Introduction
Curcumin, a biocompatible polyphenol of the dietary
spice turmeric, has been shown to exhibit anticancer effects in
multiple cancers including those of the breast, ovary, colon and
brain (1). However, the clinical
application of curcumin has been hindered to date, not only due to
its low solubility and bioavailability, but also by limitations in
its anticancer potency. It is, therefore, important to better
understand how curcumin influences the biology of cancer cells so
that new strategies can be devised to enhance its antitumor
effect.
Recent evidence points to mitochondria as the target
for many of the beneficial effects of curcumin (2). A major function of mitochondria is to
generate energy by utilizing substrates. Although malignant cells
undergo reprogramming of energy metabolism toward aerobic
glycolysis (3), they also depend on
mitochondrial oxidative metabolism for proliferation and survival.
Thus, cancer cell glucose metabolism is gaining interest as a
promising target for cancer treatment (4), and is exploited by positron emission
tomography (PET) with 18F-fluorodeoxyglucose
(18F-FDG) for monitoring treatment efficacy (5). Another important mitochondrial
function is the production of reactive oxygen species (ROS), which
mediates crucial cellular tasks including signal transduction and
induction of apoptosis (6). Hence,
cancer-specific modifications to ROS and anti-oxidants are also
considered a potential target for therapy (6).
A link between curcumin action and glucose
metabolism is suggested by observations in skeletal muscle
(7) and myotubes (8) that curcumin can stimulate glucose
uptake. However, how cancer cell glucose metabolism is influenced
by curcumin, and whether suppressing this response affects its
anticancer action has not been investigated. Furthermore, although
curcumin is through to induce apoptosis of cancer cells through
oxidative stress (9,10), it is not known whether suppressing
the ROS scavenging activity of curcumin (11) can enhance its antitumor effect. This
could be relevant for the development of newer curcumin analogs
with strengthened pro-oxidant properties for improved therapeutic
response (12,13). In addition, since cancer cells
heavily rely on glucose utilization, understanding how
metabolism-targeting drugs modulate tumor 18F-FDG uptake
may not only help uncover new strategies for cancer treatment but
could also expand the role of PET imaging for monitoring treatment
response.
In the present study, we examined how curcumin
influences cancer cell glucose metabolism and mitochondrial
function, including 18F-FDG uptake, lactate production,
hexokinase activity, oxygen consumption, mitochondrial membrane
potential (MMP) and ROS production. We further investigated whether
restricting glycolysis with 2-deoxyglucose (2-DG) and addition of
Cu2+, known to reduce curcumin's anti-oxidant property
(14–16), can enhance the anticancer action of
the compound.
Materials and methods
Cancer cell lines and culture
MCF-7 human breast cancer and CT26 colon cancer cell
lines were from the American Type Culture Collection (ATCC;
Rockville, MD, USA), and maintained in MEM (MCF-7 cells) or
RPMI-1640 media (CT26 cells) supplemented with 10% fetal bovine
serum and 1% penicillin/streptomycin in a humidified incubator at
37°C, 5% CO2. Cells grown in a monolayer were routinely
split 2 times a week, and experiments were performed at 48 h after
seeding. For cell treatment, stock solutions of curcumin dissolved
in DMSO, 2-DG dissolved in phosphate-buffered solution (PBS), and
CuSO4 dissolved in 0.05 M Tris-HCl (pH 7.4) were
used.
Reagents and antibodies
Curcumin, 2-DG, copper sulfate (II)
(CuSO4), oligomycin, carbonyl
cyanide-4-(trifluoromethoxy) phenylhydrazone (FCCP) and antimycin-A
were from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA).
CM-H2DCFDA
(5-(and-6)-chloromethyl-29,79-dichlorodihydrofluorescein diacetate,
acetyl ester) and MitoTracker Red FM was from Invitrogen Life
Technologies (Grand Island, NY, USA). Antibodies against
phosphorylated mammalian target of rapamycin (p-mTOR) and
phosphorylated extracellular-signal-regulated kinase (p-ERK), and
anti-rabbit and anti-mouse secondary antibodies were from Cell
Signaling Technology (Danvers, MA, USA). Polyclonal antibody
against β-actin was from Santa Cruz Biotechnology (Dallas, TX,
USA). Rabbit polyclonal antibody against glucose transporter 1
(GLUT1) was from Abcam (Cambridge, MA, USA).
Sulforhodamine B (SRB) assay
Surviving cell content following treatment was
evaluated by SRB assays. Briefly, cells seeded overnight on a
96-well plate were treated for indicated durations of time, and
cell monolayers were fixed with 10% (wt/vol) trichloroacetic acid
at 4°C. After cells were stained with SRB dye for 30 min, excess
dye was removed by washing repeatedly with 1% (v/v) acetic acid.
Protein-bound dye was finally dissolved in 10 mM Tris base solution
and subject to spectrophotometric measurement of absorbance at 510
nm using a microplate reader.
FDG uptake measurement
Cells in 12-well plates were incubated with 370 kBq
(10 µCi) of the radiolabeled glucose analogue
18F-FDG for 40 min at 5% CO2, 37°C. After
rapid washing twice with cold PBS, cells were lysed with 500
µl of 0.01 N NaOH and cell-associated radioactivity was
measured on a high-energy γ-counter (Perkin-Elmer, Waltham, MA,
USA). Radio-uptake levels were corrected for cell content as
assessed by Bradford protein assays.
Lactate production
L-lactate production was measured from 100 µl
of culture medium using a Cobas assay kit (Roche/Hitachi) following
the manufacturer's instructions. In the assay, lactate is
enzymatically converted to pyruvate and hydrogen peroxide. Hydrogen
peroxide then undergoes an enzymatic reaction to generate a colored
dye that is measured by absorbance on a microplate
spectrophotometer. Lactate concentration was calculated from a
standard curve of serially diluted standards and expressed in
mU/mg.
Hexokinase activity
Cells homogenized in homogenizing buffer (50 mM
triethanolamine and 5 mM MgCl2; pH 7.6) were centrifuged
at 1,000 × g for 5 min at 4°C. The pelleted mitochondrial fraction
was isolated and 50 µl was mixed with 2.52 ml of reaction
buffer containing 39 mM triethanolamine, 216 mM D-glucose, 0.74 mM
adenosine 5′-triphosphate, 7.8 mM magnesium chloride, 1.1 mM
β-nicotinamide adenine dinucleotide phosphate, and 2.5 units of
glucose 6-phosphate dehydrogenase. The reaction mixture was
repeatedly measured at 25°C for spectrophotometric absorbance at
340 nm. Hexokinase activity was expressed as mU/mg of protein, and
one unit of activity was defined as the amount of hexokinase
activity that phosphorylates 1 µmol of glucose per min at
25°C.
Oxygen consumption rate (OCR)
Cells were seeded in 100 µl of MEM media at a
density of 5×104 cells/well on analyzer plates. A
Seahorse XF24 Extracellular Flux analyzer (Seahorse Bioscience
Inc., North Billerica, MA, USA) with a solid state sensor probe
analyzed the concentration of oxygen dissolved in the media at
2-sec intervals. Following measurement of basal OCR level, 1.2
µM oligomycin (ATP synthase inhibitor), 4 µM FCCP
(proton gradient uncoupler), and 10 µM antimycin A
(mitochondria electron transport inhibitor) were sequentially added
to assess their effects on OCR. Cell density and working
concentrations for analysis was optimized according to the
manufacturer's manual. OCR was automatically calculated, recorded,
and plotted by Seahorse XF24 software version 1.8.
MMP measurement
Cells were seeded at densities of 5×104
per well in a 96-well black plate with a transparent bottom.
Culture media was removed and wells were replenished with 100
µl PBS containing 500 nM of MitoTracker Red FM (Invitrogen),
a fluorescent dye that stains mitochondria of live cells as a
function of mΔΨ. Cells were incubated for 30 min at 37°C in 5%
CO2, and then washed with 100 µl of warmed PBS
per well. Fluorescence remaining in each well was measured on a
microplate reader using 594 nm excitation and 642 nm emission
wavelengths.
Intracellular ROS production
To quantify intracellular ROS, cells were seeded at
densities of 5×104/well in a 96-well black plate.
Culture media with treating agents was removed and wells were
replenished with 100 µl PBS containing 10 µM of
CM-H2DCFDA (Molecular Probes-Invitrogen), a
cell-permeant fluorescent indicator for intracellular ROS. The
probe is non-fluorescent until the acetate groups are removed by
intracellular esterases and oxidation occurs within the cell, which
can be detected by monitoring the increase in fluorescence. Cells
were incubated with the probe for 30 min at 37°C in 5%
CO2, after which excessive dye was removed and cells
were washed with 100 µl of warmed PBS per well. Fluorescence
remaining in each well was measured on a GloMax®
microplate reader (Promega, Seoul, Korea) with 490 nm excitation
and 510–570 nm emission wavelengths.
Western blot analysis
After cells were lysed with cold PRO-PREP lysis
buffer (iNtRON Biotechnology, Seoul, Korea), 30 µg of total
cell lysate was separated by 10% SDS-PAGE and transferred to a
polyvinylidene difluoride membrane. The membrane was blocked with
5% non-fat milk in Tris-buffered saline with Tween-20 for 1 h at
room temperature and incubated with primary antibodies (1:1,000;
4°C, overnight). The membrane was then incubated with HRP-linked
anti-IgG (1:3,000; room temperature, 1 h). Immunoreactive protein
was detected by chemiluminescence, and band intensities were
quantified on a GS-800 densitometer using Quantity One software
(Bio-Rad Laboratories, Hercules, CA, USA).
Statistical analysis
All experiments were repeated two or three separate
times, and the mean ± SD of triplicate samples from a single
representative experiment is presented unless otherwise specified.
Student's t-tests were used to evaluate the statistical
significance of measurements between groups, and P-values <0.05
were considered statistically significant.
Results
Curcumin dose-dependently stimulates
cancer cell 18F-FDG uptake
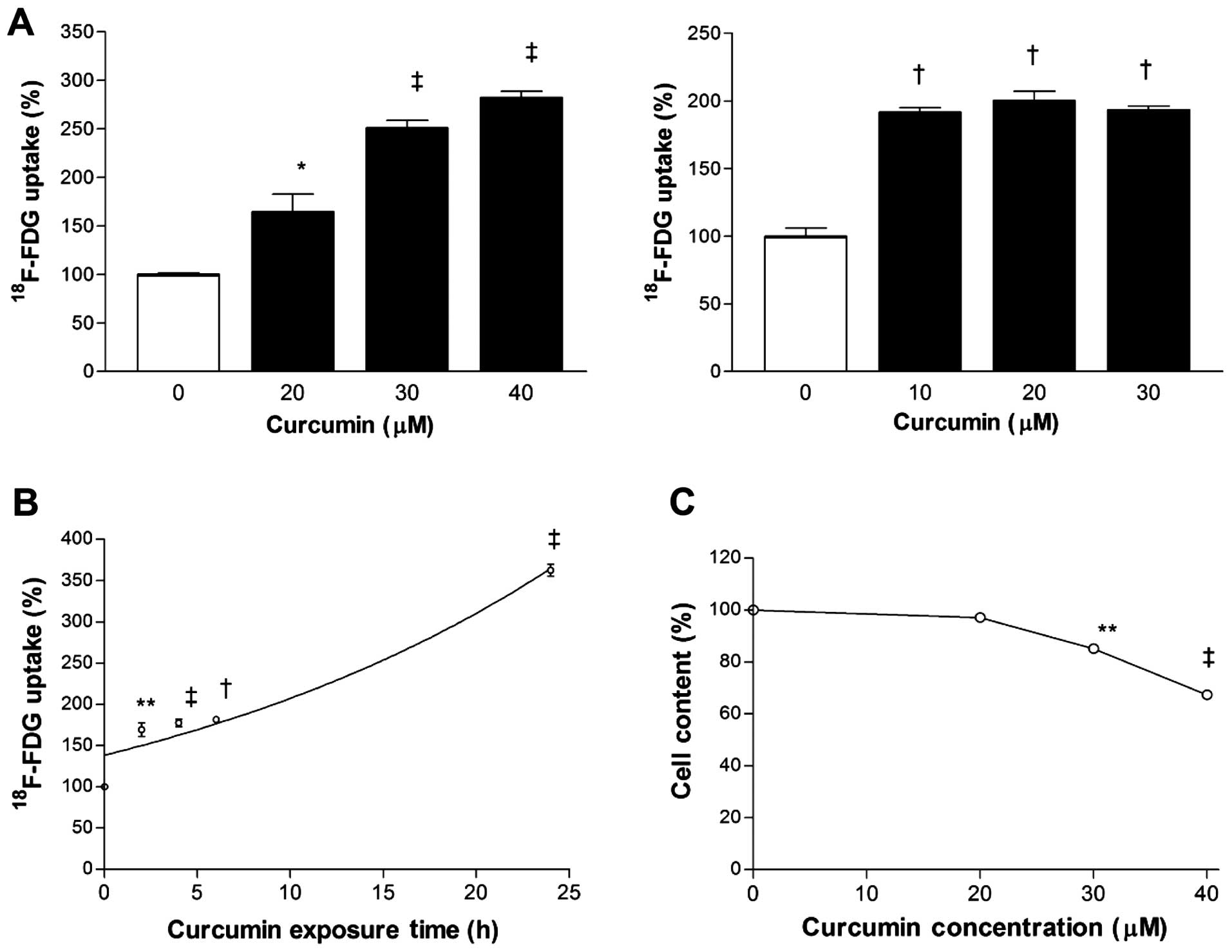
Curcumin dose-dependently stimulated
18F-FDG uptake of MCF-7 and CT26 cancer cells. Hence,
18F-FDG uptake corrected for cell content was
substantially increased to 250±13 and 193±5% of control levels by
exposure to 30 µM curcumin for MCF-7 cells and 10 µM
curcumin for CT26 cells for 24 h (Fig
1A). Time course experiments in MCF-7 cells disclosed that
stimulation of 18F-FDG uptake by 30 µM curcumin
began as soon as 2 h, with further increases that reached
362.5±12.5% of control level by 24 h (Fig. 1B). This metabolic effect occurred at
a curcumin dose that only mildly reduced surviving cell content
(85.1±2.7% of controls; Fig.
1C).
Curcumin stimulates glycolytic flux and
suppresses mitochondrial respiration
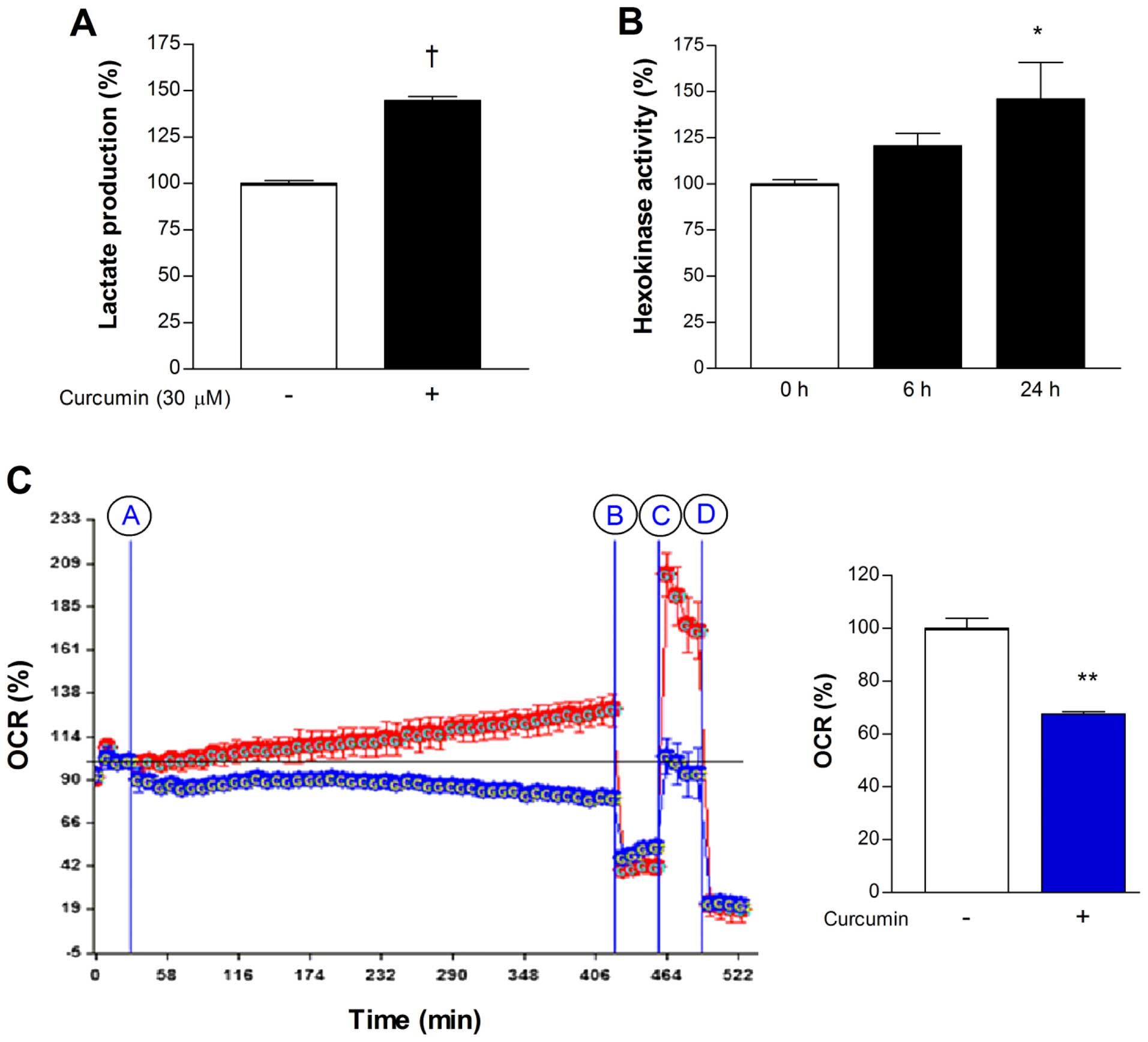
In MCF-7 cells, treatment with 30 µM curcumin
for 24 h caused a significant elevation of lactate release to
144.6±3.9% of controls (Fig. 2A),
consistent with a shift of metabolism toward glycolytic flux. To
evaluate the contribution of the major mediators of cellular
18F-FDG uptake, we measured hexokinase activity and
membrane GLUT1 expression. As a result, 30 µM curcumin for
24 h was found to significantly augment mitochondrial hexokinase
activity to 146.0±34.5% of controls (Fig. 2B). In contrast, immunoblotting
showed that curcumin had no significant influence on the level of
membrane GLUT1 expression (data not shown).
We next evaluated whether the shift toward
glycolytic flux by curcumin was associated with suppression of
mitochondrial oxidative respiration. Extracellular flux analysis of
MCF-7 cells demonstrated that treatment with 30 µM curcumin
for 6 h caused a significant reduction of basal OCR to 67.5±1.4% of
untreated controls (Fig. 2C).
Inhibition of ATP synthase with oligomycin reduced OCR of control
and curcumin-treated cells to similar levels, consistent with
comparable amounts of proton leakage. Uncoupling of the proton
gradient with FCCP caused a substantial augmentation of OCR for
control cells but not for curcumin-treated cells. This indicates
that maximal mitochondrial respiration is markedly suppressed by
curcumin (Fig. 2C). Mean OCR over 6
h following treatment with curcumin was decreased to 67.6±0.4% of
that following addition of vehicle (Fig. 2C).
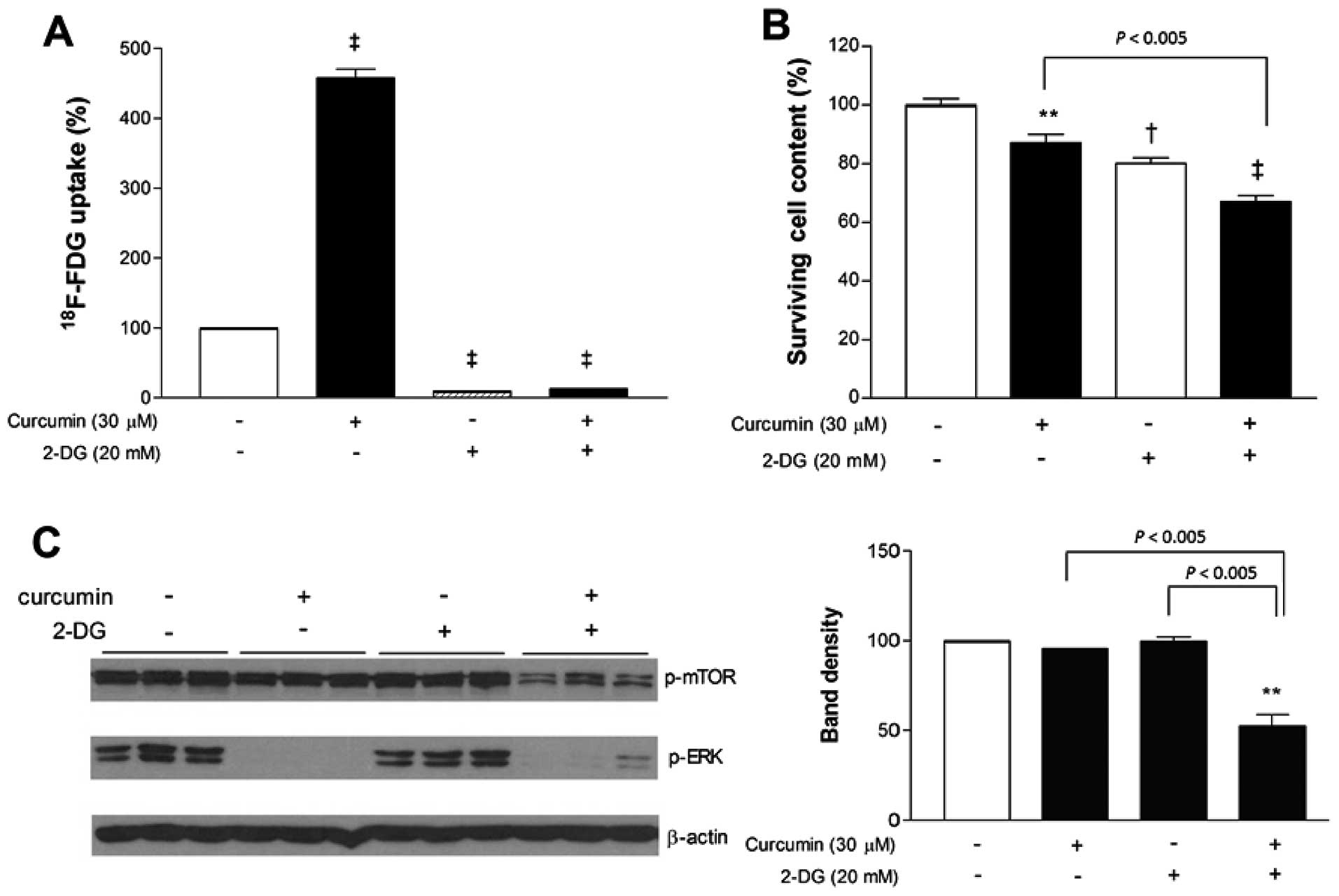
Glycolysis inhibition with 2-DG
potentiates the anticancer effect of curcumin
Given the shift toward glycolytic metabolism induced
by curcumin, we next investigated whether additional blocking of
glycolysis would potentiate its anticancer effect. 2-DG was first
confirmed, as expected, to markedly diminished MCF-7 cell
18F-FDG uptake. When used in combination, 2-DG also
completely blocked the ability of curcumin to stimulate
18F-FDG uptake. Hence, in the presence of 2-DG,
18F-FDG uptake was substantially reduced to 20.9±1.3% of
untreated controls cells even when curcumin was added (Fig. 3A).
When antitumor effects were examined, curcumin and
2-DG added separately caused only mild reductions of MCF-7 cell
content to 87.1±2.9 and 80.0±2.0% of controls, respectively. In
comparison, co-treatment with curcumin and 2-DG reduced cell
survival more efficiently to 67.1±1.9% of controls (Fig. 3B). We further evaluated how MAP
kinase and mTOR signaling are affected by treatment. Immunoblotting
revealed that curcumin, either alone or in combination with 2-DG,
completely abrogated ERK activation (Fig. 3C). Activation of mTOR was not
influenced by curcumin or 2-DG alone, but was significantly
decreased to 52.5±11.1% of untreated control level when the two
agents were combined (Fig. 3C).
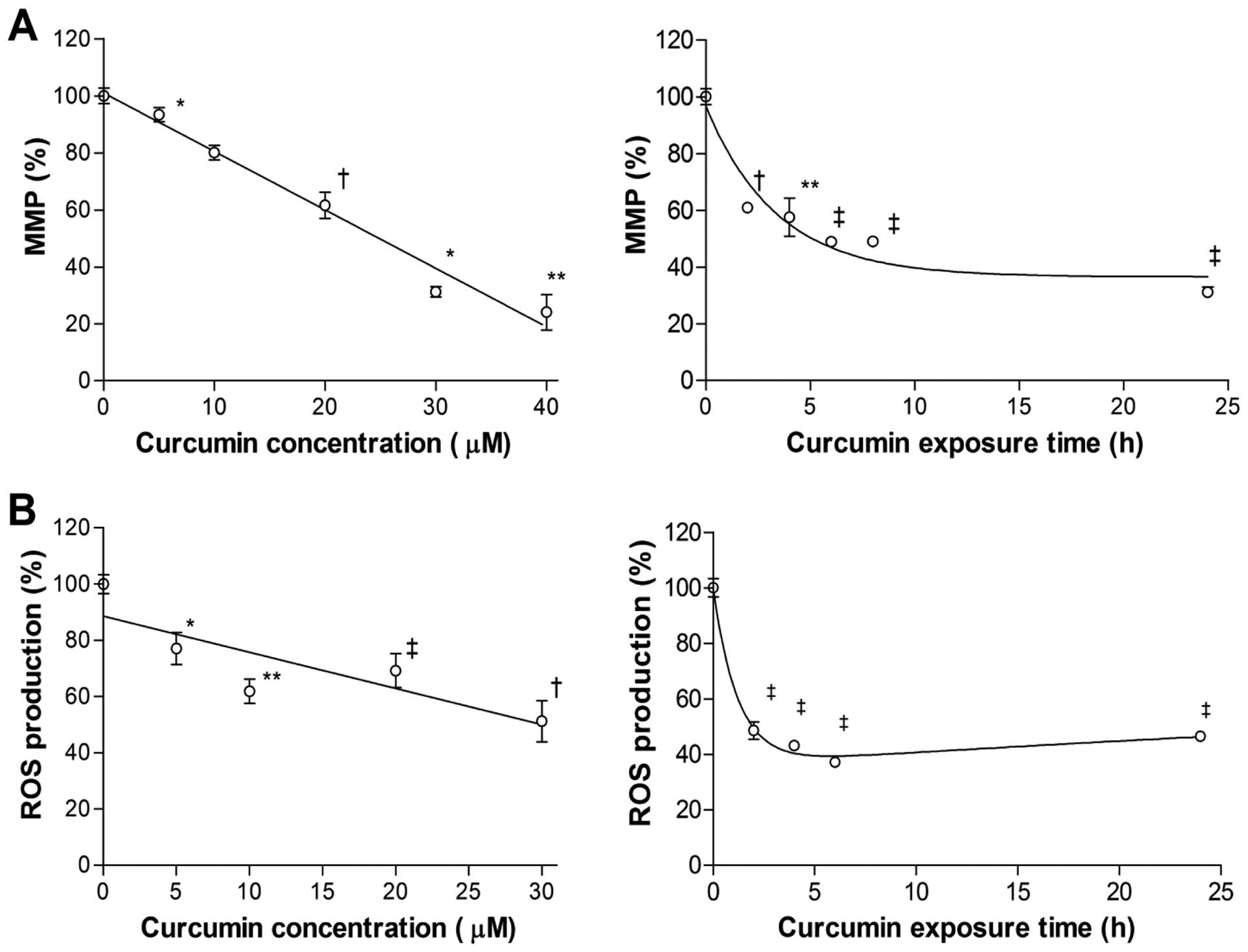
Curcumin depolarizes cancer cell MMP and
reduces intracellular ROS generation
To investigate how curcumin affects mitochondrial
function, we measured MMP, which is critically involved in
maintaining the mitochondrial respiratory chain and in avoidance of
cell death. The results showed that exposure to curcumin for 24 h
decreased MMP level of MCF-7 cells, in a manner inversely
correlating to the applied dose. With a concentration of 30
µM, MMP dropped to 31.2±3.1% of control level (Fig. 4A). The reduction of MMP began as
soon as 2 h of treatment and continued to decrease further over 24
h (Fig. 4A).
Intracellular ROS of MCF-7 cells were also
dose-dependently reduced by curcumin, and the pattern was similar
to the decrease of MMP. Treatment with 30 µM curcumin for 24
h reduced ROS level to 52.2±14.5% of controls (Fig. 4B). Time course experiments disclosed
a rapid decline of ROS level to 48.6±6.2% of controls by 2 h
(Fig. 4B). ROS decreased slightly
further to 37.1±3.2% of controls by 6 h, but slightly recovered
thereafter to 46.5±3.4% of controls by 24 h (Fig. 4B).
Blocking the anti-oxidant property of
curcumin markedly potentiates its anticancer effect
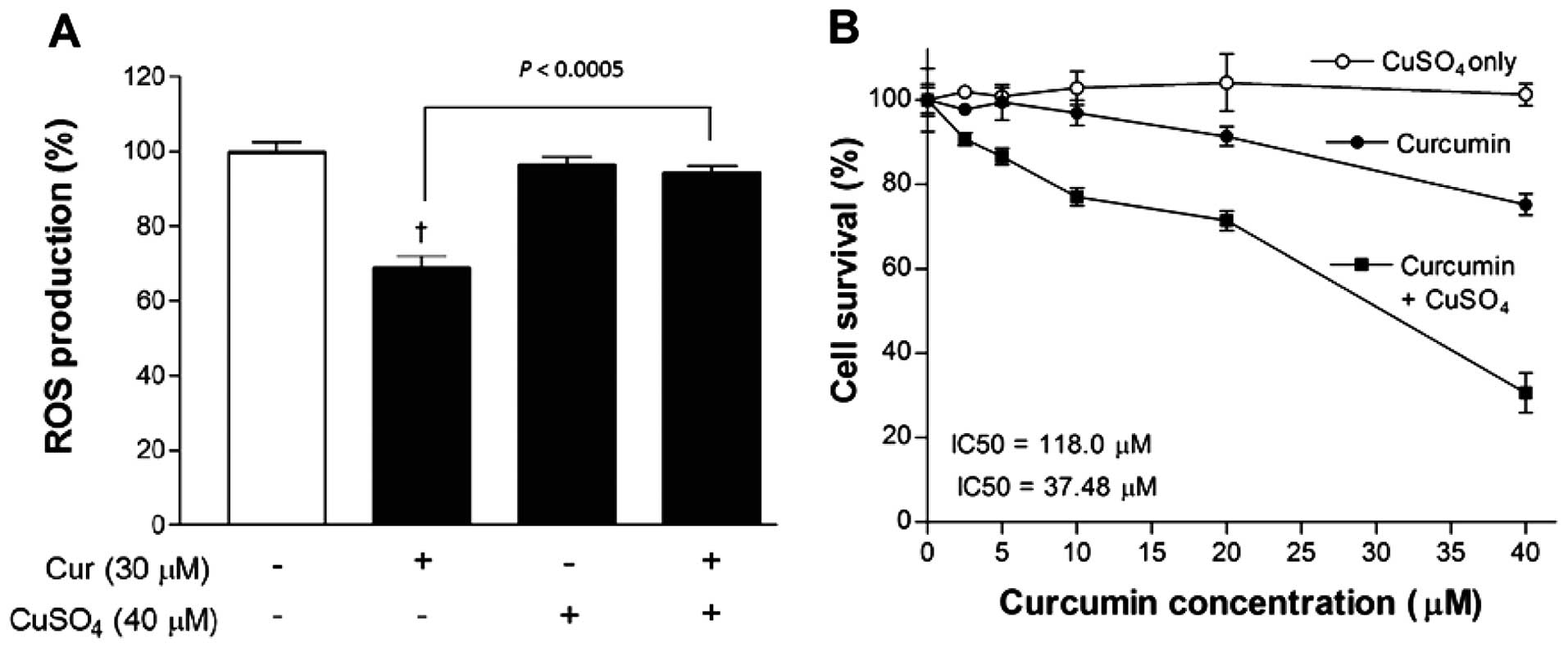
Finally, given the ROS-lowering property of curcumin
in the literature as well as in our experimental data, we evaluated
whether suppressing the anti-oxidant property of curcumin could
enhance its anticancer effect. This was achieved by adding 40
µM CuSO4, which is reported to neutralize the
anti-oxidant domains of curcumin. As a result, ROS level that was
significantly reduced by 30 µM curcumin alone, was recovered
to control levels in the presence of copper (Fig. 5A).
SRB assays showed that CuSO4 alone caused
no significant influence on MCF-7 cell survival in concentrations
of up to 40 µM. However, when combined together, 40
µM CuSO4 substantially potentiated the
dose-dependent reduction of cell survival induced by curcumin.
Hence, cell content that was reduced to 75.2±2.6% of controls by 40
µM curcumin alone was further reduced to 30.6±8.2% of
controls in the presence of 40 µM CuSO4, pointing
to a synergistic anticancer effect. Non-linear analysis of the
dose-response curve showed IC50 values of 118.0 and 37.5
µM for curcumin in the absence and presence of
CuSO4, respectively (Fig.
5B).
Discussion
The present study study demonstrates that curcumin
dose-dependently stimulates 18F-FDG uptake in MCF-7
breast cancer cells by shifting glucose metabolism toward
glycolytic flux. This response is accompanied by a suppression of
mitochondrial oxidative respiration and reduction of ROS
production.
Curcumin is well-recognized for its
anti-hyperglycemic effect through stimulation of glucose uptake in
muscle tissue. However, little is known regarding its influence on
glucose handling of malignant cells. Since curcumin is
well-recognized to exert anti-proliferative and apoptotic effects
on breast cancer cells (17), we
were particularly interested in how glucose metabolism and
mitochondrial function of these cells are affected. Our results
revealed that curcumin substantially enhances glucose uptake of
both MCF-7 and CT26 cells, but MCF-7 cells showed a more robust
dose-dependent response. As these two cell types are divergent in
genetic makeup and biological property, it is difficult to point to
a single reason explaining the different dose-responses of glucose
uptake by curcumin. Nonetheless, this finding and our interest in
breast cancer led us to select MCF-7 cells for further experiments.
Although this study did not include normal cells, a previous study
showed that curcumin and derivatives had minimal cytotoxic effects
on normal mammary epithelial MCF-10A cells (18). In MCF-7 cells, the metabolic
response began within a couple of hours of exposure, and was linked
to stimulation of glycolytic flux, as evidenced by augmentations of
lactate production and mitochondrial hexokinase activity. The
significant reduction of OCR accompanying this response indicates a
suppression of mitochondrial oxidative respiration. Curcumin
further caused a marked blunting of the normal OCR response to
FCCP, consistent with a severe reduction of maximal respiratory
capacity. Tumor cells need to balance their metabolism in a manner
that maintains a supply of energy production sufficient for
continued growth and survival. Measurements of intracellular ATP
would have strengthened our case but this was not performed in the
present study. Taken together, our stimulation of glycolytic flux
by curcumin appears to be a compensatory response of cancer cells
attempting to maintain energy homeostasis in the face of
mitochondrial dysfunction.
The dependence of malignant cells on a shift of
energy metabolism to glycolytic flux to adapt to curcumin-induced
suppression of mitochondrial respiration could provide a potential
target for enhanced therapeutic efficacy. We therefore tested the
effect of 2-DG, a glucose analogue that is taken up by transporters
and competitively inhibits glycolysis at the phosphoglucoisomerase
level (19). The results showed
that stimulation of glycolysis by curcumin was completely blocked
by 2-DG. Importantly, this had a clear additive effect on reducing
cell survival compared to that achieved by curcumin alone. This
finding is reminiscent of a recent observation that mitochondria
targeted cationic agents reduced cancer cell OCR and synergized
with 2-DG to inhibit cancer cell proliferation and survival
(20). Curcumin has the potential
to directly or indirectly influence the biological activity of a
number of signaling molecules, and its anti-proliferative effect on
cancer cells may involve modulation of MAP kinase (21,22)
and mTOR pathways (23,24). We therefore examined these pathways
by western blotting, and found that curcumin near-completely
suppressed ERK activation either alone or in combination with 2-DG.
This finding is consistent with previous reports that link
curcumin-mediated anti-proliferative actions to suppression of ERK
activity. Guo et al (21)
found in THP-1 acute monocytic leukemia cells that curcumin
inhibited proliferation and induced apoptosis by blocking
RAF/MEK/ERK and AKT/mTOR pathways. Furthermore, Xie et al
(22) observed that curcumin
inhibited nasopharyngeal carcinoma cell proliferation by reducing
ERK1/2 expression. Interestingly, mTOR activation was unaffected by
curcumin or 2-DG alone, but was significantly reduced by their
combination. Curcumin has previously been shown to inhibit
proliferation of colon cancer (23)
and rhabdomyosarcoma cells (24) by
mediating the inhibition of mTOR signaling. Furthermore, limiting
energy availability of breast cancer cells by 2-DG has been shown
to be accompanied by inhibition of mTOR activation (25). Taken together, our results suggest
that blockade of mTOR signaling in cancer cells confronting
mitochondrial dysfunction may contribute to the greater
anti-proliferative effect observed by the combination of curcumin
and 2-DG.
In addition to suppressing mitochondrial oxidative
respiration, curcumin caused a dose-dependent decline of MMP that
occurred within hours of treatment. A large amount of evidence
indicates that mitochondrial are involved in the therapeutic
properties of curcumin (3,26). When mitochondria are damaged, there
is an increase of proton leak that leads to diminished MMP, which
in turn is an early event in the apoptotic process. Curcumin has
previously been shown to exert depolarization and cause a collapse
of MMP in hepatocellular cancer cells (27). Thus, the decline of MMP observed in
our study supports a close link between the anticancer effect of
curcumin and mitochondrial dysfunction.
The decrease of MMP by curcumin in our results was
accompanied by dose- and time-dependent reductions of intracellular
ROS. Mitochondria are a main source of cellular ROS, which is
produced by electrons leaked from the mitochondrial respiratory
chain during oxidative phosphorylation (8). It is generally accepted that ROS
production has a positive relation with MMP level, and a decrease
in MMP can reduce ROS production (28). Our observation of lowered ROS
accompanying a decrease of MMP is therefore consistent with this
notion. Nevertheless, the finding may seem puzzling given that
elevated ROS plays a major role in the therapeutic effect of many
anticancer agents (29).
Furthermore, several lines of evidence indicate that it is the
pro-oxidant, rather than their anti-oxidant, action of polyphenolic
compounds that mediate their anticancer properties. It should be
noted that curcumin is an excellent ROS scavenger that is bestowed
with strong anti-oxidant activity under many circumstances. In
fact, there are reports that cancer cells treated with curcumin
display an anti-oxidant response that reduces ROS (11). Therefore, curcumin may have the
capacity to act as either an anti-oxidant or a pro-oxidant in
cancer cells, depending on the condition.
Curcumin can exhibit bifunctional anti-oxidant
properties related to its ability to directly react with ROS and to
indirectly induce expression of anti-oxidant proteins. Part of the
curcumin molecule acts as chelator of positively charged metals,
which play an important role in free radical trapping activity
(2). A factor thought to play a
major role in the exhibition of either anti-oxidant or pro-oxidant
properties by curcumin is binding of metal ions. Based on our
finding that ROS was lowered by curcumin, we examined whether
addition of Cu2+ could enhance the pro-oxidant property,
and thereby the anticancer effect, of the compound. The results
revealed that when MCF-7 cells were simultaneously treated with
CuSO4, cell content was more effectively decreased than
by curcumin alone. This finding is consistent with a previous
report of potentiated curcumin-induced cancer cell cytotoxicity by
transient metals such as Cu2+ (30,31).
Curcumin is composed of two hydrophobic phenyl domains connected by
a β-diketone moiety, which acts as a binding site for transient
metals (14). Copper, an essential
trace metal with many physiological and pathological activities, is
one of the most redox-active metal ions in living cells. Curcumin
and Cu2+ have strong interactions, and their binding
modifies the physico-chemical properties of the drug. In fact,
mobilization of endogenous copper has been proposed to stimulate
pro-oxidant action and contribute to the cytotoxic effects of
several polyphenolic compounds against cancer (16).
In our results, intracellular ROS was no longer
reduced by curcumin when Cu2+ was present. Similarly, a
recent study showed that curcumin-induced cancer cell apoptosis was
increased by Cu2+ through ROS generation, whereas it was
blocked by anti-oxidants (31).
These findings are consistent with the notion that, despite its
anti-oxidant properties, curcumin can exhibit pro-oxidant effects
in the presence of elevated Cu2+ (32). Indeed, increased ROS production was
implicated in the DNA damage caused by DNA-associated copper and
phenolic compounds (33).
ROS-mediated DNA degradation was also observed in cancer cells
treated with curcumin in the presence of Cu2+ (34,35).
These findings have led to the exploration of curcumin or synthetic
curcuminoids complexed with metal for enhanced antitumor activity
(36). While metal-curcumin
complexes may act either as anti-oxidants or pro-oxidants, curcumin
complexed with Cu2+ has been shown to exert pro-oxidant
effects by generating ROS at a high free copper level in a reducing
environment (37). Taken together,
our results suggest that CuSO4 blocks the direct ROS
scavenging effect of curcumin.
In conclusion, curcumin treatment of MCF-7 cancer
cells induces mitochondrial dysfunction that leads to a metabolic
shift to glycolytic flux and reduced ROS generation. This leads to
a significant increase in 18F-FDG uptake, suggesting the
possibility that it could serve as an imaging biomarker of tumor
response to curcumin. Furthermore, glycolysis inhibition with 2-DG
and suppression of the compound's anti-oxidant property with
Cu2+ may enhance the anticancer action of curcumin.
Acknowledgments
The present study was supported by the Basic Science
Research Program through the National Research Foundation of Korea
(NRF) funded by the Ministry of Science, ICT & Future Planning
(grant no. 2014050612).
References
|
1
|
Anand P, Sundaram C, Jhurani S,
Kunnumakkara AB and Aggarwal BB: Curcumin and cancer: An 'old-age'
disease with an 'age-old' solution. Cancer Lett. 267:133–164. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Trujillo J, Granados-Castro LF, Zazueta C,
Andérica-Romero AC, Chirino YI and Pedraza-Chaverrí J: Mitochondria
as a target in the therapeutic properties of curcumin. Arch Pharm
(Weinheim). 347:873–884. 2014. View Article : Google Scholar
|
|
3
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhao Y, Butler EB and Tan M: Targeting
cellular metabolism to improve cancer therapeutics. Cell Death Dis.
4:e5322013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Larson SM and Schwartz LH:
18F-FDG PET as a candidate for 'qualified biomarker':
Functional assessment of treatment response in oncology. J Nucl
Med. 47:901–903. 2006.PubMed/NCBI
|
|
6
|
Sullivan LB and Chandel NS: Mitochondrial
reactive oxygen species and cancer. Cancer Metab. 2:172014.
View Article : Google Scholar
|
|
7
|
Cheng TC, Lin CS, Hsu CC, Chen LJ, Cheng
KC and Cheng JT: Activation of muscarinic M-1 cholinoceptors by
curcumin to increase glucose uptake into skeletal muscle isolated
from Wistar rats. Neurosci Lett. 465:238–241. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kim JH, Park JM, Kim EK, Lee JO, Lee SK,
Jung JH, You GY, Park SH, Suh PG and Kim HS: Curcumin stimulates
glucose uptake through AMPK-p38 MAPK pathways in L6 myotube cells.
J Cell Physiol. 223:771–778. 2010.PubMed/NCBI
|
|
9
|
Hail N Jr: Mitochondrial reactive oxygen
species affect sensitivity to curcumin-induced apoptosis. Free
Radic Biol Med. 44:1382–1393. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Su CC, Lin JG, Li TM, Chung JG, Yang JS,
Ip SW, Lin WC and Chen GW: Curcumin-induced apoptosis of human
colon cancer colo 205 cells through the production of ROS,
Ca2+ and the activation of caspase-3. Anticancer Res.
26(6B): 4379–4389. 2006.
|
|
11
|
Ak T and Gülçin I: Antioxidant and radical
scavenging properties of curcumin. Chem Biol Interact. 174:27–37.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kunwar A, Jayakumar S, Srivastava AK and
Priyadarsini KI: Dimethoxycurcumin-induced cell death in human
breast carcinoma MCF7 cells: Evidence for pro-oxidant activity,
mitochondrial dysfunction, and apoptosis. Arch Toxicol. 86:603–614.
2012. View Article : Google Scholar
|
|
13
|
Li YB, Gao JL, Zhong ZF, Hoi PM, Lee SM
and Wang YT: Bisdemethoxycurcumin suppresses MCF-7 cells
proliferation by inducing ROS accumulation and modulating
senescence-related pathways. Pharmacol Rep. 65:700–709. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Priyadarsini KI: The chemistry of
curcumin: From extraction to therapeutic agent. Molecules.
19:20091–20112. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Verma SP and Goldin BR: Copper modulates
activities of genistein, nitric oxide, and curcumin in breast tumor
cells. Biochem Biophys Res Commun. 310:104–108. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hadi SM, Asad SF, Singh S and Ahmad A:
Putative mechanism for anticancer and apoptosis-inducing properties
of plant-derived polyphenolic compounds. IUBMB Life. 50:167–171.
2000. View Article : Google Scholar
|
|
17
|
Liu D and Chen Z: The effect of curcumin
on breast cancer cells. J Breast Cancer. 16:133–137. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Reddy CA, Somepalli V, Golakoti T,
Kanugula AK, Karnewar S, Rajendiran K, Vasagiri N, Prabhakar S,
Kuppusamy P, Kotamraju S, et al: Mitochondrial-targeted
curcuminoids: A strategy to enhance bioavailability and anticancer
efficacy of curcumin. PLoS One. 9:e893512014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wick AN, Drury DR, Nakada HI and Wolfe JB:
Localization of the primary metabolic block produced by
2-deoxyglucose. J Biol Chem. 224:963–969. 1957.PubMed/NCBI
|
|
20
|
Cheng G, Zielonka J, McAllister D, Hardy
M, Ouari O, Joseph J, Dwinell MB and Kalyanaraman B:
Antiproliferative effects of mitochondria-targeted cationic
antioxidants and analogs: Role of mitochondrial bioenergetics and
energy-sensing mechanism. Cancer Lett. 365:96–106. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guo Y, Shan Q, Gong Y, Lin J, Shi F, Shi R
and Yang X: Curcumin induces apoptosis via simultaneously targeting
AKT/mTOR and RAF/MEK/ERK survival signaling pathways in human
leukemia THP-1 cells. Pharmazie. 69:229–233. 2014.PubMed/NCBI
|
|
22
|
Xie YQ, Wu XB and Tang SQ: Curcumin
treatment alters ERK-1/2 signaling in vitro and inhibits
nasopharyngeal carcinoma proliferation in mouse xenografts. Int J
Clin Exp Med. 7:108–114. 2014.PubMed/NCBI
|
|
23
|
Johnson SM, Gulhati P, Arrieta I, Wang X,
Uchida T, Gao T and Evers BM: Curcumin inhibits proliferation of
colorectal carcinoma by modulating Akt/mTOR signaling. Anticancer
Res. 29:3185–3190. 2009.PubMed/NCBI
|
|
24
|
Beevers CS, Li F, Liu L and Huang S:
Curcumin inhibits the mammalian target of rapamycin-mediated
signaling pathways in cancer cells. Int J Cancer. 119:757–764.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jiang W, Zhu Z and Thompson HJ: Modulation
of the activities of AMP-activated protein kinase, protein kinase
B, and mammalian target of rapamycin by limiting energy
availability with 2-deoxyglucose. Mol Carcinog. 47:616–628. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wu SH, Hang LW, Yang JS, Chen HY, Lin HY,
Chiang JH, Lu CC, Yang JL, Lai TY, Ko YC, et al: Curcumin induces
apoptosis in human non-small cell lung cancer NCI-H460 cells
through ER stress and caspase cascade- and mitochondria-dependent
pathways. Anticancer Res. 30:2125–2133. 2010.PubMed/NCBI
|
|
27
|
Cao J, Liu Y, Jia L, Zhou HM, Kong Y, Yang
G, Jiang LP, Li QJ and Zhong LF: Curcumin induces apoptosis through
mitochondrial hyperpolarization and mtDNA damage in human hepatoma
G2 cells. Free Radic Biol Med. 43:968–975. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Suski JM, Lebiedzinska M, Bonora M, Pinton
P, Duszynski J and Wieckowski MR: Relation between mitochondrial
membrane potential and ROS formation. Methods Mol Biol.
810:183–205. 2012. View Article : Google Scholar
|
|
29
|
Gorrini C, Harris IS and Mak TW:
Modulation of oxidative stress as an anticancer strategy. Nat Rev
Drug Discov. 12:931–947. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lou JR, Zhang XX, Zheng J and Ding WQ:
Transient metals enhance cytotoxicity of curcumin: Potential
involvement of the NF-kappaB and mTOR signaling pathways.
Anticancer Res. 30:3249–3255. 2010.PubMed/NCBI
|
|
31
|
Lu JJ, Cai YJ and Ding J: The short-time
treatment with curcumin sufficiently decreases cell viability,
induces apoptosis and copper enhances these effects in
multidrug-resistant K562/A02 cells. Mol Cell Biochem. 360:253–260.
2012. View Article : Google Scholar
|
|
32
|
Urbina-Cano P, Bobadilla-Morales L,
Ramírez-Herrera MA, Corona-Rivera JR, Mendoza-Magaña ML,
Troyo-Sanromán R and Corona-Rivera A: DNA damage in mouse
lymphocytes exposed to curcumin and copper. J Appl Genet.
47:377–382. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li Y and Trush MA: Reactive
oxygen-dependent DNA damage resulting from the oxidation of
phenolic compounds by a copper-redox cycle mechanism. Cancer Res.
54(Suppl): 1895s–1898s. 1994.PubMed/NCBI
|
|
34
|
Ahsan H and Hadi SM: Strand scission in
DNA induced by curcumin in the presence of Cu(II). Cancer Lett.
124:23–30. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ahsan H, Parveen N, Khan NU and Hadi SM:
Pro-oxidant, anti-oxidant and cleavage activities on DNA of
curcumin and its derivatives demethoxycurcumin and
bisdemethoxycurcumin. Chem Biol Interact. 121:161–175. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
John VD, Kuttan G and Krishnankutty K:
Anti-tumour studies of metal chelates of synthetic curcuminoids. J
Exp Clin Cancer Res. 21:219–224. 2002.PubMed/NCBI
|
|
37
|
Leung MH, Harada T and Kee TW: Delivery of
curcumin and medicinal effects of the copper(II)-curcumin
complexes. Curr Pharm Des. 19:2070–2083. 2013.
|