Introduction
Endometrial cancer (EC) is a group of endometrial
malignant tumours that often occur in perimenopausal and
postmenopausal women. With >300,000 newly diagnosed tumours
worldwide every year, EC is the third most common gynaecological
malignant tumour (1). In the last 10
years, the incidence of EC has been increasing in China, ranking
second only to cervical cancer among female reproductive system
malignant tumours; however, the molecular mechanism of its
pathogenesis has not yet been elucidated (2).
Currently, EC lacks specific markers for diagnosis
and treatment and clinical diagnosis depends on endometrial biopsy,
which is obtained using invasive methods. When EC is in the early
stages of the disease the cure rate is as high as 83% but the
survival rate decreases sharply to <20% after late diagnosis
(2). Only ~17% of patients with
distant metastasis can be cured (3,4).
Previous data have demonstrated a significant increase in the
proportion of patients with advanced EC where survival rate has
been minimal despite improved treatments (5).
Proteomics have been extensively studied in various
types of cancer and potential biomarkers of EC have been studied,
including serum amyloid A and α-1-β glycoprotein (1,6,7). In the present study, proteomics
technology was used to detect the differential protein expression
profiles in human EC and paracancerous tissues.
DPT is a tyrosine-rich extracellular matrix protein
originally isolated from bovine dermal extracts that increases the
formation of collagen fibres (8).
DPT has a variety of biological functions in pathophysiological
processes, such as regulating the interaction between decorin and
transforming growth factor-β (TGF-β) and increasing the biological
activity of TGF-β (9). In addition,
DPT has an R-G-A-T sequence that can interact with integrins and is
similar to the R-G-D sequence that binds integrins (10). DPT combines with the extracellular
matrix receptor integrin α3β1 and promotes the formation of
abnormal blood vessels in the tumour microenvironment by regulating
TGF-β and integrin α3β1 (11).
The present study purposed to detect the
differential proteins in EC using a label-free quantification (LFQ)
method based on liquid chromatography-tandem mass spectrometry
(LC-MS/MS) and to investigate the proteins associated with the
development of EC. It was first elucidated the expression of DPT in
EC by RT-qPCR, western blotting and IHC. Furthermore, the current
study provided evidence demonstrating that the expression level of
DPT might be intimately related to the pathogenesis of EC.
Materials and methods
Tissue specimens
Tissue samples from EC resection operations were
collected from the Department of Obstetrics and Gynaecology of
Xuzhou Central Hospital (Xuzhou, China) between January 2017 to
June 2020. Postoperative pathology performed by independent
pathologists confirmed endometrial carcinoma in 15 cases of
cancerous tissue and paracancerous tissue samples. A total of 75 EC
tissue wax blocks were collected from female patients with a mean
age of 51.7 years (range, 42–76 years), and staged according to the
International Federation of Gynaecology and Obstetrics
[International Federation of Gynaecology and Obstetrics (FIGO)
2009] (12). Thirty normal
endometrial tissue wax blocks were taken from female patients with
an average age of 50.9 years (range, 46–58 years) who underwent
total hysterectomy due to uterine fibroids or adenomyomas during
the same time period. The postoperative pathological identification
of these tissues was performed by independent pathologists and
showed a normal proliferative phase of the endometrium.
Tissue samples were collected within 30 min after
the removal of the uterine appendages. Paracancerous tissues were
defined as the tissue within 3-cm outside of the 1-cm edge of the
cancer tissue. Paracancerous tissues were collected first, followed
by cancer tissues. Then, the samples were cut into multiple tissue
blocks with a diameter of ~0.5-cm and stored in a −80°C freezer.
All the cancer tissue samples were pathologically diagnosed as
endometrioid carcinoma and all the paracancerous tissue samples
were pathologically diagnosed as non-tumour tissue invasion in the
pathology department of Xuzhou Central Hospital. All subjects met
the following criteria: i) Pathological diagnosis of endometrioid
carcinoma, ii) complete clinical characteristics, iii) no
preoperative chemoradiotherapy or endocrine therapy and iv) no
history of chronic disease or other malignant tumour. This study
was approved by The Ethics Committee of Xuzhou Central Hospital
(Xuzhou, China) and all patients provided informed written
consent.
Proteomics analysis
A total of three cancer and three paracancerous
tissue samples were selected from the 15 tissues collected from the
Department of Obstetrics and Gynaecology. After weighing and lysis
using 8 M urea (Sigma-Aldrich; Merck KGaA) and 1% protease
inhibitor cocktail (Thermo Fisher Scientific, Inc.)], the
supernatant was collected to determine the protein concentration of
the samples. Equal amounts of proteins (~50 µg) from samples were
reduced with 10 mM dithiothreitol (Thermo Fisher Scientific, Inc.)
and alkylated with 50 mM iodoacetamide (Bio-Rad Laboratories,
Inc.). Solution digestion was then performed with sequencing grade
modified trypsin at 37°C overnight. The peptides were acidified
with a final concentration of 0.5–1% trifluoroacetic acid (Thermo
Fisher Scientific, Inc.). Sodium deoxycholate (Sigma-Aldrich; Merck
KGaA) was removed using high-speed centrifugation (14,000 × g at
4°C for 20 min). Tryptic peptides were desalted and centrifuged in
a SpeedVac to dry. Then, tryptic peptides were redissolved in 0.1%
formic acid (Thermo Fisher Scientific, Inc.).
After the enzymatic hydrolysis and pre-treatment of
the samples, the LFQ method based on LC-MS/MS technology was used
for peptide separation and mass spectrometry analysis of each
sample by higher-energy collisional dissociation (HCD) (ionization
mode, positive). LC-MS/MS was conducted to obtain primary mass
spectrometry of all ions and the first 20 peptides with the signal
strength of the primary spectrum peak were identified using
secondary MS. For LC-MS/MS analysis, the peptides were separated
using 90 min gradient elution at a flow rate of 2.2ⅹ10−7
l/min (nitrogen gas temperature, 320°C; nebulizer pressure, 120
psi) using a Thermo Scientific™ EASY-nLC™ 1000 HPLC system (Thermo
Fisher Scientific, Inc.), which was directly interfaced with a
Thermo Scientific Q Exactive™ mass spectrometer. Mobile phase A
consisted of 0.1% formic acid and mobile phase B consisted of
acetonitrile with 0.1% formic acid. The Q Exactive™ mass
spectrometer was operated in the data-dependent acquisition mode
using Xcalibur 2.2 SP1 software (Thermo Fisher Scientific, Inc.)
and there was a single full-scan mass spectrum in the orbitrap
(300–2,000 m/z; 70,000 resolution) followed by 20 data-dependent
MS/MS scans at 27% normalized collision energy (HCD). The mass
spectrometry proteomics data have been deposited to the
ProteomeXchange Consortium via the PRIDE (13) partner repository with the dataset
identifier PXD0242499.
Bioinformatics and statistical
analysis
A total of 74,811 sequence entries from the human
proteome database, 20,350 entries from Swiss-Prot and 544,618
entries from TrEMBL were downloaded from the UniProt Knowledgebase
(https://www.uniprot.org/proteomes/UP000005640) on
December 20, 2019 and imported into the MaxQuant-associated
Andromeda search engine (version 1.6) (14). Following protein identification, the
intensity of each identified protein was calculated using peptide
signal intensities. A fold-change (FC) ≥2 and P<0.01 were set as
the screening benchmark of significantly enhanced or weakened
differential expression. Then, the LogFC value was drawn on the
abscissa and the -log10 (P-value) value was drawn on the ordinate
to generate a volcano map. TopGO software (15) (version 2.3.0) was used to conduct the
Gene Ontology (GO) (http://geneontology.org/) analysis of the differential
proteins from the three levels of biological process, cell
component and molecular function. Then, using KOBAS software
(16) (version 3.0), the gene ID
corresponding to the differential protein was searched using the
gene name via BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi). Genes
enriched in the corresponding pathways were identified using the
Kyoto Encyclopaedia of Genes and Genomes (KEGG) database
(http://www.genome.jp/kegg/) and
differential proteins were analysed based on KEGG pathway
enrichment.
Reverse transcription quantitative PCR
(RT-qPCR)
In total, ~50 mg of cancer and paracancerous tissue
samples were weighed and RNAiso Plus (Takara Biotechnology Co.,
Ltd.) was added to extract total RNA from the tissue. The
concentration and purity of RNA were determined on a NanoDrop
ND-1000 Spectrophotometer (Thermo Fisher Scientific, Inc.). Then,
the PrimeScript™ RT kit (Takara Biotechnology Co., Ltd.) was used
to perform reverse transcription according to the manufacturer's
instructions on an ABI PCR instrument 2720 (Applied Biosystems;
Thermo Fisher Scientific, Inc.). A LightCycler® 480 II
fluorescence quantitative PCR instrument (Roche Diagnostics) and TB
Green® Premix Ex Taq™ II (Takara Biotechnology Co.,
Ltd.) were used for PCR according to the manufacturers' protocols.
The reaction system comprised a mixture of 2.66 µl cDNA, 0.6 µl
upstream primer, 0.67 µl downstream primer and 5 µl fluorescent
dye. The amplified genes for the PCR reaction and all required
primers synthesized by Sangon Biotech Co., Ltd. were shown in
Table I. The experiment was repeated
thrice and the 2−ΔΔCq method (17) was used to analyse the data.
 | Table I.Primer sequences for amplification of
IFIT3, PARP9, CYB5R1, PTPN1, SLC34A2, DPT, SLPI and GAPDH. |
Table I.
Primer sequences for amplification of
IFIT3, PARP9, CYB5R1, PTPN1, SLC34A2, DPT, SLPI and GAPDH.
| Genes | Forward primers,
5-3 | Reverse primers,
5-3 |
|---|
| IFIT3 |
GCTGCAAGCAGCCAAATGTT |
CTCTGGGACTGGAGCTGACT |
| PARP9 |
AGGGAAGAGTGAGCTGGGACAAG |
TCTGCCGTCTGCCATTCAATGTG |
| CYB5R1 |
TGGCTGTGGGCTCCTACTTGG |
GGGCAAAGCGGAACCTCTTGG |
| PTPN1 |
TCAAAGTCCGAGAGTCAGGGTCAC |
CATCAGCAAGAGGCAGGTATCAGC |
| SLC34A2 |
GATGCCGTCGTCTCCAAGTTCAC |
TCCTCCAAGTCCTCGCAGCAC |
| DPT |
TGGGGCCAGTATGGCGATTA |
CTGGTAGCTGAAGCCTTGCC |
| SLPI |
GCTGTGGAAGGCTCTGGAAAGTC |
CAGTCACTCTGGCACTCAGGTTTC |
| GAPDH |
CAGGAGGCATTGCTGATGAT |
GAAGGCTGGGGCTCATTT |
Western blotting
The main antibodies used included DPT (1:500; cat.
no. DF12196; Affinity Biosciences), GAPDH (1:500; cat. no. AP0063;
Bioworld Technology, Inc) and horseradish peroxidase-conjugated
secondary anti-rabbit IgG (1:1,000; cat. no. BS13278; Bioworld
Technology, Inc). Total cancer and paracancerous tissues were lysed
with RIPA buffer (Beyotime Institute of Biotechnology), and the
total proteins were extracted and determinated using a BCA protein
assay kit. Then, 10% SDS-PAGE was performed to separate proteins,
and each lane was loaded with an equal amount of protein (100 µg).
Subsequently, the protiens were transferred onto the nitrocellulose
membrane and blocked with albumin bovine V (Beijing Solarbio
Science & Technology Co., Ltd.) at room temperature for 1.5 h.
The strips were incubated with DPT and GAPDH antibodies at 4°C
overnight. On the second day, the strips were removed, washed
thrice with TBST (1ⅹ TBS and 0.1% Tween-20) and incubated with
horseradish peroxidase-conjugated secondary antibody at room
temperature for 1 h. After incubation, the strips were cleaned
thrice and tested and the experiment was repeated thrice for
quantitative analysis. The densities and intensities of the strips
were quantified using Odyssey® SA (LI-COR;
Biosciences).
Immunohistochemical staining
All EC and normal endometrial tissues were fixed in
10% neutral buffered formalin solution at room temperature for 24
h, then were routinely embedded in paraffin and cut into 4-µm thick
sections. The sections were quenched with 3% hydrogen peroxide at
room temperature for 10 min, then rinsed in phosphate-buffered
saline. Following antigen retrieval in 10 mM citrate buffer for
95°C 5 min, 40°C 15 min, the sections were incubated with primary
polyclonal antibody DPT (1:100; cat. no. DF12196; Affinity
Biosciences) at 4°C overnight. The negative control was treated
with PBS instead of primary antibody. Then, the secondary antibody,
goat anti-rabbit IgG (PV-9001; Beijing Zhongshan Jinqiao
Biotechnology Co., Ltd) was applied to the sections at 37°C for 20
min. At room temperature, the sections were dyed using
diaminobenzidine (DAB) for 60 sec, re-stained with haematoxylin for
20 sec, and sealed for observation. DPT-positive cells showed
yellow-brown granules in the cytoplasm. Images were captured using
an Olympus VS120® digital slice scanning microscope
(Olympus Corporation). Then, linear measurement data were acquired
using Image-Pro® Plus 6.0 (Media Cybernetics Inc.).
Statistical analysis
All data were presented as the mean ± the standard
error of the mean. The statistical analyses were performed using
SPSS 22.0 (IBM Corp.) and GraphPad Prism software version 5.0
(GraphPad Software, Inc.). A paired t-test was used for comparisons
between cancer tissues and paracancerous tissues from the same
patient, and an independent sample t-test was used for comparisons
between normal endometrial and EC cases. The associations between
the expression of DPT and clinicopathological features were
evaluated using χ2 and Fisher's exact tests. P<0.05
was considered to indicate a statistically significant
difference.
Results
Proteomic analysis of EC
After the cancer and paracancerous tissue samples
were hydrolysed and pre-treated, the total differential proteins
were determined by LC-MS/MS and analysed by bioinformatics and
statistical analysis. A majority of the identified peptides were
distributed in 7–25 amino acids, which was consistent with the
general laws of enzymatic hydrolysis and HCD fragmentation. Through
quantitative screening using MaxQuant a total of 3,401 proteome
samples were retrieved from the two groups, among which the
quantifiable number of proteins were 3,245 (Table SI). Student's t-tests were conducted
after the two groups were classified and screened. In total, 579
proteins were significantly up-regulated and 346 proteins were
significantly down-regulated (Tables
II and SII). Eventually, 925
differential proteins were screened as differentially expressed
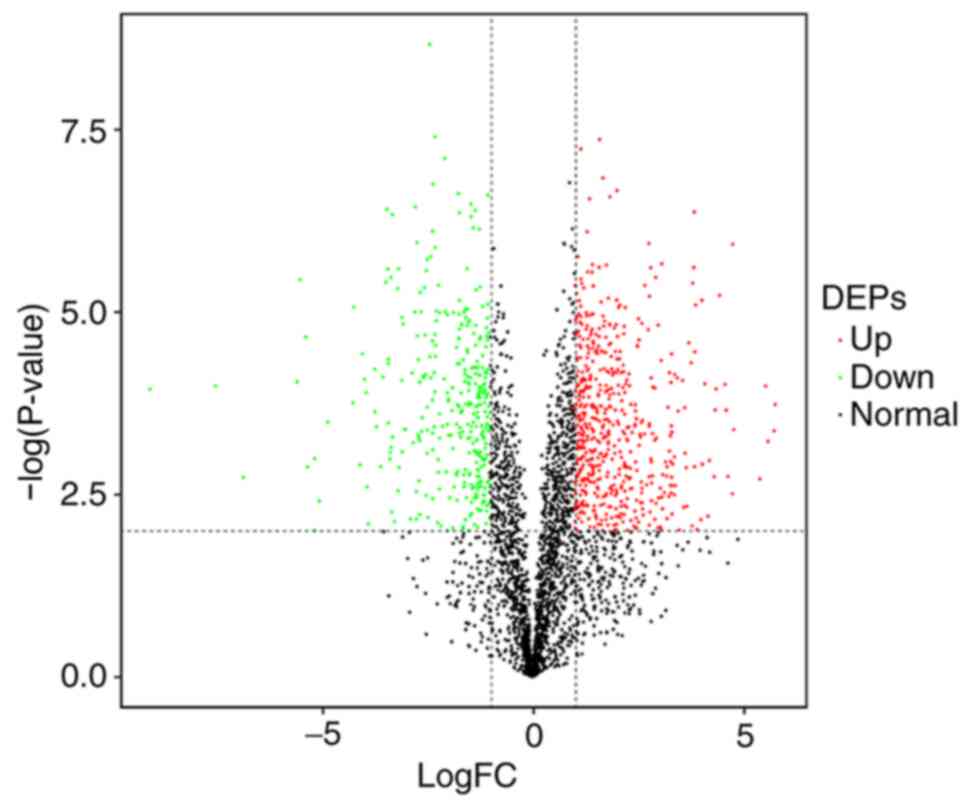
proteins (DEPs) as shown in a volcano map (Fig. 1). The red dots represent 579
significantly up-regulated proteins, whereas the green dots
represent 346 significantly down-regulated proteins.
 | Table II.Total up-regulated or down-regulated
proteins. |
Table II.
Total up-regulated or down-regulated
proteins.
| Compared groups | Regulation type | Fold-changes ≥2 and
P<0.01 |
|---|
| EC/AP | Up-regulated | 579 |
|
| Down-regulated | 346 |
To analyse the molecular functions of the identified
proteins, DEPs were clustered into GO categories, including
biological process, cellular components and molecular functions.
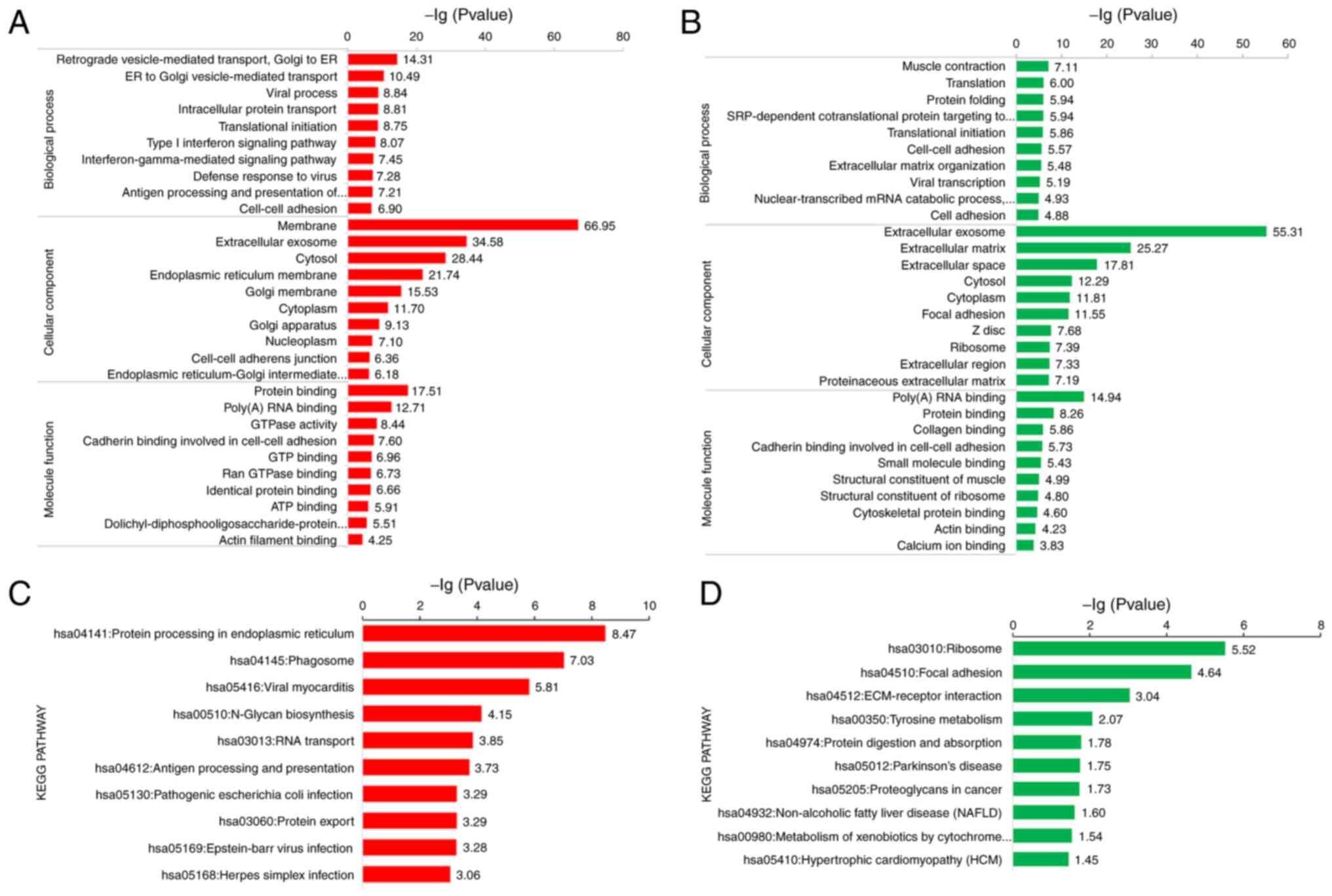
The up-regulated DEPs were primarily distributed in the ‘membrane‘
and ‘extracellular exosome’, while the down-regulated DEPs were
mainly located in ‘extracellular exosome’ and ‘extracellular
matrix’, as shown in Fig. 2A and B,
respectively. The functional enrichment of DEPs was also analysed
using the KEGG database. Up-regulated DEPs are mainly involved in
‘protein processing in the endoplasmic reticulum’ and ‘phagosome’,
while down-regulated DEPs played a crucial role in ‘ribosome’ and
‘focal adhesion’, as seen in Fig. 2C and
D, respectively. This finding is consistent with the data
described in the subsequent paragraphs. As aforementioned, DPT is
an extracellular matrix protein, which principally takes a
significant role in focal adhesion, and its down-regulation can
accelerate tumour invasion and progression (18).
Identification of diagnostic and
prognostic biomarkers
The seven potential predictors of EC were chosen
according to the difference ratio of proteins (>5; P<0.01)
protein function and literatures and classified into two groups:
Paracancerous (AP) or EC. The up-regulated proteins included
interferon-induced protein with tetratricopeptide repeats 3
(IFIT3), poly(ADP-ribose) polymerase family member 9 (PARP9),
solute carrier family 34 member 2 (SLC34A2), cytochrome b5
reductase 1 (CYB5R1) and protein tyrosine phosphatase non-receptor
type 1 (PTPN1), while down-regulated proteins contained DPT and
secretory leukocyte peptidase inhibitor (SLPI) (Table III). The RT-qPCR results of
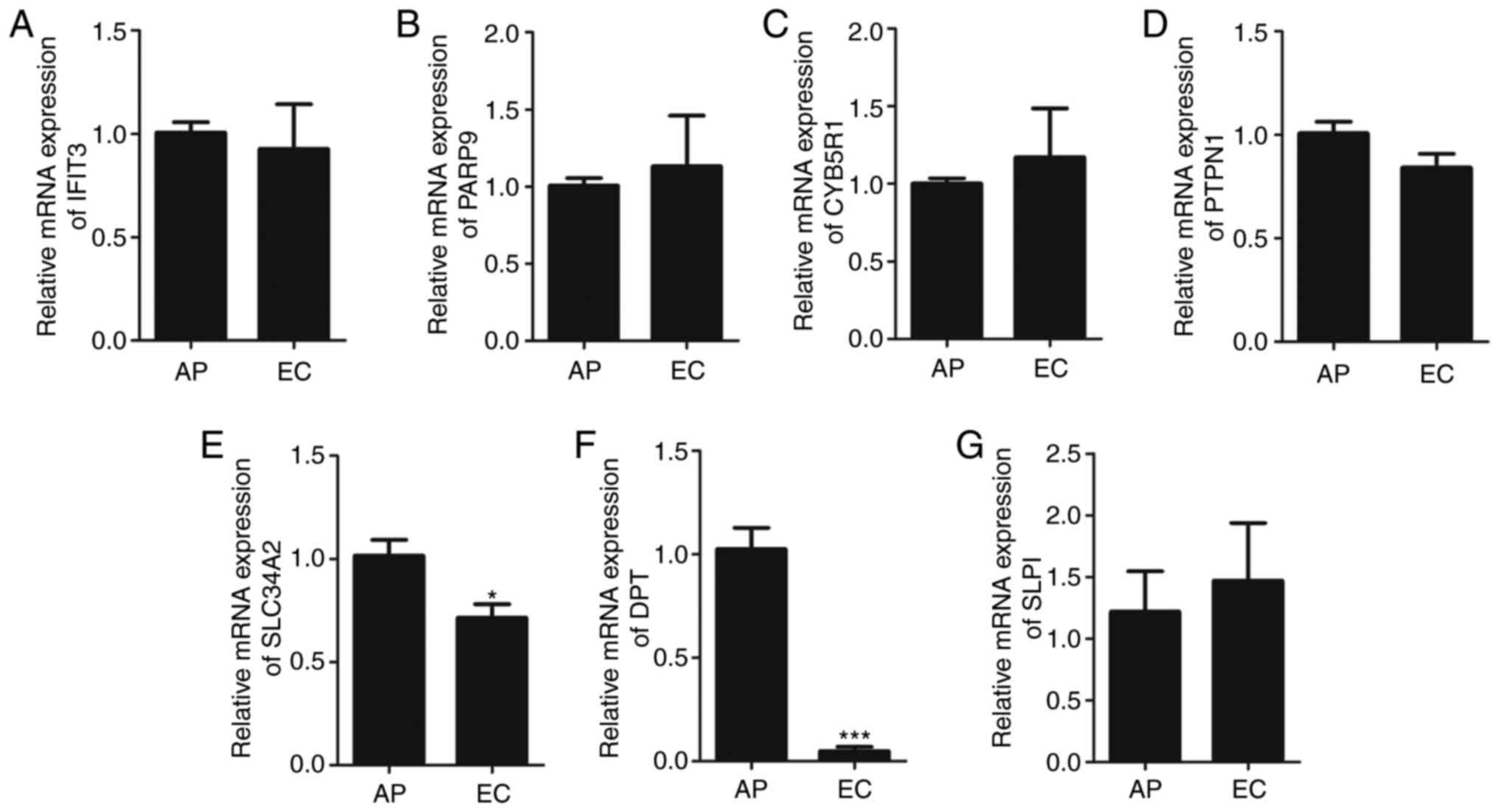
differential proteins are presented in Fig. 3. IFIT3, PARP9, CYB5R1, PTPN1 and SLPI
expressions were not conspicuously up-regulated or down-regulated,
whereas SLC34A2 and DPT expressions in EC were significantly
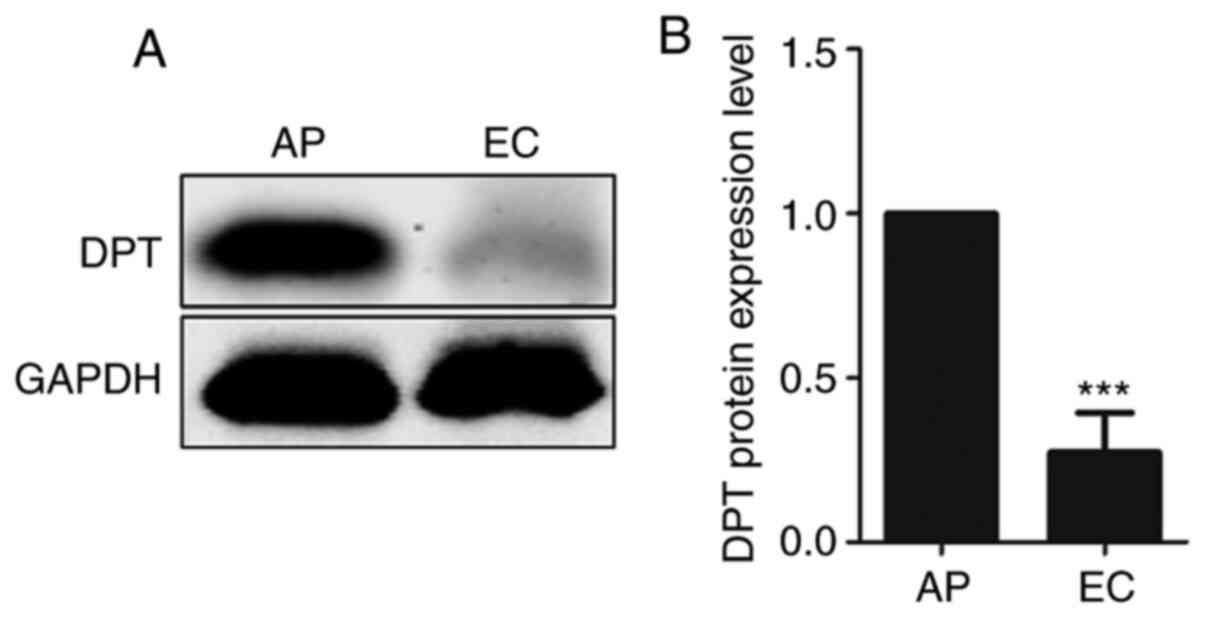
decreased (P<0.05 and P<0.001, respectively; Fig. 3E and F). Western blotting results
showed that the DPT in endometrial carcinoma was significantly
lower compared with that in paracancerous tissues (P<0.001;
Fig. 4). In summary, the DPT results
were consistent in LC-MS/MS, RT-qPCR and western blotting DPT was
shown to be significantly down-regulated in EC. These findings
indicated that the expression of DPT was markedly decreased in EC,
which might be closely associated with the pathogenesis of EC.
 | Table III.Seven differential proteins enriched
in bioinformatics analysis. |
Table III.
Seven differential proteins enriched
in bioinformatics analysis.
| Registration number
in UniProt database | Name of
differential proteins | Gene | Regulation | P-value |
|---|
| O14879 | Interferon-induced
protein with tetratricopeptide repeats 3 | IFIT3 | Up | 0.00474 |
| Q8IXQ6 | Poly(ADP-ribose)
polymerase family member 9 | PARP9 | Up | 0.00178 |
| Q9UHQ9 | Cytochrome b5
reductase 1 | CYB5R1 | Up | 0.00460 |
| P18031 | Protein tyrosine
phosphatase, nonreceptor type 1 | PTPN1 | Up | 0.00239 |
| O95436 | Solute carrier
family 34 member 2 | SLC34A2 | Up | 0.00630 |
| Q07507 | Dermatopontin | DPT | Down | 0.00001 |
| P03973 | Secretory leukocyte
peptidase inhibitor | SLPI | Down | 0.00390 |
Association between DPT and
clinicopathological characteristics
A total of 75 EC tissue wax blocks included 54
patients with stage I–II EC and 21 patients with stage III–IV.
There were 49 patients with high and medium differentiation and 26
patients with low differentiation. The aforementioned results
indicated that DPT might play a role in inhibiting progression in
EC and that its reduction promotes cancer migration and
progression. The immunohistochemical staining results of the wax
blocks of 30 normal endometrial (normal group) and 75 EC tissues
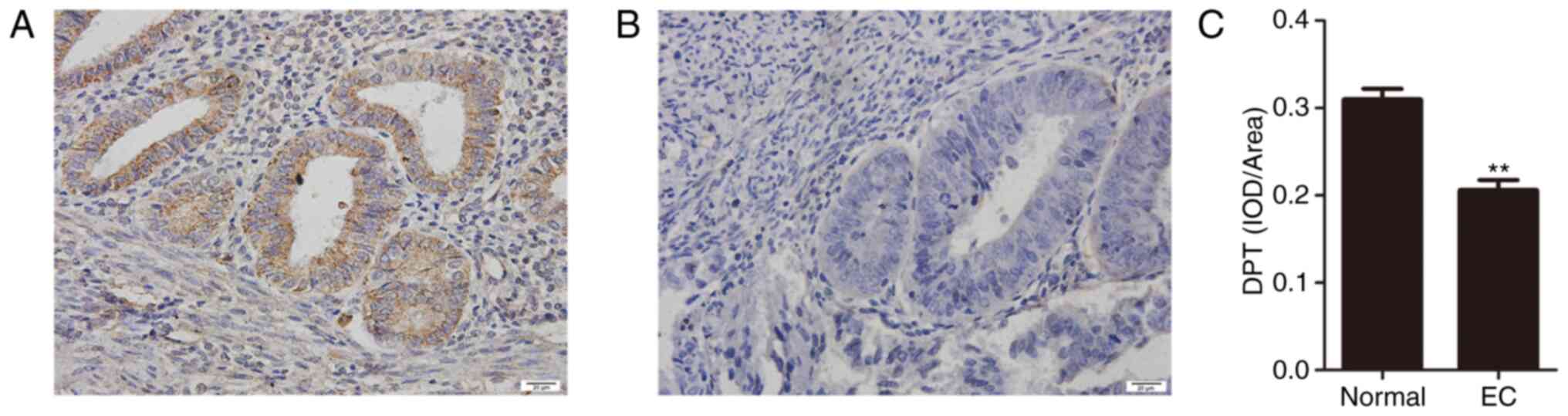
(EC group) are provided in Fig. 5.
In 30 cases of normal endometrial tissues, 25 cases were positive
and the rest were absent. Among the 75 EC tissues, 58 cases showed
low expression and only 17 cases showed high-expression. Compared
with normal endometrium, DPT was markedly down-regulated in EC
tissues. Afterwards, the χ2 and Fisher's exact tests
were applied to evaluate the association between DPT and
clinicopathological features (Table
IV). Staging, differentiation, depth of myometrial invasion and
lymph node metastasis were found to be associated with DPT
expression. As shown in Table IV,
the positive expression of DPT was markedly decreased in tissues
with late stage, poor differentiation, myometrial invasion depth
≥1/2 and lymph node metastasis (P<0.05). The above results
demonstrated that the positive expression of DPT decreased with
increasing tumour malignancy.
 | Table IV.Association between DPT expression
and clinicopathological features of patients with endometrial
cancer. |
Table IV.
Association between DPT expression
and clinicopathological features of patients with endometrial
cancer.
|
|
| DPT |
|
|
|---|
|
|
|
|
|
|
|---|
| Clinical
features | Cases, n | High | Low | χ2 | P-value |
|---|
| FIGO stage |
|
|
| – | 0.029 |
|
I–II | 54 | 16 | 38 |
|
|
|
III–IV | 21 | 1 | 20 |
|
|
|
Differentiation |
|
|
| 5.091 | 0.024 |
| High
and medium | 49 | 15 | 34 |
|
|
|
Poor | 26 | 2 | 24 |
|
|
| Depth of muscular
invasion |
|
|
| 6.799 | 0.009 |
|
<1/2 | 41 | 14 | 27 |
|
|
|
≥1/2 | 34 | 3 | 31 |
|
|
| Lymph node
metastasis |
|
|
| – | 0.031 |
| No | 55 | 16 | 39 |
|
|
|
Yes | 20 | 1 | 19 |
|
|
Discussion
The proteome of EC was detected based on LC-MS/MS
and DEPs were analysed using bioinformatics. In total, seven DEPs
were identified, including IFIT3, PARP9, SLC34A2, CYB5R1, PTPN1,
DPT and SLPI. RT-qPCR and western blotting showed that DPT
expression was significantly decreased in EC. DPT expression in 75
cases of endometrial carcinoma and 30 cases of normal non-cancerous
endometrium was detected using immunohistochemical staining, and
the association between the expression and clinicopathological
factors was analysed. Compared with normal endometrium, DPT
expression in cancer tissues was decreased, and DPT was
significantly associated with differentiation, FIGO stage, muscle
infiltration depth and lymph node metastasis.
The extracellular matrix is a macromolecular dynamic
reticular structure composed of collagen, proteoglycans and other
glycoproteins (19). The migration
of cancer cells depends on the biochemical characteristics of the
ECM, while integrin α3β1 can promote cancer cell migration and
invasion (20). After destroying the
basement membrane, the tumour cells move along the track of
collagen fibres, invade the vascular endothelial basement membrane
and finally reach distant organs through the circulation (21). Tumour cells can also migrate through
the network around the extracellular matrix. Even in primary
tumours, the extracellular matrix is constantly altered under the
influence of the tumour microenvironment, thus promoting tumour
migration and disease progression (21,22).
DPT is an extracellular matrix protein that plays an
important role in matrix remodelling and metastasis of cancer
tissues. DPT is expressed at a low level in oral squamous cell
carcinoma, hepatocellular carcinoma and thyroid papillary carcinoma
and can promote the proliferation of prostate cancer cells
(18,23). DPT not only increases the biological
activity of transforming growth factor-β but also combines with
integrin α3β1 to promote the germination of tumour
neovascularization and ducts by enhancing the movement ability of
tumour cells and inducing the formation of endothelial cell
adhesion (9,11). Fu et al (24) found that low DPT expression in
hepatocellular carcinoma is mainly mediated by DNA methylation and
also demonstrated that DPT enhances the stability of focal adhesion
through α3β1 integrin-Rho GTPase signalling, thus inhibiting the
metastasis of hepatocellular carcinoma. In thyroid carcinoma,
ectopic expression of DPT hinders thyroid cancer cell proliferation
(25). DPT also down-regulates the
expression of c-Myc and regulates the expression of cell
cycle-dependent kinases (CDK4 and CDK6) and cyclin-dependent kinase
inhibitors (P21) through the MEK-ERK-MYC signalling pathway
(25).
Despite a low expression pattern of DPT in numerous
types of cancer, no related studies have been reported in
endometrial carcinoma, to the best of our knowledge. In the present
study, DPT expression in endometrial carcinoma was studied and its
association with clinicopathological factors was analysed.
LC-MS/MS, RT-qPCR and western blotting results all confirmed a low
level of DPT expression in endometrial carcinoma. The expression of
DPT was decreased in poorly differentiated tumours, more advanced
clinicopathological stages, deeper myometrial invasion and in cases
of lymph node metastasis. Integrins are a type of cell adhesion
molecule that depend on Ca2+ or Mg2+ to
mediate recognition and adhesion between cells and the
extracellular matrix and DPT mediates cell adhesion by binding to
integrin α3β1, thus inhibiting the proliferation and migration of
tumour cells (24). Therefore, it
was hypothesised that DPT might be involved in the pathogenesis of
endometrial carcinoma and that its exact molecular mechanism needs
to be further studied.
However, the present study only contained the most
common pathological types of endometrial carcinoma and not rare
pathological types, such as serous carcinoma, mucinous carcinoma,
clear cell carcinoma and carcinosarcoma. Due to the limitation of
the sample size and single pathological type, further research
should be conducted to reveal more prevalent phenomena. Notably,
the current study confirmed for the first time that decreased DPT
levels were associated with EC, so identifying the expression of
DPT in EC might contribute to identifying novel biomarkers and
providing future prognostic guidance. In-depth exploration of DPT
may reveal the molecular mechanism of EC and provide new ideas for
targeted therapy to increase the success of treatment and survival
rate of patients with advanced EC.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
This work was supported by The Jiangsu Provincial
Commission of Health and Family Planning (grant no. H2017079) and
The Jiangsu Science and Technology Planning Project (grant no.
BE2019636).
Availability of data and materials
The datasets used and analysed during the current
study are available from the corresponding author on reasonable
request. The LC-MS/MS datasets generated during the current study
are available in the PRIDE repository (http://www.ebi.ac.uk/pride). The datasets generated
and/or analysed during the current study are available in the
UniProt knowledgebase (https://www.uniprot.org/proteomes/UP000005640).
Authors' contributions
XZ, HH and BZ participated in the conception and
design of this study. HH, ZH, LL and CW carried out the
experiments. HH and ZH collected the experimental data. HH, ZH and
ZY analysed and interpreted the data. HH, ZY, XZ and BZ took charge
of writing the article and revising it. HH, ZH and XZ confirm the
authenticity of all raw data. All the authors read and approved the
final manuscript.
Ethics approval and consent to
participate
This study was approved by The Ethics Committee of
Xuzhou Central Hospital. The patients included in the study
provided written consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Njoku K, Chiasserini D, Whetton AD and
Crosbie EJ: Proteomic biomarkers for the detection of endometrial
cancer. Cancers (Basel). 11:15722019. View Article : Google Scholar
|
|
2
|
Miller KD, Nogueira L, Mariotto AB,
Rowland JH, Yabroff KR, Alfano CM, Jemal A, Kramer JL and Siegel
RL: Cancer treatment and survivorship statistics, 2019. CA Cancer J
Clin. 69:363–385. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Martinez-Garcia E, Lopez-Gil C, Campoy I,
Vallve J, Coll E, Cabrera S, Ramon Y, Cajal S, Matias-Guiu X, Van
Oostrum J, Reventos J, et al: Advances in endometrial cancer
protein biomarkers for use in the clinic. Expert Rev Proteomics.
15:81–99. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Maxwell GL, Hood BL, Day R, Chandran U,
Kirchner D, Kolli VS, Bateman NW, Allard J, Miller C, Sun M, et al:
Proteomic analysis of stage I endometrial cancer tissue:
Identification of proteins associated with oxidative processes and
inflammation. Gynecol Oncol. 121:586–594. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ueda SM, Kapp DS, Cheung MK, Shin JY,
Osann K, Husain A, Teng NN, Berek JS and Chan JK: Trends in
demographic and clinical characteristics in women diagnosed with
corpus cancer and their potential impact on the increasing number
of deaths. Am J Obstet Gynecol. 198:218.e1–218.e6. 2008. View Article : Google Scholar
|
|
6
|
Giorgianni F, Koirala D and
Beranova-Giorgianni S: Proteomics of the human pituitary tissue:
Bioanalytical methods and applications. Bioanalysis. 6:1989–2003.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Peng L, Cantor DI, Huang C, Wang K, Baker
MS and Nice EC: Tissue and plasma proteomics for early stage cancer
detection. Mol Omics. 14:405–423. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Neame PJ, Choi HU and Rosenberg LC: The
isolation and primary structure of a 22-kDa extracellular matrix
protein from bovine skin. J Biol Chem. 264:5474–5479. 1989.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Okamoto O, Fujiwara S, Abe M and Sato Y:
Dermatopontin interacts with transforming growth factor beta and
enhances its biological activity. Biochem J. 337:537–541. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Okamoto O, Hozumi K, Katagiri F, Takahashi
N, Sumiyoshi H, Matsuo N, Yoshioka H, Nomizu M and Fujiwara S:
Dermatopontin promotes epidermal keratinocyte adhesion via α3β1
integrin and a proteoglycan receptor. Biochemistry. 49:147–155.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Krishnaswamy VR, Balaguru UM, Chatterjee S
and Korrapati PS: Dermatopontin augments angiogenesis and modulates
the expression of transforming growth factor beta 1 and integrin
alpha 3 beta 1 in endothelial cells. Eur J Cell Biol. 96:266–275.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mutch D: I231 FIGO Staging of Endometrial
Cancer 2009. Int J Gynaecol Obstet. 107:S58. 2009. View Article : Google Scholar
|
|
13
|
Perez-Riverol Y, Csordas A, Bai J,
Bernal-Llinares M, Hewapathirana S, Kundu DJ, Inuganti A, Griss J,
Mayer G, Eisenacher M, et al: The PRIDE database and related tools
and resources in 2019: Improving support for quantification data.
Nucleic Acids Res. 47:D442–D450. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cox J, Neuhauser N, Michalski A, Scheltema
RA, Olsen JV and Mann M: Andromeda: A peptide search engine
integrated into the MaxQuant environment. J Proteome Res.
10:1794–1805. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Alexa A, Rahnenführer J and Lengauer T:
Improved scoring of functional groups from gene expression data by
decorrelating GO graph structure. Bioinformatics. 22:1600–1607.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xie C, Mao X, Huang J, Ding Y, Wu J, Dong
S, Kong L, Gao G, Li CY and Wei L: KOBAS 2.0: A web server for
annotation and identification of enriched pathways and diseases.
Nucleic Acids Res. 39 (Suppl 2):W316–W322. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−ΔΔC(T)) method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Takeuchi T, Suzuki M, Kumagai J, Kamijo T,
Sakai M and Kitamura T: Extracellular matrix dermatopontin
modulates prostate cell growth in vivo. J Endocrinol. 190:351–361.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Theocharis AD, Skandalis SS, Gialeli C and
Karamanos NK: Extracellular matrix structure. Adv Drug Deliv Rev.
97:4–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cavaco ACM, Rezaei M, Caliandro MF, Lima
AM, Stehling M, Dhayat SA, Haier J, Brakebusch C and Eble JA: The
interaction between laminin-332 and α3β1 integrin determines
differentiation and maintenance of CAFs, and supports invasion of
pancreatic duct adenocarcinoma cells. Cancers (Basel). 11:142018.
View Article : Google Scholar
|
|
21
|
Eble JA and Niland S: The extracellular
matrix in tumor progression and metastasis. Clin Exp Metastasis.
36:171–198. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kalluri R: The biology and function of
fibroblasts in cancer. Nat Rev Cancer. 16:582–598. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yamatoji M, Kasamatsu A, Kouzu Y, Koike H,
Sakamoto Y, Ogawara K, Shiiba M, Tanzawa H and Uzawa K:
Dermatopontin: A potential predictor for metastasis of human oral
cancer. Int J Cancer. 130:2903–2911. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Fu Y, Feng M-X, Yu J, Ma MZ, Liu XJ, Li J,
Yang XM, Wang YH, Zhang YL, Ao JP, et al: DNA methylation-mediated
silencing of matricellular protein dermatopontin promotes
hepatocellular carcinoma metastasis by α3β1 integrin-Rho GTPase
signaling. Oncotarget. 5:6701–6715. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Guo Y, Li H, Guan H, Ke W, Liang W, Xiao H
and Li Y: Dermatopontin inhibits papillary thyroid cancer cell
proliferation through MYC repression. Mol Cell Endocrinol.
480:122–132. 2019. View Article : Google Scholar : PubMed/NCBI
|