Introduction
Prostate cancer (PC) is the most common cancer in
men over the age of 50 years. PC represents one of the leading
causes of cancer-related mortality in Western countries (1–3) and is
associated with the most rapidly increasing rate of diagnosis in
Japanese men (4). Thus, it is
essential to clearly delineate the risk factors, diagnosis,
treatment options and emerging therapies to better understand and
to detect the onset of PC. A diet rich in fruits and vegetables has
been reported to reduce the risk of common types of cancer and may
prove useful in cancer prevention. Moreover, since
less-differentiated tumors become resistant to a wide variety of
cytotoxic drugs, considerable attention has been focused on
chemoprevention with natural compounds as a new and alternative
approach to cancer control. Epidemiological studies have shown the
ability of dietary compounds to act epigenetically against cancer
cells and to influence an individual's risk of developing cancer
(5). Several natural antioxidants,
in particular polyphenols, have been reported to exhibit
chemotherapeutic activity both in vivo and in
vitro(6–10). EA
(2,3,7,8-tetrahydroxy[1]-benzopyrano[5,4,3-cde][1]benzopyran-5,10-dione)
is a natural polyphenol found, as both free and bound forms, in
numerous fruits and vegetables, particularly in pomegranate
(11–15). EA exhibits antioxidant and
anticarcinogenic properties including inhibition of tumor formation
and growth both in vitro and in vivo(16–25).
Moreover, EA was found to inhibit human prostate cancer cell
invasion (23).
Our previous research demonstrated a dose-dependent
cytotoxic effect of EA, resulting in a reduction in the
proliferation rate and a marked increase in DNA damage in prostatic
cancer cell lines (26). Moreover,
EA reduced chromogranin A (CgA) levels and increased p75 nerve
growth factor receptor (p75NGFR) expression, resulting in the
reversion of prostatic cancer cell lines from a proliferating to a
differentiated state (26).
Therefore, EA with both anti-proliferative and pro-differentiation
properties is promising as a cancer therapeutic agent.
Tumorigenesis is a multistep process activated by
various environmental carcinogens, inflammatory agents and tumor
promoters. These carcinogens modulate transcription factors,
anti-apoptotic proteins, pro-apoptotic proteins, protein kinases,
cell cycle proteins, cell adhesion molecules and growth factor
signaling pathways. EA was found to inhibit cell growth and induce
apoptosis in a variety of cell cultures (16,18,23). A
multitude of factors modulate apoptosis including growth factors,
intracellular mediators of signal transduction, nuclear proteins
regulating gene expression, DNA replication and cell cycle
regulatory genes (27–31). Moreover, the implications for ROS
regulation are highly significant for cancer therapy. Commonly used
radio-therapeutic and chemotherapeutic drugs influence cancer
outcome through ROS modulation.
The present study examined the involvement of
apoptotic markers in the cytotoxic effects exerted by EA on the
LNCaP human prostatic cancer cell line. In particular, we
investigated the anti-carcinogenic properties of EA by evaluating
its ability to induce cell cycle arrest and apoptosis. We evaluated
mTOR, SIRT1, β-catenin, HUR, AIF, caspase-3, p21, IL-6 and TGF-β.
In addition we examined the effects of EA on the cell cycle and
showed that EA regulates apoptosis in the LNCaP prostatic cancer
cell line.
Materials and methods
Cell culture and treatments
Frozen LNCaP cells were purchased from the American
Type Culture Collection (Rockville, MD, USA). After thawing, LNCaP
cells were re-suspended in RPMI-1640 medium (Sigma-Aldrich, St.
Louis, MO, USA), supplemented with 10% heat inactivated fetal
bovine serum (FBS) and 1% antibiotic/antimycotic solution (both
from Invitrogen Life Technologies, Carlsbad, CA, USA). The cells
were plated at a density of 1–5×106 cells/T75 flask.
Cell cultures were maintained at 37°C in a 5% CO2
incubator, and the medium was changed after 3–4 days. Subconfluent
cells were treated for 48 h with 2 different concentrations (25 and
50 μM) of freshly prepared EA dissolved in dimethyl sulfoxide
(DMSO). Control groups received DMSO alone.
Immunoblot analysis
Cells were cultured in T75 flasks for 48 h. They
were then washed with PBS and trypsinized (0.05% trypsin w/v with
0.02% EDTA). The pellets were lysed in buffer (50 mM Tris-HCl, 10
mM EDTA, 1% v/v Triton X-100, 1% PMSF, 0.05 mM pepstatin A and 0.2
mM leupeptin) and after mixing with sample loading buffer (50 mM
Tris-HCl, 10% w/v SDS, 10% v/v glycerol, 10% v/v 2-mercaptoethanol
and 0.04% bromophenol blue) at a ratio of 4:1, were boiled for 5
min. Samples (20 μg proteins) were loaded onto 8 or 12%
SDS-polyacrylamide (SDS-PAGE) gels and subjected to electrophoresis
(120 V, 90 min). The separated proteins were transferred to
nitrocellulose membranes (Bio-Rad, Hercules, CA, USA). After
transfer, the blots were incubated with Li-Cor blocking buffer for
1 h, followed by overnight incubation with a 1:1,000 dilution of
the primary antibody. Primary polyclonal antibodies directed
against AIF, β-catenin, p-mTOR, SIRT-1, caspase-3 and p21 were
purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA)
while HuR and TGF-β were purchased from Santa Cruz Biotechnology,
Inc. (Dallas, TX, USA). After washing with TBS, the blots were
incubated for 1 h with the secondary antibody (1:1,000). Protein
detection was carried out using a secondary infrared fluorescent
dye-conjugated antibody absorbing at 800 and 700 nm as described
below. The blots were visualized using an Odyssey Infrared imaging
scanner (LI-COR Biosciences) and quantified by densitometric
analysis performed after normalization with β-actin (Santa Cruz).
Results are expressed as arbitrary units (A.U.).
Cell cycle analysis
Cells were cultured as previously described, fixed
in 70% ethanol overnight at −20°C and washed with
phosphate-buffered saline (PBS). Aliquots of 1×106 cells
were re-suspended in 1 ml of PBS containing 1 mg/ml of RNase A and
0.5 mg/ml propidium iodide (PI). After a 30-min incubation, the
cells were analyzed by flow cytometry using a FACScan flow
cytometer (FACSCalibur; BD Biosciences, Franklin Lakes, NJ, USA)
and evaluated by fluorescence-activated cell sorting (FACS)
analysis to identify the cells at different stages of the cell
cycle.
IL-6 measurements
IL-6 levels were determined in the culture
supernatant using an ELISA kit (AssayGate Inc., Ijamsville, MD,
USA). The assays were performed according to the manufacturer's
guidelines. Results are expressed as pg/ml.
In-cell western blotting
Cells were seeded in a 96-well cell culture plate.
After a 48-h treatment with 25 or 50 μM EA, cells were washed in
PBS and directly fixed with 4% of paraformaldehyde (PFA) in PBS for
20 min. Cells were permeabilized with 0.2% Triton X-100, blocked
with Li-Cor blocking buffer for 60 min at room temperature,
followed by overnight incubation with rabbit HO-1 (1:500) and mouse
β-actin primary antibody (1:1,000). β-actin was used as a
housekeeping gene to normalize the HO-1 signal for the cell number.
After 3 washes, protein detection was carried out using a secondary
infrared fluorescent dye-conjugated antibody absorbing at 800 or
700 nm. The whole plate was visualized using an Odyssey Infrared
imaging scanner with a 700-nm fluorophore (red dye) and 800-nm
fluorophore (green dye). Relative fluorescence units from the
scanning allowed a quantitative analysis of the proteins.
ROS measurement
Determination of ROS was performed using a
fluorescent probe 2′,7′-dichlorofluorescein diacetate (DCFH-DA), as
previously described (32). The
fluorescence [corresponding to the oxidized radical species
2′,7′-dichlorofluorescein (DCF)] was monitored
spectrofluorometrically (excitation, λ=488 nm; emission, λ=525 nm).
Thus, the intensity of fluorescence was proportional to the levels
of oxidant species. The results are reported as fluorescence
intensity/mg protein. Protein content was measured according to the
method described by Bradford (33).
Statistical analyses
Statistical significance between experimental groups
was determined by the Fisher's method of analysis of multiple
comparisons (p<0.05). For comparisons among treatment groups,
the null hypothesis was tested by a 2-factor ANOVA for multiple
groups or unpaired t-test for 2 groups. Data are presented as means
± SD.
Results
Effect of EA on β-catenin, p-mTOR, HuR
and SIRT1 expression
We assessed the levels of β-catenin, p-mTOR, and
SIRT1 after 48 h of culture in the presence of EA. As shown in
Fig. 1, EA treatment (25 and 50 μM)
resulted in a decrease in both β-catenin and p-mTOR protein
expression (p<0.05). Similarly, EA exposure showed a significant
(p<0.05) reduction in the RNA-binding protein HuR and
consequently in SIRT1 (p<0.05). m-TOR protein expression
remained unchanged upon EA treatment (data not shown).
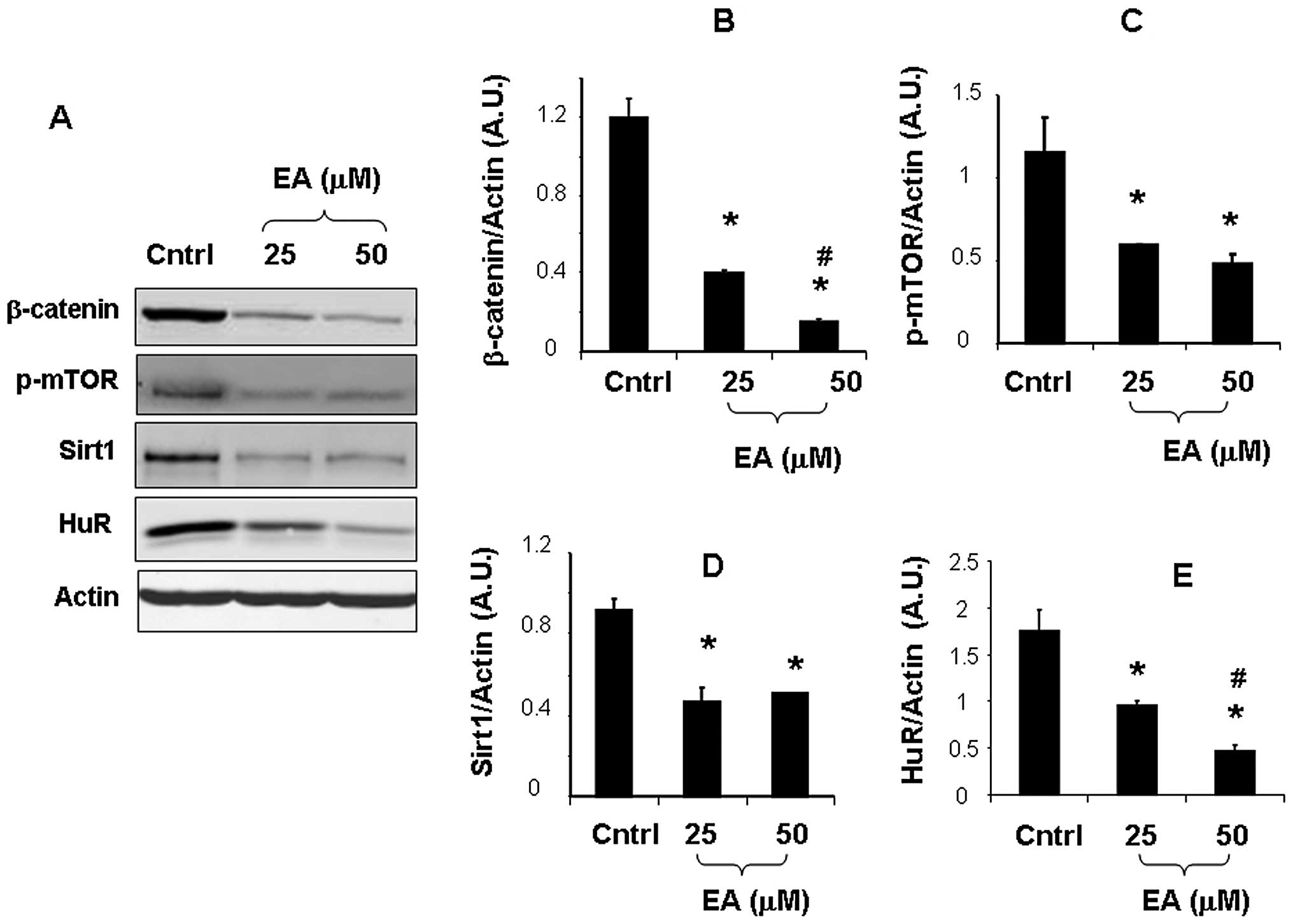 | Figure 1(A) Representative western blotting
of β-catenin, p-mTOR, Sirt1 and HuR protein expression in cultured
LNCaP cells. (B–E) Effect of EA (25 and 50 μM) on β-catenin,
p-mTOR, Sirt1 and HuR expression in cultured LNCaP cells. Results
are expressed as arbitrary units (A.U.), and represent the means ±
SD of 4 experiments performed in triplicate. *p<0.05,
significant result of 25 and 50 μM EA vs. control.
#p<0.005, significant result of 50 μM EA vs. 25 μM
EA. EA, ellagic acid; Sirt1, silent information regulator 1; HuR,
human antigen R; Cntrl, control. |
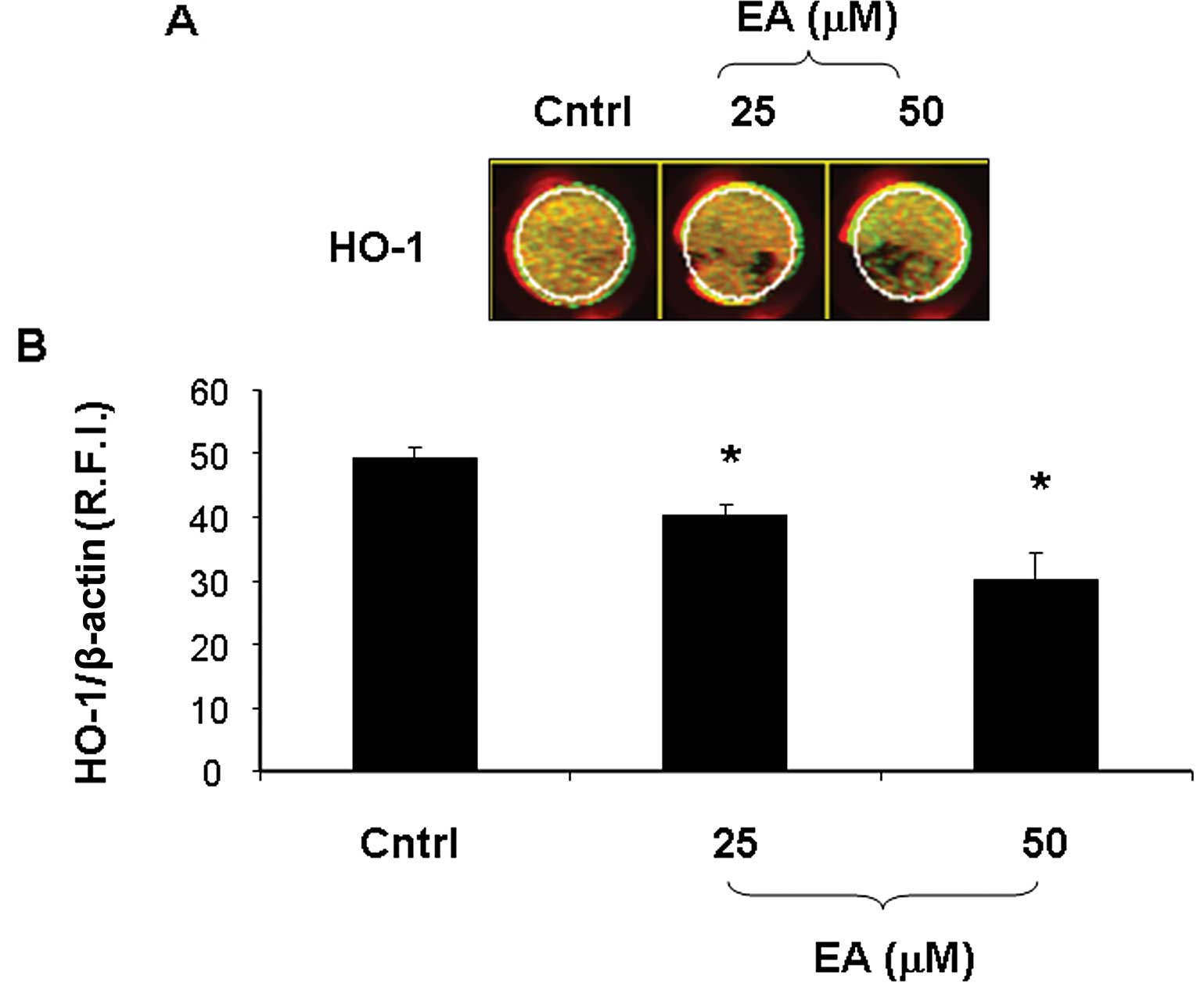
Effect of EA on HO-1 expression
HO-1 protein levels were examined by in-cell western
blot analysis, to quantify total endogenous cellular protein
(Fig. 2). LNCaP cells treated with
EA (25 and 50 μM) showed a marked decrease (p<0.005) in HO-1
fluorescence. HO-1 values were background subtracted from wells
treated only with secondary antibody, and then normalized to the
cell number by dividing the total actin fluorescence signal.
Effect of EA on AIF, caspase-3, p21 and
TGF-β expression
Fig. 3 shows the
densitometric analysis of AIF, caspase-3, p21 and TGF-β expression
in untreated and EA-treated LNCaP cells. EA treatment induced a
significant (p<0.05) decrease in total AIF, caspase-3 and TGF-β
expression. In contrast p21 expression was increased (p<0.05) in
the EA-treated group when compared to the controls. Immunoblot
analysis showed an increase (p<0.05) in cleaved-AIF and
cleaved-caspase-3 in cells treated with EA.
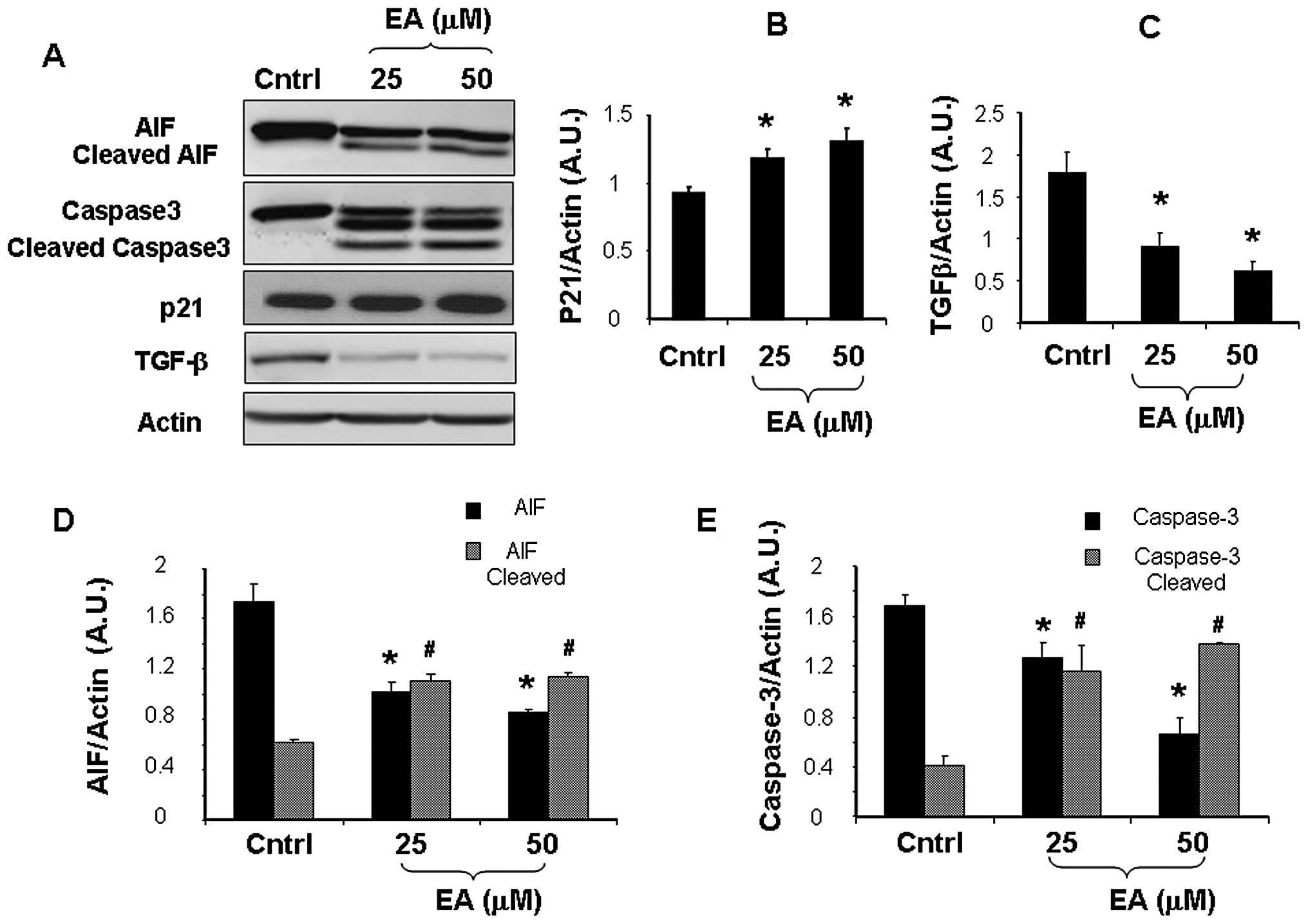 | Figure 3(A) Representative western blotting
of AIF, caspase-3, p21, TGF-β protein expression in cultured LNCaP
cells. (B–E) Effect of EA (25 and 50 μM) on AIF, caspase-3, p21,
TGF-β expression in cultured LNCaP cells. Results are expressed as
arbitrary units (A.U.), and represent the means ± SD of 4
experiments performed in triplicate. *p<0.05,
significant result of 25 and 50 μM EA vs. (black) control;
#p<0.05, significant result of 25 and 50 μM EA vs.
(grey) control. EA, ellagic acid; AIF, apoptosis-inducing factor;
TGF-β, transforming growth factor-β; Cntrl, control. |
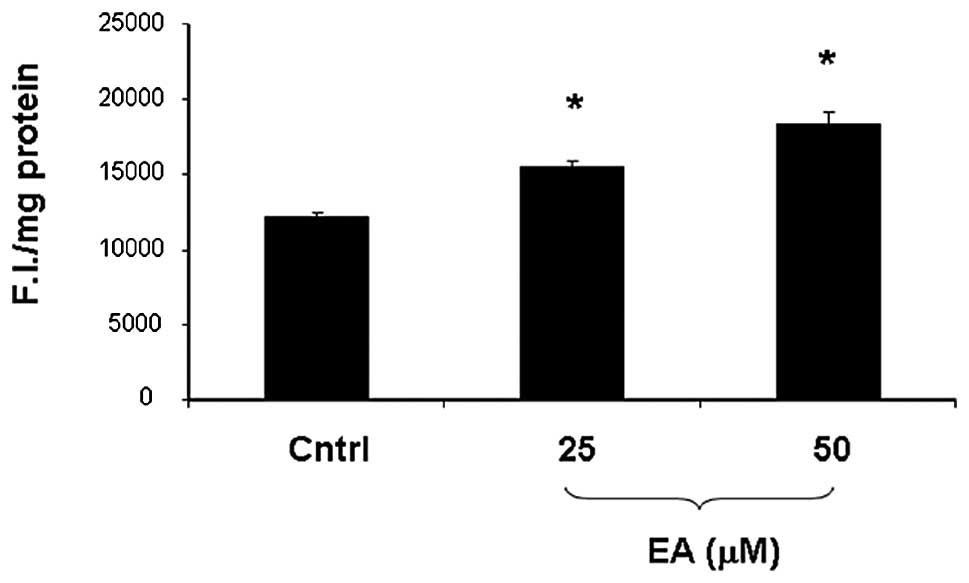
ROS levels
As shown in Fig. 4,
exposure of LNCaP cells to both 25 and 50 μM EA resulted in a
significant increase (p<0.005) in ROS levels when compared to
the untreated cells. The effect appeared to be dose dependent and
did not reach a plateau at the doses examined.
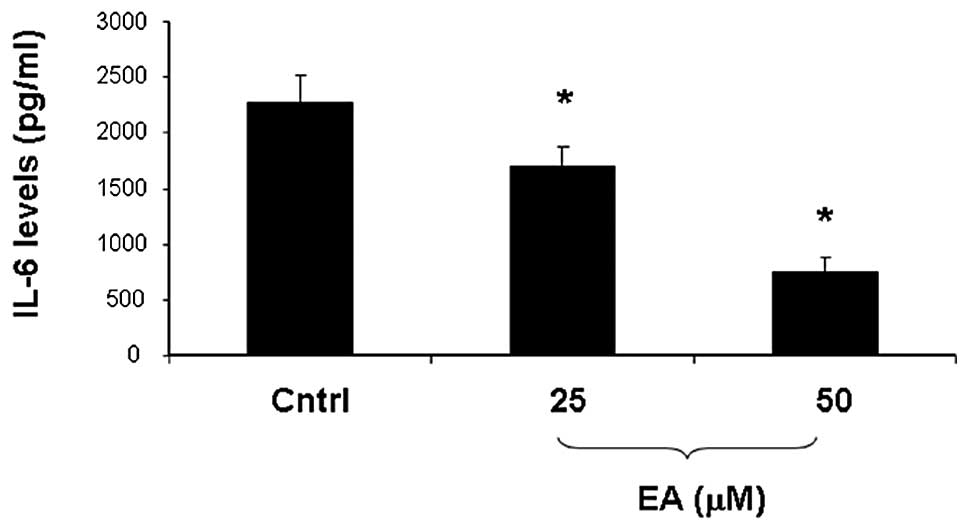
Effect of EA on IL-6 levels
The effect of EA on IL-6 levels is shown in Fig. 5. EA treatment induced a significant
(p<0.05) concentration-dependent decrease IL-6 in levels when
compared to the control cells.
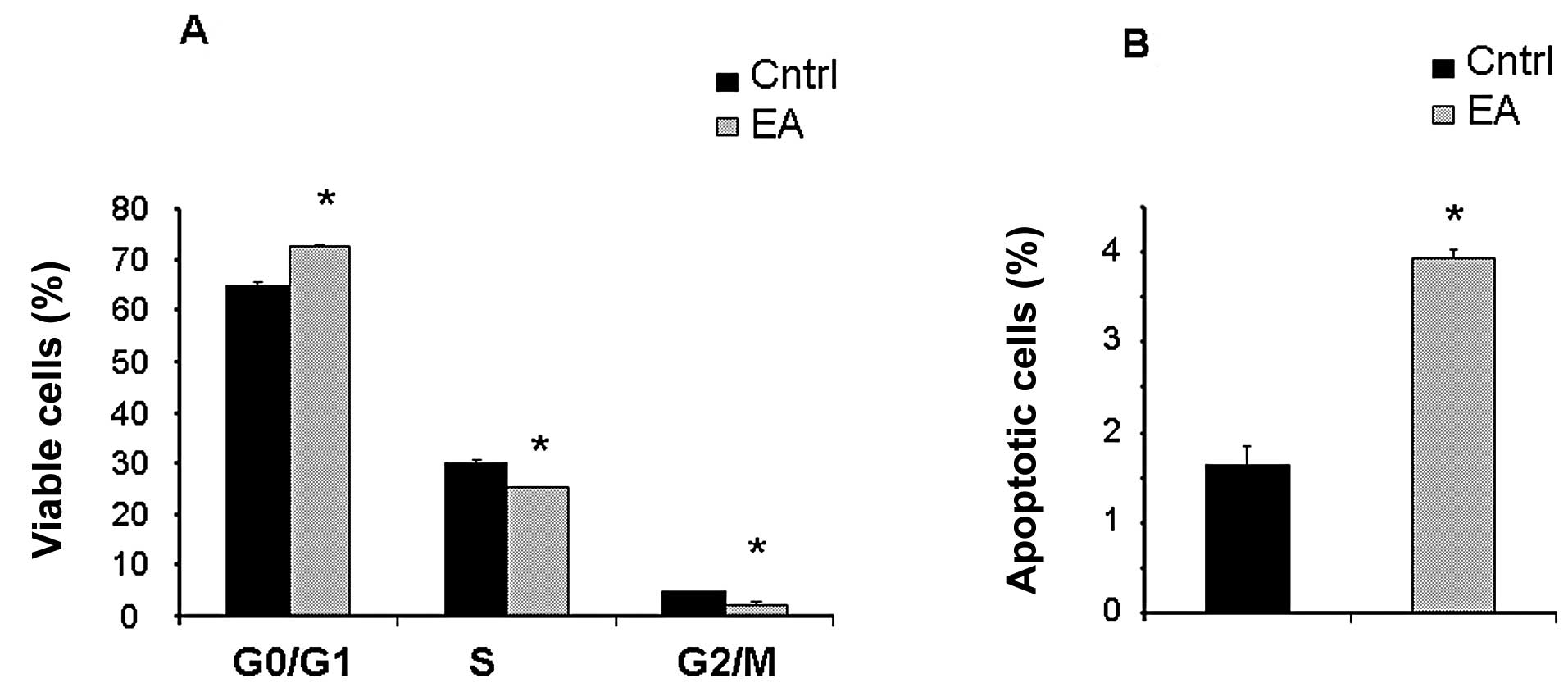
Cell cycle analysis
Flow cytometric analysis of the cell cycle
distribution is shown in Fig. 6.
Our results revealed that EA (50 μM) induced a slight but
significant (p<0.005) increase in the percentage of cells in the
G0/G1 or resting phase, a limited but
significant (p<0.005) decrease in the percentage of cells in the
S (or synthetic) and in the G2/M (or mitotic) phases
with concomitant increase (p<0.005) in the percentage of
apoptotic cells. EA at 25 μM did not induce any modification in
cell cycle distribution (data not shown).
Discussion
Prostrate cancer is a chronic disease that develops
from a small lesion to clinical manifestation over an extended
period of time. However, once the disease is metastatic, patient
prognosis is poor. Thus, the development of new strategies to fight
PC has become an important therapeutic mission. The administration
of both synthetic and naturally occurring agents to suppress,
reverse and delay carcinogenesis, is increasingly being touted as
an effective approach for the management of prostatic neoplasia
(34–37). In recent years, naturally occurring
antioxidant compounds present in the human diet have gained
considerable attention as cancer-chemopreventive and
chemotherapeutic agents (34,36–38).
As previously reported (26),
naturally occurring polyphenol EA is regarded as a promising new
class of cancer therapeutic agents, with both anti-proliferative
and pro-differentiating properties.
β-catenin is a subunit of a protein complex acting
as a signal transducer, and aberrant accumulation of intracellular
β-catenin is a well-recognized characteristic of several types of
cancers, including prostate, colon and liver (39–42).
The reduction in β-catenin levels, found in the present study,
strongly suggests that this represents a potential mechanism
implicated in the anti-proliferative effects of EA.
We showed that EA treatment exerts
anti-proliferative effects by reducing intracellular levels of
β-catenin. We previously demonstrated that EA reduced Akt
activation/phosphorylation in prostate cancer cell lines. The
capacity of p-Akt to phosphorylate/activate mTOR has been described
in several cancer cell lines (43–45).
Dysregulation of the mTOR pathway occurs in many types of cancers
including prostate (43). The
inhibition of mTOR activation is considered as a potential target
for the development of anticancer therapeutics (43–49).
In view of these observations, the significant reduction in mTOR
activation, observed in the present study, indicates that EA exerts
an anti-proliferative effect by reducing PI3K/Akt downstream
signaling through inhibition of mTOR phosphorylation. In addition,
acetylation of Akt blocks Akt binding to PIP3, thereby
preventing membrane localization and phosphorylation of Akt.
Deacetylation by SIRT1 enhances Akt binding to PIP3 and
promotes activation/phoshorylation (50).
SIRT1 functions as an oncogenic protein and plays a
role in tumorigenesis (51). SIRT1
is overexpressed in human PC cells (DU145, LNCaP, 22Rν1 and PC3)
when compared to normal prostate epithelial cells (PrEC), and
inhibition of SIRT1 results in anti-proliferative effects in human
PC cells. SIRT1 was found to be overexpressed in human PC tissues
when compared with adjacent normal prostate tissue (52). A link between SIRT1 and HuR levels
and the anti-apoptotic influence of these 2 proteins (53) has been reported. The RNA-binding
protein HuR regulates the stability of many target mRNAs, including
SIRT1 mRNA. The reduction in SIRT1 and HuR, observed in the present
study following EA treatment, suggests that EA may induce apoptosis
via the reduction of the anti-apoptotic proteins SIRT1 and HuR.
Moreover, HO-1 translation was found to be diminished in
HuR-depleted cells (54). An
increase in HO-1 protein levels is associated with a parallel
increase in EC-SOD, eNOS (55),
increased activation of Akt and an increase in mitochondrial
function. HuR promotes HO-1 expression through mRNA stabilization
and translational upregulation. Our results confirmed that
decreased levels of HuR, after EA treatment, were associated with a
reduction in HO-1 protein expression suggesting that HO-1 is a
focal target in cancer treatment. Thus, the anti-proliferative
effect of EA includes the inactivation of PI3K/Akt signaling
cascade, i.e. the inhibition of Akt, mTOR and SIRT1, a decrease in
HO-1 levels and, consequently, the activation of the
mitochondrial-mediated apoptotic pathway.
AIF is a flavoprotein anchored to the mitochondrial
inner membrane. Under physiological conditions, AIF exhibits NADH
oxidase activity, important for mitochondrial respiration (56). A previous study with mice
characterized by reduced AIF expression, the ‘Harlequin’ (Hq) mice,
suggested that AIF may also protect against oxidative stress
(57). Although mislocalization of
the AIF protein leads to nuclear condensation and apoptosis, in
vivo models of AIF loss result in oxidative stress and neuronal
degeneration (57). Upon a specific
death signal, AIF is cleaved at the N-terminus releasing the
protein from its membrane anchor (58). AIF translocation from the cytosol to
the nucleus results in chromatin condensation and large-scale DNA
fragmentation, and apoptotic cell death (59) in a caspase-independent manner
(60). We noted a decrease in AIF
expression and an increase in cleaved AIF upon exposure of the
LNCaP cells to EA. The observation that EA modulates AIF expression
has at least 2 important conceptual implications. Firstly, EA has a
pro-oxidant potential; secondly, a direct link exists between EA
and the induction of cleaved AIF followed by activation of the
apoptotic pathways. In addition, cancer chemopreventive agents
induce apoptosis through ROS generation and disruption of redox
homeostasis leading to the activation of the caspase cascade
(61,62). In agreement with previous findings
(63), our results showed that EA
increased ROS levels and activation of caspases, as demonstrated by
a decrease in caspase-3 with a concomitant increase of its cleaved
form. Therefore, EA-induced ROS production and activation of
caspase-3 may be due to AIF cleavage.
IL-6 is a multifunctional cytokine and a major
activator of different signaling pathways. It regulates growth of
prostate cancer (64) and inhibits
apoptosis in several prostate cancer cell lines (65). Increased IL-6 levels, found in
prostate cancer, may be due to enhanced expression of TGF-β
(66), which regulates cell
proliferation, phenotype and matrix synthesis. In cancer cells,
TGF-β acts on the surrounding stromal, immune, endothelial and
smooth-muscle cells, causing immunosuppression and angiogenesis,
resulting in a more invasive form of cancer (67). We demonstrated in the present study
that EA reduces both TGF-β and IL-6 levels. Overall, EA induces
both caspase-independent and caspase-dependent apoptotic cell
death. EA caused a significant increase in p21 expression, a
negative regulator of the cell cycle. In addition, FACS analysis
revealed that EA treatment induced a different distribution of
cells in the various phases of the cell cycle, with a concomitant
increase in apoptotic cells. Moreover, in addition to
pro-differentiating properties, the cytotoxic effects of EA may be
due to its ability to modulate the expression of multiple apoptotic
markers. In conclusion, the present study strongly supports the
hypothesis that EA regulates apoptosis through
activation/inactivation of several proteins involved in tumor
growth and cell invasiveness. EA may represent a new approach and a
highly effective strategy for reducing the occurrence of prostate
cancer.
References
|
1
|
Ferlay J, Parkin DM and Steliarova-Foucher
E: Estimates of cancer incidence and mortality in Europe in 2008.
Eur J Cancer. 46:765–781. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
American Cancer Society. Prostate Cancer
Statistics. 2013, http://www.cancer.org.
Accessed: May 20, 2013
|
|
3
|
American Society of Clinical Oncology
Prostate Cancer Statistics. http://www.cancer.net/prostate.
Accessed: May 20, 2013
|
|
4
|
Imamoto T, Suzuki H, Akakura K, Komiya A,
Nakamachi H, Ichikawa T, Igarashi T and Ito H: Pretreatment serum
level of testosterone as a prognostic factor in Japanese men with
hormonally treated stage D2 prostate cancer. Endocr J. 48:573–578.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen J and Xu X: Diet, epigenetic, and
cancer prevention. Adv Genet. 71:237–255. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kang NJ, Shin SH, Lee HJ and Lee KW:
Polyphenols as small molecular inhibitors of signaling cascades in
carcinogenesis. Pharmacol Ther. 130:310–324. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Weng CJ and Yen GC: Chemopreventive
effects of dietary phytochemicals against cancer invasion and
metastasis: phenolic acids, monophenol, polyphenol, and their
derivatives. Cancer Treat Rev. 38:76–87. 2012. View Article : Google Scholar
|
|
8
|
Russo A, Piovano M, Lombardo L, Vanella L,
Cardile V and Garbarino J: Pannarin inhibits cell growth and
induces cell death in human prostate carcinoma DU-145 cells.
Anticancer Drugs. 17:1163–1169. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yim D, Singh RP, Agarwal C, Lee S, Chi H
and Agarwal R: A novel anticancer agent, decursin, induces G1
arrest and apoptosis in human prostate carcinoma cells. Cancer Res.
65:1035–1044. 2005.PubMed/NCBI
|
|
10
|
Cardile V, Scifo C, Russo A, Falsaperla M,
Morgia G, Motta M, Renis M, Imbriani E and Silvestre G: Involvement
of HSP70 in resveratrol-induced apoptosis of human prostate cancer.
Anticancer Res. 23:4921–4926. 2003.PubMed/NCBI
|
|
11
|
Aviram M, Dornfield L, Rosenblat M,
Volkova N, Kaplan M, Coleman R, Hayek T, Presser D and Fuhrman B:
Pomegranate juice consumption reduces oxidative stress, atherogenic
modifications to LDL, and platelet aggregation: studies in humans
and in atherosclerotic apolipoprotein E-deficient mice. Am J Clin
Nutr. 71:1062–1076. 2000.
|
|
12
|
Kaplan M, Hayek T, Raz A, Coleman R,
Dornfeld L, Vaya J and Aviram M: Pomegranate juice supplementation
to atherosclerotic mice reduces macrophage lipid peroxidation,
cellular cholesterol accumulation and development of
atherosclerosis. J Nutr. 131:2082–2089. 2001.
|
|
13
|
Kim ND, Mehta R, Yu W, Neeman I, Livney T,
Amichay A, Poirier D, Nicholls P, Kirby A, Jiang W, Mansel R,
Ramachandran C, Rabi T, Kaplan B and Lansky E: Chemopreventive and
adjuvant therapeutic potential of pomegranate (Punica
granatum) for human breast cancer. Breast Cancer Res Treat.
71:203–217. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Cerdá B, Cerón JJ, Tomás-Barberán FA and
Espín JC: Repeated oral administration of high doses of pomegranate
ellagitannin punicalagin to rats for 37 days is not toxic. J Agric
Food Chem. 51:3493–3501. 2003.PubMed/NCBI
|
|
15
|
Cerdá B, Llorach R, Cerón JJ, Espín JC and
Tomás-Barberán FA: Evaluation of the bioavailability and metabolism
in the rat of punicalagin, an antioxidant polyphenol from
pomegranate juice. Eur J Nutr. 42:18–28. 2003.PubMed/NCBI
|
|
16
|
Narayanan BA, Geoffrey O, Willingham MC,
Re GG and Nixon DW: p53/p21(WAF1/CIP1) expression and its possible
role in G1 arrest and apoptosis in ellagic acid-treated cancer
cells. Cancer Lett. 136:215–221. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Khanduja KL, Gandhi RK, Pathania V and
Syanl N: Prevention of N-nitrosodiethylamine-induced lung
tumorigenesis by ellagic acid and quercetin in mice. Food Chem
Toxicol. 37:313–318. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mertens-Talcott SU, Talcott ST and
Percival SS: Low concentrations of quercetin and ellagic acid
synergistically influence proliferation, cytotoxicity and apoptosis
in MOLT-4 human leukemia cells. J Nutr. 133:2669–2674. 2003.
|
|
19
|
Heber D: Multitargeted therapy of cancer
by ellagitannins. Cancer Lett. 269:262–268. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Seeram NP, Adams LS, Henning SM, Niu Y,
Zhang Y, Nair MG and Heber D: In vitro antiproliferative, apoptotic
and antioxidant activities of punicalagin, ellagic acid and a total
pomegranate tannin extract are enhanced in combination with other
polyphenols as found in pomegranate juice. J Nutr Biochem.
16:360–367. 2005. View Article : Google Scholar
|
|
21
|
Seeram NP, Aronson WJ, Zhang Y, Henning
SM, Moro A, Lee RP, Sartippour M, Harris DM, Rettig MB, Suchard MA,
Pantuck AJ, Belldegrun A and Heber D: Pomegranate
ellagitannin-derived metabolites inhibit prostate cancer growth and
localize to the mouse prostate gland. J Agric Food Chem.
55:7732–7737. 2007. View Article : Google Scholar
|
|
22
|
Falsaperla M, Morgia G, Tartarone A,
Ardito R and Romano G: Support ellagic acid therapy in patients
with hormone refractory prostate cancer (HRPC) on standard
chemotherapy using vinorelbine and estramustine phosphate. Eur
Urol. 47:449–455. 2005. View Article : Google Scholar
|
|
23
|
Lansky EP, Harrison G, Froom P and Jiang
WG: Pomegranate (Punica granatum) pure chemicals show
possible synergistic inhibition of human PC-3 prostate cancer cell
invasion across Matrigel. Invest New Drugs. 23:121–122.
2005.PubMed/NCBI
|
|
24
|
Sartippour MR, Seeram NP, Rao JY, Moro A,
Harris DM, Henning SM, Firouzi A, Rettig MB, Aronson WJ, Pantuck AJ
and Heber D: Ellagitannin-rich pomegranate extract inhibits
angiogenesis in prostate cancer in vitro and in vivo.
Int J Oncol. 32:475–480. 2008.PubMed/NCBI
|
|
25
|
Umesalma S and Sudhandiran G: Ellagic acid
prevents rat colon carcinogenesis induced by 1,2 dimethyl hydrazine
through inhibition of AKT-phosphoinositide-3 kinase pathway. Eur J
Pharmacol. 660:249–258. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Vanella L, Barbagallo I, Acquaviva R, Di
Giacomo C, Cardile V, Abraham NG and Sorrenti V: Ellagic acid:
cytodifferentiating and antiproliferative effects in human
prostatic cancer cell lines. Curr Pharm Des. 19:2728–2736. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fisher ED: Apoptosis in cancer therapy:
crossing the threshold. Cell. 78:539–542. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hartwell HL and Kastan MB: Cell cycle
control and cancer. Science. 266:1821–1828. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Thompson CB: Apoptosis in the pathogenesis
and treatment of disease. Science. 267:1456–1462. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Steller H: Mechanisms and genes of
cellular suicide. Science. 267:1445–1449. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hunter T: Oncoprotein networks. Cell.
88:333–346. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Acquaviva R, Di Giacomo C, Sorrenti V,
Galvano F, Santangelo R, Cardile V, Gangia S, D'Orazio N, Abraham
NG and Vanella L: Antiproliferative effect of oleuropein in
prostate cell lines. Int J Oncol. 41:31–38. 2012.PubMed/NCBI
|
|
33
|
Bradford MM: A rapid and sensitive method
for the quantitation of microgram quantities of protein utilizing
the principle of protein-dye binding. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hong WK and Sporn MB: Recent advances in
chemoprevention of cancer. Science. 278:1073–1077. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Parnes HL, Thompson IM and Ford LG:
Prevention of hormone-related cancers: prostate cancer. J Clin
Oncol. 23:368–377. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mukhtar H and Ahmad N: Cancer
chemoprevention: future holds in multiple agents. Toxicol Appl
Pharmacol. 158:207–210. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Surh YJ: Cancer chemoprevention with
dietary phytochemicals. Nat Rev Cancer. 3:768–780. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Greenwald P: Lifestyle and medical
approaches to cancer prevention. Recent Results Cancer Res.
166:1–15. 2005. View Article : Google Scholar
|
|
39
|
Barker N and Clevers H: Catenins, Wnt
signaling and cancer. Bioessays. 22:961–965. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fearnhead NS, Britton MP and Bodmer WF:
The ABC of APC. Hum Mol Genet. 10:721–733. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Karim R, Tse G, Putti T, Scolyer R and Lee
S: The significance of the Wnt pathway in the pathology of human
cancers. Pathology. 36:120–128. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gwak J, Lee JH, Chung YH, Song GY and Oh
S: Small molecule-based promotion of PKCα-mediated β-catenin
degradation suppresses the proliferation of CRT-positive cancer
cells. PLoS One. 7:e466972012.PubMed/NCBI
|
|
43
|
Pratheeshkumar P, Budhraja A, Son YO, Wang
X, Zhang Z, Ding S, Wang L, Hitron A, Lee JC, Xu M, Chen G, Luo J
and Shi X: Quercetin inhibits angiogenesis mediated human prostate
tumor growth by targeting VEGFR-2 regulated AKT/mTOR/P70S6K
signaling pathways. PLoS One. 7:e475162012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Diersch S, Wenzel P, Szameitat M, Eser P,
Paul MC, Seidler B, Eser S, Messer M, Reichert M, Pagel P, Esposito
I, Schmid RM, Saur D and Schneider G: Efemp1 and p27Kip1
modulate responsiveness of pancreatic cancer cells towards a dual
PI3K/mTOR inhibitor in preclinical models. Oncotarget. 4:277–288.
2013.
|
|
45
|
Vinayak S and Carlson RW: mTOR inhibitors
in the treatment of breast cancer. Oncology. 27:38–44.
2013.PubMed/NCBI
|
|
46
|
Fasolo A and Sessa C: mTOR inhibitors in
the treatment of cancer. Expert Opin Investig Drugs. 17:1717–1734.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jo MJ, Kim HR and Kim GD: The anticancer
effects of Saccharina japonica on 267B1/K-ras human prostate
cancer cells. Int J Oncol. 41:1789–1797. 2012.
|
|
48
|
Adhami VM, Syed DN, Khan N and Mukhtar H:
Dietary flavonoid fisetin: a novel dual inhibitor of PI3K/Akt and
mTOR for prostate cancer management. Biochem Pharmacol.
84:1277–1281. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Fagone P, Donia M, Mangano K, Quattrocchi
C, Mammana S, Coco M, Libra M, McCubrey JA and Nicoletti F:
Comparative study of rapamycin and temsirolimus demonstrates
superimposable anti-tumour potency on prostate cancer cells. Basic
Clin Pharmacol Toxicol. 112:63–69. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Sundaresan NR, Pillai VB, Wolfgeher D,
Samant S, Vasudevan P, Parekh V, Raghuraman H, Cunningham JM, Gupta
M and Gupta MP: The deacetylase SIRT1 promotes membrane
localization and activation of Akt and PDK1 during tumorigenesis
and cardiac hypertrophy. Sci Signal. 4:ra462011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Chen HC, Jeng YM, Yuan RH, Hsu HC and Chen
YL: SIRT1 promotes tumorigenesis and resistance to chemotherapy in
hepatocellular carcinoma and its expression predicts poor
prognosis. Ann Surg Oncol. 19:2011–2019. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jung-Hynes B, Nihal M, Zhong W and Ahmad
N: Role of sirtuin histone deacetylase SIRT1 in prostate cancer. A
target for prostate cancer management via its inhibition? J Biol
Chem. 284:3823–3832. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Abdelmohsen K, Pullmann R Jr, Lal A, Kim
HH, Galban S, Yang X, Blethrow JD, Walker M, Shubert J, Gillespie
DA, Furneaux H and Gorospe M: Phosphorylation of HuR by Chk2
regulates SIRT1 expression. Mol Cell. 25:543–557. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Kuwano Y, Rabinovic A, Srikantan S,
Gorospe M and Demple B: Analysis of nitric oxide-stabilized mRNAs
in human fibroblasts reveals HuR-dependent heme oxygenase 1
upregulation. Mol Cell Biol. 29:2622–2635. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kruger AL, Peterson S, Turkseven S,
Kaminski PM, Zhang FF, Quan S, Wolin MS and Abraham NG: D-4F
induces heme oxygenase-1 and extracellular superoxide dismutase,
decreases endothelial cell sloughing, and improves vascular
reactivity in rat model of diabetes. Circulation. 111:3126–3134.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Miramar MD, Costantini P, Ravagnan L,
Saraiva LM, Haouzi D, Brothers G, Penninger JM, Peleato ML, Kroemer
G and Susin SA: NADH oxidase activity of mitochondrial
apoptosis-inducing factor. J Biol Chem. 276:16391–16398. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Klein JA, Longo-Guess CM, Rossmann MP,
Seburn KL, Hurd RE, Frankel WN, Bronson RT and Ackerman SL: The
harlequin mouse mutation downregulates apoptosis-inducing factor.
Nature. 419:367–374. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Otera H, Ohsakaya S, Nagaura Z, Ishihara N
and Mihara K: Export of mitochondrial AIF in response to
proapoptotic stimuli depends on processing at the intermembrane
space. EMBO J. 24:1375–1386. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Susin SA, Lorenzo HK, Zamzami N, Marzo I,
Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler
M, Larochette N, Goodlett DR, Aebersold R, Siderovski DP, Penninger
JM and Kroemer G: Molecular characterization of mitochondrial
apoptosis-inducing factor. Nature. 397:441–446. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Cregan SP, Fortin A, MacLaurin JG,
Callaghan SM, Cecconi F, Yu SW, Dawson TM, Dawson VL, Park DS,
Kroemer G and Slack RS: Apoptosis-inducing factor is involved in
the regulation of caspase-independent neuronal cell death. J Cell
Biol. 158:507–517. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ling YH, Liebes L, Zou Y and Perez-Soler
R: Reactive oxygen species generation and mitochondrial dysfunction
in the apoptotic response to Bortezomib, a novel proteasome
inhibitor, in human H460 non-small cell lung cancer cells. J Biol
Chem. 278:33714–33723. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Paradies G, Petrosillo G, Pistolese M and
Ruggiero FM: Reactive oxygen species affect mitochondrial electron
transport complex I activity through oxidative cardiolipin damage.
Gene. 286:135–141. 2002. View Article : Google Scholar
|
|
63
|
Malik A, Afaq S, Shahid M, Akhtar K and
Assiri A: Influence of ellagic acid on prostate cancer cell
proliferation: a caspase-dependent pathway. Asian Pac J Trop Med.
4:550–555. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Smith PC, Hobisch A, Lin DL, Culig Z and
Keller ET: Interleukin-6 and prostate cancer progression. Cytokine
Growth Factor Rev. 12:33–40. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Cavarretta IT, Neuwirt H, Untergasser G,
Moser PL, Zaki MH, Steiner H, Rumpold H, Fuchs D, Hobisch A, Nemeth
JA and Culig Z: The antiapoptotic effect of IL-6 autocrine loop in
a cellular model of advanced prostate cancer is mediated by Mcl-1.
Oncogene. 26:2822–2832. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Culig Z and Puhr M: Interleukin-6: a
multifunctional targetable cytokine in human prostate cancer. Mol
Cell Endocrinol. 360:52–58. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Yang F, Strand DW and Rowley DR:
Fibroblast growth factor-2 mediates transforming growth factor-β
action in prostate cancer reactive stroma. Oncogene. 27:450–459.
2008.
|