Introduction
The dried fruits of S. chinensis Baill, a
member of the Magnoliaceae family, are used extensively in the
traditional medicinal systems of Korea, China and Japan (1). In particular, these fruits have been
employed in the treatment and prevention of some chronic diseases,
including hepatitis and cancer (2,3), which
may result from damage to biomolecules by free radicals and
reactive oxygen species. Additionally, the seeds and fruits of
S. chinensis are enriched in lignans, and >40 lignans
have been isolated from this plant (4).
αIC and α-iso-cubebene, which have very similar
structures, were first purified from the dried fruits of S.
chinensis using n-hexane (5). In an early study, nuclear magnetic
resonance (NMR), mass spectrometry and circular dichroism results
showed that the molecular formula of α-iso-cubebene was
C15H24NH4, and harbors a cubebene
sesquiterpene skeleton (5).
α-iso-cubebene strongly stimulates intracellular calcium signaling
and CXCL8 production in human neutrophils (5). This stimulation increases in a
concentration-dependent manner, with maximal activity being evident
at ~200 μg/ml. α-iso-cubebene also attenuates the activities of
adhesion molecules, including vascular cell adhesion molecule-1,
E-selectin, and intracellular adhesion molecule-1 in tumor necrosis
factor-α-stimulated human vein endothelial cells (6). These features have revealed the
potential of α-iso-cubebene as an effective novel anti-inflammatory
agent for the prevention and treatment of vascular diseases.
However, the principal biological function of αIC remains unclear,
with the exception of the anti-inflammatory effect against
bacterial infection or endotoxin recently reported by our group
(7). The present study sought to
clarify the function of αIC in the context of therapy for
hepatocellular carcinoma.
Hepatocellular carcinoma is a primary malignancy of
hepatocytes, and generally leads to death within 6–20 months. This
disease is the fifth most common cancer in men and the eighth most
common cancer in women worldwide (8). Cirrhosis of any etiology is a major
risk factor for hepatocellular carcinoma (9). Approximately 80% of patients with
newly diagnosed hepatocellular carcinomas have pre-existing
cirrhosis in the liver, caused primarily by excessive alcohol use,
as well as hepatitis B or C infection (10). The variety of therapeutic strategies
for hepatocellular carcinoma include surgical resection and liver
transplantation, although the treatment options available depend on
the characteristics of the tumor (11,12).
However, in many cases the drugs available for hepatocellular
carcinoma therapy carry some disadvantages, such as toxicity, that
hinder their clinical application.
Presently, in an effort to identify more efficacious
therapeutic compounds, we investigated the anti-tumor functions of
αIC newly isolated from S. chinensis using HepG2
hepatocellular carcinoma cells. Apoptosis in the αIC-treated cells
was assessed using an established
3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT;
Sigma-Aldrich)-based assay and western blotting. The present
results indicate that high concentrations of αIC can induce
significant cell death of hepatocellular carcinoma cells. Apoptosis
induced by αIC is not correlated with Bax and p53 protein.
Materials and methods
Preparation of αIC compound
The αIC compound used in this study was prepared via
the methods established in our laboratory (5). Lyophilized powders of αIC were
dissolved in dimethylsulfoxide (DMSO, Sigma Aldrich, St. Louis, MO,
USA) to a final concentration of 4 mM.
Cell culture and treatment
HepG2 human hepatoblastoma cells were purchased from
the Korean Cell Line Bank (Seoul, Korea). Cells were grown in
monolayers in minimum essential medium (MEM; Gibco, Grand Island,
NY, USA) supplemented with 10% fetal bovine serum (FBS; Invitrogen,
Carlsbad, CA, USA) and antibiotics (100 U/ml penicillin and 100
μg/ml streptomycin; Gibco) during incubation at 37°C in a
humidified incubator containing 5% CO2 in air. All other
chemicals were purchased from Sigma-Aldrich.
In order to evaluate the dose-dependent effects, a
stock solution (4 mM) of αIC was applied to the HepG2 cell line
with different final concentrations (40, 80, 160 and 320 μM) of αIC
for another 24 h. Additionally, the time-dependent effects were
analyzed in the HepG2 cells treated for different times (3, 6, 12
and 24 h) with 320 μM of αIC.
Assay of cell proliferation
HepG2 cells were seeded at a density of
4×104 cells/200 μl in the wells of 96-well plates and
were then grown for 24 h in a 37°C incubator. When the cells
attained 70–80% confluence, they remained untreated (vehicle) or
were exposed to 40, 80, 160 or 320 μM αIC dissolved in DMSO for
another 24 h. Cell proliferation was determined using the
tetrazolium compound MTT. After the supernatants in the αIC- or
vehicle-treated wells were discarded, 200 μl of fresh MEM and 50 μl
of MTT solution (2 mg/ml in phosphate buffered saline; PBS) were
added to each well. The cells were then incubated in a 37°C
incubator. The reduction of MTT to insoluble purple formazan dye
crystals by viable cells was assessed in 220 μl recovered after 4
h. The formazan precipitate was dissolved in DMSO and the
absorbance was directly read at 570 nm in the well using a SoftMax
Pro5 spectrophotometer (Molecular Devices, Sunnyvale, CA, USA).
Additionally, the data were analyzed in terms of cell number versus
absorbance, allowing for the quantification of changes in cell
proliferation.
Flow cytometry analysis
The percentage of cells undergoing apoptosis and
dead cells were detected via staining with fluorescein
isothiocyanate (FITC) Annexin V (BD Bioscience, Franklin Lakes, NJ,
USA). HepG2 cells were seeded at a density of 2×106
cells in 100 mm-diameter dishes and grown for 24–48 h in an
incubator at 37°C. When the cells attained 70–80% confluence,
vehicle, 40, 80, 160 and 320 μM of αIC were added to each culture
dish and the cells were incubated for an additional 24 h. The cells
were harvested, washed twice in ice-cold PBS, and resuspended in 1×
binding buffer at a concentration of 1×106 cells/ml.
Cells (~1×105) in 100 μl of solution were transferred to
round-bottomed culture tubes. FITC Annexin V (5 μl) was added to
stain the cells. After 15 min of incubation at room temperature,
400 μl of 1× binding buffer was added to each tube and each sample
was analyzed with a FACS Calibur apparatus (BD Biosciences) within
1 h.
Western blot analyses
HepG2 cells harvested from 100-mm diameter culture
dishes were solubilized with 1% Nonidet P-40 in 150 mM NaCl, 10 mM
Tris HCl (pH 7.5), and 1 mM EDTA, and supplemented with a protein
inhibitor mixture (Roche, Basel, Switzerland). They were then
centrifuged for 10 min at 10,000 × g at 4°C. The homogenized
proteins were separated by 10% sodium dodecyl
sulfate-polyacrylamide gel electrophoresis for 3 h and transferred
to nitrocellulose membranes over 2 h at 40 V. The membranes were
then incubated with primary antibodies [anti-Bcl-2 (SC-7382),
anti-Bax (SC-493), anti-p53 (SC-6243) and anti-α-tubulin
(Sigma-Aldrich), anti-caspase-3 (Cell Signaling Technology Inc.)]
were used to detect Bcl-2, Bax, p53 and anti-α-tubulin. Each
antigen-antibody complex was visualized with a biotinylated
secondary antibody (goat anti-rabbit)-conjugated horseradish
peroxidase streptavidin Histostain-Plus Kit (Zymed, South San
Francisco, CA, USA) diluted to 1:1,500 in PBS.
Statistical analysis
Tests for significance between the vehicle- and
αIC-treated groups were performed via one-way ANOVA tests of
variance (SPSS version 10.10, SPSS Inc., Chicago, IL, USA). If a
statistically significant difference was observed, the Student
Newman-Keuls post hoc test was used. All the values are
expressed as the means ± standard deviation (SD). A p<0.05 was
considered significant.
Results
Effects of αIC on cell proliferation
Several lignans isolated from S. chinensis
induce anti-proliferative and anti-apoptotic effects in hepatic
carcinoma cells (13). Thus, we
initially attempted to determine whether αIC exhibited an
anti-proliferative effect in HepG2 hepatocellular carcinoma cells
by determining the cell viability using an established MTT-based
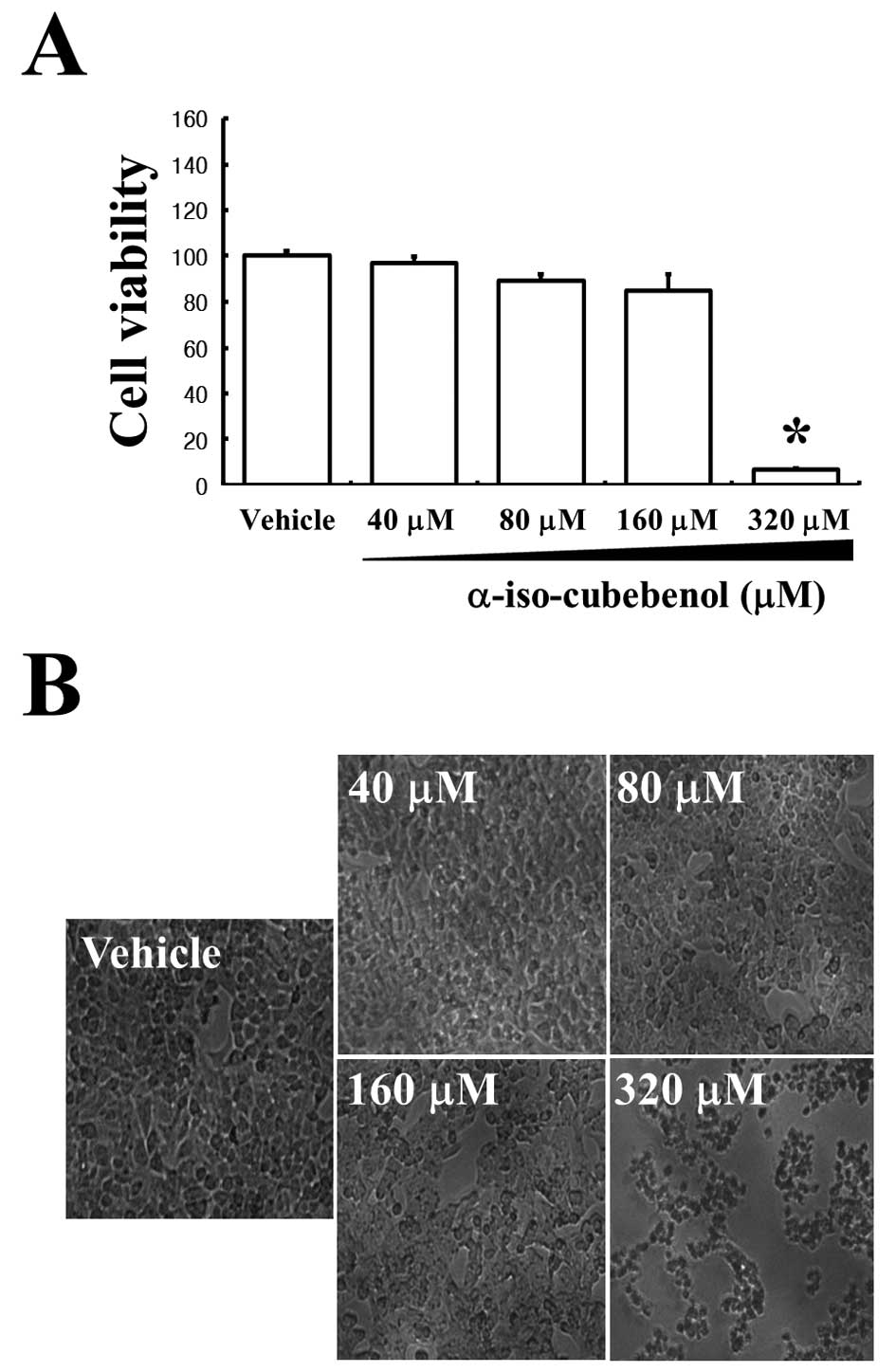
assay. The assay results confirmed that αIC significantly induced
cell death at 320 μM (Fig. 1A).
However, 40–160 μM αIC did not appreciably alter cell death.
Additionally, HepG2 cells were observed via phase-contrast
microscopy after 24 h of treatment with the same concentrations of
αIC to determine whether cell death was concurrent with cell
morphological changes. αIC concentrations of 40–160 μM did not
affect HepG2 cell morphology, and these results were similar to
those obtained with vehicle-treated cells. However,
morphologically-altered cells in populations treated with 320 μM
αIC were markedly increased relative to other groups (Fig. 1B). Additionally, to investigate the
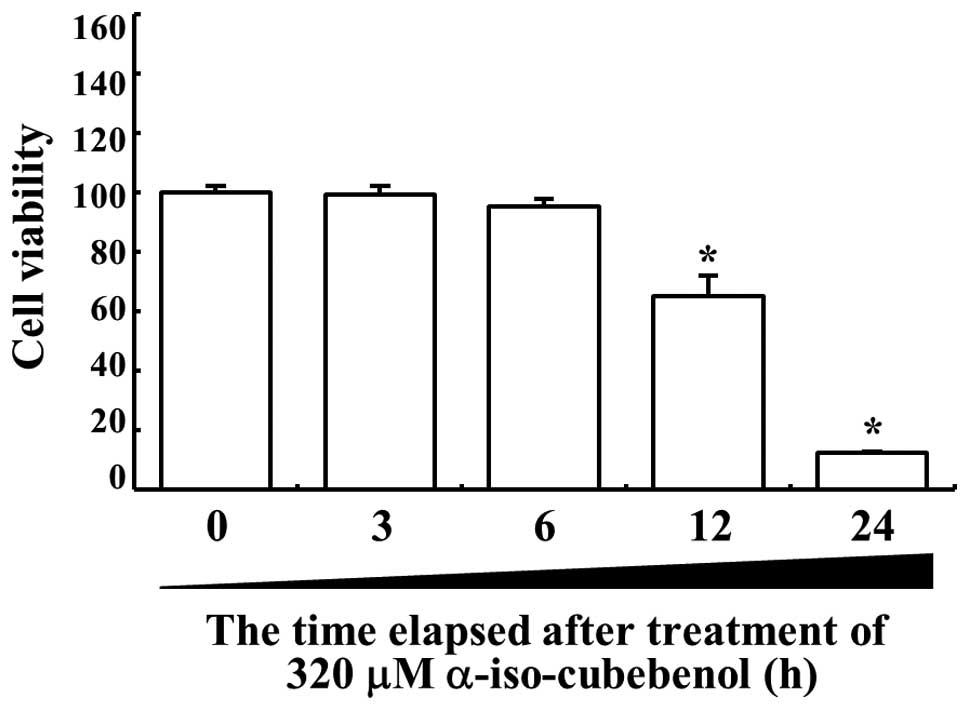
anti-proliferative effects depending on the time, HepG2 viability
was detected at various times after treatment with 320 μM αIC.
Significant changes in the death of HepG2 cells were initially
detected 12 h after αIC treatment. At 24 h after αIC treatment,
more than 80% of HepG2 cells were detected as dead cells (Fig 2). Therefore, these results showed
that high concentrations of αIC and incubation for 24 h could
effectively induce the death of hepatic carcinoma cells.
Effects of αIC on apoptosis
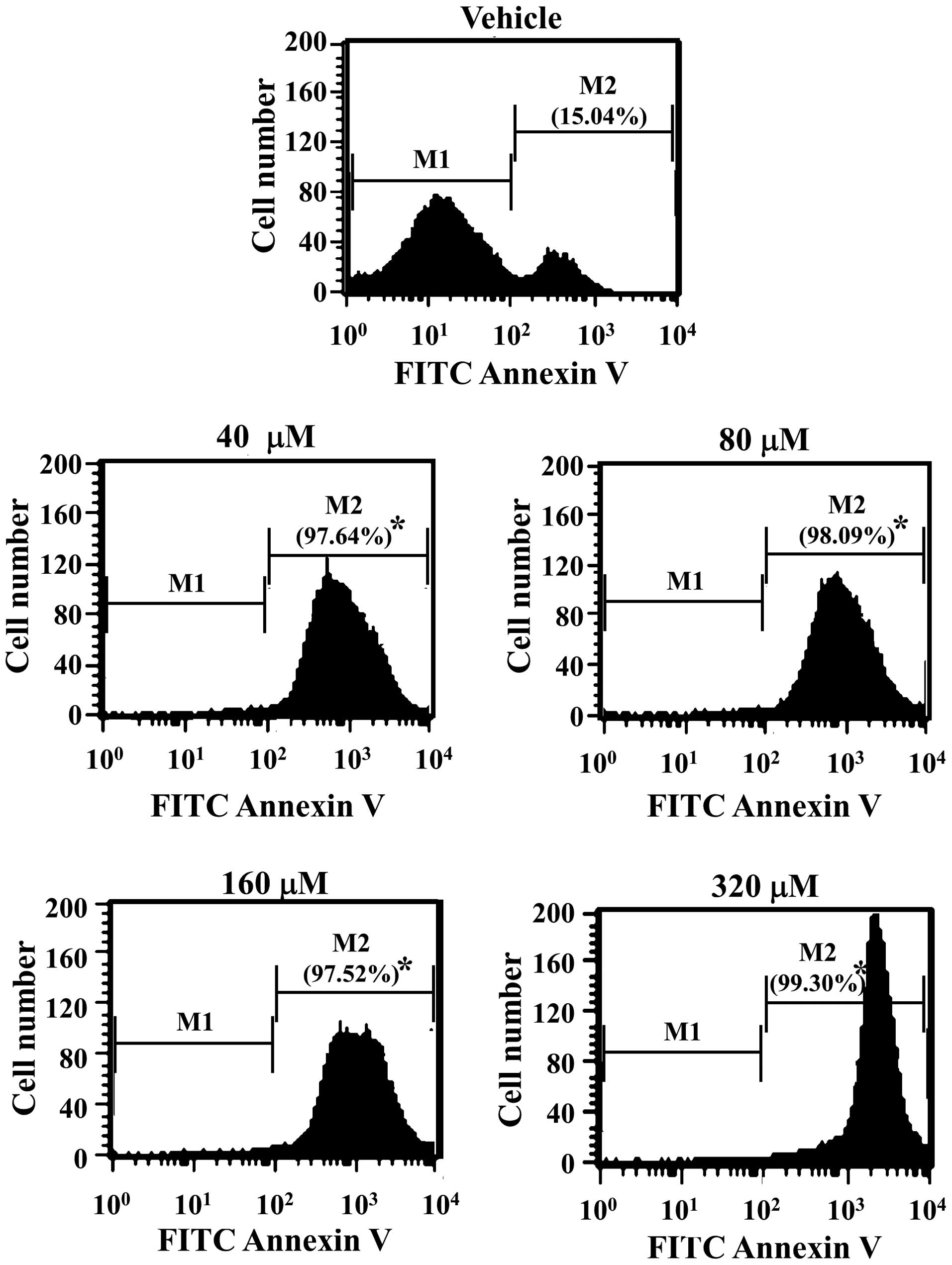
FITC Annexin V is used to quantitatively determine
the percentage of cells within a population that are actively
undergoing apoptosis (14,15). In order to evaluate the effects of
αIC on apoptosis, HepG2 cells treated with various concentrations
of αIC were stained with FITC Annexin V, and fluorescence was
detected by flow cytometry. As shown in Fig. 3, αIC induced a significant increase
in the number of cells undergoing apoptosis from 15 to 98–99% in 24
h in all treated groups. In particular, cells treated with 320 μM
αIC evidenced the highest numbers of apoptotic cells. Therefore,
these results indicated that early events such as the loss of
plasma membrane asymmetry in the apoptotic process may be induced
by αIC in the majority of treated cells, at all concentrations.
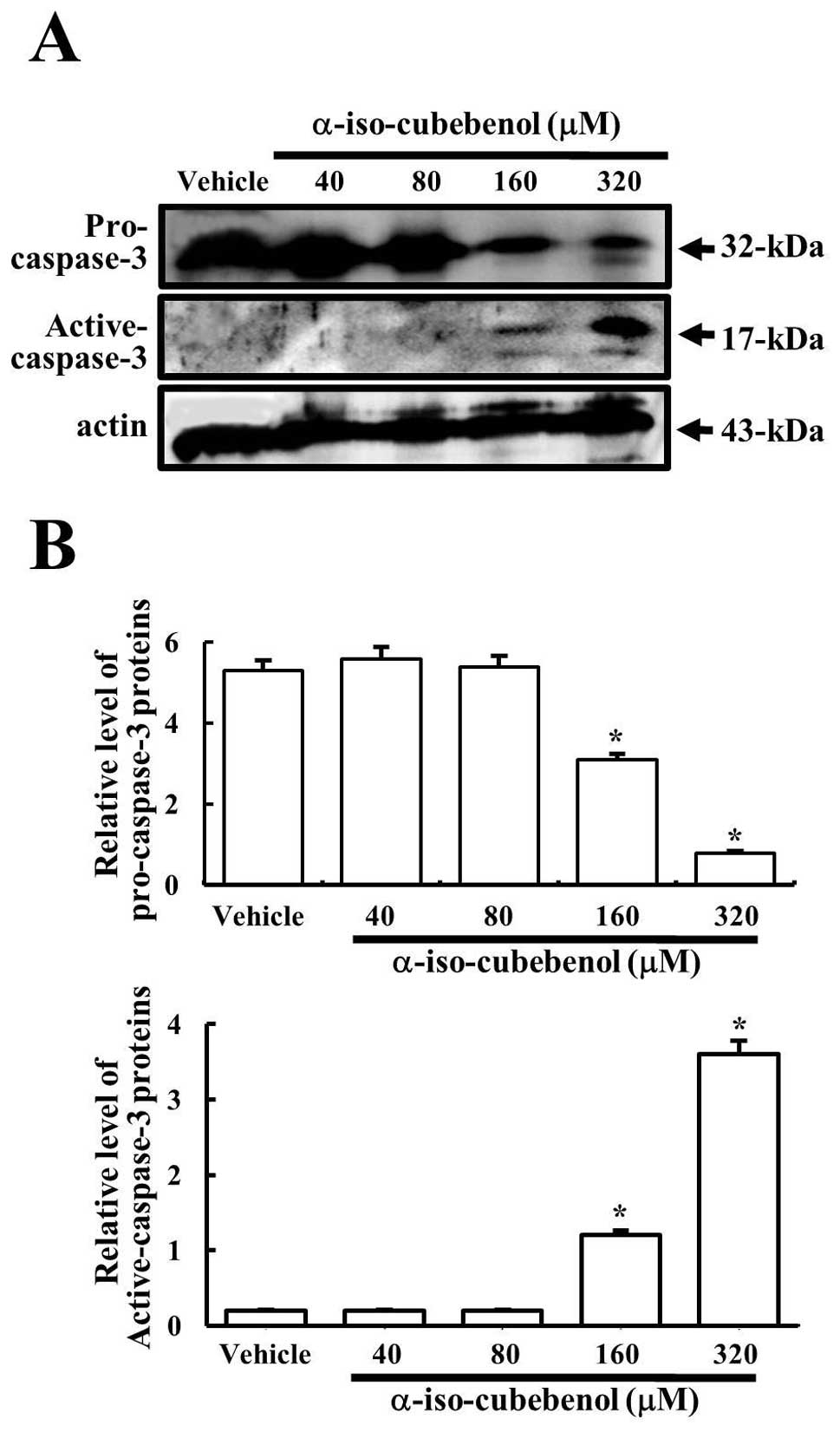
Furthermore, in order to demonstrate the activation
of the apoptosis pathway, the cleavage of caspase-3 was detected in
HepG2 cells treated with various concentrations of αIC.
Pro-caspase-3 levels were reduced significantly from the 160
μM-treated group to the 320 μM-treated group, whereas they were
maintained in both the 40- and 80 μM-treated groups. In particular,
the reduction of pro-caspase-3 levels induced increases in the
levels of active caspase-3, because the apoptosis signal induced
the cleavage of pro-caspase-3 (32 kDa) into two small fragments (12
and 17 kDa) (16). The highest
levels of active caspase-3 were detected in the 320 μM-treated
group (Fig. 4). Therefore, these
results also confirmed that αIC can activate caspase-3, a key
regulator of the apoptosis process, at high concentration.
Effects of αIC on the apoptotic
pathway
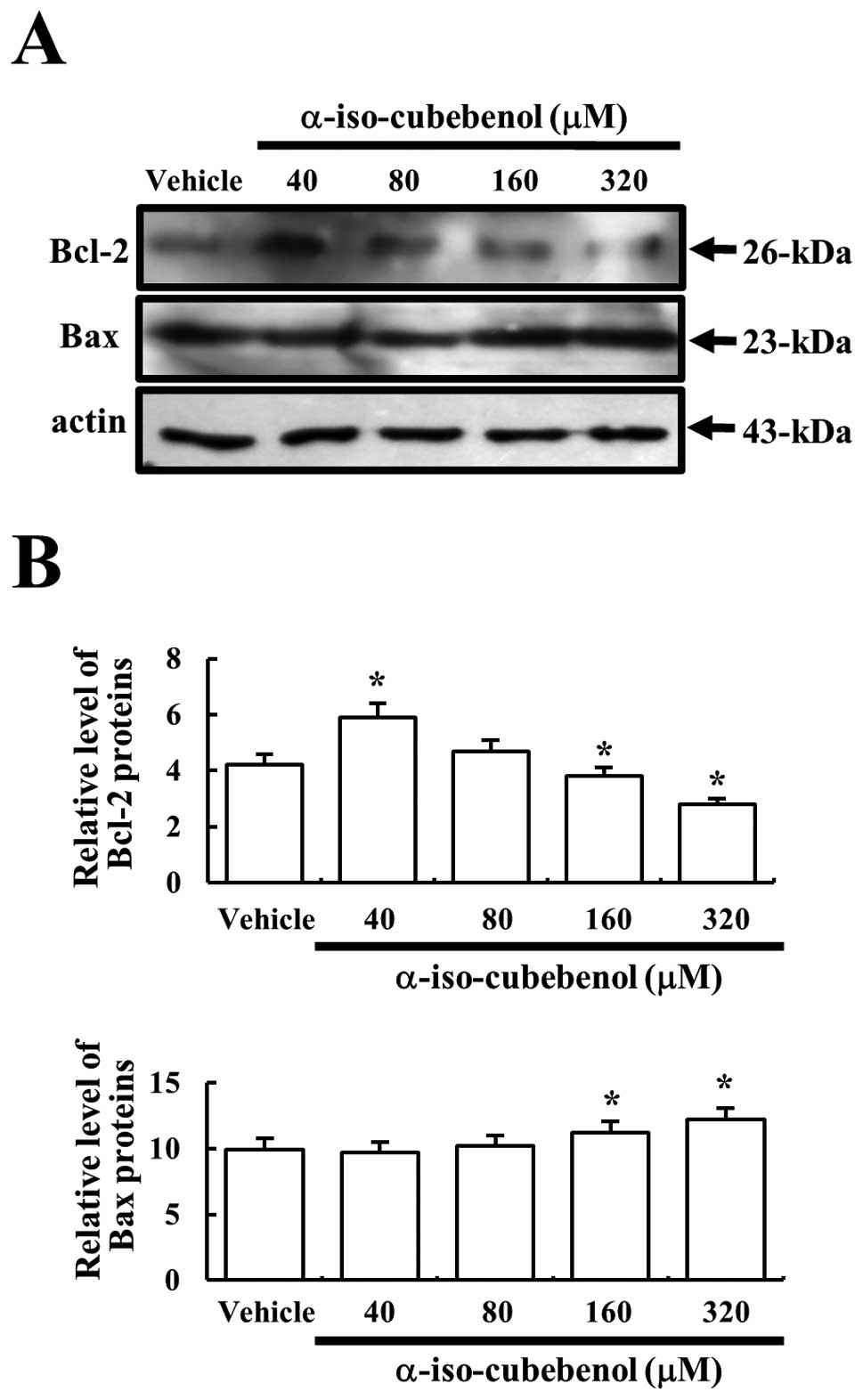
Bcl-2 belongs to the family of proteins that
includes both pro- and anti-apoptotic members. Among these members,
Bcl-2 proteins stimulate the anti-apoptotic process and the Bax
protein significantly inhibits the anti-apoptotic actions of the
Bcl-2 protein (17,18). In order to assess the effects of αIC
treatment on proteins associated with the apoptosis signaling
pathway, the expression levels of Bcl-2 and Bax proteins were
determined in the vehicle and αIC-treated groups using western blot
analysis. The expression level of Bcl-2 protein was markedly
increased only in the 40-μM αIC-treated group as compared to the
vehicle-treated group, and the protein level gradually decreased in
a dose-dependent pattern. Cells treated with 320 μM αIC exhibited
the lowest level of Bcl-2 expression. Bax protein expression was
increased slightly in a dose-dependent pattern. The highest level
of this protein was observed in cells treated with 320 μM αIC,
although their levels were quite similar to those of the
vehicle-treated group (Fig. 5).
These results indicated that αIC could simultaneously induce the
reduction of the protein levels associated with anti-apoptotic and
the increase in protein levels associated with the pro-apoptotic
process at high concentrations.
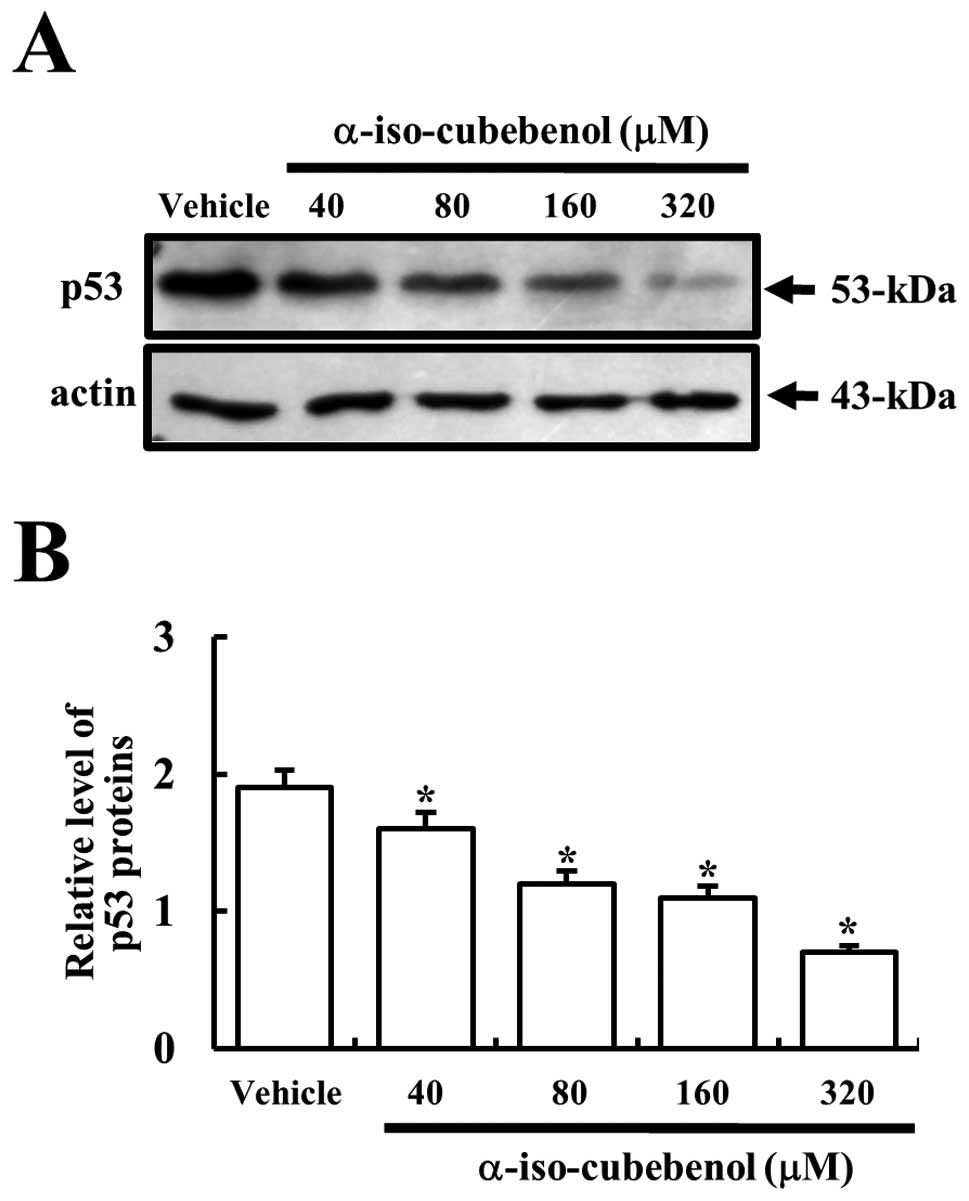
Effects of αIC on the expression of p53
tumor suppressor protein
The p53 protein is considered an important regulator
in the mechanism of apoptosis (19). In particular, the upregulation of
Bax is stimulated by activated p53 (20). Therefore, in order to determine
whether the p53 tumor suppressor protein would be affected by αIC
in HepG2 cells, the expression levels of p53 protein were assessed
in the vehicle- and αIC-treated groups. The highest levels of p53
expression were detected in vehicle-treated cells. After αIC
treatment, the levels of p53 expression decreased gradually in a
dose-dependent pattern. In particular, 320 μM αIC-treated HepG2
cells evidenced the lowest levels of p53 expression (Fig. 6). These results indicated that αIC
could induce a reduction in p53 associated with tumor suppression.
Furthermore, the expression of p53 did not contribute to the
upregulation of Bax protein in HepG2 cells.
Discussion
S. chinensis has a well-recognized history in
traditional Chinese medicine. Also referred to as ‘Chinese
Magnolia’, the plant is widely distributed throughout the Eastern
regions of Siberia, China, Japan, and Korea (3). Over the past 20 years, many active
lignans including Gomisin A, B, C, D, E, F, G, K3, N and J,
Schisandrol B, Schisandrin, and Schisandrin C were isolated from
this plant (3,21). The ligands have four thrapeutic
properties: adaptogenic action, hepatic protection, hepatic
stabilization and hepatic regeneration. Adaptogenic action involves
specific action mechanisms such as energizing, nervous system
stimulation, oxygenation, immune-modulation, anti-oxidation and
skin protection (3,22–25).
Additionally, these lignans reduce hyperlipemia and significantly
inhibit the development of arteriosclerosis (26). With regard to hepatic protection,
the lignans are correlated tightly with the increase in the hepatic
levels of ascorbic acid, as well as the inhibition of NADPH
oxidization and lipid peroxidation, which occur as the consequence
of the induction effect of the hepatic cytochrome P-450 enzymatic
system of the microsome. These lignans also protect against the
cholestasis induced by toxic substances (27–30).
The aforementioned ligands contribute to the hepatic regeneration
process via the induction of mRNA transcription for hepatocyte
growth factor, as well as the stimulation of hepatocyte
proliferation and increased blood flow to the liver. Additionally,
in an effort to improve regenerative capacity via reduced glutation
and the synthesis of hepatic glucogen, the addition of these
lignans induces increases in the levels of mitochondrial hepatic
glutation and mitochondrial reductase glutation activity (31,32).
However, relatively few functional studies have been conducted to
investigate in detail whether the novel lignans isolated from S.
chinensis influence cell death of the hepatocellular carcinoma
cells in vitro. In this study, we investigated the novel
function of αIC on the apoptosis of hepatocellular carcinoma
cells.
Lignans are the most common constituents of S.
chinensis. The major lignans in European seeds are
deoxyschisandrin (0.07–1.09%), gomisin N (0.24–1.49%), schisandrin
(0.75–1.86%), wuweizisu C (0.01–0.34%) and gomisin A (0.13–0.90%)
(33). Furthermore, several C18
dibenzocyclooctadiene lignans isolated from S. chinensis
evidence a variety of therapeutic activities involving
anti-tumorigenesis (34,35), anti-hepatocarcinogenesis (36,37),
anti-hepatotoxic (38), anti-human
immunodeficiency virus (39),
anti-oxidant (2,40), anti-inflammatory (35) and vascular relaxation (41) properties. Our laboratory previously
evaluated the anti-cancer ability of a hexane extract of S.
chinensis fruit indigenous to Korea, Japan and China (2). As part of an effort to identify new
compounds, we recently purified two novel compounds-αIC and
α-iso-cubebene-which have been employed in a variety of human
disease therapies (5,6). These compounds have profound potential
for development as novel drugs in the treatment of infection and
inflammation. However, only a relatively minimal body of literature
regarding the pharmacological mechanisms of action of αIC
exists.
Apoptosis (programmed cell death) is characterized
by certain morphological features involving the loss of plasma
membrane asymmetry and attachment, the fragmentation and
condensation of the nucleus, and the formation of cytoplasmic
vesicles (42,43). The loss of plasma membrane asymmetry
is one of the earliest detectable features, and can be
distinguished by Annexin V conjugated FITC (44,45).
We stained HepG2 cells with Annexin V to evaluate the novel
functions of αIC on hepatocellular carcinoma. Although significant
cell death was detected only at high concentrations using an MTT
assay and morphological analysis, the results of flow cytometry
suggested that the majority of the living cells were undergoing
apoptosis. Therefore, our results indicate that αIC can markedly
induce the apoptosis of HepG2 cells even after treatment at low
concentrations.
Apoptosis plays a critical role in a variety of
physiological processes during fetal development and in adult life.
Defects in the apoptosis process can lead to the progression of
many diseases involving progressive cell accumulation, as well as
cancer (46). The apoptotic process
involves many families of proteins. Among these, the Bcl-2 proteins
are key to the induction of the anti-apoptotic process (46). This protein is overexpressed in many
solid tumors, and contributes to chemotherapy resistance and
radiation-induced apoptosis (46,47).
Unlike many other known human oncogenes, Bcl-2 exerts its influence
by enhancing cell survival rather than by stimulating cell division
(47). In this study, we attempted
to determine whether the expression level of Bcl-2 protein could
affect αIC treatment in hepatocellular carcinoma cells. As shown in
Fig. 4, the expression of the Bcl-2
protein was markedly reduced with increases in the concentration of
αIC. These results bear substantial similarities to the alteration
pattern of apoptosis induced by other compounds, including gomisin
N (13).
In addition, the Bax protein, another member of the
Bcl-2 family, inhibits the anti-apoptotic actions of Bcl-2.
However, Bax may function as a classic tumor suppressor gene in
vivo. Several human tumors involved the loss of functional
mutations for this protein, and the knockout of the Bax gene
induces tumorigenesis in mice (48–51).
When we evaluated the effects of αIC treatment on the expression
level of Bax protein via western blotting, their expression was
increased slightly at a high αIC concentration. These results
indicate that the effects of αIC may be so minimal as to have a
negligible effect on Bax expression.
Genetic mutations of the p53 gene are the most
common alteration noted in hepatocellular carcinoma (52). Generally, the p53 protein functions
as a transcriptional activator, which induces the transcription of
many genes associated with cell metabolism. Among this variety of
genes, apoptosis-related genes are considered important factors of
the regulation process of balance between death and survival
(53). In particular, Bax protein
is a transcriptional target that can be regulated in response to a
variety of p53-dependent apoptosis triggers. Zakaria et
al(20) previously determined
that the upregulation of Bax is stimulated by activated p53 after
eurycomanone treatment. Although p53 activation is an important
step in the regulation of apoptosis, the effects of p53 on the
sensitivity of HepG2 cells to αIC have not been hitherto rigorously
assessed. In this study, we showed that the expression levels of
p53 were reduced dramatically by treatment with high concentrations
of αIC. However, the expression level of Bax upregulated by
activated p53 was slightly increased in HepG2 cells, as αIC was
increased from 40 to 320 μM. Therefore, these results indicate that
the apoptosis of HepG2 cells induced by αIC is regulated via a
p53-independent pathway.
All of the aforementioned results suggest that αIC
may be an excellent novel candidate for the therapeutic treatment
of hepatocellular carcinoma. Furthermore, these compounds regulated
the apoptotic process via a p53-independent pathway, which includes
a slight increase in Bax expression and reduction in p53
expression. However, intensive work remains necessary to define
precisely the role of this compound in inducing cell death of
hepatocellular carcinomas for future clinical applications.
Acknowledgements
This study was supported by Technology Development
Program for Agriculture and Forestry (106048031CG00).
References
|
1
|
Liu GT: Pharmacological actions and
clinical use of Fructus schizandrae. Chin Med J.
102:740–749. 1989.
|
|
2
|
Choi YW, Takamatsu S, Khan SI, Srinivas
PV, Ferreira D, Zhao J and Khan IA: Schisandrene, a
dibenzocyclooctadiene lignan from Schisandra chinensis:
structure-antioxidant activity relationships of
dibenzocyclooctadiene lignans. J Nat Pros. 69:356–359. 2006.
|
|
3
|
Panossian A and Wikman G: Pharmacology of
Schisandra chinensis Bail: an overview of Russian research
and uses in medicine. J Ethnopharmacol. 118:183–212. 2008.
|
|
4
|
Lu Y and Chen DF: Analysis of
Schisandra chinensis and Schisandra sphenanthera. J
Chromatogr A. 1216:1980–1990. 2009.
|
|
5
|
Lee YJ, Shim JW, Lee YJ, Park YH, Lee HY,
Kim SD, Choi YW and Bae YS: Identification of a novel compound that
stimulates intracellular calcium increase and CXCL8 production in
human neutrophils from Schisandra chinensis. Biochem Biophys
Res Commun. 379:928–932. 2009.
|
|
6
|
Choi YW, Kim HJ, Park SS, Chung JH, Lee
HW, Oh SO, Kim BS, Kim JB, Chung HY, Yu BP, Kim CD and Yoon S:
Inhibition of endothelial cell adhesion by the new
anti-inflammatory agent alpha-iso-cubebene. Vascul Pharmacol.
51:215–224. 2009.
|
|
7
|
Lee YJ, Park SY, Kim SG, Park DJ, Kang JS,
Lee SJ, Yoon S, Kim YH, Bae YS and Choi YW: Identification of a
novel compound that inhibits iNOS and COX-2 expression in
LPS-stimulated macrophages from Schisandra chinensis.
Biochem Biophys Res Commun. 391:1687–1692. 2010.
|
|
8
|
Bosch FX, Ribes J, Díaz M and Cléries R:
Primary liver cancer: worldwide incidence and trends.
Gastroenterology. 127:S5–S16. 2004.
|
|
9
|
Adami A, Hunter D and Trichopoulos D:
Cancer of the liver and biliary tract. Textbook of Cancer
Epidemiology. 2nd edition. Oxford University Press; Oxford: pp.
308–332. 2008
|
|
10
|
El-Serag HB and Mason AC: Risk factors for
the rising rates of primary liver cancer in the United States. Arch
Intern Med. 160:3227–3230. 2000.
|
|
11
|
Bruix J and Sherman M: Management of
hepatocellular carcinoma. Hepatology. 42:1208–1236. 2005.
|
|
12
|
Thomas MB and Zhu AX: Hepatocellular
carcinoma: the need for progress. J Clin Oncol. 23:2892–2899.
2005.
|
|
13
|
Yim SY, Lee YJ, Lee YK, Jung SE, Kim HJ,
Kim JE, Kim HJ, Son BG, Park YH, Lee YG, Choi YW and Hwang DY:
Gomisin N isolated from Schisandra chinensis significantly
induces anti-proliferative and pro-apoptotic effects in hepatic
carcinoma. Mol Med Rep. 2:725–732. 2009.
|
|
14
|
Andree HA, Reutelingsperger CP, Hauptmann
R, Hemker HC, Hermens WT and Willems GM: Binding of vascular
anticoagulant alpha (VAC alpha) to planar phospholipid bilayers. J
Biol Chem. 265:4923–4928. 1990.
|
|
15
|
Casciola-Rosen L, Rosen A, Petri M and
Schlissel M: Surface blebs on apoptotic cells are sites of enhanced
procoagulant activity: implications for coagulation events and
antigenic spread in systemic lupus erythematosus. Proc Natl Acad
Sci USA. 93:1624–1629. 1996.
|
|
16
|
Mazumder S, Plesca D and Almasan A:
Caspase-3 activation is a critical determinant of genotoxic
stress-induced apoptosis. Methods Mol Biol. 414:13–21. 2008.
|
|
17
|
Tsujimoto Y: Role of Bcl-2 family proteins
in apoptosis: apoptosomes or mitochondria? Genes Cells. 3:697–707.
1998.
|
|
18
|
Tsujimoto Y and Shimizu S: Bcl-2 family:
life-or-death switch. FEBS Lett. 466:6–10. 2000.
|
|
19
|
McKay BC, Ljungman M and Rainbow AJ:
Potential roles for p53 in nucleotide excision repair.
Carcinogenesis. 20:1389–1396. 1999.
|
|
20
|
Zakaria Y, Rahmat A, Pihie AH, Abdullah NR
and Houghton PJ: Eurycomanone induce apoptosis in HepG2 cells via
up-regulation of p53. Cancer Cell Int. 9:162009.
|
|
21
|
Azzam HS, Goertz C, Fritts M and Jonas WB:
Natural products and chronic hepatitis C virus. Liver Int.
27:17–25. 2007.
|
|
22
|
Chiu PY, Mak DH and Poon MK: In vivo
antioxidant action of a lignan-enriched extract of
Schisandra fruit and an anthraquinone-containing extract of
Polygonum root in comparison with schisandrin B and emodin.
Planta Med. 68:951–956. 2002.
|
|
23
|
Ko KM: Schisandrin B protects against
tert-butylhydroperoxide induced cerebral toxicity by enhancing
glutathione antioxidant status in mouse brain. Mol Cell Biochem.
238:181–186. 2002.
|
|
24
|
Nakamura K, Yoshida M and Uchiwa H:
Down-regulation of melanin synthesis by a biphenyl derivative and
its mechanism. Pigment Cell Res. 16:494–500. 2003.
|
|
25
|
Panossian AG: Effects of heavy physical
exercise and adaptogens on nitric oxide content in human saliva.
Phytomedicine. 6:17–26. 1999.
|
|
26
|
Yim TK and Ko KM: Schisandrin B protects
against myocardial ischemia-reperfusion injury by enhancing
myocardial glutathione antioxidant status. Mol Cell Biochem.
196:151–156. 1999.
|
|
27
|
Baek MS, Kim JY and Myung SW: Metabolism
of dimethyl-4,4′-dimethoxy-5,6,5′,6′-dimethylene
dioxybiphenyl-2,2′-dicarboxylate (DDB) by human liver microsomes:
characterization of metabolic pathways and of cytochrome P450
isoforms involved. Drug Metab Dispos. 29:381–388. 2001.
|
|
28
|
Iwata H, Tezuka Y and Kadota S:
Identification and characterization of potent CYP3A4 inhibitors in
Schisandra fruit extract. Drug Metab Dispos. 32:1351–1358.
2004.
|
|
29
|
Chang HF, Lin YH, Chu CC, Wu SJ, Tsai YH
and Chao JC: Protective effects of Ginkgo biloba, Panax
ginseng, and Schizandra chinensis extract on liver
injury in rats. Am J Chin Med. 35:995–1009. 2007.
|
|
30
|
Ko KM, Ip SP, Poon MK, Wu SS, Che CT, Ng
KH and Kong YC: Effect of a lignan-enriched fructus schisandrae
extract on hepatic glutathione status in rats: protection against
carbon tetrachloride toxicity. Planta Med. 61:134–137. 1995.
|
|
31
|
Chen N, Chiu PY and Ko KM: Schisandrin B
enhances cerebral mitochondrial antioxidant status and structural
integrity, and protects against cerebral ischemia/reperfusion
injury in rats. Biol Pharm Bull. 31:1387–1391. 2008.
|
|
32
|
Kubo S, Ohkura Y, Mizoguchi Y,
Matsui-Yuasa I, Otani S, Morisawa S, Kinoshita H, Takeda S, Aburada
M and Hosoya E: Effect of Gomisin A (TJN-101) on liver
regeneration. Planta Med. 58:489–492. 1992.
|
|
33
|
He XG, Lian LZ and Lin LZ: Electrospray
high performance liquid chromatography-mass spectrometry in
phytochemical analysis of kava (Piper methysticum) extract.
Planta Med. 63:70–74. 1997.
|
|
34
|
Chen DF, Zhang SX, Kozuka M, Sun QZ, Feng
J, Wang Q, Mukainaka T, Nobukuni Y, Tokuda H, Nishino H, Wang HK,
Morris-Natschke SL and Lee KH: Interiotherins C and D, two new
lignans from Kadsura interior and antitumor-promoting effects of
related neolignans on Epstein-Barr virus activation. J Nat Prod.
65:1242–1245. 2002.
|
|
35
|
Yasukawa K, Ikeya Y, Mitsuhashi H, Iwasaki
M, Aburada M, Nakagawa S, Takeuchi M and Takido M: Gomisin A
inhibits tumor promotion by 12-O-tetradecanoylphorbol-13-acetate in
two-stage carcinogenesis in mouse skin. Oncology. 49:68–71.
1992.
|
|
36
|
Nomura M, Nakachiyama M, Hida T, Ohtaki Y,
Sudo K, Aizawa T, Aburada M and Miyamoto KI: Gomisin A, a lignan
component of Schizandora fruits, inhibits development of
preneoplastic lesions in rat liver by
3′-methyl-4-dimethylamino-azobenzene. Cancer Lett. 76:11–18.
1994.
|
|
37
|
Ohtaki Y, Hida T, Hiramatsu K, Kanitani M,
Ohshima T, Nomura M, Wakita H, Aburada M and Miyamoto KI:
Deoxycholic acid as an endogenous risk factor for
hepatocarcinogenesis and effects of gomisin A, a lignan component
of Schizandra fruits. Anticancer Res. 16:751–755. 1996.
|
|
38
|
Wu MD, Huang RL, Kuo LM, Hung CC, Ong CW
and Kuo YH: The anti-HBsAg (human type B hepatitis, surface
antigen) and anti-HBeAg (human type B hepatitis, e antigen) C18
dibenzocyclooctadiene lignans from Kadsura matsudai and
Schizandra arisanensis. Chem Pharm Bull (Tokyo).
51:1233–1236. 2003.
|
|
39
|
Chen DF, Zhang SX, Xie L, Xie JX, Chen K,
Kashiwada Y, Zhou BN, Wang P, Cosentino LM and Lee KH: Anti-AIDS
agents-XXVI. Structure-activity correlations of gomisin-G-related
anti-HIV lignans from Kadsura interior and of related
synthetic analogues. Bioorg Med Chem. 5:1715–1723. 1997.
|
|
40
|
Lu H and Liu GT: Anti-oxidant activity of
dibenzocyclooctene lignans isolated from Schisandraceae. Planta
Med. 58:311–313. 1992.
|
|
41
|
Park JY, Lee SJ, Yun MR, Seo KW, Bae SS,
Park JW, Lee YJ, Shin WJ, Choi YW and Kim CD: Gomisin A from
Schisandra chinensis induces endothelium-dependent and
direct relaxation in rat thoracic aorta. Planta Med. 73:1537–1542.
2007.
|
|
42
|
Bursch W, Ellinger A, Gerner C, Fröhwein U
and Schulte-Hermann R: Programmed cell death (PCD): apoptosis,
autophagic PCD, or others? Ann NY Acad Sci. 926:1–12. 2000.
|
|
43
|
Schwartz LM, Smitho SW, Jones MEE and
Osborne BA: Do all programmed cell deaths occur via apoptosis? Proc
Natl Acad Sci USA. 90:980–984. 1993.
|
|
44
|
Homburg CH, de Haas M, von dem Borne AE,
Verhoeven AJ, Reutelingsperger CP and Roos D: Human neutrophils
lose their surface Fc gamma RIII and acquire Annexin V binding
sites during apoptosis in vitro. Blood. 85:532–540. 1995.
|
|
45
|
Koopman G, Reutelingsperger CP, Kuijten
GA, Keehnen RM, Pals ST and van Oers MH: Annexin V for flow
cytometric detection of phosphatidylserine expression on B cells
undergoing apoptosis. Blood. 84:1415–1420. 1994.
|
|
46
|
Apakama I, Robinson MC, Walter NM,
Charlton RG, Royds JA, Fuller CE, Neal DE and Hamdy FC: Bcl-2
overexpression combined with p53 accumulation correlates with
hormone refractory prostate cancer. Br J Urol. 74:1258–1262.
1996.
|
|
47
|
Joensuu H, Pylkkänen L and Toikkanen S:
Bcl-2 protein expression and long-term survival in breast cancer.
Am J Pathol. 145:1191–1198. 1994.
|
|
48
|
Baekelandt M, Holm R, Nesland JM, Tropé CG
and Kristensen GB: Expression of apoptosis-related proteins is an
independent determinant of patient prognosis in advanced ovarian
cancer. J Clin Oncol. 18:3745–3747. 2000.
|
|
49
|
Harima Y, Nagata K, Harima K, Oka A,
Ostapenko VV, Shikata N, Ohnishi T and Tanaka Y: Bax and bcl-2
protein expression following radiation therapy versus radiation
plus thermoradiotherapy in stage IIIB cervical carcinoma. Cancer.
88:132–138. 2000.
|
|
50
|
Ito T, Fujieda S, Tsuzuki H, Sunaga H, Fan
G, Sugimoto C, Fukuda M and Saito H: Decreased expression of bax is
correlated with poor prognosis in oral and oropharyngeal carcinoma.
Cancer Lett. 140:81–91. 1999.
|
|
51
|
Yin C, Knudson CM, Korsmeyer SJ and van
Dyke T: Bax suppresses tumorigenesis and stimulates apoptosis in
vivo. Nature. 385:637–640. 1997.
|
|
52
|
Fabregat I: Dysregulation of apoptosis in
hepatocellular carcinoma cells. World J Gastroenterol. 15:513–520.
2009.
|
|
53
|
McGill G and Fisher DE: Apoptosis in
tumorigenesis and cancer therapy. Front Bioscience. 2:D353–D379.
1997.
|