Introduction
Drug-induced liver injury (DILI) is an adverse drug
reaction that can result in acute liver failure (ALF) or even
death. In developed nations, acetaminophen (APAP) overdose has
become the leading cause of ALF and may require liver
transplantation (1–3). In an overdose, APAP is transformed by
cytochrome P450 into the toxic metabolite N-acetyl-p-benzoquinone
imide (NAPQI) that causes hepatic glutathione depletion.
N-acetylcysteine remains the only clinically authorized treatment
for APAP overdose (4); however,
its efficiency is significantly limited by its narrow therapeutic
window and adverse effects (5).
Therefore, understanding the mechanisms of APAP-induced cell death
and identifying novel therapeutic targets are needed to treat
APAP-induced liver injury (AILI).
The pathological process of AILI can be divided into
three stages: Injury initiation, injury progression and liver
recovery (6). Initially,
accumulated NAPQI results in an overproduction of reactive oxygen
species and mitochondrial dysfunction, which ultimately triggers
hepatocyte necrosis and apoptosis (7). Subsequently, necrotic hepatocytes
release damage-associated molecular patterns such as high-mobility
group box 1 protein and DNA fragments, which lead to recruitment
and activation of inflammatory cells like monocytes and neutrophils
at site of injury (8,9). Finally, compensatory hepatocellular
proliferation is initiated with hepatocytes surrounding the
necrotic area replacing dead cells (10).
The NLR family pyrin domain containing 3 (NLRP3)
inflammasome is one of the most extensively studied upstream
activators of pyroptosis (11).
Activation of NLRP3 leads to the assembly of a protein complex
(including NLRP3 oligomers and ASC) responsible for binding and
cleaving caspase-1, which in turn generates the cleaved gasdermin D
(GSDMD) N-terminal fragment (GSDMD-N) and forms pores in the cell
membrane to induce pyroptosis, as well as releasing proinflammatory
cytokines IL-1β and IL-18 (12–14).
Numerous studies have reported that NLRP3 activates pyroptosis in
various liver diseases (15–18).
However, the role of NLRP3 during APAP overdose has not been
elucidated. Recent studies have demonstrated that pyroptosis occurs
in APAP-induced liver injury through caspase-1 cleavage and GSDMD
activation in both hepatocytes and Kupffer cells (19,20).
Furthermore, it has been demonstrated that deficiency or knock-down
of NLRP3 reduces the pyroptotic parameters and partially rescues
APAP hepatotoxicity in mice (21,22).
However, it has also been suggested that NLRP3 activation is not
essential for APAP-induced liver injury (23,24).
Nevertheless, further investigation is required to elucidate the
relevance of NLRP3-mediated pyroptosis in APAP-induced hepatotoxic
processes.
The present study aimed to investigate the effect of
NLRP3 inflammasome during AILI. Moreover, the present study aimed
to evaluated the therapeutic effects of NLRP3 and GSDMD inhibition
in AILI both in vivo and in vitro.
Materials and methods
Animals and animal models
A total of 110 C57BL/6, 28
Nlrp3fl/fl mice and 28
Nlrp3Δhep mice were used. C57BL/6 male mice
(8-week-old) weighing 20–22 g, were purchased from Beijing Vital
River Laboratory Animal Technology Co., Ltd. (Charles River
Laboratories). Alb-iCre (strain no. T003814) and
B6JNju;B6NNju-Nlrp3 (flper)tm1/Nju
[Nlrp3 locus of X-over P1 (Nlrp3loxp or
Nlrp3fl/fl)] (strain no. T001231,) mice were
purchased from GemPharmatech Co., Ltd. Hepatocyte-specific
Nlrp3 knockout mice
(Albcre+Nlrp3loxp/loxp,
Nlrp3Δhep) were generated by crossing the
Nlrp3loxp mice with Alb-cre mice and the
mice were backcrossed for at least 10 generations on a C57BL/6
background. Male mice (age, 8 weeks old, 20–22 g) were used for the
experiments. The mice were raised in a standard room with
controlled humidity (55–60%) and temperature (23–25°C) under a 12 h
light/dark cycle with ad libitum access to food and water.
All mice received two daily health observations including water and
food intake, weight and general assessment of animal activity. The
study employed humane endpoints according to AVMA Guidelines for
the Euthanasia of Animals (https://www.avma.org/resources-tools/avma-policies/avma-guidelines-euthanasia-animals),
including when the mice showed an inability to obtain food or water
on their own, had a weight loss of >20% of their starting body
weight, difficulty moving, were depressed in the absence of
anesthesia, or their body temperature was persistently below 37°C.
All animal experiments were performed in accordance with the
regulations of the ARRIVE guidelines and approved by The Ethics
Committee Medical College of Qingdao University (Qingdao, China;
approval no. QDU-AEC-2022310).
The APAP-induced liver injury model was established
as previously described, with minor modifications (25). Briefly, 25 mg/ml APAP solution was
prepared by dissolving APAP (Beijing Solarbio Science &
Technology Co., Ltd.) in pyrogen-free phosphate-buffered saline
(PBS) at 60°C and cooled to 37°C before injection. The mice were
fasted for 15–17 h and then intraperitoneally (IP) injected with a
single dose of 300 mg/kg of APAP (sublethal dose; n=5 per group) or
500 mg/kg (lethal dose; n=10 per group). To evaluate the
therapeutic effects of NLRP3 and GSDMD inhibition, NLRP3 inhibitor
MCC950 (TargetMol Chemicals Inc.) was administered IP into mice at
50 mg/kg 2 h after APAP and GSDMD inhibitor disulfiram (DSF;
TargetMol Chemicals Inc.) was administered IP into mice at 50 mg/kg
4 and 24 h before APAP (n=5 per group), with PBS or vehicle (sesame
oil) as control group respectively. At 0, 6,12, 24, 36 or 48 h),
the mice were euthanized using 5% inhalational isoflurane for >3
min and death was verified by a lack of heartbeat and breathing.
Liver tissue and serum samples were harvested for subsequent
experiments.
Survival assays
The survival rates of Nlrp3fl/fl
and Nlrp3Δhep mice were evaluated within 72 h
after 500 mg/kg APAP treatment (lethal dose; n=10 per group) as
previously described (26). Mice
were housed under pathogen-free conditions and were monitored every
6 h with free access to food and water. All of the mice were
euthanized at the end of the study.
Cell culture and treatment
Mouse primary hepatocytes were isolated from C57BL/6
mice (n=5), Nlrp3fl/fl and
Nlrp3Δhep mice (n=3) as previously described by
two-step perfusion methods with minor modifications (27,28).
In brief, the liver was perfused with Hank's Balanced Salt Solution
(Wuhan Servicebio Technology Co., Ltd.) containing EGTA (Beijing
Solarbio Science & Technology Co., Ltd.) and digested with
collagenase type IV (Gibco; Thermo Fisher Scientific, Inc.). Then,
the liver was dissolved in residual digestive fluid and filtered
with a 100 µm cell strainer. Hepatocytes were obtained by low speed
centrifugation (100 × g) for 2 min at 4°C. Cell culture plates were
coated with 0.03 mg/ml rat tail collagen I 2 h in advance at room
temperature and washed three times with sterile PBS before use. The
cells were seeded onto 6-well or 96-well plates with
8×104 cells/ml precoated with rat tail tendon collagen I
(Shengyou Biotechnology Co., Ltd.). After overnight incubation, the
cells were treated with 1, 2.5, 5 and 10 mM APAP for 12 h at
37°C.
The α mouse liver 12 (AML12) cell line was purchased
from The Cell Bank of Type Culture Collection of The Chinese
Academy of Sciences. The cells were cultured in DMEM/F12 (1:1)
(Gibco; Thermo Fisher Scientific, Inc.) medium containing 10% fetal
bovine serum (Shanghai ExCell Biology, Inc.),
Insulin-Transferrin-Selenium (Beyotime Institute of Biotechnology)
and 40 ng/ml dexamethasone (MilliporeSigma) with 5% CO2
at 37°C. Upon reaching a confluency of 70 to 80%, the cells were
incubated with 1, 5, 10 and 20 mM APAP for 24 h, subsequently total
protein and RNA were extracted for western blotting and reverse
transcription-quantitative PCR (RT-qPCR) respectively.
Cell Counting Kit-8 (CCK-8) assay
To investigate the therapeutic effects of NLRP3 and
GSDMD inhibition in vitro, AML12 cells were seeded onto
96-well plates at a density of 1,000 cells per well, 0, 1, 5, 10 µM
MCC950 or 0, 1, 2.5, 5 µM DSF were administrated to the AML12 cell
complete medium, with or without 10 mM APAP. To evaluate the
toxicity of APAP on hepatocytes, cell viability was measured using
a CCK-8 assay (Shanghai Yeasen Biotechnology Co., Ltd.). Briefly,
10 µl of CCK-8 solution was added to each well and incubated for 1
h at 37°C. Absorbance at 450 nm was measured by SpectraMax
Absorbance Reader (Molecular Devices, LLC).
RT-qPCR
Total RNA was extracted from liver tissue, primary
hepatocytes or AML12 cells using FastPure Cell/Tissue Total RNA
Isolation kit (Vazyme Biotech Co., Ltd.). RNA was reverse
transcribed into cDNA using specific primers for Nlrp3, Caspase
1, Il-1β, Tnf-α, Il-6, Mcp-1, Cxcl-1, Cxcl-2, CyclinA2,
CyclinD1 and CyclinE1 (Table I) using the PrimeScript First
Strand cDNA Synthesis kit (Takara Biotechnology Co., Ltd.)
according to the manufacturer's protocol. As previously described
(29), qPCR was performed using
SYBR Premix Ex Taq (Vazyme Biotech Co., Ltd.) on a CFX96 Real-Time
Systems (Bio-Rad Laboratories, Inc.). The thermocycling conditions
were as follows: Pre-incubation at 95°C for 2 min; amplification at
95°C for 10 sec and 60°C for 20 sec, followed by 40 cycles; melting
curve at 65°C for 5 sec and 95°C for 15 sec. The mRNA levels were
quantified using the 2−ΔΔCq method and normalized to the
internal reference gene GADPH (30).
 | Table I.Gene-specific primers used in reverse
transcription-quantitative PCR. |
Table I.
Gene-specific primers used in reverse
transcription-quantitative PCR.
| Gene | Accession no. | Direction | Primer sequence,
5′-3′ |
|---|
| m-Gapdh | NM_001289726.1 | Forward |
GCCACCCAGAAGACTGTGGAT |
|
|
| Reverse |
GGAAGGCCATGCCAGTGA |
| m-Nlrp3 | NM_145827.4 | Forward |
CTGCGGACTGTCCCATCAAT |
|
|
| Reverse |
AGGTTGCAGAGCAGGTGCTT |
|
m-Caspase1 | NM_009807.2 | Forward |
CTGGGACCCTCAAGTTTTGC |
|
|
| Reverse |
CCCTCGGAGAAAGATGTTGAAA |
| m-Il-1β | NM_008361.4 | Forward |
CTTTCCCGTGGACCTTCCA |
|
|
| Reverse |
CTCGGAGCCTGTAGTGCAGTT |
| m-Tnf-α | NM_013693.3 | Forward |
ACAAGGCTGCCCCGACTAC |
|
|
| Reverse |
TGGGCTCATACCAGGGTTTG |
| m-Il-6 | NM_031168.2 | Forward |
ACCACTCCCAACAGACCTGTCT |
|
|
| Reverse |
CAGATTGTTTTCTGCAAGTGCAT |
| m-Mcp-1 | NM_011333.3 | Forward |
CAGCAAGATGATCCCAATGAGTAG |
|
|
| Reverse |
TTTTTAATGTATGTCTGGACCCATTC |
|
m-Cxcl-1 | NM_008176.3 | Forward |
CGCTTCTCTGTGCAGCGCTGCTGCT |
|
|
| Reverse |
AAGCCTCGCGACCATTCTTGAGT |
|
m-Cxcl-2 | NM_009140.2 | Forward |
CCTGGTTCAGAAAATCATCCA |
|
|
| Reverse |
CTTCCGTTGAGGGACAGC |
|
m-CyclinA2 | NM_009828.3 | Forward |
TCAAGACTCGACGGGTTGCT |
|
|
| Reverse |
GCTCAGCTGGCCTCTTCTGA |
|
m-CyclinD1 | NM_001379248.1 | Forward |
TGCTGCAAATGGAACTGCTT |
|
|
| Reverse |
CCACAAAGGTCTGTGCATGCT |
|
m-CyclinE1 | NM_007633.2 | Forward |
TGTTACAGATGGCGCTTGCT |
|
|
| Reverse |
ACCTCACCCGTGTCGTTGAC |
Western blotting
Total protein from liver tissue, primary hepatocytes
or AML12 cells was extracted using RIPA buffer(Wuhan Servicebio
Technology Co., Ltd.) as described previously (31). BCA Protein Assay kit (Epizyme;
ZJ102) to confirm the protein concentration. Briefly, the protein
samples (40 µg) were separated by 7.5, 10 and 12.5% sodium dodecyl
sulfate-polyacrylamide gel electrophoresis and transferred to a
polyvinylidene fluoride membrane (MilliporeSigma). After blocking
with 5% skimmed milk powder for an hour at room temperature, the
membranes were incubated with primary antibodies including: NLRP3
(1:1,000; AG-20B-0014-C100; Adipogen Life Sciences, Inc.), IL-1β
(1:1,000; A11369; ABclonal Biotech Co., Ltd.), Caspase-1 (1:1,000;
A0964; ABclonal Biotech Co., Ltd.), GSDMD (1:1,000; cat. no.
AF4012; Affinity Biosciences), β-actin (1:1,000; AC026; ABclonal
Biotech Co., Ltd.), Caspase-3 (1:1,000; 9662S; Cell Signaling
Technology, Inc.), PCNA (1:1,000, AF0239; Affinity Biosciences),
CyclinD1 (1:1,000; A0310; ABclonal Biotech Co., Ltd.) at 4°C
overnight. Next day, these membranes were washed three times using
Tris-buffered saline with 0.1% Tween-20 for ten minutes each and
then incubated with Goat Anti-Mouse IgG (H+L) HRP or Goat
Anti-Rabbit IgG (H+L) HRP (1:4,000; S0002; S0001; Affinity
Biosciences,) for 1 h at room temperature. Finally, protein bands
were washed three times again and visualized using an ultra
sensitive chemiluminescence assay kit (Shanghai Epizyme Biotech
Co., Ltd.) and imaged using an automatic chemiluminescence image
analysis system (Tanon Science and Technology Co., Ltd.). Protein
levels were quantified using ImageJ software (version 1.43;
National Institutes of Health) with β-actin as the loading
control.
Biochemical analysis and ELISA assay. Blood serum
aspartate transaminase (AST) and serum alanine aminotransferase
(ALT) levels in mice were measured using a Chemray 800 automatic
biochemical analyzer (Rayto Life and Analytical Sciences Co., Ltd.)
according to the manufacturer's instructions (S03040; S03030; Rayto
Life and Analytical Sciences Co., Ltd.). Additionally, levels of
inflammatory cytokines IL-6, IL-18, IL-1β and TNF-α were measured
using mouse interleukin-6(IL-6) ELISA kit (ml098430); mouse
interleukin-18 (IL-18) ELISA kit (ml002294); mouse interleukin-1β
(IL-1β) ELISA kit (ml098416); mouse tumor necrosis factor-α (TNF-α)
ELISA kit (ml002095)(Shanghai Enzyme-linked Biotechnology Co.,
Ltd.) according to the manufacturer's instructions. The absorbance
values were measured at 450 nm using a SpectraMax Absorbance Reader
(Molecular Devices, LLC).
Immunohistochemical staining (IHC) and
TUNEL assay
Histological sectioning and IHC were performed as
previously described (32).
Briefly, liver samples from the left lateral lobes of mice were
fixed with 4% paraformaldehyde (Wuhan Servicebio Technology Co.,
Ltd., G1101) for 24 h at room temperature and embedded in paraffin
or optimal cutting temperature (OCT) compound (Wuhan Servicebio
Technology Co., Ltd.) and stored at −80°C. The paraffin was sliced
into 5 µm sections and frozen OCT compound samples were sliced into
8 µm sections. Sections were hematoxylin and eosin (H&E)
stained to examine the pathological changes in liver tissue after
deparaffinization and dehydration through descending ethyl
alcohol). For IHC, the sections were subjected to antigen retrieval
by microwaving in citrate buffer (pH 6.0, Biogenex, CA) for 20 min
at 99–100°C. After washing with PBS for 15 min, the sections were
incubated with 3% hydrogen peroxide for 10 min at room temperature.
IHC was carried out by incubating the sections with antibodies
including: NLRP3 (1:200; DF7834; Affinity Biosciences), IL-1β
(1:200; ab9722; Abcam), Caspase-1 (1:200; ab207802; Abcam), GSDMD
(1:200; AF4012; Affinity Biosciences), PCNA (1:200; cat. no.
ab92552; Abcam), Ki67 (1:200, ab15580; Abcam), CD68 (1:200;
GB113109; Wuhan Servicebio Technology Co., Ltd.), Ly6G (1:200;
GB11229; Wuhan Servicebio Technology Co., Ltd.) and Caspase-3
(1:200; 9662S; Cell Signaling Technology, Inc.) at 4°C overnight.
Next day, according to the manufacturer's instructions for
immunochromogenic reagent (EliVision plus mouse/rabbit) (KIT9901,
Maixing Company, China), the sections were incubated with
R1(reaction enhancement solution) for 20 min, and then incubated
with R2(HRP-conjugated anti-mouse/rabbit IgG polymer) in the dark
for 30 min at room temperature. Finally, the sections were stained
with DAB chromogenic kit (Wuhan Servicebio Technology Co., Ltd.)
for 2 min at room temperature. The sections were images of the
sections were captured under an BX50 light microscope (Olympus
Corporation).
The frozen sections were subjected to TUNEL staining
using a TUNEL kit (Shanghai Yeasen Biotechnology Co., Ltd.),
following the manufacturer's instructions. Briefly, the sections
were fixed with 4% paraformaldehyde at room temperature for 30 min.
After washing with PBS for 30 min, Proteinase K (20 µg/ml) were
incubated for 10 min at room temperature and TDT incubation buffer
were incubated at 4°C overnight. Subsequently, nuclei were
counterstained with 2 µg/ml 4′,6-diamidino-2-phenylindole (Beyotime
Institute of Biotechnology) for 5 min at room temperature. The
sections were sealed with PBS. The TUNEL-positive cells were
visualized under a fluorescence microscope (Olympus Corporation)
and 5 numbers of field were calculated.
Statistical analysis
All experiments were conducted at least three times,
and statistical analyses were performed using GraphPad Prism 8.0
software (Dotmatics). Differences between groups were assessed
using an unpaired Student's t-test for two groups or one-way ANOVA
followed by Tukey's post hoc test for multiple groups. Survival
curves were compared using the log-rank (Mantel-Cox) test.
P<0.05 was considered to indicate a statistically significant
difference.
Results
APAP overdose induces NLRP3 activation
in hepatocytes in vivo and in vitro
As previously described (25), an APAP-induced liver injury mouse
model was established by IP injection of a single dose of 300 mg/kg
APAP. APAP treatment resulted in liver injury characterized by
increased hepatic necrosis (Fig. 1A
and B) and elevated serum ALT (Fig. 1C) and AST levels (Fig. 1D). To investigate the potential
role of NLRP3 activation in APAP-induced hepatotoxicity, expression
levels of NLRP3 in AILI mice were examined. IHC demonstrated an
increase in NLRP3, Caspase-1, IL-1β and GSDMD in mice liver after
APAP treatment compared with control group (sham) (Fig. 1E). Likewise, increased protein
levels of NLRP3, cleaved IL-1β and GSDMD were shown by western
blotting compared with the control group (sham) (Fig. 1F). Additionally, APAP treatment
increased the levels of proinflammatory cytokines IL-1β (Fig. 1G) and IL-18 (Fig. 1H) in mouse serum compared with 0 h,
indicating the activation of NLRP3 and GSDMD. To determine the
cellular source of NLRP3 expression after 24 or 48 h post-treatment
with APAP, mouse liver parenchymal cells were isolated for western
blotting, which showed increased levels of NLRP3, cleaved
caspase-1, cleaved IL-1β and GSDMD-N at 24 h post APAP treatment
(Fig. 1I).
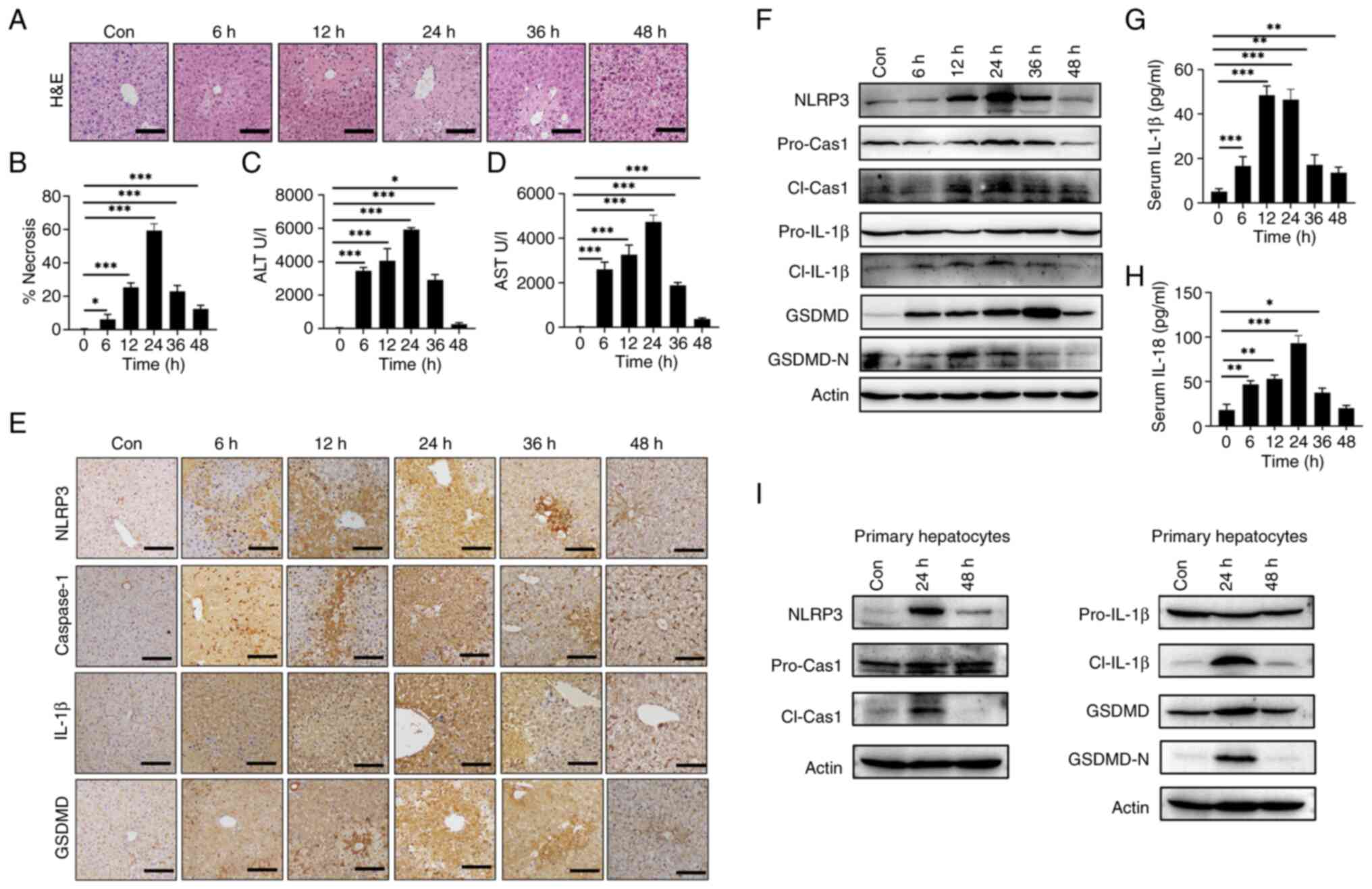 | Figure 1.APAP overdose induces hepatic NLRP3
activation in mice. C57BL/6 mice were intraperitoneally injected
with 300 mg/kg APAP at the indicated time points (n=5). The
histopathological dynamic changes of liver tissues shown by (A)
H&E staining in liver tissues and (B) statistical
quantification of hepatic necrosis area. Serum levels of (C) ALT
and (D) AST. (E) IHC staining of NLRP3, caspase-1, IL-1β and GSDMD
in liver tissues. (F) Western blotting of NLRP3, pro-cas-1,
cl-cas-1, pro-IL-1β, cl-IL-1β, GSDMD and GSDMD-N in liver tissues.
Serum levels of (G) IL-1β and (H) IL-18. (I) Mouse liver
parenchymal cells were isolated after 24 or 48 h post-treatment
with APAP for western blotting of NLRP3, pro-cas-1, cl-cas-1,
pro-IL-1β, cl-IL-1β, GSDMD and GSDMD-N. Data are presented as the
mean ± SD. *P<0.05, **P<0.01, ***P<0.001. pro-cas-1,
pro-caspase-1; cl-cas-1, cl-caspase-1; GSDMD, gasdermin D; GSDMD-N,
cleaved GSDMD N-terminal fragment; cl-IL-1β, cleaved IL-1β; APAP,
acetaminophen; NLRP3, NLR family pyrin domain containing 3; ALT,
alanine aminotransferase; AST, aspartate transaminase; con,
control; IHC, immunohistochemistry. |
To investigate whether APAP overdose induces NLRP3
activation in mouse hepatocytes in vitro, AML12 cells and
primary hepatocytes were subjected to APAP treatment for 24 and 12
h, respectively. These results showed that both cell types
exhibited dose-dependent decrease in cell viability with APAP
treatment (Fig. 2A). Likewise,
APAP treatment increased the mRNA expression levels of Nlrp3
(Fig. 2B), Caspase-1
(Fig. 2C) and Il-1β
(Fig. 2D) in AML12 cells and
primary hepatocytes compared with the control. Furthermore, the
protein levels of NLRP3 and the active fragments of
pyroptosis-related proteins, cleaved caspase-1, cleaved IL-1β and
GSDMD-N were elevated upon APAP treatment in both AML12 and primary
hepatocytes compared with the control (Fig. 2E-F). Taken together, the
aforementioned findings suggested that APAP overdose triggers NLRP3
activation leading to hepatocyte pyroptosis both in vivo and
in vitro.
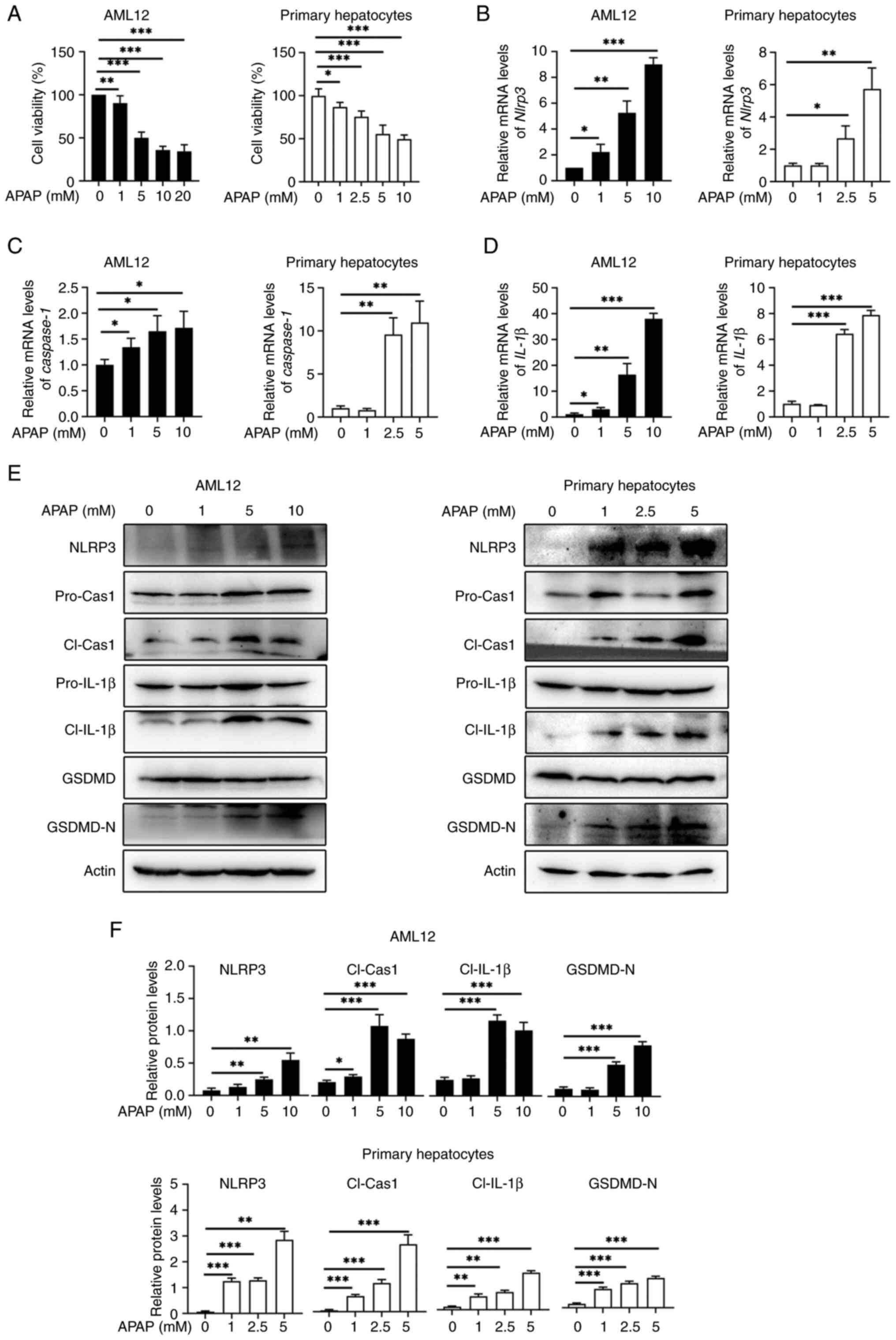 | Figure 2.APAP induces NLRP3 activation and
pyroptosis in hepatocytes in vitro (n=3). AML12 cells and
mouse primary hepatocytes were stimulated with different
concentrations of APAP for 24 h or 12 h. (A) Viability of both cell
types. RT-qPCR analysis of (B) Nlrp3, (C) caspase-1
and (D) Il-1β in these two cell types. (E) Western blotting
and (F) quantification of NLRP3, pro-cas-1, cl-cas-1, pro-IL-1β,
cl-IL-1β, GSDMD and GSDMD-N in both cell types. Data are presented
as the mean ± SD. *P<0.05, **P<0.01, ***P<0.001.
pro-cas-1, pro-caspase-1; cl-cas-1, cl-caspase-1; GSDMD, gasdermin
D; GSDMD-N, cleaved GSDMD N-terminal fragment; APAP, acetaminophen;
NLRP3, NLR family pyrin domain containing 3; ALT, alanine
aminotransferase; AST, aspartate transaminase; RT-qPCR, reverse
transcription-quantitative PCR. |
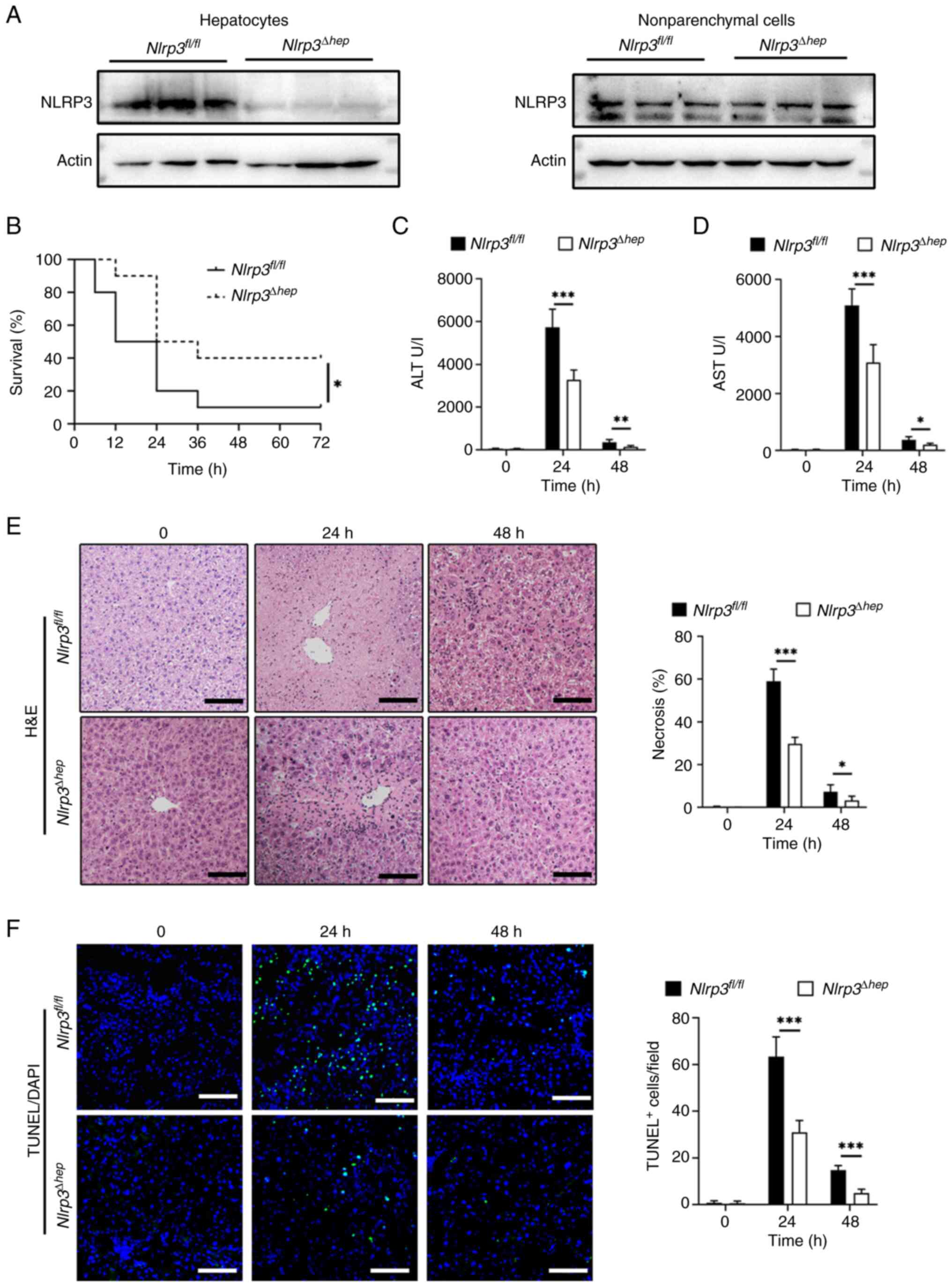
Hepatic Nlrp3 deficiency protects mice
from APAP-induced acute liver injury
As NLRP3 is crucial for APAP-induced hepatocyte
pyroptosis, Nlrp3Δhep mice were generated by
crossing Nlrp3fl/fl mice with
Albcre+ mice. Western blotting confirmed the
specific knockout of Nlrp3 in hepatocytes and not in
non-parenchymal cells (Fig. 3A).
To investigate the impact of hepatic Nlrp3 deficiency on
APAP-induced liver injury, AILI models were created using
Nlrp3fl/fl and Nlrp3Δhep mice.
Survival assays were conducted through IP injection of a lethal
dose (500 mg/kg) of APAP, revealing significantly lower mortality
in Nlrp3Δhep mice compared with
Nlrp3fl/fl mice (Fig. 3B). Liver damage was assessed by
injecting 300 mg/kg APAP and measuring alanine aminotransferase
(ALT) (Fig. 3C) and aspartate
aminotransferase (AST) levels (Fig.
3D), as well as the level of necrosis in liver tissue (Fig. 3E), all of which showed significant
reductions in Nlrp3Δhep mice when compared with
Nlrp3fl/fl mice. Consistently, livers from
Nlrp3Δhep mice exhibited lower TUNEL-positive
staining after APAP treatment compared with the
Nlrp3fl/fl mice (Fig. 3F). These findings indicate that
hepatic blockade of NLRP3 not only protects against APAP-induced
liver injury but also improves survival.
Hepatic Nlrp3 deficiency attenuates
hepatic inflammatory response to APAP overdose
During the pathogenesis of AILI, the immune response
serves a crucial role, and a timely decrease in inflammation is
believed to be important in repairing acute liver injury in mice
(33). To investigate whether the
protective effect from liver injury in Nlrp3Δhep
mice is related to hepatic decrease in inflammation, the levels of
inflammation between the two groups were analyzed after treatment
with 300 mg/kg APAP. IHC staining for Ly6G and CD68, which
represent infiltration of liver neutrophils and macrophages,
respectively, were significantly reduced in
Nlrp3Δhep mice at 24 and 48 h compared with the
Nlrp3fl/fl mice (Fig. 4A). Furthermore, there was a
significant decrease in mRNA expression levels of pro-inflammatory
cytokines Tnf-α and Il-6 at 24 h, and for
Il-1β at both 24 and 48 h post-APAP treatment compared with
the Nlrp3fl/fl mice. Furthermore, a significant
decrease in proinflammatory chemokine mRNA levels of Mcp-1
and Cxcl-1 at 24 h and of Cxcl-2 at 24 and 48 h post
APAP treatment was observed in Nlrp3Δhep mice
compared with Nlrp3fl/fl mice (Fig. 4B).
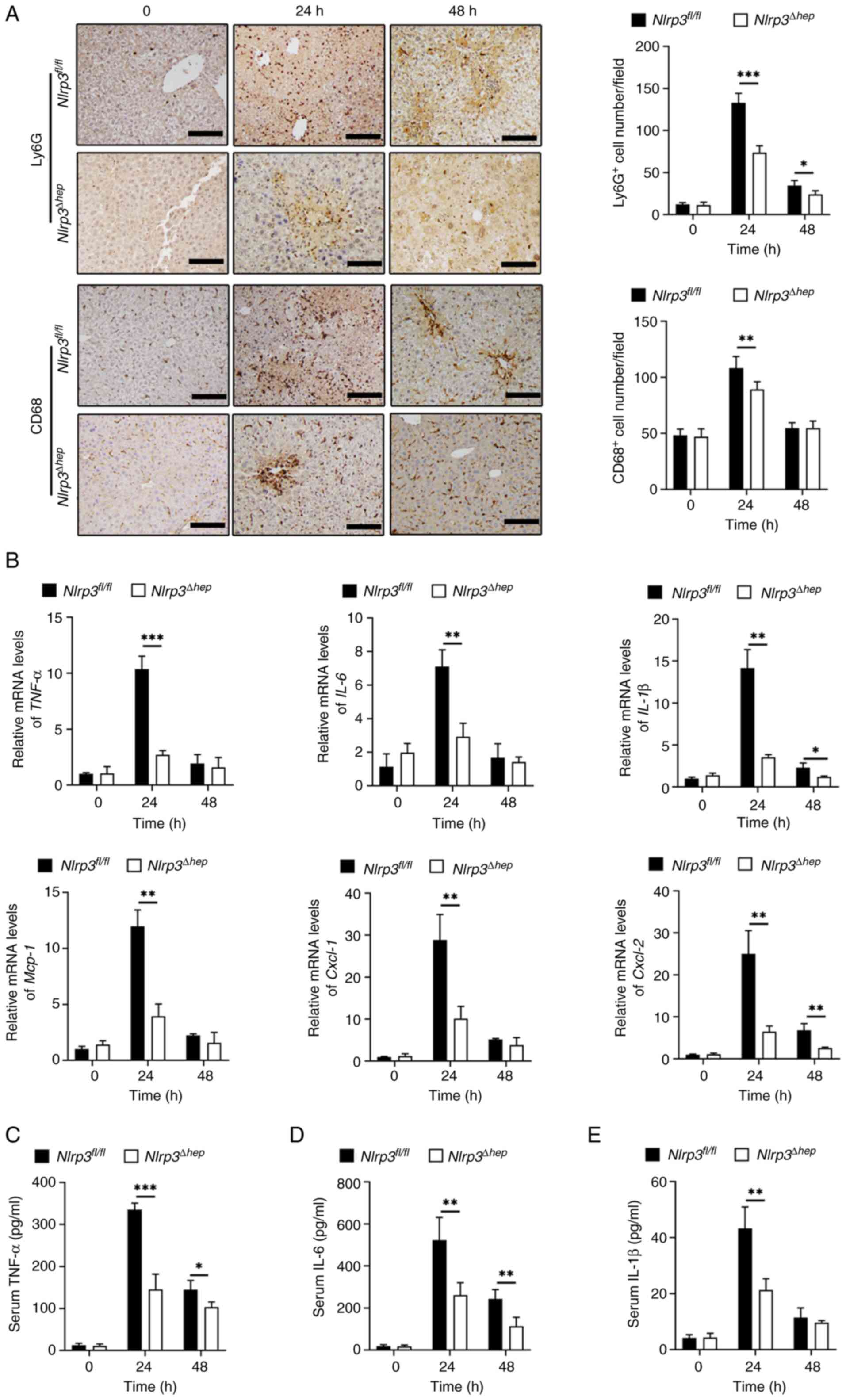 | Figure 4.Hepatic NLRP3 deficiency attenuates
the inflammatory reaction to APAP overdose. Both
Nlrp3fl/fl and Nlrp3Δhep mice
were intraperitoneally injected with 300 mg/kg APAP (n=5). (A)
Representative hepatic Ly6G and CD68 immunohistochemical staining
of liver sections and positive cell number/field were shown. (B)
The relative mRNA levels of inflammatory cytokines Tnf-α, Il-6,
Il-1β and chemokines Mcp-1, Cxcl-1, Cxcl-2 in liver
tissues were determined by RT-qPCR. ELISA analyses of the serum
levels of (C) TNF-α, (D) IL-6 and (E) IL-1β. Data are presented as
the mean ± SD. *P<0.05, **P<0.01, ***P<0.001. APAP,
acetaminophen; NLRP3, NLR family pyrin domain containing 3;
RT-qPCR, reverse transcription-quantitative PCR;
Nlrp3fl/fl, B6JNju;B6NNju-Nlrp3
(flper)tm1/Nju mice;
Nlrp3Δhep, hepatocyte-specific Nlrp3
knockout mice. |
Additionally, compared with
Nlrp3fl/fl mice, significantly decreased protein
levels of TNF-α (Fig. 4C), IL-6
(Fig. 4D) at 24 and 48 h, and
IL-1β (Fig. 4E) at 24 h post-APAP
treatment were measured in Nlrp3Δhep serum
samples. These findings suggest that ablation of hepatic NLRP3 may
alleviate APAP-induced liver injury by reducing inflammation.
NLRP3/GSDMD inhibition alleviates
APAP-induced acute liver injury
To assess the therapeutic potential of NLRP3
inhibition in APAP-induced liver injury, MCC950, an NLRP3
inhibitor, was used as previously described (34). In vivo, C57BL/6 mice were
administered with MCC950 or PBS following APAP treatment and serum
ALT, AST were evaluated at 24 and 48 h post-APAP treatment. The
proportion of liver necrosis (Fig.
5A) and TUNEL-positive cell labeling (Fig. 5B) were significantly decreased with
MCC950 treatment at 24 and 48 h compared with the PBS controls.
Likewise, serum ALT (Fig. 5C) and
AST (Fig. 5D) levels were
significantly reduced in the MCC950 treated group at 24 h compared
with the PBS treatment. In vitro experiments using AML12
cells treated with or without APAP showed that MCC950 improved cell
viability, although the increase was small (Fig. 5E).
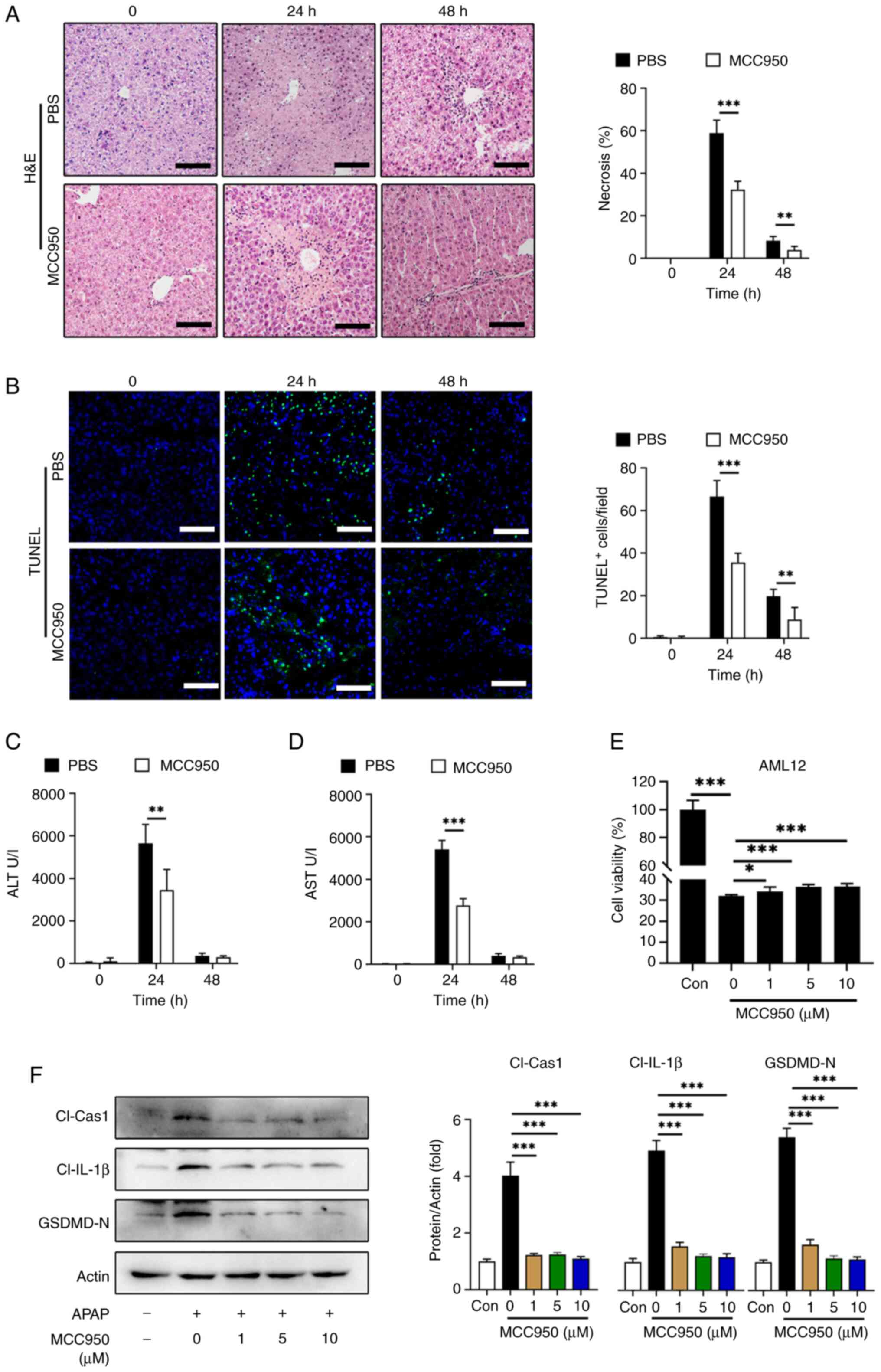 | Figure 5.NLRP3 inhibitor MCC950 alleviates
APAP-induced liver injury and hepatocyte pyroptosis. C57BL/6 mice
were treated with MCC950 or PBS following APAP treatment (n=5). The
therapeutic effect of MCC950 in APAP induced liver injury, with PBS
treated mice as control. (A) Representative images of H&E
staining and the statistical quantification of hepatic necrosis in
each group. (B) Representative images of TUNEL staining and the
statistical quantification of TUNEL-positive cells/field in two
groups. Serum (C) ALT and (D) AST levels for two groups. (E)
Viability of AML12 cells after co-treatment with APAP and different
concentrations of MCC950 for 24 h. (F) Western blotting of
cl-cas-1, cl-IL-1β, GSDMD-N and the statistical quantification of
each protein level in AML12 cells after co-treatment with 10 mM
APAP and 0, 1, 5, 10 µM MCC950 for 24 h. Data are presented as the
mean ± SD. *P<0.05, **P<0.01, ***P<0.001. APAP,
acetaminophen; NLRP3, NLR family pyrin domain containing 3;
cl-cas-1, cl-caspase-1; GSDMD-N, cleaved gasdermin D N-terminal
fragment; cl-IL-1β, cleaved IL-1β. |
Additionally, western blotting demonstrated that 1
µM MCC950 treatment significantly decreased the levels of the
active fragments from pyroptosis-related proteins including cleaved
caspase-1, cleaved IL-1β and GSDMD-N in AML12 cells compared with
the 0 µM MCC950 control (Fig.
5F).
To further validate the association between
NLRP3-mediated pyroptosis and its protective effect on AILI, DSF, a
GSDMD inhibitor, was used as previously described (35,36).
Mice were administered with either DSF or vehicle prior to APAP
treatment. Area of liver necrosis in the DSF group was decreased
compared with vehicle (Fig. 6A).
The results also demonstrated a significant reduction in serum
ALT/AST levels in the group treated with DSF compared with the
vehicle controls. (Fig. 6C and D).
Moreover, IHC revealed a significant decrease in caspase-3 positive
cells at 24 and 48 h post APAP treatment compared with the vehicle
controls. (Fig. 6B), which is
considered an indicator of cell death. Moreover, DSF significantly
decreased the levels of GSDMD-N and cleaved caspase-3 with 1 µM DSF
treatment with compared with the 0 µM DSF control suggesting that
this treatment prevented pyroptosis of AML12 cells (Fig. 6E).
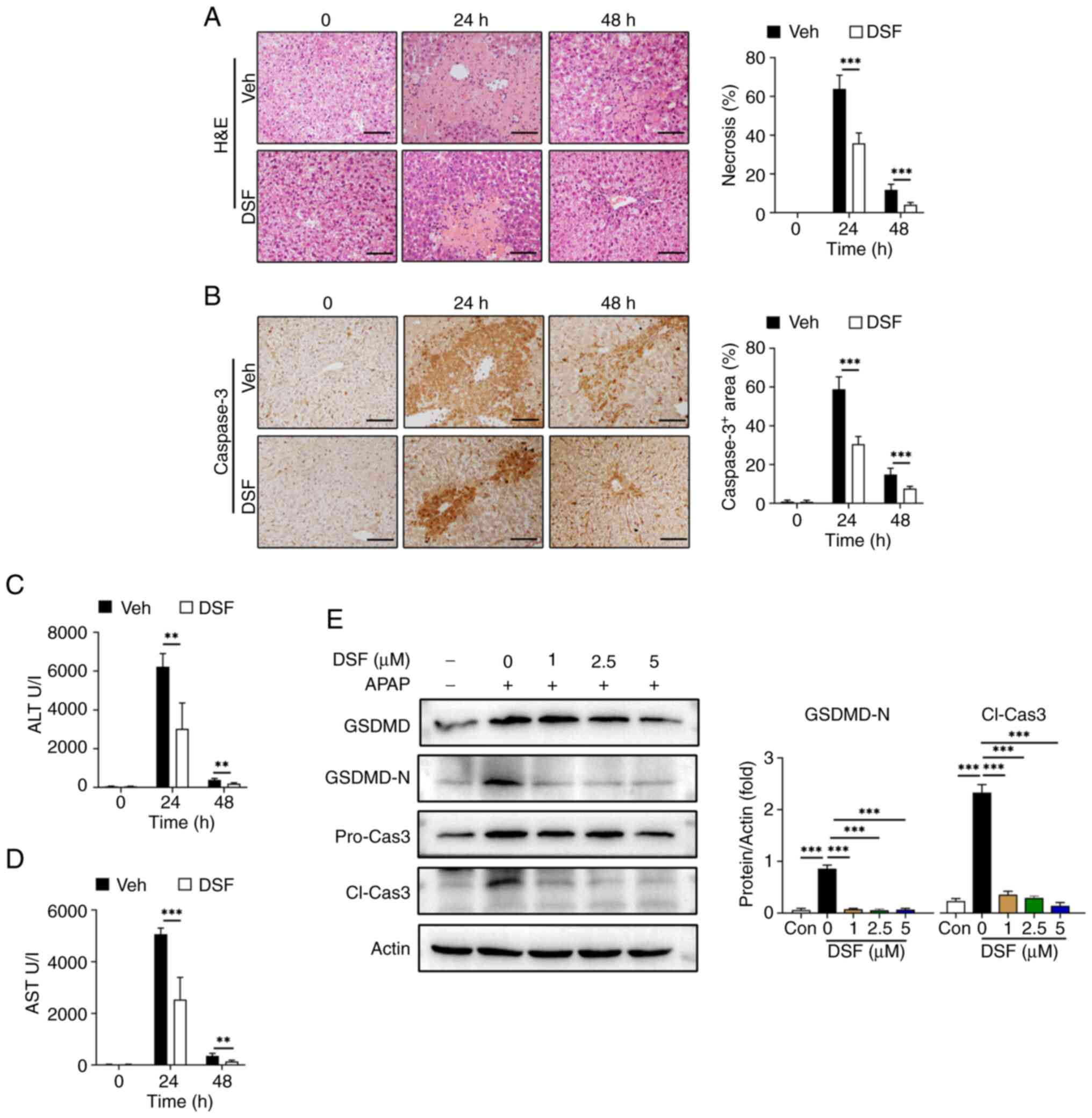 | Figure 6.GSDMD inhibitor DSF mitigates
APAP-induced liver injury and hepatocyte pyroptosis. C57BL/6 mice
were treated with DSF or vehicle prior to APAP treatment (n=5). (A)
Representative images of H&E staining and the statistical
quantification of hepatic necrosis. (B) IHC staining of hepatic
Caspase-3 and the statistical quantification of the positive area.
Serum (C) ALT and (D) AST levels for two groups. (E) Western
blotting of GSDMD-N, Pro-Cas-3/Cl-Cas-3 and the statistical
quantification of each protein level in AML12 cells after
co-culture with 10 mM APAP and 0, 1, 2.5, 5 µM DSF for 24 h. Data
were presented as the mean ± SD. **P<0.01, ***P<0.001. APAP,
acetaminophen; cl-cas-1, cl-caspase-1; pro-cas-1, pro-caspase-1;
GSDMD, gasdermin D; GSDMD-N, cleaved GSDMD N-terminal fragment,
DSF. disulfiram; IHC, immunohistochemical staining; ALT, alanine
aminotransferase; AST, aspartate transaminase. |
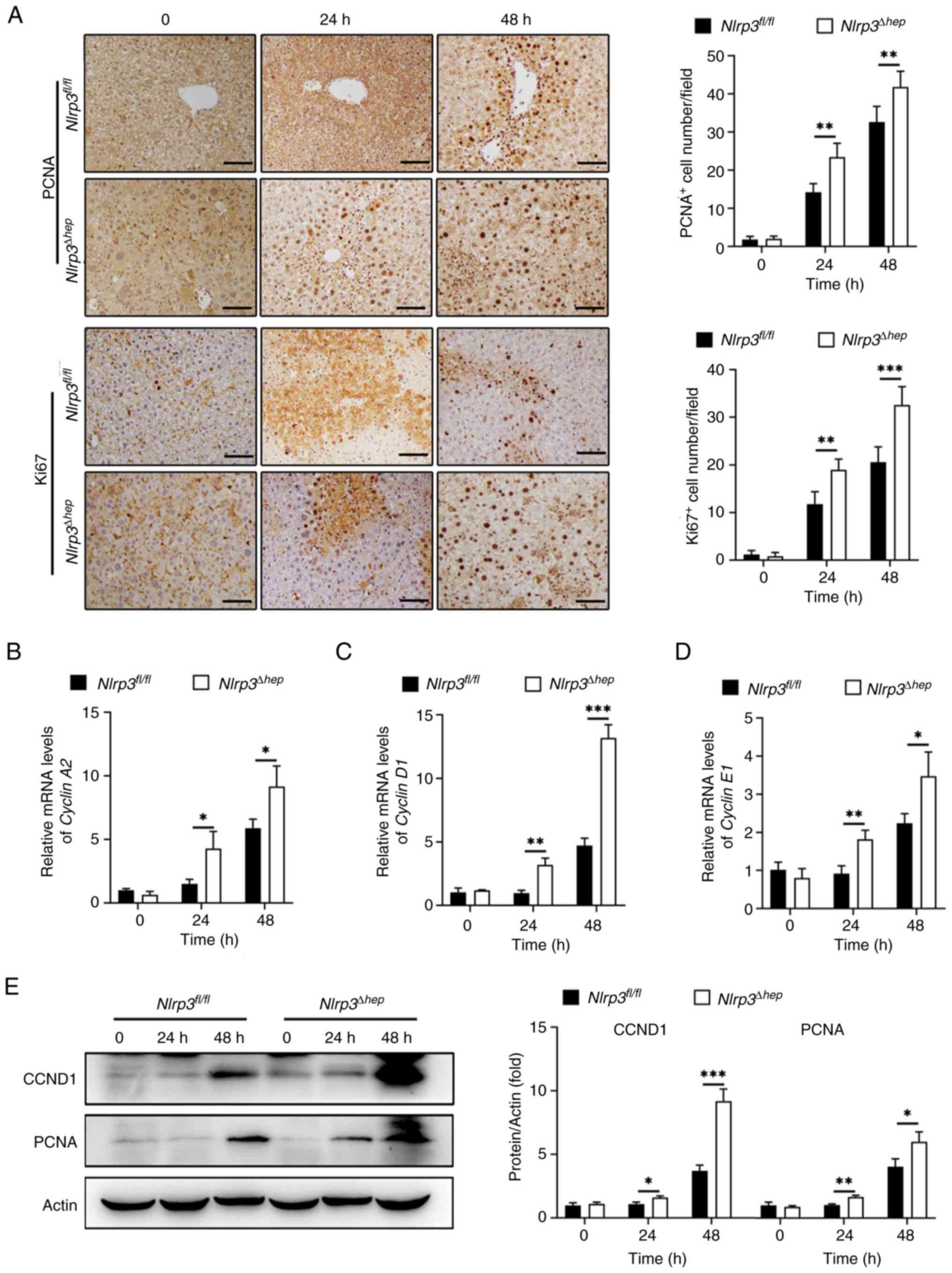
Hepatic Nlrp3 deficiency promotes
liver repair following APAP-induced liver injury
Evidence suggests that the timely initiation of
liver regeneration is crucial for recovery from APAP-induced
hepatotoxicity (6). In the present
study, the regenerative capacity of Nlrp3Δhep and
NLRP3fl/fl mice at 24 and 48 h post APAP
treatment were assessed. A significant increase in the number of
PCNA-positive and Ki67-positive hepatocytes in
Nlrp3Δhep mice at 24 and 48 h compared with the
Nlrp3fl/fl mice were observed (Fig. 7A).
Moreover, mRNA expression levels of Cyclin A2
(Fig. 7B), Cyclin D1
(Fig. 7C) and Cyclin E1
(Fig. 7D), which are pivotal
regulators of cell proliferation, were significantly increased at
24 and 48 h post APAP treatment in Nlrp3Δhep mice
compared with the NLRP3fl/fl mice.
Furthermore, western blotting confirmed elevated
protein levels of CCND1 and PCNA in Nlrp3Δhep
mice at 24 and 48 h post APAP treatment compared with the
NLRP3fl/fl mice (Fig. 7E), consistent with the
aforementioned results (Fig.
7B-D). These findings suggest that hepatic NLRP3 deficiency
promotes liver repair following APAP-induced liver injury.
Discussion
APAP is one of the most commonly used
over-the-counter analgesics and antipyretics (37). While it demonstrates beneficial
effects when used as directed, excessive doses can lead to
hepatotoxicity and dose-dependent ALF. The occurrence of AILI
remains a significant challenge in clinical practice. In the
present study, the activation of NLRP3 in hepatocytes following
APAP overdose was demonstrated. Functionally, these findings
indicate that deficiency of NLRP3 in hepatocytes protects against
liver damage in mice treated with APAP, potentially by reducing
inflammation and promoting hepatic restoration. Furthermore, these
results suggest that this protective effect may be associated with
GSDMD-mediated pyroptosis.
Pyroptosis is a form of programmed cell death that
is mediated by inflammatory caspases (38,39).
While it has been suggested that NLRP3-mediated pyroptosis is
closely associated with acute or chronic liver injury, the role it
serves in AILI remains controversial (20). Some studies have indicated that
NLRP3 activation and IL-1β maturation have little impact on APAP
hepatotoxicity (40), but previous
research has shown that the NLRP3/GSDMD signaling pathway serves a
critical role in APAP-induced hepatocyte death (41–43).
Inhibition of NLRP3-mediated pyroptosis by caveolin-1 or
peroxiredoxin 3 has been shown to alleviate APAP-induced liver
injury (20,44). In the present study,
histopathological analysis was performed by calculating necrotic
areas of liver tissue, as previously described (45–52).
It was demonstrated that administration of APAP (300 mg/kg) caused
severe liver injury with a time-dependent profile characterized by
extensive centrilobular necrosis and elevated serum ALT and AST
activities. It was also observed that NLRP3 activation was induced
as early as 6 h post-APAP treatment, peaked at 12–24 h and then
gradually decreased, similar to the observed time-course of liver
pathology following APAP administration. Furthermore, the levels of
active fragments of pyroptosis-related proteins, cleaved caspase-1,
GSDMD-N and cleaved IL-1β were increased after APAP treatment both
in vivo and in vitro, indicating direct involvement
of NLRP3-mediated pyroptosis in APAP hepatotoxicity.
Growing evidence suggest that Kupffer cells and
neutrophil infiltration are significant in liver injury (53–55).
Notably, this data showed significantly decreased liver injury in
hepatocyte-specific Nlrp3 knockout mice. Moreover, the
pharmacological inhibition of NLRP3/GSDMD signaling by MCC950 or
DSF had similar outcomes to those observed in
Nlrp3Δhep mice, implying its potential
applications in the treatment of acetaminophen overdose.
An increasing number of previous studies have
revealed that sterile inflammation is a significant factor in the
pathogenesis of APAP hepatotoxicity, secondary to oxidative stress
and necrotic cell death (56–58).
It is recognized that the NLRP3 inflammasome serves a crucial role
in initiating and sustaining sterile inflammation in various
diseases (59–61). Upon exposure to stimuli, the NLRP3
inflammasome activates and cleaves caspase-1 by interacting with
the apoptosis-associated speck-like protein containing a caspase
recruitment domain, which leads to the maturation of IL-1β and
IL-18, ultimately exacerbating the inflammatory response (62). The NLRP3 inflammasome is implicated
in the release of pro-inflammatory cytokines in APAP treated mice,
and Nlrp3 knockout mice exhibit less severe AILI and
inflammation (22). Consistently,
the results of the present study showed significantly reduced
infiltration of neutrophils (Ly6G+) and monocytes
(CD68+), as well as decreased levels of pro-inflammatory
cytokines and chemokines in Nlrp3Δhep mice. These
findings suggest that the NLRP3 inflammasome expressed by
hepatocytes might contribute to immune responses during AILI.
Previous studies have demonstrated that liver
compensatory regeneration is a pivotal determinant of survival
after APAP overdose, and enhancing liver regeneration has been
reported to improve the final outcome of AILI (63–66).
The inflammatory response is involved in the initiation and
regulation of the liver regeneration (67), but excess inflammation serves as a
detrimental factor that aggravates liver damage (68). Although the NLRP3 inflammasome is
known to contribute to initiating inflammation during the early
stage of liver injury (43), its
potential involvement in liver regeneration after APAP overdose
remains unclear. A previous study has shown that dexmedetomidine
promotes liver regeneration by suppressing the NLRP3 inflammasome
in a partial hepatectomy model (69). Similarly, GSDMD-mediated pyroptosis
has been found to suppress liver regeneration using the same model
(35). However, another study has
revealed that NLRP3 inflammasome activation accelerates hepatocyte
proliferation after AILI (70). In
the present study, by utilizing hepatocyte Nlrp3 knockout
mice, it was observed that hepatocyte NLRP3 deficiency increased
the number of mitotic hepatocytes and increased the expression of
hepatic PCNA and CCND1 at 24 and 48 h after APAP overdose. From
this it can be speculated that more hepatocytes would survive
pyroptosis in the context of NLRP3 deficiency, where mitogenic
genes are upregulated. However, further research is needed to
elucidate the underlying mechanism.
In conclusion, the present study demonstrated that
NLRP3 deficiency inhibited hepatocyte pyroptosis, alleviated
inflammatory response, and promoted hepatocyte proliferation in
AILI. These results identify the critical regulatory role of
NLRP3-mediated pyroptosis in APAP-induced hepatotoxicity and
provides novel insights into the pathological mechanisms of
AILI.
Acknowledgements
Not applicable.
Funding
The present work was supported by the National Key R&D
Program of China Grants (grant no. 2018YFA0109800), the Shandong
Provincial Natural Science Foundation grant (grant no. ZR2022MH183)
and the Shandong Provincial Youth Entrepreneurship Program for
Colleges and Universities (grant no. 2022KJ148).
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author upon
reasonable request.
Authors' contributions
XY, PC contributed to sample testing, data analysis
and study design. XL, CY, LM and YZ contributed to sample
preparation. AL and TS contributed to study design. GD directed the
project, contributed to the conception and design of the work. GD
had full access to all the data in the study and had final
responsibility for the decision to submit for publication. XY, PC
and GD confirm the authenticity of all the raw data. All authors
have read and approved the final version of the manuscript.
Ethics approval and consent to
participate
This study was approved by The Ethics Committee
Medical College of Qingdao University (approval no.
QDU-AEC-2022310).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Gulmez SE, Larrey D, Pageaux GP, Lignot S,
Lassalle R, Jové J, Gatta A, McCormick PA, Metselaar HJ, Monteiro
E, et al: Transplantation for acute liver failure in patients
exposed to NSAIDs or paracetamol (acetaminophen): The multinational
case-population SALT study. Drug Saf. 36:135–144. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Larson AM, Polson J, Fontana RJ, Davern
TJ, Lalani E, Hynan LS, Reisch JS, Schiødt FV, Ostapowicz G, Shakil
AO, et al: Acetaminophen-induced acute liver failure: Results of a
United States multicenter, prospective study. Hepatology.
42:1364–1372. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gow PJ, Jones RM, Dobson JL and Angus PW:
Etiology and outcome of fulminant hepatic failure managed at an
Australian liver transplant unit. J Gastroenterol Hepatol.
19:154–159. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jaeschke H, Adelusi OB, Akakpo JY, Nguyen
NT, Sanchez-Guerrero G, Umbaugh DS, Ding WX and Ramachandran A:
Recommendations for the use of the acetaminophen hepatotoxicity
model for mechanistic studies and how to avoid common pitfalls.
Acta Pharm Sin B. 11:3740–3755. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Du K, Ramachandran A and Jaeschke H:
Oxidative stress during acetaminophen hepatotoxicity: Sources,
pathophysiological role and therapeutic potential. Redox Biol.
10:148–156. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bhushan B and Apte U: Liver regeneration
after acetaminophen hepatotoxicity: Mechanisms and therapeutic
opportunities. Am J Pathol. 189:719–729. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jaeschke H, McGill MR and Ramachandran A:
Oxidant stress, mitochondria, and cell death mechanisms in
drug-induced liver injury: Lessons learned from acetaminophen
hepatotoxicity. Drug Metab Rev. 44:88–106. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jaeschke H, Williams CD, Ramachandran A
and Bajt ML: Acetaminophen hepatotoxicity and repair: The role of
sterile inflammation and innate immunity. Liver Int. 32:8–20. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kubes P and Mehal WZ: Sterile inflammation
in the liver. Gastroenterology. 143:1158–1172. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bajt ML, Knight TR, Farhood A and Jaeschke
H: Scavenging peroxynitrite with glutathione promotes regeneration
and enhances survival during acetaminophen-induced liver injury in
mice. J Pharmacol Exp Ther. 307:67–73. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang L, Jiao XF, Wu C, Li XQ, Sun HX, Shen
XY, Zhang KZ, Zhao C, Liu L, Wang M, et al: Trimetazidine
attenuates dexamethasone-induced muscle atrophy via inhibiting
NLRP3/GSDMD pathway-mediated pyroptosis. Cell Death Discov.
7:2512021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shi J, Zhao Y, Wang K, Shi X, Wang Y,
Huang H, Zhuang Y, Cai T, Wang F and Shao F: Cleavage of GSDMD by
inflammatory caspases determines pyroptotic cell death. Nature.
526:660–665. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sborgi L, Ruhl S, Mulvihill E, Pipercevic
J, Heilig R, Stahlberg H, Farady CJ, Müller DJ, Broz P and Hiller
S: GSDMD membrane pore formation constitutes the mechanism of
pyroptotic cell death. EMBO J. 35:1766–1778. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
He WT, Wan H, Hu L, Chen P, Wang X, Huang
Z, Yang ZH, Zhong CQ and Han J: Gasdermin D is an executor of
pyroptosis and required for interleukin-1beta secretion. Cell Res.
25:1285–1298. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu C, Chen P, Miao L and Di G: The Role of
the NLRP3 inflammasome and programmed cell death in acute liver
injury. Int J Mol Sci. 24:30672023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yang W, Tao K, Zhang P, Chen X, Sun X and
Li R: Maresin 1 protects against
lipopolysaccharide/d-galactosamine-induced acute liver injury by
inhibiting macrophage pyroptosis and inflammatory response. Biochem
Pharmacol. 195:1148632022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Han D, Kim H, Kim S, Le QA, Han SY, Bae J,
Shin HW, Kang HG, Han KH, Shin J and Park HW: Sestrin2 protects
against cholestatic liver injury by inhibiting endoplasmic
reticulum stress and NLRP3 inflammasome-mediated pyroptosis. Exp
Mol Med. 54:239–251. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ruan S, Han C, Sheng Y, Wang J, Zhou X,
Guan Q, Li W, Zhang C and Yang Y: Antcin A alleviates pyroptosis
and inflammatory response in Kupffercells of non-alcoholic fatty
liver disease by targeting NLRP3. Int Immunopharmacol.
100:1081262021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu T, Yang L, Gao H, Zhuo Y, Tu Z, Wang
Y, Xun J, Zhang Q, Zhang L and Wang X: 3,4-dihydroxyphenylethyl
alcohol glycoside reduces acetaminophen-induced acute liver failure
in mice by inhibiting hepatocyte ferroptosis and pyroptosis. PeerJ.
10:e130822022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang Y, Zhao Y, Wang Z, Sun R, Zou B, Li
R, Liu D, Lin M, Zhou J, Ning S, et al: Peroxiredoxin 3 inhibits
acetaminophen-induced liver pyroptosis through the regulation of
mitochondrial ROS. Front Immunol. 12:6527822021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang JC, Shi Q, Zhou Q, Zhang LL, Qiu YP,
Lou DY, Zhou LQ, Yang B, He QJ, Weng QJ and Wang JJ: Sapidolide A
alleviates acetaminophen-induced acute liver injury by inhibiting
NLRP3 inflammasome activation in macrophages. Acta Pharmacol Sin.
43:2016–2025. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Imaeda AB, Watanabe A, Sohail MA, Mahmood
S, Mohamadnejad M, Sutterwala FS, Flavell RA and Mehal WZ:
Acetaminophen-induced hepatotoxicity in mice is dependent on Tlr9
and the Nalp3 inflammasome. J Clin Invest. 119:305–314.
2009.PubMed/NCBI
|
|
23
|
Zhang C, Feng J, Du J, Zhuo Z, Yang S,
Zhang W, Wang W, Zhang S, Iwakura Y, Meng G, et al:
Macrophage-derived IL-1alpha promotes sterile inflammation in a
mouse model of acetaminophen hepatotoxicity. Cell Mol Immunol.
15:973–982. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen CJ, Kono H, Golenbock D, Reed G,
Akira S and Rock KL: Identification of a key pathway required for
the sterile inflammatory response triggered by dying cells. Nat
Med. 13:851–856. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li M, Sun X, Zhao J, Xia L, Li J, Xu M,
Wang B, Guo H, Yu C, Gao Y, et al: CCL5 deficiency promotes liver
repair by improving inflammation resolution and liver regeneration
through M2 macrophage polarization. Cell Mol Immunol. 17:753–764.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang B, Li J, Jiao J, Xu M, Luo Y, Wang F,
Xia Q, Gao Y, Feng Y, Kong X and Sun X: Myeloid DJ-1 deficiency
protects acetaminophen-induced acute liver injury through
decreasing inflammatory response. Aging (Albany NY).
13:18879–18893. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wen Y, Feng D, Wu H, Liu W, Li H, Wang F,
Xia Q, Gao WQ and Kong X: Defective initiation of liver
regeneration in osteopontin-deficient mice after partial
hepatectomy due to insufficient activation of IL-6/Stat3 Pathway.
Int J Biol Sci. 11:1236–1247. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ni HM, Du K, You M and Ding WX: Critical
role of FoxO3a in alcohol-induced autophagy and hepatotoxicity. Am
J Pathol. 183:1815–1825. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Luan X, Chen P, Li Y, Yuan X, Miao L,
Zhang P, Cao Q, Song X and Di G: TNF-α/IL-1β-licensed hADSCs
alleviate cholestatic liver injury and fibrosis in mice via
COX-2/PGE2 pathway. Stem Cell Res Ther. 14:1002023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu J, Chen P, Yu C, Shi Q, Wei S, Li Y, Qi
H, Cao Q, Guo C, Wu X and Di G: Hypoxic bone marrow mesenchymal
stromal cells-derived exosomal miR-182-5p promotes liver
regeneration via FOXO1-mediated macrophage polarization. FASEB J.
36:e225532022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wei S, Li Z, Shi Q, Luan X, Yuan X, Li Y,
Guo C, Wu X, Shi C and Di G: Collagen-binding vascular endothelial
growth factor (CBD-VEGF) promotes liver regeneration in murine
partial hepatectomy. Mol Med Rep. 26:3262022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Triantafyllou E, Pop OT, Possamai LA,
Wilhelm A, Liaskou E, Singanayagam A, Bernsmeier C, Khamri W, Petts
G, Dargue R, et al: MerTK expressing hepatic macrophages promote
the resolution of inflammation in acute liver failure. Gut.
67:333–347. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mridha AR, Wree A, Robertson AAB, Yeh MM,
Johnson CD, Van Rooyen DM, Haczeyni F, Teoh NC, Savard C, Ioannou
GN, et al: NLRP3 inflammasome blockade reduces liver inflammation
and fibrosis in experimental NASH in mice. J Hepatol. 66:1037–1046.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lv X, Chen J, He J, Hou L, Ren Y, Shen X,
Wang Y, Ji T and Cai X: Gasdermin D-mediated pyroptosis suppresses
liver regeneration after 70% partial hepatectomy. Hepatol Commun.
6:2340–2353. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hu JJ, Liu X, Xia S, Zhang Z, Zhang Y,
Zhao J, Ruan J, Luo X, Lou X, Bai Y, et al: FDA-approved disulfiram
inhibits pyroptosis by blocking gasdermin D pore formation. Nat
Immunol. 21:736–745. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chowdhury A, Nabila J, Adelusi Temitope I
and Wang S: Current etiological comprehension and therapeutic
targets of acetaminophen-induced hepatotoxicity. Pharmacol Res.
161:1051022020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kovacs SB and Miao EA: Gasdermins:
Effectors of pyroptosis. Trends Cell Biol. 27:673–684. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Broz P and Dixit VM: Inflammasomes:
Mechanism of assembly, regulation and signalling. Nat Rev Immunol.
16:407–420. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Williams CD, Farhood A and Jaeschke H:
Role of caspase-1 and interleukin-1beta in acetaminophen-induced
hepatic inflammation and liver injury. Toxicol Appl Pharmacol.
247:169–178. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Cai C, Huang H, Whelan S, Liu L, Kautza B,
Luciano J, Wang G, Chen G, Stratimirovic S, Tsung A, et al: Benzyl
alcohol attenuates acetaminophen-induced acute liver injury in a
Toll-like receptor-4-dependent pattern in mice. Hepatology.
60:990–1002. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yu Y, Zhou S, Wang Y, Di S, Wang Y, Huang
X and Chen Y: Leonurine alleviates acetaminophen-induced acute
liver injury by regulating the PI3K/AKT signaling pathway in mice.
Int Immunopharmacol. 120:1103752023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Gao Z, Zhan H, Zong W, Sun M, Linghu L,
Wang G, Meng F and Chen M: Salidroside alleviates
acetaminophen-induced hepatotoxicity via Sirt1-mediated activation
of Akt/Nrf2 pathway and suppression of NF-κB/NLRP3 inflammasome
axis. Life Sci. 327:1217932023. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jiang X, Li Y, Fu D, You T, Wu S, Xin J,
Wen J, Huang Y and Hu C: Caveolin-1 ameliorates
acetaminophen-aggravated inflammatory damage and lipid deposition
in non-alcoholic fatty liver disease via the ROS/TXNIP/NLRP3
pathway. Int Immunopharmacol. 114:1095582023. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xu L, Yang Y, Jiang J, Wen Y, Jeong JM,
Emontzpohl C, Atkins CL, Kim K, Jacobsen EA, Wang H and Ju C:
Eosinophils protect against acetaminophen-induced liver injury
through cyclooxygenase-mediated IL-4/IL-13 production. Hepatology.
77:456–465. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen L, Dong J, Liao S, Wang S, Wu Z, Zuo
M, Liu B, Yan C, Chen Y, He H, et al: Loss of Sam50 in hepatocytes
induces cardiolipin-dependent mitochondrial membrane remodeling to
trigger mtDNA release and liver injury. Hepatology. 76:1389–1408.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Gao RY, Wang M, Liu Q, Feng D, Wen Y, Xia
Y, Colgan SP, Eltzschig HK and Ju C: Hypoxia-Inducible Factor-2α
reprograms liver macrophages to protect against acute liver injury
through the production of interleukin-6. Hepatology. 71:2105–2117.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chen Y, Liu K, Zhang J, Hai Y, Wang P,
Wang H, Liu Q, Wong CCL, Yao J, Gao Y, et al: c-Jun NH2-Terminal
protein kinase phosphorylates the Nrf2-ECH Homology 6 domain of
nuclear factor erythroid 2-Related Factor 2 and downregulates
cytoprotective genes in acetaminophen-induced liver injury in mice.
Hepatology. 71:1787–1801. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Park S, Park J, Kim E and Lee Y: The
Capicua/ETS translocation variant 5 axis regulates liver-resident
memory CD8(+) T-Cell development and the pathogenesis of liver
injury. Hepatology. 70:358–371. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Chen D, Ni HM, Wang L, Ma X, Yu J, Ding WX
and Zhang L: p53 Up-regulated modulator of apoptosis induction
mediates acetaminophen-induced necrosis and liver injury in mice.
Hepatology. 69:2164–2179. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sun Y, Li TY, Song L, Zhang C, Li J, Lin
ZZ, Lin SC and Lin SY: Liver-specific deficiency of unc-51 like
kinase 1 and 2 protects mice from acetaminophen-induced liver
injury. Hepatology. 67:2397–2413. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang C, Lin J, Zhen C, Wang F, Sun X,
Kong X and Gao Y: Amygdalin protects against acetaminophen-induced
acute liver failure by reducing inflammatory response and
inhibiting hepatocyte death. Biochem Biophys Res Commun.
602:105–112. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Pu JL, Huang ZT, Luo YH, Mou T, Li TT, Li
ZT, Wei XF and Wu ZJ: Fisetin mitigates hepatic
ischemia-reperfusion injury by regulating GSK3β/AMPK/NLRP3
inflammasome pathway. Hepatobiliary Pancreat Dis Int. 20:352–360.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Huang Z, Mou T, Luo Y, Pu X, Pu J, Wan L,
Gong J, Yang H, Liu Y, Li Z, et al: Inhibition of miR-450b-5p
ameliorates hepatic ischemia/reperfusion injury via targeting
CRYAB. Cell Death Dis. 11:4552020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen YX, Sato M, Kawachi K and Abe Y:
Neutrophil-mediated liver injury during hepatic
ischemia-reperfusion in rats. Hepatobiliary Pancreat Dis Int.
5:436–442. 2006.PubMed/NCBI
|
|
56
|
James LP, Simpson PM, Farrar HC, Kearns
GL, Wasserman GS, Blumer JL, Reed MD, Sullivan JE and Hinson JA:
Cytokines and toxicity in acetaminophen overdose. J Clin Pharmacol.
45:1165–1171. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Shen K, Chang W, Gao X, Wang H, Niu W,
Song L and Qin X: Depletion of activated hepatic stellate cell
correlates with severe liver damage and abnormal liver regeneration
in acetaminophen-induced liver injury. Acta Biochim Biophys Sin
(Shanghai). 43:307–315. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Woolbright BL, Nguyen NT, McGill MR,
Sharpe MR, Curry SC and Jaeschke H: Generation of pro-and
anti-inflammatory mediators after acetaminophen overdose in
surviving and non-surviving patients. Toxicol Lett. 367:59–66.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Toldo S, Mezzaroma E, Buckley LF, Potere
N, Di Nisio M, Biondi-Zoccai G, Van Tassell BW and Abbate A:
Targeting the NLRP3 inflammasome in cardiovascular diseases.
Pharmacol Ther. 236:1080532022. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Takahashi M: NLRP3 inflammasome as a key
driver of vascular disease. Cardiovasc Res. 118:372–385. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Sharma BR and Kanneganti TD: NLRP3
inflammasome in cancer and metabolic diseases. Nat Immunol.
22:550–559. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Kelley N, Jeltema D, Duan Y and He Y: The
NLRP3 Inflammasome: An overview of mechanisms of activation and
regulation. Int J Mol Sci. 20:33282019. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Nejak-Bowen KN and Monga SP: Beta-catenin
signaling, liver regeneration and hepatocellular cancer: Sorting
the good from the bad. Semin Cancer Biol. 21:44–58. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Donahower BC, McCullough SS, Hennings L,
Simpson PM, Stowe CD, Saad AG, Kurten RC, Hinson JA and James LP:
Human recombinant vascular endothelial growth factor reduces
necrosis and enhances hepatocyte regeneration in a mouse model of
acetaminophen toxicity. J Pharmacol Exp Ther. 334:33–43. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Schmidt LE and Dalhoff K:
Alpha-fetoprotein is a predictor of outcome in
acetaminophen-induced liver injury. Hepatology. 41:26–31. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Hu B and Colletti LM: Stem cell factor and
c-kit are involved in hepatic recovery after acetaminophen-induced
liver injury in mice. Am J Physiol Gastrointest Liver Physiol.
295:G45–G53. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Hu C, Wu Z and Li L: Mesenchymal stromal
cells promote liver regeneration through regulation of immune
cells. Int J Biol Sci. 16:893–903. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Lazcanoiturburu N, García-Sáez J,
González-Corralejo C, Roncero C, Sanz J, Martín-Rodríguez C,
Valdecantos MP, Martínez-Palacián A, Almalé L, Bragado P, et al:
Lack of EGFR catalytic activity in hepatocytes improves liver
regeneration following DDC-induced cholestatic injury by promoting
a pro-restorative inflammatory response. J Pathol. 258:312–324.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Lv M, Zeng H, He Y, Zhang J and Tan G:
Dexmedetomidine promotes liver regeneration in mice after 70%
partial hepatectomy by suppressing NLRP3 inflammasome not
TLR4/NFκB. Int Immunopharmacol. 54:46–51. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Shi L, Zhang S, Huang Z, Hu F, Zhang T,
Wei M, Bai Q, Lu B and Ji L: Baicalin promotes liver regeneration
after acetaminophen-induced liver injury by inducing NLRP3
inflammasome activation. Free Radic Biol Med. 160:163–177. 2020.
View Article : Google Scholar : PubMed/NCBI
|