Introduction
Cancer immunotherapy is emerging as a beneficial
tool for cancer treatment by activating the immune system to
produce antitumor effects (1).
Recently, cancer immunotherapy, particularly immune checkpoint
therapy, has progressed and provided novel strategies for the
treatment of cancer. The most advanced approach to therapeutically
utilize the antitumor activity is via immune checkpoint inhibitors.
This strategy has recently achieved notable clinical success in
patients with numerous malignant cancer types; for example, in
patients with advanced melanoma, the blockade of cytotoxic T
lymphocyte-associated antigen 4 (CTLA-4) via the antibody
ipilimumab and the inhibition of the programmed death 1 (PD-1)
receptor via the antibody nivolumab resulted in improved overall
survival time (2,3). In comparison to conventional therapies
for cancer, including radiation and chemotherapy, cancer
immunotherapy primarily targets the immune system or tumor
microenvironment rather than tumor cells themselves, and can induce
a synergistic effect in combination therapies (4). However, the efficacy of cancer
immunotherapy is limited to only certain patients, due to not all
patients responding to these immunomodulatory maneuvers, and there
are notable differences between individuals (5). The manner in which to improve the
efficacy of patients with cancer is fast becoming the focus of
cancer immunotherapy.
There is increasing evidence demonstrating that the
differences in the outcome of cancer immunotherapy are attributed
to the heterogeneity of the tumor microenvironment (6). The tumor microenvironment consists of
tumor cells, tumor-infiltrating immune cells, cancer-associated
fibroblasts (CAFs), the tumor vasculature and the extracellular
matrix (ECM), which collectively can promote tumor transformation,
protect the tumor from host immunity, support tumor growth and
invasion, foster therapeutic resistance and provide niches for
dormant metastases to thrive (7). The
presence of malignant tumor cells initiates a series of changes
that can transform the tumor environment into one that can promote
cancer progression. The orchestration of these changes involves
recruitment and activation of CAFs, migration of immune cells,
stroma remodeling, development of tumor vascular networks,
upregulation of the suppressive receptors on tumor cells and
reprogramming of cell metabolism (8).
The complexity of these changes results in the heterogeneity of the
tumor microenvironment. Furthermore, the tumor immunosuppressive
microenvironment is formed with the development of tumor
proliferation and the increasing heterogeneity of the tumor
microenvironment, which may influence the cancer immunotherapy.
In the present review, the progression of tumor
microenvironment heterogeneity, its development and the effect on
immunotherapy, and the present and future of cancer immunotherapy
from the perspective of the tumor microenvironment are
summarized.
Formation and development of the tumor
immuno-suppressive microenvironment
Although tumor cells initially instigate the
formation of the tumor microenvironment, the mutual influence and
co-evolution among tumor cells, stroma components and immune cells
continuously promote the development of immunosuppressive progress
(7). Tumor cells utilize the negative
regulatory mechanism of the immune system, in order to establish a
full range of immunosuppressive states in the tumor
microenvironment, which will create the conditions for their
survival and development (9).
Effects of tumor cells on the tumor
microenvironment
Tumor cells promote immune escape by forming an
immunosuppressive microenvironment. Antigens expressed on the
surface of tumor cells are usually in a defective state. The
decreased or absent expression of major histocompatibility complex
class I restricts the activation of the tumor-infiltrating
lymphocytes (TILs) (9). The existing
suppressive signal transduction in the immune system could be
utilized by tumor cells, including PD-1, ligand programmed
death-ligand 1 (PD-L1), CTLA-4, cytotoxic T lymphocyte activation
gene 3 (LAG-3), T cell immunoglobulin domain 3 mucin domain protein
3 (Tim-3) and cluster of differentiation (CD)160 (10), which may gradually result in T cell
exhaustion (11); therefore, the
inhibited function of TILs in the tumor microenvironment results in
tumor immunosuppression. Tumor cells can also secrete
immunosuppressive factors, including transforming growth factor-β
(TGF-β), interleukin-6 (IL-6), IL-10, vascular endothelial growth
factor (VEGF) and matrix metalloproteinase (5,9), in order
to cause tumor-infiltrating immune cells to inhibit their antitumor
effect.
Furthermore, abnormal metabolism of tumor cells can
also enhance the immunosuppressive effect of the tumor
microenvironment. Normal cells acquire energy primarily through
oxidative phosphorylation, and a limited number use glycolysis,
which can be inhibited under aerobic conditions; however, the
method by which tumor cells acquire energy is different, and is
termed aerobic glycolysis or ‘the Warburg effect’ (12). In this condition, tumor cells maintain
an increased rate of glycolysis even in the presence of adequate
oxygen. This was initially considered to be a strategy to adapt to
hypoxia, but it is now widely accepted that this shift of energy
metabolism is not only to produce the necessary resources for the
biosynthetic activities of tumor cells, but also to generate
numerous acidic products to form the acidic tumor microenvironment,
which results in immunosuppression (13). In addition to aerobic glycolysis, in
order to rapidly proliferate, tumor cells are also required to
increase the demand for amino acids. Among them, glutamine,
methionine, tryptophan, arginine and leucine are essential for the
tumor cells as metabolic regulators in supporting cancer cell
growth (14), and the tumor cells are
more competitive for these metabolic resources compared to
tumor-infiltrating immune cells. Additionally, indoleamine
2,3-dioxygenase (IDO), which is highly expressed by tumor cells, is
a rate-limiting enzyme of tryptophan metabolism that has regulatory
effects on T cells resulting from tryptophan depletion in tumor
microenvironments (15). In addition,
the tryptophan metabolites, including 3-hydroxyquinolinic acid via
the kynurenine pathway, can also directly inhibit T effector cells
(16). Furthermore, the metabolic
interplay between tumor cells and immune cells can contribute to
the exhaustion of TILs and immunosuppression (17).
Effect of CAF on the tumor
microenvironment
Fibroblasts are the dominant component of the tumor
stroma (18). The important functions
of fibroblasts include deposition of ECM, regulation of epithelial
differentiation, regulation of inflammation and participation in
wound healing. Activated fibroblasts, termed CAFs, are also
critical for the formation of the tumor microenvironment,
particularly for solid tumor types (19). Fibroblasts are a critical determinant
in the tumor malignant progression and represent an important
target for cancer therapies.
Fibroblast activation protein α (FAPα) is
selectively expressed on the surface of CAF, and the majority of
epithelial tumor types exhibit high expression of FAPα (20). FAPα has a dual role as a protease and
in signal transduction. The former refers to its involvement in
remodeling the construction of the microenvironment stroma by
degrading fibronectin and changing the structure of collagen, in
order to enhance the invasion ability of tumor cells along the
fibers (21). The latter refers to
its involvement with TGF-β, VEGF, stromal cell-derived factor-1,
platelet-derived growth factor, hepatocyte growth factor and other
cytokines, which could conduct signals to promote tumor growth,
prevent immune cell recruitment, inhibit the function of
tumor-infiltrating immune cells and enhance ECM proliferation for
the formation of a tumor biological barrier (22). Furthermore, the desmoplastic stroma
could then surround the tumor cells and prevent access of antitumor
drugs (23). Kraman et al
(24) confirmed that depleting
FAP-expressing cells could reduce the occurrence of hypoxic
necrosis in vitro and permit the immunological control of
growth in vivo; therefore, FAP-expressing cells are an
important immunosuppressive component of the tumor
microenvironment.
Effect of the tumor abnormal vascular
structure on the tumor microenvironment
Tumor angiogenesis is an important process in the
tumor microenvironment. Emerging evidence indicates that
angiogenesis and immunosuppression frequently occur simultaneously
in response to different stimuli (25). Tumor neovascularization is primarily
leaky, tortuous, dilated and saccular. The structural and
functional abnormalities of tumor blood vessels result in the
impaired blood supply and interstitial hypertension or high
interstitial fluid pressure (IFP) (26). The perfusion of tumor tissues is
further hampered by the formation of hypoxia and high IFP in the
microenvironment of malignant tumor types. The imbalance between
the promotion and inhibition of angiogenic factors contributes to
the abnormal structure of the tumor vasculature (27). Among a whole range of pro-angiogenic
factors that participate in physiological or pathological
angiogenesis, VEGF is the most important and also a potent
angiogenic factor that can increase the density of tumor blood
vessels (28). The sufficient
expression of VEGF depends on the oxygen concentration in tissues.
There are a variety of transcription factors in tumor tissues,
including hypoxia inducible factor (HIF), which can upregulate the
VEGF under hypoxic conditions (29).
Therefore, combined with the abnormal metabolism of
tumor cells, the suitable microenvironment for tumor survival is
characterized by low pH, hypoxia and high IFP, which is considered
to aid in rendering tumor microenvironments hostile to the immune
cells. The low pH or acidic microenvironment can accelerate the
differentiation of regulatory T cells (Tregs) and the development
of myeloid-derived suppressor cells by promoting the production of
IL-2, inhibiting the infiltration of T cells and inducing their
apoptosis, and activating tumor-associated macrophages (TAMs), in
order to secrete a large number of cytokines to promote tumor
angiogenesis (30). Hypoxia can
promote the formation of tumor blood vessels by upregulating the
expression of pro-angiogenic factors such as HIF-1, VEGF, IL-6,
TNFα, and tyrosine kinase receptor Tie2 (31), and increase the malignancy and trigger
tumor metastasis by inducing epithelial-mesenchymal transition
(EMT) (32). High IFP can prevent
immune cells from recruiting to the tumor tissue and interfere with
drug delivery. Additionally, high IFP and pro-angiogenic factors
can also evoke lymphangiogenesis, which is the important
physiological basis of lymphatic metastasis (26). Collectively, these vascular
abnormalities result in a complex immunosuppressive
microenvironment, which may promote the survival and metastasis of
tumor cells.
Heterogeneity of the tumor
microenvironment
The formation of the tumor immunosuppressive
microenvironment is a dynamic and complex process. In addition to
the significant heterogeneity of tumor cells, heterogeneity of
stroma components and immune cells can also increase the complexity
of the tumor microenvironment (8).
Additionally, tumor progression, pathological stage, treatment
efficacy and prognosis are also associated with the tumor
microenvironment, which determines the antitumor response and
remains a notable obstacle for the treatment of cancer (33). Therefore, due to the presence of tumor
microenvironmental heterogeneity, the degree of the antitumor
immune response in different individuals is variable.
The heterogeneity of stroma components in the tumor
microenvironment is common. In pancreatic, breast and prostate
cancer, and other solid tumor types with a high content of CAF, the
formation of high-density ECM will increase the tumor IFP, and
hinder the absorption of chemotherapy drugs and the infiltration of
immune cells (19). Additionally,
tumors with different types, locations and stages also exhibit
tumor vascular heterogeneity (34).
For instance, pancreatic ductal adenocarcinoma (PDA) is a
stroma-rich cancer type and its tumor environment has been
demonstrated to consist of an abundance of stroma containing
numerous cells types, but predominantly pancreatic stellate cells
(PSCs) (35). The vasculature in PDA
is notably influenced by the excessive desmoplasia caused by the
secretion of PSCs and finally leads to hypovascularity and
perfusion impairment (36), which
indicates that the role of tumor angiogenesis in the progression of
pancreatic cancer is less notable compared with that of other
hypervascular tumor types, including liver cancer. Kashiwagi et
al (37) reported that murine
melanoma cells were intracranially and subcutaneously implanted
into mice, and the results determined that the vascular density of
the intracranial tumor types was increased compared with that of
subcutaneous tissues, but that the diameter was reduced. Compared
with early renal cell carcinoma, advanced renal cell carcinoma
exhibits an increased endothelial cell proliferation fraction,
while presenting with a reduced microvessel density, which
indicates that the heterogeneity in angiogenic activity is
associated with tumor stage (38).
Additionally, these structural abnormalities of tumor vasculature
contribute to the spatial and temporal heterogeneity in tumor blood
flow, and solid pressure generated by proliferating tumor cells
compresses intratumor blood and lymphatic vessels, which further
impairs not only the blood flow, but also the lymphatic flow
(39); therefore, the heterogeneity
in the stroma of tumor microenvironment requires consideration
regarding the efficacy of immunotherapies.
The recruitment, differentiation and location of
immune cells in the tumor microenvironment are variable among
different tumor types, and their heterogeneity is also affected.
Chevrier et al (40) conducted
mass cytometry for high-dimensional single-cell analysis in order
to produce an in-depth human atlas of the tumor immune
microenvironment in patients with clear cell renal cell carcinoma,
and this demonstrated the immune cell diversity in the tumor
ecosystem and the fact that a number of specific immune signatures
could function as biomarkers associated with progression-free
survival. Additionally, the innate immune landscape in early lung
adenocarcinoma indicates that the heterogeneity of immune cells may
begin to form at an early tumor stage and evolve with the
progression of the tumor stage, gradually compromising the
antitumor immunity (41).
Furthermore, the dominant types of immune cells infiltrated in
tumor microenvironments are also different. TAMs are among the most
frequently located cells in the pancreatic tumor microenvironment,
while the majority of other tumor types are primarily dominated by
TILs (42); therefore, the usage of
agonist CD40 monoclonal antibody in PDA can activate and recruit a
large number of macrophages, which are tumoricidal and could
facilitate the depletion of the tumor stroma (43). The tumor microenvironment includes a
complex network of immune T-cell subpopulations, and the state of
activation, the location of infiltration and the density of the
tumor stroma could be different (44). Therefore, due to the heterogeneity of
immune cells in the tumor immune microenvironment, Chen et
al (45) divided it into three
phenotypes according to the distribution of immune cells, as
follows: i) Immune-inflamed phenotype, where intratumor
infiltration of CD4+ and CD8+ T cells, and
parenchyma and stroma can be observed in a large number of immune
cells; ii) immune-excluded phenotype, where immune cells cannot
penetrate the parenchyma and only exist in the stroma; and iii)
immune-desert phenotype, which a paucity of tumor-specific T cells
are located in the parenchyma or stroma. Among them, the first two
phenotypes are associated with non-inflamed tumor types. This
classification explains the heterogeneity of immune cells in the
tumor microenvironment, indicating that the tumor microenvironment
can be modified by changing the immune phenotype, and providing a
theoretical basis for personalized immunotherapy.
Immunotherapies targeted to the tumor
microenvironment
The complexity and heterogeneity of the tumor
immunosuppressive microenvironment increases the difficulty of
cancer immunotherapy and is an important reason for the variable
efficacy in immunotherapies. Unlike directly targeting the tumor
cells, the tumor microenvironment represents an increasingly
popular therapeutic target with a decreased risk of resistance and
recurrence due to the genetically stable stromal cells (46). Recently, the immune checkpoint
inhibitors have provided new hope and have become a focus of
current cancer immunotherapy (47).
The antitumor immunity points of the tumor microenvironment also
have other beneficial targets, and application together with
conventional cancer therapies can also provide a survival benefit
for increased numbers of patients with cancer. The association
between the heterogeneity of the tumor microenvironment and the
main microenvironment-targeted therapies is demonstrated in
Fig. 1; therefore, the combination or
‘cocktail’ therapy for cancer provides an increased number of
advantages compared with monotherapy, and has become an important
method to improve the efficacy of tumor immunotherapy (48).
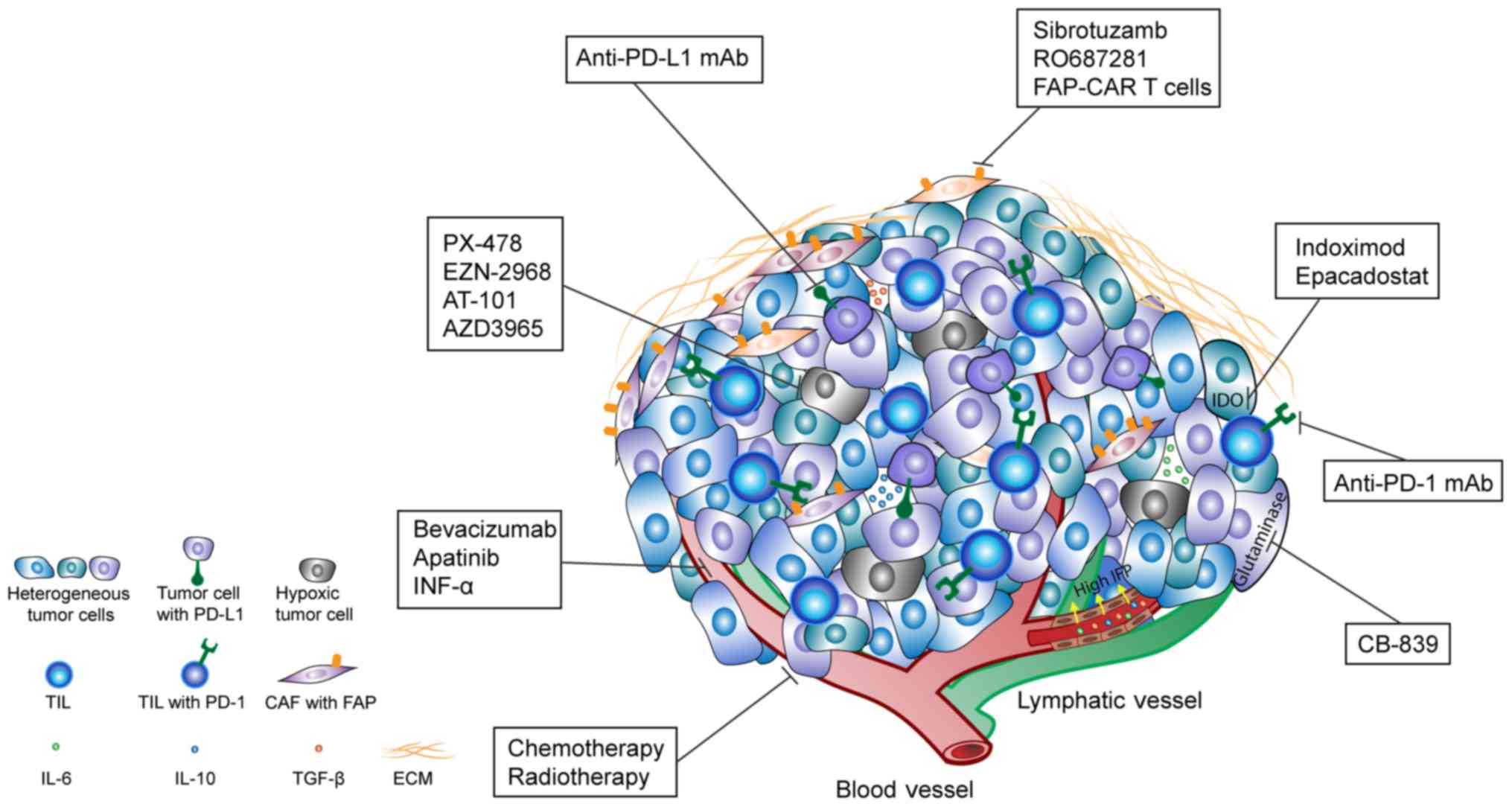 | Figure 1.Heterogeneity of the tumor
microenvironment and the main microenvironment-targeted therapies.
Tumor cells, expression of biomarkers, oxygen concentration, pH,
IFP, angiogenesis, metabolism, ECM and other intra- and extra-tumor
characters exhibit notable heterogeneity. Tumor cells can secrete
factors into the ECM, including TGF-β, IL-6 and IL-10, in order to
inhibit the function of TIL and result in tumor immunosuppression.
These corresponding therapies include: Anti-PD-1 and anti-PD-L1
antibody targeting the immunosuppressive microenvironment; IDO
inhibitor (epacadostat and indoximod) and glutaminase inhibitor
(CB-839) targeting the tumor abnormal amino acids metabolism;
hypoxia inducible factor 1α inhibitors (PX-478 and EZN-2968),
B-cell lymphoma 2 inhibitor (AT-101) and monocarboxylate
transporter inhibitor (AZD3965) targeting the hypoxic tumor cells
in the hypoxic tumor microenvironment; anti-angiogenic inhibitors
(bevacizumab, INF-α and apatinib) and FAPα inhibitor (sibrotuzumab,
RO6874281 and FAP-CAR T cells) targeting the regulation of tumor
stroma; and combination therapies with chemotherapy, radiotherapy
and other therapies. IFP, interstitial fluid pressure; TGF-β,
transforming growth factor-β; IL, interleukin; PD-1, programmed
death 1; PD-L1, PD-ligand 1; IDO, indoleamine 2,3-dioxygenase;
INF-α, interferon-α; FAP, fibroblast activation protein; FAP-CAR,
FAP-specific chimeric antigen receptor; TIL, tumor-infiltrating
lymphocyte; CAF, cancer-associated fibroblast; ECM, extracellular
matrix. |
Immune checkpoint blockade
Immune checkpoint inhibitors are strategies for
activating immune function and normalizing the tumor
microenvironment. Immune checkpoint inhibitors have become an
effective means of treating numerous tumor types (49). The anti-PD-1/PD-L1 monoclonal antibody
has been successfully used in clinical application and has already
been approved for use in numerous cancer types, including melanoma,
non-small cell lung cancer, kidney cancer and bladder cancer
(50). Clinical trials are currently
being used to determine the success of the application of the
anti-PD-1 antibody (nivolumab) for different malignant tumor types,
and the objective response rate (ORR) has been found to be
variable: 32% of melanoma, 29% of renal cell carcinoma, 17% of
non-small cell lung cancer (33% of squamous cell carcinoma and 12%
of non-squamous cell carcinoma) (51), and only 13.3% of head and neck cancer
(52). The mechanism of inhibitor
therapy is the activation of T cells, which requires an adequate
number of TILs; therefore, the heterogeneity of the antitumor
immune response is directly associated with the density of TILs.
According to Teng et al (53),
the tumor microenvironment could be stratified into four different
types based on the presence or absence of TILs and PD-L1
expression, as follows: i) Type I
(TILs+PD-L1+), where the tumor
microenvironment is PD-L1+, with TILs driving adaptive
immune resistance, indicating that it may benefit from a
single-agent anti-PD-1/L1 blockade; ii) type II
(TILs−PD-L1−), where the tumor
microenvironment is PD-L1−, with no TILs, indicating
immune ignorance; iii) type III
(TILs−PD-L1+), where the tumor
microenvironment is PD-L1+, with no TILs, indicating
intrinsic induction and that the recruitment of TILs is necessary;
and iv) type IV (TILs+PD-L1−), where the
tumor microenvironment is PD-L1−, with TILs, indicating
the role of other suppressor pathways in promoting immune
tolerance. For type II tumors, due to their inability to produce an
antitumor immune response in the tumor microenvironment, the
recruitment of T cells should be a priority. This stratification of
the tumor microenvironment can predict the clinical efficacy of
anti-PD-1/PD-L1 therapies and enable the optimal combination of
cancer therapies tailored to target different tumor
microenvironments (54). Furthermore,
the latest research demonstrated that the expression of PD-L1 on
the minimal residual disease would increase when the tumor recurs
and acquires treatment resistance, while the proportion of effector
cells could consistently express increased PD-1 and Tim-3
expression in the tumor microenvironment (55). This indicates that the expression of
immune checkpoints should be monitored dynamically, and that the
combination treatments may be valuable for improving efficacy and
preventing recurrence in patients with tumors.
Recently, anti-PD-1 antibody Keytruda
(pembrolizumab) has been approved by the FDA to treat solid tumor
with microsatellite instability-high or mismatch repair-deficient
(56). This approval confirms the
important position of the immunotherapy-targeted tumor
microenvironment in the cancer therapies and produces a foundation
for cancer immunotherapy as a major part of the combination therapy
strategy for different tumor types. With the success of PD-1/PD-L1
inhibitors in cancer immunotherapy, combination therapy with other
immune checkpoints has received increasing attention in order to
achieve greater clinical benefit. The combination therapies,
including dual immune checkpoint inhibitors, are undergoing
clinical trials. Clinical studies have demonstrated that the
anti-PD-1/PD-L1 antibodies integrated with CTLA-4 inhibitors can
increase the therapeutic efficacy and the percentage of responders
in the treatment of advanced melanoma, indicating that the
combination of immune checkpoint inhibitors can significantly
enhance antitumor immunity (57).
Currently, the indications and safety of PD-1/PD-L1 inhibitors in
combination with CTLA-4 inhibitors for potential usage have also
been investigated (58), and this
combination was approved by the US Food and Drug Administration
(FDA) for patients with BRAF V600 wild-type, unresectable or
metastatic melanoma. Recently, a phase II clinical trial (CheckMate
069) indicated that the combination of first-line nivolumab plus
ipilimumab could result in improved outcomes compared with
first-line ipilimumab alone in patients with advanced melanoma, and
the 2-year survival rates were 63.8 and 53.6%, respectively
(59). Additionally, Wei et al
(60) demonstrated the distinct
underlying mechanisms of anti-PD-1 and anti-CTLA-4 checkpoint
blockade therapies using a mass cytometry-based systems approach to
identify different subsets of exhausted T cells, which could
improve the understanding of why the combination checkpoint
blockade therapies are more effective than monotherapy. In other
words, this combination could overcome the heterogeneity of TILs.
Additionally, anti-PD-1/PD-L1 treatment can be used in combination
with the inhibition of other immune checkpoints. Notably, the
inhibition of PD-1 can stimulate other immune checkpoints expressed
on T cells and increase the resistance of anti-PD-1 therapies,
including Tim-3 (61,62) and LAG-3 (63), providing a theoretical basis for the
combination therapy. Currently, all 5 clinical trials regarding the
combined inhibition of Tim-3 and PD-1 are undergoing recruitment
(NCT02817633, NCT03099109, NCT02608268, NCT03066648 and
NCT02947165). Additionally, 8 clinical trials containing the
combination of LAG-3 and PD-1 antibodies are also recruiting
(NCT02658981, NCT01968109, NCT02061761, NCT03005782, NCT02676869,
NCT02966548, NCT02488759 and NCT02060188) (https://clinicaltrials.gov/). Claudin-low breast
cancer, an aggressive subtype that confers poor prognosis and
exhibits a high expression level of EMT genes, has been reported to
recruit Tregs to the tumor microenvironment, which inhibits an
effective antitumor immune response (64), and this study indicated that future
clinical trials should target the immunosuppressive elements in the
tumor microenvironment in combination with immune checkpoint
blockades in order to increase the efficacy.
However, the combinations of immunotherapies are not
always successful. Two independent studies (65,66)
demonstrated that the concurrent administration of the anti-PD-1
antibody and the agonist antibody to OX40, a tumor necrosis factor
family costimulatory receptor that could promote the activation and
expansion of T cells, had an adverse effect on the antitumor
response of OX40 stimulation and resulted in poor outcomes in mice.
Additionally, the antitumor effect of sequential anti-OX40 and
anti-PD-1 combination is controversial between the two studies;
therefore, the sequence and timing of immunotherapies are critical
to the success of combination therapy, and require further
investigation prior to clinical use.
The appropriate selection of immune checkpoints
inhibitors or other immunotherapies for personalized combination
therapy is an indispensable option and the underlying mechanism
requires further investigation.
Tumor metabolism regulation
Improving the immunosuppression of the tumor
microenvironment by regulating tumor metabolism is a popular
research topic. Immunotherapy with inhibition of IDO to inhibit
tumor metabolism has achieved notable results. Currently, there are
two primary drug types directed against IDO: i) Highly potent IDO
inhibitor that directly inhibits the degradation of tryptophan,
such as the drug epacadostat (67);
and ii) IDO pathway inhibitor that inhibits the degradation of
tryptophan and also reverses IDO-mediated immune suppression, such
as the drug indoximod (68).
Additionally, the safety and clinical efficacy of these two drug
types have also been confirmed in recent clinical trials (69,70).
Significant breakthroughs in the studies of tumor cell metabolism
have also provided novel options to combine with immunotherapies.
IDO inhibitor epacadostat and anti-PD-1 antibody pembrolizumab have
been demonstrated to have a promising clinical efficacy and safety
for advanced cancer types in clinical trials improved objective
response rate and disease control rate (71). Furthermore, a clinical trial has been
initiated to evaluate the preliminary efficacy of indoximod
combined with immune checkpoint inhibitors (NCT02073123);
therefore, it is possible to conclude that IDO inhibitors have a
potential synergistic effect with immune checkpoint inhibitors.
Additionally, due to the success of IDO inhibitors, other
tumor-associated amino acids with abnormal metabolism are gaining
increasing attention. For example, clinical trials of glutaminase
inhibitor CB-839 alone (NCT02861300) and combined with nivolumab
(NCT02771626) for the treatment of solid tumor types are also under
recruitment.
Hypoxic and acidic microenvironments are associated
with the consequence of tumor metabolism, and reversing them is
also being used as a strategy to regulate the tumor
microenvironment. PX-478 is a selective inhibitor that can suppress
hypoxia-induced HIF-1α levels (72).
In a previous clinical trial, patients with refractory solid tumor
types were treated with EZN-2968, a locked nucleic acid antisense
oligonucleotide against HIF-1α (73).
The number of cases was too small to veritably reflect the
efficacy, but even so, there were a number of patients who
responded to the treatment. Additionally, lactate can specifically
upregulate B-cell lymphoma 2 (Bcl-2) through translational control
and can promote resistance to the glucose starvation of tumor cells
(74). In the clinical trial of
abnormal lactate metabolism, it was determined that the use of
cisplatin and etoposide in combination with Bcl-2 inhibitor AT-101
could enhance the antitumor effect (75). Furthermore, the clinical trial
regarding the the transport of lactic acid, pyruvate and other
metabolites, and monocarboxylate transporter inhibitor AZD3965,
which could prevent the release of lactic acid by hypoxic tumor
cells and then inhibit their growth and survival, is also
recruiting (NCT01791595) (76).
Tumor stroma regulation
Regulation of the heterogenous stroma components in
the tumor microenvironment could modulate its immunosuppressive
conditions. Promoting normalization of tumor blood vessels and
weakening the function of CAFs are the key roles in effectively
transporting oxygen, drugs or immune cells and other components to
tumor tissues, reducing the tumor proliferation and invasion
(77).
The first anti-angiogenic therapy, Avastin
(bevacizumab), was approved by the FDA in 2003. Considerable effort
into the development of anti-angiogenic therapies has been
undertaken, and a number of these inhibitory agents have been
approved for clinical use against a number of cancer types;
however, tumors can frequently escape the effects of these agents,
causing the disease to eventually progress (78). Therefore, anti-angiogenic therapy may
serve a role in vessel normalization, in order to increase immune
cell infiltration and enhance the efficacies of immunotherapies
(79). The combined treatment of
bevacizumab and interferon-α has also entered phase 2 and 3
clinical trials and demonstrated improved clinical efficacy in
metastatic renal cell carcinoma, confirming the clinical value of
combined application of anti-vascular therapy and immunotherapy
when compared with monotherapy (80,81).
Furthermore, the clinical trials combined with VEGF receptor
tyrosine kinase inhibitor apatinib and PD-1 inhibitor are currently
recruiting, in order to evaluate the efficacy for the treatment of
gastric cancer types (NCT03092895 and NCT02942329).
Additionally, there are several studies have also
attempted to improve the immunosuppressive condition by modulating
the function of CAFs in the tumor microenvironment. The humanized
monoclonal antibody sibrotuzumab, which is directed against the
specific antigen FAPα on CAFs, could block its dual function of
protease and signal transduction, then inhibit tumor progression,
invasion and metastasis progression, and reduce its negative
regulation of antitumor immunity. In phase 1 and 2 clinical trials
using sibrotuzumab alone (82,83), only
a limited number of patients achieved stable disease and the
expected clinical response rate was not met; however, whether the
efficacy of the treatment could be improved by combining it with
other immunotherapies requires further investigation. RO6874281 is
a bispecific antibody containing an IL-2 variant targeting FAPα.
The IL-2 variant does not bind to Tregs, which could prevent the
immunosuppressive capacity of the Tregs (84). By specifically targeting FAPα, the
antibody could not only increase the local IL-2 concentration, in
order to activate the immune effector cells in the tumor
microenvironment, but also inhibit the deterioration of the tumor
microenvironment by directly blocking FAPα. Due to the consistent
expression of FAP in the tumor stroma, modified T cells that
express a FAP-specific chimeric antigen receptor have also been
engineered to inhibit the tumor proliferation and augment host
immunity (85–87). Clinical trials regarding monotherapy
(NCT02627274) and combination with other immunotherapies
(NCT03063762) are currently being conducted to evaluate the safety,
tolerability and preliminary therapeutic efficacy.
Therefore, studies regarding the stroma components
in the tumor microenvironment will continue to be conducted in
order to improve the effectiveness of the tumor immunosuppression
and provide a novel alternative approach for personalized
combination therapy.
Combination with chemotherapy and
radiotherapy
Immunotherapy combined with traditional radiotherapy
and chemotherapy has received increasing attention. Different
chemotherapeutic drugs have different immunological mechanisms
underlying the efficacy of the cancer therapy (88), including: i) Increased immunogenicity
resulting in tumor cell apoptosis from drugs such as anthracycline,
5-fluorouracil (5-Fu) and oxaliplatin; ii) direct immunostimulation
activating the tumor immunity of immune effector cells from such as
gemcitabine, paclitaxel and pemetrexed; and iii) indirect
immunostimulation inhibiting the immunosuppressive cells from drugs
such as 5-Fu, cyclophosphamide and oxaliplatin. Additionally,
radiotherapy can also influence the tumor immune response. Tumor
cell death from irradiation can enhance the antitumor immunity by
inducing antigen expression on tumor cells and activating
lymphocytes (89,90), and by generating the abscopal effect
(91). Chemotherapy or radiotherapy
can eliminate a number of the tumor cells in advance, then expose a
large number of the tumor antigens and neoantigen products in the
microenvironment, which could recruit increased numbers of immune
effector cells, and finally improve the immunosuppressive state of
the tumor microenvironment. Currently, the rationale for combining
immunotherapy with chemotherapy and radiotherapy has been verified
(92), and preclinical studies have
also been well investigated (93,94);
therefore, it is plausible that combining immunotherapy with
standard conventional therapies, including chemotherapy or
radiotherapy, will provide synergistic antitumor effects (95), but the most beneficial dose and the
appropriate time requires investigation (89,96).
Conclusions and perspectives
The tumor immunosuppressive microenvironment is in a
dynamic status and is coordinated by multiple immunosuppressive
signals in the regulatory network. In the course of clinical cancer
treatment, due to the tumor types, stages, histological features
and other microenvironment-associated factors, heterogeneity of the
tumor microenvironment will cause immunosuppression and then result
in the differences in the efficacy of immunotherapies. Despite the
success in targeting non-tumor cell components, including immune
checkpoint blockade, focusing on a single immunosuppressive target
is ineffective in the majority of patients with cancer. Even among
the cancer types that do respond to the immune checkpoint
inhibitors, including melanoma, non-small cell lung cancer and
renal cell cancer, few patients exhibit objective control of tumor
progression. Following blocking or inhibiting of one
immunosuppressive signal, the tumor will compensate through other
mechanisms to generate the resistance and reduce the efficacy of
this immunotherapy. The association between heterogeneity of the
tumor microenvironment and the immunotherapy response remains a
significant challenge.
In the future, immunotherapy may be required to be
tailored for each patient with cancer according to the tumor
microenvironment. The application of novel immune biomarkers and
the ability to monitor and evaluate the tumor microenvironment by
novel strategies, in order to improve early cancer diagnosis and
predict the therapeutic efficacy and prognosis, requires further
investigation. Personalized immunotherapy based on individual
genetic, molecular and immune profiling has the potential to
produce the most optimized outcomes for patients with cancer, but
healthcare costs must be kept in an affordable range. Notably, the
combinations must be designed in a rational and safe manner, and
further clinical trials should be conducted, in order to verify the
combination therapies prior to progressing to clinical use.
Acknowledgments
Not applicable.
Funding
The present study was supported by the National Key
Research and Development Program of China (grant no.
2016YFC1303800), the National Health and Family Planning Commission
of China (grant no. ZX-07-C2016004), the Key Laboratory
Construction Project of Science and Technology Department in Jilin
Province (grant no. 20170622011JC), the Industrial Technology
Research and Development Special Project of Development and Reform
Commission in Jilin Province (grant no. 2017C022) and the
Development and Reform Commission project in Jilin Province (grant
no. 2014N147).
Availability of data and materials
Not applicable.
Authors' contributions
YY was responsible for the literature search and
manuscript preparation. JC was responsible for study design,
manuscript co-writing and correction. Both authors revised the
article and approved the final version for publication.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Whiteside TL, Demaria S, Rodriguez-Ruiz
ME, Zarour HM and Melero I: Emerging opportunities and challenges
in cancer immunotherapy. Clin Cancer Res. 22:1845–1855. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hodi FS, O'Day SJ, McDermott DF, Weber RW,
Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel
JC, et al: Improved survival with ipilimumab in patients with
metastatic melanoma. N Engl J Med. 363:711–723. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Topalian SL, Sznol M, McDermott DF, Kluger
HM, Carvajal RD, Sharfman WH, Brahmer JR, Lawrence DP, Atkins MB,
Powderly JD, et al: Survival, durable tumor remission, and
long-term safety in patients with advanced melanoma receiving
nivolumab. J Clin Oncol. 32:1020–1030. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tang H, Qiao J and Fu YX: Immunotherapy
and tumor microenvironment. Cancer Lett. 370:85–90. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Beatty GL and Gladney WL: Immune escape
mechanisms as a guide for cancer immunotherapy. Clin Cancer Res.
21:687–692. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Klemm F and Joyce JA: Microenvironmental
regulation of therapeutic response in cancer. Trends Cell Biol.
25:198–213. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Swartz MA, Iida N, Roberts EW, Sangaletti
S, Wong MH, Yull FE, Coussens LM and DeClerck YA: Tumor
microenvironment complexity: Emerging roles in cancer therapy.
Cancer Res. 72:2473–2480. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Junttila MR and de Sauvage FJ: Influence
of tumour micro-environment heterogeneity on therapeutic response.
Nature. 501:346–354. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Becker JC, Andersen MH, Schrama D and
Straten Thor P: Immune-suppressive properties of the tumor
microenvironment. Cancer Immunol Immunother. 62:1137–1148. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen L and Flies DB: Molecular mechanisms
of T cell co-stimulation and co-inhibition. Nat Rev Immunol.
13:227–242. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wherry EJ: T cell exhaustion. Nat Immunol.
12:492–499. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Warburg O, Wind F and Negelein E: The
metabolism of tumors in the body. J Gen Physiol. 8:519–530. 1927.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vander Heiden MG, Cantley LC and Thompson
CB: Understanding the Warburg effect: The metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Walls J, Sinclair L and Finlay D: Nutrient
sensing, signal transduction and immune responses. Semin Immunol.
28:396–407. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fallarino F, Grohmann U, Vacca C, Bianchi
R, Orabona C, Spreca A, Fioretti MC and Puccetti P: T cell
apoptosis by tryptophan catabolism. Cell Death Differ. 9:1069–1077.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Munn DH and Mellor AL: Indoleamine 2,3
dioxygenase and metabolic control of immune responses. Trends
Immunol. 34:137–143. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jiang Y, Li Y and Zhu B: T-cell exhaustion
in the tumor microenvironment. Cell Death Dis. 6:e17922015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kalluri R and Zeisberg M: Fibroblasts in
cancer. Nat Rev Cancer. 6:392–401. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kalluri R: The biology and function of
fibroblasts in cancer. Nat Rev Cancer. 16:582–598. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fearon DT: The carcinoma-associated
fibroblast expressing fibroblast activation protein and escape from
immune surveillance. Cancer Immunol Res. 2:187–193. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lee HO, Mullins SR, Franco-Barraza J,
Valianou M, Cukierman E and Cheng JD: FAP-overexpressing
fibroblasts produce an extracellular matrix that enhances invasive
velocity and directionality of pancreatic cancer cells. BMC Cancer.
11:2452011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shimoda M, Mellody KT and Orimo A:
Carcinoma-associated fibroblasts are a rate-limiting determinant
for tumour progression. Semin Cell Dev Biol. 21:19–25. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rucki AA and Zheng L: Pancreatic cancer
stroma: Understanding biology leads to new therapeutic strategies.
World J Gastroenterol. 20:2237–2246. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kraman M, Bambrough PJ, Arnold JN, Roberts
EW, Magiera L, Jones JO, Gopinathan A, Tuveson DA and Fearon DT:
Suppression of antitumor immunity by stromal cells expressing
fibroblast activation protein-alpha. Science. 330:827–830. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Motz GT and Coukos G: The parallel lives
of angiogenesis and immunosuppression: Cancer and other tales. Nat
Rev Immunol. 11:702–711. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rofstad EK, Galappathi K and Mathiesen BS:
Tumor interstitial fluid pressure-a link between tumor hypoxia,
microvascular density, and lymph node metastasis. Neoplasia.
16:586–594. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Goel S, Duda DG, Xu L, Munn LL, Boucher Y,
Fukumura D and Jain RK: Normalization of the vasculature for
treatment of cancer and other diseases. Physiol Rev. 91:1071–1121.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Carmeliet P and Jain RK: Molecular
mechanisms and clinical applications of angiogenesis. Nature.
473:298–307. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shweiki D, Itin A, Soffer D and Keshet E:
Vascular endothelial growth factor induced by hypoxia may mediate
hypoxia-initiated angiogenesis. Nature. 359:843–845. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Calcinotto A, Filipazzi P, Grioni M, Iero
M, De Milito A, Ricupito A, Cova A, Canese R, Jachetti E, Rossetti
M, et al: Modulation of microenvironment acidity reverses anergy in
human and murine tumor-infiltrating T lymphocytes. Cancer Res.
72:2746–2756. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Balamurugan K: HIF-1 at the crossroads of
hypoxia, inflammation, and cancer. Int J Cancer. 138:1058–1066.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu MZ, Tsai YP, Yang MH, Huang CH, Chang
SY, Chang CC, Teng SC and Wu KJ: Interplay between HDAC3 and WDR5
is essential for hypoxia-induced epithelial-mesenchymal transition.
Mol Cell. 43:811–822. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pottier C, Wheatherspoon A, Roncarati P,
Longuespée R, Herfs M, Duray A, Delvenne P and Quatresooz P: The
importance of the tumor microenvironment in the therapeutic
management of cancer. Expert Rev Anticancer Ther. 15:943–954. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jain RK: Normalizing tumor
microenvironment to treat cancer: Bench to bedside to biomarkers. J
Clin Oncol. 31:2205–2218. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Apte M, Pirola RC and Wilson JS:
Pancreatic stellate cell: Physiologic role, role in fibrosis and
cancer. Curr Opin Gastroenterol. 31:416–423. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Feig C, Gopinathan A, Neesse A, Chan DS,
Cook N and Tuveson DA: The pancreas cancer microenvironment. Clin
Cancer Res. 18:4266–4276. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kashiwagi S, Izumi Y, Gohongi T, Demou ZN,
Xu L, Huang PL, Buerk DG, Munn LL, Jain RK and Fukumura D: NO
mediates mural cell recruitment and vessel morphogenesis in murine
melanomas and tissue-engineered blood vessels. J Clin Invest.
115:1816–1827. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Baldewijns MM, Thijssen VL, Van den Eynden
GG, Van Laere SJ, Bluekens AM, Roskams T, van Poppel H, De Bruine
AP, Griffioen AW and Vermeulen PB: High-grade clear cell renal cell
carcinoma has a higher angiogenic activity than low-grade renal
cell carcinoma based on histomorphological quantification and
qRT-PCR mRNA expression profile. Br J Cancer. 96:1888–1895. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Padera TP, Stoll BR, Tooredman JB, Capen
D, di Tomaso E and Jain RK: Pathology: Cancer cells compress
intratumour vessels. Nature. 427:6952004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chevrier S, Levine JH, Zanotelli VRT,
Silina K, Schulz D, Bacac M, Ries CH, Ailles L, Jewett MAS, Moch H,
et al: An immune atlas of clear cell renal cell carcinoma. Cell.
169(736–749): e182017.
|
|
41
|
Lavin Y, Kobayashi S, Leader A, Amir ED,
Elefant N, Bigenwald C, Remark R, Sweeney R, Becker CD, Levine JH,
et al: Innate immune landscape in early lung adenocarcinoma by
paired single-cell analyses. Cell. 169(750–765): e172017.
|
|
42
|
Mielgo A and Schmid MC: Impact of tumour
associated macrophages in pancreatic cancer. BMB Rep. 46:131–138.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Beatty GL, Chiorean EG, Fishman MP,
Saboury B, Teitelbaum UR, Sun W, Huhn RD, Song W, Li D, Sharp LL,
et al: CD40 agonists alter tumor stroma and show efficacy against
pancreatic carcinoma in mice and humans. Science. 331:1612–1616.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tosolini M, Kirilovsky A, Mlecnik B,
Fredriksen T, Mauger S, Bindea G, Berger A, Bruneval P, Fridman WH,
Pages F, et al: Clinical impact of different classes of
infiltrating T cytotoxic and helper cells (Th1, th2, treg, th17) in
patients with colorectal cancer. Cancer Res. 71:1263–1271. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen DS and Mellman I: Elements of cancer
immunity and the cancer-immune set point. Nature. 541:321–330.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Quail DF and Joyce JA: Microenvironmental
regulation of tumor progression and metastasis. Nat Med.
19:1423–1437. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Li Y, Li F, Jiang F, Lv X, Zhang R, Lu A
and Zhang G: A Mini-review for cancer immunotherapy: Molecular
understanding of PD-1/PD-L1 pathway & translational blockade of
immune checkpoints. Int J Mol Sci. 17:pii: E1151. 2016.
|
|
48
|
Ledford H: Cocktails for cancer with a
measure of immunotherapy. Nature. 532:162–164. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Sharma P and Allison JP: The future of
immune checkpoint therapy. Science. 348:56–61. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Page DB, Bourla AB, Daniyan A, Naidoo J,
Smith E, Smith M, Friedman C, Khalil DN, Funt S, Shoushtari AN, et
al: Tumor immunology and cancer immunotherapy: Summary of the 2014
SITC primer. J Immunother Cancer. 3:252015. View Article : Google Scholar
|
|
51
|
Gunturi A and McDermott DF: Nivolumab for
the treatment of cancer. Expert Opin Investig Drugs. 24:253–260.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ferris RL, Blumenschein G Jr, Fayette J,
Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE,
Even C, et al: Nivolumab for recurrent squamous-cell carcinoma of
the head and neck. N Engl J Med. 375:1856–1867. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Teng MW, Ngiow SF, Ribas A and Smyth MJ:
Classifying cancers based on t-cell infiltration and pd-l1. Cancer
Res. 75:2139–2145. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Smyth MJ, Ngiow SF, Ribas A and Teng MW:
Combination cancer immunotherapies tailored to the tumour
microenvironment. Nat Rev Clin Oncol. 13:143–158. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kottke T, Evgin L, Shim KG, Rommelfanger
D, Boisgerault N, Zaidi S, Diaz RM, Thompson J, Ilett E, Coffey M,
et al: Subversion of NK-cell and TNFα immune surveillance drives
tumor recurrence. Cancer Immunol Res. 5:1029–1045. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
U.S. Food and drug administration:
Treatment approved for Any Solid tumor with biomarker. Onco Times.
39:52–53. 2017. View Article : Google Scholar
|
|
57
|
Baumeister SH, Freeman GJ, Dranoff G and
Sharpe AH: Coinhibitory pathways in immunotherapy for cancer. Annu
Rev Immunol. 34:539–573. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Boutros C, Tarhini A, Routier E, Lambotte
O, Ladurie FL, Carbonnel F, Izzeddine H, Marabelle A, Champiat S,
Berdelou A, et al: Safety profiles of anti-CTLA-4 and anti-PD-1
antibodies alone and in combination. Nat Rev Clin Oncol.
13:473–486. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Hodi FS, Chesney J, Pavlick AC, Robert C,
Grossmann KF, McDermott DF, Linette GP, Meyer N, Giguere JK,
Agarwala SS, et al: Combined nivolumab and ipilimumab versus
ipilimumab alone in patients with advanced melanoma: 2-year overall
survival outcomes in a multicentre, randomised, controlled, phase 2
trial. Lancet Oncol. 17:1558–1568. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wei SC, Levine JH, Cogdill AP, Zhao Y,
Anang NAS, Andrews MC, Sharma P, Wang J, Wargo JA, Pe'er D and
Allison JP: Distinct cellular mechanisms underlie anti-CTLA-4 and
anti-PD-1 checkpoint blockade. Cell. 170(1120–1133): e172017.
|
|
61
|
Shayan G, Srivastava R, Li J, Schmitt N,
Kane LP and Ferris RL: Adaptive resistance to anti-PD1 therapy by
Tim-3 upregulation is mediated by the PI3K-Akt pathway in head and
neck cancer. Oncoimmunology. 6:e12617792016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Koyama S, Akbay EA, Li YY, Herter-Sprie
GS, Buczkowski KA, Richards WG, Gandhi L, Redig AJ, Rodig SJ,
Asahina H, et al: Adaptive resistance to therapeutic PD-1 blockade
is associated with upregulation of alternative immune checkpoints.
Nat Commun. 7:105012016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Foy SP, Sennino B, dela Cruz T, Cote JJ,
Gordon EJ, Kemp F, Xavier V, Franzusoff A, Rountree RB and Mandl
SJ: Poxvirus-based active immunotherapy with PD-1 and LAG-3 dual
immune checkpoint inhibition overcomes compensatory immune
regulation, Yielding complete tumor regression in mice. PLoS One.
11:e01500842016. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Taylor NA, Vick SC, Iglesia MD, Brickey
WJ, Midkiff BR, McKinnon KP, Reisdorf S, Anders CK, Carey LA,
Parker JS, et al: Treg depletion potentiates checkpoint inhibition
in claudin-low breast cancer. J Clin Invest. 127:3472–3483. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Messenheimer DJ, Jensen SM, Afentoulis ME,
Wegmann KW, Feng Z, Friedman DJ, Gough MJ, Urba WJ and Fox BA:
Timing of PD-1 blockade is critical to effective combination
immunotherapy with anti-OX40. Clin Cancer Res. 23:6165–6177. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Shrimali RK, Ahmad S, Verma V, Zeng P,
Ananth S, Gaur P, Gittelman RM, Yusko E, Sanders C, Robins H, et
al: Concurrent PD-1 blockade negates the effects of OX40 agonist
antibody in combination immunotherapy through inducing T-cell
apoptosis. Cancer Immunol Res. 5:755–766. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Yue EW, Sparks R, Polam P, Modi D, Douty
B, Wayland B, Glass B, Takvorian A, Glenn J, Zhu W, et al:
INCB24360 (Epacadostat), a highly potent and selective
indoleamine-2,3-dioxygenase 1 (IDO1) inhibitor for immuno-oncology.
ACS Med Chem Lett. 8:486–491. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zhai L, Spranger S, Binder DC, Gritsina G,
Lauing KL, Giles FJ and Wainwright DA: Molecular pathways:
Targeting IDO1 and other tryptophan dioxygenases for cancer
immunotherapy. Clin Cancer Res. 21:5427–5433. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Beatty GL, O'Dwyer PJ, Clark J, Shi JG,
Bowman KJ, Scherle PA, Newton RC, Schaub R, Maleski J, Leopold L,
et al: First-in-human phase I study of the oral inhibitor of
indoleamine 2,3-dioxygenase-1 epacadostat (INCB024360) in patients
with advanced solid malignancies. Clin Cancer Res. 23:3269–3276.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Soliman HH, Minton SE, Han HS, Ismail-Khan
R, Neuger A, Khambati F, Noyes D, Lush R, Chiappori AA, Roberts JD,
et al: A phase I study of indoximod in patients with advanced
malignancies. Oncotarget. 7:22928–22938. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Gangadhar TC, Hamid O, Smith DC, Bauer TM,
Wasser JS, Luke JJ, Balmanoukian AS, Kaufman DR, Zhao Y, Maleski J,
et al: Preliminary results from a Phase I/II study of epacadostat
(incb024360) in combination with pembrolizumab in patients with
selected advanced cancers. J Immunother Cancer. 3 Suppl 2:O72015.
View Article : Google Scholar
|
|
72
|
Zhu Y, Zang Y, Zhao F, Li Z, Zhang J, Fang
L, Li M, Xing L, Xu Z and Yu J: Inhibition of HIF-1α by PX-478
suppresses tumor growth of esophageal squamous cell cancer in vitro
and in vivo. Am J Cancer Res. 7:1198–1212. 2017.PubMed/NCBI
|
|
73
|
Jeong W, Rapisarda A, Park SR, Kinders RJ,
Chen A, Melillo G, Turkbey B, Steinberg SM, Choyke P, Doroshow JH,
et al: Pilot trial of EZN-2968, an antisense oligonucleotide
inhibitor of hypoxia-inducible factor-1 alpha (HIF-1α), in patients
with refractory solid tumors. Cancer Chemother Pharmacol.
73:343–348. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Huang C, Sheng S, Li R, Sun X, Liu J and
Huang G: Lactate promotes resistance to glucose starvation via
upregulation of Bcl-2 mediated by mTOR activation. Oncol Rep.
33:875–884. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Schelman WR, Mohammed TA, Traynor AM,
Kolesar JM, Marnocha RM, Eickhoff J, Keppen M, Alberti DB, Wilding
G, Takebe N and Liu G: A phase I study of AT-101 with cisplatin and
etoposide in patients with advanced solid tumors with an expanded
cohort in extensive-stage small cell lung cancer. Invest New Drugs.
32:295–302. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Kershaw S, Cummings J, Morris K, Tugwood J
and Dive C: Optimisation of immunofluorescence methods to determine
MCT1 and MCT4 expression in circulating tumour cells. BMC Cancer.
15:3872015. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Tarallo V and De Falco S: The vascular
endothelial growth factors and receptors family: Up to now the only
target for anti-angiogenesis therapy. Int J Biochem Cell Bio.
64:185–189. 2015. View Article : Google Scholar
|
|
78
|
Bueno MJ, Mouron S and Quintela-Fandino M:
Personalising and targeting antiangiogenic resistance: A complex
and multifactorial approach. Br J Cancer. 116:1119–1125. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Tian L, Goldstein A, Wang H, Lo Ching H,
Kim Sun I, Welte T, Sheng K, Dobrolecki LE, Zhang X, Putluri N, et
al: Mutual regulation of tumour vessel normalization and
immunostimulatory reprogramming. Nature. 544:250–254. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Melichar B, Bracarda S, Matveev V,
Alekseev B, Ivanov S, Zyryanov A, Janciauskiene R, Fernebro E,
Mulders P, Osborne S, et al: A multinational phase II trial of
bevacizumab with low-dose interferon-α2a as first-line treatment of
metastatic renal cell carcinoma: BEVLiN. Ann Oncol. 24:2396–2402.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Rini BI, Bellmunt J, Clancy J, Wang K,
Niethammer AG, Hariharan S and Escudier B: Randomized phase III
trial of temsirolimus and bevacizumab versus interferon alfa and
bevacizumab in metastatic renal cell carcinoma: INTORACT trial. J
Clin Oncol. 32:752–759. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Scott AM, Wiseman G, Welt S, Adjei A, Lee
FT, Hopkins W, Divgi CR, Hanson LH, Mitchell P, Gansen DN, et al: A
phase I dose-escalation study of sibrotuzumab in patients with
advanced or metastatic fibroblast activation protein-positive
cancer. Clin Cancer Res. 9:1639–1647. 2003.PubMed/NCBI
|
|
83
|
Hofheinz RD, al-Batran SE, Hartmann F,
Hartung G, Jäger D, Renner C, Tanswell P, Kunz U, Amelsberg A,
Kuthan H and Stehle G: Stromal antigen targeting by a humanised
monoclonal antibody: An early phase II trial of sibrotuzumab in
patients with metastatic colorectal cancer. Onkologie. 26:44–48.
2003.PubMed/NCBI
|
|
84
|
Arenas-Ramirez N, Woytschak J and Boyman
O: Interleukin-2: Biology, design and application. Trends Immunol.
36:763–777. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Kakarla S, Chow KK, Mata M, Shaffer DR,
Song XT, Wu MF, Liu H, Wang LL, Rowley DR, Pfizenmaier K, et al:
Antitumor effects of chimeric receptor engineered human T cells
directed to tumor stroma. Mol Ther. 21:1611–1620. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Tran E, Chinnasamy D, Yu Z, Morgan RA, Lee
CC, Restifo NP and Rosenberg SA: Immune targeting of fibroblast
activation protein triggers recognition of multipotent bone marrow
stromal cells and cachexia. J Exp Med. 210:1125–1135. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Wang LC, Lo A, Scholler J, Sun J, Majumdar
RS, Kapoor V, Antzis M, Cotner CE, Johnson LA, Durham AC, et al:
Targeting fibroblast activation protein in tumor stroma with
chimeric antigen receptor T cells can inhibit tumor growth and
augment host immunity without severe toxicity. Cancer Immunol Res.
2:154–166. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Galluzzi L, Zitvogel L and Kroemer G:
Immunological mechanisms underneath the efficacy of cancer therapy.
Cancer Immunol Res. 4:895–902. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Teng F, Kong L, Meng X, Yang J and Yu J:
Radiotherapy combined with immune checkpoint blockade
immunotherapy: Achievements and challenges. Cancer Lett. 365:23–29.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Kalbasi A, June CH, Haas N and Vapiwala N:
Radiation and immunotherapy: A synergistic combination. J Clin
Invest. 123:2756–2763. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Park B, Yee C and Lee KM: The effect of
radiation on the immune response to cancers. Int J Mol Sci.
15:927–943. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Dalgleish AG: Rationale for combining
immunotherapy with chemotherapy. Immunotherapy. 7:309–316. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Wargo JA, Reuben A, Cooper ZA, Oh KS and
Sullivan RJ: Immune effects of chemotherapy, radiation, and
targeted therapy and opportunities for combination with
immunotherapy. Semin Oncol. 42:601–616. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Sharabi AB, Lim M, DeWeese TL and Drake
CG: Radiation and checkpoint blockade immunotherapy:
Radiosensitisation and potential mechanisms of synergy. Lancet
Oncol. 16:e498–e509. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Shahabi V, Postow MA, Tuck D and Wolchok
JD: Immune-priming of the tumor microenvironment by radiotherapy:
Rationale for combination with immunotherapy to improve anticancer
efficacy. Am J Clin Oncol. 38:90–97. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Hughes PE, Caenepeel S and Wu LC: Targeted
therapy and checkpoint immunotherapy combinations for the treatment
of cancer. Trends Immunol. 37:462–476. 2016. View Article : Google Scholar : PubMed/NCBI
|















