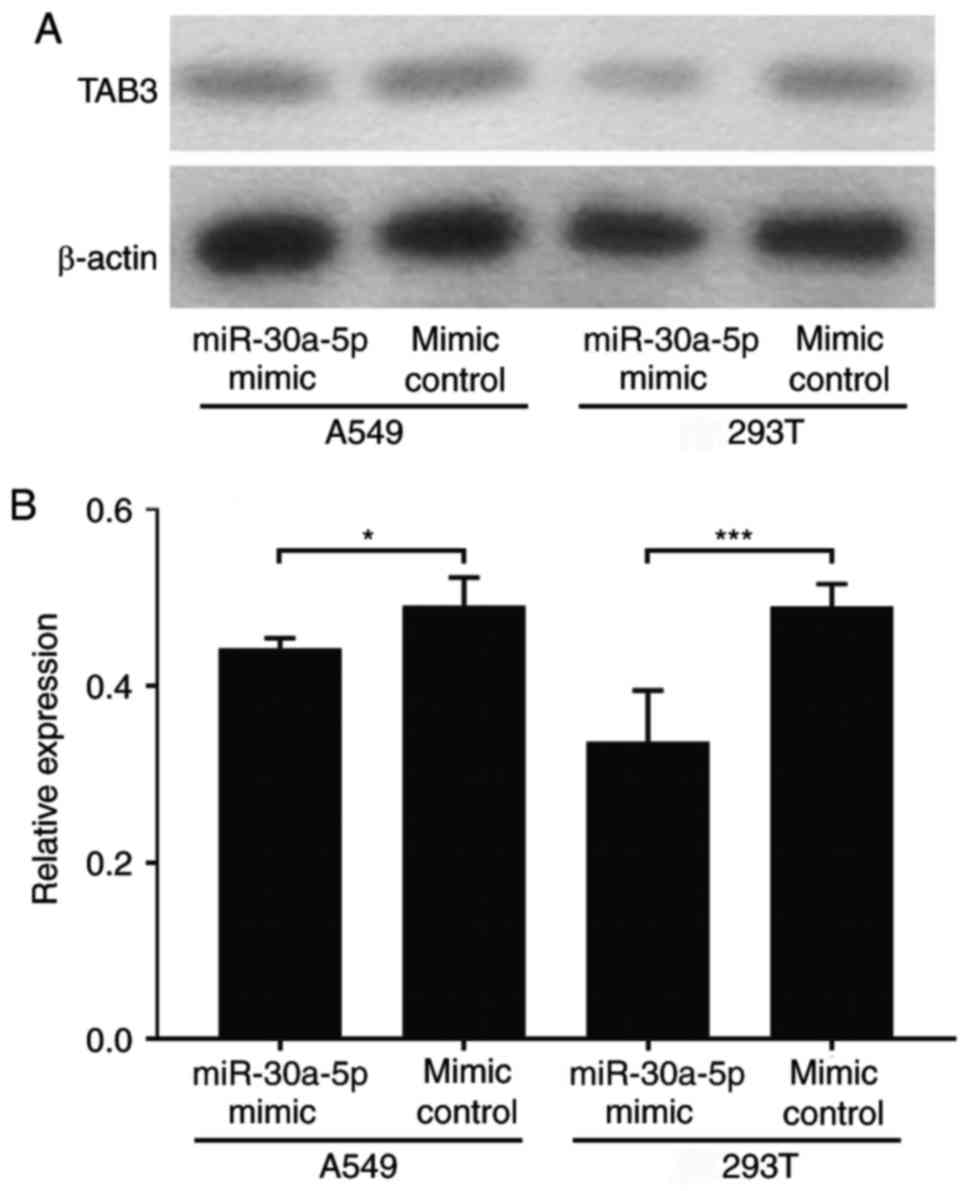
control. This mechanism was further verified by western blotting
analysis evaluating the expression level of TAB3 in A549 and 293T
cells with or without miR-30a-5p mimic transfection (Fig. 4). miR-30a-5p mimic transfection
significantly decreased the TAB3 protein expression level compared
with non-targeting mimic control transfection in A549 (P<0.05)
and 293T cells (P<0.001). Together, these data suggested that
TAB3 mRNA is a direct target of miR-30a-5p.
miR-30a-5p overexpression attenuates
TGF-β1 stimulated α-SMA and fibronectin expression
Considering the potential association between
miR-30a-5p, TAB3 and TGF-β signaling, a preliminary study was
performed on the impact of miR-30a-5p overexpression on the mRNA
expression level of TAB3, α-SMA and fibronectin in A549 cells, the
last two of which have been suggested biomarkers of TGF-β1 induced
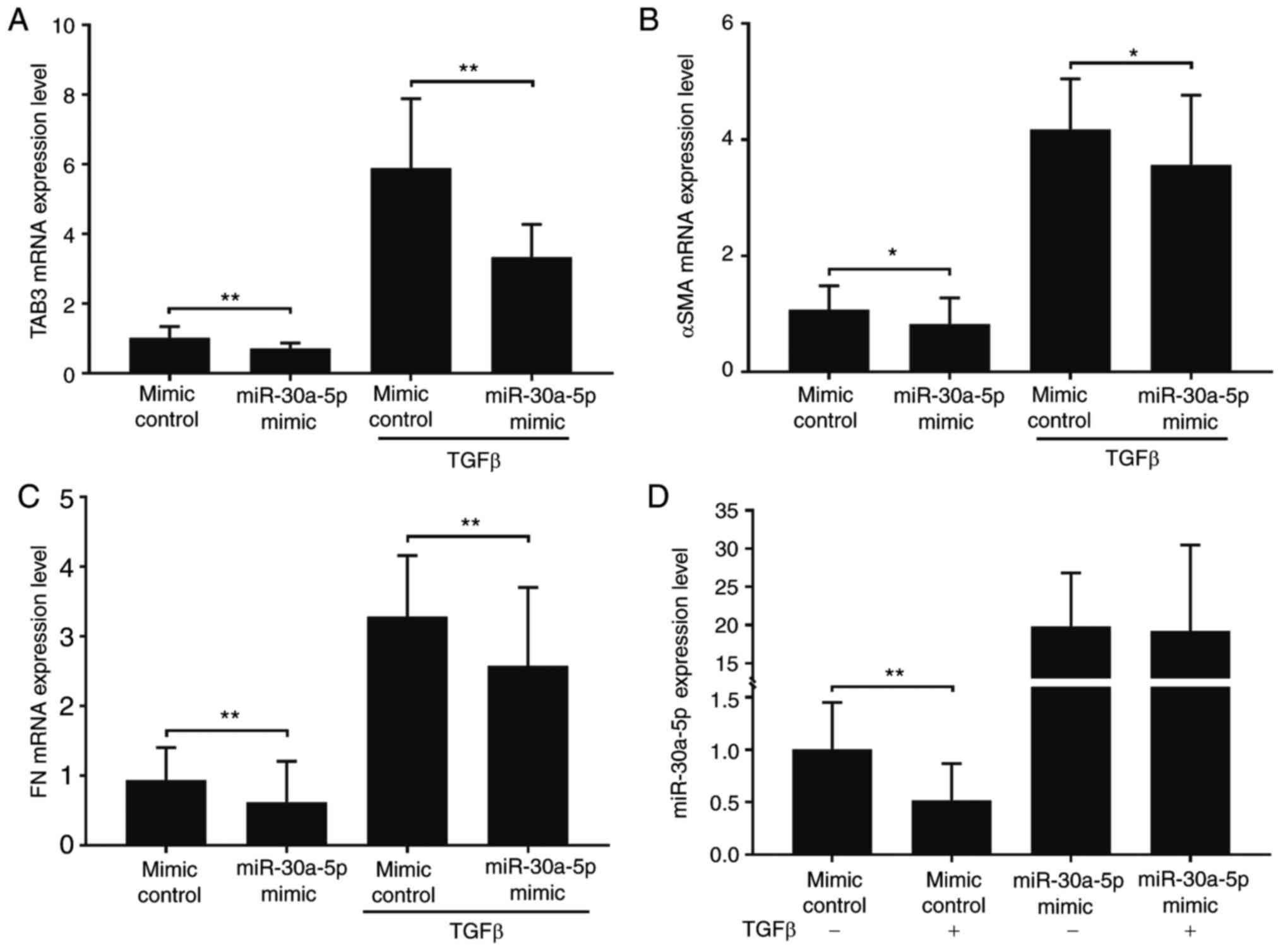
fibrogenesis in lung epithelial cells (Fig. 5) (28–30).
The results of the present study demonstrated that 5 ng/ml TGF-β1
treatment for 24 h increased TAB3, α-SMA and fibronectin
expression, which was significantly decreased by artificial
miR-30a-5p overexpression (P<0.05; Fig. 5A-C). Notably, it was demonstrated
that TGF-β1 treatment could also significantly decrease the
miR-30a-5p expression level in A549 cells with the mimic control
but not in the cells transfected with the miR-30a-5p mimic, which
implied that TGF-β1 signaling may inhibit miR-30a-5p expression in
an indirect fashion (Fig. 5D).
Discussion
miRs are small non-coding RNAs that normally serve
negative regulatory roles in different cellular events by inducing
post-transcriptional silencing of their target genes (31). The lungs of patients with are
characterized by phenotypic alterations of fibroblasts and
epithelial cells, due to alterations in gene expression.
The aim of the present study was to detect
differentially expressed exosomal miRs in BALF from patients with
IPF and healthy participants that may be involved in IPF
pathogenesis or progression. miRs were extracted from exosomes in
BALF from 30 patients with IPF and 16 healthy participants, and miR
profiling was subsequently performed. Microarray and
next-generation sequencing (NGS) are the two major high-throughput
approaches for genome and transcriptome analysis. The former was
chosen to analyze miR expression in the present study, as previous
experiences demonstrated that NGS results could not be well
replicated by qPCR, and it was hypothesized that this was due to
the inevitable sample loss and subsequent proportional alteration
in composition during sequencing library construction, which may
distort the results. The microarray chip method, however,
frequently yields a more accurate outcome because of its simpler
process compared with NGS. The major advantage of NGS lies in
identifying novel sequences or mutations; however, this was not
within the scope of the present study.
Significant differences in miR expression profile
were observed between the two groups. Among the 68 differentially
expressed miRs, let-7d-5p, miR-103-3p, miR-27b-3p and miR-30a-5p
were downregulated >2-fold in patients with IPF compared with
the control, which is consistent with previous studies that these
miRs may serve an important role in regulating IPF pathogenesis and
IPF-associated carcinogenesis (20,32).
For instance, according to a previous study (33), let-7d may target insulin-like
growth factor 1 (IGF-1) and insulin-like growth factor receptor 2.
The increased expression of IGF-1/2 in interstitial macrophages,
cells and epithelial cells may stimulate collagen production
(34,35). Downregulation of let-7d results in
overexpression of IGF-1 and, therefore, accelerates fibrosis.
Although certain miRs are downregulated in IPF, specific others are
increased compared with the control, including miR-125b-5p,
miR-128-3p, miR-21-5p, miR-100-5p, miR-140-3p and miR-374b-5p
(36). The target genes of these
increased miRs were also predicted using FunRich software (ver. 3)
with an enrichment analysis of these predicted target genes,
demonstrating that these genes are involved in the epidermal growth
factor receptor, liver kinase B1 (LKB1), vascular endothelial
growth factor, sphingosine 1-phosphate (S1P) and TNF-related
apoptosis-inducing ligand (TRAIL) signaling network (data not
shown), among which the LKB1, S1P and TRAIL signaling network have
been reported to serve negative roles in regulating fibrosis
pathogenic processes in different tissues (37–39).
These reports supported the veracity of the miRNA array assay
results in the present study. The expression profiles of miRs in
the lung of patients with pulmonary diseases have frequently been
reported to be different to that of healthy lungs. In the present
study, the differences in miR expression profiles between patients
with IPF and healthy participants have the potential to serve as
non-invasive or mini-invasive biomarkers for the screening and
prognosis of IPF, discriminating early stage IPF patients from
control subjects with high sensitivity and specificity. Using miRs
from BALF exosome as biomarkers is more practical and reliable
compared with using invasive lung tissue biopsies for IPF
diagnosis.
Based on the analysis of the miR expression profile
in the present study, miR-30a-5p was chosen as a research focus.
The miR-30a-5p family has been reported to suppress cell migration
and invasion, and have an anti-fibrotic role in liver fibrosis
(40). The miR-30a-5p expression
level in the 46 BALF samples from patients with IPF and healthy
participants was measured by RT-qPCR. The results of the present
study indicated that miR-30a-5p was downregulated by 71.8% in
patients with IPF compared with the control. Furthermore, the
microarray analysis indicated that the average expression level of
miR-30a-5p in patients with IPF was >2-fold lower than that in
healthy participants.
TAB3, which was selected for further investigation
has been reported to be involved in multiple signaling events in
IPF pathogenesis or progression, including chronic inflammatory
responses and tissue remodeling, altered fibrosis and tissue
repair, and immune responses. It was further hypothesized that
miR-30a-5p inhibits TAB3 expression by binding to the 3′UTR region
of TAB3 mRNA, and a dual-luciferase reporter assay was performed to
confirm this association (41).
Following co-transfection of 293T cells with the miR-30a-5p mimic
and vectors encoding WT TAB3 3′UTR flanked by the firefly
luciferase sequence, the luciferase activity was observed to be
decrease by up to 46.3% compared with the negative controls;
however, the miR-30a-5p mimic did not influence the firefly
luciferase activity when MT TAB3 3′UTR use used. The direct
interaction between the miR-30a-5p gene and TAB3 3′UTR indicates
that miR-30a-5p overexpression may decrease TAB3 protein
expression. As expected, when A549 and 293T cells were transfected
with miR-30a-5p, a decrease in TAB3 protein expression level was
observed compared with the control. These data suggested that TAB3
upregulation due to decreased miR-30a-5p expression may be an
important factor in IPF progression.
As targeting the TGF-β1 signaling pathway is being
considered as a strategy in IPF management (42,43),
the impact of miR-30a-5p overexpression on the expression level of
two TGF-β1 signaling-inducible fibrogenesis biomarkers, α-SMA and
fibronectin (44,45) was determined in response to
treatment of A549 cells with the potent fibrogenic cytokine, TGF-β1
(46,47). The results of the present study
demonstrate that enforced miR-30a-5p expression inhibited
TGF-β1-induced α-SMA and fibronectin upregulation, demonstrating a
potential inhibitory role in IPF progression. Notably, the results
of the present study also demonstrated that TGF-β1 treatment
decreased the miR-30a-5p expression level in A549 cells, presumably
in an indirect manner, which may be involved in the
TGF-β1-stimulated fibrogenesis mechanism.
In conclusion the results of the present study
suggested that decreased miR-30a-5p in BALF exosome may be a
potential biomarker for IPF diagnosis, and artificially increasing
miR-30a-5p level may be a potential strategy for IPF
management.
Acknowledgements
Not applicable.
Funding
The present study was funded by the Henan Natural
Science Foundation (grant no. 162300410312).
Availability of data and materials
The analyzed data sets generated during the study
are available from the corresponding author on reasonable
request.
Authors' contributions
TJ and XH completed the experiments including the
collection of exosomes and extraction of miRNAs. ZDL and LZ
performed the experiments including cell culture, RT-qPCR and
western blotting. HL and ZHL participated in the Dual-luciferase
reporter assay. BL and LM participated in statistical analysis and
bioinformatics analysis. BL wrote and revised the manuscript. All
authors read and approved the final the manuscript.
Ethics approval and consent to
participate
The present study was approved by the institutional
review board of the People's Hospital of Zhengzhou University
(Zhengzhou, China). Informed consent was obtained from each
participant in written form prior to undergoing bronchoalveolar
lavage fluid collection following the Chinese medical association
alveolar lavage operation instruction.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zibrak JD and Price D: Interstitial lung
disease: Raising the index of suspicion in primary care. NPJ Prim
Care Respir Med. 24:140542014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Matsumoto A and Azuma A: Idiopathic
pulmonary fibrosis (IPF). Nihon Rinsho. 64:1354–1360. 2006.(In
Japanese). PubMed/NCBI
|
|
3
|
Tzouvelekis A, Tzilas V, Papiris S,
Aidinis V and Bouros D: Diagnostic and prognostic challenges in
idiopathic pulmonary Fibrosis: A patient's ‘Q and A’ approach. Pulm
Pharmacol Ther. 42:21–24. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Selman M, King JTE and Pardo A: American
Thoracic Society; European Respiratory Society; American College of
Chest Physicians: Idiopathic pulmonary fibrosis: Prevailing and
evolving hypotheses about its pathogenesis and implications for
therapy. Ann Intern Med. 134:136–151. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xaubet A, Ancochea J and Molina-Molina M:
Idiopathic pulmonary fibrosis. Med Clin (Barc). 148:170–175.
2017.(In English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hostettler K: Idiopathic pulmonary
fibrosis-Pathogenesis and therapeutic concepts. Ther Umsch.
73:19–24. 2016.(In German). View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bargagli E, Lavorini F, Pistolesi M, Rosi
E, Prasse A, Rota E and Voltolini L: Trace metals in fluids lining
the respiratory system of patients with idiopathic pulmonary
fibrosis and diffuse lung diseases. J Trace Elem Med Biol.
42:39–44. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Levänen B, Bhakta NR, Paredes Torregrosa
P, Barbeau R, Hiltbrunner S, Pollack JL, Sköld CM, Svartengren M,
Grunewald J, Gabrielsson S, et al: Altered microRNA profiles in
bronchoalveolar lavage fluid exosomes in asthmatic patients. J
Allergy Clin Immunol. 131:894–903. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Michael A, Bajracharya SD, Yuen PS, Zhou
H, Star RA, Illei GG and Alevizos I: Exosomes from human saliva as
a source of microRNA biomarkers. Oral Dis. 16:34–38. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gallo A, Tandon M, Alevizos I and Illei
GG: The majority of microRNAs detectable in serum and saliva is
concentrated in exosomes. PLoS One. 7:e306792012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Javeed N and Mukhopadhyay D: Exosomes and
their role in the micro-/macro-environment: A comprehensive review.
J Biomed Res. 31:386–394. 2017.PubMed/NCBI
|
|
12
|
Chinese Medical Association Respiratory
Diseases Branch: Chinese expert consensus on detection of pathogens
in bronchoalveolar lavage of pulmonary infectious diseases. Chin J
Tuberc Respir Dis. 40:578–583. 2017.
|
|
13
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Raghu G, Collard HR, Egan JJ, Martinez FJ,
Behr J, Brown KK, Colby TV, Cordier JF, Flaherty KR, Lasky JA, et
al: An official ATS/ERS/JRS/ALAT statement: Idiopathic pulmonary
fibrosis: Evidence-based guidelines for diagnosis and management.
Am J Respir Crit Care Med. 183:788–824. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wells AU, Desai SR, Rubens MB, Goh NS,
Cramer D, Nicholson AG, Colby TV, du Bois RM and Hansell DM:
Idiopathic pulmonary fibrosis: A composite physiologic index
derived from disease extent observed by computed tomography. Am J
Respir Crit Care Med. 167:962–969. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sun X, Zhang S and Ma X: Prognostic value
of MicroRNA-125 in various human malignant Neoplasms: A
meta-analysis. Clin Lab. 61:1667–1674. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hu J, Cheng Y, Li Y, Jin Z, Pan Y, Liu G,
Fu S, Zhang Y, Feng K and Feng Y: microRNA-128 plays a critical
role in human non-small cell lung cancer tumourigenesis,
angiogenesis and lymphangiogenesis by directly targeting vascular
endothelial growth factor-C. Eur J Cancer. 50:2336–2350. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lin L, Tu HB, Wu L, Liu M and Jiang GN:
MicroRNA-21 regulates non-small cell lung cancer cell invasion and
chemo-sensitivity through SMAD7. Cell Physiol Biochem.
38:2152–2162. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jeon YJ, Middleton J, Kim T, Laganà A,
Piovan C, Secchiero P, Nuovo GJ, Cui R, Joshi P, Romano G, et al: A
set of NF-kappaB-regulated microRNAs induces acquired TRAIL
resistance in lung cancer. Proc Natl Acad Sci USA. 112:E3355–E3364.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pandit KV, Corcoran D, Yousef H,
Yarlagadda M, Tzouvelekis A, Gibson KF, Konishi K, Yousem SA, Singh
M, Handley D, et al: Inhibition and role of let-7d in idiopathic
pulmonary fibrosis. Am J Respir Crit Care Med. 182:220–229. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang D, Wang JJ, Li JS and Xu QY: miR-103
functions as a tumor suppressor by directly targeting programmed
cell death 10 in NSCLC. Oncol Res. 2017.
|
|
22
|
Zhang S, Liu H, Liu Y, Zhang J, Li H, Liu
W, Cao G, Xv P, Zhang J, Lv C and Song X: miR-30a as potential
therapeutics by targeting TET1 through regulation of Drp-1 promoter
hydroxymethylation in idiopathic pulmonary fibrosis. Int J Mol Sci.
18:E6332017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou H, Liu Y, Xiao L, Hu Z and Xia K:
Overexpression of MicroRNA-27b inhibits proliferation, migration,
and invasion via suppression of MET expression. Oncol Res.
25:147–154. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Rajasekaran S, Rajaguru P and Gandhi
Sudhakar PS: MicroRNAs as potential targets for progressive
pulmonary fibrosis. Front Pharmacol. 6:2542015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Willis BC and Borok Z: TGF-beta-induced
EMT: Mechanisms and implications for fibrotic lung disease. Am J
Physiol Lung Cell Mol Physiol. 293:L525–L534. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kis K, Liu X and Hagood JS: Myofibroblast
differentiation and survival in fibrotic disease. Expert Rev Mol
Med. 13:e272011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Choi ME, Ding Y and Kim SI: TGF-β
signaling via TAK1 pathway: Role in kidney fibrosis. Semin Nephrol.
244–252. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Usuki J, Matsuda K, Azuma A, Kudoh S and
Gemma A: Sequential analysis of myofibroblast differentiation and
transforming growth factor-β1/Smad pathway activation in murine
pulmonary fibrosis. J Nippon Med Sch. 79:46–59. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Crosby LM and Waters CM: Epithelial repair
mechanisms in the lung. Am J Physiol Lung Cell Mol Physiol.
298:L715–L731. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hsu E, Yasuoka H and Feghali-Bostwick CA:
Gene expression in pulmonary fibrosis. Crit Rev Eukaryot Gene Expr.
18:47–56. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu W, Wang Z and Liu Y: The
characterization of microRNA-mediated gene regulation as impacted
by both target site location and seed match type. PLoS One.
9:e1082602014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Oglesby I, McElvaney N and Greene C:
MicroRNAs in inflammatory lung disease-master regulators or target
practice? Respir Res. 11:1482010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Barh D, Malhotra R, Ravi B and Sindhurani
P: Microrna let-7: An emerging next-generation cancer therapeutic.
Curr Oncol. 17:70–80. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vannella KM and Wynn TA: Mechanisms of
organ injury and repair by macrophages. Annu Rev Physiol.
79:593–617. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Han ZC, Zhang HN, Wang YZ, Lv CY and Xu
ZY: Effect of the human insulin-like growth factor 1 gene
transfection to human umbilical cord blood mesenchymal stem cells.
Saudi Med J. 35:435–441. 2014.PubMed/NCBI
|
|
36
|
Pandit KV, Milosevic J and Kaminski N:
MicroRNAs in idiopathic pulmonary fibrosis. Transl Res.
157:191–199. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Du C, Ren Y, Yao F, Duan J, Zhao H, Du Y,
Xiao X, Duan H and Shi Y: Sphingosine kinase 1 protects renal
tubular epithelial cells from renal fibrosis via induction of
autophagy. Int J Biochem Cell Biol. 90:17–28. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ozcan C, Battaglia E, Young R and Suzuki
G: LKB1 knockout mouse develops spontaneous atrial fibrillation and
provides mechanistic insights into human disease process. J Am
Heart Assoc. 4:e0017332015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang J, Lesko M, Badri MH, Kapoor BC, Wu
BG, Li Y, Smaldone GC, Bonneau R, Kurtz ZD, Condos R and Segal LN:
Lung microbiome and host immune tone in subjects with idiopathic
pulmonary fibrosis treated with inhaled interferon-gamma. ERJ Open
Res. 3:00008–2017. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhong M, Bian Z and Wu Z: miR-30a
Suppresses cell migration and invasion through downregulation of
PIK3CD in colorectal carcinoma. Cell Physiol Biochem. 31:209–218.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jin Y, Chen Z, Liu X and Zhou X:
Evaluating the microRNA targeting sites by luciferase reporter gene
assay. Methods Mol Biol. 936:117–127. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Clarke DL, Murray LA, Crestani B and
Sleeman MA: Is personalised medicine the key to heterogeneity in
idiopathic pulmonary fibrosis? Pharmacol Ther. 169:35–46. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wolters PJ, Collard HR and Jones KD:
Pathogenesis of idiopathic pulmonary fibrosis. Ann Rev Pathol.
9:157–179. 2014. View Article : Google Scholar
|
|
44
|
Zhou B, Liu Y, Kahn M, Ann DK, Han A, Wang
H, Nguyen C, Flodby P, Zhong Q, Krishnaveni MS, et al: Interactions
between β-catenin and transforming growth factor-β signaling
pathways mediate epithelial-mesenchymal transition and are
dependent on the transcriptional co-activator cAMP-response
element-binding protein (CREB)-binding protein (CBP). J Biol Chem.
287:7026–7038. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Marmai C, Sutherland RE, Kim KK, Dolganov
GM, Fang X, Kim SS, Jiang S, Golden JA, Hoopes CW, Matthay MA, et
al: Alveolar epithelial cells express mesenchymal proteins in
patients with idiopathic pulmonary fibrosis. Am J Physiol Lung Cell
Mol Physiol. 301:L71–L78. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kasai H, Allen J, Mason R, Kamimura T and
Zhang Z: TGF-beta1 induces human alveolar epithelial to mesenchymal
cell transition (EMT). Respir Res. 6:562005. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Pardo A, Gibson K, Cisneros J, Richards
TJ, Yang Y, Becerril C, Yousem S, Herrera I, Ruiz V, Selman M and
Kaminski N: Up-regulation and profibrotic role of osteopontin in
human idiopathic pulmonary fibrosis. PLoS Med. 2:e2512005.
View Article : Google Scholar : PubMed/NCBI
|