Introduction
Breast cancer is the most commonly diagnosed cancer
among women and is second only to lung cancer in terms of
cancer-related mortality (1).
Despite advances in breast cancer research and therapy, the
mortality rate remains high (2);
therefore, the development of new agents for breast cancer therapy
is clinically important.
Phytochemicals are a promising source for the
development of novel cancer therapeutics. Due to their potential
effectiveness and low toxicity profiles (3), phytochemicals generally have been
successful in clinical drug development to treat many diseases
(4,5). Hirsutine is one of the major alkaloids
found in plants of the Uncaria genus and is known for its
cardioprotective (6),
anti-hypertensive and anti-arrhythmic activities (7). Recently, we identified hirsutine as an
antimetastatic phytochemical by targeting NF-κB activation in a
murine breast cancer model. We demonstrated the significant
anti-metastatic activity of hirsutine both in vitro and
in vivo (8).
In the present study, we further explored the
clinical utility of hirsutine against breast cancer by testing the
effect of hirsutine against six human breast cancer cell lines.
Notably, hirsutine showed selective anticancer activity against
human breast cancer cell lines with HER2-expressing and p53-mutated
phenotypes. We further identified that the induction of DNA damage
response can be a primary target of hirsutine, leading to apoptotic
cell death of breast cancer cells.
Materials and methods
Reagents
Hirsutine and a Cell Counting kit were purchased
from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Muse
Annexin V and Dead Cell Assay kits were purchased from EMD
Millipore Corporation (Merck Millipore, Billerica, MA, USA). The
reagent for the Caspase-Glo3/7 Assay was obtained from Promega
(Madison, WI, USA). Irinotecan was purchased from Sigma-Aldrich
(St. Louis, MO, USA).
Cells
All cell lines used in the present study were
obtained from the American Type Culture Collection (ATCC). Human
breast cancer cells ZR-75-1 and BT474 were maintained in RPMI-1640
medium containing 10% bovine serum (Nissui, Tokyo, Japan). MCF-7,
MDA-MB-231, MDA-MB-468 and MDA-MB-453 cells were maintained in
Dulbecco’s modified Eagle’s medium (DMEM) containing 10% bovine
serum (Nissui). Cells were incubated at 37°C in a humidified
atmosphere of 95% air and 5% CO2.
Cell viability assay
For detection of the neoplastic activity of cells
grown in 96-well plates, the human breast cancer cells in
exponential growth were placed at a final concentration of
2×104 cells/well in a 96-well plate. After a 3-h
incubation, the cells were treated with hirsutine (6.25, 12.5, 25
and 50 μM) or with the vehicle (vehicle control, 0.5% DMSO) for 24
h. After treatment, 10 μl of WST-1 reagent was added. The
microplate was incubated for another 2 h in a humidified atmosphere
(37°C, 5% CO2) to allow the formation of formazan dye
and to get a higher sensitivity. The absorbance was measured in a
microplate reader at 450/620 nm. Cell viability was determined from
the absorbance of soluble formazan dye generated by the living
cells.
Detection of apoptosis
Cells were grown in 24-well plates and treated with
50 μM of hirsutine for 15 h before assaying using Muse™ Cell
Analyzer (Merck Millipore). Floating and adherent cells were
collected after the treatments. The cells were washed twice with
PBS and resuspended in PBS. The apoptosis profiling and apoptotic
cell counts were obtained with Muse Annexin V and Dead Cell Assay
kit. The assay was conducted in triplicate and in accordance with
the manufacturer’s instructions.
Measurement of caspase activity
For measurements of the activities of caspase-3 and
caspase-7, the Caspase-Glo® 3/7 Assay was carried out
according to the manufacturer’s instructions. This kit is based on
the cleavage of the amino acid sequence DEVD of a luminogenic
substrate by caspase-3 and caspase-7, which results in a
luminescent signal. Human breast cancer cells in exponential growth
were placed at a final concentration of 1×104 cells/well
in 90 μl in a white-walled multi-well plate. After 3 h of
incubation, the cells were treated with hirsutine (25 and 50 μM) or
with the vehicle (vehicle control, 0.5% DMSO) for another 3 h. Then
100 μl of Caspase-Glo® 3/7 reagent was added, and the
plates were incubated for another 30 min. Caspase-3 and -7
activities were recorded in a Glomax Multi-Detection system.
Western blot analysis
Cells were exposed to 50 μM hirsutine for 0, 1, 3
and 6 h. Treated cells were collected, washed with PBS, and lysed
in lysis buffer [25 mM HEPES (pH 7.7), 0.3 M NaCl, 1.5 mM
MgCl2, 0.2 mM EDTA, 0.1% Triton X-100, 20 mM
β-glycerophosphate, 0.1 mM sodium orthovanadate, 0.5 mM
phenylemethylsulfonyl fluoride (PMSF), 1 mM dithiothreitol, 10
mg/ml aprotinin, and 10 mg/ml leupeptin]. The cell lysates were
separated by 5–10% SDS-PAGE and transferred to PVDF membranes using
a glycine transfer buffer [192 mM glycine, 25 mM Tris-HCl (pH 8.8)
and 20% (v/v) methanol]. After blocking with Block Ace for 4 h at
room temperature, the membranes were incubated overnight with the
primary antibodies, and then for 60 min with the secondary
antibodies. Primary antibodies were used at a dilution of 1:1,000.
The secondary antibodies were used at a dilution of 1:2,000 and
visualized with an enhanced chemiluminescence system (Amersham
Biosciences, Amersham, UK).
The following antibodies from Cell Signaling
Technology (Danvers, MA, USA) were used for western blot analysis
according to the manufacturer’s recommendations: anti-phospho-Akt
(Ser473, #9271L), anti-phospho-p65 (Ser536, #3033S); anti-caspase-3
(#9662S), anti-phospho-p38 (p-p38) (Thr-180/Tyr-182, #4511),
anti-phospho-histone H2AX (Ser139, #9947). Anti-actin (sc-1615,
#G1312), anti-caspase-9 (sc-7885, #B0807), anti-ErbB2 (sc-284,
#L0706), anti-p65 (sc-109G, #J1003), anti-Akt1 (sc-1618, #131705)
and anti-p38α (sc-535, #K1010) were purchased from Santa Cruz
Biotechnology (Santa Cruz, CA, USA).
Statistical analysis
All the data are expressed as mean ± SD of at least
3 independent experiments and were analyzed for statistical
significance using the Student t-test. P-values <0.05 were
considered statistically significant.
Results
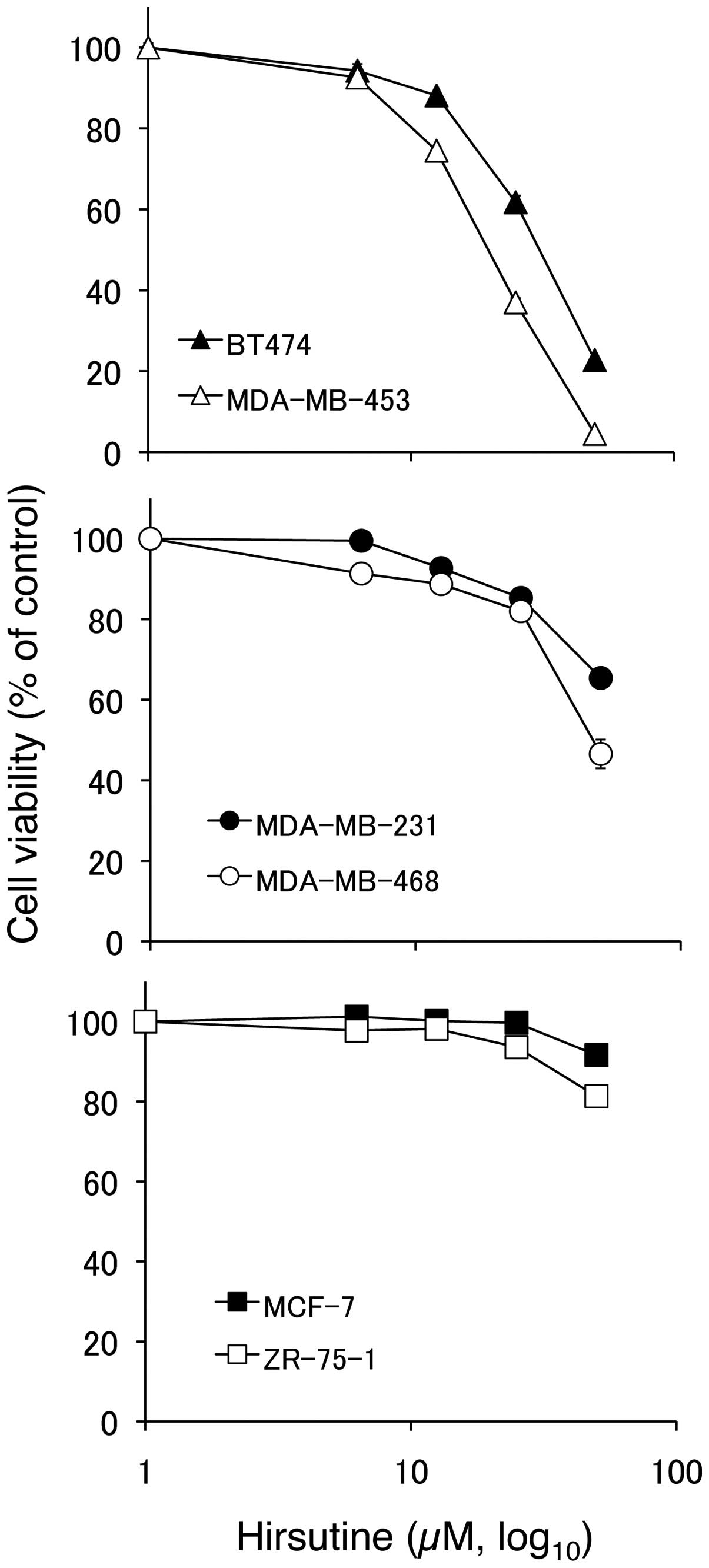
Selective cytotoxicity of hirsutine
against human breast cancer cells
We recently found hirsutine as an anti-metastatic
phytochemical by targeting NF-κB activation in a murine breast
cancer model. To further extend the utility of hirsutine, we
examined the anticancer effect of hirsutine against human breast
cancer cell lines with distinct molecular background. Among the 6
tested human breast cancer cell lines, hirsutine showed strong
cytotoxicity against the MDA-MB-453 and BT474 cell lines that have
HER2-positive and p53-mutated characteristics (Fig. 1, top panel). While triple-negative
breast cancer cell lines MDA-MB-231 and MDA-MB-468 showed a
moderate response (Fig. 1, middle
panel), HER2-negative and p53 wild-type MCF-7 and ZR-75-1 cells
showed significant resistance against hirsutine-induced
cytotoxicity (Fig. 1, bottom
panel).
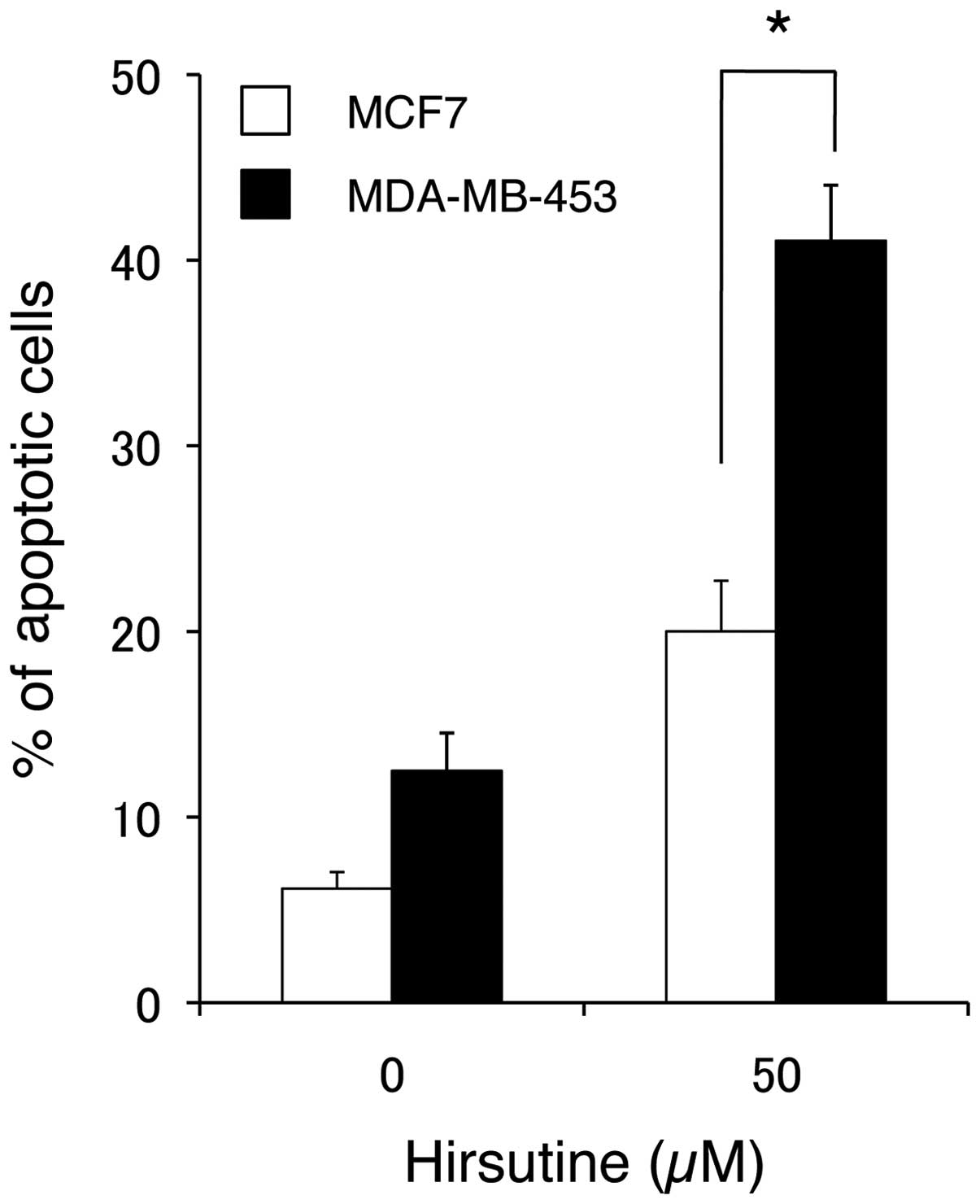
Induction of breast cancer cell apoptosis
by hirsutine
To examine whether the cytotoxic effect of hirsutine
is a result of the induction of apoptosis, both hirsutine sensitive
(MDA-MB-453) and resistant (MCF-7) cells were subjected to Annexin
V staining 15 h after treatment with hirsutine. In concert with the
hirsutine-induced cytotoxicity (Fig.
1), hirsutine significantly induced apoptosis in the MDA-MB-453
cells, but not in the MCF-7 cells (Fig.
2). Such induction of apoptosis by hirsutine was related to the
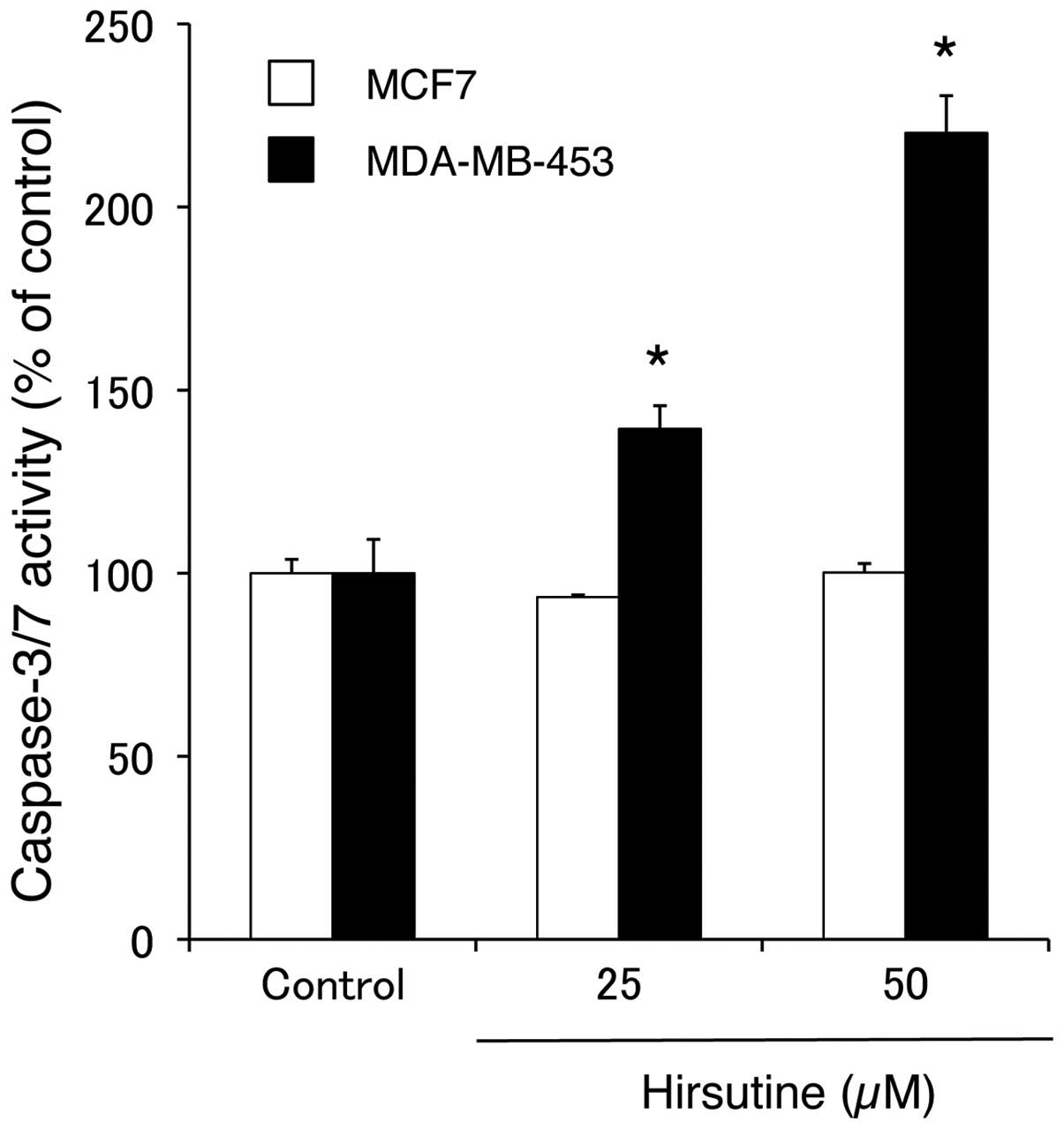
activation of caspases, as noted in the elevated caspase-3/7
activity in the MDA-MB-453 cells, but not in the MCF-7 cells, 3 h
after treatment with hirsutine (Fig.
3). This demonstrated that caspases played an important role in
hirsutine-induced apoptosis in the MDA-MB-453 cells. These results
clearly show that the induction of apoptotic cell death can be a
potential mechanism for the selective anticancer effect of
hirsutine against human breast cancer cells.
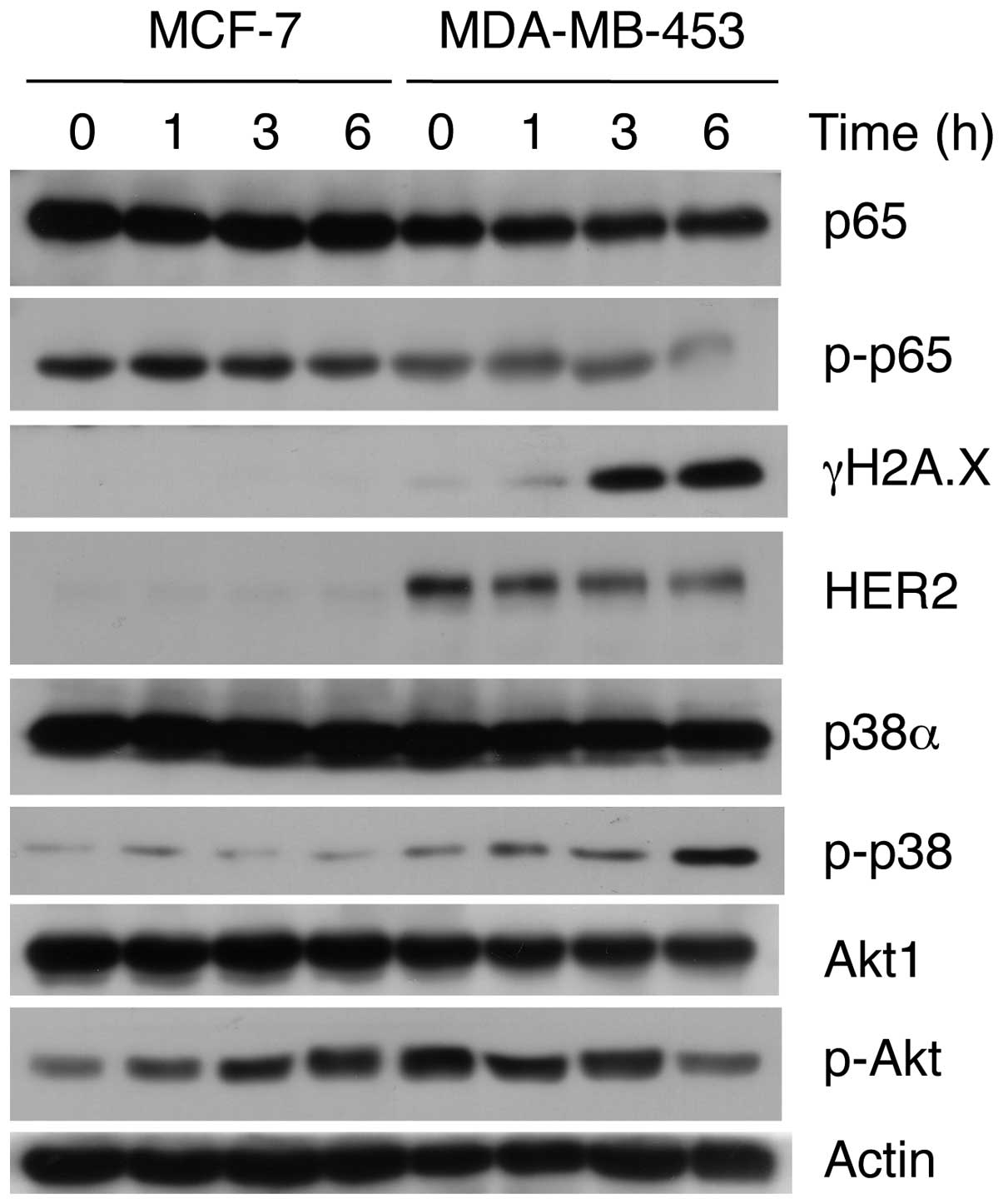
Hirsutine induces a DNA damage response
in breast cancer cells
To further explore the possible mechanism of
hirsutine to induce apoptosis in breast cancer cells, we assessed
its effect on the MAPK, HER2, NF-κB, and Akt signaling pathways, as
well as the DNA damage response. As shown in Fig. 4, hirsutine significantly suppressed
the HER2, NF-κB, and Akt signaling pathways in the MDA-MB-453
cells. Meanwhile, the activation of the p38 MAPK stress pathway and
a DNA damage response, as noted in the expression of p-p38 and
γH2AX, respectively, were observed upon hirsutine treatment in the
MDA-MB-453 cells. Conversely, hirsutine did not show any
significant effect on those molecular pathways or DNA damage
response in the MCF-7 cells. In order to determine the selectivity
of hirsutine to induce the DNA damage response in HER2-positive and
p53-mutant breast cancer cells, we used irinotecan as a typical DNA
damage-inducing agent to assess its effects on the MCF-7 and
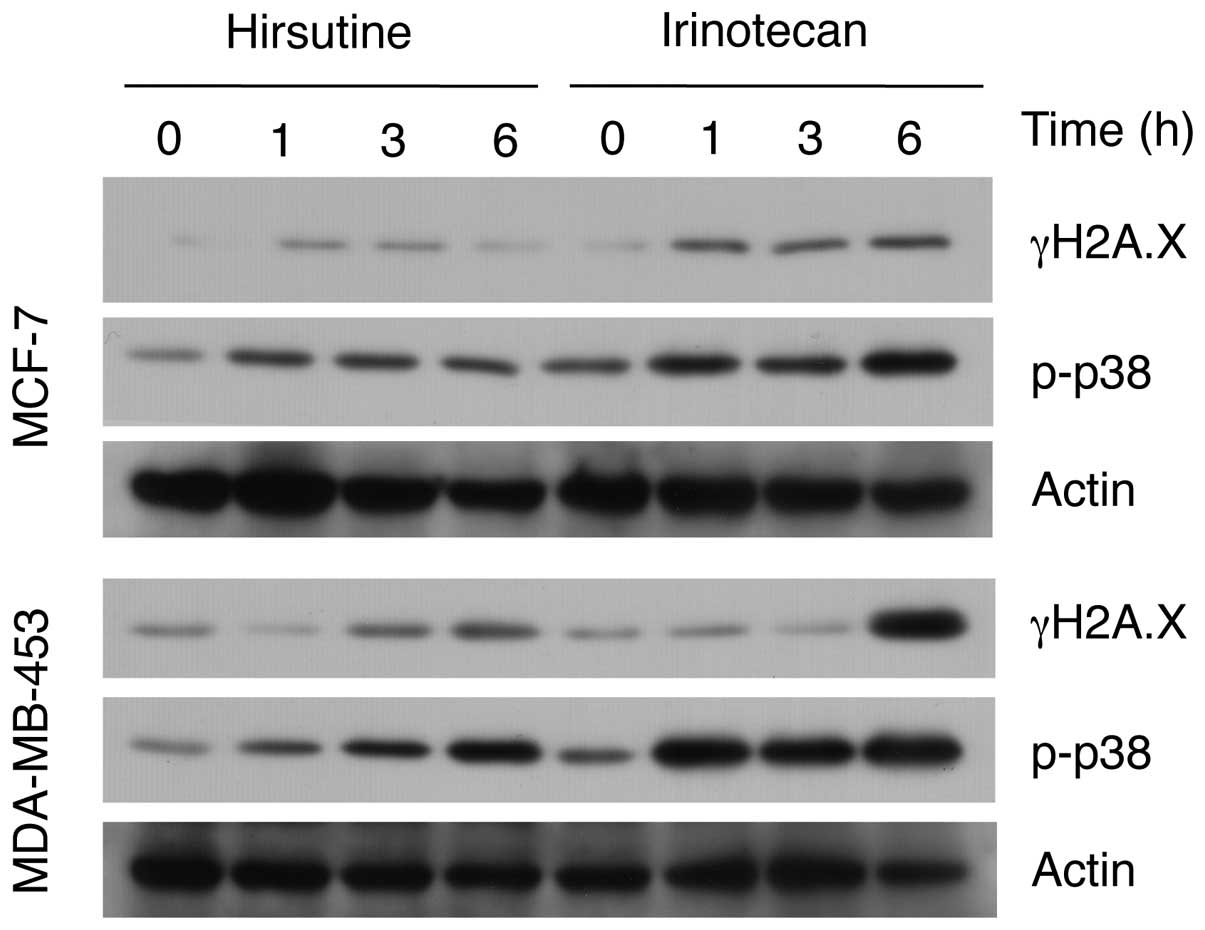
MDA-MB-453 cells. As shown in Fig.
5, irinotecan significantly upregulated γH2AX and p-p38
expression in both the MCF-7 and MDA-MB-453 cells, in contrast to
hirsutine. Collectively, these results suggest the selective
activity of hirsutine to induce DNA damage response in
HER2-positive and the p53-mutated human breast cancer cells.
Discussion
In the present study, we investigated the clinical
utility of hirsutine, an indol alkaloid phytochemical compound
present in plants of the Uncaria genus, as an anticancer
agent for human breast cancer cells. We found that the
HER2-positive/p53-mutated MDA-MB-453 and BT474 cell lines were
relatively sensitive to hirsutine-induced cytotoxicity among the 6
tested human breast cancer cell lines (Fig. 1). To exert its cytotoxic activity
against breast cancer cells, hirsutine induced apoptosis along with
the activation of caspases (Figs. 2
and 3). Importantly, the expression
of γH2AX, a hallmark of DNA damage response, was significantly
upregulated after treatment of hirsutine along with the p38/MAPK
stress signaling pathways (Figs. 4
and 5) implying that a DNA damage
response was involved in the hirsutine-induced apoptosis.
Apoptosis is a programmed cell death occurring in
multicellular organisms (9). In
general, drug-induced apoptosis is one major mechanism of action
for treating cancer, and various signaling pathways are involved in
the process (10). Among these, a
DNA damage response is one of the molecular events leading to
apoptosis, and indeed many anticancer agents induce a DNA damage
response (11–15). The mechanism involved in DNA
damage-induced apoptotic cell death is known to closely correlate
with the DNA repair response (16–18).
The phosphorylation of the Ser-139 residue of the histone variant
H2AX, forming γH2AX, is known as an early cellular response
associated with the induction of DNA double-strand breaks.
Therefore, induction of γH2AX has been considered as a hallmark of
a DNA damage response (19–22). We originally identified hirsutine to
inhibit NF-κB activation (8), and
the NF-κB pathway is known to play an important role in cancer
progression (23–30). The NF-κB pathway can be activated as
part of the DNA damage response by orchestrating cellular survival
pathways (31,32). Indeed, we observed the
downregulation of p-p65 in the MDA-MB-453 cells, but not in the
MCF-7 cells, in response to hirsutine treatment along with the
inhibition of Akt activity (Fig.
4), which has been widely recognized as an important
determinant for DNA damage-induced apoptosis (33,34)
and double-strand break repair (35–37).
Considering the upregulation of p-Akt in MCF-7 cells following
hirsutine treatment along with resistance to its cytotoxicity, the
NF-κB/Akt pathway may be involved in the selective resistance of
MCF-7 cells against hirsutine.
In addition to the NF-κB/Akt pathway, p38 MAPK
activity is also known to play an important role in the DNA damage
response induced by genotoxic stress with DNA-damaging
chemotherapeutic agents through the activation of p53 (38). Inhibition of the p38 MAPK pathway
reduced γH2AX expression (39),
diminished the apoptotic fraction of cells and increased cell
survival following exposure to chemotherapeutic agents (15,38,40),
thus suggesting a role for p38 activation in the apoptotic response
against genotoxic stress. Consistently, we observed the
upregulation of p-p38 upon hirsutine treatment in the MDA-MB-453
cells, but not in the MCF-7 cells. In the context of selective
cytotoxicity, hirsutine treatment downregulated the expression of
HER2 in the MDA-MB-453 cells (Fig.
4), which has been known to be involved in the repair response
of DNA damage induced by chemotherapeutic agents (41,42).
Importantly, we obtained similar results of the DNA damage response
in HER2-positive BT474 cells (data not shown). Furthermore, by
using the anticancer drug irinotecan (CPT-11), which is a
topoisomerase I inhibitor (43–47),
we did not observe a selective induction of DNA damage response
between the MDA-MB-453 and MCF-7 cells (Fig. 5). Considering the differential
status of HER2 expression and/or p53 mutation between the
MDA-MB-453 and MCF-7 cells (48–50),
HER2 expression and/or p53 status is presumably involved in
determining the cellular response to hirsutine treatment. Although
further study is clearly required to determine the exact mechanisms
of action by which hirsutine targets cancer cells, our present
study clearly demonstrated the selective anticancer activity of
hirsutine against HER2-positive breast cancer cells by inducing DNA
damage, implying its clinical utility as a novel anticancer
drug.
Acknowledgements
This research was supported by a Grant-in-Aid for
the 2012 and 2013 Cooperative Research Project I from the Institute
of Natural Medicine, University of Toyama. C.L. is a graduate
student supported by the Campus Asian Program of the University of
Toyama.
References
|
1
|
DeSantis C, Ma J, Bryan L and Jemal A:
Breast cancer statistics, 2013. CA Cancer J Clin. 64:52–62. 2014.
View Article : Google Scholar
|
|
2
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Crowell JA: The chemopreventive agent
development research program in the Division of Cancer Prevention
of the US National Cancer Institute: an overview. Eur J Cancer.
41:1889–1910. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cragg GM, Newman DJ and Snader KM: Natural
products in drug discovery and development. J Natl Prod. 60:52–60.
1997. View Article : Google Scholar
|
|
5
|
Harvey AL: Natural products in drug
discovery. Drug Discov Today. 13:894–901. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu LX, Gu XF, Zhu YC and Zhu YZ:
Protective effects of novel single compound, hirsutine on hypoxic
neonatal rat cardiomyocytes. Eur J Pharmacol. 650:290–297. 2011.
View Article : Google Scholar
|
|
7
|
Horie S, Yano S, Aimi N, Sakai S and
Watanabe K: Effects of hirsutine, an antihypertensive indole
alkaloid from Uncaria rhynchophylla, on intracellular calcium in
rat thoracic aorta. Life Sci. 50:491–498. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lou C, Takahashi K, Irimura T, Saiki I and
Hayakawa Y: Identification of hirsutine as an anti-metastatic
phytochemical by targeting NF-κB activation. Int J Oncol.
45:2085–2091. 2014.PubMed/NCBI
|
|
9
|
Elmore S: Apoptosis: a review of
programmed cell death. Toxicol Pathol. 35:495–516. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Saddoughi SA, Song P and Ogretmen B: Roles
of bioactive sphingolipids in cancer biology and therapeutics.
Subcell Biochem. 49:413–440. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhu H, Huang M, Yang F, et al: R16, a
novel amonafide analogue, induces apoptosis and G2-M arrest via
poisoning topoisomerase II. Mol Cancer Ther. 6:484–495. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cai Y, Lu J, Miao Z, Lin L and Ding J:
Reactive oxygen species contribute to cell killing and
P-glycoprotein downregulation by salvicine in multidrug resistant
K562/A02 cells. Cancer Biol Ther. 6:1794–1799. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cai B, Lyu H, Huang J, et al: Combination
of bendamustine and entinostat synergistically inhibits
proliferation of multiple myeloma cells via induction of apoptosis
and DNA damage response. Cancer Lett. 335:343–350. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kudoh T, Kimura J, Lu ZG, Miki Y and
Yoshida K: D4S234E, a novel p53-responsive gene, induces apoptosis
in response to DNA damage. Exp Cell Res. 316:2849–2858. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rudolf E, Kralova V, Rudolf K and John S:
The role of p38 in irinotecan-induced DNA damage and apoptosis of
colon cancer cells. Mutat Res. 741–742:27–34. 2013. View Article : Google Scholar
|
|
16
|
Norbury CJ and Zhivotovsky B: DNA
damage-induced apoptosis. Oncogene. 23:2797–2808. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ljungman M: The DNA damage response -
repair or despair? Environ Mol Mutagen. 51:879–889. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Roos WP and Kaina B: DNA damage-induced
cell death by apoptosis. Trends Mol Med. 12:440–450. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Mah LJ, El-Osta A and Karagiannis TC:
gammaH2AX: a sensitive molecular marker of DNA damage and repair.
Leukemia. 24:679–686. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Valdiglesias V, Giunta S, Fenech M, Neri M
and Bonassi S: γH2AX as a marker of DNA double strand breaks and
genomic instability in human population studies. Mutat Res.
753:24–40. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Redon CE, Nakamura AJ, Zhang YW, et al:
Histone gammaH2AX and poly(ADP-ribose) as clinical pharmacodynamic
biomarkers. Clin Cancer Res. 16:4532–4542. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ivashkevich AN, Martin OA, Smith AJ, et
al: γH2AX foci as a measure of DNA damage: a computational approach
to automatic analysis. Mutat Res. 711:49–60. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xiao G and Fu J: NF-κB and cancer: a
paradigm of Yin-Yang. Am J Cancer Res. 1:192–221. 2011.
|
|
24
|
Basseres DS and Baldwin AS: Nuclear
factor-kappaB and inhibitor of kappaB kinase pathways in oncogenic
initiation and progression. Oncogene. 25:6817–6830. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bours V, Bonizzi G, Bentires-Alj M, et al:
NF-kappaB activation in response to toxical and therapeutical
agents: role in inflammation and cancer treatment. Toxicology.
153:27–38. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huang CY, Fong YC, Lee CY, et al: CCL5
increases lung cancer migration via PI3K, Akt and NF-kappaB
pathways. Biochem Pharmacol. 77:794–803. 2009. View Article : Google Scholar
|
|
27
|
Nottingham LK, Yan CH, Yang X, et al:
Aberrant IKKα and IKKβ cooperatively activate NF-κB and induce
EGFR/AP1 signaling to promote survival and migration of head and
neck cancer. Oncogene. 33:1135–1147. 2014. View Article : Google Scholar :
|
|
28
|
Zhang W, Tan W, Wu X, et al: A NIK-IKKα
module expands ErbB2-induced tumor-initiating cells by stimulating
nuclear export of p27/Kip1. Cancer Cell. 23:647–659. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ryan KM, Ernst MK, Rice NR and Vousden KH:
Role of NF-kappaB in p53-mediated programmed cell death. Nature.
404:892–897. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kaltschmidt B, Kaltschmidt C, Hofmann TG,
Hehner SP, Droge W and Schmitz ML: The pro- or anti-apoptotic
function of NF-kappaB is determined by the nature of the apoptotic
stimulus. Eur J Biochem. 267:3828–3835. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Janssens S and Tschopp J: Signals from
within: the DNA-damage-induced NF-kappaB response. Cell Death
Differ. 13:773–784. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Volcic M, Karl S, Baumann B, et al: NF-κB
regulates DNA double-strand break repair in conjunction with
BRCA1-CtIP complexes. Nucleic Acids Res. 40:181–195. 2012.
View Article : Google Scholar :
|
|
33
|
Kandel ES, Skeen J, Majewski N, et al:
Activation of Akt/protein kinase B overcomes a G(2)/m cell cycle
checkpoint induced by DNA damage. Mol Cell Biol. 22:7831–7841.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Henry MK, Lynch JT, Eapen AK and Quelle
FW: DNA damage-induced cell-cycle arrest of hematopoietic cells is
overridden by activation of the PI-3 kinase/Akt signaling pathway.
Blood. 98:834–841. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Toulany M, Kasten-Pisula U, Brammer I, et
al: Blockage of epidermal growth factor
receptor-phosphatidylinositol 3-kinase-AKT signaling increases
radiosensitivity of K-RAS mutated human tumor cells in vitro by
affecting DNA repair. Clin Cancer Res. 12:4119–4126. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Toulany M, Kehlbach R, Florczak U, et al:
Targeting of AKT1 enhances radiation toxicity of human tumor cells
by inhibiting DNA-PKcs-dependent DNA double-strand break repair.
Mol Cancer Ther. 7:1772–1781. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Toulany M, Kehlbach R, Rodermann HP and
Mozdarani H: Radiocontrast media affect radiation-induced DNA
damage repair in vitro and in vivo by affecting Akt signalling.
Radiother Oncol. 94:110–116. 2010. View Article : Google Scholar
|
|
38
|
Sanchez-Prieto R, Rojas JM, Taya Y and
Gutkind JS: A role for the p38 mitogen-activated protein kinase
pathway in the transcriptional activation of p53 on genotoxic
stress by chemotherapeutic agents. Cancer Res. 60:2464–2472.
2000.PubMed/NCBI
|
|
39
|
Lu C, Shi Y, Wang Z, et al: Serum
starvation induces H2AX phosphorylation to regulate apoptosis via
p38 MAPK pathway. FEBS Lett. 582:2703–2708. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Bulavin DV, Saito S, Hollander MC, et al:
Phosphorylation of human p53 by p38 kinase coordinates N-terminal
phosphorylation and apoptosis in response to UV radiation. EMBO J.
18:6845–6854. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Madson JG, Lynch DT, Svoboda J, et al:
Erbb2 suppresses DNA damage-induced checkpoint activation and
UV-induced mouse skin tumorigenesis. Am J Pathol. 174:2357–2366.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Boone JJ, Bhosle J, Tilby MJ, Hartley JA
and Hochhauser D: Involvement of the HER2 pathway in repair of DNA
damage produced by chemotherapeutic agents. Mol Cancer Ther.
8:3015–3023. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wasserman E, Sutherland W and Cvitkovic E:
Irinotecan plus oxaliplatin: a promising combination for advanced
colorectal cancer. Clin Colorectal Cancer. 1:149–153. 2001.
View Article : Google Scholar
|
|
44
|
Ikeda M, Kurebayashi J, Sonoo H, et al:
Evaluation of irinotecan hydrochloride (CPT-11) and trastuzumab
combination therapy as salvage treatment in patients with HER2
overexpressing metastatic breast cancer. Gan To Kagaku Ryoho.
36:773–777. 2009.(In Japanese). PubMed/NCBI
|
|
45
|
Anders C, Deal AM, Abramson V, et al:
TBCRC 018: phase II study of iniparib in combination with
irinotecan to treat progressive triple negative breast cancer brain
metastases. Breast Cancer Res Treat. 146:557–566. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen AY and Liu LF: DNA topoisomerases:
essential enzymes and lethal targets. Annu Rev Pharmacol Toxicol.
34:191–218. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Pommier Y: Eukaryotic DNA topoisomerase I:
genome gatekeeper and its intruders, camptothecins. Semin Oncol.
23:3–10. 1996.PubMed/NCBI
|
|
48
|
Neve RM, Chin K, Fridlyand J, et al: A
collection of breast cancer cell lines for the study of
functionally distinct cancer subtypes. Cancer Cell. 10:515–527.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Holliday DL and Speirs V: Choosing the
right cell line for breast cancer research. Breast Cancer Res.
13:2152011. View
Article : Google Scholar : PubMed/NCBI
|
|
50
|
Hermanto U, Zong CS and Wang LH:
ErbB2-overexpressing human mammary carcinoma cells display an
increased requirement for the phosphatidylinositol 3-kinase
signaling pathway in anchorage-independent growth. Oncogene.
20:7551–7562. 2001. View Article : Google Scholar : PubMed/NCBI
|