Introduction
Hepatocellular carcinoma (HCC; liver cancer) is one
of the most frequently occuring cancers in the world (1). The incidence rates (newly occurring
cases per unit of population) and mortality rates in sub-Saharan
Africa were 6.2 and 8.3%, respectively (2). HCC is the third primary cause of
cancer-related deaths worldwide, with an estimated 749,000 new
cases annually, particularly in low income countries (3). Moreover, only a few HCC patients are
indicated to receive systemic chemotherapy treatment (4). However, HCC patients receiving
chemotherapy with sorafenib, 5-fluorouracil and cisplatin may also
present with severe side effects and multidrug drug resistance.
Thus, therapy for HCC patients is still a severe challenge.
Therefore, it is imperative to discover and develop natural
anticarcinogens which are safe and effective. At present, natural
products from medicinal plants such as, pure compounds such as
sasanquasaponin (5) guggulsterone
(6), xanthohumol (7) and isoliensinine polyphyllin VII
(8) are used as alternative
treatments due to their wide range of antiproliferative activities
in human tumor cell lines.
Gigantol is a biphenyl phenolic compound primarily
extracted from the stem of Dendrobium aurantiacum (9). Previous studies have confirmed that
the phenolic compound extracted from Crataegus monogyna
(10) inhibited the cell growth of
human tumor cells including breast adenocarcinoma MCF-7, non-small
cell lung cancer NCI-H460, cervical carcinoma HeLa and HCC HepG2
cell lines. Furthermore, it has been reported that gigantol
possesses a variety of bioactivities including anti-osmotic
(11), antitumor in lung cancer
(12), antimutagenic (13) and immunomodulatory (14). In addition, gigantol has been
described as a potent compound used in the recovery of sight in
diabetic cataract disease (15).
However, there have been no studies οn the effects of gigantol on
growth inhibition and apoptosis in human liver cancer.
One of the most significant intracellular signal
transduction pathways is the phosphoinositol-3-kinase (PI3K/Akt)
signaling pathway (16). When
activated, the pathway regulates important cellular functions,
including the promotion of cancer cell proliferation and inhibition
of apoptosis by influencing the activity of downstream effector
molecules (17). Moreover, it has
been reported that when tumor cells are treated with wortmannin or
LY294002, PI3K/Akt inhibitors (18), Akt phosphorylation not only leads to
phosphorylation of blocked BAD, but also phosphorylates Ser 196 of
caspase-9 and Ser 184 of Bax, which leads to the inactivation and
inhibition of pro-apoptotic effects (19). NF-κB is a common dimeric
transcription factor (20) that
plays a key role in the regulation of the expression of a variety
of critical genes that regulate cell survival, proliferation,
apoptosis, immune responses and changes in the intracellular redox
balance (21). Several
apoptosis-associated genes or regulatory proteins, such as p53,
Bax, Bcl-2 and caspase-3, are reportedly activated or regulated by
NF-κB in various tumor cells (22).
In the present study, the anticancer effects of
gigantol-induced growth inhibition and apoptosis of human liver
cancer via the PI3K/Akt/NF-κB signaling pathway were
elucidated.
Materials and methods
Chemicals and materials
The human liver cancer HepG2 cell line was obtained
from Guangzhou Jini Ou Biological Technology Co., Ltd. (Guangzhou,
China). High glucose Dulbecco's modified Eagle's medium (H-DMEM),
fetal bovine serum (FBS), penicillin-streptomycin, trypsin, trypsin
(non-EDTA) and phosphate-buffered saline (PBS) were acquired from
Gibco Chemical Co. (Rockville, MD, USA).
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide
(MTT) and dimethyl sulfoxide (DMSO) were acquired from
Sigma-Aldrich (St. Louis, MO, USA). Gigantol was purchased from
Chinese Materials Research Center (Daxing, Beijing, China). The
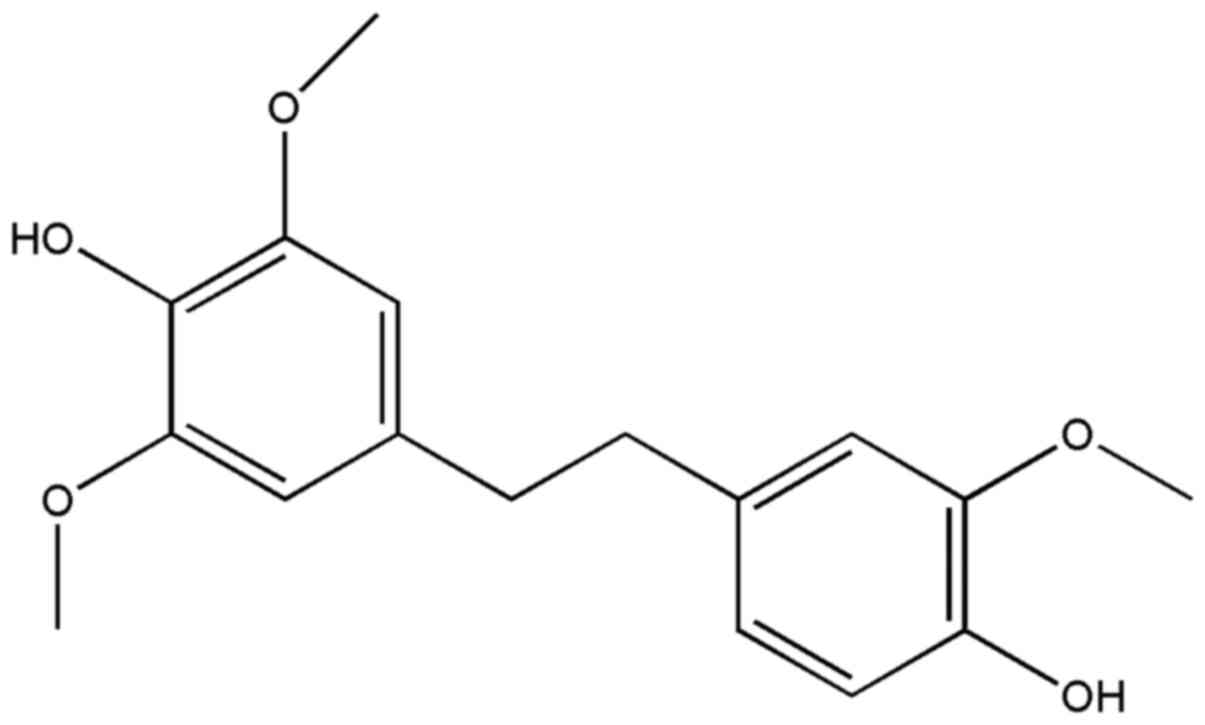
chemical structure of gigantol is shown in Fig. 1. The Annexin V-FITC apoptosis assay
and Hoechst 33258 assay kits were provided by KeyGen Biotech Co.,
Ltd. (Jiangsu, China). The PI3K/AKT inhibitor LY294002 was
purchased from Selleck Chemicals (Houston, TX, USA). All other
chemicals used were of analytical grade.
Cell culture
HepG2 cells were cultured in DMEM supplemented with
10% (v/v) FBS and 1% penicillin-streptomycin and incubated at 37°C
in a humidified atmosphere containing 5% CO2. The
control group included HepG2 cells in the absence of gigantol in
serum-free medium. To study the effects from the intervention of
the inhibitor, the cells were pretreated with PI3K/AKT inhibitor
LY294002 (25 µmol/l) for 1 h, followed by treatment with
gigantol.
Cell viability assay
The cytotoxicity was measured using MTT assays.
Briefly, HepG2 cells were seeded in a 96-well plate
(5×104 cells/well) and serum-starved for 24 h using
H-DMEM (without FBS). Then the culture medium was replaced with
H-DMEM containing a series of concentrations of gigantol (1, 10,
40, 80 and 150 µM) for different time intervals (12, 24 and 48 h).
After treatment, 20 µl of MTT (5 mg/ml) was added to each well and
continuously cultured for an additional 4 h at 37°C. The cell
supernatant was removed by aspiration and the formazan was
dissolved with 150 µl DMSO. The absorbance was read at 490 nm using
a microplate reader (Bio-Rad Laboratories, Inc., Hercules, CA,
USA). The cell viability was calculated as follows: cell viability
= (A490 experimental/A490 control) ×
100%.
Morphological changes
The effect of gigantol on the morphological changes
of HepG2 cells was assessed using the chromatin dye Hoechst 33258.
The HepG2 cells were cultured in 24-well plates (5×104
cells/well) overnight and then exposed to gigantol with
concentrations of 0, 1, 40 and 150 µM for 48 h. Subsequently, the
cells were fixed with 4% paraformaldehyde for 10 min at 4°C and
then dyed with Hoechst 33258 for 5 min in the dark after being
washed by PBS twice. Morphological changes were observed by
phase-contrast microscopy and the images of the apoptotic cells
were visualized using a fluorescence microscope (Leica,
Microsystems GmbH, Wetzlar, Germany).
Flow cytometric analysis of cell
apoptosis
HepG2 cells were seeded in 6-well plates
(5×105 cells/well). After the cells had attached to the
culture plate, gigantol was added at concentrations of 0, 1, 40 and
150 µM for 48 h. According to the manufacturer's instructions,
HepG2 cells which were digested with trypsin (non-EDTA) were washed
with PBS and harvested and then suspended in 500 µl binding buffer.
Following staining with 5 µl Annexin V-FITC and 5 µl propidium
iodide (PI) for 10 min at room temperature in the dark, the samples
were immediately analyzed using BD FACSCanto II flow cytometer.
Protein extraction and western blot
analysis
HepG2 cells were seeded in 6-well plates
(1×106 cells/well) overnight. After treatment with or
without gigantol or in combination with the inhibitor LY294002, the
cells were collected, washed with cold PBS, and resuspended in cold
RIPA buffer with a protein inhibitor. Then the cellular protein was
collected and the protein concentration was determined using the
Bradford assay. Equal amounts of protein (40 µg/lane) were
separated on 15% SDS-polyacrylamide gels and transferred onto
polyvinylidene difluoride membranes via electrophoresis. After
blocking with Tris-buffered saline (TBS) containing 5% skimmed milk
and 0.05% Tween-20 for 1 h, the membranes were incubated with
primary antibodies Akt, p-Akt, PARP, p53 and β-actin at 4°C
overnight. Following three washes with TBS for 5 min, the membranes
were incubated with horseradish peroxidase-conjugated with goat
anti-rabbit immunoglobulin G for 1 h at room temperature. Finally,
the protein bands were washed with TBST three times and visualized
using an ECL western blot analysis kit according to the
manufacturer's instructions.
Statistical analysis
Data shown in this study are presented as the mean ±
SEM and the results from each group were obtained using triplicate
samples independently. A one-way ANOVA was used to determine the
differences among the groups. A probability value of *P<0.05 was
considered to be statistically significant, while **P<0.01 was
considered to be highly statistically significant and are indicated
in the figures. All statistical analyses were carried out using
Prism5 software package (GraphPad Software Inc., La Jolla, CA,
USA).
Results
Anticancer effect of gigantol on the
inhibition of HepG2 cell growth
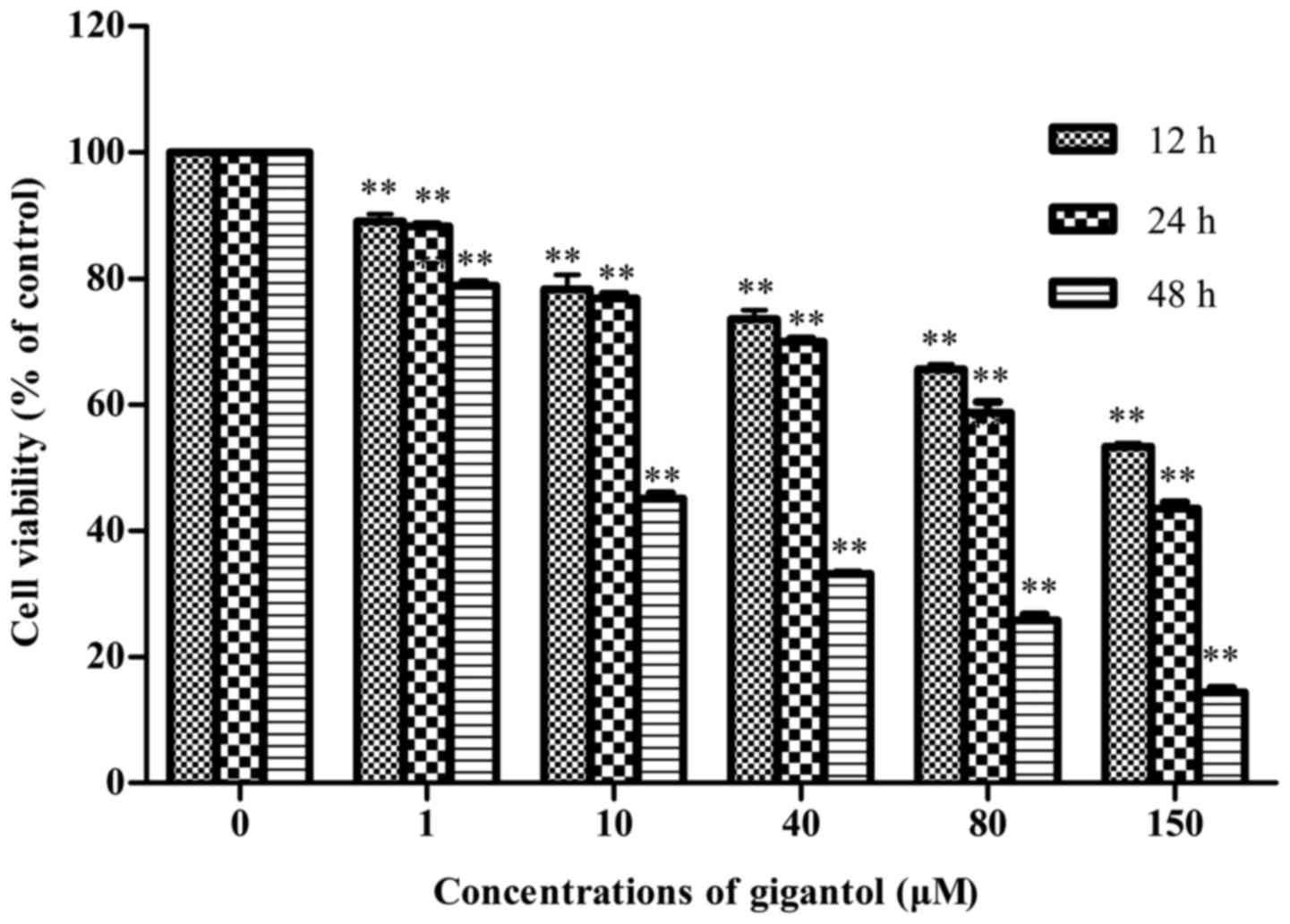
The effect of gigantol on the viability of the HepG2
cells was measured by MTT assay. As indicated in Fig. 2, gigantol inhibited the growth of
HepG2 cells in a dose- and time-dependent manner, particularly at a
concentration beyond 10 µM at 48 h. Gigantol at concentrations of
1, 40 and 150 µM markedly decreased the cell viability by 11.7,
30.0 and 56.4% at 24 h and 21.1, 66.8 and 85.5% at 48 h,
respectively. The IC50 (48 h) value was 9.30 µM. Thus,
the concentrations of 1, 40 and 150 µM and the incubation period of
48 h were the optimal parameters to carry out further
experiments.
Anticancer effect of gigantol on the
morphological changes of HepG2 cells
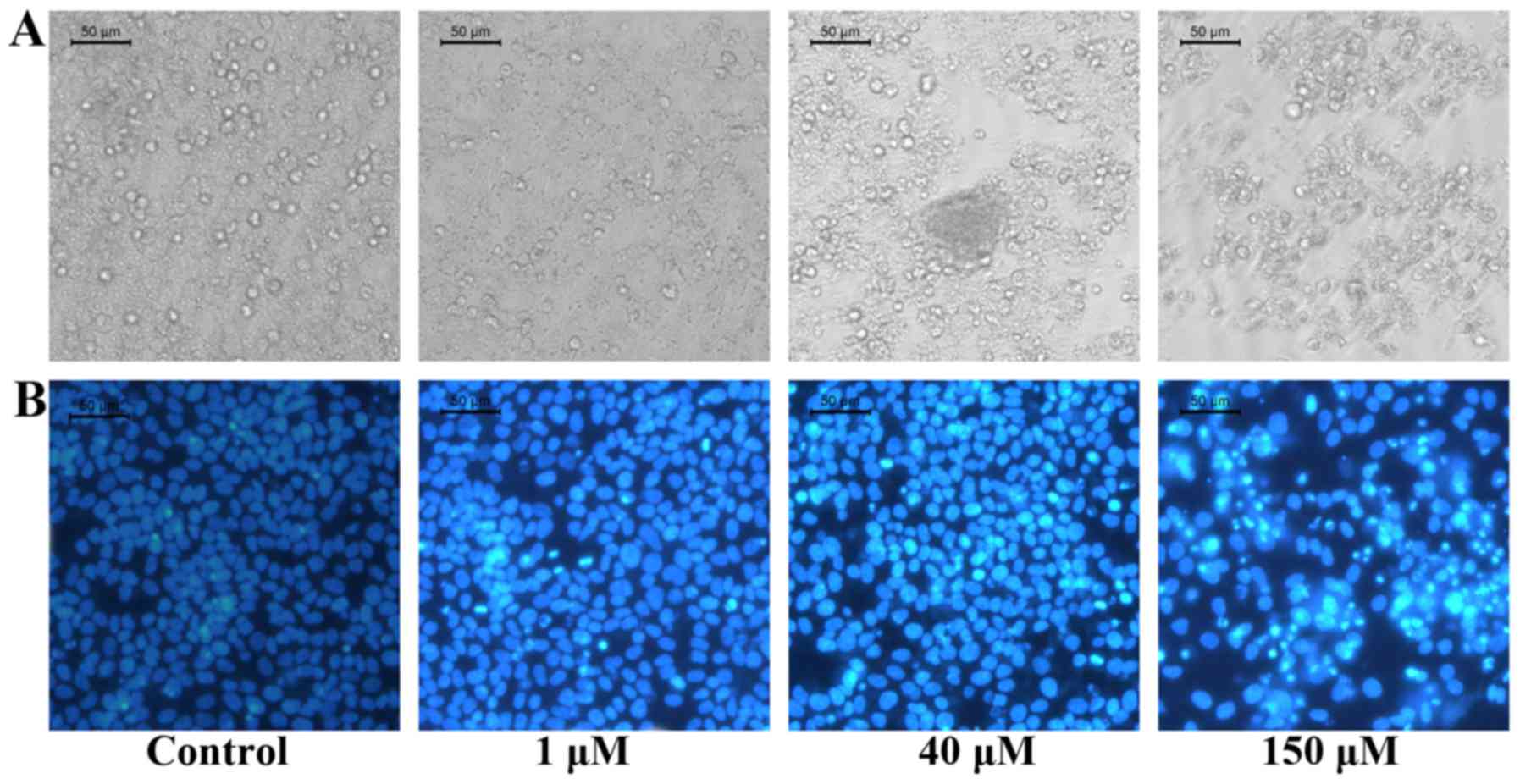
The morphological changes and apoptotic morphology
of HepG2 cells were investigated by fluorescence microscopy, as
shown in Fig. 3. Following
treatment with gigantol (1, 40 and 150 µM) for 48 h, the cells
exhibited membrane blebbing, cytoplasm condensation, and nuclear
fragmentation with condensed chromatin typical characteristics of
apoptosis. This suggests that gigantol inhibits the growth of HepG2
cells by inducing the apoptotis of tumor cells.
Anticancer effect of gigantol on the
apoptosis rate of HepG2 cells
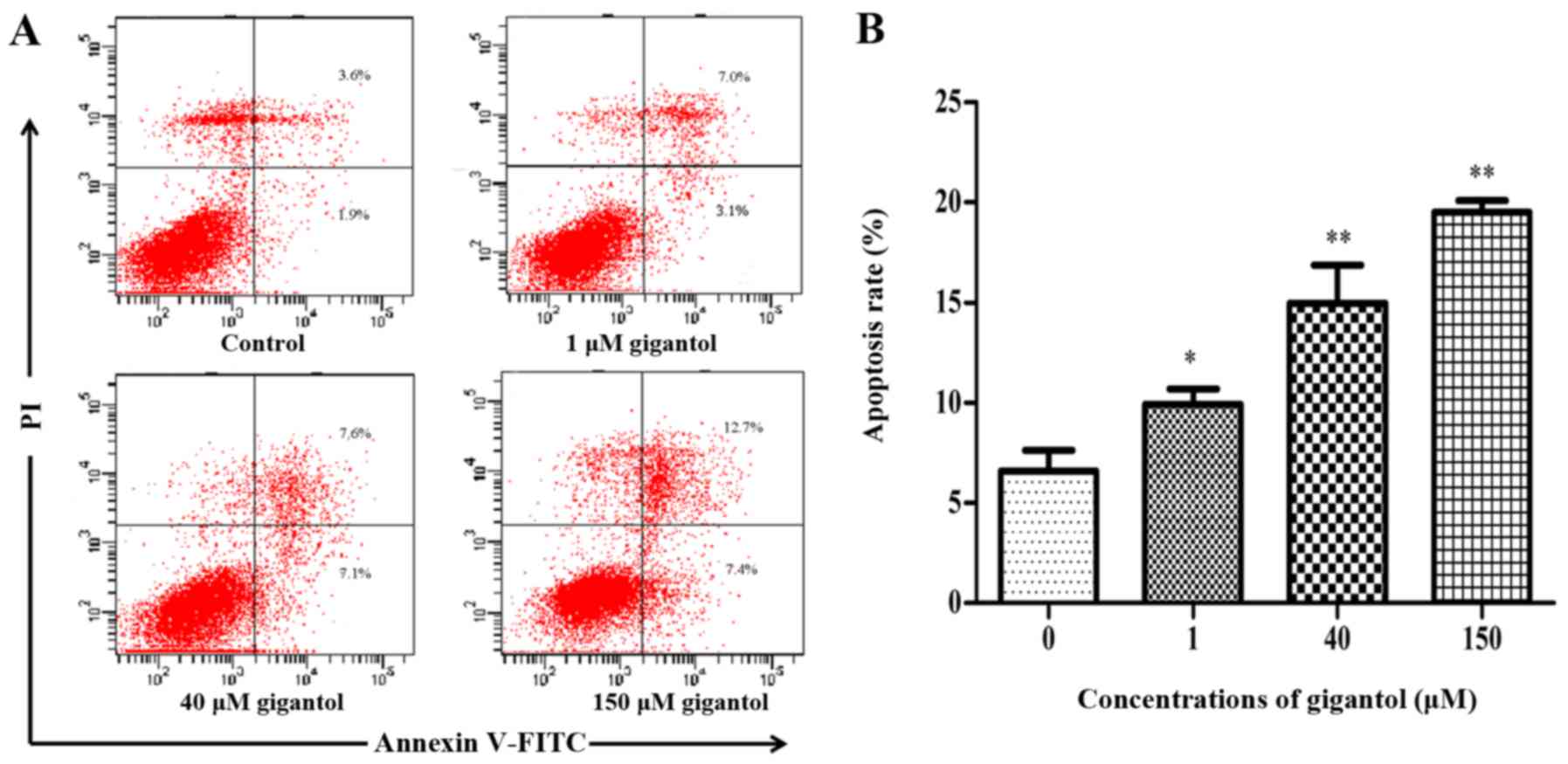
We evaluated cell apoptosis using the Annexin V and
PI double-staining assay using flow cytometry. As depicted in
Fig. 4, after incubation with
gigantol (1, 40 and 150 µM) for 48 h, the apoptosis rates were 9.9,
14.9 and 20.7%, respectively. All of the values were higher than
that in the control group, which demonstrated a significant
increase in the cell apoptosis rate.
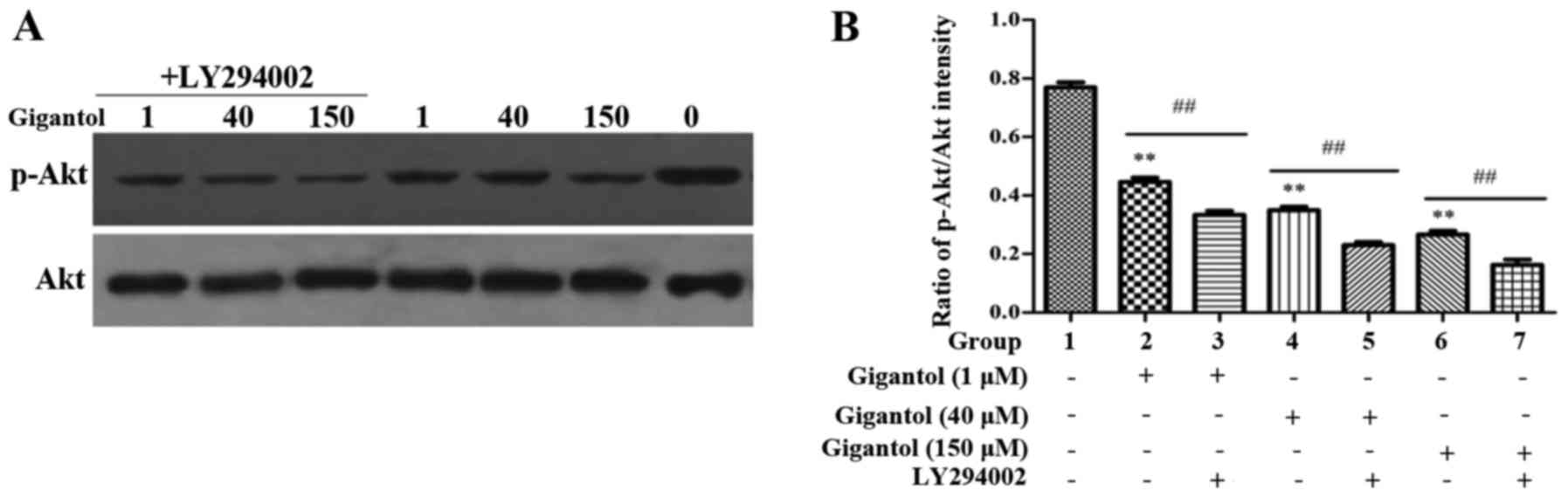
Anticancer effect of gigantol involves
inhibition of p-Akt/Akt in human liver cancer cells
As previously reported, the PI3K/Akt pathway plays
an important role in cell survival under stimulation (23). Therefore, to investigate whether
gigantol inhibits the phosphorylation of Akt, HepG2 cells were
treated with gigantol at concentrations of 1, 40 and 150 µM for 48
h (Fig. 5). Western blot analysis
showed that the p-Akt protein expression level was decreased in a
dose-dependent manner. In addition, there was a significant
decrease in the ratio of p-Akt/Akt following treatment with
gigantol, which was further suppressed by LY294002.
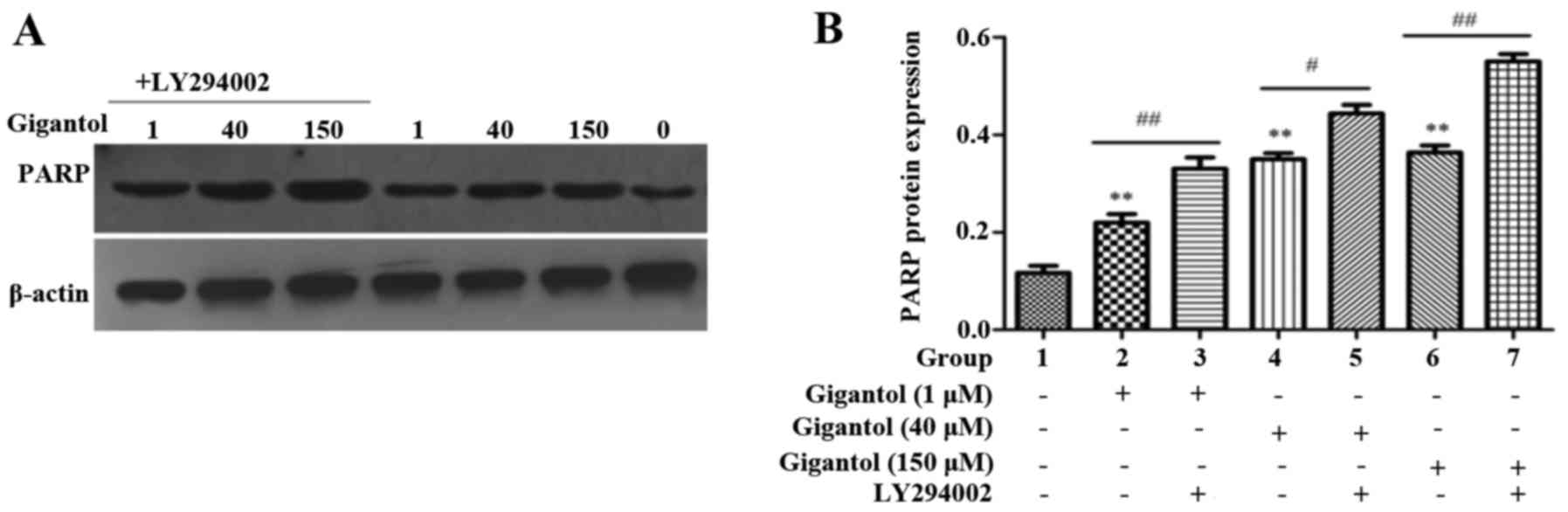
Anticancer effect of gigantol involves the induction
of PARP signaling in human liver cancer cells. Western blot
analysis was performed to detect PARP protein expression in HepG2
cells which were treated with gigantol. As shown in Fig. 6, significant upregulation of PARP
protein expression was observed in the HepG2 cells treated with
gigantol (1, 40 and 150 µM) and LY294002 further increased the
expression of PARP.
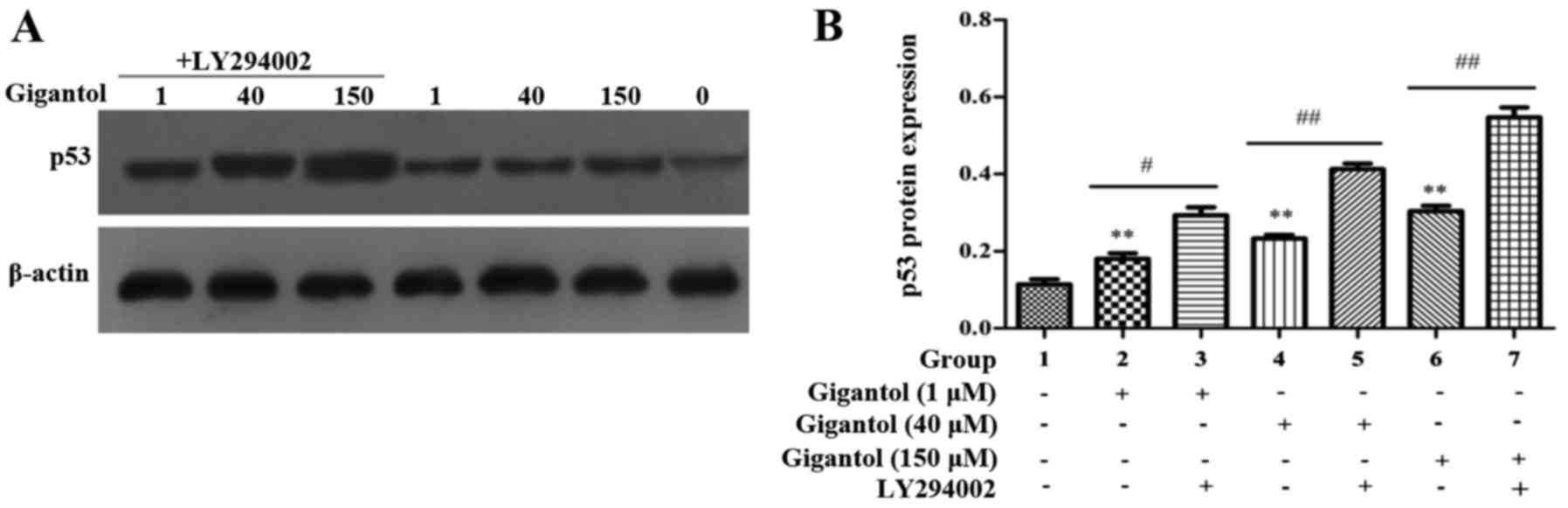
Anticancer effect of gigantol involves
the induction of p53 signaling in human liver cancer cells
We investigated whether the anticancer effect of
gigantol involves p53 signaling in human liver cancer cells by
western blot analysis. As shown in Fig.
7, following gigantol treatment, p53 protein expression was
increased in the HepG2 cells and LY294002 further increased the
expression of p53.
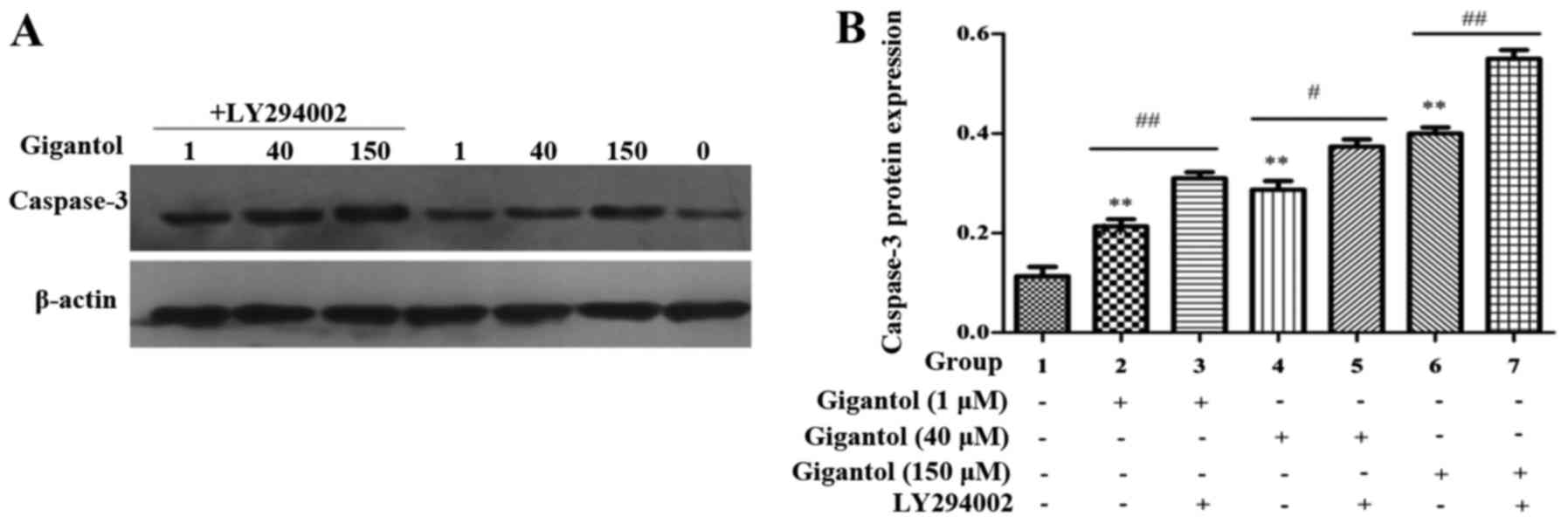
Anticancer effect of gigantol involves
the induction of caspase-3 signaling in human liver cancer
cells
To determine whether gigantol induces the apoptosis
of HepG2 cells, caspase-3 activity was assessed. Fig. 8 demonstrates that gigantol
significantly increased caspase-3 activity in the HepG2 cells.
Discussion
Apoptosis, which is a process of cell suicide, is
associated with characteristic morphological changes, such as
condensation of chromatin, blebbing formation and biochemical
changes (24). In apoptotic cells,
the membrane phospholipid phosphatidylserine (PS) is translocated
from the inner to the outer leaflet of the plasma membrane, thereby
exposing PS to the external cellular environment. Annexin V has a
high affinity for PS, and binds to cells with exposed PS. Annexin V
is conjugated to FITC and serves as a sensitive probe for flow
cytometric analysis. Thus, since externalization of PS occurs in
the earlier stages of apoptosis, FITC Annexin V staining can
identify apoptosis at earlier stages (25,26).
The present study revealed that HepG2 cells were increased in a
dose-dependent manner when exposed to PS, which was manifested when
gigantol-induced cells entered the early stages of apoptosis.
Previous studies have shown that the PI3K/Akt
pathway is crucial to many aspects of cell growth, survival and
apoptosis and implicated in inducing cell metabolism,
proliferation, migration and survival (27), while NF-κB is usually associated
with suppression of cell proliferation, invasion, tumor metastasis
and apoptosis (28). Simultaneous
activation with multiple signaling pathways may lead to undesirable
therapy, therefore, the PI3K/Akt/NF-κB signaling pathway is
important in the stimulation of tumor cell survival,
chemosensitivity, and invasive behavior in a variety of cancer
signaling pathways (29). In a
previous study, Shan et al (30) indicated that the inhibition of
Aurora-B suppressed HepG2 cells by decreasing the protein
expression levels of p-Akt, Akt, NF-κB p65, MMP-2 and MMP-9 via the
modulation of the PI3K/Akt/NF-κB signaling pathway in
vitro.
PARP plays a significant role in genomic stability
and in the process of apoptosis in the modulation of DNA
replication and transcription (31). When DNA is broken and damaged,
activated PARP participates in combining with DNA breakage, which
may protect the terminal of bare DNA from the catabolic reaction by
nuclease (32). Also, in response
to DNA damage, p53, one of the most significant members of the
tumor-suppressor proteins, markedly promotes the genes responsible
for cell proliferation and migration (33). Zhao et al (7) reported that xanthohumol induced growth
inhibition and apoptosis through an increase in cleaved PARP and
p53 in human liver cancer.
In summary, the present study revealed for the first
time, that gigantol, a monomer compound isolated from Dendrobium
aurantiacum, exerted growth inhibition and apoptosis against
human liver cancer cells through the PI3K/Akt/NF-κB signaling
pathway and the anticancer effects of gigantol induced growth
inhibition and apoptosis through an increase in cleaved PARP, p53,
caspase-3 and a decreased ratio of p-Akt/Akt in human liver cancer
cells. Our findings provide confirmation of the anticancer effect
of gigantol in human liver cancer. However, it is still necessary
to investigate further in order to elucidate its pro-apoptotic
effects and the underlying biochemical mechanisms involved.
Acknowledgements
The authors are grateful for the financial support
by the Special Foundation of 2015 High Level University
Construction (no. 2050205).
References
|
1
|
Yu F, Shen X, Fan L and Yu Z: Analysis of
histone modifications at human ribosomal DNA in liver cancer cell.
Sci Rep. 5:181002015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bosch FX, Ribes J, Díaz M and Cléries R:
Primary liver cancer: Worldwide incidence and trends.
Gastroenterology. 127:(Suppl 1). S5–S16. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Palliyaguru DL and Wu F: Global
geographical overlap of aflatoxin and hepatitis C: Controlling risk
factors for liver cancer worldwide. Food Addit Contam Part A Chem
Anal Control Expo Risk Assess. 30:534–540. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lin L, Fang Z, Lin H, You H, Wang J, Su Y,
Wang F and Zhang ZY: Depletion of Cks1 and Cks2 expression
compromises cell proliferation and enhance chemotherapy-induced
apoptosis in HepG2 cells. Oncol Rep. 35:26–32. 2016.PubMed/NCBI
|
|
5
|
Zeng J, Chen S, Li N, Chen L, Su J, Niu G,
Zhu S and Liang Y: Sasanquasaponin from Camellia oleifera Abel.
induces apoptosis via Bcl-2, Bax and caspase-3 activation in HepG2
cells. Mol Med Rep. 12:1997–2002. 2015.PubMed/NCBI
|
|
6
|
Shi JJ, Jia XL, Li M, Yang N, Li YP, Zhang
X, Gao N and Dang SS: Guggulsterone induces apoptosis of human
hepatocellular carcinoma cells through intrinsic mitochondrial
pathway. World J Gastroenterol. 21:13277–13287. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhao X, Jiang K, Liang B and Huang X:
Anticancer effect of xanthohumol induces growth inhibition and
apoptosis of human liver cancer through NF-κB/p53-apoptosis
signaling pathway. Oncol Rep. 35:669–675. 2016.PubMed/NCBI
|
|
8
|
Shu G, Yue L, Zhao W, Xu C, Yang J, Wang S
and Yang X: Isoliensinine, a bioactive alkaloid derived from
embryos of Nelumbo nucifera, induces hepatocellular carcinoma cell
apoptosis through suppression of NF-κB signaling. J Agric Food
Chem. 63:8793–8803. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yang L, Wang Z and Xu L: Phenols and a
triterpene from Dendrobium aurantiacum var. denneanum
(Orchidaceae). Biochem Syst Ecol. 34:658–660. 2006. View Article : Google Scholar
|
|
10
|
Rodrigues S, Calhelha RC, Barreira JCM,
Dueñas M, Carvalho AM, Abreu RMV, Buelga CS and Ferreira ICFR:
Crataegus monogyna buds and fruits phenolic extracts: Growth
inhibitory activity on human tumor cell lines and chemical
characterization by HPLC–DAD–ESI/MS. Food Res Int. 49:516–523.
2012. View Article : Google Scholar
|
|
11
|
Fang H, Hu X, Wang M, Wan W, Yang Q, Sun
X, Gu Q, Gao X, Wang Z, Gu L, et al: Anti-osmotic and antioxidant
activities of gigantol from Dendrobium aurantiacum var. denneanum
against cataractogenesis in galactosemic rats. J Ethnopharmacol.
172:238–246. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bhummaphan N and Chanvorachote P: Gigantol
suppresses cancer stem cell-like phenotypes in lung cancer cells.
Evid-Based Compl Al. 2015:1–10. 2015. View Article : Google Scholar
|
|
13
|
Miyazawa M, Shimamura H, Nakamura S and
Kameoka H: Antimutagenic activity of gigantol from Dendrobium
nobile. J Agric Food Chem. 45:2849–2853. 1997. View Article : Google Scholar
|
|
14
|
Won JH, Kim JY, Yun KJ, Lee JH, Back NI,
Chung HG, Chung SA, Jeong TS, Choi MS and Lee KT: Gigantol isolated
from the whole plants of Cymbidium goeringii inhibits the
LPS-induced iNOS and COX-2 expression via NF-κB inactivation in RAW
264.7 macrophages cells. Planta Med. 72:1181–1187. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu J, Lu C, Li X, Fang H, Wan W, Yang Q,
Sun X, Wang M, Hu X, Chen CY, et al: Synthesis and biological
evaluation of novel gigantol derivatives as potential agents in
prevention of diabetic cataract. PLoS One. 10:e01410922015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Song G, Ouyang G and Bao S: The activation
of Akt/PKB signaling pathway and cell survival. J Cell Mol Med.
9:59–71. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rychahou PG, Jackson LN, Silva SR,
Rajaraman S and Evers BM: Targeted molecular therapy of the PI3K
pathway: Therapeutic significance of PI3K subunit targeting in
colorectal carcinoma. Ann Surg. 243:833–842. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Vara JA Fresno, Casado E, de Castro J,
Cejas P, Belda-Iniesta C and González-Barón M: PI3K/Akt signalling
pathway and cancer. Cancer Treat Rev. 30:193–204. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xin M and Deng X: Nicotine inactivation of
the proapoptotic function of Bax through phosphorylation. J Biol
Chem. 280:10781–10789. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dey A, Tergaonkar V and Lane DP:
Double-edged swords as cancer therapeutics: Simultaneously
targeting p53 and NF-κB pathways. Nat Rev Drug Discov. 7:1031–1040.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chariot A: The NF-κB-independent functions
of IKK subunits in immunity and cancer. Trends Cell Biol.
19:404–413. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim SM, Lee SY, Yuk DY, Moon DC, Choi SS,
Kim Y, Han SB, Oh KW and Hong JT: Inhibition of NF-κB by
ginsenoside Rg3 enhances the susceptibility of colon cancer cells
to docetaxel. Arch Pharm Res. 32:755–765. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Maddika S, Ande SR, Panigrahi S,
Paranjothy T, Weglarczyk K, Zuse A, Eshraghi M, Manda KD, Wiechec E
and Los M: Cell survival, cell death and cell cycle pathways are
interconnected: Implications for cancer therapy. Drug Resist Updat.
10:13–29. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hu Q, Pan R, Wang L, Peng B, Tang J and
Liu X: Platycodon grandiflorum induces apoptosis in SKOV3 human
ovarian cancer cells through mitochondrial-dependent pathway. Am J
Chin Med. 38:373–386. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
van Engeland M, Nieland LJ, Ramaekers FC,
Schutte B and Reutelingsperger CP: Annexin V-affinity assay: A
review on an apoptosis detection system based on phosphatidylserine
exposure. Cytometry. 31:1–9. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Andree HA, Reutelingsperger CP, Hauptmann
R, Hemker HC, Hermens WT and Willems GM: Binding of vascular
anticoagulant α (VACα) to planar phospholipids bilayers. Blood
Coagul Fibrinolysis. 5:921–928. 1994.PubMed/NCBI
|
|
27
|
Stambolic V and Woodgett JR: Functional
distinctions of protein kinase B/Akt isoforms defined by their
influence on cell migration. Trends Cell Biol. 16:461–466. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim HJ, Kim JC, Min JS, Kim MJ, Kim JA,
Kor MH, Yoo HS and Ahn JK: Aqueous extract of Tribulus terrestris
Linn induces cell growth arrest and apoptosis by down-regulating
NF-κB signaling in liver cancer cells. J Ethnopharmacol.
136:197–203. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Azijli K, Weyhenmeyer B, Peters GJ, de
Jong S and Kruyt FA: Non-canonical kinase signaling by the death
ligand TRAIL in cancer cells: Discord in the death receptor family.
Cell Death Differ. 20:858–868. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Shan RF, Zhou YF, Peng AF and Jie ZG:
Inhibition of Aurora-B suppresses HepG2 cell invasion and migration
via the PI3K/Akt/NF-κB signaling pathway in vitro. Exp Ther Med.
8:1005–1009. 2014.PubMed/NCBI
|
|
31
|
Booth L, Cruickshanks N, Ridder T, Dai Y,
Grant S and Dent P: PARP and CHK inhibitors interact to cause DNA
damage and cell death in mammary carcinoma cells. Cancer Biol Ther.
14:458–465. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kulkarni A, Oza J, Yao M, Sohail H,
Ginjala V, Tomas-Loba A, Horejsi Z, Tan AR, Boulton SJ and Ganesan
S: Tripartite motif-containing 33 (TRIM33) protein functions in the
poly(ADP-ribose) polymerase (PARP)-dependent DNA damage response
through interaction with Amplified in Liver Cancer 1 (ALC1)
protein. J Biol Chem. 288:32357–32369. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Brown CJ, Lain S, Verma CS, Fersht AR and
Lane DP: Awakening guardian angels: Drugging the p53 pathway. Nat
Rev Cancer. 9:862–873. 2009. View
Article : Google Scholar : PubMed/NCBI
|