Introduction
Osteoarthritis (OA) is a severe degenerative disease
involving multiple etiologies (1).
Its main features include articular cartilage erosion, marginal
bone hyperplasia, osteophyte formation, subchondral bone sclerosis,
and varieties of biochemical and morphological changes in the
synovium and joint cavity (2). OA
lesions not only occur in the cartilage but also the entire
synovial joint (3,4). However, previous studies
investigating OA have focused on the role of cartilage (5), therefore synovial sourced pathology
in the development of OA has not been fully examined. The
physiological function of the synovium also plays a key role in
maintaining a healthy joint (6).
Moreover, nutrition and lubrication of the cartilage are supported
by the synovium. Matrix conversion plays an important role in the
joint microenvironment, and the synovial membrane can transform the
matrix (7). Fibroblast-like
synoviocytes (FLS) are located in the synovial lining layer and can
secrete cytokines and catabolic factors, of which the latter can
trigger articular cartilage degradation as a result of inflammatory
trauma and oxidative stress (8).
Synovial vascularization and synovial hyperplasia are observed in
the early stage of OA (9). A
previous morphological study of the OA synovium at different stages
observed the accumulation of large amounts of cellulose in the
synovium, which is related to disease severity (10). Moreover, synovial inflammation is
an important driver of the early pathology in OA (11). Therefore, the synovial membrane is
likely to be a potential target for OA prevention and
treatment.
The joint is frequently located in a dynamic
fluctuating oxygen environment (12). During daily activities, joints
repeatedly undergo tissue hypoxia and reoxygenation (H/R) (13). H/R under physiological and
pathological conditions has minor effects on hypoxic-tolerant
chondrocytes, but may lead to respiratory dysfunction in aerobic
synovial cells (14). Furthermore,
overproduction of reactive oxygen species (ROS) is initiated by
tissue swelling and temporary hypoxia. H/R can regulate the
activity of inducible nitric oxide synthase (iNOS) and nitrous
oxides, which are induced by interleukin (IL)-1β and tumor necrosis
factor (TNF)-α in synovial cells under such pathological conditions
(15). The local accumulation of
free radicals participates in intracellular and extracellular
oxidative damages, which compromise mitochondrial function, and
thus a detrimental cycle is initiated in the joint (16).
Cellular senescence can be induced by a variety of
stressors and different genotoxic stressors induce different
phenotypes of cellular senescence (17). With aging, large numbers of
senescent cells (SnCs) accumulate in the human body, which can
induce a drastic modification in gene expression and secretion of
multiple of factors to cause a senescence-associated secretory
phenotype (SASP) (18,19). Furthermore, the actions of SnCs
strengthen senescence via autocrine signaling, which stimulates
neighboring cells to undergo senescence (18,19).
When joint organs are unable to replace SnCs or reduce damage,
intracellular damage accumulates and exerts deleterious effects on
both the host cells and other cell types, which impairs their
function and contributes to the advanced stage of OA (20). The SASP includes a varieties of
chemical factors, pro-inflammatory factors, extracellular
matrix-degrading proteins and growth factors, which play an
important role in the local microenvironment of tissues (21–23).
OA is a typical age-related disease, characterized by typical
senescent features. Moreover, the characteristics of catabolic
mediators and inflammation in the pathogenesis of OA are similar to
those observed in ‘classic’ SnCs. The presence of inflammatory
factors among the SASP phenotype can induce stress-induced
premature senescence. Furthermore, SnCs can enhance the level of
inflammation and form a positive feedback loop (24).
The JNK signaling pathway includes important members
of the mitogen-activated protein kinase (MAPK) family. This pathway
is widely involved in regulation of cell proliferation, metabolism,
apoptosis and DNA damage repair (25–27).
Furthermore, the JNK pathway can be activated by stress stimulation
(28,29). Previous studies have shown that
activation of the JNK signaling pathway is associated with
senescence and various degenerative diseases (29–31).
Since the JNK signaling pathway can regulate several inflammatory
pathways, blocking this mechanism may effectively inhibit
inflammation and prevent joint destruction in experimental models
of arthritis (32,33). Therefore, the present study
hypothesized that H/R activation of the JNK pathway in synovium may
promote cell damage and senescence, which could lead to OA
lesions.
Patients and methods
Patients and tissue
The present study collected synovial tissue
specimens from 20 patients (age, 33–72 years; mean age, 52 years;
men, 10; women, 10) each with acute knee injury and OA-total knee
replacement in the Department of Orthopedics, Wuhan University
People's Hospital within 4 h of acute knee joint trauma operation.
The specimens were collected between September 2017 and January
2019. Prior to the study, all patients voluntarily agreed to
participate. The present study was approved by the Ethics Committee
of Renmin Hospital of Wuhan University.
Tissue mass culture
Tissues were soaked in PBS solution for 5 min and
then rinsed thrice with PBS at room temperature. Then, the synovium
was chopped into small pieces that were placed at 37°C in high
glucose DMEM (Gibco; Thermo Fisher Scientific, Inc.) supplemented
with 15% FBS (Gibco; Thermo Fisher Scientific, Inc.) and 1%
penicillin/streptomycin in a humidified atmosphere containing 5%
CO2. The medium was changed once after 3–4 days.
Culture and isolation of FLS
Fresh synovial tissue was soaked in PBS solution for
5 min and rinsed thrice with PBS. Fat and other fibrous tissue were
removed and placed in a petri dish. The synovial was cut into
pieces with ophthalmic shears and placed into a culture bottle.
Then, 0.2% collagenase II (Sigma-Aldrich; Merck KGaA) with 10% FBS
was added and placed into a CO2 incubator for digestion
(37°C, 5% CO2). After digesting for 6 h, the supernatant
was removed by centrifugation at 1,000 × g at 37°C for 5 min, and
then 4 ml culture medium (15% FBS; 1% penicillin/streptomycin) was
transferred into a clean culture bottle and cultured in an
incubator (37°C, 5% CO2). The following day, the
impurities were gently washed away with PBS buffer and replaced
with DMEM containing 10% FBS and 1% mixture of penicillin and
streptomycin. The medium was changed once every 2–3 days.
Hypoxia/reoxygenation (H/R)
intervention, synovial tissue block and FLS experimental
grouping
The patients were sub-divided into four groups: i)
Control, with healthy synovium; ii) H/R intervention; iii) TNF-α
(PeproTech, Inc.) intervention (20 ng/ml TNF-α incubated at 37°C
for 24 h); and iv) H/R+TNF-α intervention, with healthy tissue mass
incubated at 37°C with 20 ng/ml TNF-α for 24 h before H/R
intervention. The normal culture conditions (5% CO2;
37°C) and hypoxic culture conditions (5% CO2; 1%
O2; 37°C) were established by regulating several
conventional cell incubators (14). The synovial tissue mass and FLS
culture vessel were placed in anoxic conditions for 2 h and
conventional conditions for 2 h, and this process was repeated for
three cycles. The present study synchronously changed the liquid in
each experimental group after 24 h in order to minimize the
experimental errors among the groups.
Hematoxylin and eosin (H&E)
staining
The cultured tissue block was embedded in paraffin
and sliced at a thickness of 4 µm, and then rinsed with tap water
at room temperature for 5 min. Tissues were differentiated with 1%
hydrochloric acid and 75% alcohol for 3 sec, rinsed with water for
10 min at room temperature. Then, 0.5% alcohol-soluble eosin stain
was added to stain for 30 sec at room temperature, and the sample
was dehydrated with gradient ethanol (95 and 00%). After the xylene
treatment became transparent for 1 min, the sample was sealed with
neutral glue. The sample was observed under a light microscope at
magnification, ×400.
Immunofluorescence and
immunohistochemistry
The cultured tissue blocks were embedded in paraffin
and sectioned at the thickness of 4 µm. Samples were fixed with 4%
paraformaldehyde for 15 min and washed in PBS at room temperature.
Then, samples were permeabilized with 5% BSA (Calico LLC) and 0.1%
Triton X-100 for 1 h at room temperature. Subsequently, samples
were incubated the following primary antibodies: Rabbit vimentin (1
mg/ml; Abcam; cat. no. ab63379), CD90 (1:100; Abcam; cat. no.
ab125215), IL-1β (1:150; Abcam; cat. no. 1743-1), p16 (1:200;
Abcam; cat. no. 1712-1) and matrix metalloproteinase-13 (MMP-13;
1:200; Abcam; cat. no. ab9128) at 4°C overnight. Samples were then
incubated with anti-rabbit FITC (1:200; Abcam; cat. no. ab27912)
and anti-rabbit CY3 (1:200; Abcam; cat. no. ab6939) conjugated
secondary antibody at room temperature for 2 h. After washing with
PBS, the sections were sealed with fluorescent sealing tablets and
observed under an Olympus fluorescent microscope at magnification,
×400 (Olympus Corporation) after counterstaining with DAPI (Nanjing
KeyGen Biotech Co. Ltd.), which was used to counterstain the nuclei
for 5 min at room temperature.
ELISA
FLS were divided into the specified four groups.
Cells and cell fragments were removed by centrifugation during the
supernatant analysis, which was at 1,000 × g at room temperature
for 15 min. ELISA kits (R&D Systems. Inc.) were used to assess
the expression levels of IL-6 (R&D Systems, Inc.; cat. no.
D6050) and IL-1β (R&D Systems, Inc.; cat no. DLB50) in cell
culture supernatants according to the manufacturer's
instructions.
Western blot analysis
High mobility group box 1 (HMGB1), caspase-8
(Casp8), p16, p21, matrix metalloproteinase (MMP)-3 and MMP-13
protein expression levels in FLS were analyzed by western blotting.
After digested by 0.25% trypsin (Servicebio, Inc.) and
centrifugation at 1,000 × g at room temperature for 5 min, FLS were
collected and washed twice with PBS. Cell precipitation was
collected by centrifugation at 1,000 × g at room temperature for 5
min. Phosphatase inhibitor: Protease inhibitor: RIPA lysate
(Beyotime Institute of Biotechnology) was mixed at a ratio of
(1:1:50) and added to cells for 30 min on ice. Total protein was
collected by centrifugation for 15 min at 4°C 12,000 × g. For
denaturation, proteins were boiled for 15 min at 100°C. A
bicinchoninic acid kit was used to detect protein concentration
(Beyotime Institute of Biotechnology). The aliquots of protein (40
µg per lane) were separated by SDS-PAGE on a 12% gel and then
transferred to PVDF membranes. The membranes were blocked with 5%
non-fat milk in TBST (0.05% Tween-20) for 1 h at 37°C. After being
blocked, membranes were incubated overnight at 4°C with antibodies:
HMGB1 (1:1,000; cat. no. ab77302; Abcam), Casp8 (1:1,000; cat. no.
ab25901; Abcam), p16 (1:1,000; cat. no. ab211542; Abcam), p21
(1:1,000; cat. no. ab109520; Abcam), MMP-3 (1:1,000; cat. no.
ab52915; Abcam), MMP-13 (1:1,000; cat. no. ab39012; Abcam) and
GAPDH (1:5,000; cat. no. GB12002; Servicebio, Inc.). After
incubation, the PVDF membranes were washed thrice with TBST buffer
and incubated with horseradish peroxidase-conjugated goat
anti-rabbit (cat. no. GB23303; Servicebio, Inc.; 1:3,000) secondary
antibodies at room temperature for 1 h. After washing three times
with TBST at 37°C for 30 min, a chemiluminescence luminol reagent
(cat. no. G2014, Servicebio, Inc.) was used to visualize the
protein bands using the Image Lab 5.2 quantitative assay system
(Bio-Rad Laboratories, Inc.). The relative protein levels were
determined by normalizing to GAPDH.
Reverse transcription-quantitative PCR
(RT-qPCR)
TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) was used to extract total RNA from FLS.
The RevertAid First Strand cDNA Synthesis kit (Fermentas; Thermo
Fisher Scientific, Inc.) was used to reverse transcribe cDNA from
the RNA. The cDNAs were produced with the kit and incubated at 37°C
for 15 min and at 85°C for 5 sec. qPCR was carried out using Hieff
qPCR SYBR Green Master Mix (No Rox; Shanghai Yeasen Biotechnology
Co., Ltd.) and was performed on a StepOnePlus device (Applied
Biosystems; Thermo Fisher Scientific, Inc.) with the following
thermocycling conditions: Initial denaturation at 95°C for 10 sec,
followed by 40 cycles at 95°C for 5 sec and at 60°C for 20 sec. The
following primers were used: HMGB1 forward,
5′-ATGGGCAAAGGAGATCCTAA-3′ and reverse,
5′-TTAATCATCATCATCTTCTTCTTCA-3′; p16 Ink4a forward,
5′-GATCCAGGTGGGTAGAAGGTC-3′ and reverse, 5′-CCCCTGCAAACTTCGTCCT-3′;
p21 forward, 5′-GCGATGGAACTTCGACTTTGT-3′ and reverse,
5′-GGGCTTCCTCTTGGAGAAGAT-3′; MMP-3 forward,
5′-GGGTGAGGACACCAGCATGA-3′ and reverse,
5′-CAGAGTGTCGGAGTCCAGCTTC-3′; MMP-13 forward,
5′-TTGAGGATACAGGCAAGACT-3′ and reverse, 5′-TGGAAGTATTACCCCAAATG-3′;
IL-1β forward, 5′-AAACAGATGAAGTGCTCCTTCCAGG-3′ and reverse,
5′-TGGAGAACACCACTTGTTGCTCCA-3′; IL-6 forward,
5′-TGCTGGTGTGTGACGTTCCC-3′ and reverse,
5′-CCATCTTTGGAAGGTTCAGGTTG-3′; Casp8 forward,
5′-CGGGGTACCATGGACTTCAGCAGAAATC-3′ and reverse,
5′-TCAATCAGAAGGGAAGACAA-3′; JNK forward,
5′-ATGAGCAGAAGCAAGCGTGAC-3′ and reverse,
5′-CTGGGCTTTAAGTCCCGATG-3′; Bax forward,
5′-ACCAAGAAGCTGAGCGAGTGT-3′ and reverse,
5′-ACAAACATGGTCACGGTCTGC-3′; Bcl-2 forward,
5′-AGATGTCCAGCCAGCTGCAC-3′ and reverse, 5′-TGTTGACTTCACTTGTGGCC-3′;
p53 forward, 5′-CAGCCAAGTCTGTGACTTGCACGTAC-3′ and reverse,
5′-CTATGTCGAAAAGTGTTTCTGTCATC-3′; and GAPDH forward,
5′-GAGTCAACGGATTTGGTCGT-3′ and reverse,
5′-TGAGGCCCAAGGCCACAGGTA-3′. GADPH was used as an internal
reference. The 2−ΔΔCq method was used to calculate the
relative mRNA expression levels (34).
Senescence-associated β-galactosidase
(SA-β-gal) staining
According to the manufacturer's instructions, FLS
with the cell density of 1×106 cells/well were washed
twice and then fixed by adding 1.5 ml of 1X SA-β-gal fixative,
followed by incubation for 10 min at 25°C. Then, the fixed cells
were washed three times with PBS and stained using an
SA-β-galactosidase staining kit (BioVision, Inc.; cat. no.
K320-250). The fixed FLS were incubated overnight at 37°C. Then,
cells were observed and image under a light microscope at
magnification, ×200 (Olympus Corporation); SnCs were identified as
blue-stained cells. Image-Pro Plus 6.0 (Media Cybernetics, Inc.)
was used to analyze the number of positive cells in the
sections.
Microscopic assessment of
mitochondrial membrane potential (ΔΨm)
ΔΨm was evaluated using an ΔΨm assay kit (Beijing
Solarbio Science & Technology Co., Ltd.; cat. no. M8650) with
JC-1. FLS were washed with PBS and stained with 1 ml JC-1 staining
solution for 20 min at 37°C. After staining, the ratio of red and
green light was observed under a fluorescence microscope at
magnification, ×200 after adding medium with 10% FBS.
Flow cytometric determination of
ΔΨm
ΔΨm was determined using a JC-1 containing ΔΨm kit
(Beijing Solarbio Science & Technology Co., Ltd.; cat. no.
M8650). FLS were washed with PBS. After digestion using 0.25%
trypsin (Servicebio, Inc.) and centrifugation at 1,000 × g at room
temperature for 5 min, FLS were soaked at 37°C for 20 min with 800
µl of JC-1 working solution. After washing with PBS, FLS were
resuspended in 1 ml JC-1buffer (1X) and measured by a flow
cytometer FACSCalibur (Becton, Dickinson and Company). Then FlowJo
10 software was used for analysis (FlowJo LLC).
Detection of the openness of the
mitochondrial permeability transition pore (MPTP)
The openness of MPTP was tested using an MPTP
staining kit (BestBio Co., Ltd.; cat. no. BB-48122). After
digestion with0.25% trypsin (Servicebio, Inc.) and centrifugation
at 1,000 × g at room temperature for 5 min, 3 ml probe working
solution (BBcellP robe™ CA1 Probe Diluted with 500X in 1X mPTP
Staining Buffer and 5 ml fluorescence quencher agent) was added to
each sample and incubated at 37°C in the dark for 15 min. After
this step and after washing with 3 ml MPTP Staining Buffer (1X),
FLS were suspended with 400 ml of Hanks Balanced Salt solution at
room temperature. The openness of MPTP was calculated by measuring
the fluorescence intensity of the sample by flow cytometry
FACSCalibur (Becton, Dickinson and Company). Then FlowJo v10
software was used for analysis (FlowJo LLC).
Measurement of intracellular ROS
The amount of ROS produced in cells was measured by
2′,7′-dichlorodihydrofluorescein diacetate (DCHF-DA) according to
the manufacturer's instructions. FLS cell culture medium was
stained with 10 µm DCHF-DA (Invitrogen; Thermo Fisher Scientific,
Inc.) for 2 h at 37°C. FLS samples were collected, washed twice
with pre-chilled PBS and assayed by flow cytometry FACSCalibur
(Becton Dickinson and Company). Then FlowJo v10 software was used
for analysis (FlowJo LLC).
Ultrastructural detection of SnCs
The ultrastructure of cells was observed by
transmission electron microscopy (TEM). Sections were sliced into
80 nm pieces and fixed in 2.5% glutaraldehyde solution at 4°C for
>2 h. After being washed with PBS (pH 7.2), synoviocytes were
fixed with 1% osmic acid at 4°C for 2 h and dehydrated with a
series of ethanol concentrations (50, 70, 90 and 100%) for 10 min
at 4°C. After being embedded in epoxy resin, stained with saturated
uranium acetate for 30 min and lead citrate staining for 5–8 min,
the synoviocytes were observed using a TEM (Hitachi, Ltd.) at
magnification, ×100,000.
Statistical analysis
Data are expressed as the mean ± standard deviation
for each group. Intra-group differences were assessed using
Student's t-test and one-way analysis of variance using SPSS 16.0
software (SPSS, Inc.), followed by Bonferroni post-hoc correction
for multiple testing using GraphPad Prism software (version 7.04;
GraphPad Software, Inc.) P<0.05 was considered to indicate a
statistically significant difference.
Results
H/R increases TNF-α-induced IL-1β, P16
and MMP-13 in synovial tissue
Pathological and senescent phenotypic differences
were assessed between healthy individuals and patients with OA. It
was determined that expression levels of pathological and senescent
markers in the synovial tissue mass before and after H/R were
different. The experiment was divided into two stages. Firstly, the
difference in synovial tissue masses from healthy controls and
patients with OA were examined with H&E staining (Fig. 1A), and IL-1β (Fig. 1B), p16 (Fig. 1C) and MMP-13 (Fig. 1D) staining. It was revealed that
the pathological features of H&E staining in OA tissues were
more evident, compared with the healthy tissues (Fig. 1A). Furthermore, in the healthy
tissue group, no inflammation, edema or other pathological changes
were observed. However, in the OA group, synovial edema was clear
and there were higher levels of inflammatory cell infiltration
(Fig. 1A). Moreover, IL-1β, p16
and MMP-13 expression levels were significantly upregulated in the
OA group (Fig. 1B-D). In the
second stage, the experiment was divided into four groups. The
present results indicated that the expression levels of IL-1β
(Fig. 1E), p16 (Fig. 1F) and MMP-13 (Fig. 1G) were significantly increased
after H/R compared with the control group (Fig. 1E-I).
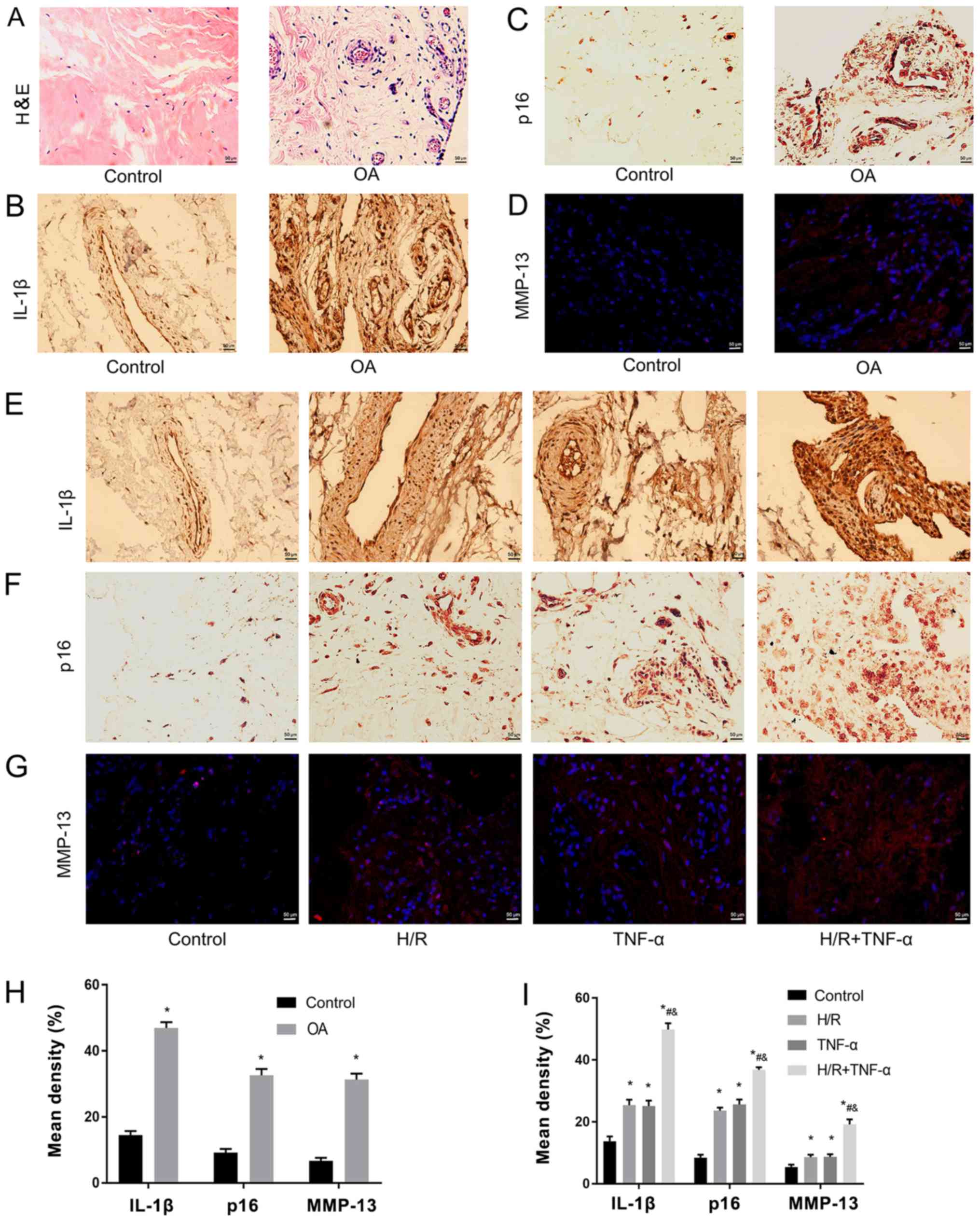 | Figure 1.H/R increases TNF-α-induced IL-1β,
p16 and MMP-13 in synovial tissue. (A) Pathological phenotypic
H&E staining of health patients and patients with OA.
Magnification, ×400. (B) Immunohistochemistry of IL-1β and (C) p16.
Magnification, ×400. (D) Immunofluorescence was used to detect the
expression level of MMP-13. Magnification, ×400. Expression levels
of (E) IL-1β, (F) p16 and (G) MMP-13 as senescent phenotype markers
in the normal synovium group, H/R group, TNF-α group and H/R +
TNF-α group. (H) Quantitative analysis of the expression levels of
IL-1β, p16 and MMP-13 in healthy individuals and patients with OA.
Magnification, ×400. (I) Quantification of the expression levels of
IL-1β, p16 and MMP-13 in the normal synovium group, H/R group,
TNF-α group and H/R + TNF-α group. Data are presented as the mean ±
SD. n=3. *P<0.05 vs. Control group; #P<0.05 vs.
H/R group; &P<0.05 vs. TNF-α group. N=3. IL,
interleukin; TNF-α, tumor necrosis factor α; MMP, matrix
metalloproteinase; OA, osteoarthritis; H/R, Hypoxia/reoxygenation;
H&E, hematoxylin and eosin. |
Primary culture of FLS, identification
of cellular immunofluorescence and detection of SA-β-gal
It was demonstrated that most FLS extracted from the
synovial tissue were spindle-shaped. After 4 days of growth, FLS
were sub-passaged to the third generation at a ratio of 1:2
(Fig. 2A). Moreover, positive
immunocytochemical identification of CD90 and vimentin protein
staining was performed for the identification of FLS (Fig. 2B). In addition, the expression
level of SA-β-gal in FLS after H/R was studied (Fig. 2C), and the positive cells (blue)
expressing SA-β-galactosidase were quantitatively assessed
(Fig. 2D). It was determined that
the expression level SA-β-gal in FLS increased with H/R
intervention compared to the control group. Furthermore, in the H/R
+ TNF-α group, the expression level of SA-β-gal was further
increased compared with the control group, H/R group and TNF-α
group, which indicated a larger proportion of senescent cells.
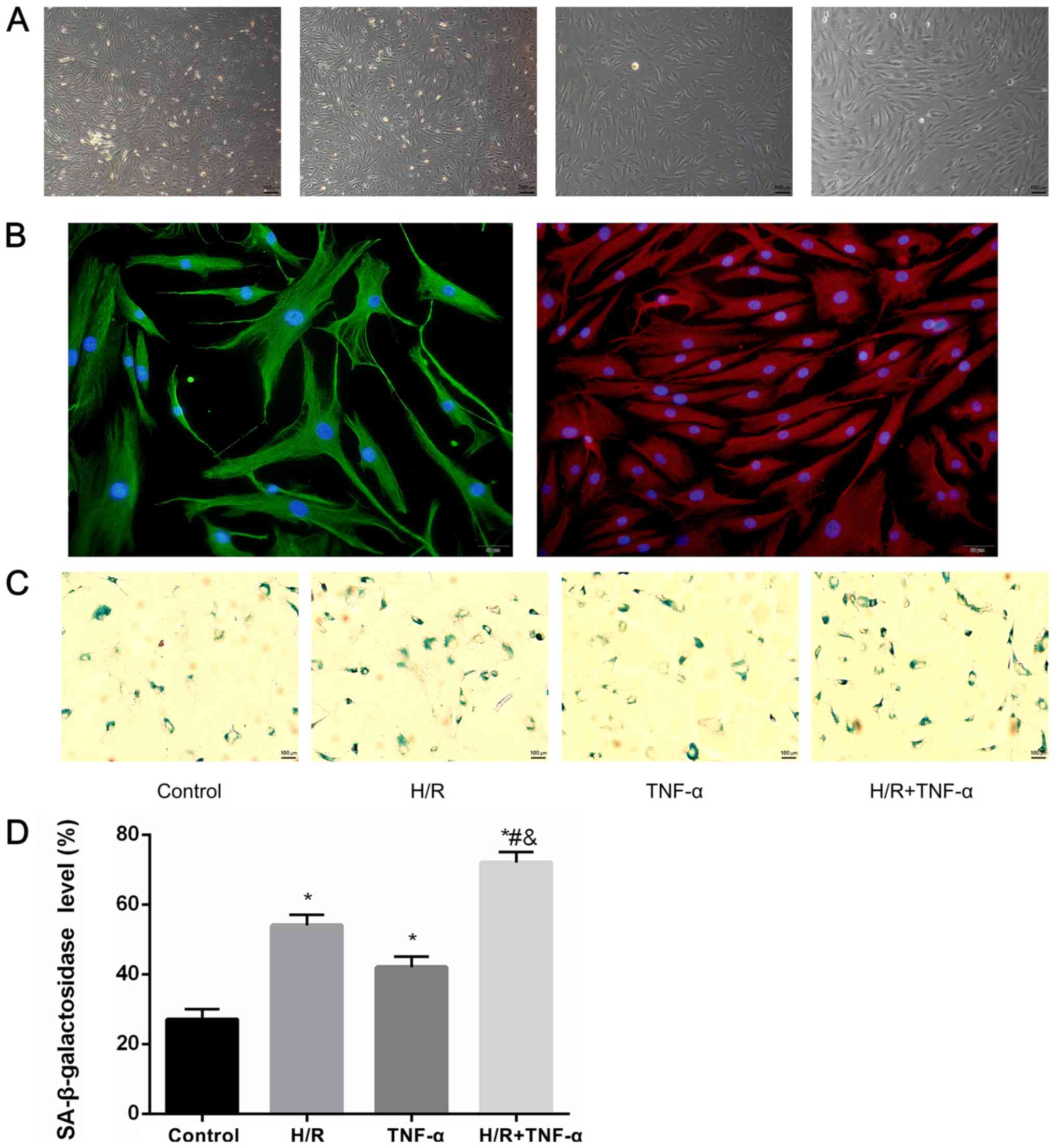 | Figure 2.Primary culture of FLS,
identification of cellular immunofluorescence and detection of
SA-β-gal. (A) FLS extracted from synovial; a large number of the
FLS were spindle-shaped. Magnification, ×40. The 3–5 generation FLS
had a uniform arc arrangement, which were fusiform. Magnification,
×100. (B) Positive expression of CD90, which stains red, vimentin,
which stains green, and the nucleus, which stains blue.
Magnification, ×200. (C) Detection of senescence marker SA-β-gal
blue staining and (D) quantitative analysis of positive cells.
Magnification, ×100. Data are presented as the mean ± SD. n=3.
*P<0.05 vs. Control group; #P<0.05 vs. H/R group;
&P<0.05 vs. TNF-α group. N=3. SA-β-gal,
senescence-associated β-galactosidase; FLS, fibroblast-like
synoviocytes; H/R, hypoxia/reoxygenation; TNF-α, tumor necrosis
factor α. |
Relative expression levels of SASP
proteins in FLS after H/R
The expression levels of the senescent phenotype
proteins and inflammatory factors HMGB1, Casp8, p16, p21, MMP-3,
MMP-13, IL-6 and IL-1β in FLS before and after H/R were studied.
IL-1β (Fig. 3A) and IL-6 (Fig. 3B) levels in FLS were assessed using
ELISA, and HMGB1, Casp8, p16, p21, MMP-3 and MMP-13 proteins were
detected by western blotting (Fig. 3C
and D). In addition, RT-qPCR was used to detect the mRNA
expression levels of HMGB1, Casp8, p16, p21, MMP-3, MMP-13, IL-1β
and IL-6 (Fig. 3E). The results
indicated that the protein and mRNA expression levels of HMGB1,
Casp8, p16, p21, MMP-3, MMP-13, IL-1β and IL-6 were significantly
increased in the H/R environment compared with the control.
Moreover, the expression levels of all these factors were further
significantly increased after pre-incubation with TNF-α before 24 h
of H/R compared with the control group, H/R group and TNF-α
group.
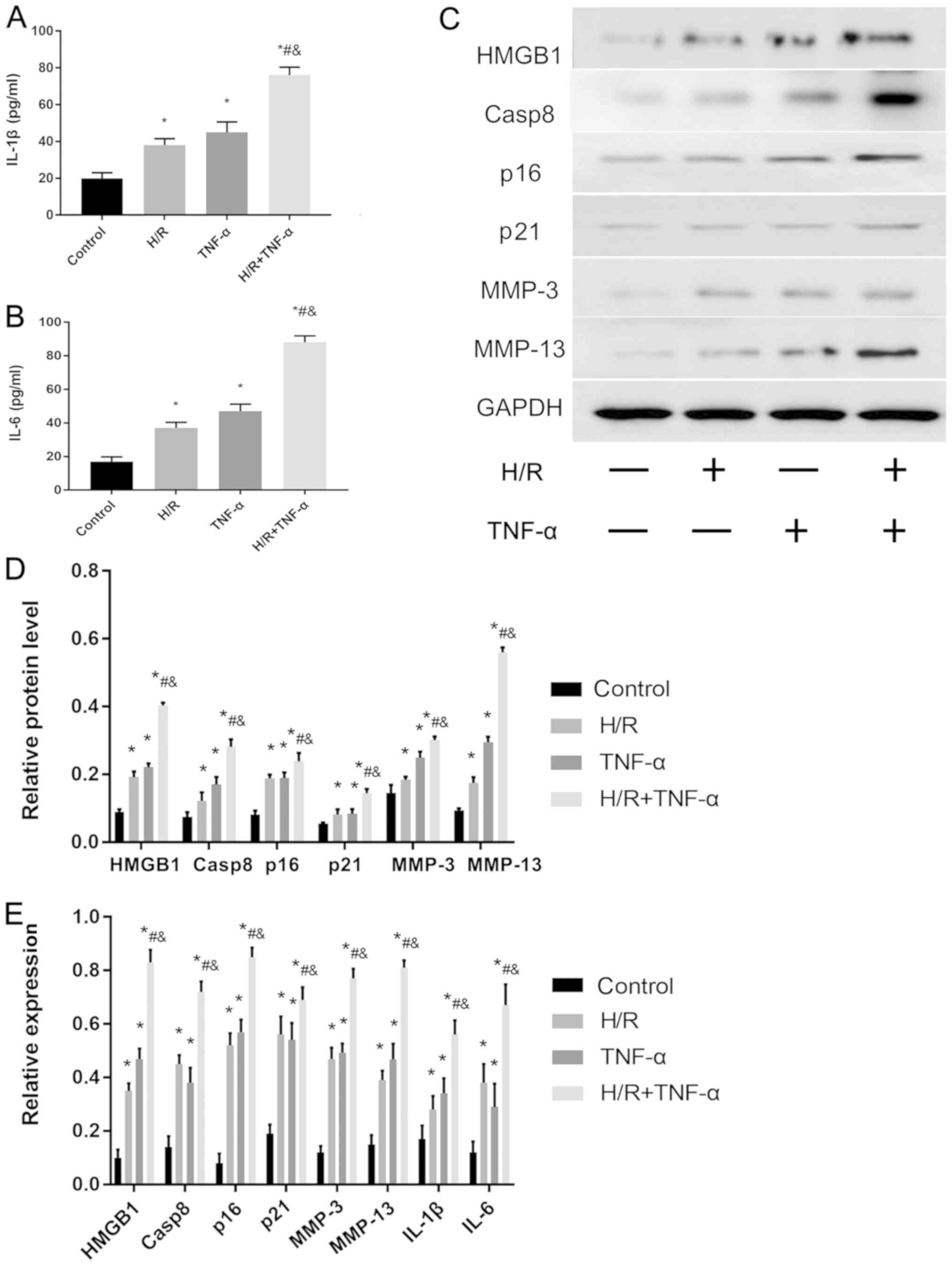 | Figure 3.Effect of H/R on the expression
levels of senescence-associated secretory phenotype-associated
proteins in FLS. Protein expression levels of (A) IL-1β and (B)
IL-6 protein levels in the cell supernatant were assessed and
quantified. (C) Western blotting results indicated the expression
levels of HMGB1, Casp8, p16, p21, MMP-3 and MMP-13. (D)
Quantitative analysis of HMGB1, Casp8, p16, p21, MMP-3 and MMP-13
protein expression levels. (E) mRNA expression levels of HMGB1,
Casp8, p16, p21, MMP-3, MMP-13, IL-6 and IL-1β were assessed by
reverse transcription-quantitative PCR. Data are presented as the
mean ± SD. *P<0.05 vs. Control group; #P<0.05 vs.
H/R group; &P<0.05 vs. TNF-α group. N=3. FLS,
fibroblast-like synoviocytes; H/R, hypoxia/reoxygenation; TNF-α,
tumor necrosis factor α; IL, interleukin; MMP, matrix
metalloproteinase; HMGB1, high mobility group box 1; Casp8,
caspase-8. |
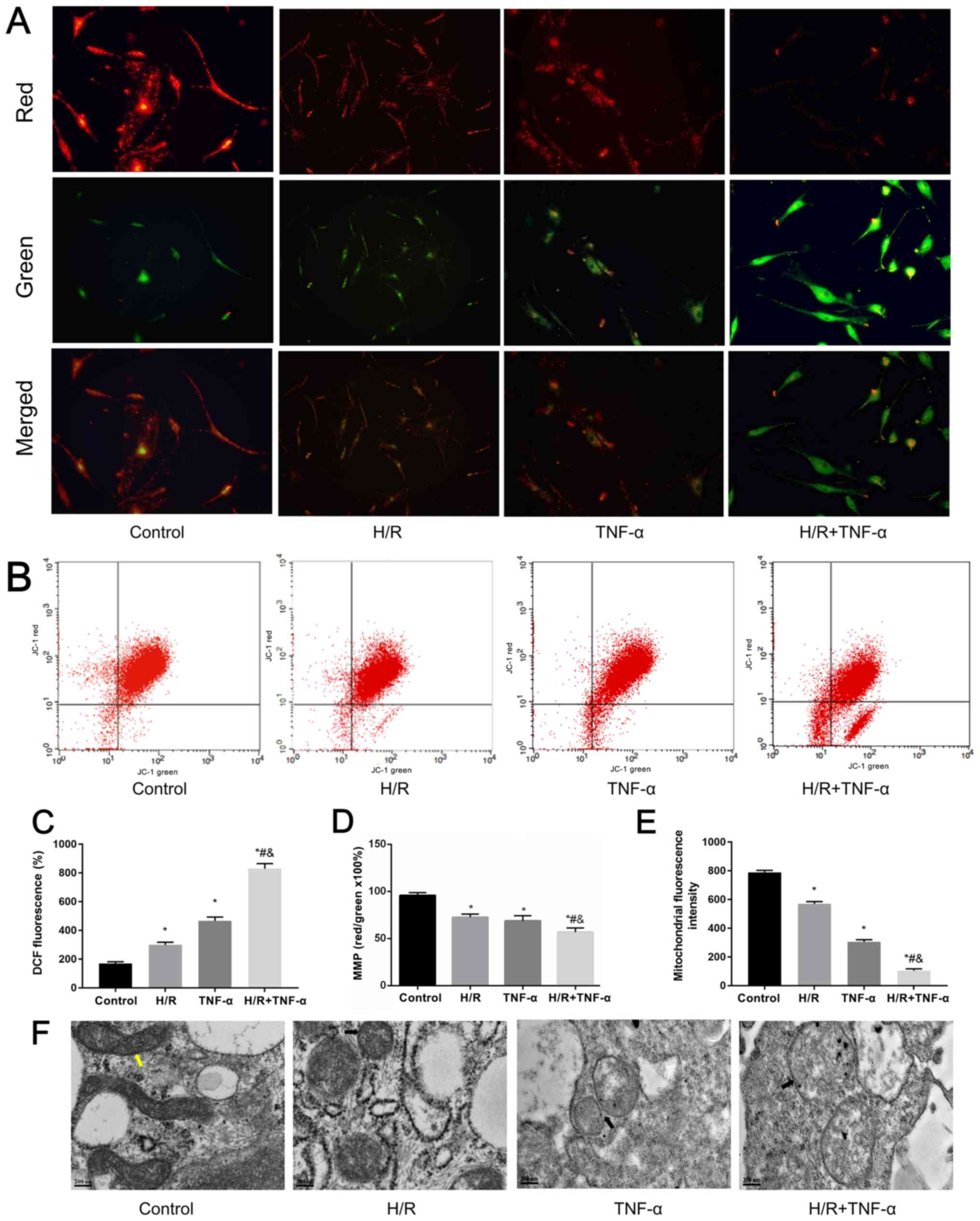
Mitochondrial dysfunction induced by
H/R and its manifestations
H/R induces morphological changes and mitochondrial
dysfunction in FLS (14).
Furthermore, overproduction of ROS is the initial sign of cellular
inflammation (35). The loss of
ΔΨm and the opening of the MPTP often cause excessive accumulation
of ROS, which significantly increases ROS levels in cells (36). Therefore, a JC-1 MMP assay kit was
used to detect changes in ΔΨm in FLS under H/R conditions, where
the red/green ratio decreased as the level of ΔΨm decreased
(Fig. 4A). The changes in ΔΨm were
detected by flow cytometry (Fig. 4B
and D). It was demonstrated that H/R decreased ΔΨm. Moreover,
flow cytometric results indicated that the MPTP in mitochondria was
opened under H/R conditions in FLS (Fig. 4E). Flow cytometry was also used to
estimate intracellular ROS levels by detecting the relative
increase of fluorescent units after DCFH-DA staining. It was
determined that H/R significantly increased intracellular ROS
levels (Fig. 4C). In addition,
changes in mitochondrial ultrastructure were observed under TEM;
FLS morphology of the control group was normal as the mitochondria
were rod-shaped and the mitochondrial ridges were clearly visible.
Compared with the control group, small and short rod-shaped
mitochondria were observed in the H/R group. Moreover, mitochondria
ridges were ruptured and mitochondria became swollen in the TNF-α
and TNF-α + H/R groups (Fig.
4F).
H/R-activates the JNK pathway to
reduce Bcl-2 expression, and increase JNK, p53 and Bax expression
levels to promote senescence
Western blotting was used to assess the protein
expression levels of JNK, p53, Bcl-2 and Bax (Fig. 5A and B). In addition, the mRNA
expression levels of JNK, p53, Bax and Bcl-2 were detected by
RT-qPCR (Fig. 5C). It was
demonstrated that, compared with the control group, H/R
intervention decreased the expression level of Bcl-2, and enhanced
the expression levels of JNK, p53 and Bax. Therefore, the present
results indicated that H/R may be important in activating the JNK
signaling pathway to promote senescence.
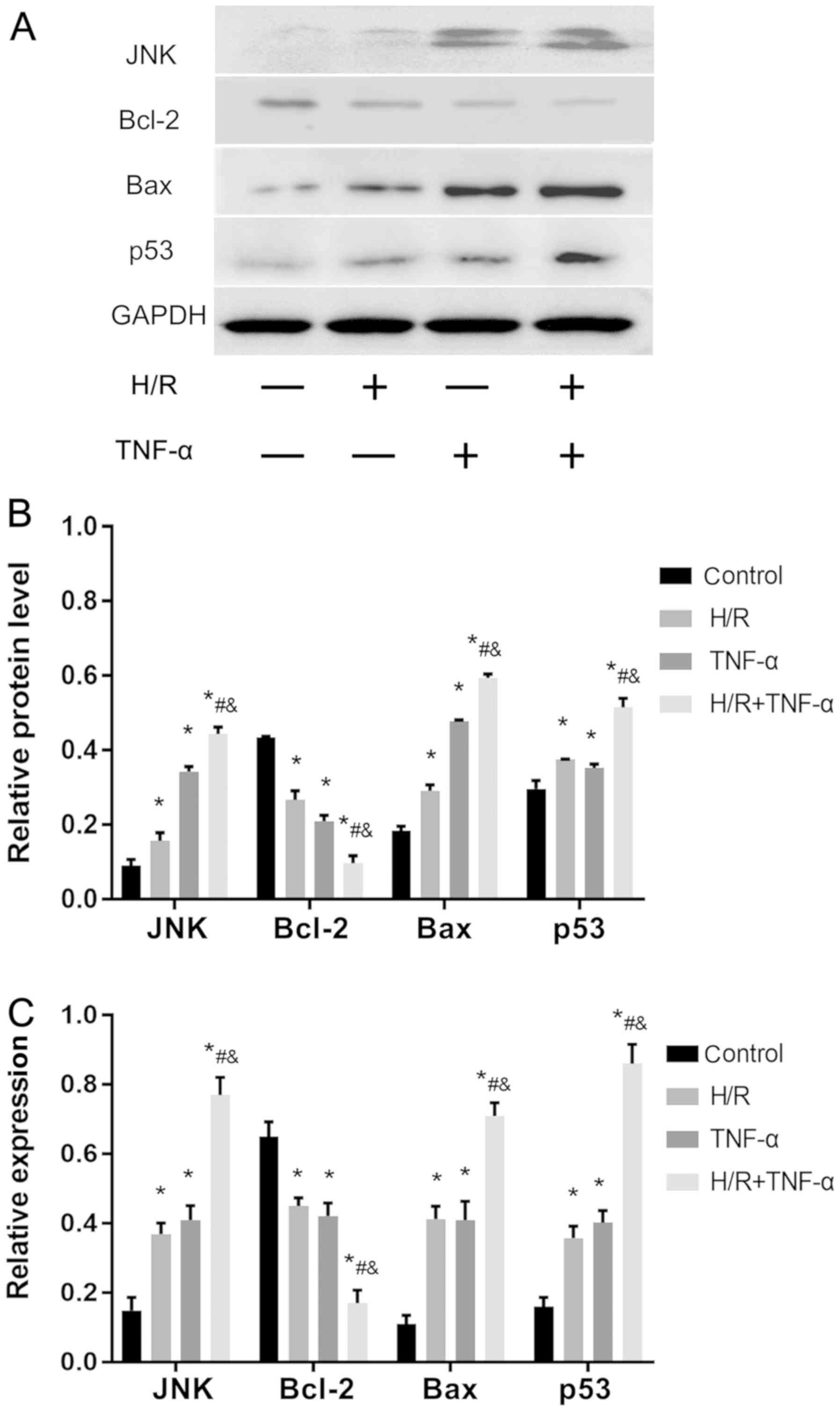 | Figure 5.H/R activates the JNK signaling
pathway in FLS, promotes Bax and p53 expression level and decreases
Bcl-2. (A) Protein expression levels of JNK, Bax, p53 and Bcl-2 in
FLS. (B) Quantitative analysis of JNK, Bax, p53 and Bcl-2 protein
expression levels. (C) mRNA expression levels of JNK, Bax, p53 and
Bcl-2. Data are presented as the mean ± SD. *P<0.05 vs. Control
group; #P<0.05 vs. H/R group;
&P<0.05 vs. TNF-α group. N=3. FLS,
fibroblast-like synoviocytes; H/R, hypoxia/reoxygenation; TNF-α,
tumor necrosis factor α. |
Discussion
The pathological structures involved in the
development of OA include both the cartilage and the entire joint
(2), among which the synovial
membrane has an abundant blood supply that is oxygen-dependent
(37,38). Therefore, due to the
characteristics of the synovial blood supply, inflammatory
cytokines in the circulation can accumulate in the synovium more
frequently and earlier than chondrocytes (39).
Joint motion is an H/R process (40). Excessive H/R damage can cause
damage to synovial cells that are sensitive to changes in oxygen
concentration (15). Synovial cell
stimulation induced by oxidative stress or excessive mechanical
action primarily results in a SASP phenotype, via which the
synovial membrane enters a ‘stress-induced senescence state’ that
leads to the development of OA (41,42).
The present study established a repetitive H/R environment to
simulate mild hypoxia. Moreover, to improve the simulated OA
environment and the experimental integrity, samples were pretreated
with TNF-α prior to H/R induction, in order to detect the changes
of inflammatory cytokines and SASP in FLS in a fluctuating oxygen
environment.
Increases in inflammatory factors in the blood of
the aging population is a marker of chronic, low-grade systemic
inflammation related to senescence, and plays an important role in
the occurrence and development of chronic senescence diseases
(43). H/R can create a chronic
anoxic environment in the joint organs (13,16).
Furthermore, anoxia increases ROS levels in FLS, which leads to
mitochondrial damage and induces the production of inflammatory
factors (8). In addition, by
inhibiting the synthesis of respiratory complexes III and IV in
synovial cells, mitochondrial dysfunction is worsened and
inflammation is escalated (44).
OA synovial cells are special; in the context of excessive ROS,
these cells may exhibit additional senescent characteristics
(45). Moreover, SnCs secrete
various factors to alter the SASP phenotype, which destroy the
microenvironment of neighboring tissues, thus spreading the
imbalance and dysfunction to the whole tissue, and ultimately
triggering the onset of OA (46).
The present results indicated that the pathological characteristics
of synovial tissue in OA were clear. It was revealed that the
expression levels of IL-1β, p16 and MMP-13 in the OA group and H/R
intervention group exhibited a similar trend, and these secretions
were greatly increased compared with the control group. In
addition, the expression levels of SASP-related proteins and
inflammatory factors were higher in cells cultured under a H/R
environment, and the proportion of SA-β-gal-positive FLS was also
higher compared with the control. It was demonstrated that TNF-α
pre-intervention before H/R cοuld further increase the expression
levels of SASP-related proteins and the proportion of
SA-β-gal-positive FLS in a H/R environment. Collectively, the
present results suggested that H/R induced FLS dysfunction,
promoted SASP phenotype changes and consequently promoted
senescence, and thus may play an important role in the development
of OA lesions.
Mitochondria are the main target of oxidative stress
and damages to mitochondrion in FLS in a hypoxic environment may be
caused by repeated H/R (14).
Furthermore, mitochondria are downstream of ROS stimulation
(47). Continuous mitochondrial
damage can increase ROS levels, and ROS stimulation can cause
mitochondrial DNA damage, inflammatory cytokine release and DNA
mutations (48). Moreover, certain
inflammatory cytokines may cause permanent changes in the
mitochondrial complex, thus these factors may lead to further
dysfunction of mitochondrial activity (49). The accumulation of ROS acts as an
intracellular signal, which locally produces oxygen free radicals
that participate in intracellular and extracellular oxidative
damage response, stimulate synovial cells, trigger the inflammatory
response and lead to cartilage destruction by altering the
extracellular matrix of cartilage tissue (16). ROS, ATP production, oxygen uptake
and membrane potential, are important to maintain the mitochondrial
redox balance (50). Accumulation
of senescent mitochondria are associated with decreased membrane
potential and simultaneously increased ROS levels, which contribute
to organelle dysfunction (51).
ROS can affect numerous cell signaling pathways, such as MAPK
(52). The JNK signaling pathway
is an important member of the MAPK family, which are widely
involved in cellular processes, including cell proliferation,
metabolism and DNA damage repair (53). JNK is located in the cytoplasm, and
once the transcription factor is activated it will combine with the
cis-acting element to increase gene expression, and it also
has an important relationship with inflammation (54,55).
It has been previously reported that senescence in numerous tissues
involves the JNK signaling pathway (30,56).
Moreover, JNK activity is an indicator of mitochondrial dysfunction
in the liver of an elderly individual (57). Similarly, due to its role in stress
responses, JNK is important in the development of age-related
muscular degeneration (58). The
present study revealed that H/R enhanced oxidative stress and
increased ROS in FLS. Furthermore, as a result of JNK signaling
pathway activation in FLS, it was demonstrated that the expression
levels of p53 and Bax were promoted, and Bcl-2 was decreased.
Therefore, this process may be an important contributor to the
development of OA.
Previous studies investigating synovial-caused OA
pathologies have several limitations (59–63):
i) Cells are cultured only under normal oxygen partial pressure,
thus physiological or pathological oxygen fluctuations are not
considered; and ii) the effect on hypoxic tolerance of chondrocytes
by fluctuating oxygen have been studied, while mitochondrial damage
and respiratory dysfunction in oxygen dependent FLS have not been
investigated in depth. Therefore, due to physiological and
pathological H/R processes in joint organs, previous studies may
have a certain level of bias (15). Therefore, synovial pathology is
likely to be initiated prior to primary articular cartilage damage.
The present study established a cycling hypoxic environment to
observe the expression and effects of inflammatory mediators
between senescence and OA in FLS (22). On the basis of previous studies on
the pathophysiological mechanism of OA (64,65),
the importance of synovial injury induced by H/R injury in the
pathogenesis of OA was emphasized in the present study. It was
revealed that H/R could damage the mitochondria of synovial cells,
and subsequently upregulate the response of joint tissue structure
to inflammatory factors and oxidative stress, resulting in great
number of SnCs. Mitochondrial dysfunction and increased ROS
production in SnCs can induce DNA damage in adjacent proliferating
cells (33). In addition, SnCs
secrete pro-inflammatory factors to alter SASP in synovial cells,
destroy the tissue microenvironment, activate the JNK pathway, and
promote cell senescence and apoptosis (30). Therefore, even if only a small
proportion of SnCs exist, these can lead to the imbalance and
dysfunction of the entire organ. Furthermore, these interactions
escalate cell destruction and inflammation, further damaging
dysfunctional chondrocytes and creating an irreversible vicious
cycle, which eventually leads to advanced OA lesions (20).
In conclusion, the present results revealed that H/R
can stimulate FLS to release inflammatory cytokines and increase
the proportion of cells with senescent characters. Furthermore, the
secretion of SASP-related and catabolic factors were elevated,
together with dysfunction of mitochondria, by H/R intervention.
This effect may be via the JNK signaling pathway, which will lead
to synovium senescence. Therefore, the present results indicated
that H/R may increase TNF-α-induced synovitis, and play an
important role in inflammation-induction pathogenesis of OA. Thus,
future studies should be performed to aid in the understanding of
the role of H/R in the pathogenesis of OA.
Acknowledgements
The authors would like to acknowledge the help from
Ms. Qiong Ding, Dr Pengchen Yu, Ms. Yingxia Jin and Ms. Lina Zhou
(Central Laboratory, Renmin Hospital of Wuhan University) in
assisting with the FCM analyses.
Funding
The present study was supported by The National
Natural Science Foundation of China (grant no. 81171760) and The
Natural Science Foundation of Hubei Province (grant no.
ZRMS2017000057).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YZ and HL designed the research study. SZ, WC, GH,
JL and MC performed the research and analyzed the data. YZ, HL and
WC contributed the reagents or tools, and wrote the main manuscript
text. All authors contributed to revising the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was conducted in strict in
accordance with the Helsinki Declaration. This project had approval
from both the Wuhan University and Renmin Hospital of Wuhan
University (Wuhan, China). All patients participating in the
experiment have signed informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Xia B, Di Chen, Zhang J, Hu S, Jin H and
Tong P: Osteoarthritis pathogenesis: A review of molecular
mechanisms. Calcif Tissue Int. 95:495–505. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bijlsma JW, Berenbaum F and Lafeber FP:
Osteoarthritis: An update with relevance for clinical practice.
Lancet. 377:2115–2126. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Loeser RF, Goldring SR, Scanzello CR and
Goldring MB: Osteoarthritis: A disease of the joint as an organ.
Arthritis Rheum. 64:1697–1707. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Scanzello CR and Goldring SR: The role of
synovitis in osteoarthritis pathogenesis. Bone. 51:249–257. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Charlier E, Deroyer C, Ciregia F, Malaise
O, Neuville S, Plener Z, Malaise M and de Seny D: Chondrocyte
dedifferentiation and osteoarthritis (OA). Biochem Pharmacol.
165:49–65. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nakamura H, Shimamura S, Yasuda S, Kono M,
Kono M, Fujieda Y, Kato M, Oku K, Bohgaki T, Shimizu T, et al:
Ectopic RASGRP2 (CalDAG-GEFI) expression in rheumatoid synovium
contributes to the development of destructive arthritis. Ann Rheum
Dis. 77:1765–1772. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chubinskaya S, Frank BS, Michalska M,
Kumar B, Merrihew CA, Thonar EJ, Lenz ME, Otten L, Rueger DC and
Block JA: Osteogenic protein 1 in synovial fluid from patients with
rheumatoid arthritis or osteoarthritis: Relationship with disease
and levels of hyaluronan and antigenic keratan sulfate. Arthritis
Res Ther. 8:R732006. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yin S, Zhang L, Ding L, Huang Z, Xu B, Li
X, Wang P and Mao J: Transient receptor potential ankyrin 1 (trpa1)
mediates il-1β-induced apoptosis in rat chondrocytes via calcium
overload and mitochondrial dysfunction. J Inflamm (Lond).
15:272018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Del RM MJ, Izquierdo E, Caja S, Usategui
A, Santiago B, Galindo M and Pablos JL: Human inflammatory synovial
fibroblasts induce enhanced myeloid cell recruitment and
angiogenesis through a hypoxia-inducible transcription factor
1alpha/vascular endothelial growth factor-mediated pathway in
immunodeficient mice. Arthritis Rheum. 60:2926–2934. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Riis RG, Gudbergsen H, Simonsen O,
Henriksen M, Al-Mashkur N, Eld M, Petersen KK, Kubassova O, Bay
Jensen AC, Damm J, et al: The association between histological,
macroscopic and magnetic resonance imaging assessed synovitis in
end-stage knee osteoarthritis: A cross-sectional study.
Osteoarthritis Cartilage. 25:272–280. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang Y, Liu Y, Fan Z, Liu D, Wang F and
Zhou Y: IGFBP2 enhances adipogenic differentiation potentials of
mesenchymal stem cells from Wharton's jelly of the umbilical cord
via JNK and Akt signaling pathways. PLoS One. 12:e1841822017.
|
|
12
|
Mathy-Hartert M, Burton S, Deby-Dupont G,
Devel P, Reginster JY and Henrotin Y: Influence of oxygen tension
on nitric oxide and prostaglandin E2 synthesis by bovine
chondrocytes. Osteoarthritis Cartilage. 13:74–79. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
June RK, Liu-Bryan R, Long F and Griffin
TM: Emerging role of metabolic signaling in synovial joint
remodeling and osteoarthritis. J Orthop Res. 34:2048–2058. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhou S, Wen H, Cai W, Zhang Y and Li H:
Effect of hypoxia/reoxygenation on the biological effect of IGF
system and the inflammatory mediators in cultured synoviocytes.
Biochem Biophys Res Commun. 508:17–24. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chenevier-Gobeaux C, Simonneau C,
Lemarechal H, Bonnefont-Rousselot D, Poiraudeau S, Rannou F,
Ekindjian OG, Anract P and Borderie D: Effect of
hypoxia/reoxygenation on the cytokine-induced production of nitric
oxide and superoxide anion in cultured osteoarthritic synoviocytes.
Osteoarthritis Cartilage. 21:874–881. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Grishko VI, Ho R, Wilson GL and Pearsall
AW IV: Diminished mitochondrial DNA integrity and repair capacity
in OA chondrocytes. Osteoarthritis Cartilage. 17:107–113. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Toussaint O, Dumont P, Dierick JF, Pascal
T, Frippiat C, Chainiaux F, Magalhaes JP, Eliaers F and Remacle J:
Stress-induced premature senescence as alternative toxicological
method for testing the long-term effects of molecules under
development in the industry. Biogerontology. 1:179–183. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Campisi J: Senescent cells, tumor
suppression, and organismal aging: Good citizens, bad neighbors.
Cell. 120:513–522. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Campisi J: Aging, cellular senescence, and
cancer. Annu Rev Physiol. 75:685–705. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Peilin W, Songsong T, Chengyu Z, Zhi C,
Chunhui M, Yinxian Y, Lei Z, Min M, Zongyi W, Mengkai Y, et al:
Directed elimination of senescent cells attenuates development of
osteoarthritis by inhibition of c-IAP and XIAP. Biochim Biophys
Acta Mol Basis Dis. 1865:2618–2632. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Koobatian MT, Liang MS, Swartz DD and
Andreadis ST: Differential effects of culture senescence and
mechanical stimulation on the proliferation and leiomyogenic
differentiation of MSC from different sources: Implications for
engineering vascular grafts. Tissue Eng Part A. 21:1364–1375. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Biniecka M, Kennedy A, Fearon U, Ng CT,
Veale DJ and O'Sullivan JN: Oxidative damage in synovial tissue is
associated with in vivo hypoxic status in the arthritic joint. Ann
Rheum Dis. 69:1172–1178. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Krizhanovsky V, Yon M, Dickins RA, Hearn
S, Simon J, Miething C, Yee H, Zender L and Lowe SW: Senescence of
activated stellate cells limits liver fibrosis. Cell. 134:657–667.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Baker DJ, Wijshake T, Tchkonia T,
LeBrasseur NK, Childs BG, van de Sluis B, Kirkland JL and van
Deursen JM: Clearance of p16Ink4a-positive senescent cells delays
ageing-associated disorders. Nature. 479:232–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Acosta JC, Banito A, Wuestefeld T,
Georgilis A, Janich P, Morton JP, Athineos D, Kang TW, Lasitschka
F, Andrulis M, et al: A complex secretory program orchestrated by
the inflammasome controls paracrine senescence. Nat Cell Biol.
15:978–990. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Basu S, Rajakaruna S, Reyes B, Van
Bockstaele E and Menko AS: Suppression of MAPK/JNK-MTORC1 signaling
leads to premature loss of organelles and nuclei by autophagy
during terminal differentiation of lens fiber cells. Autophagy.
10:1193–1211. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang X, Hunter DJ, Jin X and Ding C: The
importance of synovial inflammation in osteoarthritis: Current
evidence from imaging assessments and clinical trials.
Osteoarthritis Cartilage. 26:165–174. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yao Y, Chen R, Ying C, Zhang G, Rui T and
Tao A: Interleukin-33 attenuates doxorubicin-induced cardiomyocyte
apoptosis through suppression of ASK1/JNK signaling pathway.
Biochem Biophys Res Commun. 493:1288–1295. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kyriakis JM and Avruch J: Mammalian
mitogen-activated protein kinase signal transduction pathways
activated by stress and inflammation. Physiol Rev. 81:807–869.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yang LW, Song M, Li YL, Liu YP, Liu C, Han
L, Wang ZH, Zhang W, Xing YQ and Zhong M: L-Carnitine inhibits the
senescence-associated secretory phenotype of aging adipose tissue
by JNK/p53 pathway. Biogerontology. 20:203–211. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Das M, Jiang F, Sluss HK, Zhang C, Shokat
KM, Flavell RA and Davis RJ: Suppression of p53-dependent
senescence by the JNK signal transduction pathway. Proc Natl Acad
Sci USA. 104:15759–15764. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lanna A, Gomes DC, Muller-Durovic B,
McDonnell T, Escors D, Gilroy DW, Lee JH, Karin M and Akbar AN: A
sestrin-dependent Erk-Jnk-p38 MAPK activation complex inhibits
immunity during aging. Nat Immunol. 18:354–363. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nelson G, Wordsworth J, Wang C, Jurk D,
Lawless C, Martin-Ruiz C and von Zglinicki T: A senescent cell
bystander effect: Senescence-induced senescence. Aging Cell.
11:345–349. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xia G, Wang X, Sun H, Qin Y and Fu M:
Carnosic acid (CA) attenuates collagen-induced arthritis in db/db
mice via inflammation suppression by regulating ROS-dependent p38
pathway. Free Radic Biol Med. 108:418–432. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xin R, Pan YL, Wang Y, Wang SY, Wang R,
Xia B, Qin RN, Fu Y and Wu YH: Nickel-refining fumes induce NLRP3
activation dependent on mitochondrial damage and ROS production in
Beas-2B cells. Arch Biochem Biophys. 676:1081482019. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kim JH, Jeon J, Shin M, Won Y, Lee M, Kwak
JS, Lee G, Rhee J, Ryu JH, Chun CH and Chun JS: Regulation of the
catabolic cascade in osteoarthritis by the zinc-ZIP8-MTF1 axis.
Cell. 156:730–743. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
de Lange-Brokaar BJ, Ioan-Facsinay A, van
Osch GJ, Zuurmond AM, Schoones J, Toes RE, Huizinga TW and
Kloppenburg M: Synovial inflammation, immune cells and their
cytokines in osteoarthritis: A review. Osteoarthritis Cartilage.
20:1484–1499. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mathiessen A and Conaghan PG: Synovitis in
osteoarthritis: Current understanding with therapeutic
implications. Arthritis Res Ther. 19:182017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Schneider N, Mouithys-Mickalad AL, Lejeune
JP, Deby-Dupont GP, Hoebeke M and Serteyn DA: Synoviocytes, not
chondrocytes, release free radicals after cycles of
anoxia/re-oxygenation. Biochem Biophys Res Commun. 334:669–673.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Harper ME, Bevilacqua L, Hagopian K,
Weindruch R and Ramsey JJ: Ageing, oxidative stress, and
mitochondrial uncoupling. Acta Physiol Scand. 182:321–331. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Musumeci G, Szychlinska MA and Mobasheri
A: Age-related degeneration of articular cartilage in the
pathogenesis of osteoarthritis: Molecular markers of senescent
chondrocytes. Histol Histopathol. 30:1–12. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Schulz E, Wenzel P, Munzel T and Daiber A:
Mitochondrial redox signaling: Interaction of mitochondrial
reactive oxygen species with other sources of oxidative stress.
Antioxid Redox Signal. 20:308–324. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cillero-Pastor B, Martin MA, Arenas J,
López-Armada MJ and Blanco FJ: Effect of nitric oxide on
mitochondrial activity of human synovial cells. BMC Musculoskelet
Disord. 12:422011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lepetsos P and Papavassiliou AG:
ROS/oxidative stress signaling in osteoarthritis. Biochim Biophys
Acta. 1862:576–591. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Passos JF, Nelson G, Wang C, Richter T,
Simillion C, Proctor CJ, Miwa S, Olijslagers S, Hallinan J, Wipat
A, et al: Feedback between p21 and reactive oxygen production is
necessary for cell senescence. Mol Syst Biol. 6:3472010. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Strandberg TE and Tilvis RS: C-reactive
protein, cardiovascular risk factors, and mortality in a
prospective study in the elderly. Arterioscler Thromb Vasc Biol.
20:1057–1060. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kaarniranta K, Pawlowska E, Szczepanska J,
Jablkowska A and Blasiak J: Role of Mitochondrial DNA Damage in
ROS-mediated pathogenesis of age-related macular degeneration
(AMD). Int J Mol Sci. 20(pii): E23742019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Shay JW and Wright WE: Hayflick, his
limit, and cellular ageing. Nat Rev Mol Cell Biol. 1:72–76. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Dalle PP, Nelson G, Otten EG, Korolchuk
VI, Kirkwood TB, von Zglinicki T and Shanley DP: Dynamic modelling
of pathways to cellular senescence reveals strategies for targeted
interventions. PLoS Comput Biol. 10:e10037282014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Dhawan P and Richmond A: A novel NF-kappa
B-inducing kinase-MAPK signaling pathway up-regulates NF-kappa B
activity in melanoma cells. J Biol Chem. 277:7920–7928. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Schattauer SS, Bedini A, Summers F,
Reilly-Treat A, Andrews MM, Land BB and Chavkin C: Reactive oxygen
species (ROS) generation is stimulated by κ opioid receptor
activation through phosphorylated c-Jun N-terminal kinase and
inhibited by p38 mitogen-activated protein kinase (MAPK)
activation. J Biol Chem. 294:16884–16896. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Craig R, Larkin A, Mingo AM, Thuerauf DJ,
Andrews C, McDonough PM and Glembotski CC: p38 MAPK and NF-kappa B
collaborate to induce interleukin-6 gene expression and release.
Evidence for a cytoprotective autocrine signaling pathway in a
cardiac myocyte model system. J Biol Chem. 275:23814–23824. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lories RJ, Derese I, Luyten FP and de Vlam
K: Activation of nuclear factor kappa B and mitogen activated
protein kinases in psoriatic arthritis before and after etanercept
treatment. Clin Exp Rheumatol. 26:96–102. 2008.PubMed/NCBI
|
|
55
|
Hsieh CC, Rosenblatt JI and
Papaconstantinou J: Age-associated changes in SAPK/JNK and p38 MAPK
signaling in response to the generation of ROS by 3-nitropropionic
acid. Mech Ageing Dev. 124:733–746. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kanatsu-Shinohara M, Yamamoto T, Toh H,
Kazuki Y, Kazuki K, Imoto J, Ikeo K, Oshima M, Shirahige K, Iwama
A, et al: Aging of spermatogonial stem cells by Jnk-mediated
glycolysis activation. Proc Natl Acad Sci USA. 116:16404–16409.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Ge HX, Zou FM, Li Y, Liu AM and Tu M: JNK
pathway in osteoarthritis: Pathological and therapeutic aspects. J
Recept Signal Transduct Res. 37:431–436. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Twumasi-Boateng K, Wang TW, Tsai L, Lee
KH, Salehpour A, Bhat S, Tan MW and Shapira M: An age-dependent
reversal in the protective capacities of JNK signaling shortens
Caenorhabditis elegans lifespan. Aging Cell. 11:659–667. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Olivotto E, Merli G, Assirelli E, Cavallo
C, Belluzzi E, Ramonda R, Favero M, Filardo G, Roffi A, Kon E and
Grigolo B: Cultures of a human synovial cell line to evaluate
platelet-rich plasma and hyaluronic acid effects. J Tissue Eng
Regen Med. 12:1835–1842. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Huhtakangas JA, Veijola J, Turunen S,
Karjalainen A, Valkealahti M, Nousiainen T, Yli-Luukko S,
Vuolteenaho O and Lehenkari P: 1,25(OH)2D3
and calcipotriol, its hypocalcemic analog, exert a long-lasting
anti-inflammatory and anti-proliferative effect in synoviocytes
cultured from patients with rheumatoid arthritis and
osteoarthritis. J Steroid Biochem Mol Biol. 173:13–22. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhang L, Zhang L, Huang Z, Xing R, Li X,
Yin S, Mao J, Zhang N, Mei W, Ding L and Wang P: Increased HIF-1α
in Knee Osteoarthritis aggravate synovial fibrosis via
fibroblast-like synoviocyte pyroptosis. Oxid Med Cell Longev.
2019:63265172019.PubMed/NCBI
|
|
62
|
Hsu HC, Chang WM, Wu JY, Huang CC, Lu FJ,
Chuang YW, Chang PJ, Chen KH, Hong CZ, Yeh RH, et al: Folate
deficiency triggered apoptosis of synoviocytes: Role of
overproduction of reactive oxygen species generated via NADPH
Oxidase/Mitochondrial Complex II and calcium perturbation. PLoS
One. 11:e1464402016.
|
|
63
|
Gale AL, Mammone RM, Dodson ME, Linardi RL
and Ortved KF: The effect of hypoxia on chondrogenesis of equine
synovial membrane-derived and bone marrow-derived mesenchymal stem
cells. BMC Vet Res. 15:2012019. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Fernandes JC, Martel-Pelletier J and
Pelletier JP: The role of cytokines in osteoarthritis
pathophysiology. Biorheology. 39:237–246. 2002.PubMed/NCBI
|
|
65
|
Park J, Lee J, Kim KI, Lee J, Jang S, Choi
HT, Son Y, Kim HJ, Woo EJ, Lee E and Oh TI: A pathophysiological
validation of collagenase II-induced biochemical osteoarthritis
animal model in rabbit. Tissue Eng Regen Med. 15:437–444. 2018.
View Article : Google Scholar : PubMed/NCBI
|