Introduction
Green tea is a popular beverage consumed worldwide,
which is obtained from the dried leaves of the plant Camellia
sinensis (CS). Accumulating evidence has shown that green tea
is beneficial to health. It was demonstrated that green tea
polyphenols were able to act as direct antioxidants by scavenging
reactive oxygen species or chelating transition metals (1). Clinical studies showed that green tea
consumption was inversely associated with cardiovascular disease
mortality (2). Epidemiological
studies have shown that green tea extract induced thermogenesis and
resulted in weight loss (3). A
number of studies report the antitumor effects of CS and its
polyphenolic components. The CS extract or tea polyphenol induced
apoptosis and resulted in significant inhibition of tumor growth in
various types of cancer including leukemia (4), prostate (5) and breast cancer (6). Recently, tea polyphenol EGCG from CS
was used as adjuvant cancer therapy in combination with other
chemotherapies (7). EGCG-induced
chemosensitization of cancer cells through additive or synergistic
effects with anticancer drugs has been identified in a number of
preclinical, in vitro and in vivo studies (7). The effects of drugs such as
5-fluorouracil (8), doxorubicin
(9) or tamoxifen (10) have been shown to be significantly
increased when combined with EGCG in various types of cancer. In a
previous study, we demonstrated that CS aqueous extract (mimic tea
beverage) significantly inhibited 4T1 cell proliferation by
inducing apoptosis and caspase-3, -8 and PARP activation, and
increasing the expression of Bax-to-Bcl-2 ratio (11). CS also inhibited 4T1 cell migration
and invasion dose dependently. In animal studies, the CS extract
(0.6 g/kg, orally-fed daily for 4 weeks) was effective in
decreasing tumor weight, as well as lung and liver metastasis in
mice bearing 4T1 tumors. Furthermore, micro-computed tomography
(µ-CT) and in vitro osteoclast staining analysis
demonstrated that CS extract was effective in bone protection
against breast cancer-induced bone destruction (11). However, to the best of our
knowledge, few studies have investigated the herb-drug interaction
between CS extract and conventional anticancer agents.
Zoledronate (ZOL), the potent third-generation
nitrogen-containing bisphosphonate, is effective in the prevention
and treatment of bone destruction caused by the metastatic spread
of primary cancer to the skeleton (12). ZOL exerted potential direct and
indirect antitumor effects on in vitro and in vivo
models. ZOL dose-dependently inhibited the proliferation of breast
and prostate cancer, and osteosarcoma cells in vitro and
induced apoptosis in these tumor cells (13,14).
Additionally, ZOL induced tumor cell adhesion, invasion and
angiogenesis activities (15).
Recent findings have shown that ZOL administered in a metronomic
(repeated low-dose) manner appeared to be more effective than the
conventional regimen in breast cancer patients as shown by the
reduction of biomarkers (16,17).
Metronomic administration of ZOL also exhibited greater antitumor
effects in a mouse model of breast cancer (18). Furthermore, Facchini et al
demonstrated that the metronomic administration of ZOL and taxotere
combination in castration-resistant prostate cancer patients showed
promising antitumor activity (19).
In a previous study, we demonstrated that metronomic ZOL [0.0125
mg/kg intraperitoneally (i.p.) injected twice a week for 4 weeks]
was more effective than the conventional regimen (0.1 mg/kg i.p.
injected once only) in reducing breast cancer tumor burden, and
decreasing lung and liver metastasis in primary and metastatic
breast cancer (20).
The present study aimed to investigate the
antitumor, anti-metastatic and anti-osteolytic effects of the
combined use of CS aqueous extract and metronomic ZOL. The
metronomic dose of ZOL used in the present study was 0.0125 mg/kg,
i.p. injected 8 times in 4 weeks, and the total amount was
identical to that of the single conventional treatment of 0.1
mg/kg, although the metronomic method decreased the daily dose and
increased the frequency. The 4T1 mouse mammary tumor model was
employed to evaluate the antitumor, anti-metastatic and
anti-osteolytic effects of the combination of CS and metronomic
ZOL, together with the individual treatment of CS or metronomic
ZOL. Furthermore, the antiproliferative and apoptotic induction
effects as well as the anti-migration and anti-invasion abilities
of the combined use of CS and ZOL on mouse breast cancer 4T1 cells
were assessed in vitro. Gelatin zymography analysis was
employed to determine the underlying mechanism of the
antimetastatic activities of CS and ZOL.
Materials and methods
Preparation of CS aqueous extract and
high-performance liquid chromatography (HPLC) analysis
The dried leaves of green tea CS, whose origin is in
the Hainan Province, China, were purchased from the herbal supplier
of Hong Kong. A voucher specimen (no. 2011-3336) was kept in the
museum of the Institute of Chinese Medicine, The Chinese university
of Hong Kong. One kilogram of dried leaves of CS was soaked in one
liter of boiled water for 15 min twice. Following the filtration,
the water extracts were combined and evaporated under reduced
pressure at 60°C to dryness.
The chemical composition of the CS water extract was
analyzed by HPLC (Agilent, Santa Clara, CA, USA) as previously
described (21). Briefly,
two-gradient elution system including mobile phase A [85%
orthophosphoric acid and water (0.05:99.95, v:v)] and mobile phase
B (acetonitrile) was employed. The gradient was running as: 0–4
min, 2% B; 4–21 min, linear gradient from 2 to 9% B; 21–32 min,
linear gradient from 9 to 23% B; 32–45 min, 23% B. The
concentration of catechins in the CS extract was determined by
HPLC.
Cells and reagents
Mouse mammary tumor cells (4T1) were obtained from
the American Type Culture Collection (ATCC; rockville, MD, usa),
and cultured in rPMI-1640 medium containing 10% (v/v) fetal bovine
serum (FBS) and 1% (v/v) penicillin-streptomycin (Life
Technologies, Grand Island, NY, usa) at 37°C in 5% CO2
humidified incubator.
ZOL was purchased from Novartis Pharma Stein,
Switzerland. Transwell plates for Transwell migration assay were
obtained from Corning Incorporated Life Sciences (Tewksbury, Ma,
usa). Alanine transaminase (ALT), aspartate transaminase (AST) and
alkaline phosphatase (ALP) kits were purchased from Stanbio
(Boerne, TX, USA). The
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
was obtained from sigma (st. Louis, MO, USA). The caspase-3
activity assay kit was purchased from Life Technologies. The TUNEL
assay kit (in situ cell death POD kit) was obtained from
roche, Mannheim, Germany. The Annexin V/PI double staining kit was
purchased from BD Pharmingen (San Jose, CA, USA).
4T1 mouse mammary tumor model
The animal experiments were approved by the Animal
Experimentation Ethics Committee of The Chinese University of Hong
Kong (ref. no. 12/042/MIs). Female Balb/c mice (6–8 weeks old) were
supplied by the Laboratory Animal Services Centre, The Chinese
University of Hong Kong. The 4T1 cells (4×105) were
resuspended in 100 µl phosphate-buffered saline (PBS) and
subcutaneously (s.c.) inoculated at the mammary fat pad of each
mouse. Treatments were initiated 1 week after cancer cell
implantation and lasted for 4 weeks. After 4T1 cell implantation,
the tumor-bearing mice were randomly assigned into 4 groups (n=10):
untreated control (orally fed with distilled water everyday); CS
(0.6 g/kg CS extract, orally fed everyday); ZOL (0.0125 mg/kg ZOL,
i.p. injected twice a week); CS+ZOL (0.6 g/kg CS, orally fed
everyday + 0.0125 mg/kg ZOL, i.p. injected twice a week). Naive
mice without tumor and treatment were set as the normal standard.
The body weight of each mouse was measured once a week during the
treatment period. At day 28, the mice were sacrificed, and tumors,
lungs and livers were removed for quantification of the tumor
weight and burden. Tibias of mice from different groups were
removed for µ-CT analysis.
Hemato-biochemical markers were assessed by
measuring the activities of liver-specific (ALT and AST) and
bone-related (ALP) enzymes in the plasma using assay kits, after
mice were treated with CS and/or ZOL. The average absorbance/minute
was assessed, and then multiplied by a factor for results in
units/liter (U/L).
TUNEL assay
After mice were sacrificed, the tumors were removed
and fixed in 10% formalin for 2 weeks at room temperature, embedded
in paraffin, and then sectioned at a thickness of 5-µm. The
level of cell apoptosis was determined with a Roche In Situ
Cell Death POD kit according to the manufacturer’s protocols. After
de-waxation and rehydration, the tumor section was pretreated with
proteinase K and 3% H2O2 solution. The
stained tumor sections were photographed using an Olympus IX71
microscope (Japan) and analyzed using software ImageJ. Five fields
of tumor sections (magnification, x100) were randomly selected, and
the area of apoptotic cells in which nuclei stained as brown
particles was calculated in each field. The apoptotic index (AI %)
was calculated as: Area of apoptotic cells/area of total tumor
cells (22).
Histological analysis
Lungs and livers of tumor-bearing mice were fixed in
10% buffered formalin for 7 days at room temperature. The samples
were paraffin embedded, sectioned longitudinally at 5 µm,
and stained with hematoxylin and eosin (H&E). The stained
sections were examined and photographed using an Olympus IX71
microscope and SPOT advanced software (version 3.5.6). Five fields
of lung or liver sections were randomly selected, and the area with
4T1 tumors was calculated in each field. Tumor burden, defined as
tumor area divided by the lung or liver area, was measured using
ImageJ and expressed as an average percentage of the tumor area to
lung or liver area.
µ-CT analysis
Tibias removed from tumor-bearing mice were scanned
with a high resolution microtomographic system, µ-CT 40
(Scanco Medical AG, Switzerland). Each three-dimensional image data
comprised an ~500 micro-CT slide image (8 µm/slide) starting
from the growth plate of tibial interface and moving down the
tibia. Bone volume (BV) (mm3) was generated from
µ-CT analysis and compared with the control tibia for each
animal (18).
Cell viability assay
Cells (1×104/well) in 100 µl
culture medium were seeded in 96-well flat-bottomed plates
(Corning) and incubated with CS (0, 0.025, 0.05, 0.1 and 0.2 mg/ml)
and ZOL (0, 10, 20 and 30 µM) for 48 h. Following
incubation, the cells were subjected to MTT assay (20). Results were expressed as percentage
of cell viability with respect to the untreated control cells (as
100%). Only the most effective dose of ZOL was selected in
subsequent studies and combined with CS at various concentrations
(0, 0.05 and 0.1 mg/ml).
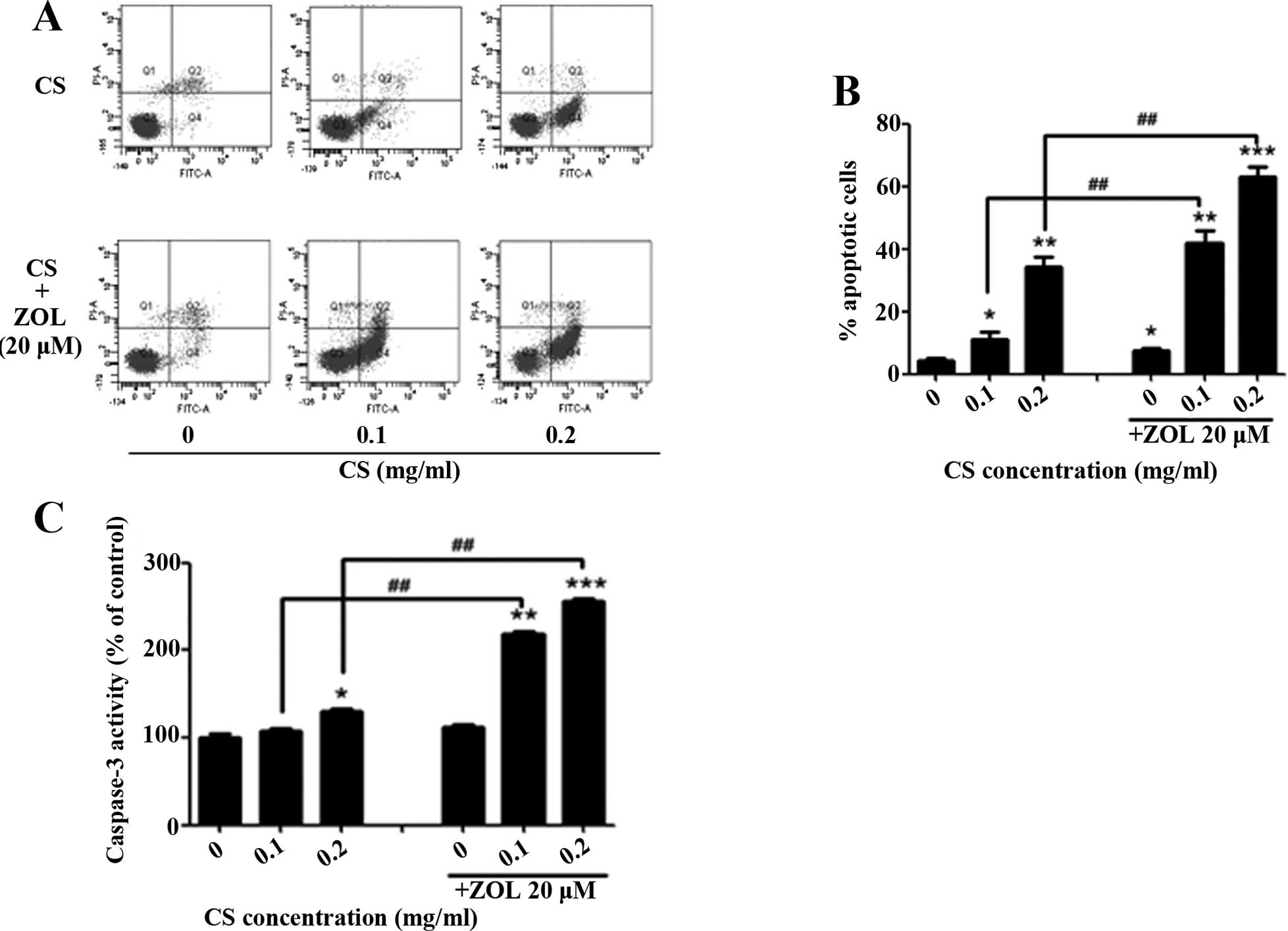
Annexin V-FITC/PI double staining
The 4T1 cells (3×105) in 1 ml culture
medium were seeded in 6-well plates and incubated at 37°C for 24 h.
The cells were then treated with the combined use of CS (0, 0.05
and 0.1 mg/ml) and ZOL (20 µM) for 24 h. After incubation,
the 4T1 cells were collected and washed with PBS. Then the cells
were stained in binding buffer containing Annexin V-FITC and PI for
15 min in the dark at room temperature. Subsequently, fluorescent
signaling in the cells was detected by flow cytometry (FACSCanto
II; Becton-Dickinson). Cell debris, characterized by a low forward
scatter/side scatter, was excluded from the analysis. Positioning
of quadrants on Annexin V/PI plots was performed to distinguish
living cells (FITC−/PI−), early apoptotic
cells (FITC+/PI−) and late apoptotic or
necrotic cells (FITC+/PI+). The separation of
the quadrants was adjusted by gating >95% cells of negative
control (i.e., 0 mg/ml) into the left lower quadrant.
Caspase-3 activity assay
The 4T1 cells (3×105) in 1 ml culture
medium were seeded in 6-well plates and incubated at 37°C for 24 h.
The cells were treated with the combination of CS (0, 0.05 and 0.1
mg/ml) and ZOL (20 µM) for 24 h, and subsequently collected
and lysed in lysis buffer to determine the caspase-3 activity,
using the caspase-3 activity assay kit (Invitrogen) according to
the manufacturer’s instructions. The plate was then read at 405 nm
in a microplate reader (µQuant; BioTek, Winooski, VT, USA).
An increase of caspase-3 activity was determined by direct
comparison to the level of the untreated control.
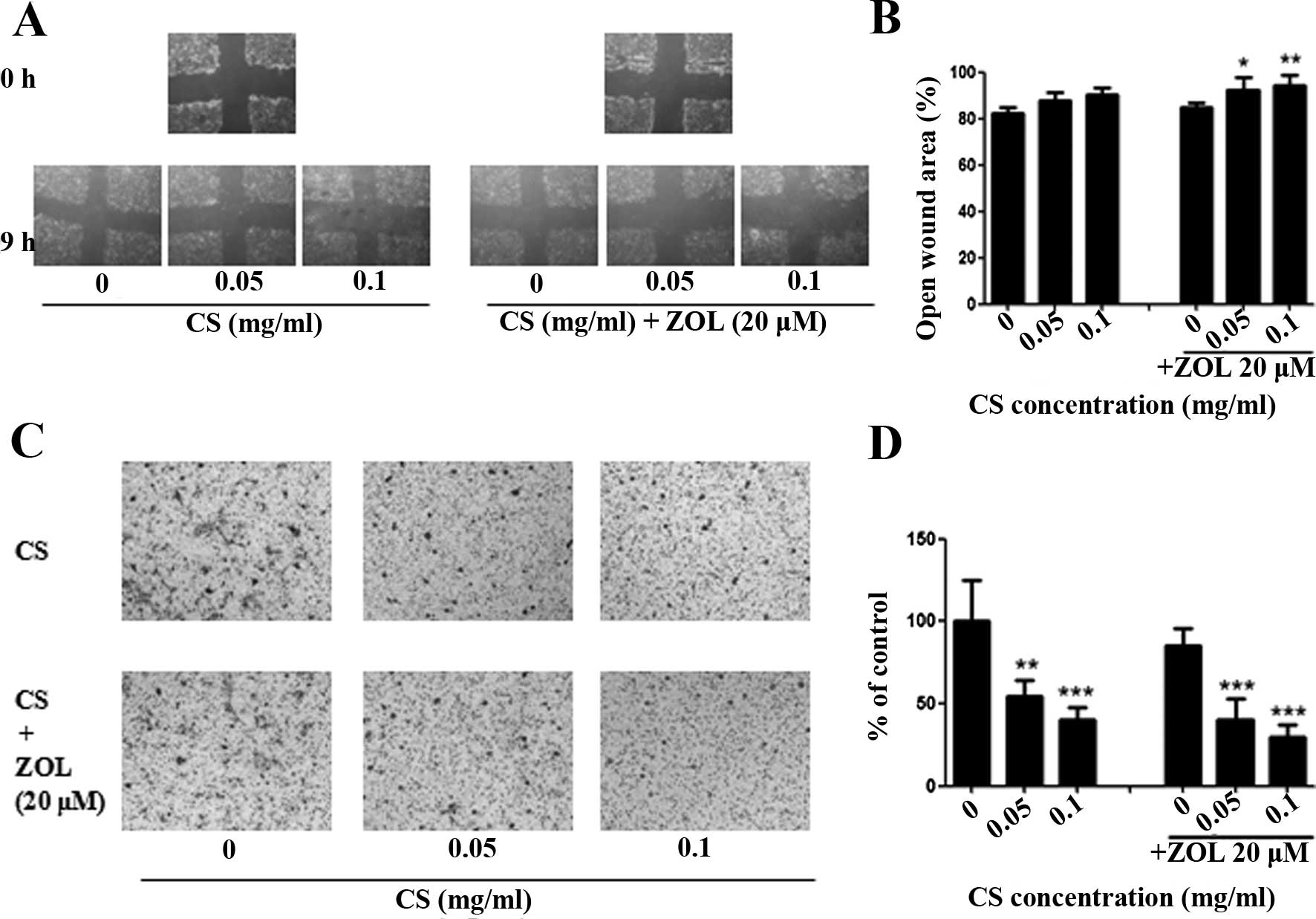
Scratch wound-healing assay
The 4T1 cells (1×105/well) in 1 ml
culture medium were seeded in 24-well plates and incubated at 37°C
for 24 h. After being starved in medium without FBS for 24 h, the
cells were scraped using 200 µl pipette tips and the scratch
was shaped like a cross. The medium was then replaced with fresh
medium with CS (0, 0.05 and 0.1 mg/ml) and ZOL (20 µM). The
cells were incubated for 9 h, and each well was photographed under
a microscope (Nikon Eclipse TS100). The percentages of open wound
area were measured and calculated using the TScratch software
(23). Motility was determined by
the decrease in the open wound area.
Transwell migration assay
The 4T1 cells (5×104 in 100 µl)
were added into each Transwell filter chamber with 1% v/v FBS. At
the same time, 100 µl of medium containing CS (0,0.05 and
0.1 mg/ml) and ZOL (20 µM) (with 1% v/v FBS) was added to
the upper Transwell chamber. Then, 500 µl complete medium
(with 10% v/v FBS), served as chemoattractant medium, was added
into the lower chamber. The cells were allowed to migrate through
the Boyden chamber membrane to the lower chamber for 6 h at 37°C,
under 5% CO2 conditions. After incubation, the cells
were fixed with methanol, stained with hematoxylin and images were
captured under a microscope (Nikon Eclipse TS100). The non-migrated
cells on the top surface of the filter membrane were scraped with
cotton swab. Stained filters were then photographed under
microscope (Nikon Eclipse TS100). The migrated cells were
quantified by manual counting in a blinded manner (24). Changes in cell numbers were
presented as a percentage of control values (as 100%).
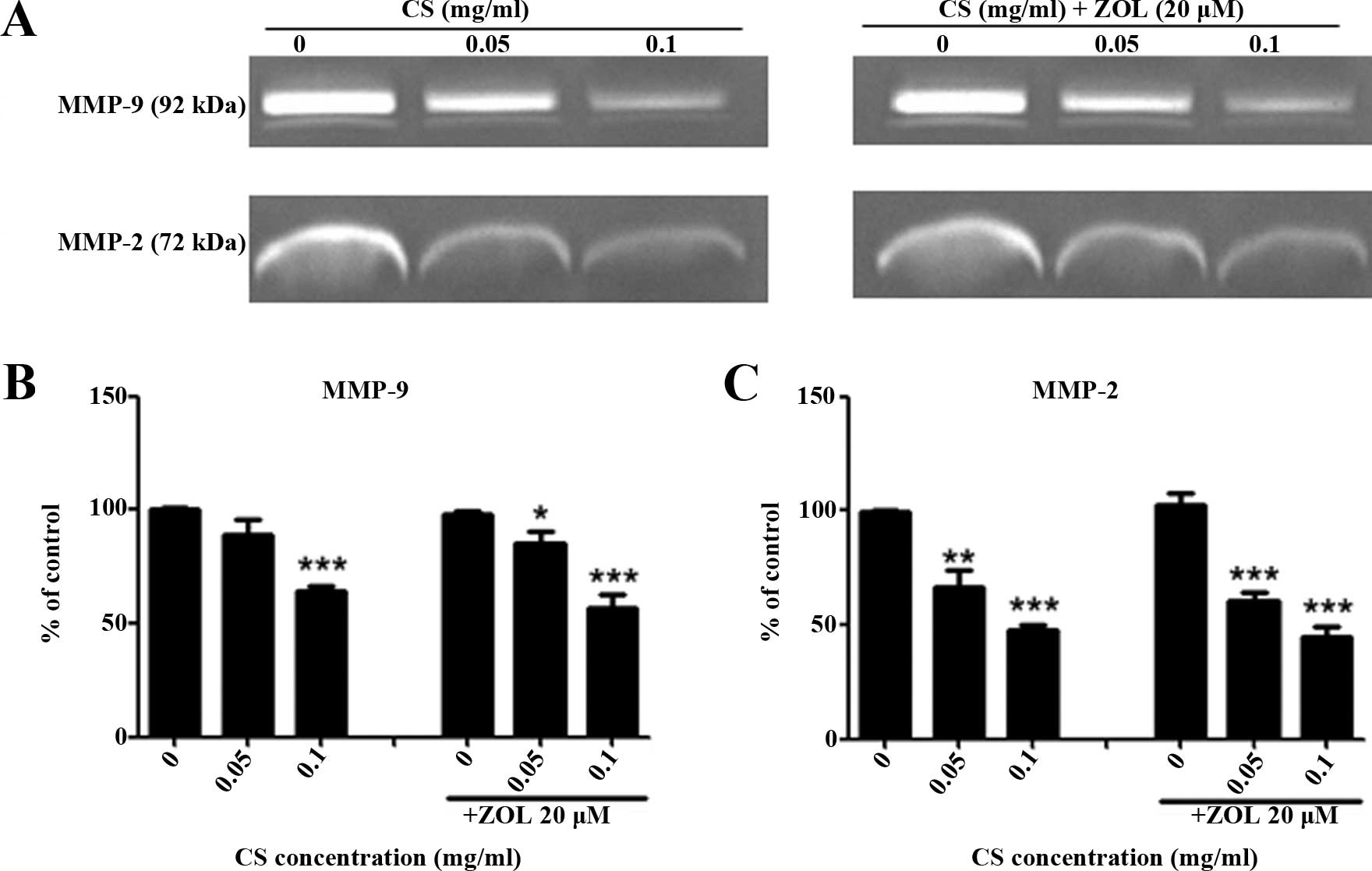
Gelatin zymography
Cells (1×105/well) in 1 ml culture medium
were seeded in 24-well plates and incubated with various
concentrations of CS (0, 0.05 and 0.1 mg/ml) at 37°C for 24 h. The
supernatant was collected and stored at −80°C. Protein sample (20
µg) from the supernatant was fractionated in 10%
SDS-polyacrylamide gel with 0.1% gelatin substrate. Following
electrophoresis, the gels were washed three times in 2.5% Triton
X-100 in PBS for 30 min at room temperature. The gels were then
incubated overnight at room temperature in developing buffer (50 mM
Tris base, 200 mM naCl, 0.005 mM ZnCl2, 5 mM
CaCl2·2H2O and 0.02% NaN3, pH 7.5)
and then stained with 0.125% (w/v) Coomassie brilliant blue for
20–30 min and destained in destaining buffer (10% acetic acid and
5% ethanol in distilled water) for 1–2 days. Visualization of bands
was performed on a Bio-Rad, XBS+ Imaging System (Bio-Rad, Hercules,
CA, USA).
Statistical analysis
Data were presented as mean ± SD (for in
vitro studies) or sEM (for in vivo studies). Statistical
analysis was performed using one-way ANOVA. P<0.05 was
considered to indicate a statistically significant result.
Results
HPLC analysis of polyphenols of CS
aqueous extract
The chemical composition of tea polyphenols in CS
extract was analyzed by HPLC. The CS water extract contained large
amount of tea polyphenol of ~25%, with EGCG as the most abundant,
followed by EGC, ECG and EC. Detailed information is provided in a
previous study (11).
Effect of combination therapy on 4T1
tumor growth and apoptotic induction
To investigate the activities of the combined use of
CS plus metronomic ZOL on tumor growth and metastasis, a mammary
fat pad model was employed, in which cells were injected into the
mammary fat pad of female BalB/c mice. The results showed that no
significant body weight loss was found in any of the treatment
groups (data not shown). Tumors from different groups shown in
Fig. 1A, reveal that 4T1 mammary
tumors were decreased significantly in the CS and CS+ZOL groups by
36.2 and 45.1%, respectively, as compared to the untreated control
(Fig. 1B). The combination of
CS+ZOL showed the lowest tumor weight among the three groups, and a
significant difference was observed between the CS+ZOL and ZOL
monotherapy groups (Fig. 1B). In
addition, the apoptotic index (%) in tumor, defined as the
percentage of apoptotic cell area to the total tumor area, was
assessed using TUNEL assay. As shown in Fig. 1C and D, treatment of CS, ZOL and
CS+ZOL resulted in an increase in tumor cell death in 4T1 tumors.
The combination of CS+ZOL showed a higher apoptotic index against
the individual treatment of CS or ZOL alone and significant
difference was identified (p<0.05).
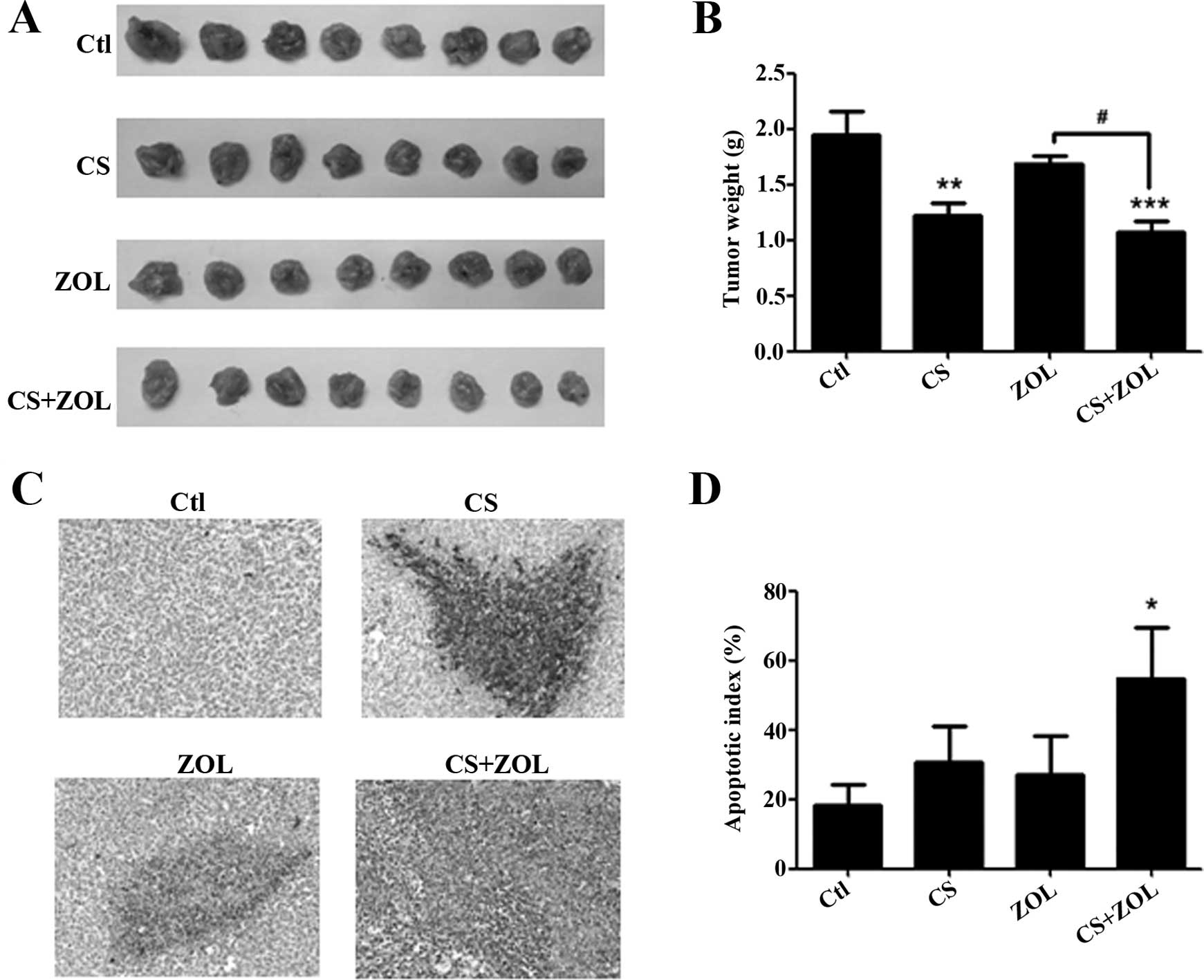 | Figure 1Effect of CS and/or ZOL on tumor
weight in 4T1 mouse mammary tumor model. (A) Representative tumor
images from different groups. (B) The graph shows the tumor weight
from different groups in 4T1 mouse mammary tumor model. (C)
Treatments of CS, ZOL and CS+ZOL−induced tumor cell death as
assessed by TunEl assay. (D) Quantification of apoptotic cells in
tumor sections according to the apoptotic index (%), defined as the
percentage of apoptotic cell area to the total tumor area. Data
were presented as mean + SEM, n=10. *P<0.05,
**p<0.01 and ***p<0.001 as compared
with control; #p<0.05. Ctl, control group; CS, CS
water extract treatment group in which mice were treated with 0.6
g/kg CS extract, orally fed everyday; ZOL, metronomic ZOL treatment
group in which mice were treated with 0.0125 mg/kg ZOL, i.p.
injected twice a week for 4 weeks; CS+ZOL, the combination
treatment group in which mice were treated with CS plus ZOL, and
the dose was identical to the individual treatment of CS or ZOL.
CS, Camellia sinensis; ZOL, zoledronate. |
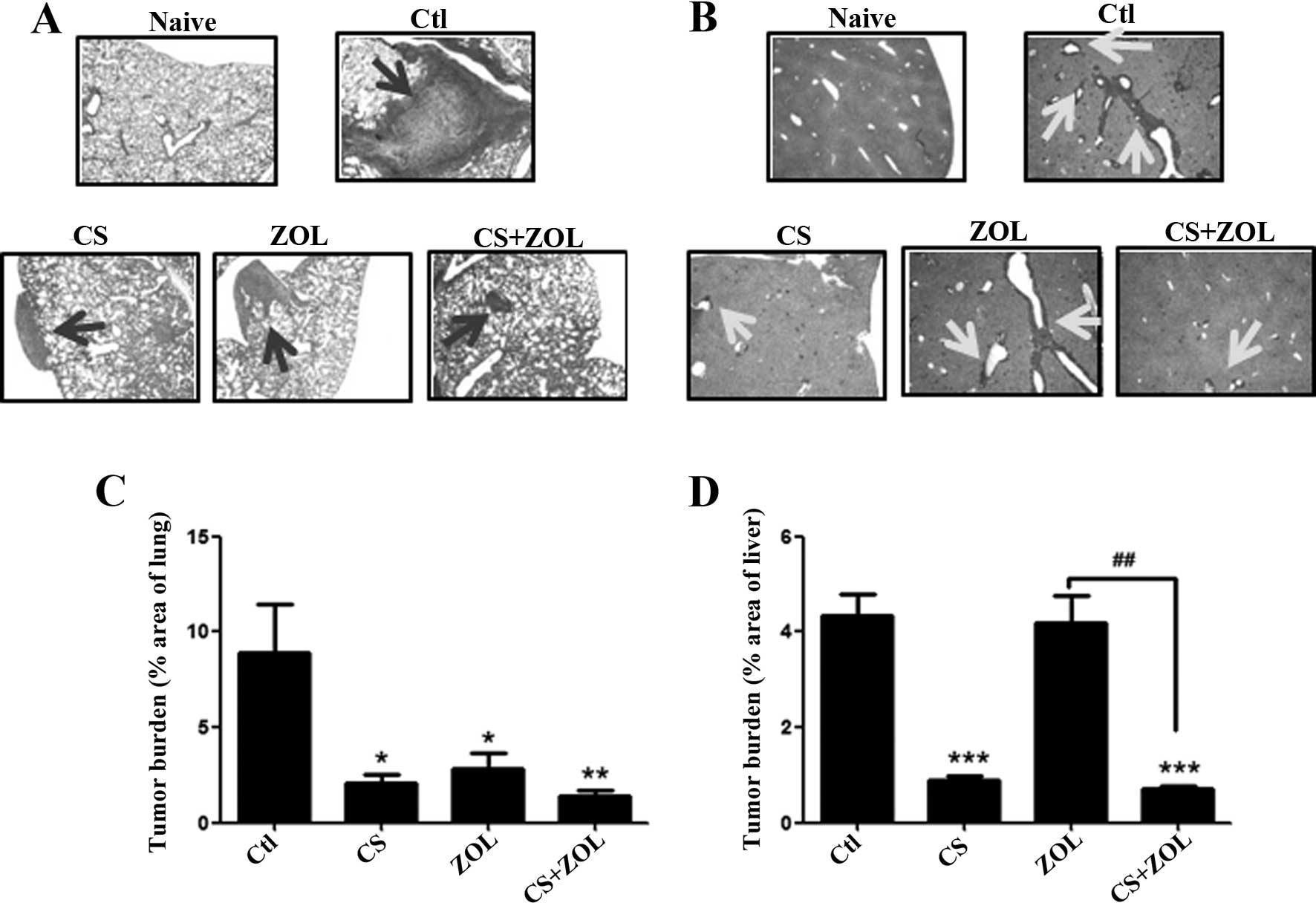
Effect of combination therapy on lung and
liver metastasis
Lungs and livers from each mouse were removed for
histological analysis and assessment of lung and liver metastasis.
Fig. 2A and B shows the
representative histological sections of lungs and livers from
different groups. Large 4T1 tumors were found in the untreated
control group (with arrows shown in lung and liver in the image),
while the tumor area and nodules were decreased in all the
treatment groups. Tumor burden in lung was found to be decreased by
76.4% (lung metastasis decreased from 8.9 to 2.1%), 68.5%
(decreased from 8.9 to 2.8%) and 80.8% (decreased from 8.9 to 1.7%)
in CS−, ZOL− and (CS+ZOL)-treated groups respectively, against
untreated control group (Fig. 2C).
Of the three treatment groups, the combination of CS+ZOL showed the
highest inhibition of lung metastasis. Similar results were found
in liver metastasis. Liver metastasis was decreased significantly
following CS and CS+ZOL treatments, while no obvious effect was
evident in the ZOL-treated group. A significant difference was
observed between ZOL and the combination of CS+ZOL (p<0.01)
(Fig. 2D).
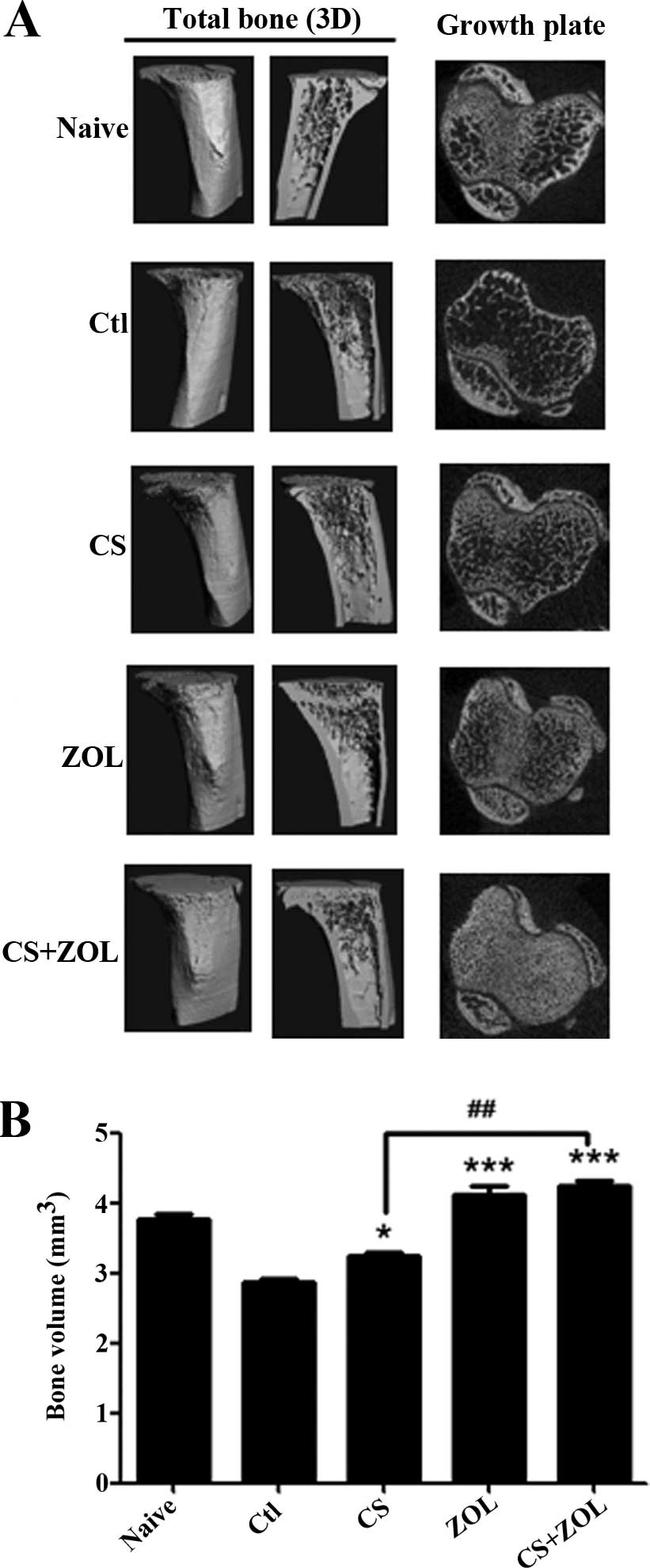
Effect of combination therapy on breast
cancer-induced osteolysis
At the late stage of cancer propagation, breast
cancer cells metastasize to bone and induce severe bone destruction
(25). To assess the efficacy of CS
and/or ZOL against breast cancer-induced bone destruction,
µ-CT analysis was employed. The 3D µ-CT images of
representative tibia from the control group demonstrated
significant bone destruction when compared to the corresponding
tibia from the non-tumor-bearing naive group (Fig. 3A). Animals treated with CS, ZOL and
CS+ZOL showed significant preservation of the bone structure. As
shown in Fig. 3B, the bone volume
(BV) in the control group was 2.87 mm3, lost nearly 18%
of BV when compared to the BV of the naive group, indicating severe
bone destruction in the control group. By contrast, treatments with
CS, ZOL and CS+ZOL resulted in a marked increase of BV in
tumor-bearing mice of 15.1, 43.2 and 45.1%, respectively. No
significant difference was shown between ZOL and CS+ZOL, while a
significant difference was identified between CS and the
combination of the CS+ZOL treatment groups. The marked
anti-osteolytic effect of CS+ZOL may be due to the effect of ZOL,
which is a clinical drug that is commonly used in the treatment of
bone disorders by inhibiting osteoclastic bone resorption.
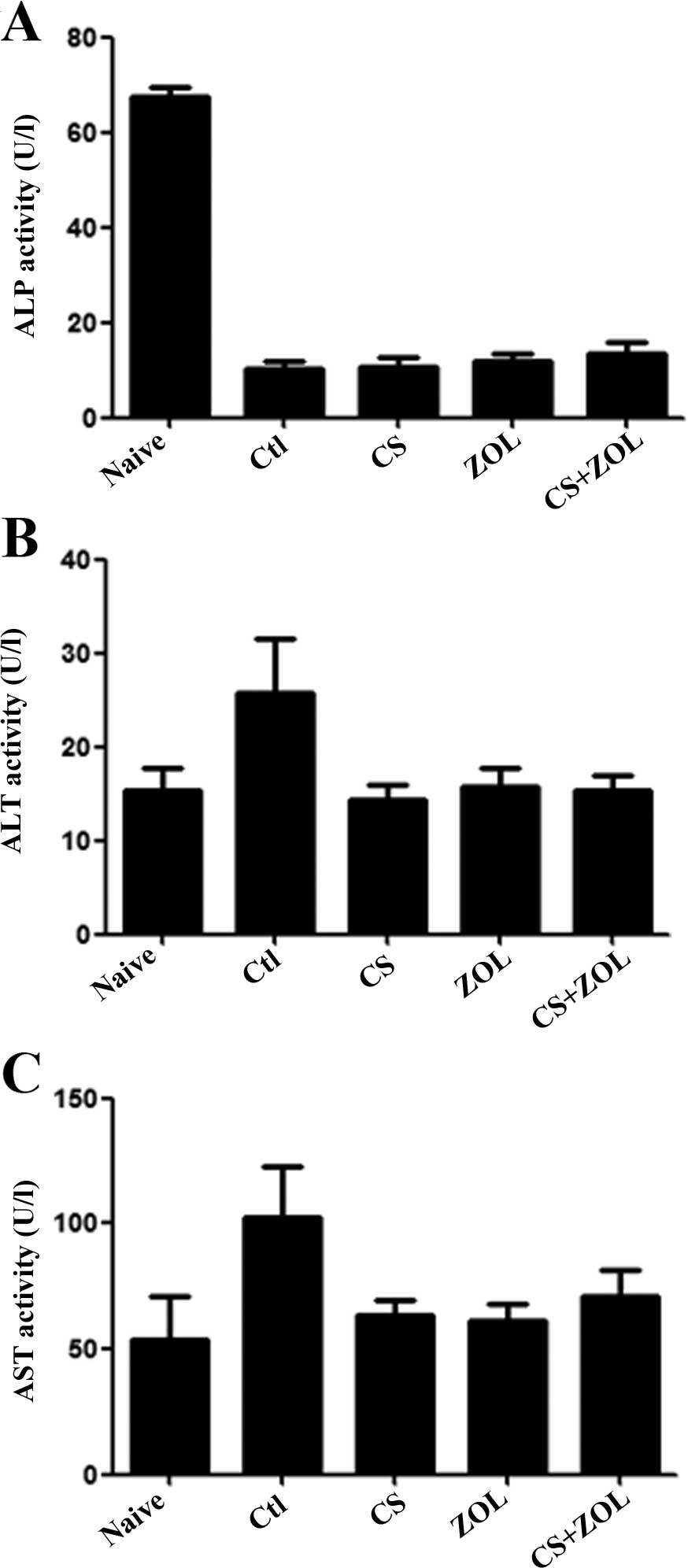
The bone-related enzyme ALP in plasma of mice was
assessed after treatments with CS and/or ZOL. As shown in fig. 4a, no significant difference was
shown in the treatment groups and the untreated control. However,
the ALP level in the non-tumor-bearing naive group was higher than
that in the tumor-bearing mice groups, indicating the damage of the
4T1 tumor to the bone.
Effect of the combination therapy on
hemato-biochemical markers
Hemato-biochemical markers in mice plasma were
tested following treatment with CS and/or ZOL by measuring the
plasma activities of liver-specific (ALT and AST) enzymes. As shown
in Fig. 4B and C, no significant
difference was shown in CS−, ZOL− and CS+ZOL−treated groups on
plasma activities of liver-specific enzymes, indicating no obvious
damage of the treatments to the liver.
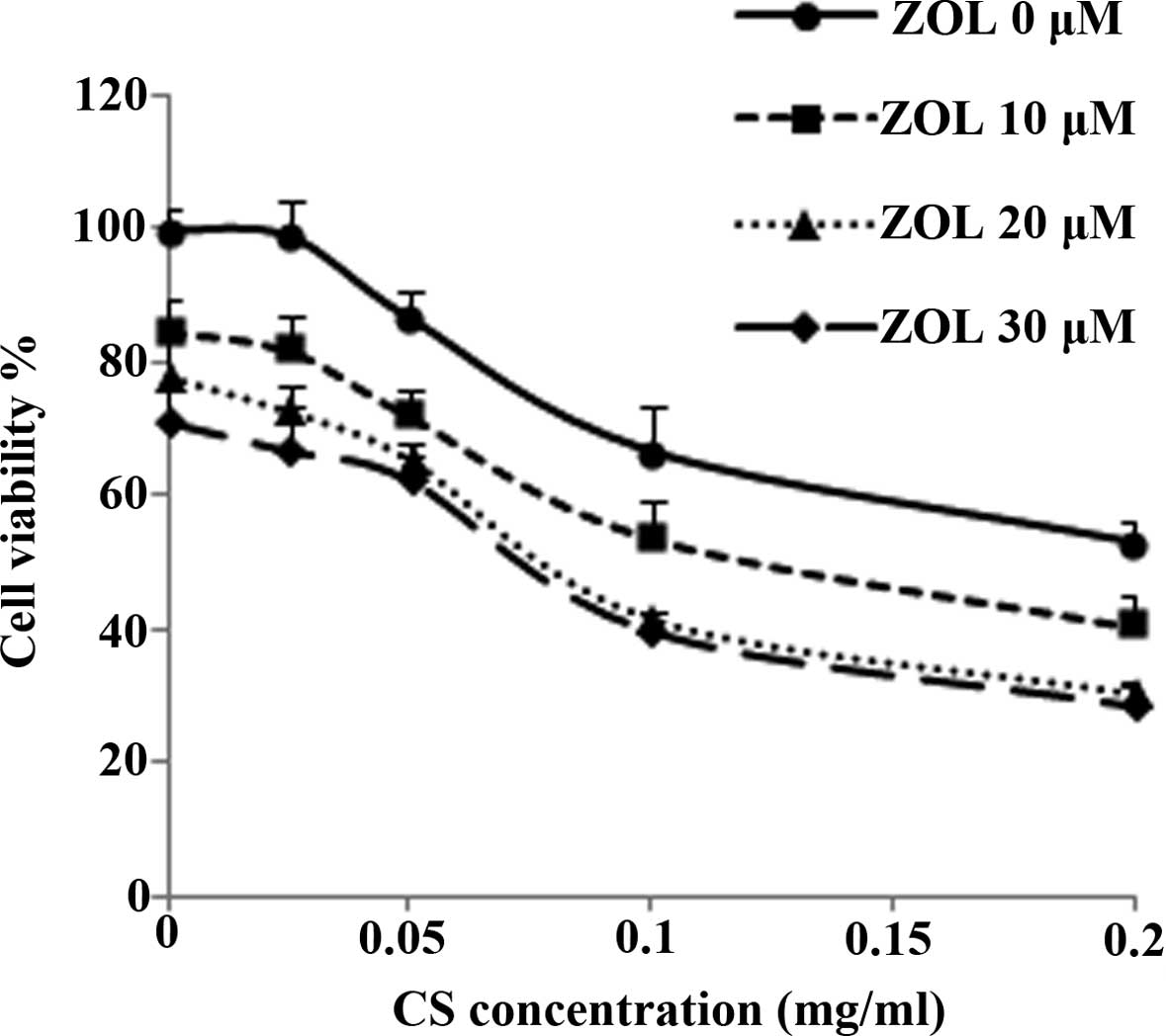
Effect of the combined use of CS and ZOL
on the viability of 4T1 cells
An MTT assay was performed to assess the effect of
ZOL as a single agent and in combination with CS at different
concentrations on the viability of 4T1 breast cancer cells, and the
most effective dose of the combination treatment was selected for
subsequent studies. As shown in Fig.
5, the growth of 4T1 cells was suppressed by 10, 20 and 30
µM ZOL in the presence of various concentrations of CS in a
dose-dependent manner. The combination of ZOL and CS demonstrated
additive cytotoxic effects, particularly at the doses of 20
µM ZOL plus 0.1 mg/ml of CS, which produced an inhibition of
58.5% cell viability, while ZOL and CS added alone caused
inhibition rates of 22.1 and 33.3%, respectively. Since the
combination of ZOL (20 µM) and CS (0.1 mg/ml) has the best
additive inhibition activity on cell viability, this combinatorial
dose was selected for subsequent experiments.
Effect of the combined use of CS and ZOL
on apoptotic induction
Treatment of 4T1 cells with CS extract resulted in
increasing number of apoptotic cells (early and late stages of
apoptotic cells) when compared to the untreated control (Fig. 6A). The percentage of apoptotic cells
following treatment with 0.1 or 0.2 mg/ml of CS extract for 24 h
were found to be 11 or 34.1%, respectively. ZOL (20 µM)
alone resulted in 7.6% of apoptotic cells. However, greater level
of apoptosis was observed after CS (0.1 or 0.2 mg/ml) was combined
with ZOL (20 µM), and the percentage of apoptotic cells was
found to be 43 and 66%, respectively, with significant differences
being observed for the individual treatment of CS (p<0.05)
(Fig. 6B). Furthermore, CS-induced
4T1 cell apoptosis was concomitant with a dose-associated increase
in caspase-3 activity, and ZOL synergistically enhanced the
CS-induced apoptosis in 4T1 cells, while ZOL (20 µM) alone
resulted in no obvious increase of caspase-3 activity (Fig. 6C).
Effect of combined use of CS and ZOL on
4T1 cell migration and invasion
Treatment with CS alone had some effect on the
inhibition of cell migration of 4T1 cells. However, no significant
difference was observed in the CS-treated group. ZOL (20 µM)
alone induced no significant increase in the open wound area.
However, a greater level of increase in the open wound area on 4T1
cells was observed when the cells were treated with CS+ZOL, and
significant differences were observed (p<0.05) (Fig. 7A and B), indicating the combination
of CS+ZOL inhibited 4T1 cell migration and thus increased the open
wound area. Thus ZOL strengthened the inhibitory effect of CS
against 4T1 cell migration.
In order to determine the efficacy of CS and ZOL
against cancer cell invasion in vitro, the Transwell
migration assay was performed. Treatment of CS at 0.05 and 0.1
mg/ml resulted in significant inhibition of 4T1 cell invasion. ZOL
(20 µM) led to a non-significant decrease (16%) of the
4T1-invasive ability, and the addition of ZOL to CS did not improve
the anti-invasive properties of CS. A greater level of inhibition
on cell invasion was observed after CS was combined with ZOL, the
cell invasion was inhibited by 61 and 72% at 0.05 and 0.1 mg/ml,
respectively, of Cs+ZOL (20 µM). However, no significant
difference was identified between the Cs+ZOL and the individual
treatments of CS or ZOL alone.
Effect of the combined use of CS and ZOL
on MMP-2 and MMP-9 activities
Gelatin zymography was carried out to assess the
effect of CS and/or ZOL on the activity of matrix
metalloproteinases (MMP)-2 and -9, which played an important role
in extracellular matrix and basement membrane degradation (26). As shown in fig. 8a, MMP-2 and MMP-9 in the 4T1 cell
culture supernatant were detected in the gel at molecular weight of
72 and 92 kDa, respectively. The MMP-2 and MMP-9 digested the
gelatin substrate significantly, and clear bands were presented in
control. Treatment with CS or CS+ZOL resulted in the reduction of
the bands, indicating the suppression of the enzyme activities of
MMP-2 or MMP-9. The enzyme activity of MMP-2 and MMP-9 was
significantly suppressed by Cs, and greater levels of suppression
on MMP-9 and MMP-2 were observed in the combination of CS and ZOL.
However, no significant difference was identified between the
combination of CS+ZOL and the individual treatment of CS alone
(Fig. 8B and C).
Discussion
Combination therapy, in which one or more medication
or therapies are used simultaneously, is an effective cure for
cancer treatment (27). An optimal
combination therapy of antitumor agents results in synergistic or
additive therapeutic efficacy, including increased activity,
decreased side-effects and minimal or delayed drug resistance,
whereas some unsatisfactory combinations may produce antagonistic
efficacy (28). Patients with
cancer frequently use herbal medicine along with conventional
medical treatments to enhance the efficacy of drugs, remove the
unwanted side-effects and obtain additional protection. Over half
(53.9%) of cancer patients in Hong Kong took Chinese herbal
medicines along with chemotherapeutic agents (29). A clinical study from mainland China
showed that treatment with integrated traditional Chinese and
Western medicines for advanced gastric cardia carcinoma in 26 cases
resulted in 57% remission in patients when compared to untreated
individual (30). The Chinese
herbal medicine Yun Zhi (Coriolus versicolor) preparation
used in combination with chemotherapy in patients with breast,
gastric or colorectal cancer, resulted in a significant survival
advantage when compared with the conventional anticancer
chemotherapy alone (31). Green tea
polyphenol EGCG acted synergistically in combination with clinical
anticancer drugs cisplatin and designed trans-palladiums in ovarian
cancer cells (32). There is an
increasing trend in cancer treatment and research to use
combination therapy for advanced types of cancer (31–33).
In the present study, we aimed to determine whether a synergistic
or additive effect would be identified in the combination treatment
of the Chinese herbal extract of Camellia sinensis (CS) and
metronomic regimen of zoledronate (ZOL) against tumor growth,
metastasis and bone destruction in a 4T1 mouse mammary tumor model.
The metronomic dose of ZOL used in the present study was 0.0125
mg/kg, i.p. injected 8 times in 4 weeks, and the total amount was
identical to that of the single conventional treatment of 0.1
mg/kg. However, the metronomic manner decreased the daily dose and
increased the frequency (20).
In the 4T1 mouse mammary tumor model, the 4T1 mouse
mammary carcinoma cells were injected orthotopically into the
mammary fat pad of the female BALB/c mice directly. Due to the high
aggressivity and metastatic characteristics of 4T1 cells, it is
easy to establish metastasis to lung and liver, as well as in bone
(34). Therefore, the antitumor and
antimetastatic effects were assessed in this model. After
treatments with CS or CS+ZOL, tumor weights in the orthotopic site
were decreased significantly, and no significant body weight loss
was identified. The combined use of CS and metronomic ZOL showed
improved antitumor effect against the individual treatments of CS
or metronomic ZOL. In order to determine whether the apoptotic
process occurred in mammary tumors, TUNEL assay was employed.
Treatment of CS, ZOL and CS+ZOL resulted in cell death in 4T1
tumors, and the combination of CS and metronomic ZOL showed the
most potent apoptotic induction among the three groups. The
findings suggest that CS and metronomic ZOL exerted better effects
than the individual treatments. Apoptosis plays a crucial role in
cancer propagation, which was considered as a protective mechanism
against the development and progression of cancer (35).
The 4T1 cells are documented to be highly invasive
and to develop tumor metastasis to the lung, liver and bone after
primary tumor was established for 2–3 weeks in BALB/c mice
(34). Following treatment with CS,
ZOL and CS+ZOL, tumor burden in lung was decreased significantly
against the untreated control group. Consistent results were also
identified in liver metastasis, except that metronomic ZOL had no
obvious effect on inhibiting liver metastasis. Previous findings
showed that tea polyphenols from CS were effective in decreasing
metastasis of tumor cells to lungs in mice bearing 4T1 tumors
(36). The present study has
demonstrated that the combination of CS and metronomic ZOL had
improved results in decreasing lung and liver metastasis in
contrast to the individual treatment only, suggesting that CS and
metronomic ZOL cooperated in inhibiting tumor metastasis.
Apart from lung and liver metastasis, bone
destruction was also assessed using µ-CT analysis. It was
determined that treatment with CS, ZOL and CS+ZOL resulted in a
marked increase of BV in mice of 15.1, 43.2 and 45.1%,
respectively, and the combined use had an optimal result among the
3 groups. It has been demonstrated that CS ingredients such as tea
polyphenols EGCG and EGC showed significant effects in inhibiting
formation of osteoclasts (37,38). A
recent animal study further confirmed the finding that EGCG
decreased ovariectomy-induced bone loss in mice via inhibition of
osteoclasts (39). In addition, ZOL
is a clinical drug that is commonly used in the treatment of bone
disorders by inhibiting osteoclastic bone resorption (40). ZOL inhibits osteoclastic bone
resorption by preventing prenylation of GTPases and ultimately
induces cell death in osteoclasts (41). Our previous study demonstrated that
metronomic ZOL also had significant anti-osteolytic effect
(20). In the present study, the
combination of CS and metronomic ZOL was firstly demonstrated to
have great potential in anti-osteolytic effects.
In addition, hemato-biochemical marker (ALP, ALT and
AST) tests were performed following treatment with CS and/or
metronomic ZOL. No significant difference was identified on the
enzymes level of ALT and AST in the treatment groups, suggesting
that CS and metronomic ZOL had no obvious damage to the liver. The
ALP activities in normal naive mice were higher than those of the
control, which may be due to the growth of the 4T1 tumor. It was
demonstrated that 4T1 cells are highly invasive and have metastatic
characteristics exhibiting primary tumor metastases to lung, liver
and bone as early as 2 weeks after inoculation (34). Our results have shown significant
bone destruction induced by 4T1 cancer, particularly in the control
group. Any condition of bone growth may cause the elevation of ALP
levels (42). The high level of ALP
in normal naive mice may be due to bone growth. While in the 4T1
tumor-bearing mice, the bone was destroyed by the metastasized
breast cancer cells and the bone growth was retarded, the ALP level
was decreased in tumor-bearing groups as compared to the naive
group.
Furthermore, the in vitro apoptosis induction
of the combination of CS and ZOL was assessed. The combination of
ZOL (20 µM) and CS (0.1 mg/ml) in 4T1 cells have the best
additive inhibition on cell viability, and the doses of the
combination were selected in the subsequent studies. The results
from Annexin V-FITC/PI double staining and caspase-3 activity
assays demonstrated that 4T1 cells treated with the CS water
extract resulted in an increased number of apoptotic cells in a
dose-dependent manner. A greater level of apoptosis was observed
after CS was combined with ZOL, and significant differences were
shown between individual CS treatment and the combination of CS and
ZOL (Fig. 6). The results suggest
that ZOL synergistically enhanced CS-induced apoptosis in 4T1
cells. The in vitro findings demonstrated by Annexin
V-FITC/PI staining and caspase-3 activity assays were completely in
concordance with the TUNEL results from tumor sections showing that
ZOL synergistically enhanced CS-induced apoptosis in 4T1 cells.
The anti-migration and anti-invasion abilities of CS
and/or ZOL were also assessed. The combination of CS plus ZOL
inhibited 4T1 cell migration, and a significant difference was
observed, indicating that ZOL strengthened the inhibitory effect of
CS on cell migration against 4T1 cells. Previous studies showed
that ZOL significantly prevented cell migration in MDA-MB-231
breast cancer cells (43), and CS
water extract was effective in inhibiting the proliferation and
migration of human SW620 colon cancer cells (44). Our results show that the combination
of CS and ZOL had a greater level of inhibition on 4T1 cell
migration against the individual treatment of CS or ZOL. In the
invasion assay, no additive or synergistic effect was shown in the
combined use of CS and ZOL on 4T1 cell invasion. The reason for
this finding may be due to the high dose of CS herbal extract used
in this assay, and that treatment of CS alone already resulted in
significant inhibition of cell invasion. In addition, results of
the mechanistic study showed that the enzyme activity of MMP-2 and
MMP-9 was significantly suppressed by CS plus ZOL, indicating the
potent antimetastatic effect of the combined use of CS and ZOL. The
in vitro findings showed that the combined use of CS+ZOL
inhibited 4T1 cell migration and invasion significantly, which may
partly explain the activity of CS+ZOL in the inhibition of tumor
metastasis to lung and liver in vivo. As demonstrated by
Oppenheimer, cell migration is the first step of the invasive
process and plays an important role in tumor metastasis (45).
Although the underlying mechanism of the antitumor
and antimetastatic effects of the combined use of CS+ZOL was not
well investigated in the present study, studies have demonstrated
the apoptotic induction and antimetastatic effects of CS or ZOL.
Baliga et al found that green tea CS extract inhibited 4T1
cell proliferation and induced apoptosis in this cell line by
activation of caspase-3 and PARP (36). In addition, our results revealed
that CS suppressed the enzyme levels of MMP-2 and MMP-9
significantly in 4T1 cells, suggesting that CS had potent
antimetastatic effects. On the other hand, previous studies have
demonstrated that ZOL induced apoptosis in 4T1 cells via the
activation of caspase-3, and inhibited visceral metastasis in a 4T1
mouse breast cancer model (46).
ZOL also prevented cell migration and invasion in MDA-MB-231 breast
cancer cells (43).
In summary, to the best of our knowledge, our
results present the first evidence on the combination of Chinese
herbal extract CS and conventional drug ZOL against tumor growth,
metastasis and bone destruction in a 4T1 mouse tumor model. The
combination of CS and metronomic ZOL cooperated in decreasing tumor
burden and metastasis, and significantly inhibited the breast
cancer-induced bone destruction in a 4T1 mouse model. Additionally,
the combined use of CS and ZOL inhibited cell migration and
invasion, and induced apoptosis in 4T1 cells. The novel application
of herbal extract CS plus metronomic ZOL promoted the antitumor,
antimetastatic and anti-osteolytic effects of ZOL against
metastatic breast cancer. The metronomic dose means more frequent
and low-dose drug administration compared with conventional
therapy, which was first experimented previously with positive
results for patients with metastatic breast and recurrent ovarian
cancer, advanced multiple myeloma and other types of carcinomas
(47,48). The metronomic use of ZOL was found
to be effective in inhibiting tumor growth and metastasis to lung
and liver in both of primary and metastatic breast cancer (20). The present study results reveal that
metronomic ZOL was effective in decreasing tumor burden, inhibiting
metastasis to lung and inhibiting osteolysis in 4T1 mouse
carcinoma. In addition, the combination of metronomic CS and ZOL
shows the most potent antitumor and antimetastasis effects among
the three groups. Such findings reveal promising results of
metronomic ZOL in clinical trials, and may play influential roles
in the future production of CS as a supplement in breast cancer
treatment with ZOL.
Based on results of the present study, more detailed
molecular mechanisms, for instance, genomic and proteomic responses
on antimetastasis and anti-osteolysis should be studied. Further
investigation is needed to determine the clinical efficacy and
safety of the combination therapy of CS and metronomic ZOL in human
subjects with breast cancer metastasis. Our observations revealed
promising results in breast cancer treatment of the combined use of
herbal medicine CS and metronomic ZOL, suggesting the promising
application in breast cancer patients clinically.
Acknowledgments
This study was supported by the Focused Innovations
Scheme (Major area scheme a, Phase 2) of The Chinese university of
Hong Kong. The authors would like to thank Dr Sammy Siu and Ms.
Anita Shum for their technical support on the micro-CT
analysis.
References
|
1
|
Hara Y: Advances in Food Science and
Technology. Nippon shokuhin Kogyo Gakkai; Korin, Tokyo: 1990
|
|
2
|
Kuriyama S, Shimazu T, Ohmori K, Kikuchi
N, Nakaya N, Nishino Y, Tsubono Y and Tsuji I: Green tea
consumption and mortality due to cardiovascular disease, cancer,
and all causes in Japan: The Ohsaki study. JAMA. 296:1255–1265.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shixian Q, VanCrey B, Shi J, Kakuda Y and
Jiang Y: Green tea extract thermogenesis-induced weight loss by
epigallocat-echin gallate inhibition of
catechol-O-methyltransferase. J Med Food. 9:451–458. 2006.
View Article : Google Scholar
|
|
4
|
Nakazato T, Ito K, Miyakawa Y, Kinjo K,
Yamada T, Hozumi N, Ikeda Y and Kizaki M: Catechin, a green tea
component, rapidly induces apoptosis of myeloid leukemic cells via
modulation of reactive oxygen species production in vitro and
inhibits tumor growth in vivo. Haematologica. 90:317–325.
2005.PubMed/NCBI
|
|
5
|
Bettuzzi S, Brausi M, Rizzi F, Castagnetti
G, Peracchia G and Corti A: Chemoprevention of human prostate
cancer by oral administration of green tea catechins in volunteers
with high-grade prostate intraepithelial neoplasia: A preliminary
report from a one-year proof-of-principle study. Cancer Res.
66:1234–1240. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gu JW, Makey KL, Tucker KB, Chinchar E,
Mao X, Pei I, Thomas EY and Miele L: EGCG, a major green tea
catechin suppresses breast tumor angiogenesis and growth via
inhibiting the activation of HIF-1α and NFκB, and VEGF expression.
Vasc Cell. 5:92013. View Article : Google Scholar
|
|
7
|
Lecumberri E, Dupertuis YM, Miralbell R
and Pichard C: Green tea polyphenol epigallocatechin-3-gallate
(EGCG) as adjuvant in cancer therapy. Clin nutr. 32:894–903. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Qiao J, Gu C, shang W, Du J, Yin W, Zhu M,
Wang W, Han M and Lu W: Effect of green tea on pharmacokinetics of
5-fluorouracil in rats and pharmacodynamics in human cell lines in
vitro. Food Chem Toxicol. 49:1410–1415. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liang G, Tang A, Lin X, Li L, Zhang S,
Huang Z, Tang H and Li QQ: Green tea catechins augment the
antitumor activity of doxorubicin in an in vivo mouse model for
chemoresistant liver cancer. Int J Oncol. 37:111–123.
2010.PubMed/NCBI
|
|
10
|
Farabegoli F, Papi A and Orlandi M:
(−)-Epigallocatechin-3-gallate down-regulates EGfr, MMP-2, MMP-9
and EMMPrIn and inhibits the invasion of MCf-7 tamoxifen-resistant
cells. Biosci Rep. 31:99–108. 2011. View Article : Google Scholar
|
|
11
|
Luo KW, Ko CH, Yue GGL, Lee JKM, Li KK,
Lee M, Li G, Fung KP, Leung PC and Lau CB: Green tea (Camellia
sinensis) extract inhibits both the metastasis and osteolytic
components of mammary cancer 4T1 lesions in mice. J Nutr Biochem.
25:395–403. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Labrinidis A, Hay S, liapis V, findlay DM
and Evdokiou A: Zoledronic acid protects against
osteosarcoma-induced bone destruction but lacks efficacy against
pulmonary metastases in a syngeneic rat model. Int J Cancer.
127:345–354. 2010.
|
|
13
|
Senaratne SG and Colston KW: Direct
effects of bisphosphonates on breast cancer cells. Breast Cancer
Res. 4:18–23. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Evdokiou A, Labrinidis A, Bouralexis S,
Hay S and Findlay DM: Induction of cell death of human osteogenic
sarcoma cells by zoledronic acid resembles anoikis. Bone.
33:216–228. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wood J, Bonjean K, Ruetz S, Bellahcène A,
Devy L, Foidart JM, Castronovo V and Green JR: Novel antiangiogenic
effects of the bisphosphonate compound zoledronic acid. J Pharmacol
Exp Ther. 302:1055–1061. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao X, Xu X, Guo L, et al: Biomarker
alterations with metronomic use of low-dose zoledronic acid for
breast cancer patients with bone metastases and potential clinical
significance. Breast Cancer Res Treat. 124:733–743. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhao X, Xu X, Zhang Q, Jia Z, Sun S, Zhang
J, Wang B, Wang Z and Hu X: Prognostic and predictive value of
clinical and biochemical factors in breast cancer patients with
bone metastases receiving ‘metronomic’ zoledronic acid. BMC Cancer.
11:4032011. View Article : Google Scholar
|
|
18
|
Daubiné F, Le Gall C, Gasser J, Green J
and Clézardin P: Antitumor effects of clinical dosing regimens of
bisphosphonates in experimental breast cancer bone metastasis. J
Natl Cancer Inst. 99:322–330. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Facchini G, Caraglia M, Morabito A, et al:
Metronomic administration of zoledronic acid and taxotere
combination in castration resistant prostate cancer patients: Phase
I ZANTE trial. Cancer Biol Ther. 10:543–548. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Luo KW, Ko CH, Yue GGL, et al: Anti-tumor
and anti-osteolysis effects of the metronomic use of zoledronic
acid in primary and metastatic breast cancer mouse models. Cancer
Lett. 339:42–48. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Peng L, Song XH, Shi XG, Li JX and Ye CX:
An improved HPLC method for simultaneous determination of phenolic
compounds, purine alkaloids and theanine in Camellia species. J
Food Compos Anal. 21:559–563. 2008. View Article : Google Scholar
|
|
22
|
Staibano S, Lo Muzio L, Mezza E,
argenziano G, Tornillo L, Pannone G and De Rosa G: Prognostic value
of apoptotic index in cutaneous basal cell carcinomas of head and
neck. Oral Oncol. 35:541–547. 1999. View Article : Google Scholar
|
|
23
|
Vigl B, Zgraggen C, Rehman N,
Banziger-Tobler NE, Detmar M and Halin C: Coxsackie- and adenovirus
receptor (CAR) is expressed in lymphatic vessels in human skin and
affects lymphatic endothelial cell function in vitro. Exp Cell Res.
315:336–347. 2009. View Article : Google Scholar
|
|
24
|
Xiao D and Singh SV: Phenethyl
isothiocyanate inhibits angio-genesis in vitro and ex vivo. Cancer
res. 67:2239–2246. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Azim HA, Kamal NS and Azim HA Jr: Bone
metastasis in breast cancer: The story of RANK-ligand. J Egypt Natl
Cancer Inst. 24:107–114. 2012. View Article : Google Scholar
|
|
26
|
Chaudhary AK, Pandya S, Ghosh K and
Nadkarni A: Matrix metalloproteinase and its drug targets therapy
in solid and hematological malignancies: An overview. Mutat Res.
753:7–23. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Fitzgerald JB, Schoeberl B, Nielsen UB and
Sorger PK: Systems biology and combination therapy in the quest for
clinical efficacy. Nat Chem Biol. 2:458–466. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yeh P and Kishony R: Networks from
drug-drug surfaces. Mol Syst Biol. 3:852007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lam YC, Cheng CW, Peng H, Law CK, Huang X
and Bian Z: Cancer patients’ attitudes towards Chinese medicine: A
Hong Kong survey. Chin Med. 4:252009. View Article : Google Scholar
|
|
30
|
Wang JA, Zhou FX and Zhang ML: Treatment
for advanced gastric cardia carcinoma in 26 cases of integrated
traditional Chinese and Western Medicine. Shanxi J Tradit Chin Med.
25:772–773. 2004.
|
|
31
|
Eliza WL, Fai CK and Chung LP: Efficacy of
Yun zhi (Coriolus versicolor) on survival in cancer patients:
Systematic review and meta-analysis. Recent Pat Inflamm allergy
Drug Discov. 6:78–87. 2012. View Article : Google Scholar
|
|
32
|
Mazumder ME, Beale P, Chan C, Yu JQ and
Huq F: Epigallo-catechin gallate acts synergistically in
combination with cisplatin and designed trans-palladiums in ovarian
cancer cells. Anticancer Res. 32:4851–4860. 2012.PubMed/NCBI
|
|
33
|
Jackman AL, Kaye S and Workman P: The
combination of cytotoxic and molecularly targeted therapies - can
it be done. Drug Discov Today. 1:445–454. 2004.
|
|
34
|
Pulaski BA and Ostrand-Rosenberg S:
Reduction of established spontaneous mammary carcinoma metastases
following immunotherapy with major histocompatibility complex class
II and B7.1 cell-based tumor vaccines. Cancer Res. 58:1486–1493.
1998.PubMed/NCBI
|
|
35
|
Kerr JF, Wyllie AH and Currie AR:
Apoptosis: A basic biological phenomenon with wide-ranging
implications in tissue kinetics. Br J Cancer. 26:239–257. 1972.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Baliga MS, Meleth S and Katiyar SK: Growth
inhibitory and antimetastatic effect of green tea polyphenols on
metastasis-specific mouse mammary carcinoma 4T1 cells in vitro and
in vivo systems. Clin Cancer Res. 11:1918–1927. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ko CH, lau KM, Choy WY and leung PC:
Effects of tea catechins, epigallocatechin, gallocatechin, and
gallocatechin gallate, on bone metabolism. J Agric Food Chem.
57:7293–7297. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lee JH, Jin H, Shim HE, Kim HN, Ha H and
Lee ZH: Epigallo-catechin-3-gallate inhibits osteoclastogenesis by
down-regulating c-Fos expression and suppressing the nuclear
factor-kappaB signal. Mol Pharmacol. 77:17–25. 2010. View Article : Google Scholar
|
|
39
|
Lee SH, Kim BJ, Choi HJ, et al:
(−)-Epigallocathechin-3-gallate, an aMPK activator, decreases
ovariectomy-induced bone loss by suppression of bone resorption.
Calcif Tissue Int. 90:404–410. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Deeks ED and Perry CM: Zoledronic acid: A
review of its use in the treatment of osteoporosis. Drugs aging.
25:963–986. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang FL and Casey PJ: Protein
prenylation: Molecular mechanisms and functional consequences. Annu
Rev Biochem. 65:241–269. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Tobiume H, Kanzaki S, Hida S, Ono T,
Moriwake T, Yamauchi S, Tanaka H and Seino Y: Serum bone alkaline
phosphatase isoenzyme levels in normal children and children with
growth hormone (GH) deficiency: A potential marker for bone
formation and response to GH therapy. J Clin Endocrinol Metab.
82:2056–2061. 1997.PubMed/NCBI
|
|
43
|
Rachner TD, Singh SK, Schoppet M, Benad P,
Bornhäuser M, Ellenrieder V, Ebert R, Jakob F and Hofbauer LC:
Zoledronic acid induces apoptosis and changes the TRAIL/OPG ratio
in breast cancer cells. Cancer Lett. 287:109–116. 2010. View Article : Google Scholar
|
|
44
|
Zhou F, Zhou H, Wang T, Mu Y, Wu B, Guo
DL, Zhang XM and Wu Y: Epigallocatechin-3-gallate inhibits
proliferation and migration of human colon cancer SW620 cells in
vitro. Acta Pharmacol Sin. 33:120–126. 2012. View Article : Google Scholar
|
|
45
|
Oppenheimer SB: Cellular basis of cancer
metastasis: A review of fundamentals and new advances. Acta
Histochem. 108:327–334. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hiraga T, Williams PJ, Ueda A, Tamura D
and Yoneda T: Zoledronic acid inhibits visceral metastases in the
4T1/luc mouse breast cancer model. Clin Cancer Res. 10:4559–4567.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Orlando L, Cardillo A, Ghisini R, Rocca A,
Balduzzi A, Torrisi R, Peruzzotti G, Goldhirsch A, Pietri E and
Colleoni M: Trastuzumab in combination with metronomic
cyclophosphamide and methotrexate in patients with HER-2 positive
metastatic breast cancer. BMC Cancer. 6:2252006. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
de Weerdt O, van de Donk NW, Veth G, Bloem
AC, Hagenbeek A and Lokhorst HM: Continuous low-dose
cyclophosphamide-prednisone is effective and well tolerated in
patients with advanced multiple myeloma. Neth J Med. 59:50–56.
2001. View Article : Google Scholar : PubMed/NCBI
|