Introduction
Atherosclerosis (AS), a chronic inflammatory
disease, is a major cause of cardiovascular disease and has
developed to a serious challenge to the health system (1). AS is characterized by a series of
pathological changes such as endothelial damage, lipid deposition,
monocyte adhesion and immigration, the formation of foam cells,
fatty streaks and atheromatous plaques (2,3).
Macrophage migration and foam cell formation under the endothelium,
and macrophage proliferation, aggregation and apoptosis in plaques
contribute to the development of AS (4). Disruption of the homoeostasis of
cholesterol intake and efflux in macrophages leads to lipid
accumulation and formation of foam cells (5). However, there is a lack of
therapeutics that can effectively inhibit these abnormal macrophage
features during the progression of AS (6). Therefore, improved understanding of
the potential biological mechanisms underlying the macrophage
response in AS could facilitate the development of novel treatments
for AS.
Long noncoding RNAs (lncRNAs), which are defined as
being >200-nucleotide in length, can regulate gene expression in
ischemic myocardial injury (for example, 2810403D21Rik/macrophage
Ia-positive recruiting factor) (7), pathological cardiac hypertrophy
[cardiac hypertrophy-associated regulator, (CHAR)] (8), acute myocardial infarction (CHAR,
ZNFX1 antisense RNA 1) (9) and
other cardiovascular diseases (10). Previous studies have indicated that
lncRNAs are involved in AS. For example, Chen et al
(11) demonstrated that knockdown
of lncRNA growth arrest specific 5 suppresses atherogenesis by
regulating the apoptosis of macrophages and endothelial cells via
exosomes. In addition, lncRNA-FA2H-2 has been shown to alleviate
the inflammatory response, which acts as an independent risk factor
of atherogenesis induced by oxidized low-density lipoprotein
(ox-LDL), via the induction of the autophagic flux (12). These previous studies showed that
lncRNAs act as promoting or inhibiting factors in AS development.
Mechanistically, lncRNAs exert ‘sponge-like’ effects on specific
microRNAs (miRNAs/miRs) to affect miRNA binding to target genes
(13).
Metastasis associated lung adenocarcinoma transcript
1 (MALAT1), an 8.5-kB lncRNA located at 11q13, is recognized as a
biomarker for various cancer types (14,15).
Recent studies have shown that MALAT1 is lowly expressed in AS
plaques (16) and that
hematopoietic deficiency of MALAT1 promotes atherosclerotic lesion
formation in mice via enhanced accumulation of hematopoietic cells
(17). Therefore, MALAT1 may
possess an important role in AS; however, the potential molecular
mechanism of MALAT1 regulation of cholesterol accumulation in
macrophages, which is a key event of AS progression (18), remains to be elucidated. In
addition, our previous study has demonstrated that miR-17-5p was
highly expressed in the peripheral blood of patients with AS, and
the suppression of miR-17-5p could alleviate AS in mice (19). The present study hypothesized that
MALAT1 may have a conserved miR-17-5p binding site, and ATP-binding
cassette transporter A1 (ABCA1) could be a target of miR-17-5p.
Moreover, ABCA1 has been reported to mediate cholesterol efflux
from macrophages (20,21). In addition, the present study
hypothesized that MALAT1 may regulate the miR-17-5p/ABCA1 axis to
affect cholesterol accumulation in macrophages.
Materials and methods
Patient and control specimens
Peripheral blood samples were obtained from 30
patients with AS (age, 40–84 years; male patients, 13; female
patients, 7) and 30 healthy controls (HC; age, 37–80 years; males,
9; females, 21) who had no coronary artery disease, diabetes or
cardiac insufficiency. Peripheral venous whole-blood samples
(volume, 2 ml) were collected from each subject after 12 h fasting
and stored in EDTA anticoagulant vacutainers. The participants were
consecutively recruited from The Second Affiliated Hospital of
Shenyang Medical College between May 2018 and March 2019. The
protocol was approved by The Ethics Committee of The Second
Affiliated Hospital of Shenyang Medical College (approval no.
2019-002) and conformed to the recommendations in The Declaration
of Helsinki. In addition, all patients signed an informed consent
form. Characteristics of the patients are presented in Table SI.
Cell culture
Human monocytic THP-1 cells were purchased from
Shanghai Zhong Qiao Xin Zhou Biotechnology Co., Ltd. and suspended
in RPMI-1640 medium (Shanghai Zhong Qiao Xin Zhou Biotechnology
Co., Ltd.) containing 10% FBS (GEHealthcare Life Sciences)
maintained at 37°C in a humidified atmosphere containing 5%
CO2. THP-1 cells were induced to differentiate into
macrophages using the phorbol 12-myristate 13-acetate (PMA;
MedChemExpress LLC; 100 nM) (22),
for 48 h at 37°C prior to subsequent experimentation.
293T cells (Shanghai Zhong Qiao Xin Zhou
Biotechnology Co., Ltd.) were cultured in DMEM (Gibco; Thermo
Fisher Scientific, Inc.) with 10% FBS and maintained at 37°C in a
humidified atmosphere containing 5% CO2.
Cell treatment and transfection
For ox-LDL induction, THP-1 macrophages were
incubated with 50 mg/ml ox-LDL (Peking Union-Biology Co., Ltd.;
http://www.unionbiol.com.cn/) for 48 h
at room temperature. Negative control (NC) small interfering
(si)RNA and MALAT1 siRNA were purchased from JTS Scientific
(http://www.jtsbio.com/). The following siRNA
sequences were used: MALAT1 siRNA forward,
5′-CCAGAGAACUUAAAGUCUUTT-3′, and reverse,
5′-AAGACUUUAAGUUCUCUGGTT-3′; and NC siRNA forward,
5′-UUCUCCGAACGUGUCACGUTT-3′, and reverse,
5′-ACGUGACACGUUCGGAGAATT-3′. All transfections were performed using
the Lipofectamine® 2000 reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) following the manufacturer's
instructions.
For MALAT1 knockdown, macrophages at ~70% confluence
were transfected with 100 pmol NC siRNA (50 pmol/ml) or 100 pmol
MALAT1 siRNA (50 pmol/ml) for 24 or 48 h at room temperature. Then,
cells were stimulated with 50 mg/ml ox-LDL for 48 h at 37°C or
directly collected for subsequent assays.
NC mimics forward, 5′-UUCUCCGAACGUGUCACGUTT-3′ and
reverse, 5′-ACGUGACACGUUCGGAGAATT-3′; miR-17-5p mimics forward,
5′-CAAAGUGCUUACAGUGCAGGUAG-3′ and reverse,
5′-ACCUGCACUGUAAGCACUUUGUU-3′; NC inhibitor
5′-UUGUACUACACAAAAGUACUG-3′; and miR-17-5p inhibitor
5′-CUACCUGCACUGUAAGCACUUUG-3′ were purchased from JTS Scientific.
For knockdown or overexpression of miR-17-5p, NC mimics, miR-17-5p
mimics, NC inhibitor or miR-17-5p inhibitor (100 pmol) were
transfected into macrophages (~70% confluence) for 48 h at room
temperature.
For co-transfection, macrophages were co-transfected
with NC siRNA or MALAT1 siRNA (50 pmol), and NC inhibitor or
miR-17-5p inhibitor (50 pmol). At 24 h after transfection,
macrophages were stimulated with 50 mg/ml ox-LDL for 48 h at room
temperature.
ox-LDL uptake assay
At 48 h post-transfection with NC siRNA or MALAT1
siRNA, macrophages were stimulated with 20 µg/ml Dil-ox-LDL (Peking
Union-Biology Co., Ltd.) for 4 h at room temperature. After being
washed twice with PBS, macrophages were fixed with 4%
paraformaldehyde (0.5 ml) for 15 min at room temperature, and then
stained with Hoechst staining solution (0.5 ml) for 5 min at room
temperature. Dil-ox-LDL uptake was observed under a fluorescence
microscope (magnification, ×400; Olympus Corporation).
Lipid accumulation and T-CHO
content
Oil Red O staining was used for detecting lipid
accumulation. After transfection with NC siRNA or MALAT1 siRNA for
24 h, and treatment with 50 mg/ml ox-LDL for 48 h, the macrophages
(1×106/ml) were washed twice with PBS and then fixed in
4% paraformaldehyde for 20 min at room temperature. After being
washed twice with PBS, macrophages were stained with 0.5% Oil Red O
(Sangon Biotech Co., Ltd.) for 15 min at room temperature.
Macrophages were then observed under a light microscope at ×400
magnification (Olympus Corporation).
T-CHO content in the macrophages was determined
using a Total cholesterol test kit (cat. no. A111-1; Nanjing
Jiancheng Bioengineering Institute Co., Ltd.) according to the
manufacturer's instructions.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA from human peripheral whole-blood or
macrophages was extracted using TRIpure reagent (BioTeke
Corporation) and cDNAs were synthesized using Super M-MLV reverse
transcriptase (BioTeke Corporation) according to the manufacturer's
instructions. For the RT of mRNA, the RT mixture contained 4 µl 5X
RT buffer, 2 µl 2.5 mM dNTP, 0.5 µl RNAse inhibitor (BioTeke
Corporation). After adding M-MLV reverse transcriptase, mixed
liquid was incubated at 25°C for 10 min, at 42°C for 50 min and
heated at 80°C for 10 min to terminate the reaction. RT-qPCR for
mRNA was performed using SYBR Green reaction mix (Sigma-Aldrich;
Merck KGaA) on an Exicycler 96 system (Bioneer Corporation) with
the following thermocycling parameters: Initial denaturation at
94°C for 5 min, followed by 40 cycles of 94°C for 15 sec, 60°C for
20 sec and 72°C for 30 sec. For the RT of miRNA, the reverse
transcription mixture contained 4 µl 5X RT buffer, 0.75 µl 2.5 mM
dNTP and 0.25 µl RNAse inhibitor (BioTeke Corporation). After
adding M-MLV reverse transcriptase, the mixed liquid was incubated
at 37°C for 30 min, at 42°C for 30 min and heated at 70°C for 10
min to terminate the reaction. RT-qPCR for miRNA was performed
using SYBR Green reaction mix (Sigma-Aldrich; Merck KGaA) on an
Exicycler 96 system (Bioneer Corporation) with the following
thermocycling parameters: Initial denaturation at 94°C for 2 min,
followed by 40 cycles of 94°C for 10 sec, 60°C for 15 sec and 72°C
for 15 sec. Gene expression levels were quantified via the
2−ΔΔCq method (23).
Expression level of miRNA was normalized to U6 and expression level
of mRNA was normalized to GAPDH. All primers for RT-qPCR were
synthesized by GenScript Biotech Corporation and listed in Table I.
 | Table I.Primer sequences used for
reverse-transcription-quantitative PCR. |
Table I.
Primer sequences used for
reverse-transcription-quantitative PCR.
| Gene | Primer sequence
(5′→3′) |
|---|
| MALAT1 | F:
ATACCTAACCAGGCATAACA |
|
| R:
AGTAGACCAACTAAGCGAAT |
| miR-17-5p | F:
CAAAGTGCTTACAGTGCAGGTAG |
|
| R:
GCAGGGTCCGAGGTATTC |
| U6 | F:
GCTTCGGCAGCACATATACT |
|
| R:
GCAGGGTCCGAGGTATTC |
| SR-A | F:
CACTGATTGCCCTTTACCTC |
|
| R:
TTCCTCTTCGCTGTCATTTC |
| SR-B1 | F:
CGGCGGTGATGATGGAGAAT |
|
| R:
AGAGCCCAGAGTCGGAGTTG |
| ApoE | F:
CAGCAGACCGAGTGGCAGAG |
|
| R:
TGTTCCTCCAGTTCCGATTTGT |
| ABCA1 | F:
GGCATCGTGTATGAGAAGG |
|
| R:
CTGTAGGGCAGCAGGTTT |
| GAPDH | F:
GACCTGACCTGCCGTCTAG |
|
| R:
AGGAGTGGGTGTCGCTGT |
Western blotting
Total proteins prepared from macrophages were lysed
in RIPA buffer (Beijing Solarbio Science & Technology Co.,
Ltd.) containing 1 mM PMSF (Beijing Solarbio Science &
Technology Co., Ltd.). Protein concentrations were determined by a
bicinchoninic acid kit (Beijing Solarbio Science & Technology
Co., Ltd.). Equal quantities of protein (20 µg) were separated via
8–10% SDS-PAGE and transferred onto PVDF membranes (EMD Millipore).
After blocking with 5% (w/v) skim milk in TBS-0.1% Tween-20 buffer
for 1 h at room temperature, the membranes were incubated overnight
at 4°C with the following primary antibodies: Anti-scavenger
receptor class A (SR-A; 1:3,000; cat. no. A14187; ABclonal Biotech
Co., Ltd.); anti-SR-class B member 1 (SR-B1; 1:1,000; cat. no.
A10799; ABclonal Biotech Co., Ltd.); anti-apolipoprotein E (ApoE;
1:500; cat. no. A16344; ABclonal Biotech Co., Ltd.); anti-ABCA1
(1:1,000; cat. no. ab7360; Abcam); and anti-GAPDH (1:10,000; cat.
no. 60004-1-Ig; ProteinTech Group, Inc.). On the next day, the
membranes were incubated with horseradish peroxidase-conjugated
goat anti-rabbit (1:3,000; cat. no. SE134; Beijing Solarbio Science
& Technology Co., Ltd.) or goat anti-mouse (1:3,000; cat. no.
SE131; Beijing Solarbio Science & Technology Co., Ltd.)
immunoglobulin G secondary antibody for 1 h at 37°C. Then, ECL
solution (Beijing Solarbio Science & Technology Co., Ltd.) was
used to visualize these membranes, and a gel image processing
system (Gel-Pro-Analyzer software; cat. no. WD-9413B; Beijing LIUYI
Biotechnology Co., Ltd.) was used to analyze the relative
intensities.
Dual-luciferase reporter assay
MALAT1 or the ABCA1 3′-untranslated region (3′UTR)
containing the predicted potential miR-17-5p binding sites
(StarBase prediction software; V3.0; http://starbase.sysu.edu.cn/index.php) (24) or corresponding mutant (mut)
sequences were inserted into a pmirGLO vector constructed by
GenScript Biotech Corporation. The plasmids were referred to as
‘wild-type (wt)-MALAT1’, ‘mut-MALAT1’, ‘wt-ABCA1-3′UTR’ or
‘mut-ABCA1-3′UTR’ reporter plasmid. 293T cells were co-transfected
with these constructed reporter vectors, and NC mimics or miR-17-5p
mimics (75 pmol) using Lipofectamine® 2000 reagent (9
µl; Invitrogen; Thermo Fisher Scientific, Inc.). At 48 h after
transfection, the relative luciferase activities were measured
using a dual-luciferase reporter assay kit (cat. no. KGAF040;
Nanjing KeyGen Biotech Co., Ltd.) according to the manufacturer's
protocols. The firefly luciferase expression was normalized to
Renilla.
Statistical analysis
Data are presented as the mean ± SD of three
independent replicates. Differences in sex, histories of
hypertension, diabetes and smoking were compared using a
χ2 test. Differences between two groups were analyzed
with an unpaired Student's t-test and differences between multiple
groups were analyzed using one-way ANOVA with Bonferroni post hoc
test. All statistical analyses were performed using GraphPad Prism
software (version 6.0; GraphPad Software, Inc.). P<0.05 was
considered to indicate a statistically significant difference.
Results
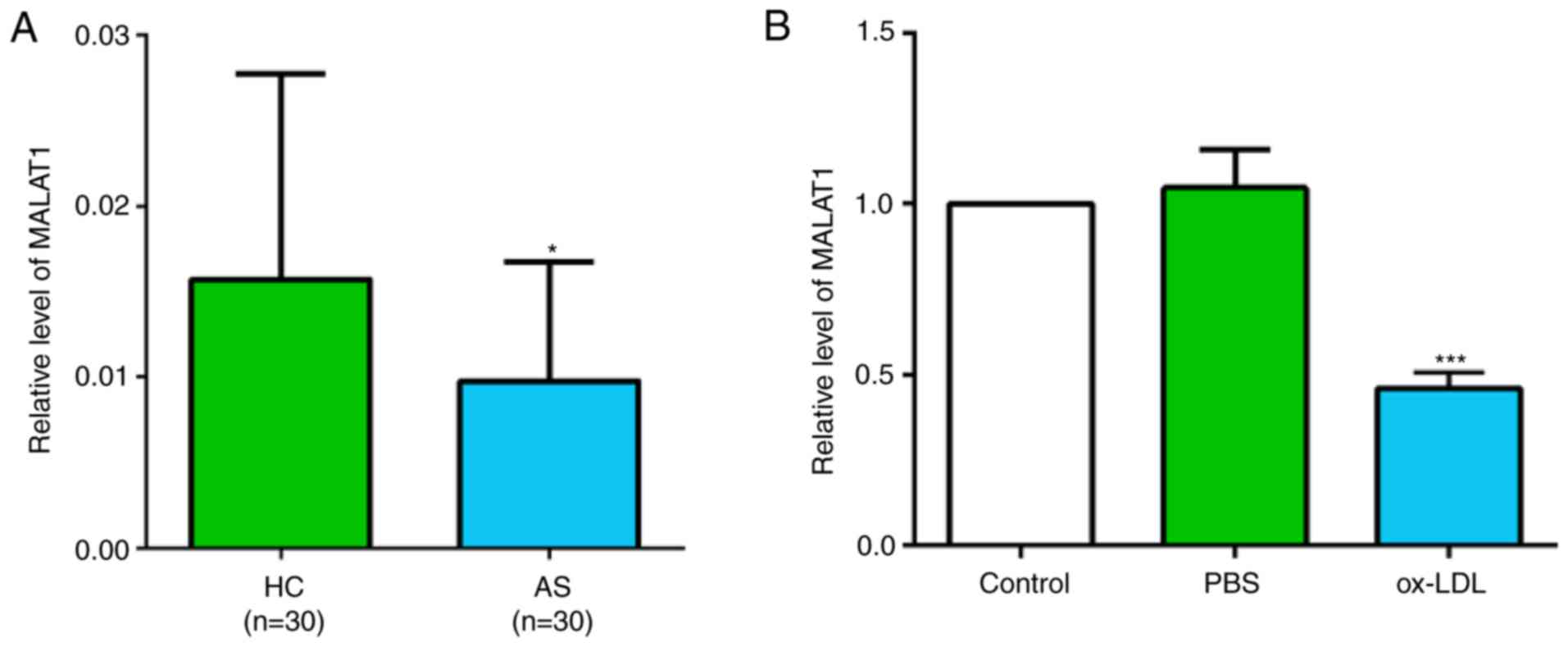
Expression of MALAT1 in patients with
AS and ox-LDL-stimulated macrophages is decreased
There were significant differences in age,
hypertension incidence, diabetes incidence, T-CHO level and
C-reactive protein level between patients with AS and healthy
controls (Table SI). The relative
expression level of MALAT1 was investigated in the peripheral blood
of 30 patients with AS and 30 healthy volunteers. The results
suggested that the MALAT1 expression level was significantly
decreased in patients with AS (Fig.
1A). Similarly, the expression level of MALAT1 was
significantly decreased in ox-LDL-stimulated THP-1 macrophages
(Fig. 1B).
Knockdown of MALAT1 increases ox-LDL
uptake, and causes changes in the expression levels of SR-A, SR-B1,
ApoE and ABCA1
To investigate the effect of MALAT1 on ox-LDL uptake
in macrophages, fluorescence staining with Dil-labeled ox-LDL,
which stains red for intracellular lipid, and Hoechst staining
solution, which stains nuclei blue, was performed. It was revealed
that Dil-ox-LDL can be taken up by macrophages, and MALAT1 siRNA
transfection significantly increased Dil-ox-LDL uptake (Fig. 2A and B). The efficiency of MALAT1
siRNA in suppressing MALAT1 expression was confirmed by RT-qPCR
(Fig. 2C). At the molecular level,
knockdown of MALAT1 resulted in changes in the expression levels of
key genes implicated in the uptake and efflux of cholesterol in
THP-1 macrophages (25). The mRNA
and protein expression levels of SR-B1, ApoE and ABCA1 were
significantly decreased, but the mRNA and protein expression levels
of SR-A were significantly upregulated in MALAT1-inhibited
macrophages (Fig. 2D-F).
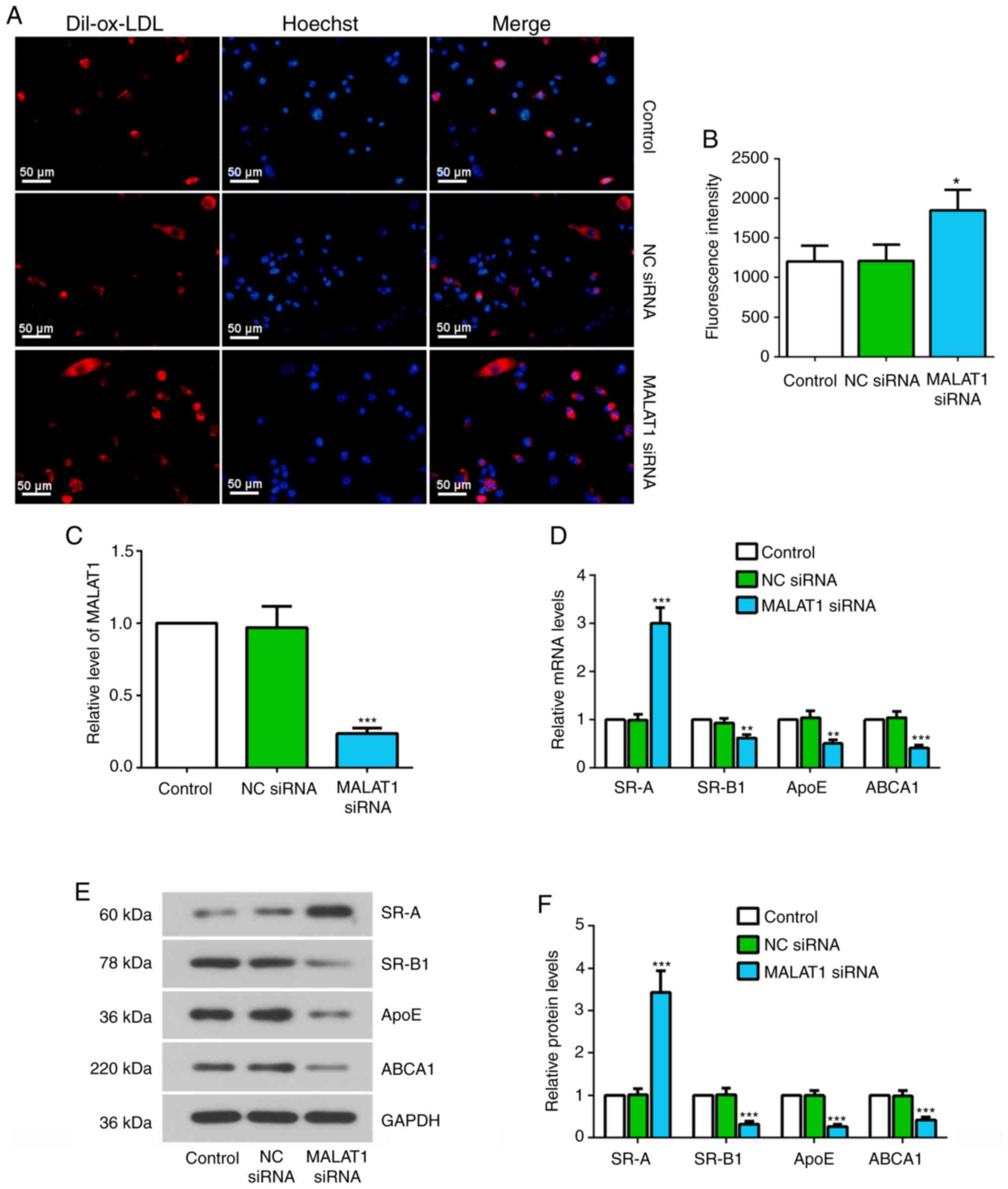 | Figure 2.Knockdown of MALAT1 increases ox-LDL
uptake, and causes changes in the expression levels of SR-A, SR-B1,
ApoE and ABCA1. (A) At 24 h after transfection with NC siRNA or
MALAT1 siRNA, macrophages were stimulated with 20 µg/ml Dil-ox-LDL
for 4 h and the ox-LDL uptake was observed. Representative
fluorescent images. Scale bar, 50 µm. (B) Quantification of mean
fluorescence intensity for ox-LDL uptake assay. (C) Relative
expression level of MALAT1 in macrophages transfected with NC siRNA
or MALAT1 siRNA. (D) Relative mRNA expression levels of SR-A,
SR-B1, ApoE and ABCA1 in macrophages transfected with NC siRNA or
MALAT1 siRNA for 48 h. (E) Western blotting, and (F) quantification
of the relative protein expression levels of SR-A, SR-B1, ApoE and
ABCA1 in macrophages transfected with NC siRNA or MALAT1 siRNA for
48 h. Data are presented as the mean ± SD. *P<0.05, **P<0.01,
***P<0.001 vs. NC siRNA group. NC, negative control; siRNA,
small interfering RNA; MALAT1, metastasis associated lung
adenocarcinoma transcript 1; ox-LDL, oxidized low-density
lipoprotein; SR-A, scavenger receptor class A; SR-B1, scavenger
receptor class B member 1; ApoE, apolipoprotein E; ABCA1,
ATP-binding cassette transporter A1. |
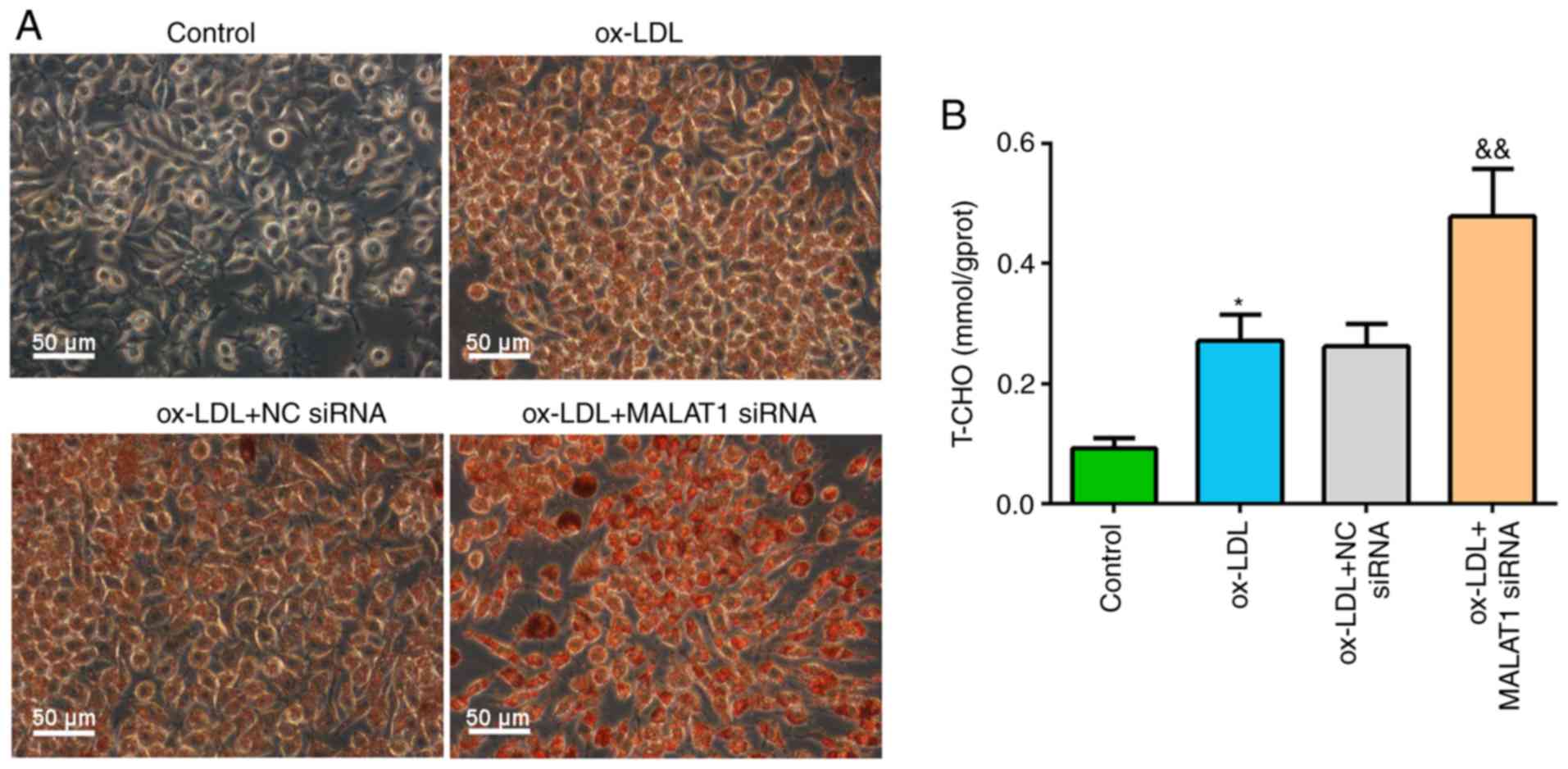
Knockdown of MALAT1 increases lipid
accumulation and T-CHO level in ox-LDL-induced macrophages
After transfection with NC siRNA or MALAT1 siRNA for
24 h, macrophages were stimulated with ox-LDL. It was demonstrated
that ox-LDL stimulation induced foam cell formation, and MALAT1
siRNA cotreatment significantly increased lipid droplet
accumulation in macrophage foam cells (Fig. 3A). Similar results were observed
for T-CHO levels; ox-LDL stimulation significantly increased
intracellular T-CHO level compared with control macrophages. In
addition, ox-LDL + MALAT1 siRNA co-treatment induced increased
T-CHO levels in macrophages compared with ox-LDL-induced
macrophages with no co-transfection (Fig. 3B).
MALAT1 regulates the miR-17-5p/ABCA1
axis
To investigate the functional mechanism of MALAT1 in
macrophages, the present bioinformatic prediction analysis
identified miR-17-5p as a potential target gene of MALAT1 and
ABCA1. The present study investigated the effect of silencing
MALAT1 on miR-17-5p and ABCA1 expression levels. The present
results suggested that silencing MALAT1 significantly increased
miR-17-5p expression and decreased ABCA1 mRNA expression in
macrophages (Fig. 4A and B).
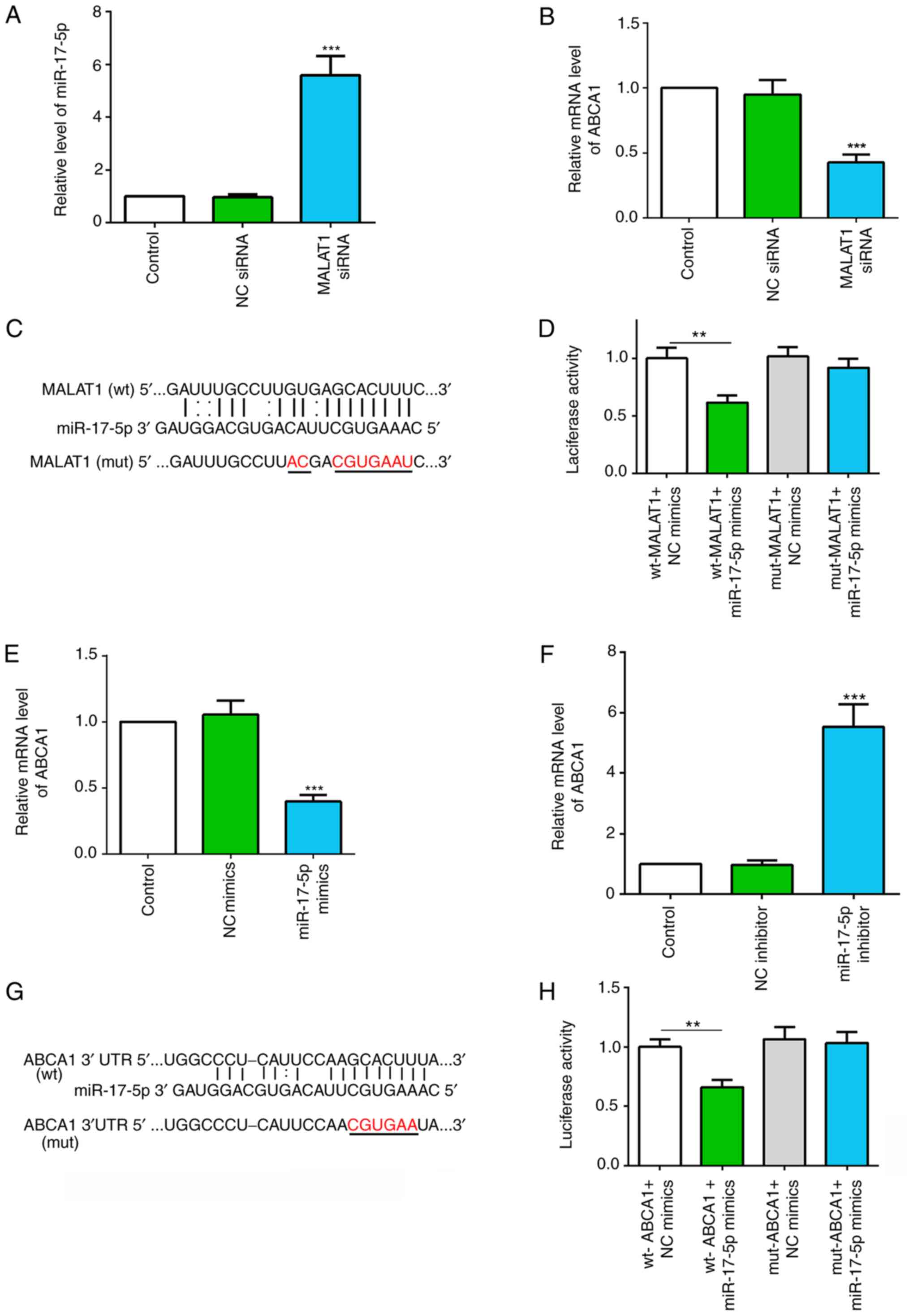 | Figure 4.MALAT1 regulates the miR-17-5p/ABCA1
axis. (A) Relative expression level of miR-17-5p in macrophages
with NC or MALAT1 siRNA transfection. (B) Relative expression level
of ABCA1 in macrophages with NC or MALAT1 siRNA transfection. (C)
Putative binding sites between MALAT1 and miR-17-5p, and the mutant
sites in the mut-MALAT1 reporter vector. (D) Luciferase activity
analysis of 293T cells co-transfected with wt-MALAT1 or mut-MALAT1,
and NC mimics or miR-17-5p mimics. (E) Relative mRNA expression
level of ABCA1 in macrophages transfected with NC mimics or
miR-17-5p mimics. (F) Relative mRNA expression level of ABCA1 in
macrophages transfected with NC inhibitor or miR-17-5p inhibitor.
(G) Putative binding sites between miR-17-5p and ABCA1 3′UTR, and
the mutant sites in mut-ABCA1 3′UTR reporter vector. (H) Luciferase
activity analysis in 293T cells co-transfected with wt-ABCA1 or
mut-ABCA1, and NC mimics or miR-17-5p mimics. Data are presented as
the mean ± SD. **P<0.01, ***P<0.001 vs. NC group [(A and B)
NC siRNA; (D) wt-MALAT1 + NC mimics; (F) NC inhibitor; (H) wt-ABCA1
+ NC mimics]. wt, wild-type; mut, mutant; NC, negative control;
miR, microRNA; ABCA1, ATP-binding cassette transporter A1; MALAT1,
metastasis associated lung adenocarcinoma transcript 1; siRNA,
small interfering RNA; 3′UTR, 3′-untranslated region. |
Computational analysis revealed complementary
sequences between MALAT1 and miR-17-5p (Fig. 4C). The dual-luciferase reporter
assay revealed that co-transfection of the luciferase vector with
the wt-MALAT1 and miR-17-5p mimics into 293T cells significantly
decreased luciferase activity compared with co-transfection with NC
mimics. However, there were no significant effects on the relative
luciferase activity following co-transfection with mut-MALAT1 and
miR-17-5p mimics (Fig. 4D).
Therefore, the present results suggested that MALAT1 may target
miR-17-5p.
The transfection efficiencies of miR-17-5p mimics
and inhibitor were demonstrated via RT-qPCR (Fig. S1A and B). It was shown that
miR-17-5p overexpression significantly decreased the mRNA
expression levels of ABCA1, and miR-17-5p suppression using a
miR-17-5p inhibitor significantly increased ABCA1 mRNA expression
in macrophages (Fig. 4E and F).
The putative miR-17-5p binding sites within the ABCA1 3′UTR are
shown in Fig. 4G. Furthermore,
dual-luciferase assay results revealed that miR-17-5p mimics and
wt-ABCA1 3′UTR co-transfection significantly inhibited relative
luciferase activity; however, luciferase activity was unchanged
after mut-ABCA1 3′UTR and miR-17-5p mimic co-transfection (Fig. 4H). Therefore, the data suggested
that miR-17-5p may directly target ABCA1. Collectively, the present
results suggested that MALAT1 may regulate the miR-17-5p/ABCA1
axis.
Inhibition of miR-17-5p expression
reverses the effect of MALAT1 in ox-LDL-stimulated macrophages
The present study investigated the effect of MALAT1
and miR-17-5p on T-CHO levels, and the protein expression levels of
ApoE and ABCA1 in ox-LDL-stimulated THP-1 macrophages. It was
revealed that inhibiting MALAT1 increased T-CHO level in
ox-LDL-stimulated macrophages, but this effect was significantly
suppressed after co-transfection with the miR-17-5p inhibitor
(Fig. 5A). Furthermore, the
protein expression levels of ApoE and ABCA1 were significantly
decreased in ox-LDL stimulated macrophages transfected with MALAT1
siRNA, while addition of the miR-17-5p inhibitor attenuated the
effect of MALAT1 knockdown on the expression level of these
proteins (Fig. 5B and C).
Collectively, the present results suggested that MALAT1 may
regulate macrophage features by regulating the miR-17-5p/ABCA1 axis
(Fig. 6).
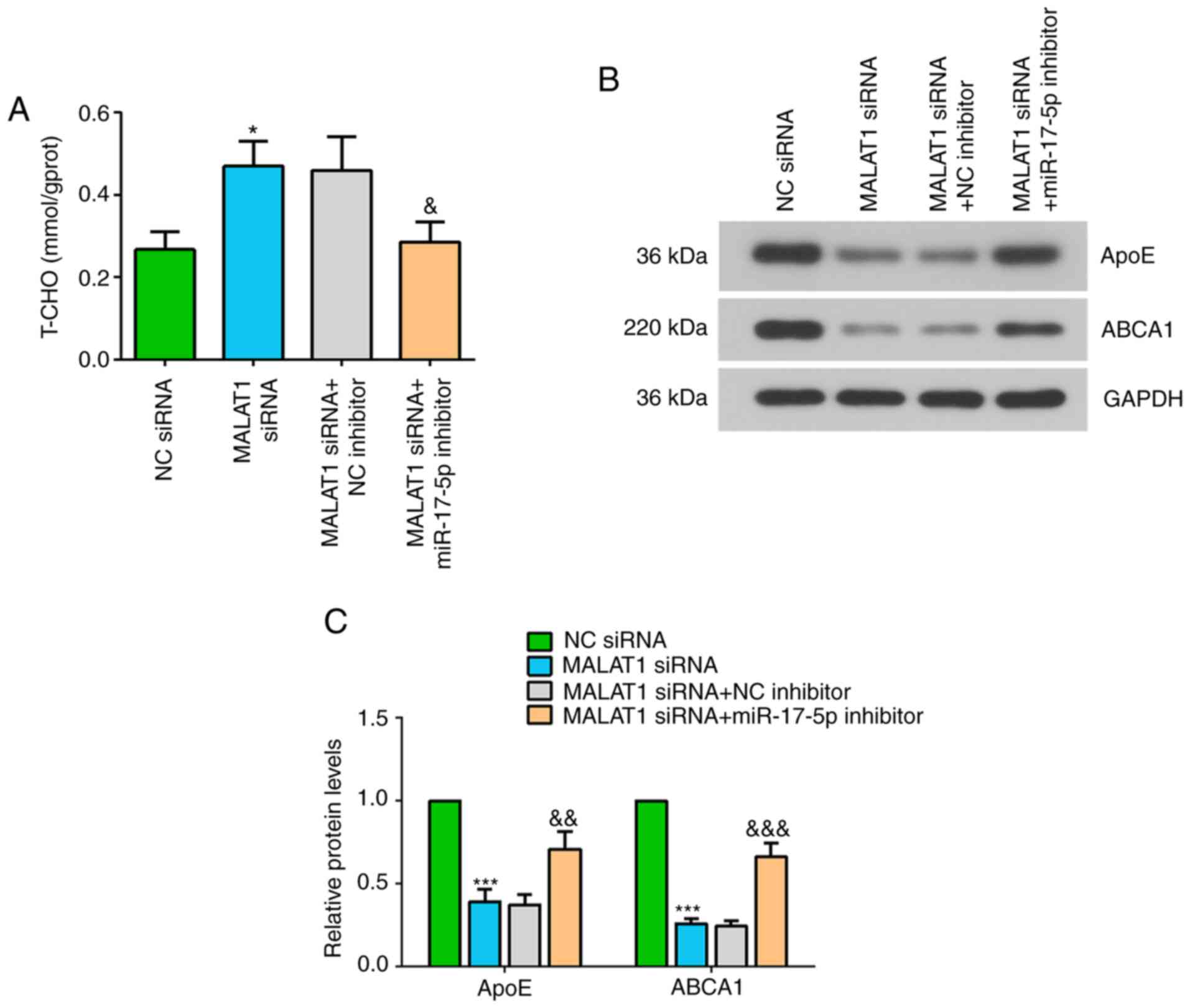 | Figure 5.Effects of MALAT1 inhibition on
ox-LDL-stimulated macrophages are partly reversed by suppressing
miR-17-5p expression. After co-transfection of NC siRNA or MALAT1
siRNA, and NC inhibitor or miR-17-5p inhibitor for 24 h,
macrophages were stimulated with 50 mg/ml ox-LDL for 48 h. (A)
Level of T-CHO in THP-1 macrophages. (B) Western blot analysis, and
(C) quantification of the expression levels of ApoE and ABCA1. Data
are presented as the mean ± SD. *P<0.05, ***P<0.001 vs. NC
siRNA group. &P<0.05,
&&P<0.01,
&&&P<0.001 vs. MALAT1 siRNA + NC
inhibitor. NC, negative control; miR, microRNA; ox-LDL, oxidized
low-density lipoprotein; ABCA1, ATP-binding cassette transporter
A1; MALAT1, metastasis associated lung adenocarcinoma transcript 1;
siRNA, small interfering RNA; ApoE apolipoprotein E; T-CHO, total
cholesterol. |
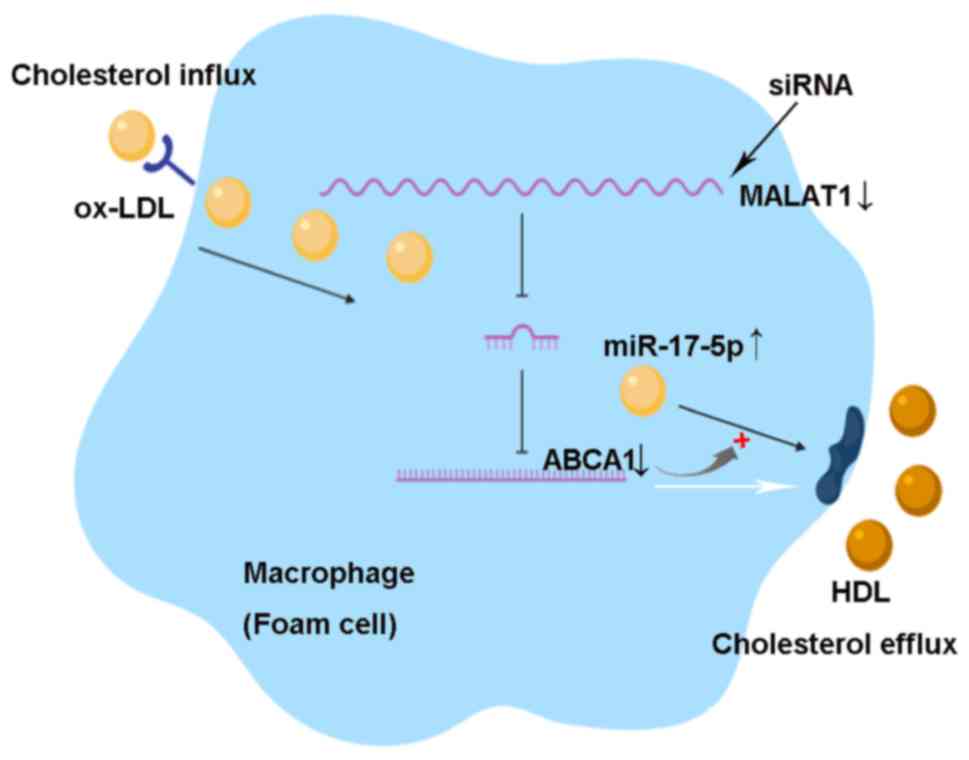 | Figure 6.Knockdown of MALAT1 promotes
cholesterol accumulation by regulating the miR-17-5p/ABCA1 axis in
ox-LDL-induced THP-1 macrophages. ABCA1 is a cell membrane protein
that mediates the transport of cholesterol, phospholipids and other
metabolites from cells to form HDL apolipoproteins. Thus, ABCA1
promotes cholesterol efflux and inhibits macrophage foam cell
formation. MALAT1 can sponge miR-17-5p, which targets the
3′-untranslated region of the ABCA1 mRNA, and facilitates its mRNA
degradation or translational repression. Knockdown of MALAT1 by
small interfering RNA increases ox-LDL uptake, lipid accumulation
and total cholesterol levels via the miR-17-5p/ABCA1 axis in
ox-LDL-induced macrophages. miR, microRNA; ABCA1, ATP-binding
cassette transporter A1; MALAT1, metastasis associated lung
adenocarcinoma transcript 1; HDL, high-density lipoprotein; ox-LDL,
oxidized low-density lipoprotein. |
Discussion
lncRNA MALAT1 has been identified as a transcript
expressing conserved sequences across several species and
demonstrated to be significantly associated with metastasis in
human non-small cell lung cancer (26). MALAT1 has been reported to be
upregulated in various types of cancer, such as bladder cancer,
gastric cancer and osteosarcoma (27–29),
but was reported to have reduced expression in AS plaques (16). It was revealed that MALAT1
expression was significantly downregulated in patients with AS and
ox-LDL-induced macrophages. The present results suggested that
knockdown of MALAT1 increased ox-LDL uptake, lipid accumulation and
T-CHO levels in ox-LDL induced macrophages, therefore indicating a
suppressive role for MALAT1 in AS, which was in line with previous
results from Li et al (30).
AS is a progressive and chronic inflammatory disease
associated with the involvement of lipid metabolism (31). One of the most important events of
early-stage AS is the accumulation of lipid-laden foam cells
derived from macrophages under the endothelium, which initiates the
formation of fatty streaks (32).
Macrophage-derived foam cell formation is caused by abnormal
cholesterol deposition and transport (33). To investigate the effects of MALAT1
on cholesterol accumulation, the present study evaluated ox-LDL
uptake by assessing the fluorescence intensity of Dil-ox-LDL, lipid
accumulation and intracellular T-CHO in PMA-activated THP-1
monocytes after transfection with MALAT1 siRNA and ox-LDL
stimulation. The incubation of macrophages with ox-LDL leads to
cholesterol ester accumulation (34,35),
and the present results suggested that ox-LDL may play a central
role in AS plaque formation. The present results indicated the
potential risk factor of MALAT1 inhibition in AS.
Macrophages regulate cholesterol homeostasis via
several factors including scavenger receptors, cholesterol
metabolism enzymes and cholesterol transporters (25,36).
A series of scavenger receptors, including SR-A and SR-B1, mediate
the binding of modified LDL (37).
Smooth muscle cells within AS plaques, and smooth muscle cells
co-incubated with macrophages or ox-LDL from macrophage-conditioned
medium both express SR-A (38,39).
The present findings indicated that MALAT1 silencing upregulated
the expression of SR-A in ox-LDL-stimulated macrophages.
SR-A-mediated uptake of modified LDL by macrophages leads to
deposition of cholesterol and foam cell formation during
atherogenesis (40). It was
demonstrated that the mRNA and protein expression levels of SR-B1,
ApoE and ABCA1 were significantly downregulated in macrophages
co-treated with MALAT1 siRNA and ox-LDL. SR-B1 plays an
antiatherogenic role, and is responsible for selective uptake of
cholesterol esters from high-density lipoprotein (HDL) and LDL, and
free cholesterol efflux to lipoprotein acceptors (41). ABCA1, a major protective factor
against AS, is involved in directing cholesterol efflux from
macrophages (42). ABCA1 can
transfer excess free cholesterol to cholesterol acceptors such as
ApoA-I or ApoE, thus promoting cholesterol efflux and inhibiting
macrophage foam cell formation (42). The reduced SR-B1, ABCA1 and ApoE
expression levels, and elevated SR-A expression were suggestive of
cholesterol homeostasis disturbance at the molecular level in the
MALAT1-silenced macrophages.
lncRNAs play important regulatory roles in the
expression and function of target mRNAs by adsorbing specific
miRNAs (43). To detect the
molecular mechanism of MALAT1 involved in the regulation of
cholesterol transport, the present study predicted miR-17-5p
binding to MALAT1 using bioinformatics prediction software. Our
previous studies have shown that miR-17-5p was elevated in patients
with AS and also macrophages of ApoE−/− mice with AS,
and that inhibition of miR-17-5p reduced inflammation and lipid
accumulation by interacting with ABCA1 in AS (19,44).
Based on the present bioinformatics prediction and our previous
results, the present study investigated the role of MALAT1 on the
miR-17-5p/ABCA1 axis. Luciferase reporter assay results indicated
that miR-17-5p was a potential target of MALAT1. Furthermore,
reduced MALAT1 expression level by MALAT1 siRNA resulted in
increased expression of miR-17-5p, which suggested a relationship
between MALAT1 and miR-17-5p. In addition, the present results
suggested that ABCA1 may be a target of miR-17-5p. Downregulation
of miR-17-5p by interferon-stimulated gene 15 has been previously
shown to increase cholesterol efflux from THP-1 macrophage-derived
foam cells by targeting Beclin-1 (45). Tang et al (46) demonstrated that interleukin-8
affects ApoA-I-mediated ABCA1-dependent cholesterol efflux via the
miR-183/ABCA1 axis in THP-1 macrophage-derived foam cells. These
previous results suggest a risk factor role of miR-17-5p and a
beneficial role of ABCA1, and that the miRNA/mRNA axis may play an
intermediary role in cholesterol metabolism. The present study
reported that MALAT1 silencing decreased T-CHO levels and the
expression of proteins that regulate cholesterol efflux in
ox-LDL-treated macrophages, whereas miR-17-5p inhibition reversed
these effects. Hence, the present findings indicated that MALAT1
could regulate the miR-17-5p/ABCA1 axis to participate in
cholesterol accumulation in macrophages.
In conclusion, the present results suggested that
knockdown of MALAT1 may promote cholesterol accumulation by
regulating the miR-17-5p/ABCA1 axis in ox-LDL-induced THP-1
macrophages. These findings provide insight into the potential
molecular mechanism of MALAT1 in the progression of AS.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This study was supported by grants from The Guide
Project for Key Research and Development Project of Liaoning
Province (grant nos. 2019010173-JH8/103 and 2017020258-201) and The
Foundation of Shenyang Science and Technology Bureau (grant no.
18-014-4-68).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LL and LT were involved in the conception and design
of the study, administrating the project, collecting the data and
writing the manuscript. JY and LY performed the experiments and
analyzed the data. All authors read and approved the final version
of the manuscript.
Ethics approval and consent to
participate
The protocol was approved by The Ethics Committee of
The Second Affiliated Hospital of Shenyang Medical College
(approval no. 2019-002) and conformed to the Declaration of
Helsinki. All patients signed an informed consent form.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AS
|
atherosclerosis
|
|
lncRNA
|
long non-coding RNA
|
|
MALAT1
|
metastasis associated lung
adenocarcinoma transcript 1
|
|
RT-qPCR
|
reverse transcription-quantitative
PCR
|
|
ox-LDL
|
oxidized low-density lipoprotein
|
|
T-CHO
|
total cholesterol
|
|
SR-B1
|
scavenger receptor class B member
1
|
|
ApoE
|
apolipoprotein E
|
|
ABCA1
|
ATP-binding cassette transporter
A1
|
|
SR-A
|
scavenger receptor class A
|
References
|
1
|
Vieceli Dalla Sega F, Fortini F, Aquila G,
Campo G, Vaccarezza M and Rizzo P: Notch signaling regulates immune
responses in atherosclerosis. Front Immunol. 10:11302019.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Berman JW and Calderon TM: The role of
endothelial cell adhesion molecules in the development of
atherosclerosis. Cardiovasc Pathol. 1:17–28. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Geng J, Liu H, Ge P, Hu T, Zhang Y, Zhang
X, Xu B, Wang B and Xie J: PM2.5 promotes plaque vulnerability at
different stages of atherosclerosis and the formation of foam cells
via TLR4/MyD88/NFκB pathway. Ecotoxicol Environ Saf. 176:76–84.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
McLaren JE, Michael DR, Ashlin TG and
Ramji DP: Cytokines, macrophage lipid metabolism and foam cells:
Implications for cardiovascular disease therapy. Prog Lipid Res.
50:331–347. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chistiakov DA, Bobryshev YV and Orekhov
AN: Macrophage-mediated cholesterol handling in atherosclerosis. J
Cell Mol Med. 20:17–28. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Finn AV, Kramer MC, Vorpahl M, Kolodgie FD
and Virmani R: Pharmacotherapy of coronary atherosclerosis. Expert
Opin Pharmacother. 10:1587–1603. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liang H, Su X, Wu Q, Shan H, Lv L, Yu T,
Zhao X, Sun J, Yang R, Zhang L, et al: lncRNA 2810403D21Rik/Mirf
promotes ischemic myocardial injury by regulating autophagy through
targeting Mir26a. Autophagy. 12:1–15. 2019.(Epub ahead of print).
View Article : Google Scholar
|
|
8
|
Zhang M, Jiang Y, Guo X, Zhang B, Wu J,
Sun J, Liang H, Shan H, Zhang Y, Liu J, et al: Long non-coding RNA
cardiac hypertrophy-associated regulator governs cardiac
hypertrophy via regulating miR-20b and the downstream PTEN/AKT
pathway. J Cell Mol Med. 23:7685–7698. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wu T, Wu D, Wu Q, Zou B, Huang X, Cheng X,
Wu Y, Hong K, Li P, Yang R, et al: Knockdown of long non-coding
RNA-ZFAS1 protects cardiomyocytes against acute myocardial
infarction via anti-apoptosis by regulating miR-150/CRP. J Cell
Biochem. 118:3281–3289. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Uchida S and Dimmeler S: Long noncoding
RNAs in cardiovascular diseases. Circ Res. 116:737–750. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Chen L, Yang W, Guo Y, Chen W, Zheng P,
Zeng J and Tong W: Exosomal lncRNA GAS5 regulates the apoptosis of
macrophages and vascular endothelial cells in atherosclerosis. PLoS
One. 12:e01854062017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Guo FX, Wu Q, Li P, Zheng L, Ye S, Dai XY,
Kang CM, Lu JB, Xu BM, Xu YJ, et al: The role of the
lncRNA-FA2H-2-MLKL pathway in atherosclerosis by regulation of
autophagy flux and inflammation through mTOR-dependent signaling.
Cell Death Differ. 26:1670–1687. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bayoumi AS, Sayed A, Broskova Z, Teoh JP,
Wilson J, Su H, Tang YL and Kim IM: Crosstalk between long
noncoding RNAs and MicroRNAs in health and disease. Int J Mol Sci.
17:3562016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lee NK, Lee JH, Ivan C, Ling H, Zhang X,
Park CH, Calin GA and Lee SK: MALAT1 promoted invasiveness of
gastric adenocarcinoma. BMC Cancer. 17:462017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Malakar P, Shilo A, Mogilevsky A, Stein I,
Pikarsky E, Nevo Y, Benyamini H, Elgavish S, Zong X, Prasanth KV
and Karni R: Long noncoding RNA MALAT1 promotes hepatocellular
carcinoma development by SRSF1 upregulation and mTOR activation.
Cancer Res. 77:1155–1167. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Arslan S, Berkan Ö, Lalem T, Özbilüm N,
Göksel S, Korkmaz Ö, Çetin N and Devaux Y; Cardiolinc™ network, :
Long non-coding RNAs in the atherosclerotic plaque.
Atherosclerosis. 266:176–181. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cremer S, Michalik KM, Fischer A,
Pfisterer L, Jaé N, Winter C, Boon RA, Muhly-Reinholz M, John D,
Uchida S, et al: Hematopoietic deficiency of the long noncoding RNA
MALAT1 promotes atherosclerosis and plaque inflammation.
Circulation. 139:1320–1334. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Moore KJ, Sheedy FJ and Fisher EA:
Macrophages in atherosclerosis: A dynamic balance. Nat Rev Immunol.
13:709–721. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tan L, Meng L, Shi X and Yu B: Knockdown
of microRNA-17-5p ameliorates atherosclerotic lesions in
ApoE−/− mice and restores the expression of very low
density lipoprotein receptor. Biotechnol Lett. 39:967–976. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhao GJ, Mo ZC, Tang SL, Ouyang XP, He PP,
Lv YC, Yao F, Tan YL, Xie W, Shi JF, et al: Chlamydia pneumoniae
negatively regulates ABCA1 expression via TLR2-Nuclear factor-kappa
B and miR-33 pathways in THP-1 macrophage-derived foam cells.
Atherosclerosis. 235:519–525. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chinetti G, Lestavel S, Bocher V, Remaley
AT, Neve B, Torra IP, Teissier E, Minnich A, Jaye M, Duverger N, et
al: PPAR-alpha and PPAR-gamma activators induce cholesterol removal
from human macrophage foam cells through stimulation of the ABCA1
pathway. Nat Med. 7:53–58. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xie Z, Yuan Y, Shi J, Shi X, Gao X, Zhao
Y, Ye J and Feng X: Metformin inhibits THP-1 macrophage-derived
foam cell formation induced by lipopolysaccharide. Xi Bao Yu Fen Zi
Mian Yi Xue Za Zhi. 32:168–172. 2016.(In Chinese). PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42:D92–D97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Voloshyna I, Hai O, Littlefield MJ,
Carsons S and Reiss AB: Resveratrol mediates anti-atherogenic
effects on cholesterol flux in human macrophages and endothelium
via PPARg and adenosine. Eur J Pharmacol. 698:299–309. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ji P, Diederichs S, Wang W, Böing S,
Metzger R, Schneider PM, Tidow N, Brandt B, Buerger H, Bulk E, et
al: MALAT-1, a novel noncoding RNA, and thymosin beta4 predict
metastasis and survival in early-stage non-small cell lung cancer.
Oncogene. 22:8031–8041. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ying L, Chen Q, Wang Y, Zhou Z, Huang Y
and Qiu F: Upregulated MALAT-1 contributes to bladder cancer cell
migration by inducing epithelial-to-mesenchymal transition. Mol
Biosyst. 8:2289–2294. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu K, Huang J, Ni J, Song D, Ding M, Wang
J, Huang X and Li W: MALAT1 promotes osteosarcoma development by
regulation of HMGB1 via miR-142-3p and miR-129-5p. Cell Cycle.
16:578–587. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Okugawa Y, Toiyama Y, Hur K, Toden S,
Saigusa S, Tanaka K, Inoue Y, Mohri Y, Kusunoki M, Boland CR and
Goel A: Metastasis-associated long non-coding RNA drives gastric
cancer development and promotes peritoneal metastasis.
Carcinogenesis. 35:2731–2739. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li S, Sun Y, Zhong L, Xiao Z, Yang M, Chen
M, Wang C, Xie X and Chen X: The suppression of ox-LDL-induced
inflammatory cytokine release and apoptosis of HCAECs by long
non-coding RNA-MALAT1 via regulating microRNA-155/SOCS1 pathway.
Nutr Metab Cardiovasc Dis. 28:1175–1187. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fenyo IM and Gafencu AV: The involvement
of the monocytes/macrophages in chronic inflammation associated
with atherosclerosis. Immunobiology. 218:1376–1384. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kita T, Kume N, Minami M, Hayashida K,
Murayama T, Sano H, Moriwaki H, Kataoka H, Nishi E, Horiuchi H, et
al: Role of oxidized LDL in atherosclerosis. Ann N Y Acad Sci.
947:199–205; discussion 205-196. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lin YW, Liu PS, Adhikari N, Hall JL and
Wei LN: RIP140 contributes to foam cell formation and
atherosclerosis by regulating cholesterol homeostasis in
macrophages. J Mol Cell Cardiol. 79:287–294. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Quinn MT, Parthasarathy S, Fong LG and
Steinberg D: Oxidatively modified low density lipoproteins: A
potential role in recruitment and retention of monocyte/macrophages
during atherogenesis. Proc Natl Acad Sci USA. 84:2995–2998. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Parthasarathy S, Quinn MT and Steinberg D:
Is oxidized low density lipoprotein involved in the recruitment and
retention of monocyte/macrophages in the artery wall during the
initiation of atherosclerosis? Basic Life Sci. 49:375–380.
1988.PubMed/NCBI
|
|
36
|
Mangum LC, Hou X, Borazjani A, Lee JH,
Ross MK and Crow JA: Silencing carboxylesterase 1 in human THP-1
macrophages perturbs genes regulated by PPARg/RXR and RAR/RXR:
Down-regulation of CYP27A1-LXRα signaling. Biochem J. 475:621–642.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pluddemann A, Neyen C and Gordon S:
Macrophage scavenger receptors and host-derived ligands. Methods.
43:207–217. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Naito M, Suzuki H, Mori T, Matsumoto A,
Kodama T and Takahashi K: Coexpression of type I and type II human
macrophage scavenger receptors in macrophages of various organs and
foam cells in atherosclerotic lesions. Am J Pathol. 141:591–599.
1992.PubMed/NCBI
|
|
39
|
Mietus-Snyder M, Gowri MS and Pitas RE:
Class A scavenger receptor up-regulation in smooth muscle cells by
oxidized low density lipoprotein. Enhancement by calcium flux and
concurrent cyclooxygenase-2 up-regulation. J Biol Chem.
275:17661–17670. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Suzuki H, Kurihara Y, Takeya M, Kamada N,
Kataoka M, Jishage K, Ueda O, Sakaguchi H, Higashi T, Suzuki T, et
al: A role for macrophage scavenger receptors in atherosclerosis
and susceptibility to infection. Nature. 386:292–296. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Rhainds D and Brissette L: The role of
scavenger receptor class B type I (SR-BI) in lipid trafficking.
Defining the rules for lipid traders. Int J Biochem Cell Biol.
36:39–77. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ren K, Jiang T, Zhou HF, Liang Y and Zhao
GJ: Apigenin retards atherogenesis by promoting ABCA1-mediated
cholesterol efflux and suppressing inflammation. Cell Physiol
Biochem. 47:2170–2184. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yoon JH, Abdelmohsen K and Gorospe M:
Functional interactions among microRNAs and long noncoding RNAs.
Semin Cell Dev Biol. 34:9–14. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tan L, Liu L, Jiang Z and Hao X:
Inhibition of microRNA-17-5p reduces the inflammation and lipid
accumulation, and up-regulates ATP-binding cassette transporterA1
in atherosclerosis. J Pharmacol Sci. 139:280–288. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Huang C, Yu XH, Zheng XL, Ou X and Tang
CK: Interferon-stimulated gene 15 promotes cholesterol efflux by
activating autophagy via the miR-17-5p/Beclin-1 pathway in THP-1
macrophage-derived foam cells. Eur J Pharmacol. 827:13–21. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Tang XE, Li H, Chen LY, Xia XD, Zhao ZW,
Zheng XL, Zhao GJ and Tang CK: IL-8 negatively regulates ABCA1
expression and cholesterol efflux via upregulating miR-183 in THP-1
macrophage-derived foam cells. Cytokine. 122:1543852018. View Article : Google Scholar : PubMed/NCBI
|