Introduction
Telomeres have been characterized as molecular
clocks, playing a pivotal role in cell division arrest. These
chromatin structures, composed of tandem DNA sequence repeats
coated by the sheltering protein complex, are formed at chromosomal
termini as protective caps against DNA degradation and
recombination to ensure genomic stability and integrity (1). Each cell division leads to shorter
telomeres due to the inability of DNA polymerase to fully replicate
the end part of the lagging strand of DNA, described as the end
replication problem. Telomere length (TL) loss can be compensated
by telomerase, which functions as a reverse transcriptase, adding
telomeric repeats at the end of chromosomes. The telomerase complex
consists of a catalytic unit denominated as telomerase reverse
transcriptase (TERT) and the telomerase RNA component (2). This ribonucleoprotein polymerase is
abundantly expressed in highly proliferating cells, such as stem
cells; however, its expression in somatic cells is reduced or
absent (3). In cells with a low
telomerase activity, telomeres continue to shorten, and when they
reach a critically short length, cells undergo apoptosis or cell
senescence. Beyond this critical length, excessive telomere
shortening can lead to chromosomal fusions and genomic instability
that increase the risk of cancer tumorigenesis through genomic
alterations. Ultimately, cells need to reactivate telomerase to
become immortal during tumor progression (4). The replicative past and potential of
cells can be assessed through TL and telomerase activity estimation
methods, focusing on short telomere load and the rate of telomere
shortening that reflects cellular aging (5).
Telomere attrition and cellular senescence are
identified as major contributors to the physiological process of
aging (6). Furthermore, increased
aging and age-related diseases have been shown to be associated
with the disruption of telomere homeostasis, which has been mainly
attributed to increased levels of oxidative stress and inflammation
(7). To date, cumulative oxidative
stress and inflammation have been strongly associated with
age-related TL shortening (8).
Research on the associations of telomere shortening with age has
revealed that the inhibition of telomere attrition via telomere
gene therapy in mice results in delayed physical aging (9). By contrast, it has been suggested
that a short leukocyte TL is an independent risk factor that
accounts for functional decline in elderly European populations
(10). Furthermore, critically
short telomeres have been shown to be associated with higher
mortality rates and shorter lifespans (11).
The human lifespan has been considerably extended,
leading to an increase in the elderly populations globally,
stressing the need to develop ‘healthy aging’ strategies that will
prevent and treat aging-related diseases (12). Research focusing on identifying
effective natural or synthetic compounds with anti-aging properties
has intensified in recent years (13). Specifically, there is increasing
evidence to indicate that bioactive compounds, including nutrients
and vitamins present in foods and supplements with antioxidant and
anti-inflammatory properties can ameliorate age-related phenotypes,
including telomere shortening (14,15).
Of note, telomerase activity can be positively modified by natural
compounds, such as Astragalus membranaceus plant extracts,
cycloastragenol (CAG), curcumin C3 and vitamins, among others,
which enable telomerase activation (13). Moreover, in a previous study, the
authors demonstrated that a formulation containing Centella
asiatica (C. asiatica) extract markedly enhanced
telomerase activity in human peripheral blood mononuclear cells
(PBMCs) in vitro (16).
Notably, the demonstrated increase in telomerase activity was the
highest reported in vitro, to date, at least to the best of
our knowledge (16). Another study
also demonstrated that the administration of the formulation
containing C. asiatica extract, vitamin C, zinc and vitamin
D3 for a period of 3 months restored TERT expression and increased
telomerase activity in the brains of middle-aged rats. Furthermore,
a structural reversibility effect was observed in the brains of
middle-aged rats treated with the formulation, close to the
differentiation of the grey matter from a young control group
(17). Supporting evidence was
provided from a complementary behavioral study of the same treated
rats, where supplementation with the formulation improved the
locomotor activity and decreased stress significantly in a
dose-dependent manner in aged rats (12).
The present study aimed to examine the effects of
the nutraceutical formulation containing C. asiatica
extract, vitamin C, zinc and vitamin D3 on telomerase activity and
TL using a murine model. The obtained results shed light onto the
potential association of supplement administration with the abating
of the aging process.
Materials and methods
Animal model
All animals were obtained from the University of
Medicine and Pharmacy of Craiova Animal House, Craiova Romania,
authorization no. 76/20.04.2016. The Ethical Committee of the
University of Medicine and Pharmacy of Craiova, Craiova, Romania,
approved the animal study (protocol no. 102/23.09.2019). All the
procedures were according to the European directives for animal
experiments (E.U. Directive 2010/63/E.U.as amended by Regulation
E.U.2019/1010). A total of 24 Sprague-Dawley (CD-SD) male rats
divided into four groups were used in the present study. Of the
total 24 animals, 18 rats were 18 months old [body weight (bw)
range, 500–580 g]. The middle-aged animals were randomly assigned
to three groups as follows: Old group 1, old group 2 and old
control group, with 6 rats in each group. The remaining 6 rats were
3 months old, and were denominated as the young control group. Male
rats were selected over female animals to reduce the variability of
responses due to potentially asynchronous estrous cycles (18). All animals were acclimatized to
their new housing conditions for 2 weeks before the commencement of
the study. The rats were kept in cages containing 2 or 3 animals
during the study, under standard conditions with a constant
temperature (22±1°C) and humidity (50±10%) and a 12-h dark/light
cycle, receiving free access to standard animal feed and tap water.
The study animals received the Reverse™ (Natural Doctor S.A.)
supplement treatments presented in Table I for 3 months and were evaluated
daily for signs of morbidity and mortality. At the end of the
experiment (3 months), the animals received 5% sevoflurane
anesthesia via nasal administration and were monitored until they
were fully anesthetized. The rats were then sacrificed by
exsanguination from the abdominal aorta for the analysis. As stated
in current regulatory testing guidelines, when animals are
evidently in pain, exhibiting signs of severe and enduring
distress, or characterized as moribund, they were humanely
euthanized rather than allowing them to survive to the end of the
scheduled study period. However, in the present study, no animal
death was observed (19,20). The animals were examined daily for
any walking impairments that prevented access to food or water,
excessive weight loss and emaciation, a lack of physical or mental
alertness, difficulty breathing, and an inability to stand upright
for long periods of time.
 | Table I.Treatments administered to the animal
groups for 3 months daily and sampling time points (pre-and
post-treatment). |
Table I.
Treatments administered to the animal
groups for 3 months daily and sampling time points (pre-and
post-treatment).
| Animal groups | Old group 1
(n=6) | Old group 2
(n=6) | Old control group
(n=6) | Young control group
(n=6) |
|---|
| Treatments | 1 capsule per
kg/body weight of Reverse™ supplement | 2 capsules per
kg/body weight of Reverse™ supplement | 1.5 ml corn
oil | 1.5 ml corn
oil |
| Age (months)
(pre-treatment) | 18 | 18 | 18 | 3 |
| Age (months)
(post-treatment) | 21 | 21 | 21 | 6 |
Treatment dose selection and
administration
Reverse™ (Natural Doctor S.A.), notified as a food
supplement (Notification no. 6704/21 1 2020) at the Greek National
Organization for Medicines was administered at a dose of 1 or 2
capsules/kg bw/day. The supplement contains 9 mg C. asiatica
extract (consisting of a >90% high purity single chemical
entity, as assessed using high-performance liquid chromatography
and gas chromatography) (21),
vitamin C (200 mg as magnesium ascorbate), zinc (5 mg as zinc
citrate) and vitamin D3 (50 µg as cholecalciferol) per capsule.
The doses administered to the rats were extrapolated
from the human doses using the correction factor (fc) ratio for the
species used and the safety factor value for humans. The fc is
estimated as the ratio of the mean bw (kg) of the species used, to
the species' body surface area (m2), according to the
Food and Drug Administration guidelines being 6 for rats and 37 for
humans, respectively. The fc ratio for drug dose conversion from
rats to humans corresponds to a value of 6.2. The safety factor
(fs) value for converting rat doses to humans is 1043. The
reference bw for humans is 60 kg (22), which means the dose for humans (dH)
is calculated to 1/60 capsules per kg bw for a single dose of 1
capsule and 2/60 capsules per kg bw for a single dose of 2
capsules. Based on the information provided above, the dose in rats
(dR) was calculated as follows: dR=dH × fcratio × fs (Equation
1).
Based on Equation 1, the old group 1 receiving the
equivalent of the 1 capsule/day human dose, was administered
dR=1/60 capsules × 6.2×10=1.03 capsule per kg/bw. The Old Group 2,
receiving the equivalent of the 2 capsules/day human dose, was
issued with dR=2/60 capsule × 6.2×10=2.06 capsule per kg/bw.
Prior to treatment administration, the content of
the capsules was suspended in corn oil, widely used as an inert
suspension agent for non-water-soluble drugs in animal experiments
(23), as a stock suspension with
1 capsule/1 ml corn oil concentration or 2 capsules/1 ml corn oil.
Each rat received the equivalent of 1 capsule per kg/bw or 2
capsules per kg/bw, once per day at the same time for 3 months
(Table I). The respective
equivalent dose calculated according to the bw of each animal was
diluted with corn oil until the final volume of 1.5 ml and
administered by gavage.
The dose administered was within clinical
recommendations relative to bw and similar to other animal studies
evaluating the effects of vitamin C (24,25),
vitamin D3 (25-hydroxyvitamin D) (26,27)
and zinc (28). Specifically, as
regards vitamin D3, the administered dose per capsule was below the
upper limit and was safely administered without medical
supervision. Concerning the safety levels of the ingested dose for
vitamin D, the most common concern is the risk of hypercalcemia,
which may be evoked when the serum 25-hydroxyvitamin D levels
exceed 700 ng/ml, which is >7-fold higher than the levels of
sufficiency (29). Cholecalciferol
was selected over ergocalciferol since ergocalciferol is less
stable and less potent than cholecalciferol, characterized as the
only vitamin D form suitable for supplementation (21). For C. asiatica, there is no
established clinical recommendation, at least to the best of our
knowledge. The dose administered in the present study was markedly
lower than the levels reported in the literature (30).
Sample collection and preparation for
quantitative-fluorescent in situ hybridization (Q-FISH)
Peripheral blood from the tail vein (2 ml) was
collected from each rat at the start of the experiment
(pre-treatment) and after 3 months (post-treatment). The collected
heparinized blood cultured in 50-ml falcon tubes with RPMI-1640
culture medium supplemented with 10% fetal bovine serum (FBS), 1%
L-glutamine, 1% penicillin, phytohemagglutinin 100 µg/ml and
streptomycin was stimulated for 72 h in a CO2 incubator
with phytohemagglutinin. The cells were incubated at 37°C with 10
µg/ml colcemid for 2 h to obtain metaphases, followed by KCl
hypotonic shock, and then were harvested and fixed in
methanol/acetic acid (3:1). All reagents were obtained from
MilliporeSigma.
Quantitative-fluorescent in situ
hybridization (Q-FISH)
Several drops of a fixative solution containing
cells from each culture were applied to three slides. Slides with
the fixative solution containing cells were thereupon dried and
incubated on a hot plate (55°C) overnight prior to hybridization.
Fluorescence staining for telomeric DNA was performed using the
peptide nucleic acid (PNA) fluorescent probe Cy3-(C3TA2)3 obtained
from Panagene Inc. Each slide was hybridized with 20 µl of 60%
formamide (MilliporeSigma) and 0.3 mg/ml Cy3 (C3TA2)3 PNA probe
(HLB Panagene) diluted in 20 mM Tris pH 7.4 (MilliporeSigma) and
covered using a coverslip (76×26 mm). Cell DNA was denatured by
heat treatment for 10 min at 85°C. Following hybridization for 2 h
at room temperature, the slides were washed at 55°C with a washing
solution containing 0.05% Tween-20 (MilliporeSigma) (2×10 min). The
chromosome preparations were counterstained with
4′,6-diamino-2-phenylindole (DAPI) (Thermo Fisher Scientific, Inc.)
(0.5 µl/ml SSC) at room temperature for 20 min and then washed with
SSC solution (MilliporeSigma) (2×2 min). The slides were air-dried
and covered using mounting media and a coverslip before proceeding
with image acquisition.
Image analysis
The analysis of FISH Images from metaphase cells was
performed using a Leica TCS Sp8 inverted laser scanning spectral
confocal microscope (Leica Microsystems GmbH). For each slide, a
total of 20–30 optical sections were captured. Metaphase spread
images were captured using a 63X objective and a charge-coupled
device camera at a 1024×1024 pixel resolution and 8-bit depth with
a step of 250 nm. A 405-nm laser was used for the excitation and
detection of DAPI for nuclear staining. In comparison, a 568-nm
laser was utilized for the excitation and detection of Alexa Fluor
647 for telomere staining. Exposure and gain settings remained
unaltered between captures to avoid differences between the
replicates. For each slide, >10 different scanned images were
obtained. The maximum projections and deconvolution of the images
were performed using Leica Q-FISH software (Leica Application
Suite-Advanced Fluorescence version 3.1.3 for Leica TCS SP8; Leica
Microsystems, Inc.). The quantification of telomere fluorescence
intensity was performed twice by two different researchers using
ImageJ software 1.8.0 (National Institutes of Health). A total of
two calibration steps were undertaken to ensure the accuracy of
telomere fluorescence intensity quantification. First, images of
fluorescent beads (orange beads, size 0.2 µm, Thermo Fisher
Scientific, Inc.) were obtained before the samples. Fluorescence
intensities of the beads (obtained using ImageJ software) were used
to normalize and adjust the lamp intensity and alignment prior to
sample analysis. Second, for the determination of the telomere
fluorescence intensity the L5178Y-S cells (cat. no. 93050408;
European Collection of Authenticated Cell Cultures) were included
as a calibration standard in each experiment. The mean value of
L5178Y-S cell telomere fluorescence intensities was used to
normalize the respective values of samples between slides. Telomere
fluorescence values were converted into kb according to the
telomere fluorescence intensity of L5178Y-S cells that have an
established TL of ~7 kb (31).
Telomerase activity assay
Blood samples for telomerase activity assay were
collected from each animal at the beginning of the experiment
(pre-treatment) and after 3 months (post-treatment). PBMCs were
isolated from the blood samples using Ficoll-Hypaque
(MilliporeSigma) gradient centrifugation at 277 × g for 5 min at
room temperature in 15-ml falcon tubes. The extracted PBMCs were
cultured in DMEM (F0455, Biochrom AG) supplemented with 10% FBS
(10500-064, Invitrogen; Thermo Fisher Scientific, Inc.) with 4 mM
glutamine (XCT1715, Biosera) and antibiotics (gentamycin;
15710-049, Gibco; Thermo Fisher Scientific, Inc.; and 100 U/ml
penicillin/streptomycin; LMA4118, Biosera). Telomerase activity was
quantified using a TeloTAGGG telomerase PCRELISA TRAP kit
(MilliporeSigma), based on the telomeric repeat amplification
protocol, as previously described (16). All measures for each condition were
performed in triplicate.
Statistical analysis
The obtained data on TL were implemented into the
specialized spreadsheet (BIOTEL 2.4) (32) to produce TL statistics, including
percentiles, medians and telomere distribution. The TL data were
further statistically analyzed using the IBM SPSS Statistics 24.0
package (IBM Corp.), while data for telomerase activity were
processed using GraphPad Prism version 5.0 for Windows (GraphPad
Software, Inc., www.graphpad.com). TL data are presented in two forms
as follows: i) As the mean with 95% confidence intervals (CIs); and
ii) as the median and interquartile range (IQR). Parametric
(independent samples t-test) and non-parametric tests (Mann-Whitney
test) for independent samples were applied to compare the means of
two independent groups or, additionally/alternatively, the
distribution of TL data. A similar approach using one-way ANOVA
(parametric) and Kruskal-Wallis (non-parametric) for independent
samples was applied to compare means for more than two separate
groups or, additionally/alternatively, the distribution of TL data.
These analyses (ANOVA and Kruskal-Wallis) was followed by post hoc
analyses using the Bonferroni adjusted t-test, SNK, Dunnett's for
ANOVA, and Dunn's test for Kruskal-Wallis.
Differences in paired design (pre-and post-treatment
measures) were examined using a t-test for paired samples
(parametric) or a Wilcoxon ranked sum test (non-parametric).
Boxplots and scatterplots were applied for the graphical
representation of the data. A value of P<0.05 was set as a
significant statistical hypothesis.
Results
Effects of the nutraceutical
formulation on the TL of the rats
Initially, the present study evaluated TL variations
during the 3-month study period in the animals that did not receive
treatment. These animals demonstrated a significant reduction in
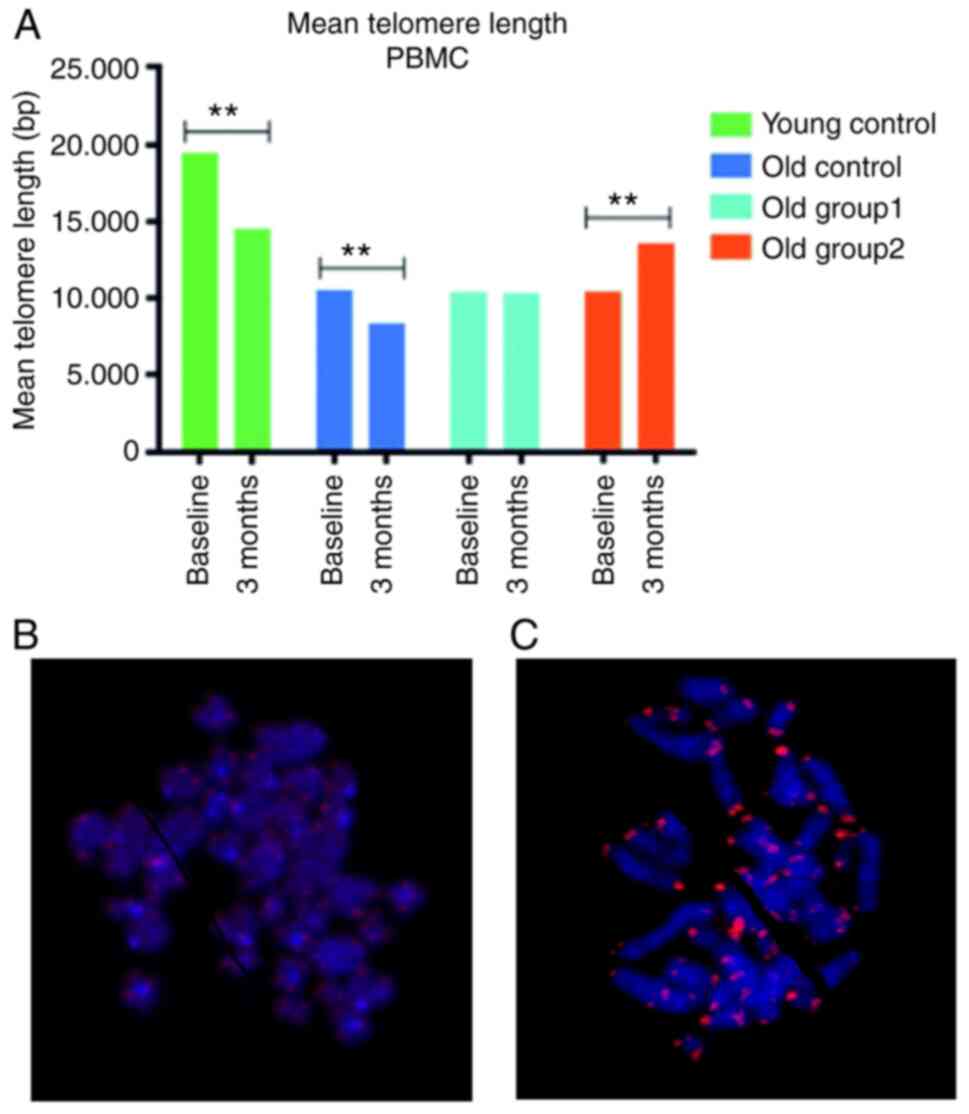
the mean and median TL during this period (Table II). Specifically, the
pre-treatment mean TL of the young control group of 19.468 bp
(19.146–19.789) was significantly longer compared to the TL mean
value [14.535 bp (14.329–14.741)] at the study termination.
Similarly, the pre-treatment mean TL values of the old control were
decreased during the 3-month period (Table II).
 | Table II.Descriptive statistics of TL and
comparisons between pre-treatment (baseline). |
Table II.
Descriptive statistics of TL and
comparisons between pre-treatment (baseline).
|
| Young control | Old control | Old group 1 | Old group 2 |
|---|
|
|
|
|
|
|
|---|
| Statistics | Pre | Post | Pre | Post | Pre | Post | Pre | Post |
|---|
| Min | 8.061 | 6.542 | 2.805 | 1.952 | 2.671 | 4.075 | 2.543 | 2.905 |
| 95% LB | 19.146 | 14.329 | 10.294 | 8.127 | 10.240 | 10.010 | 10.203 | 12.755 |
| Mean | 19.468 | 14.535 | 10.569 | 8.420 | 10.472 | 10.369 | 10.469 | 13.600 |
| 95% UB | 19.789 | 14.741 | 10.843 | 8.713 | 10.703 | 10.729 | 10.734 | 14.446 |
| 1st | 16.613 | 12.613 | 7.697 | 5.593 | 8.737 | 7.407 | 8.088 | 7.117 |
| Median | 19.297 | 14.439 | 10.068 | 7.103 | 10.221 | 9.142 | 10.292 | 9.064 |
| 3rd | 22.458 | 16.366 | 13.220 | 10.682 | 12.009 | 12.637 | 12.502 | 19.385 |
| Max | 31.379 | 22.438 | 22.178 | 21.616 | 17.833 | 22.792 | 19.305 | 34.881 |
|
P-valuea | t(587)=82.42,
P<0.001 | t(671)=76.35,
P<0.001 | t(449)=1.45,
P=0.148 | t(449)=−10.08,
P<0.001 |
|
P-valueb | z=−21.00,
P<0,001 | z=−22.46,
P<0.001 | z=−3.14,
P=0.002 | z=−1.179,
P=0.238 |
|
P-valuec |
|
|
|
| Pre: F(2,
1581)=0.19, P=0.825 |
|
|
|
|
|
|
|
| Post: F(2,
1581)=104.93, P<0.001 |
|
|
|
P-valued |
|
|
|
| Pre:
χ2(2)=0.463,
P=0.793 |
|
|
|
|
|
|
|
| Post:
χ2(2)=136.40,
P<0.001 |
|
|
Of note, a mean TL reduction was not observed in the
rats that received the treatment. Specifically, no significant
differences were detected between the mean TL values at
pre-treatment compared to post-treatment in the old group 1 [t
(449)=1.45, P=0.148] (Table II).
In the old group 2, the mean TL values at 13.600 bp (12.755–14.446)
were statistically significantly higher at post-treatment compared
to the baseline TL values of 10.469 bp (10.203–10.734), while the
distribution was not significantly altered (z=−1.179, P=0.238)
(Table II). The mean telomere
length levels at baseline and at 3 months post-treatment are
illustrated in Fig. 1 for the
control groups and the groups that received treatments and
indicative images from Q-FISH analysis referring to old group 2
pre-treatment and to old group 2 post-treatment are also
presented.
One-way ANOVA and the Kruskal-Wallis test were
applied to examine the differences between the baseline TL of the
old group 1, old group 2 and the old control group. No
statistically significant differences were determined for the
baseline mean TL among the old control groups. Similarly, no
significant differences were detected in TL distributions [median
of the overall old control group, 10.068; median old group 1,
10.221; and old group 2, 10.292; χ2(2)=0.463, P=0.793]
(Table II).
On the other hand, significant differences in the
mean and median TLs among these groups were identified
post-treatment (Table II). The
old control group displayed lower mean TL values (8.420,
8.127–8.713 bp) compared to the intervention groups [old group 1,
10.369 bp (10.010–10.729 bp); old group 2, 13.600 bp (12.755–14.446
bp); F (2.1581)=104.93, P<0.001]. The median TL values
post-treatment exhibited similar differences
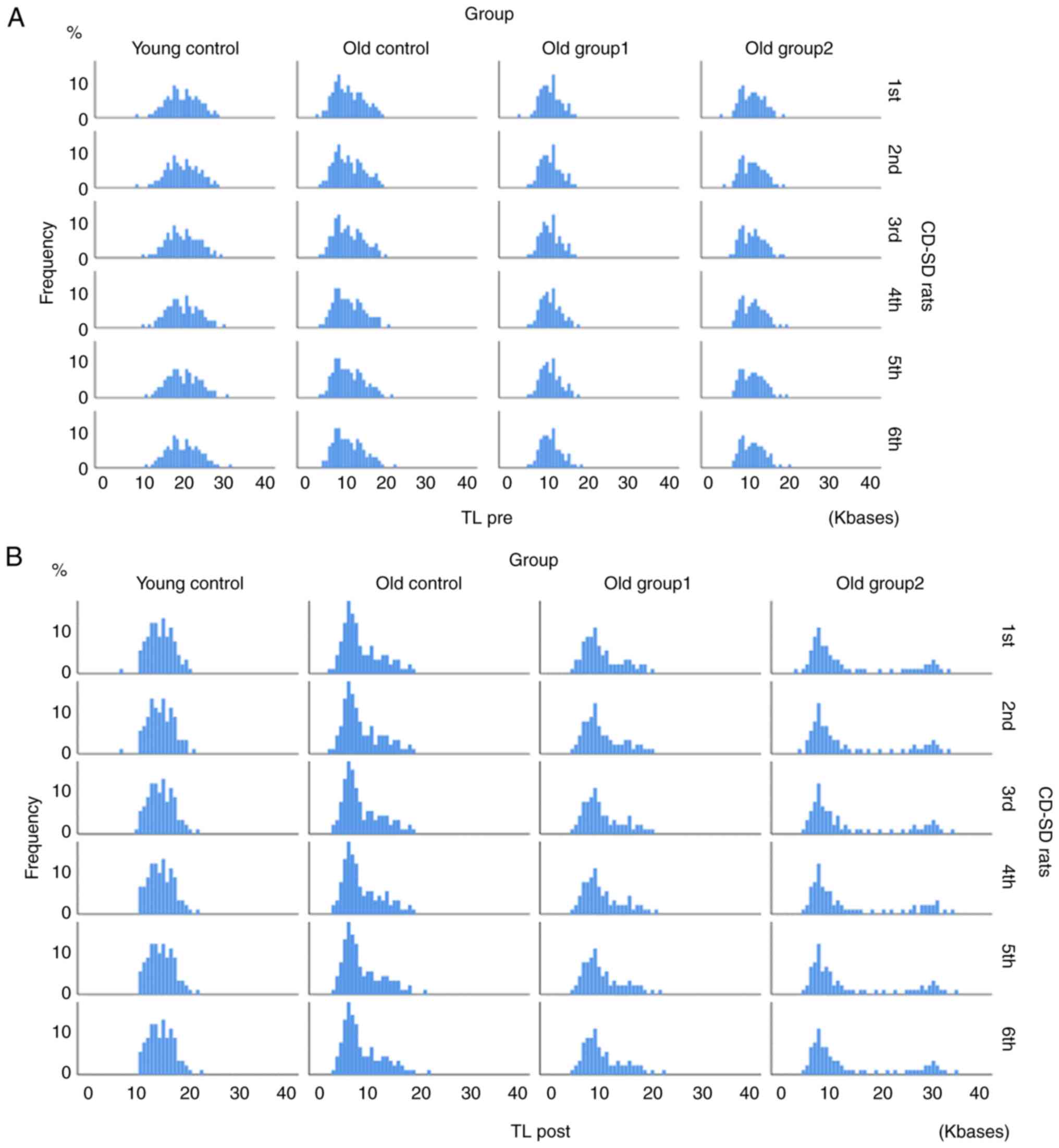
[χ2(2)=136.40, P<0.001]. The TL distribution per
animal at the two set time points (pre-and post-treatment) is
presented in Fig. 2. The results
of the Kolmogorov-Smirnov test for normality are presented in
Table SI.
Independent statistical analysis using the adjusted
t-test (LSD, Bonferroni or Dunnett's) likewise did not detect any
differences in the TL pre-treatment values between the old controls
and the old treatment groups (old group 1 and old group 2). On the
other hand, significant differences in TL values were detected
between the old controls and the rats in old group 1, and old group
1 vs. the rats in old group 2. A detailed post hoc analysis of
pairwise differences is presented in Tables SII and SIII).
Effects of treatment with the
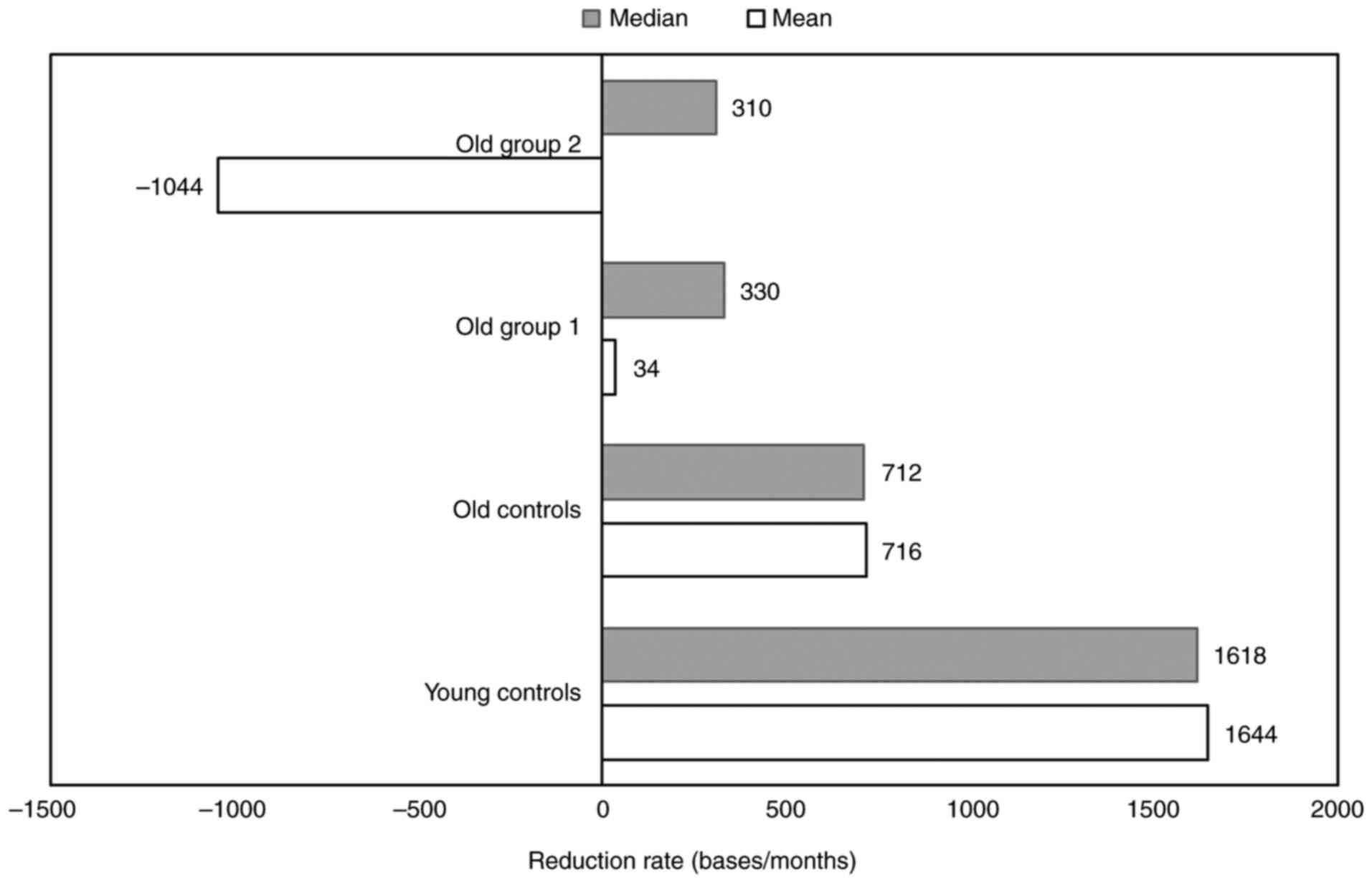
nutraceutical formulation on the TL reduction rate
To evaluate the effects of the nutraceutical
formulation treatment on the rate of TL change, the mean and median
difference in TL per month (TL pre- and TL post-treatment)/month
was calculated and represented for each group (Table SIV). A decrease in the median
difference in TL per month was detected in all the study groups. In
younger rats (untreated young control), the magnitude of the TL
reduction rate of TL values was shown to be relatively high,
whereas the reduction rate of TL values for the old untreated
control group was decreased to ~50% as compared to the reduction
rate of the young controls. Treated with supplements, rats showed a
complex pattern in TL changes. The median TL reduction rate of the
old group 1 and old group 2 was <50% compared to the reduction
rate of the old control group (Fig.
3). Nevertheless, the mean TL reduction rate was low for old
group 1, whereas the TL of old group 2 exhibited a considerable
increase with a mean of −1044 bases/month (Fig. 3).
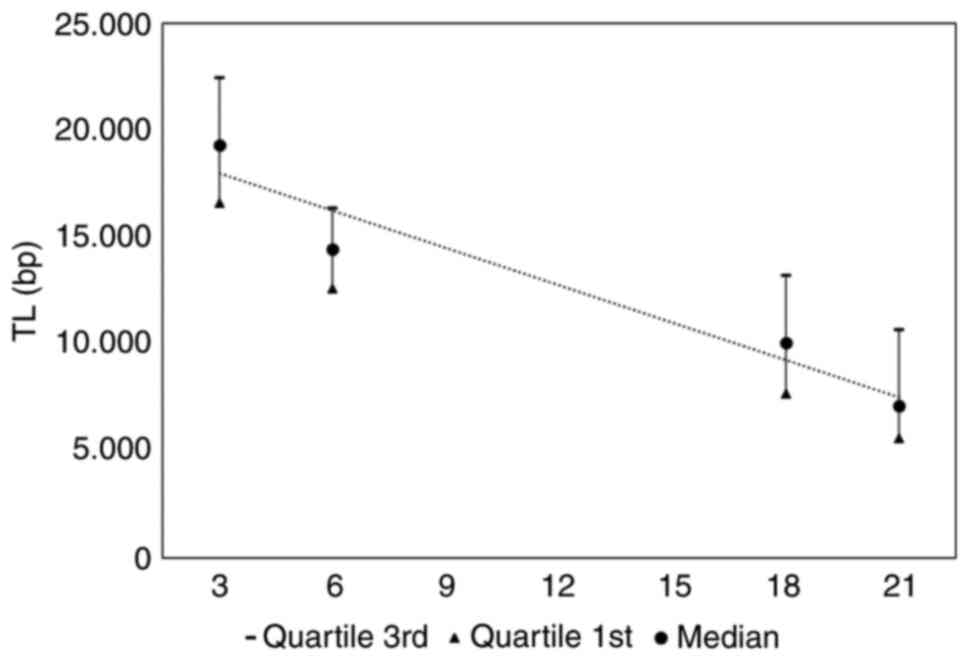
Effects of aging on rat TL
Furthermore, the effects of aging on TL were
evaluated in the old and young rat groups. The median TL and
quartile range values at 3 months (young control pre-), 6 months
(young control post), 18 months (old control pre) and 21 months
(old control post) are presented (Fig.
4). A TL decrement is apparent, and TL vs. time dependence can
be characterized by a linear regression line TL=−1743 * time +
19.700 bp with an R2=0.932.
Telomerase activity
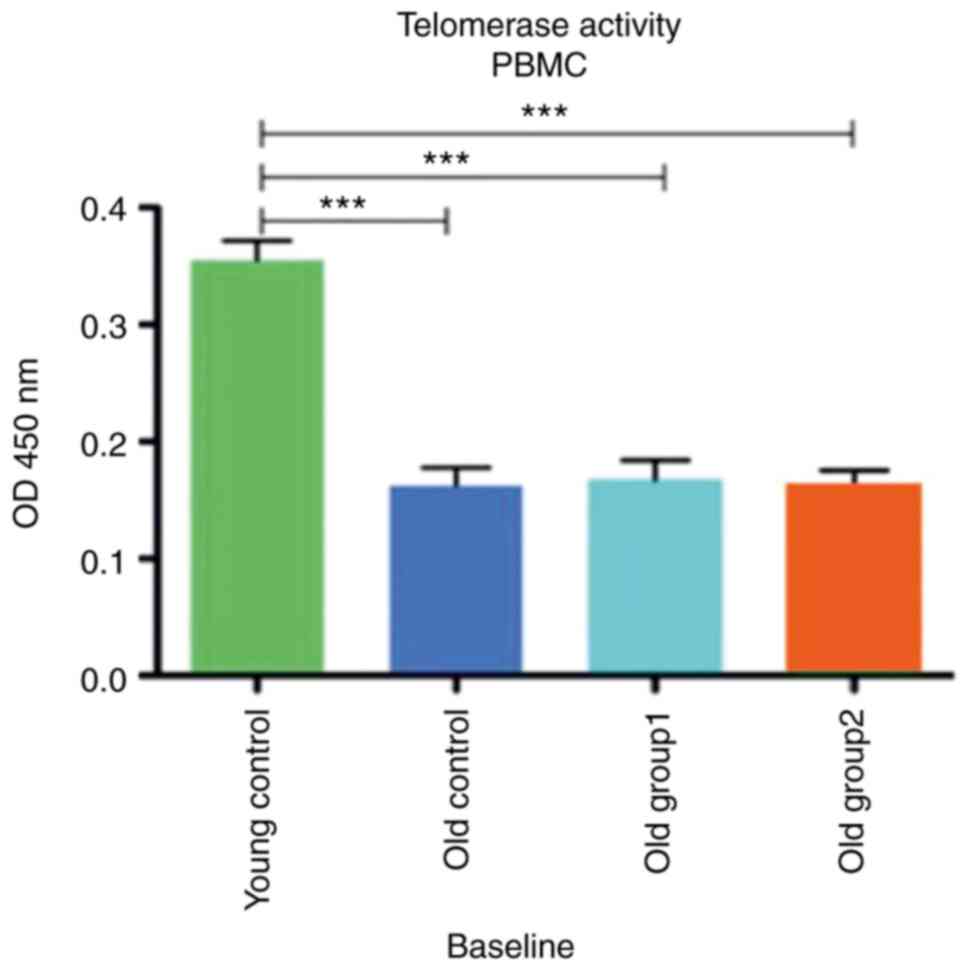
The baseline telomerase activity of the PBMCs
collected from all rat groups measured as fluorescence at 450 nm OD
is presented in Fig. 5. The mean
telomerase activity of the PBMCs was 0.355±0.033 for the young
control group, while the old control group exhibited a mean
activity of 0.163±0.03. Old group 1 and old group 2 exhibited
similar baseline telomerase activities with a mean of 0.168±0.03
and 0.163±0.03, respectively. One-way ANOVA revealed a
statistically significant difference between the young group and
all old groups [(F(3, 12)=37,94, P<0.001)]. Similar results were
obtained with the post-hoc Dunnett's test (t=8.784,
P<0.001).
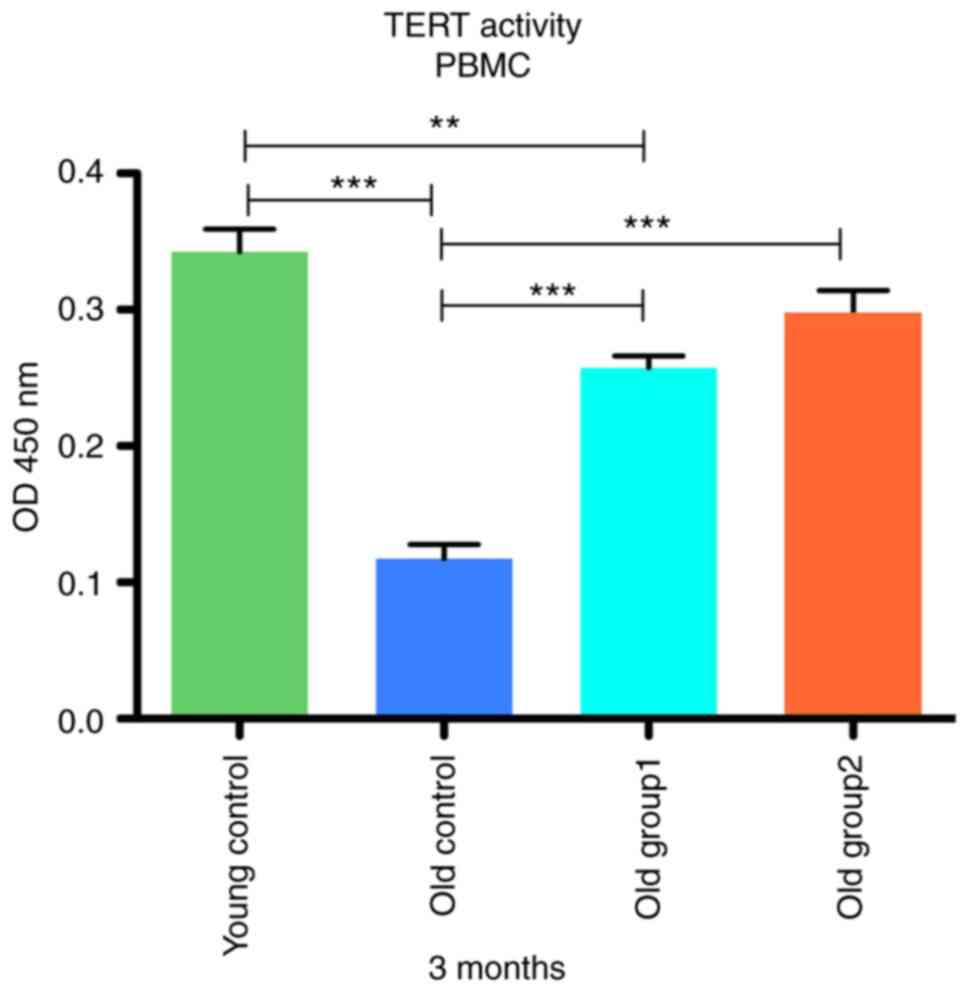
The telomerase activity of the PBMCs form the
control and treated groups at 3 months post-treatment is presented
in Fig. 6. One-way ANOVA revealed
a statistically significant difference between the young control
group (0.343±0.03), old control group (0.118±0.02), treated old
group1 (0.258±0.017) and treated old group2 (0.298±0.033) with an
F(3, 12)=52.17 and a P-value of <0.001. In continuation, the
analysis of telomerase activity values with the Dunnett's test
revealed a significant difference between the old and young
controls (t=11.82, P<0,05), and between old group 1 and the
young controls (t=4.464, P<0.05). Notably, no marked differences
in telomerase activity were detected between old group 2 and the
young controls (t=2.364, P>0.05). Statistically significant
differences at the P<0.001 level were found for old group 1 and
old group 2 vs. the old control CD-SD rats (P>0.001).
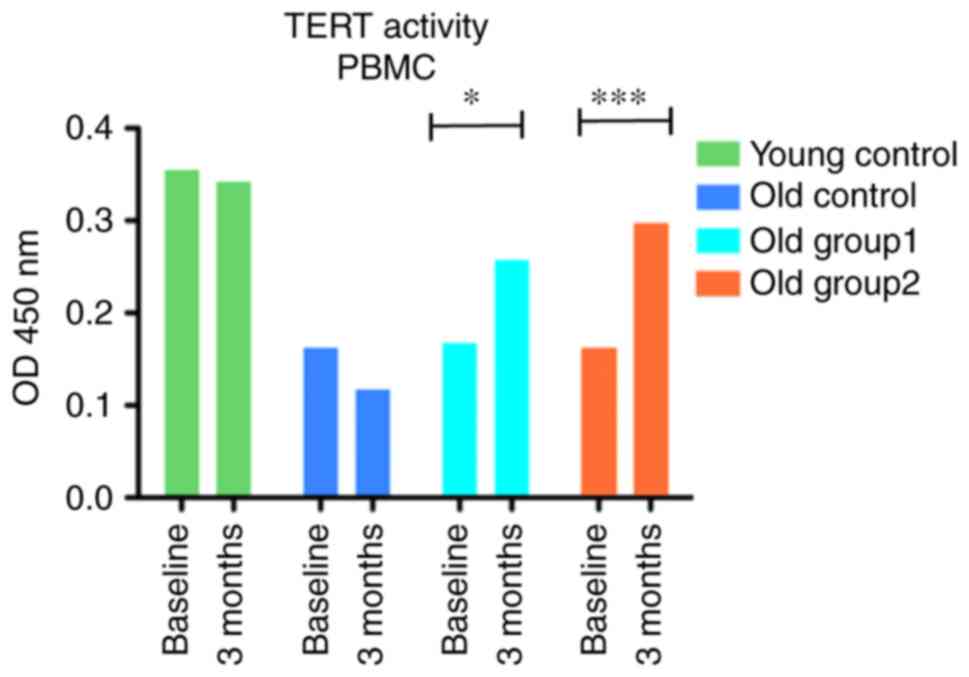
A two-way ANOVA was applied to compare the
telomerase activity of PBMCs expressed at OD450 nm fluorescence
between four groups (two controls and two treated groups) at two
time points (baseline and following 3 months of treatment). The
analysis revealed that treatment × time interaction presented a
significant effect [F(3, 12)=16.72, P=0.007]. The administration of
the C. asiatica-containing supplements induced significant
effects on PBMC telomerase activity with F(3, 12)=69.95,
P<0.001, and F(1, 12)=5.46, P=0.005, respectively (Fig. 7). Furthermore, the Bonferroni post
hoc test revealed differences in telomerase activity in old group 1
CD-SD rats (t=3.657, P<0.05) and old group 2 CD-SD rats
(t=5.485, P<0.001) when comparing the baseline and the levels at
3 months after treatment.
Discussion
Telomere shortening is strongly associated with DNA
damage, oxidative stress and inflammation (1), without omitting various other
factors, including genetic and epigenetic mediators, which affect
telomere length regulation (33).
Natural molecules with demonstrated antioxidant and
anti-inflammatory capacities are being increasingly recognized as
potent agents which can be used to restore telomere attrition for
the improvement of aging-related diseases (34–36).
The effects of natural compounds on TL regulation
have been ascribed to the central role of oxidative stress and
inflammation to telomere shortening and telomerase, suggesting that
nutrients with anti-inflammatory and antioxidant capacity can
reduce cell sensitivity to telomere loss (37). Th authors have previously
demonstrated that the nutraceutical formulation containing C.
asiatica extract, vitamin C (as magnesium ascorbate), zinc (as
zinc citrate) and vitamin D3 (as cholecalciferol) has a beneficial
effect on aging-associated telomerase decline, behavior and brain
morphology change in vivo (17). The present study examined the
potency of the formulation in middle-aged rats and revealed that it
results in telomere shortening attenuation and the activation of
telomerase of PBMCs in a dose-dependent manner. Specifically, the
treatment maintained the rat PBMC TL at the lower dose, whereas the
higher dose increased the median TLs, compared to the pre-treatment
levels. In terms of translating these findings in humans, it is
supported that two capsules of the nutraceutical formulation per
day were more potent in activating telomerase and increasing the
telomere length of PBMCs than 1 capsule. Clinical trials on the
efficacy of the nutraceutical formulation to telomere shortening
and other aging biomarkers are required to further evaluate these
effects.
Data from epidemiological studies and clinical
trials have demonstrated that the increased intake of
micronutrients, such as vitamins D, C, E and A, and dietary fiber
in the context of a balanced diet, e.g., the Mediterranean diet, is
associated with longer telomeres, as reviewed by Galiè et al
(38). In addition, the
consumption of nuts and seeds, a rich source of anti-inflammatory
polyunsaturated fatty acids, has been showon to be associated with
longer telomeres, and improved metabolic and blood lipid profiles,
possibly mediated by an antioxidant effect (39).
The formulation tested herein consisted of nutrients
that have been previously reported to improve aging-related and/or
oxidative stress markers. C. asiatica is a herb utilized in
traditional Chinese medicine for the treatment of various
pathologies (40). The extract of
this herb contains some pentacyclic triterpenoids, mainly
asiaticoside, madecasosside, asiatic acid, madecassic acid and
other components, including centellose and centelloside (41). Notably, the extracts of C.
asiatica have been shown to exert potent antioxidant and
anti-inflammatory effects. Specifically, the oral administration of
C. asiatica has been shown to attenuate inflammatory markers
and enhance the antioxidative function of diabetic rats (42). Moreover, carbon tetrachloride
(CCl4)-induced liver fibrosis has been shown to be ameliorated by
oral treatment with C. asiatica. Specifically, C.
asiatica was shown to decrease CCl4-induced inflammation,
oxidative stress and hepatocyte apoptosis, and to modify Bcl-2/Bax
signaling in the livers of rats (43).
Furthermore, vitamin D is a fat-soluble
micronutrient that participates in critical cellular functions
beyond the absorption of calcium and its deficiency has been
related to numerous inflammatory and aging-associated pathologies,
including hypertension, cognitive decline and cardiometabolic
complications (44). One of its
extraskeletal effects is the regulation of the pace of aging as a
regulator of mitochondrial function, oxidative stress and
senescence (45). The results of
an epidemiological study suggest that vitamin D can reduce the
production of inflammatory mediators, such as C-reactive protein
and interleukin-6, known to mitigate telomere shortening (46). Moreover, it was previously
demonstrated that supplementation with vitamin D for 12 months
improved cognitive function by reducing oxidative stress in older
adults with mild cognitive impairment. This was associated with an
increased TL (47). A separate,
recent epidemiological study suggested that genetic variations in
the gene encoding vitamin D binding protein (GC-rs2282679) were
associated with a low TL, suggesting that the vitamin D status may
affect TL early in life (48).
A cross-sectional study performed on a cohort of
2,483 males demonstrated that 25-hydroxyvitamin D (25(OH)D) and
1,25-dihydroxy vitamin D plasma levels were not associated with a
low TL (49). On the other hand,
higher plasma 25(OH)D levels appear to be associated with longer
telomeres in women, and this association appears to be modulated by
calcium intake (50). There is
additional epidemiological evidence linking high serum levels of
vitamin D with a longer TL. As previously demonstrated, the
treatment of patients undergoing hemodialysis with vitamin D
resulted in a longer peripheral PBMC TL compared to that of
untreated patients (51).
Additionally, epidemiological studies have positively associated
vitamin C intake with a longer TL, and the vitamin's antioxidative
properties have further been associated with abated telomere
shortening of peripheral blood cells (52–54).
There is also in vitro evidence to support the potency of
zinc to decrease the aging of mesenchymal stem cells via an
increase in TL, telomerase expression and activity, with an
additional decrease in the percentage of senescent cells and the
epigenetic modification of the hTERT gene promoter (55).
The present study further demonstrated that the
administration of the nutraceutical formulation containing C.
asiatica extract, vitamin C, zinc and vitamin D3 increased the
telomerase activity of PBMCs in a dose-dependent manner in a rat
model. Specifically, the telomerase activity of medium-aged rats
was reversed to the levels of the young controls at the higher
dose. These findings are in line with those of previous studies
demonstrating the potency of formulation and its naturally
occurring constituents on telomerase activation (17). Moreover, the authors also recently
demonstrated that C. asiatica extract increased telomerase
activity in human PBMCs (16). The
potency of the C. asiatica-containing formulation to
activate telomerase was significantly more pronounced compared to
other multi-nutrient formulations (16). Furthermore, vitamin C treatment has
been found to be capable of mesenchymal stem cell sheet formation
and tissue regeneration by inducing telomerase activity in
vitro (56). In addition,
vitamin C has been shown to activate telomerase in periodontal
ligament stem cells, resulting in their enhanced expression of
extracellular matrix components and stem cell markers (57). Moreover, the treatment of human
pluripotent stem cell-derived cardiomyocytes with vitamin C has
been found to result in the upregulation of human telomerase RNA
(57). Likewise, supplementation
with vitamin D has been demonstrated to significantly enhance the
PBMC telomerase activity in a cohort of overweight African
Americans (58). In a separate
cohort of obese African Americans, vitamin D supplementation was
associated with reduced epigenetic aging (59). Notwithstanding, it has been
highlighted that the combination of nutrients and natural compounds
exerts more significant effects than single compounds (13,16,60).
Thus, it can be hypothesized that the beneficial effects of
treatment on TL and telomerase may be due to the synergy of the
constituents of the supplement. However, further studies are
required to validate this.
The present study has several limitations which
should be mentioned. First, the relatively low number of animals in
the control and treatment groups can be mitigated by increasing
animal numbers in future studies. In addition, the present study
did not assess the putative mechanisms of action. In future
studies, this point can be addressed by examining antioxidant,
anti-inflammatory and various epigenetic markers. Finally, there is
a general limitation in extrapolating results obtained in animal
models to clinical efficacy that can be attributed to internal and
external validity (61). In the
present study, strict measures were taken to ensure internal
validity and control bias. However, the issue of external validity,
including the incapacity of animal models to emulate the complexity
of human conditions and species-related differences, remain. One
approach would be to perform the study with a larger number of
animals exposed to some level of stress, e.g., sleep interruption,
to better mimic the everyday pressures facing the majority of the
human population. Furthermore, there are several differences
between rats, as well as mice and humans regarding telomeres,
telomerase and lifespan. Small animals such as mice and rats are
characterized by longer telomeres and a higher telomerase
expression in various tissues compared to humans, albeit with a
lower lifespan (62). Still, they
are the gold standard in aging research, with numerous reports on
the impact of telomerase activation and underlying anti-aging
mechanisms (63). Collectively, it
was observed that the administration of the nutraceutical
supplement resulted in telomerase activation in both PBMCs and the
brains of rats in accordance with the amelioration of brain
structure and function and the increase in TL. However, additional
studies are required to define the association of these effects,
given that rats have long telomeres, and other non-canonical
pathways of TERT may be involved (64). Finally, epidemiological studies
would also provide a better overview of the putative role of
multi-nutrient supplements on TL and telomerase activity in
humans.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the European Union's ‘Horizon
Europe’ framework programme project name: ‘MAgnetically steerable
wireless Nanodevices for the tarGeted delivery of therapeutIc
agents in any vascular rEgion of the body’ (ANGIE
H2020-EIC-FETPROACT-2019). The study was also partially funded by
the Spin-Off Company of the University of Crete, Toxplus S.A., by
the start-up company LifePlus and by the Special Research Account
of University of Crete (ELKE nos. 3464).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author upon reasonable
request.
Authors' contributions
AT, DT, DAS, DN and AOD conceptualized the study.
AT, DT, AOD, DN and DC were involved in the supervision of the
study. AMB, AOD, DC, ER, ES, EV and PF performed the experiments,
sample treatment and analyses. ER, DN, ES, DT, AMB, EV, PF, AA,
AOD, AT and DAS were involved in the writing and editing of the
manuscript. AA performed the statistical analyses. AT and AA
confirm the authenticity of the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
All animals were obtained from the University of
Medicine and Pharmacy of Craiova Animal House, Craiova Romania,
authorization no. 76/20.04.2016. The Ethical Committee of the
University of Medicine and Pharmacy of Craiova, Craiova, Romania,
approved the animal study (protocol no. 102/23.09.2019). All the
procedures were according to the European directives for animal
experiments (E.U. Directive 2010/63/E.U.as amended by Regulation
E.U.2019/1010).
Patient consent for publication
Not applicable.
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
DT is a scientific advisor for Natural Doctor S.A. The other
authors declare that they have no competing interests.
References
|
1
|
Chakravarti D, LaBella KA and De Pinho RA:
Telomeres: History, health, and hallmarks of aging. Cell.
184:306–322. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Poole JC, Andrews LG and Tollefsbol TO:
Activity, function, and gene regulation of the catalytic subunit of
telomerase (hTERT). Gene. 269:1–12. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Razgonova MP, Zakharenko AM, Golokhvast
KS, Thanasoula M, Sarandi E, Nikolouzakis K, Fragkiadaki P,
Tsoukalas D, Spandidos DA and Tsatsakis A: Telomerase and telomeres
in aging theory and chronographic aging theory (Review). Mol Med
Rep. 22:1679–1694. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Maciejowski J and de Lange T: Telomeres in
cancer: Tumour suppression and genome instability. Nat Rev Mol Cell
Biol. 18:175–186. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hao LY, Armanios M, Strong MA, Karim B,
Feldser DM, Huso D and Greider CW: Short telomeres, even in the
presence of telomerase, limit tissue renewal capacity. Cell.
123:1121–1131. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
López-Otín C, Blasco MA, Partridge L,
Serrano M and Kroemer G: The hallmarks of aging. Cell.
153:1194–1217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fernandes SG, Dsouza R and Khattar E:
External environmental agents influence telomere length and
telomerase activity by modulating internal cellular processes:
Implications in human aging. Environ Toxicol Pharmacol.
85:1036332021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li H, Lin L, Ye S, Li H and Fan J:
Baseline Assessment of nutrient and heavy metal contamination in
the seawater and sediment of Yalujiang Estuary. Mar Pollut Bull.
117:499–506. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Derevyanko A, Whittemore K, Schneider RP,
Jiménez V, Bosch F and Blasco MA: Gene therapy with the TRF1
telomere gene rescues decreased TRF1 levels with aging and prolongs
mouse health span. Aging Cell. 16:1353–1368. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rojas DM, Nilsson A, Ponsot E, Brummer RJ,
Fairweather-Tait S, Jennings A, de Groot LCPGM, Berendsen A,
Pietruszka B, Madej D, et al: Short telomere length is related to
limitations in physical function in elderly European adults. Front
Physiol. 9:11102018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Steenstrup T, Kark JD, Verhulst S,
Thinggaard M, Hjelmborg JVB, Dalgård C, Kyvik KO, Christiansen L,
Mangino M, Spector TD, et al: Telomeres and the natural lifespan
limit in humans. Aging (Albany NY). 9:1130–1142. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tsoukalas D, Zlatian O, Mitroi M, Renieri
E, Tsatsakis A, Izotov BN, Burada F, Sosoi S, Burada E, Buga AM, et
al: A novel nutraceutical formulation can improve motor activity
and decrease the stress level in a murine model of middle-age
animals. J Clin Med. 10:6242021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Selim AM, Nooh MM, El-Sawalhi MM and
Ismail NA: Amelioration of age-related alterations in rat liver:
Effects of curcumin C3 complex, Astragalus membranaceus and
blueberry. Exp Gerontol. 137:1109822020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tsoukalas D, Fragkiadaki P, Docea AO,
Alegakis AK, Sarandi E, Vakonaki E, Salataj E, Kouvidi E, Nikitovic
D, Kovatsi L, et al: Association of nutraceutical supplements with
longer telomere length. Int J Mol Med. 44:218–226. 2019.PubMed/NCBI
|
|
15
|
Renieri E, Vakonaki E, Karzi V,
Fragkiadaki P and Tsatsakis A: Telomere length: Associations with
nutrients and xenobiotics. Toxicological Risk Assessment and
Multi-System Health Impacts from Exposure. Tsatsakis A: 1st
edition. Academic Press; 2021, View Article : Google Scholar
|
|
16
|
Tsoukalas D, Fragkiadaki P, Docea AO,
Alegakis AK, Sarandi E, Thanasoula M, Spandidos DA, Tsatsakis A,
Razgonova MP and Calina D: Discovery of potent telomerase
activators: Unfolding new therapeutic and anti-aging perspectives.
Mol Med Rep. 20:3701–3708. 2019.PubMed/NCBI
|
|
17
|
Tsoukalas D, Buga AM, Docea AO, Sarandi E,
Mitrut R, Renieri E, Spandidos DA, Rogoveanu I, Cercelaru L,
Niculescu M, et al: Reversal of brain aging by targeting
telomerase: A nutraceutical approach. Int J Mol Med. 48:1992021.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zucker I and Beery AK: Males still
dominate animal studies. Nature. 465:6902010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Environmental Protection Agency (EPA), .
Health Effects Test Guidelines OPPTS 870.3700 Prenatal
Developmental Toxicity Study. EPA; Washington, DC: 1998
|
|
20
|
Organization for Economic Co-operation and
Development (OECD), . Guidance Document on the Recognition,
Assessment, and Use of Clinical Signs as Humane Endpoints for
Experimental Animals Used in Safety Evaluation. OECD Series on
Testing and Assessment. No. 19. OECD Publishing; Paris, France:
2002
|
|
21
|
Gad SC, Cassidy CD, Aubert N, Spainhour B
and Robbe H: Nonclinical vehicle use in studies by multiple routes
in multiple species. Int J Toxicol. 25:499–521. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nair A and Jacob S: A simple practice
guide for dose conversion between animals and human. J Basic Clin
Pharm. 7:27–31. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Vieth R: Vitamin D supplementation:
Cholecalciferol, calcifediol, and calcitriol. Eur J Clin Nutr.
74:1493–1497. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Dietary Reference Intakes for Vitamin A,
Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, IronManganese,
Molybdenum, Nickel, Silicon, Vanadium, Zinc: Dietary Reference
Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper,
Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and
Zinc. National Academies Press (US); 2000
|
|
25
|
Sil S, Ghosh T, Gupta P, Ghosh R, Kabir SN
and Roy A: Dual role of vitamin C on the neuroinflammation mediated
neurodegeneration and memory impairments in colchicine induced rat
model of Alzheimer disease. J Mol Neurosci. 60:421–435. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Holick MF, Binkley NC, Bischoff-Ferrari
HA, Gordon CM, Hanley DA, Heaney RP, Murad MH and Weaver CM:
Guidelines for preventing and treating vitamin D deficiency and
insufficiency revisited. J Clin Endocrinol Metabol. 97:1153–1158.
2012. View Article : Google Scholar
|
|
27
|
Williamson L, Hayes A, Hanson ED, Pivonka
P, Sims NA and Gooi JH: High dose dietary vitamin D3 increases bone
mass and strength in mice. Bone Rep. 6:44–50. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dietary Reference Intakes for Vitamin C,
Vitamin E, Selenium, Carotenoids, Dietary Reference Intakes for
Vitamin C, Vitamin E, Selenium, and Carotenoids. National Academies
Press; 2000
|
|
29
|
Hathcock JN, Shao A, Vieth R and Heaney R:
Risk assessment for vitamin D. Am J Clin Nut. 85:6–18. 2007.
View Article : Google Scholar
|
|
30
|
Rao SB, Chetana M and Devi PU: Centella
asiatica treatment during postnatal period enhances learning and
memory in mice. Physiol Behav. 86:449–457. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
McIlrath J, Bouffler SD, Samper E,
Cuthbert A, Wojcik A, Szumiel I, Bryant PE, Riches AC, Thompson A,
Blasco MA, et al: Telomere length abnormalities in mammalian
radiosensitive cells. Cancer Res. 61:912–915. 2001.PubMed/NCBI
|
|
32
|
Tsatsakis A, Tsoukalas D, Fragkiadaki P,
Vakonaki E, Tzatzarakis M, Sarandi E, Nikitovic D, Tsilimidos G and
Alegakis AK: Developing BioTel: A semi-automated spreadsheet for
estimating telomere length and biological age. Front Genet.
10:842019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kansara K and Gupta SS: Chapter 14. DNA
damage, repair, and maintenance of telomere length: Role of
nutritional supplements. Mutagenicity: Assays and Applications.
Academic Press; pp. 287–307. 2017
|
|
34
|
Guo J, Huang X, Dou L, Yan M, Shen T, Tang
W and Li J: Aging and aging-related diseases: from molecular
mechanisms to interventions and treatments. Signal Transduct Target
Ther. 7:3912022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jacczak B, Rubiś B and Totoń E: Potential
of naturally derived compounds in telomerase and telomere
modulation in skin senescence and aging. Int J Mol Sci.
22:63812021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Grosso G, Godos J, Currenti W, Micek A,
Falzone L, Libra M, Giampieri F, Forbes-Hernández TY, Quiles JL,
Battino M, et al: The effect of dietary polyphenols on vascular
health and hypertension: Current evidence and mechanisms of action.
Nutrients. 14:5452022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Davinelli S, Trichopoulou A, Corbi G, De
Vivo I and Scapagnini G: The potential nutrigeroprotective role of
Mediterranean diet and its functional components on telomere length
dynamics. Ageing Res Rev. 49:1–10. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Galiè S, Canudas S, Muralidharan J,
García-Gavilán J, Bulló M and Salas-Salvadó J: Impact of nutrition
on telomere health: Systematic review of observational cohort
studies and randomized clinical trials. Adv Nutr. 11:576–601. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Godos J, Giampieri F, Micek A, Battino M,
Forbes-Hernández TY, Quiles JL, Paladino N, Falzone L and Grosso G:
Effect of Brazil nuts on selenium status, blood lipids, and
biomarkers of oxidative stress and inflammation: A systematic
review and meta-analysis of randomized clinical trials.
Antioxidants (Basel). 11:4032022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Sun B, Wu L, Wu Y, Zhang C, Qin L, Hayashi
M, Kudo M, Gao M and Liu T: Therapeutic potential of centella
asiatica and its triterpenes: A review. Front Pharmacol.
11:5680322020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Pizzorno M and Murray J: Textbook of
natural medicine. 4th edition. Elsevier Health Sciences; 2012
|
|
42
|
Giribabu N, Karim K, Kilari EK, Nelli SR
and Salleh N: Oral administration of Centella asiatica (L.) Urb
leave aqueous extract ameliorates cerebral oxidative stress,
inflammation, and apoptosis in male rats with type-2 diabetes.
Inflammopharmacology. 28:1599–1622. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wei L, Chen Q, Guo A, Fan J, Wang R and
Zhang H: Asiatic acid attenuates CCl4-induced liver fibrosis in
rats by regulating the PI3K/AKT/mTOR and Bcl-2/Bax signaling
pathways. Int Immunopharmacol. 60:1–8. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Meehan M and Penckofer S: The role of
vitamin D in the aging adult. J Aging Gerontol. 2:60–71. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sosa-Díaz E, Hernández-Cruz EY and
Pedraza-Chaverri J: The role of vitamin D on redox regulation and
cellular senescence. Free Radic Biol Med. 193:253–273. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mazidi M, Kengne AP and Banach M: Mineral
and vitamin consumption and telomere length among adults in the
United States. Pol Arch Intern Med. 127:87–90. 2017.PubMed/NCBI
|
|
47
|
Yang T, Wang H, Xiong Y, Chen C, Duan K,
Jia J and Ma F: Vitamin D supplementation improves cognitive
function through reducing oxidative stress regulated by telomere
length in older adults with mild cognitive impairment: A 12-month
randomized controlled trial. J Alzheimers Dis. 78:1509–1518. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Normando P, Santos-Rebouças C, Leung C,
Epel E, da Fonseca AC, Zembrzuski V, Faerstein E and Bezerra FF:
Variants in gene encoding for vitamin D binding protein were
associated with leukocyte telomere length: The Pró-Saúde study.
Nutrition. 71:1106182020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Julin B, Shui IM, Prescott J, Giovannucci
EL and De Vivo I: Plasma vitamin D biomarkers and leukocyte
telomere length in men. Eur J Nutr. 56:501–508. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu JJ, Prescott J, Giovannucci E,
Hankinson SE, Rosner B, Han J and De Vivo I: Plasma vitamin D
biomarkers and leukocyte telomere length. Am J Epidemiol.
177:1411–1417. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Borras M, Panizo S, Sarró F, Valdivielso
JM and Fernandez E: Assessment of the potential role of active
vitamin D treatment in telomere length: A case-control study in
Hemodialysis patients. Clin Ther. 34:849–856. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Lee JY, Shin C and Baik I: Longitudinal
associations between micronutrient consumption and leukocyte
telomere length. J Hum Nutr Diet. 30:236–243. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sen A, Marsche G, Freudenberger P,
Schallert M, Toeglhofer AM, Nagl C, Schmidt R, Launer LJ and
Schmidt H: Association between higher plasma lutein, zeaxanthin,
and vitamin C concentrations and longer telomere length: Results of
the Austrian stroke prevention study. J Am Geriatr Soc. 62:222–229.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Marcon F, Siniscalchi E, Crebelli R,
Saieva C, Sera F, Fortini P, Simonelli V and Palli D: Diet-related
telomere shortening and chromosome stability. Mutagenesis.
27:49–57. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Farahzadi R, Fathi E, Mesbah-Namin SA and
Zarghami N: Zinc sulfate contributes to promote telomere length
extension via increasing telomerase gene expression, telomerase
activity and change in the TERT gene promoter CpG island
methylation status of human adipose-derived mesenchymal stem cells.
PLoS One. 12:e01880522017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wei F, Qu C, Song T, Ding G, Fan Z, Liu D,
Liu Y, Zhang C, Shi S and Wang S: Vitamin C treatment promotes
mesenchymal stem cell sheet formation and tissue regeneration by
elevating telomerase activity. J Cell Physiol. 227:3216–3224. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kim YY, Ku SY, Huh Y, Liu HC, Kim SH, Choi
YM and Moon SY: Anti-aging effects of vitamin C on human
pluripotent stem cell-derived cardiomyocytes. Age (Dordr).
35:1545–1557. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhu H, Guo D, Li K, Pedersen-White J,
Stallmann-Jorgensen IS, Huang Y, Parikh S, Liu K and Dong Y:
Increased telomerase activity and vitamin D supplementation in
overweight African Americans. Int J Obes (Lond). 36:805–809. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Chen L, Dong Y, Bhagatwala J, Raed A,
Huang Y and Zhu H: Effects of Vitamin D 3 supplementation on
epigenetic aging in overweight and obese African Americans with
suboptimal Vitamin D status: A randomized clinical trial. J
Gerontol A Biol Sci Med Sci. 74:91–98. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Athanasopoulou S, Kapetanou M, Magouritsas
MG, Mougkolia N, Taouxidou P, Papacharalambous M, Sakellaridis F
and Gonos E: Anti-oxidant and antiaging properties of a novel
synergistic nutraceutical complex: Readouts from an in cellulo
study and an in vivo prospective, randomized trial. Antioxidants
(Basel). 11:4682022. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ferreira GS, Veening-Griffioen DH, Boon
WPC, Moors EHM and van Meer PJK: Levelling the translational gap
for animal to human efficacy data. Animals (Basel). 10:11992020.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Calado RT and Dumitriu B: Telomere
dynamics in mice and humans. Semin Hematol. 50:165–174. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Folgueras AR, Freitas-Rodríguez S, Velasco
G and López-Otín C: Mouse models to disentangle the hallmarks of
human aging. Circ Res. 123:905–924. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Apostolova N and Victor VM: Molecular
strategies for targeting anti-oxidants to mitochondria: Therapeutic
implications. Antioxid Redox Signal. 22:686–729. 2015. View Article : Google Scholar : PubMed/NCBI
|