Introduction
Previous studies have shown that the perturbation of
endoplasmic reticulum (ER) function causes ER stress, which has
been linked to numerous diseases. Yet, prolonged ER stress can
activate apoptotic pathways of damaged cells (1). ER stress is related to the
pathophysiology of cancer; therefore pharmacological interventions
that effectively enhance tumor cell death by activating ER stress
have attracted much attention for anticancer therapy (2,3).
Various anticancer agents, such as paclitaxel (4), farnesol (5) and polyphyllin D (6), can induce cancer cell apoptosis by ER
stress. Curcumin, a yellow compound, is isolated from the rhizome
of the herb Curcuma longa. Over the last half-century,
various bio-functions of curcumin have been revealed through
extensive research. A few clinical trials have demonstrated that
curcumin possesses anticancer effect, but was proposed as a more
native chemoprevention agent in pancreatic and colon cancer
(7). Various studies suggest that
curcumin may induce the apoptosis of several types of cancer cells
via ER stress (8–11). Curcumin was also reported to display
its pro-apoptotic effects by inducing ER stress in acute
promyelocytic leukemia (12),
non-small cell lung cancer H460 cells (13) and human liposarcoma cells (14).
However, the instability and poor metabolic property
of cucurmin have significantly limited its clinical application
(15). Therefore, in the past few
years, a series of curcumin analogs have been synthesized in our
laboratory, in order to enhance its metabolic stability and
pharmacological potency (1,16-22).
Given that the β-diketone moiety of curcumin may lead to its
instability and poor metabolic properties, by deleting this moiety,
we designed many mono-carbonyl analogs of curcumin and three of
them were discovered as the most efficient compounds against tumor
cells with enhanced stability. Their chemical structures were
respectively 06 [(1E,4E)-1,5-bis(2-bromophenyl)
penta-1,4-dien-3-one], 12 [(2E, 5E)-2,
5-bis(4-(3-(dimethylamino) propoxy)benzylidene)cyclopentanone], and
19
[(1E,4E)-1,5-bis(2,3-dimethoxyphenyl)penta-1,4-dien-3-one]
(17). To further develop new
analogs of curcumin which can enhance stability and anticancer
effects, we designed and screened novel double carbonyl analogs of
curcumin. In the present study, we identified a novel double
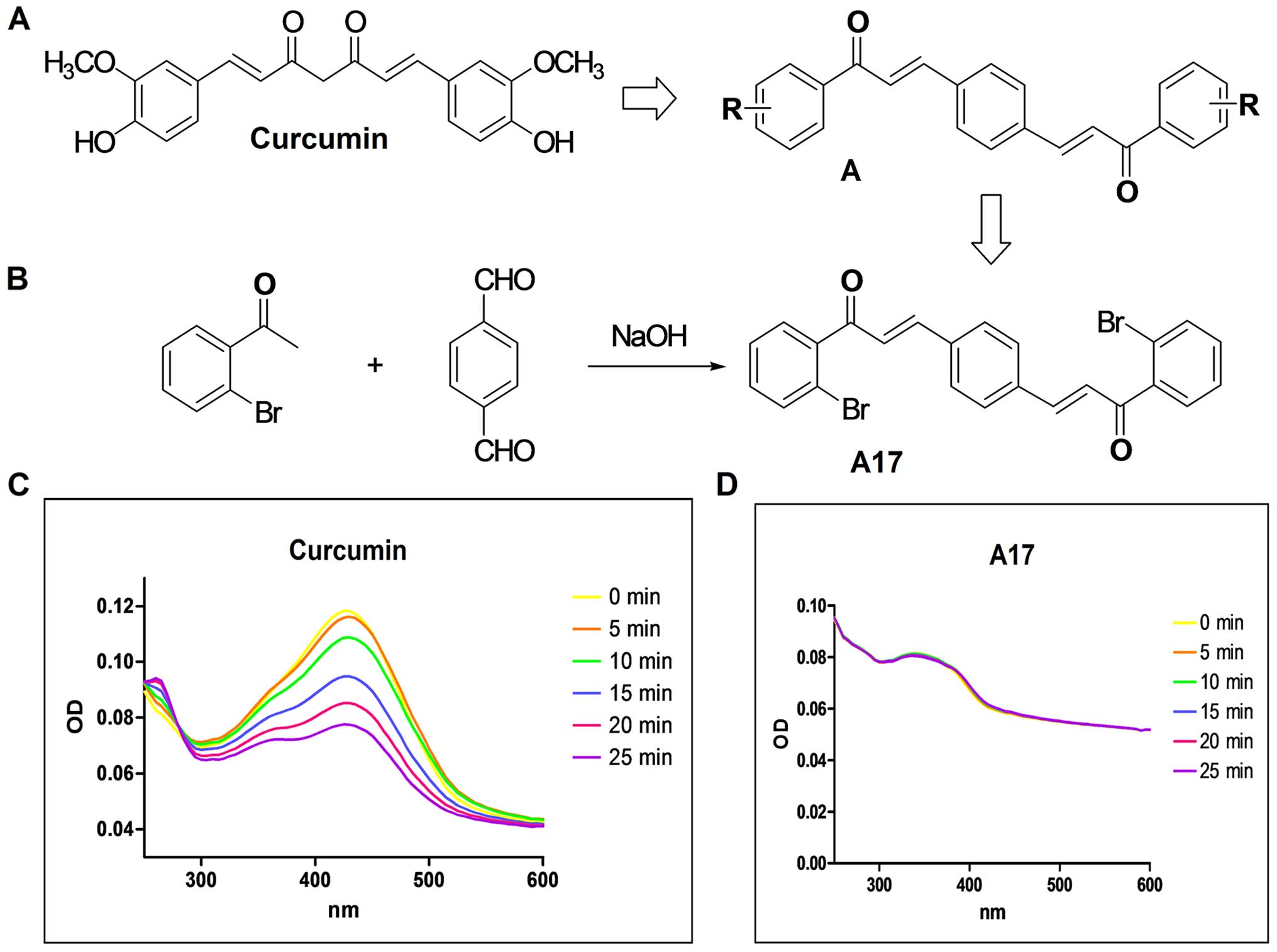
carbonyl analog of curcumin (named A17; Fig. 1), and described its stability and
antitumor activity. The results showed that A17 has better
stability and antitumor activity than curcumin.
Materials and methods
Chemicals and reagents
Curcumin was purchased from Sigma Chemical Co. (St.
Louis, MO, USA). As shown in Fig. 1A
and B, curcumin analog A17 was synthesized and recrystallized
by CH2C2/EtOH. Its structure was identified
by MS and 1H-NMR analyses and its purity (99.50%) was
determined using HPLC. The CellTiter-Glo kit was purchased from
Promega Corporation (Madison, WI, USA). FITC Annexin V Apoptosis
Detection kit I was purchased from BD Pharmingen (Franklin Lakes,
NJ, USA). Nitrocellulose membranes and enhanced chemiluminescence
reagents were obtained from Bio-Rad (Hercules, CA, USA). Antibodies
including anti-cleaved PARP, anti-pro-caspase-3, anti-Bcl-2,
anti-BAX, anti-GAPDH, anti-CHOP, anti-ATF-4, anti-XBP-1,
anti-GRP78, goat anti-mouse IgG-HRP, goat anti-rabbit IgG-HRP and
donkey anti-goat IgG-HRP were purchased from Santa Cruz
Biotechnology (Santa Cruz, CA, USA), and anti-cleaved caspase-3
antibody was purchased from Cell Signaling Technology (Danvers, MA,
USA). The Ambion RNAqueous kit was purchased from Applied
Biosystems Inc. (Foster City, CA, USA). All other reagents and
solvents used were of analytical grade.
Cell lines
Human lung carcinoma cell line NCI-H460 was
purchased from the American Type Culture Collection (ATCC;
Manassas, VA, USA). The B16-F10 (mouse B16-F10 malignant melanoma
cells), U251 (human glioma cell U251), and A549 cells were all
purchased from the Shanghai Institute of Biosciences and Cell
Resources Center (Chinese Academy of Sciences, Shanghai, China).
B16-F10 and U251 were cultured in RPMI-1640 medium, whereas
NCI-H460 and A549 were cultured in Dulbecco's modified Eagle's
medium (DMEM) (both from Invitrogen, Carlsbad, CA, USA) with
high-glucose media; the four types of cells were supplemented with
10% heat-inactivated fetal bovine serum (FBS) (Atlanta Biologicals
Inc., Lawrenceville, GA, USA) and 100 U/ml penicillin-streptomycin
(Mediatech, Inc., Manassas, VA, USA) and incubated at 37°C with 5%
CO2.
Stability test
To determine the optical density (OD) of A17 and
curcumin, 10 µl A17 or curcumin was added into each cuvette
and then the absorbance was detected at once from 200 to 600 nm. OD
was measured once every 5 min, and three independent experiments
were conducted.
Methyl thiazolyl tetrazolium (MTT)
assay
MTT assay was conducted according to the basic
specifications described by Ye et al (23), but with slight modification.
Briefly, B16-F10, U251, H460 and A549 cells (100 µl/well) at
the logarithmic phase were seeded and cultured in a 96-well plate
at 37°C. After 24 h, A17 was dissolved and diluted with dimethyl
sulfoxide (DMSO) to a final concentration of 60, 12, 2.4, 0.48 or
0.096 µg/ml, and directly added to the culture medium in a
96-well plate and incubated for 72 h before the MTT assay. Curcumin
was used as the positive control. MTT (5 mg/ml) dissolved in NaCl
solution (0.9%) was added to each well of the plate. After
incubation in a CO2 incubator for 3 h, the cells were
dissolved in 100 µl of DMSO and then analyzed in a
multi-well plate reader at 570 nm.
Cell viability assay
Freshly resuspended H460 cells were seeded into
96-well plates at 5×103 cells/well overnight, and then
the cells were treated with various concentrations of A17 or
curcumin. The compounds were dissolved in DMSO and added to the
culture medium (final concentration of 1.25, 2.5, 5, 10 and 15
µM) and incubation was carried out for 24 h in an incubator
at 37°C with 5% CO2. Cell viability was observed by
microscopy and evaluated with the CellTiter-Glo kit at different
time points. Absorbance was measured at 450 nm to determine the
number of viable cells.
Colony formation assay
H460 cells were placed into a 6-well plate at
1×103 cells/well and cultured overnight. After starved
cultivation for 24 h, the cells were treated for a week with 1 and
5 µM of A17, 5 µM of curcumin and DMSO (were used as
a control). The number of surviving colonies was counted after
staining with crystal violet solution and then photographed. Each
experiment was performed in triplicate wells 3 times.
Flow cytometric analysis
H460 cells were plated in a 6-mm plate for 6 h and
then treated with A17 (1, 5 and 10 µM), curcumin (10
µM) or DMSO for 24 h, respectively. In addition, the cells
were collected and stained with Annexin V and propidium iodide (PI)
for 30 min at 37°C when 100 mg/ml RNAse and 0.1% Triton X-100 were
present. Flow cytometric analysis was performed using FACSCalibur
(BD Biosciences, San Jose, CA, USA).
Western blot analysis
H460 cells were washed and collected with PBS after
treatment, and lysed in lysis buffer containing 50 mmol/l Tris-HCl
(pH 7.4), 150 mmol/l NaCl, 1% NP40 and 0.1% SDS. After the protein
concentration was determined by BCA protein assays, protein samples
were electrophoresed by 10% SDS-PAGE gel before proteins were
transferred to nitrocellulose membranes. Each membrane was blocked
with Tris-buffered saline (pH 7.6) containing 0.05% Tween-20 and 5%
non-fat milk for 1 h at room temperature. After being washed with
TBS 3 times, the nitrocellulose membrane was probed with the
specific primary antibody at 4°C overnight followed by incubation
with secondary antibodies conjugated with horseradish peroxidase
and visualized using enhanced chemiluminescence reagents.
Construction of lentiviral siRNA for
CHOP
To specifically target the nucleotides of CHOP,
lentiviral siRNA for CHOP was constructed according to the basic
specifications described by Liang et al (18). The sense sequence of the siRNA
cassettes was designed with the siRNA target finder (Ambion,
Austin, TX, USA), and the sequence of CHOP siRNA was
5′-GCAGGAAATCGAGCGCCTGAC-3′.
Statistical analysis
Student's t-test was employed to analyze the
differences between data sets. The data analysis was performed
using GraphPad Prism 4.0 (GraphPad, San Diego, CA, USA).
Differences were considered to indicate a statistically significant
result at p<0.05.
Results
Structures of A17 and curcumin and their
activity against human tumor cell lines
Fig. 1A and B show
the design and structural skeleton of the novel double carbonyl
analogs of curcumin (A17). According to the characteristic
absorption peak of A17 and curcumin in Fig. 1C and D, A17 is more stable than
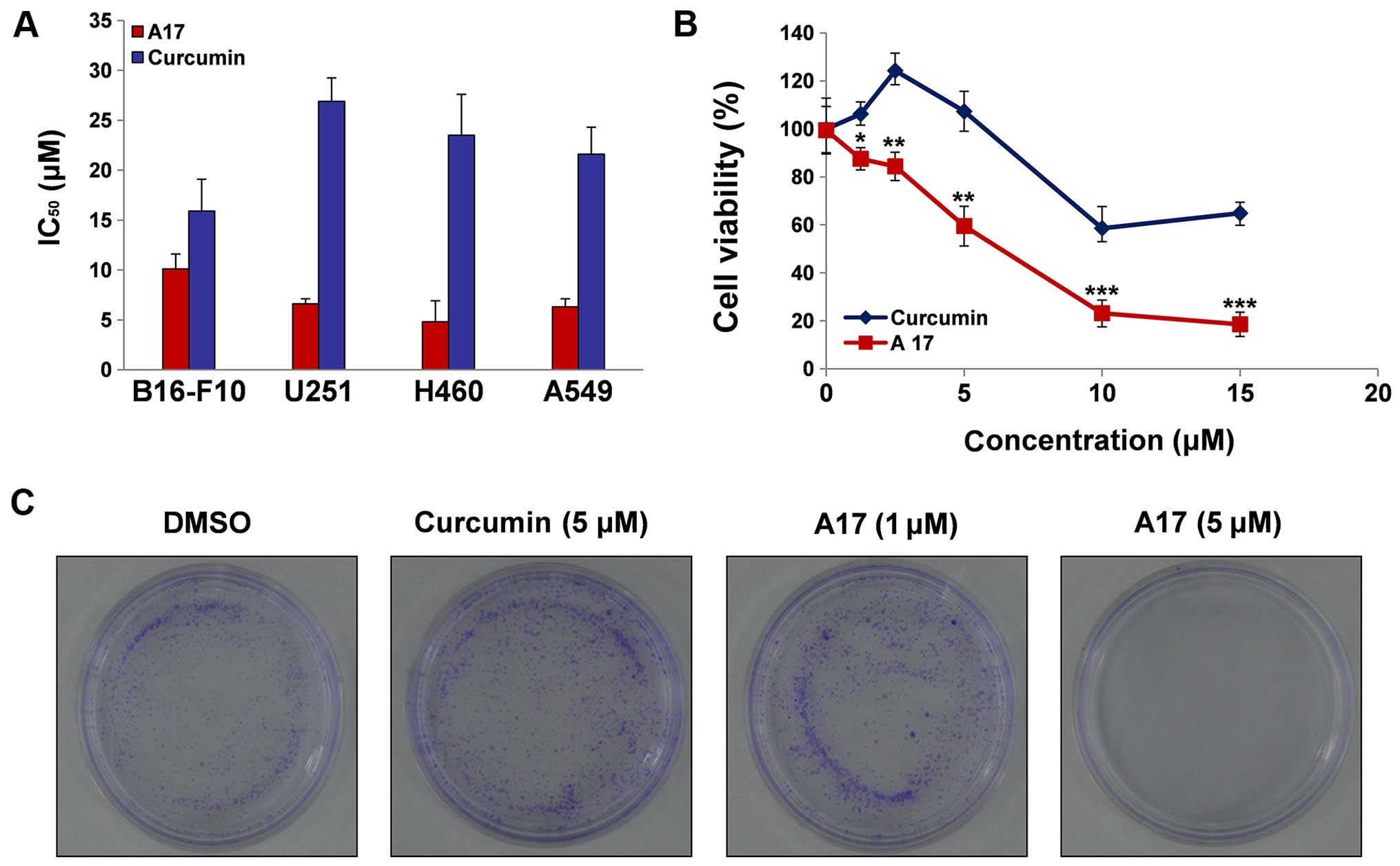
curcumin, and Fig. 2A shows that
A17 exhibited good antiproliferative effect against B16-F10, U251,
H460 and A549 cell lines, which were chosen for the present study.
IC50 values of A17 and curcumin against the above
mentioned cell lines were determined by an MTT assay (Fig. 2A). Curcumin showed cytotoxicity
against B16-F10, U251, H460 and A549 cells with IC50
values of 15.9±3.2, 26.9±23.4, 23.5±4.1 and 21.6±2.7 µM,
respectively; by contrast, A17 displayed stronger cytotoxicity than
curcumin, with IC50 values reaching 10.1±1.5, 6.6±0.1,
4.8±2.1 and 6.3±0.8 µM, respectively.
H460 cell death is induced by A17
After H460 cells were treated with A17 or curcumin
at different concentrations (0, 1.25, 2.5, 5, 10 and 15 µM)
for 24 h, cell viability was determined by the CellTiter-Glo kit
assay. A17 at different doses, induced H460 cell death more
effectively than curcumin at the same doses, as shown in Fig. 2B (p<0.01), and A17 ranging from
1.25 to 15 µM dose-dependently decreased cell survival. As
shown in Fig. 2B, after 24 h of
treatment with A17 or curcumin at 10 µM, A17 significantly
reduced H460 cell viability (22.71%) when compared with curcumin
(58.2%). Subsequently, the antiproliferative effect of H460 cells
was identified by colony formation assay. The results showed that,
compared with curcumin and DMSO, A17 significantly reduced the
number of colonies formed by the H460 cells at 5 µM
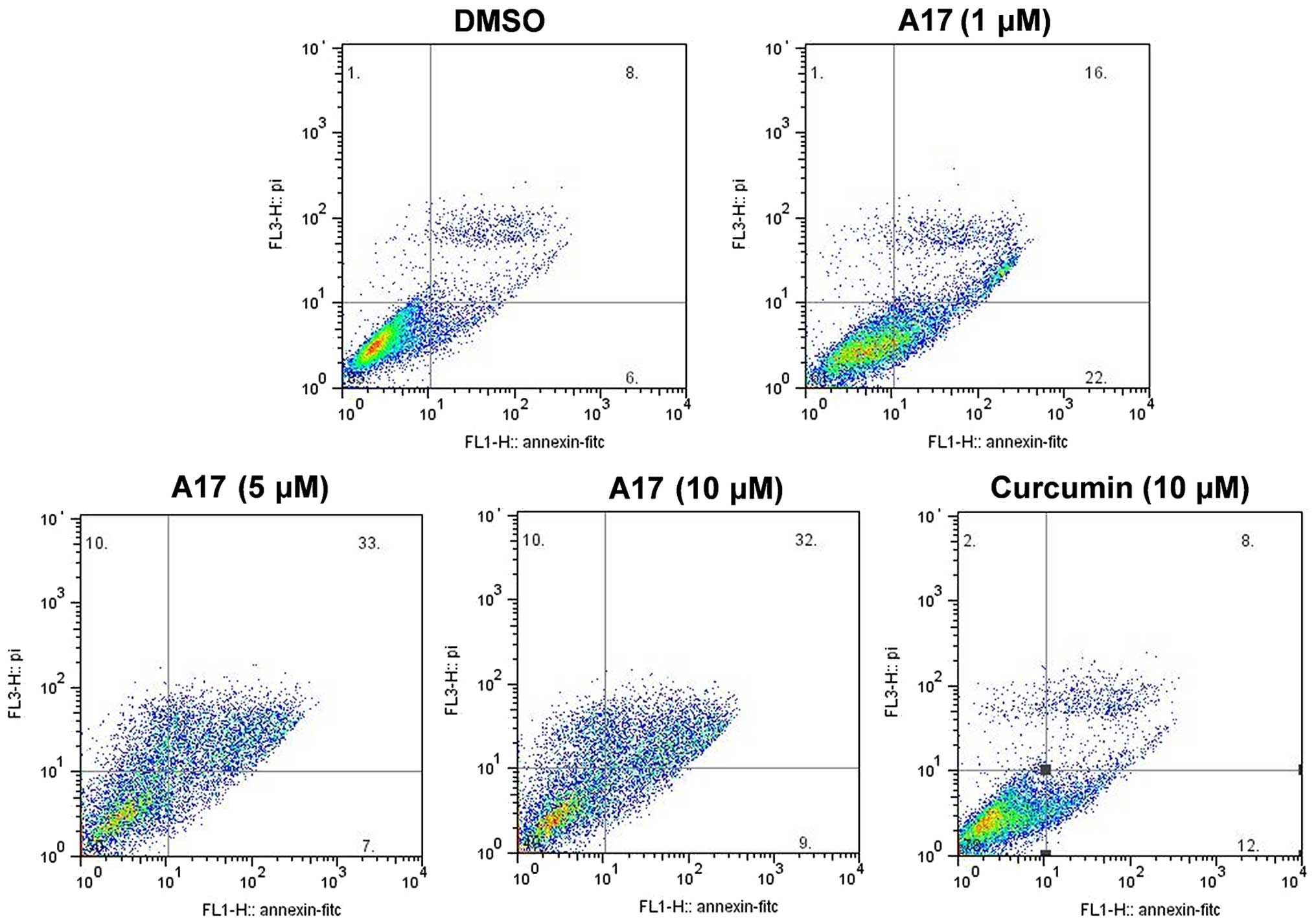
(Fig. 2C). Flow cytometric analysis
was used to assess the effects of A17 and curcumin on the induction
of apoptosis in H460 cells. Fig. 3
showed that A17 increased H460 apoptosis in a dose-dependent manner
after 6 h of treatment. After H460 cells were treated with A17 at 5
µM or curcumin at 10 µM, only a cell apoptosis rate
of 8% was detected in the curcumin group, whereas 33% H460 cell
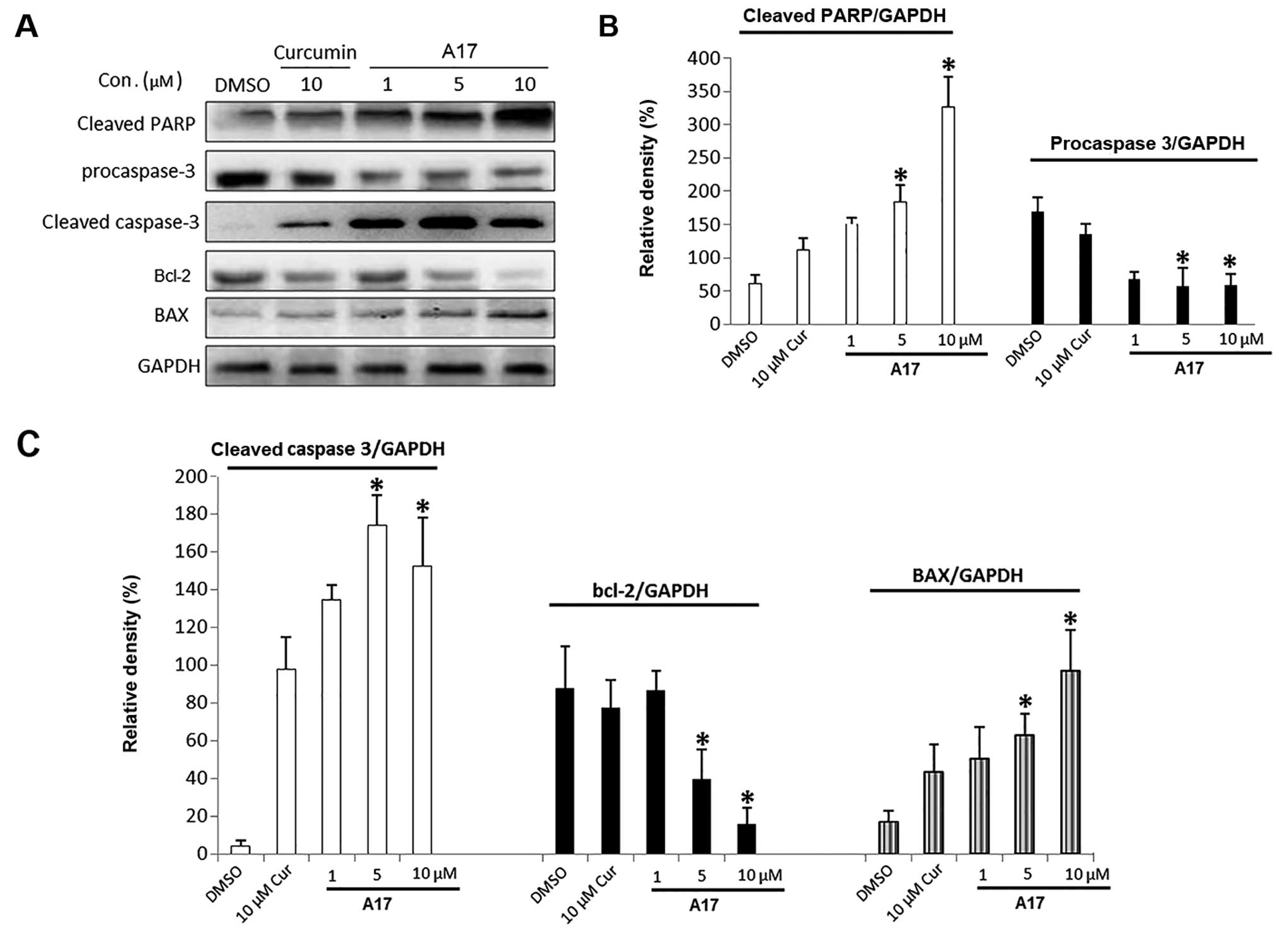
apoptosis was induced in the A17 group. We further investigated the
A17-induced H460 cell death by western blot analysis. As shown in
Fig. 4, cleaved PARP, procaspase-3,
cleaved caspase-3, Bcl-2 and BAX are apoptosis-related proteins.
A17 dose-dependently enhanced H460 cell apoptosis after 12–24 h of
treatment by increasing the expression of BAX, cleaved caspase-3,
and cleaved PARP while decreasing the expression of Bcl-2 and
procaspase-3.
A17 causes ER stress in the H460
cells
Glucose-regulated protein/immunoglobulin heavy
chain-binding protein (GRP78) is the gatekeeper to the activation
of ER stress (24). Thus, the
protein expression of GRP78 was measured after A17 treatment.
Fig. 5C and E show that the protein
expression of GRP78 was increased at 3 h following treatment with
A17 at 5 µM, whereas no significant increase was detected in
the curcumin-treated group (18).
In addition, western blot analysis revealed that the protein
expression of GRP78 increased in a time-dependent manner at 1–12 h
after the cells were treated with A17 at 5 µM. Thereafter,
we also examined the expression levels of the downstream X-box
binding protein 1 (XBP-1) and activating transcription factor 4
(ATF-4) in H460 cells treated with 5 µM A17 or curcumin
using western blot analysis. The time course results showed that
the treatment with A17 at 5 µM induced the expression of
ATF-4 (6 h after treatment) and XBP-1 (3 h after treatment) to peak
(Fig. 5C and D), whereas no
significant increase could be seen in the 5 µM
curcumin-treated H460 cells (18).
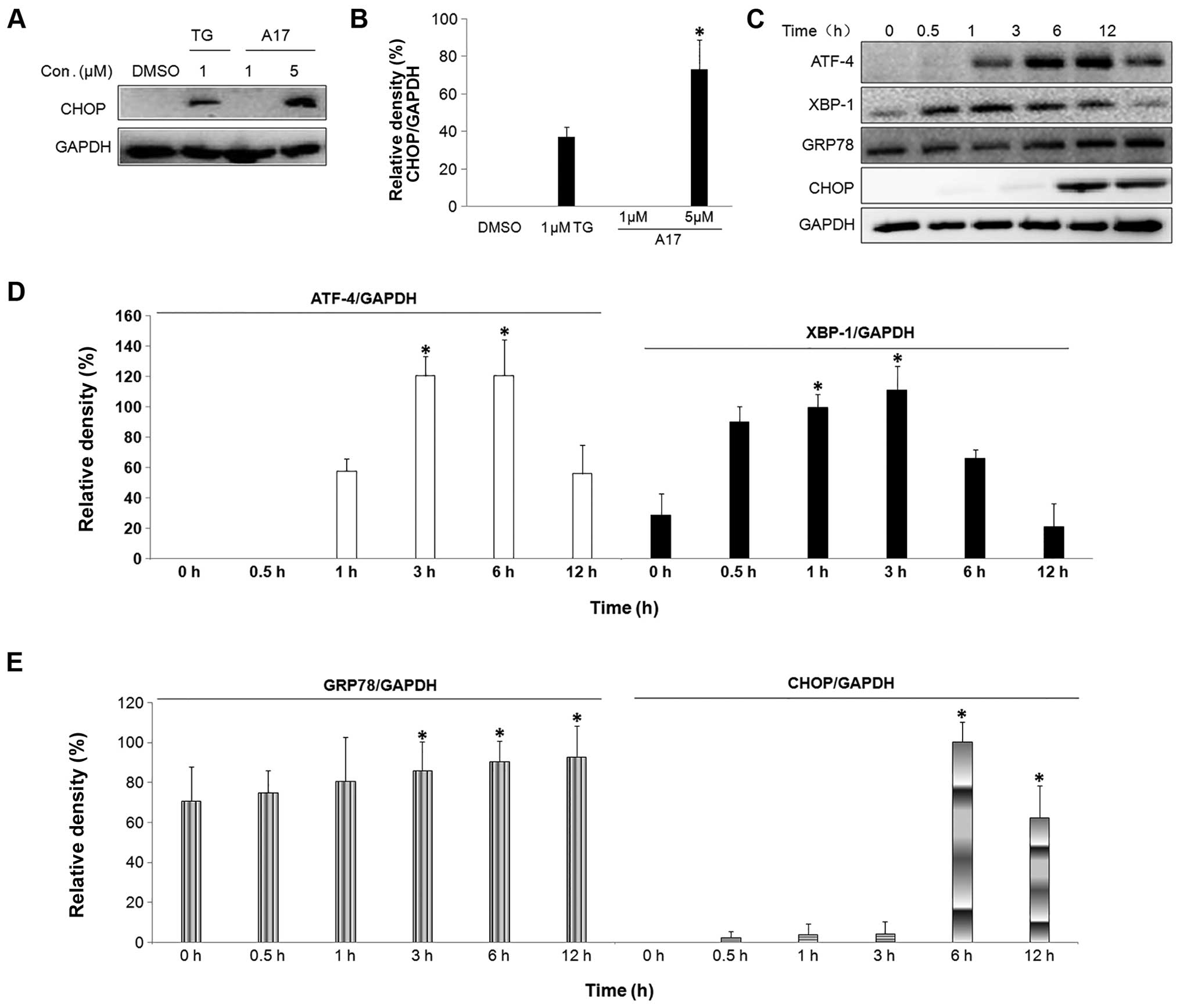 | Figure 5A17 induces GRP78, ATF-4, XBP-1 and
CHOP expression. (A) After H460 cells were treated with A17 at 1
and 5 µM for 6 h, CHOP expression was examined by western
blot analysis. GAPDH is shown as a control for equal loading. (B)
Western blot results from (A) were calculated and represented as
the percentage of the control; *p<0.02 vs. the DMSO
group. (C) Cells were treated with A17 at 5 µM for 0, 0.5,
1, 3, 6 and 12 h, and the expression of GRP78, ATF-4, XBP-1 and
CHOP was examined by western blot analysis. GAPDH is shown as a
control for equal loading. (D and E) Western blot results from (C)
were calculated and represented as the percentage of the control;
*p<0.02 vs. the 0 h group. |
CHOP induction may be most sensitive to ER stress
response, and CHOP is regarded as a marker of the commitment to ER
stress-induced apoptosis (25). The
transcription of the CHOP gene is induced by the unfolded protein
response (UPR) including ATF-4 and XBP-1 pathways in the process of
ER stress (26). In the present
study, we demonstrated that A17 obviously increased CHOP at the
protein level. Fig. 5A and B
indicated that A17 induced CHOP protein upregulation in a
dose-dependent manner. Similarly, CHOP expression was not
detectable in curcumin-treated cells. As shown in Fig. 5C and E, the CHOP protein expression
was noticeably increased after H460 cells were exposed to A17 for 6
and 12 h, and reached its peak at 12 h, but curcumin at 5 µM
had no effect on CHOP protein levels.
All upstream signals finally result in caspase
activation to finish the execution of ER stress-induced apoptosis
(27). As shown in Fig. 4, we tested the downstream events of
ER stress-mediated apoptosis after A17-induced ER stress. The
western blot analysis results showed that the cleavage of
procaspase-3 was detected after A17 treatment for 24 h. However,
caspase-3 was not able to be activated by curcumin at this
concentration. These results demonstrate that the caspase signal is
related to the ER stress-mediated apoptotic pathway induced by
A17.
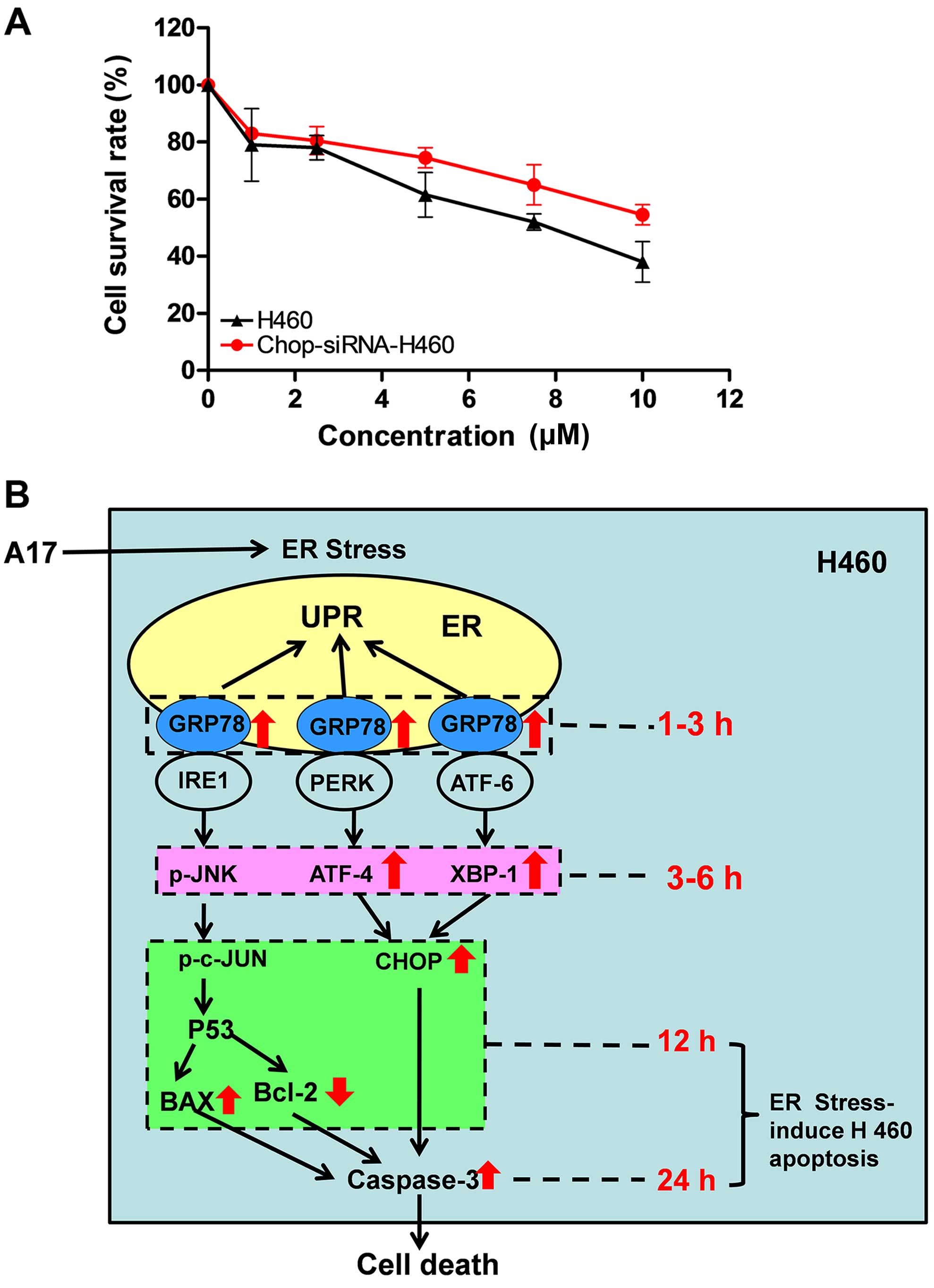
A17-induced H460 cell death is suppressed
by reducing CHOP expression
To further prove that ER stress plays an important
role in the process of A17-induced H460 cell apoptosis, we
constructed a lentiviral vector that contained an siRNA targeting
the CHOP gene and transiently transfected it into H460 cells. We
detected the titers by counting EGFP-expressing cells after 48 h
via fluorescence microscopy. As a result, we found that over 67% of
the H460 cells were transfected using the lentiviral siRNA. In
addition, to verify that A17-induced H460 cell death was suppressed
by reducing CHOP expression, CHOP siRNA-tranfected H460 cells were
treated using A17 at the indicated concentrations (0, 2, 4, 6, 8,
10 and 20 µM) for 24 h. Compared with the control group,
cell apoptosis induced by A17 was markedly decreased when CHOP
expression in H460 cells was silenced (Fig. 6A). CHOP is a key protein in ER
stress, thus, these results suggest that A17-induced cell apoptosis
is at least partially mediated by the ER stress pathway.
Discussion
Curcumin, also known as diferuloylmethane, is
extracted from the root of Curcuma longa and has been widely
used as a therapeutic agent for several different types of cancers
in clinical trials (19). However,
numerous studies indicate that its poor bioavailability and
pharmacokinetic profiles have limited its application in anticancer
therapy (15,28,29).
The chemical modification of curcumin is an effective way to obtain
potential analogs with enhanced bioavailability and antitumor
activity (16,18,19,30–34).
Given that the β-diketone moiety of curcumin may cause its
instability and poor metabolic property, we previously deleted the
moiety to design a series of mono-carbonyl analogs of curcumin
(17). Through the analysis of
cytotoxic screening experiment, we found that 06, 12 and 19 of
curcumin analogs showed enhanced toxic effects against human lung
cancer H460 cells, and the IC50 values were <10
µM (18). To further improve
the stability and anticancer effect of curcumin, we designed a
double carbonyl analog of curcumin (named A17; Fig. 1A and B). A17 had better stability
and improved pharmacokinetic profiles than curcumin. Subsequently,
an MTT assay for A17 against human lung cancer H460 cells was
performed, and A17 was a more efficient compound than curcumin
against H460 cells with IC50 <5 µM, thereby
suggesting that A17 also has much higher activity than curcumin
(Fig. 2A).
In rapidly growing tumors, when cancer cells are
continuously exposed to harsh microenvironments of hypoxia and
nutritional deprivation, this leads to ER stress (35). The UPR pathways of most normal cells
remain quiescent, since they do not undergo an active ̔stress̓
response. The difference between normal cells and tumor cells
allows for the agents that target ER stress to implement the
specificity of cancer therapy. Some data from clinical trials
support the hypothesis that the regulation of ER stress may raise
the curative effect of chemotherapy drugs and that ER
stress-mediated apoptotic pathways may provide new targets for
antitumor drugs (21). Previous
studies have shown that curcumin induces the apoptosis of human
non-small cell lung cancer (NSCLC) H460 cells through ER stress and
mitochondria-dependent pathways (13,27).
Curcumin at >30 µM caused cytotoxicity in H460 cells via
the induction of apoptosis, and apoptotic cell death was associated
with the ER stress signaling pathway (18). In the present study, to develop
novel antitumor agents via the ER stress-mediated mechanism, we
tested the ability of A17 to increase the expression of CHOP, which
is a hallmark of the ER stress-mediated apoptotic pathway. Fig. 5A shows that A17 induced CHOP
overexpression in the H460 cells, thereby suggesting that A17 may
have a different mechanism of antitumor action from that of the
other analogs. Therefore, we revealed the exact molecular mechanism
by which A17 causes ER stress-mediated cell apoptosis, as explained
below. GRP78 is a member of a glucose-regulated protein family and
is the gatekeeper of the activation of ER stress (24). ER has three transmembrane receptors:
the PKR-like ER kinase (PERK), ATF-6 and IRE1. They usually combine
with GRP78 and form a complex in the inactive state, respectively.
However, in the process of UPR, once GRP 78 is combined with
misfolded proteins, these transmembrane receptors are released from
complexes, and then they monitor their downstream molecules and
regulate ER stress (36). In the
present study, after H460 cells were treated with A17 for 3 h,
GRP78 protein expression was increased (Fig. 5C and E). Subsequently, we detected
the A17-induced activation of PERK, ATF-6, and IRE1 by monitoring
their downstream molecules: ATF-4, XBP-1, BAX and Bcl-2 (namely,
p-JNK↑ → p-c-JUN↑ → P53↑ → BAX↑ and Bcl-2↓) at 6–12 h after A17
treatment, respectively (Figs. 4A and
C; and 5C and D). These data
indicate that the ER stress-dependent apoptotic pathway was
triggered by A17.
After H460 cells were treated with A17 for 12 h,
CHOP expression was also found to increase (Fig. 5A–C and E). In fact, CHOP is the
common downstream activated protein in two signaling pathways of
PERK and ATF-6; ATF-4 and XBP-1 activation lead to the upregulation
of CHOP gene expression, thus triggering an ER stress-specific
cascade for apoptosis. In addition, several members of the Bcl-2
family proteins, regarded as main regulators in the
mitochondrial-mediated apoptotic pathway, are important in the ER
stress downstream pathway (26,38,39).
The phosphorylation of c-Jun activates the transcription factor P53
and the latter regulates Bcl-2 family proteins (26,38,39).
As shown in Fig. 4A and C, after
A17 treatment of H460 cells for 12 h, A17 dose-dependently
increased the expression of BAX, but decreased the expression of
Bcl-2.
Finally, caspase is activated by all of the upstream
signals, which leads to the ordered and sequential dismantling of
the cells until competion of the execution of ER stress-induced
apoptosis (26,37). In different studies of ER
stress-induced human cancer cell apoptosis, the cleavage and
activation of caspase-3 has been observed (26,37,38,40).
Fig. 4 suggests that A17
dose-dependently induced the cleavage and activation of caspase-3;
the cleaved caspase-3 was able to cleave PARP (namely, the
substrate of caspase-3), thereby resulting in the loss of PARP
activity consequently leading to H460 cell apoptosis. In summary,
our data confirmed the capacity of A17 to activate all key proteins
at various phases of ER stress. Therefore, according to above
relative results, we propose a signaling model of the development
of ER stress-induced apoptosis induced by A17 (Fig. 6B).
The sensitivity of H460 cells to A17 treatment at
concentrations ranging from 1.25 to 15 µM was further
confirmed by the results of the cell viability and colony formation
assays, and flow cytometric analysis, whereas curcumin did not
display adequate activity at the indicated concentrations (Figs. 2 and 3). The above mentioned data suggest that
ER stress was related to A17-induced cell apoptosis. However,
whether A17-induced cell apoptosis is ER stress-dependent remains
unknown. The transfection of cells with CHOP or GRP78 siRNA has
been reported to reduce ER stress-mediated human colon cancer cell
apoptosis (41). CHOP knockdown by
specific siRNAs attenuated γ-tocotrienol-induced (42) and desipramine-induced ER
stress-mediated apoptotic cascade (43). By comparing A17-induced
ER-stress-dependent and non-ER-stress-dependent apoptosis, the role
of ER stress in A17-induced apoptosis was verified using CHOP siRNA
transfection. Fig. 6A also
indicates that CHOP is a pro-apoptotic protein. More significantly,
the results suggest that the A17-induced apoptosis is, at least in
part, ER stress-dependent. Despite the fact that our hypothesis was
supported by the data, the A17-induced apoptosis may also involve
other apoptotic mechanisms. In mitochondria-mediated apoptotic
pathways, p53 phosphorylation and caspase-3 activation also play
important roles (37,39). Moreover, it was reported that
curcumin exerts anticancer effects by multi-targeting mechanisms
(13,44). However, the present study only
focused on ER stress-mediated apoptosis. Thus, further studies are
warranted.
In summary, novel compound A17 was found to have
better stability and antitumor effects than curcumin against human
lung cancer H460 cells via an ER stress-mediated mechanism.
Therefore, based on these properties of A17, safe and effective
agents could be further explored for the treatment of NSCLC.
Acknowledgments
The present study was supported by the Natural
Science Foundation of Zhejiang Province, China (grant nos.
13H300005 and Y4110029), the National Natural Science Foundation of
China (grant nos. 81272462 and 31300819), the China Scholarship
Council (grant no. 201208330266), the Zhejiang Provincial
Foundation for Health Department (grant no. 2015KYA150), the
Technology Foundation for Medical Science of Zhejiang Province
(grant nos. 2015KYB056 and 2012KYA129), and the Wenzhou Municipal
Science and Technology Project for Public Welfare (grant no.
Y20140662).
References
|
1
|
Liu Z, Sun Y, Ren L, Huang Y, Cai Y, Weng
Q, Shen X, Li X, Liang G and Wang Y: Evaluation of a curcumin
analog as an anti-cancer agent inducing ER stress-mediated
apoptosis in non-small cell lung cancer cells. BMC Cancer.
13:494–513. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Armstrong JL, Flockhart R, Veal GJ, Lovat
PE and Redfern CP: Regulation of endoplasmic reticulum
stress-induced cell death by ATF4 in neuroectodermal tumor cells. J
Biol Chem. 285:6091–6100. 2010. View Article : Google Scholar :
|
|
3
|
Hill DS, Martin S, Armstrong JL, Flockhart
R, Tonison JJ, Simpson DG, Birch-Machin MA, Redfern CP and Lovat
PE: Combining the endoplasmic reticulum stress-inducing agents
bortezomib and fenretinide as a novel therapeutic strategy for
metastatic melanoma. Clin Cancer Res. 15:1192–1198. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liao PC, Tan SK, Lieu CH and Jung HK:
Involvement of endoplasmic reticulum in paclitaxel-induced
apoptosis. J Cell Biochem. 104:1509–1523. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Joo JH, Liao G, Collins JB, Grissom SF and
Jetten AM: Farnesol-induced apoptosis in human lung carcinoma cells
is coupled to the endoplasmic reticulum stress response. Cancer
Res. 67:7929–7936. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Siu FM, Ma DL, Cheung YW, Lok CN, Yan K,
Yang Z, Yang M, Xu S, Ko BC, He QY, et al: Proteomic and
transcriptomic study on the action of a cytotoxic saponin
(Polyphyllin D): Induction of endoplasmic reticulum stress and
mitochondria-mediated apoptotic pathways. Proteomics. 8:3105–3117.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sun SH, Huang HC, Huang C and Lin JK:
Cycle arrest and apoptosis in MDA-MB-231/Her2 cells induced by
curcumin. Eur J Pharmacol. 690:22–30. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pae HO, Jeong SO, Jeong GS, Kim KM, Kim
HS, Kim SA, Kim YC, Kang SD, Kim BN and Chung HT: Curcumin induces
pro-apoptotic endoplasmic reticulum stress in human leukemia HL-60
cells. Biochem Biophys Res Commun. 353:1040–1045. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Scott DW and Loo G: Curcumin-induced
GADD153 upregulation: Modulation by glutathione. J Cell Biochem.
101:307–320. 2007. View Article : Google Scholar
|
|
10
|
Bakhshi J, Weinstein L, Poksay KS,
Nishinaga B, Bredesen DE and Rao RV: Coupling endoplasmic reticulum
stress to the cell death program in mouse melanoma cells: Effect of
curcumin. Apoptosis. 13:904–914. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lin SS, Huang HP, Yang JS, Wu JY, Hsia TC,
Lin CC, Lin CW, Kuo CL, Gibson Wood W and Chung JG: DNA damage and
endoplasmic reticulum stress mediated curcumin-induced cell cycle
arrest and apoptosis in human lung carcinoma A-549 cells through
the activation caspases cascade- and mitochondrial-dependent
pathway. Cancer Lett. 272:77–90. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ng AP, Chng WJ and Khan M: Curcumin
sensitizes acute promyelocytic leukemia cells to unfolded protein
response-induced apoptosis by blocking the loss of misfolded N-CoR
protein. Mol Cancer Res. 9:878–888. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wu SH, Hang LW, Yang JS, Chen HY, Lin HY,
Chiang JH, Lu CC, Yang JL, Lai TY, Ko YC, et al: Curcumin induces
apoptosis in human non-small cell lung cancer NCI-H460 cells
through ER stress and caspase cascade- and mitochondria-dependent
pathways. Anticancer Res. 30:2125–2133. 2010.PubMed/NCBI
|
|
14
|
Wang L, Wang L, Song R, Shen Y, Sun Y, Gu
Y, Shu Y and Xu Q: Targeting sarcoplasmic/endoplasmic reticulum
Ca2+-ATPase 2 by curcumin induces ER
stress-associated apoptosis for treating human liposarcoma. Mol
Cancer Ther. 10:461–471. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Anand P, Kunnumakkara AB, Newman RA and
Aggarwal BB: Bioavailability of curcumin: Problems and promises.
Mol Pharm. 4:807–818. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liang G, Yang S, Jiang L, Zhao Y, Shao L,
Xiao J, Ye F, Li Y and Li X: Synthesis and anti-bacterial
properties of mono-carbonyl analogues of curcumin. Chem Pharm Bull.
56:162–167. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Liang G, Shao L, Wang Y, Zhao C, Chu Y,
Xiao J, Zhao Y, Li X and Yang S: Exploration and synthesis of
curcumin analogues with improved structural stability both in vitro
and in vivo as cytotoxic agents. Bioorg Med Chem. 17:2623–2631.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang Y, Xiao J, Zhou H, Yang S, Wu X,
Jiang C, Zhao Y, Liang D, Li X and Liang G: A novel monocarbonyl
analogue of curcumin,
(1E,4E)-1,5-bis(2,3-dimethoxyphenyl)penta-1,4-dien-3-one, induced
cancer cell H460 apoptosis via activation of endoplasmic reticulum
stress signaling pathway. J Med Chem. 54:3768–3778. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang X, Zhang HQ, Zhu GH, Wang YH, Yu XC,
Zhu XB, Liang G, Xiao J and Li XK: A novel mono-carbonyl analogue
of curcumin induces apoptosis in ovarian carcinoma cells via
endoplasmic reticulum stress and reactive oxygen species
production. Mol Med Rep. 5:739–744. 2012.
|
|
20
|
Zhao C, Liu Z and Liang G: Promising
curcumin-based drug design: Mono-carbonyl analogues of curcumin
(MACs). Curr Pharm Des. 19:2114–2135. 2013.
|
|
21
|
Qu W, Xiao J, Zhang H, Chen Q, Wang Z, Shi
H, Gong L, Chen J, Liu Y, Cao R, et al: B19, a novel monocarbonyl
analogue of curcumin, induces human ovarian cancer cell apoptosis
via activation of endoplasmic reticulum stress and the autophagy
signaling pathway. Int J Biol Sci. 9:766–777. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li W, Wu M, Tang L, Pan Y, Liu Z, Zeng C,
Wang J, Wei T and Liang G: Novel curcumin analogue 14p protects
against myocardial ischemia reperfusion injury through
Nrf2-activating anti-oxidative activity. Toxicol Appl Pharmacol.
282:175–183. 2015. View Article : Google Scholar
|
|
23
|
Ye H, Tong J, Wu J, Xu X, Wu S, Tan B, Shi
M, Wang J, Zhao W, Jiang H, et al: Preclinical evaluation of
recombinant human IFNα2b-containing magnetoliposomes for treating
hepatocellular carcinoma. Int J Nanomedicine. 9:4533–4550.
2014.
|
|
24
|
Bertolotti A, Zhang Y, Hendershot LM,
Harding HP and Ron D: Dynamic interaction of BiP and ER stress
transducers in the unfolded-protein response. Nat Cell Biol.
2:326–332. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Oyadomari S and Mori M: Roles of
CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Di er.
2004.11:381–389. View Article : Google Scholar
|
|
26
|
Szegezdi E, Logue SE, Gorman AM and Samali
A: Mediators of endoplasmic reticulum stress-induced apoptosis.
EMBO Rep. 7:880–885. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dempke WC, Suto T and Reck M: Targeted
therapies for non-small cell lung cancer. Lung Cancer. 67:257–274.
2010. View Article : Google Scholar
|
|
28
|
Sharma RA, Steward WP and Gescher AJ:
Pharmacokinetics and pharmacodynamics of curcumin. Adv Exp Med
Biol. 595:453–470. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Appiah-Opong R, de Esch I, Commandeur JN,
Andarini M and Vermeulen NP: Structure-activity relationships for
the inhibition of recombinant human cytochromes P450 by curcumin
analogues. Eur J Med Chem. 43:1621–1631. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Selvam C, Jachak SM, Thilagavathi R and
Chakraborti AK: Design, synthesis, biological evaluation and
molecular docking of curcumin analogues as antioxidant,
cyclooxygenase inhibitory and anti-inflammatory agents. Bioorg Med
Chem Lett. 15:1793–1797. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ohori H, Yamakoshi H, Tomizawa M, Shibuya
M, Kakudo Y, Takahashi A, Takahashi S, Kato S, Suzuki T, Ishioka C,
et al: Synthesis and biological analysis of new curcumin analogues
bearing an enhanced potential for the medicinal treatment of
cancer. Mol Cancer Ther. 5:2563–2571. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mosley CA, Liotta DC and Snyder JP: Highly
active anticancer curcumin analogues. Adv Exp Med Biol. 595:77–103.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Subramaniam D, May R, Sureban SM, Lee KB,
George R, Kuppusamy P, Ramanujam RP, Hideg K, Dieckgraefe BK,
Houchen CW, et al: Diphenyl difluoroketone: A curcumin derivative
with potent in vivo anticancer activity. Cancer Res. 68:1962–1969.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang X, Lin J, Chen Y, Zhong W, Zhao G,
Liu H and Li S, Wang L and Li S: Novel fatty acid synthase (FAS)
inhibitors: Design, synthesis, biological evaluation, and molecular
docking studies. Bioorg Med Chem. 17:1898–1904. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Park IJ, Kim MJ, Park OJ, Choe W, Kang I,
Kim SS and Ha J: Cryptotanshinone induces ER stress-mediated
apoptosis in HepG2 and MCF7 cells. Apoptosis. 17:248–257. 2012.
View Article : Google Scholar
|
|
36
|
Zhang K and Kaufman RJ: Protein folding in
the endoplasmic reticulum and the unfolded protein response. Handb
Exp Pharmacol. 172:69–91. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Schleicher SM, Moretti L, Varki V and Lu
B: Progress in the unraveling of the endoplasmic reticulum
stress/autophagy pathway and cancer: Implications for future
therapeutic approaches. Drug Resist Updat. 13:79–86. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rasheva VI and Domingos PM: Cellular
responses to endoplasmic reticulum stress and apoptosis. Apoptosis.
14:996–1007. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mielke K and Herdegen T: JNK and p38
stresskinases - degenerative effectors of
signal-transduction-cascades in the nervous system. Prog Neurobiol.
61:45–60. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Loughlin DT, Artlett CM and Kango-Singh M:
Precursor of advanced glycation end products mediates
ER-stress-induced caspase-3 activation of human dermal fibroblasts
through NAD(P)H oxidase 4. PLoS One. 5:e110932010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Huang SM, Cheung CW, Chang CS, Tang CH,
Liu JF, Lin YH, Chen JH, Ko SH, Wong KL and Lu DY: Phloroglucinol
derivative MCPP induces cell apoptosis in human colon cancer. J
Cell Biochem. 112:643–652. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ma J, Qiu Y, Yang L, Peng L, Xia Z, Hou
LN, Fang C, Qi H and Chen HZ: Desipramine induces apoptosis in rat
glioma cells via endoplasmic reticulum stress-dependent CHOP
pathway. J Neurooncol. 101:41–48. 2011. View Article : Google Scholar
|
|
44
|
Bar-Sela G, Epelbaum R and Schaffer M:
Curcumin as an anti-cancer agent: Review of the gap between basic
and clinical applications. Curr Med Chem. 17:190–197. 2010.
View Article : Google Scholar : PubMed/NCBI
|