Introduction
Royal jelly (RJ) is an animal secretion produced by
hypopharyngeal and mandibular glands of worker honeybees (Apis
mellifera L.) that are from 5- to 14-day-old. It represents the
principal food source for all the larvae in the hive, until the
third day of age, and for the queen bee, for the whole life
(1). As the queen honeybee is the
only fertile organism in the hive, its life expectancies are the
highest of the colony and its morphological traits are unique and
very peculiar (i.e. big dimensions, reduction of faringeal and wax
glands, absence of pollen pocket, growth of pheromonal glands and
gonads), it appears clear that a RJ-based diet can strongly
influence honeybee's development and destiny, acting on gene
expression and metabolism (2).
RJ chemical composition varies according to honeybee
species, physiological state of the colony, environmental
conditions and production period. Generally, it contains water
(50–60%), nitrogen compounds (18%), sugars (15%), lipids (3–6%),
mineral salts (1.5%) and traces of vitamins (3). Among nitrogen compounds, free
essential amino acids, several enzymes (i.e. glucose oxidases,
phosphatases and cholinesterases) and five honeybee typical
proteins (major royal jelly proteins, MRJP) were detected (4–7).
Fructose is the main carbohydrate (50%), followed by glucose
(33–43%), sucrose (6%) and other minor saccharides (8). Finally, although in low concentration,
the lipid profile may be considered a fingerprint for RJ. This
class of molecules includes short-chain (8C-10C) fatty acids, such
as trans-10-hydroxy-2-decenoic acid (10-HDA), non-polar lipids,
sterols, including cholesterol, and a non-saponifiable fraction of
hydrocarbons (9–11).
Various studies document that RJ possesses many
biological properties on murine and human cell systems. In
particular, it was demonstrated to have antioxidant (12,13),
anti-inflammatory (14),
anticholesteremic (15),
hypoglycemic, erythropoietic (16),
antithyroidic (17),
immunostimulatory (18,19), anti-osteoporotic (20), antifungal and antibiotic properties,
especially against Escherichia coli, Salmonella ssp., Proteus
ssp., Bacillus subtilis and Staphylococcus aureus
(21,22). In addition, Taniguchi et al
(23) and Shirzad et al
(24) demonstrated that RJ had an
important role in control and regression of murine fibrosarcoma
tumors. Of note, Tamura et al (25) even suggested that RJ could exert a
significant antiproliferative activity against slow-growing cancers
but not towards fast-growing ones. However, it is surprising and
unexpected to note the scarce scientific evidence reported on RJ
antineoplastic power.
To extend our knowledge on RJ medical
potentialities, we investigated the in vitro bioactivity of
six different RJs on the growth of three mammalian cell lines:
immortalized murine myoblasts (C2C12), human prostate cancer (PC3)
and human neuroblastoma (SH-SY5Y). In fact, no data are provided in
literature on the effect of this matrix on these specific cells.
Moreover, the originality of our study also consisted in performing
separate analyses on the lipophilic and hydrophilic portions of the
RJs, an approach never performed before, in order to better
discriminate the biological role of both these fractions. Finally,
to establish a complete framework, biochemical and protein profiles
of the RJs were deeply characterized by gas chromatography-mass
spectrometry (GC-MS) and one-dimensional sodium dodecyl
sulfate-polyacrylamide gel electrophoresis (1D-SDS-PAGE),
respectively.
Materials and methods
Sample material
Six different RJs were used in the current study.
Three of them (P1, P2 and P3) were bought directly from local
producers of RJ, while the others (G1, G2 and G3) were purchased
from National Pharmaceutical Societies. The Honey Research Center
of the University of Rome ‘Tor Vergata’ certified freshness,
authenticity and quality (i.e. absence of pesticide and antibiotic
contamination) of the samples. RJs were stored at 4°C until their
analysis.
GC-MS analysis
For GC-MS study, RJs were separated in two
fractions, according to their lipophilicity, as described in
Isidorov et al (26).
Briefly, 500 mg of RJ were resuspended in 10 ml of diethyl ether
and constantly mixed for 15 min at room temperature. Then, after
centrifugation for 5 min at maximum speed (13.000 rpm) at 4°C, the
supernatant was recovered, filtered by a Millipore 0.45 µm sieve
and conserved at 4°C. On the other hand, the pellet, containing
more polar and less lipophilic compounds, was subjected to methanol
extraction, following the same identical steps previously performed
in the procedure with diethyl ether. Finally, both samples were
completely dried out under nitrogen flow and then resuspended with
500 µl of diethyl ether or methanol, respectively. Each extract (1
µl) was injected in a GC-MS instrument (QP2010 Shimadzu, Japan) and
analyzed. The chromatographic separation was performed, in a DB-5
column (30 m × 0.25 mm × 0.25 µl; Agilent Technologies, Santa
Clara, CA, USA), setting the GC oven as follows: 50°C for 10 min,
150°C (reached at a rate of 5°C/min) for 10 min, 280°C (reached at
a rate of 1°C/min) for 35 min. Helium was used as carrier gas at a
constant flow of 2.1 ml/min. MS conditions and details about the
identification of the molecules was the same of those reported in
Gismondi et al (27). The
amount of each compound was expressed as percentage of its relative
abundance in the RJ, as described in Giovannini et al
(28).
Protein study
Lipophilic and hydrophilic protein portions of RJs
were purified according to the procedure of Li et al
(29) adequately modified. In
brief, 200 µl of 1X phosphate-buffered saline (PBS) were added to
100 mg of RJ. The solution was vortexed for 5 min, sonicated for
further 5 min and centrifuged for 10 min at maximum speed at 4°C.
The supernatant, containing the hydrosoluble protein portion, was
recovered, transferred into a new Eppendorf tube and stored at 4°C.
On the contrary, the pellet was resuspended with 200 µl of
solubilization buffer (SB: 7 M urea; 2 M thiourea; 4%
3-[(3-cholamido propyl)-dimethylammonio]-1-propane sulfonate; 0.8%
IPG-buffer pI 3–10 NL; 1% DTT) and subjected to the same previous
passages. This second supernatant, including the liposoluble
protein portion, was conserved at 4°C, while the pellet was
discarded. At this point, both the extracts were enriched with 22
µl of 100% trichloroacetic acid and put on ice for 10 min, in order
to favour protein precipitation. Then, samples were centrifuged at
4°C for 10 min at maximum speed. While the supernatants were
discarded, the protein pellets, resuspended with 100 µl of SB, were
stored at −20°C until their analysis. Protein quantitation was
carried out according to Bradford (30) method, using a specific dye reagent
(Quick Start Bradford, Bio-Rad Laboratories, Milan, Italy) and
bovine serum albumin as standard (Sigma-Aldrich, Milan, Italy).
Finally, protein samples were fractionated through 1D-SDS-PAGE (12%
concentrated) for 30 min at 80 V and further 60 min at 120 V. Gels
were stained with Coomassie blue (G-250) for 2 h, destained three
times for 30 min in methanol, acetic acid and water (2:3:5; v/v/v)
and photographed by VersaDoc (Bio-Rad Laboratories) instrument
associated to Quantity One software (Bio-Rad Laboratories).
Cell growth assay
Lipophilic and hydrophilic fractions of each RJ were
purified as follows. RJ (250 mg) were resuspended in 1 ml of 1X
PBS, vortexed for 30 min and centrifuged at maximum speed for 10
min at 4°C. The supernatant, containing the hydrosoluble portion of
RJ, was transferred into a new Eppendorf tube, while the pellet was
resuspended in 1 ml of dimethyl sulfoxide (DMSO) and subjected to
the same previous protocol, in order to obtain the liposoluble
portion of RJ. Hence, the final pellet was discarded, while both
the extracts were stored at 4°C until their application as cell
treatment. Tumoral (PC3 and SH-SY5Y) or health (C2C12) mammalian
cell lines were cultivated as widely reported in Gismondi et
al (31). In particular, PC3
and C2C12 were propagated in D-MEM (Dulbecco's modified Eagle's
medium), while SH-SY5Y in D-MEM/Ham's F12 (1:1). Cells were equally
distributed in 24-well plates and treated, for 24 and 48 h, with 1,
3 or 5 µl of hydrophilic or lipophilic RJ extract per ml of culture
medium, which corresponded to the hydrosoluble or liposoluble
molecules contained in 250, 750 and 1250 µg of RJ, respectively.
Control cells (CNT) were treated with equal volumes of PBS 1X or
DMSO for the same times. Cell growth was monitored using the MTT
(3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide)
assay carried out exactly according to manufacturer's guidelines
(Sigma-Aldrich). Results were expressed as percentage variation of
the cell proliferation with respect to control (PBS or DMSO CNT),
considered as unit (100%).
Statistics
All data were expressed as mean ± standard deviation
(s.d.) of the relative results obtained in three independent
replicates (n=3). The significance of the analyses was measured by
One-way ANOVA test, using PAST software; p-values <0.05 (vs.
control) were considered significant.
Results
GC-MS profiles of RJs
GC-MS analysis detected 276 molecules in the diethyl
ether extracts of the RJs. On the contrary, the methanol
extractions appeared to be richer than the previous ones,
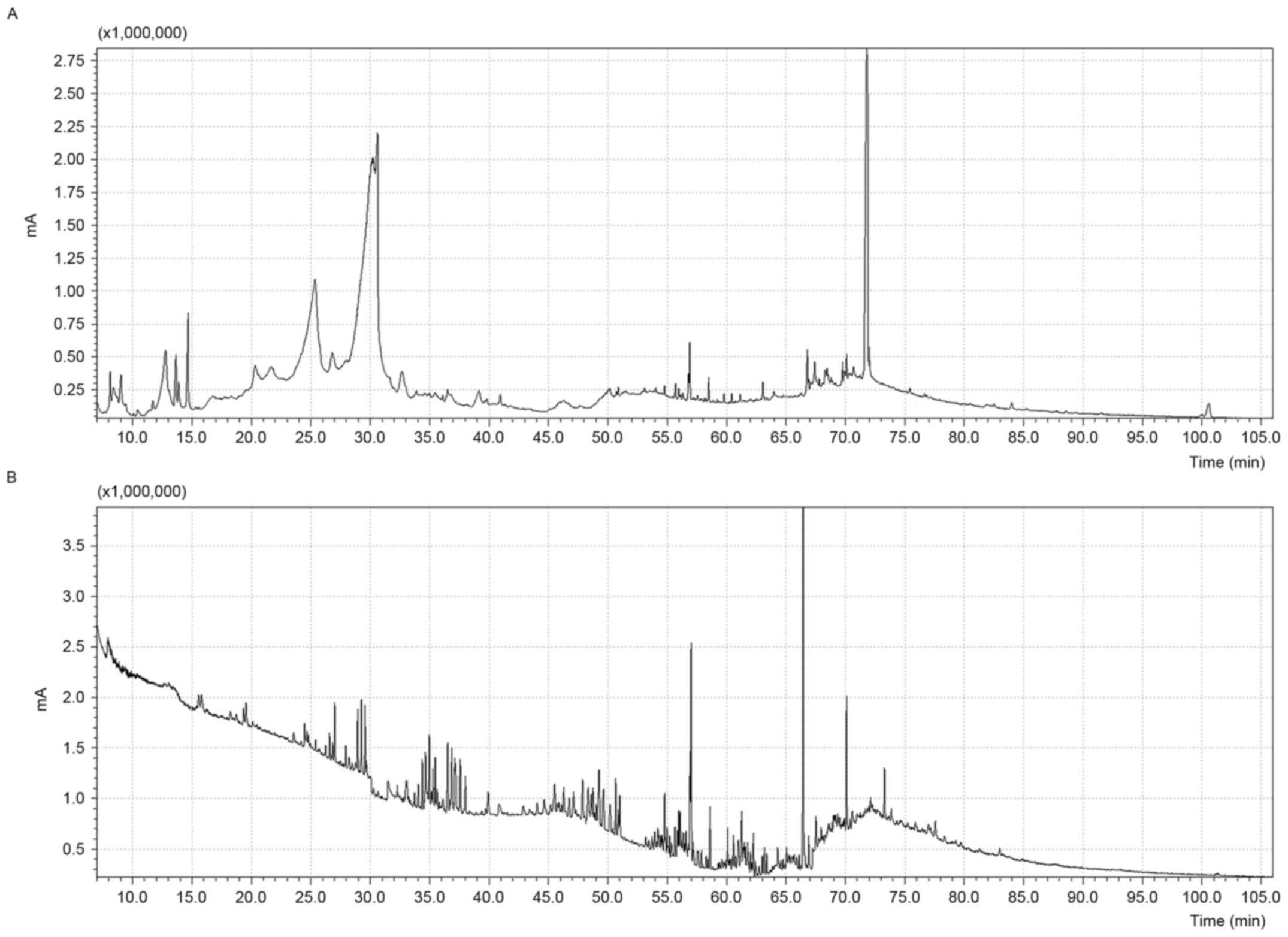
containing a total of 348 different compounds. In fact, as shown in
Fig. 1, the GC profiles of the
methanol extracts presented a higher number of peaks in comparison
with the chromatograms obtained analyzing the diethyl ether
extracts of the RJs.
The lists of molecules identified both in diethyl
ether and in methanol extracts of each RJs are shown in Table I. In this table, the amount of each
compound was reported as percentage of relative abundance (% RA) in
the RJ. In order to resume our results, all the molecules revealed
into the diethyl ether extracts were grouped in four classes of
frequency: class 1, including compounds identified in all the RJs;
class 2, containing molecules present, at least, in 4 RJs; class 3,
presenting rare compounds detectable in 2 or 3 RJs; class 4,
clustering all the substances which could be found only in one RJ.
According to this classification, in the diethyl ether samples, we
detected 4 molecules for the class 1 (eicosane; dodecane
2,6,11-trimethyl; octacosyl trifluoroacetate;
phenantrenemetil-2-phenilcinnamato) and 13, 49 and 210 other
compounds for the class 2, 3 and 4, respectively (Table I). Likewise, the chemical species
identified in the methanol extracts were grouped in similar classes
of frequency. In this case, no molecule could be included in class
1, while 15, 66 and 267 compounds were grouped in class 2, 3 and 4,
respectively (Table I).
 | Table I.Detailed GC-MS profiles of
diethylether and methanol extracts of RJs. |
Table I.
Detailed GC-MS profiles of
diethylether and methanol extracts of RJs.
To better describe the mean composition of the RJs,
we further classified all the molecules detected in the diethyl
ether extracts in different molecular groups as follows: alkanes
(25.27%), acids (including fatty acids; 19.92%), aromatic
derivatives (17.02%), alcohols (15.94%), alkenes (7.60%),
carbohydrates and sugary derivatives (1.81%), quinolinic
derivatives (1.45%), oligopeptides and amino acids (1.08%),
coumarins (0.72%) and others (i.e. aldehydes, ketones, nitrilic
derivatives; 10.50%). In contrast, on the average, the methanol
extracts of the RJs contained a high concentration of fatty acids
(37.35%) followed by alcohols (13.21%), aromatic derivatives
(10.34%), alkanes (5.45%), carbohydrates and sugary derivatives
(4.02%), oligopeptides and amino acids (4.02%), alkenes (3.44%),
quinolinic derivatives (2.01%), cholesterol derivatives (0.6%) and
others (i.e. aldehydes, ketones, nitrilic derivatives; 19.54%).
The diethyl ether extract of the P1 RJ revealed the
highest level of 10-HDA (7.41%) of the remaining ones. Moreover,
only this sample presented the squalane (1.93%), an ester of the
leucin (0.07%), a glycine dipeptide (0.22%) and the decanedioic
(0.04%) and myristoleic (0.03%) acids. P1 methanol extract was
characterized by high doses of 2-Furancarboxaldehyde,
5-(hydroxymethyl) (18.60%) and Erucylamide (24.70%). It was also
the richest of the RJs in typical molecules (class 4), including 80
different peculiar compounds (see Table
I).
P2 sample showed the greatest amounts of
2-Isopropyl-5-methyl-1-heptanol (2.64%), 8-Nonen-2-one (3.02%) and
Octanoic acid (1.17%), with respect to all the other diethyl ether
extracts. On the other hand, it was the least characteristic of the
RJs, according to the low number (n. 19) of specific compounds
(class 4) which typified it. The methanol extract of the P2 RJ was
rich in 2-furancarboxaldehyde, 5-(hydroxymethyl) (20.20%), 10-HDA
(5.92%), 2,5-dimethyl-4-hydroxy-3(2H)-furanone (4.75%), erucylamide
(4.64%) and 2-furancarboxaldehyde, 5-methyl (4.61%). Among the
substances unique in this sample we found an ester of the butanoic
acid (9.55%), the 2-decenoic acid (1.01%), the 3-Hydroxydecanoic
acid (1.07%) and traces of ribose and ascorbic acid.
P3 showed a diethyl ether extract which presented
the highest concentration of 1,6-heptadiene, 2-methyl-6-phenyl
(10.99%) among all the other RJs. Moreover, it typically contained
phthalic and butanal derivatives. The methanol preparation of this
RJ was rich in 2-furancarboxaldehyde, 5-(hydroxymethyl) (23.92%),
4H-pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl (15.77%) and
1,2-cyclopentanedione, 3-methyl (7.33%). The same extract
singularly included trans-13-octadecenoic acid (1.29%), quinoline,
8-hydrazino (1.28%), ascorbic acid (0.05%) a galactofuranoside
derivative (0.71%), a pentanoic acid ester (0.14%) and a glycin
(0.01%) ester.
The GC profile of the G1 diethyl ether extract
revealed the highest level of eicosane (16.58%) and
tetratetracontane (4.17%). Sixty-two different molecules, such as
methyl-cinnamene and capric ether, typically characterized this
sample (class 4, Table I). The
methanol extract of the G1 RJ presented the most elevated doses of
decanoic acid, 2-hydroxy- (4.74%), 2-furanmethanol (4.18%) and
2(5H)-furanone (2.13%) with respect to the other RJs. Moreover,
propanoic acid (11.81%) and pentanoic acid (12.52%) esters
represented the most abundant specific markers of this same
sample.
The diethyl ether extracts of the G2 and G3 RJs
showed the higher concentration of octacosyl trifluoroacetate
(13.68%) and benzonitrile, m-phenethyl (43.70%), respectively. In
particular, G2 sample was typified by the presence of
3-butynyl-benzene, 3,5-bis(1,1-dimethylethyl)-phenol and
2-(2-methylpropyl)-quinoline, while G3 preparation was
characterized by high doses of 2-Cyclopenten-1-one, 2-hydroxy-
(9.22%) and 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl-
(12.58%). The G2 methanol extract revealed elevated amounts of
4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- (38.22%),
benzonitrile, m-phenethyl (10.20%) and 2-cyclopenten-1-one,
2-hydroxy (19.64%). The same extract singularly included
3-butyn-1-ol (2.97%), N-dl-alanylglycine (0.15%), Stearic acid
ester (0.05%) and a mannitol derivative (0.1%). On the other hand,
the methanol extract of the G3 RJ was rich of benzonitrile,
m-phenethyl (27.67%), 2-furancarboxaldehyde, 5-(hydroxymethyl)
(32.14%) and glutaconic anhydride (2.35%). Finally,
6-oxa-bicyclo-hexan-3-one,
5-amino-6-nitroso-pyrimidine-2,4(1H,3H)-dione, 3-pentenyl-benzene,
9-hydroxy-2-nonanone and cyclopentaneundecanoic acid were the
compounds that could be identified only in G3 sample.
Protein quantitation and analysis
Hydrosoluble and liposoluble protein portions of the
RJs were purified as reported in Materials and methods and then
quantified by Bradford method (30). As shown in Table II, all the RJs contained a greater
amount of hydrosoluble proteins than liposoluble ones. In
particular, the highest and the lowest contents of hydrophilic
proteins were detected in G3 (1.174 µg/mg) and P2 (0.831 µg/mg)
samples, respectively. On the other hand, G2 was the richest sample
in lipophilic proteins (0.545 µg/mg) among all the RJs, while P3
(0.207 µg/mg) the poorest one.
 | Table II.Protein quantitation. |
Table II.
Protein quantitation.
| RJ | Hydrosoluble
protein content (µg/mg) | Liposoluble protein
content (µg/mg) |
|---|
| P1 | 0.854±0.017 | 0.262±0.005 |
| P2 | 0.831±0.024 | 0.396±0.007 |
| P3 | 0.967±0.048 | 0.207±0.006 |
| G1 | 1.072±0.053 | 0.313±0.009 |
| G2 | 1.021±0.020 | 0.545±0.010 |
| G3 | 1.174±0.035 | 0.525±0.026 |
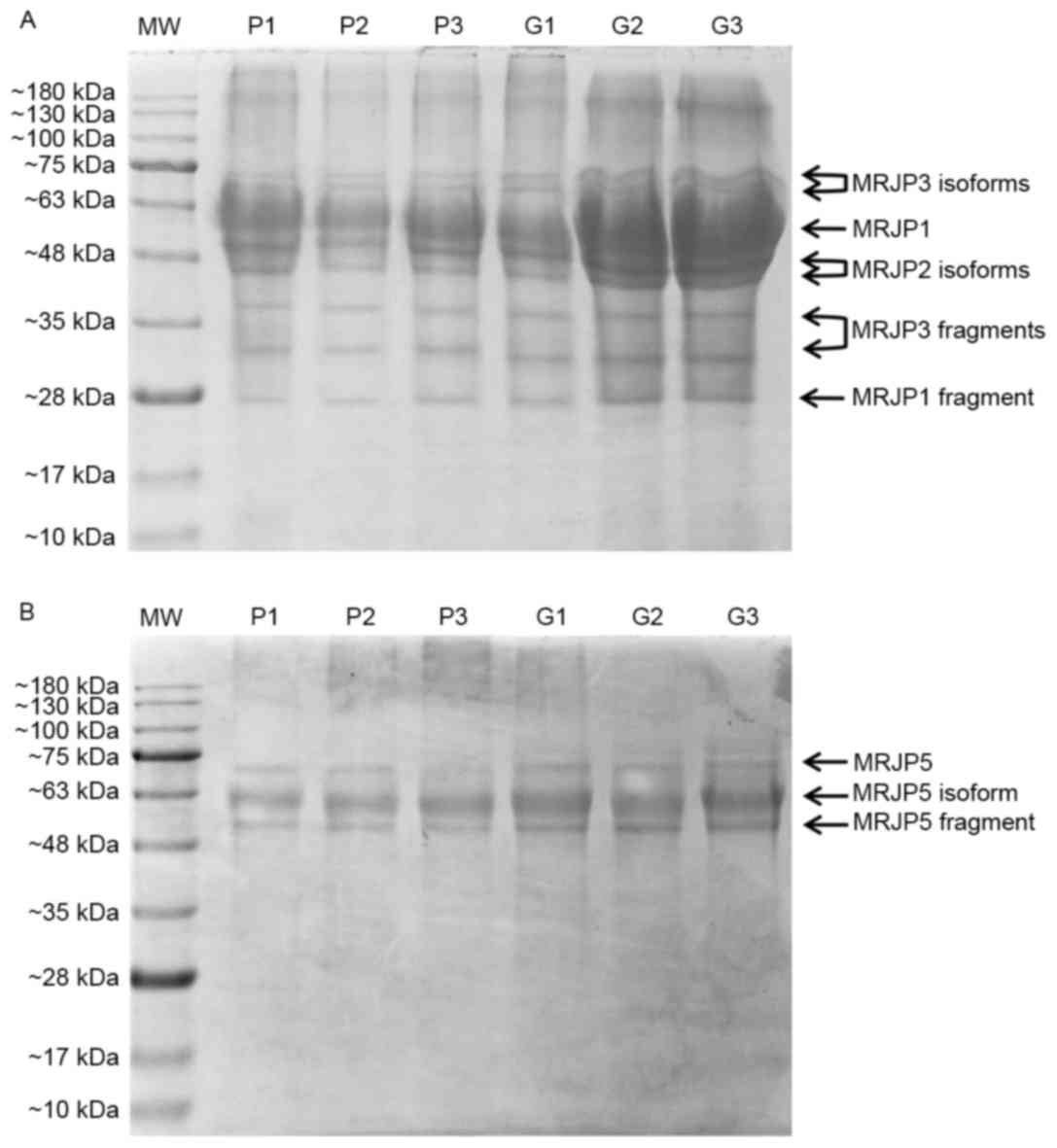
Protein extracts were then subjected to one
dimensional denaturing gel electrophoresis and Coomassie Blue
staining (Fig. 2). No qualitative
difference could be observed among the various samples. In general,
the hydrophilic protein profile of all the RJs was characterized by
8 principal bands. According to literature data (5,32) and
thanks to a standard of molecular weights (MW), we were able to
recognize each one of the protein spots. In particular, MRJP1,
MRJP2, MRJP3, their isoforms and their fragments were identified
(Fig. 2A). In the same manner, we
also identified the three protein bands detected in the gel lanes
where the lipophilic portions of the RJs were fractionated. They
corresponded to MRJP5 and to its isoform and fragment (Fig. 2B).
Biological activity of the RJs on
mammalian cells
The lipophilic and hydrophilic fractions of the RJs
were purified as described in Materials and methods and used to
treat, for 24 and 48 h, three mammalian cell lines: C2C12, PC3 and
SH-SY5Y. In particular, all the treatments were performed at three
different concentrations, that is the equivalent of the
hydrosoluble or liposoluble molecules contained in 250, 750 and
1250 µg of RJ, respectively, per ml of culture medium. Cell growth
was measured by MTT assay and the data are reported in Fig. 3A-F.
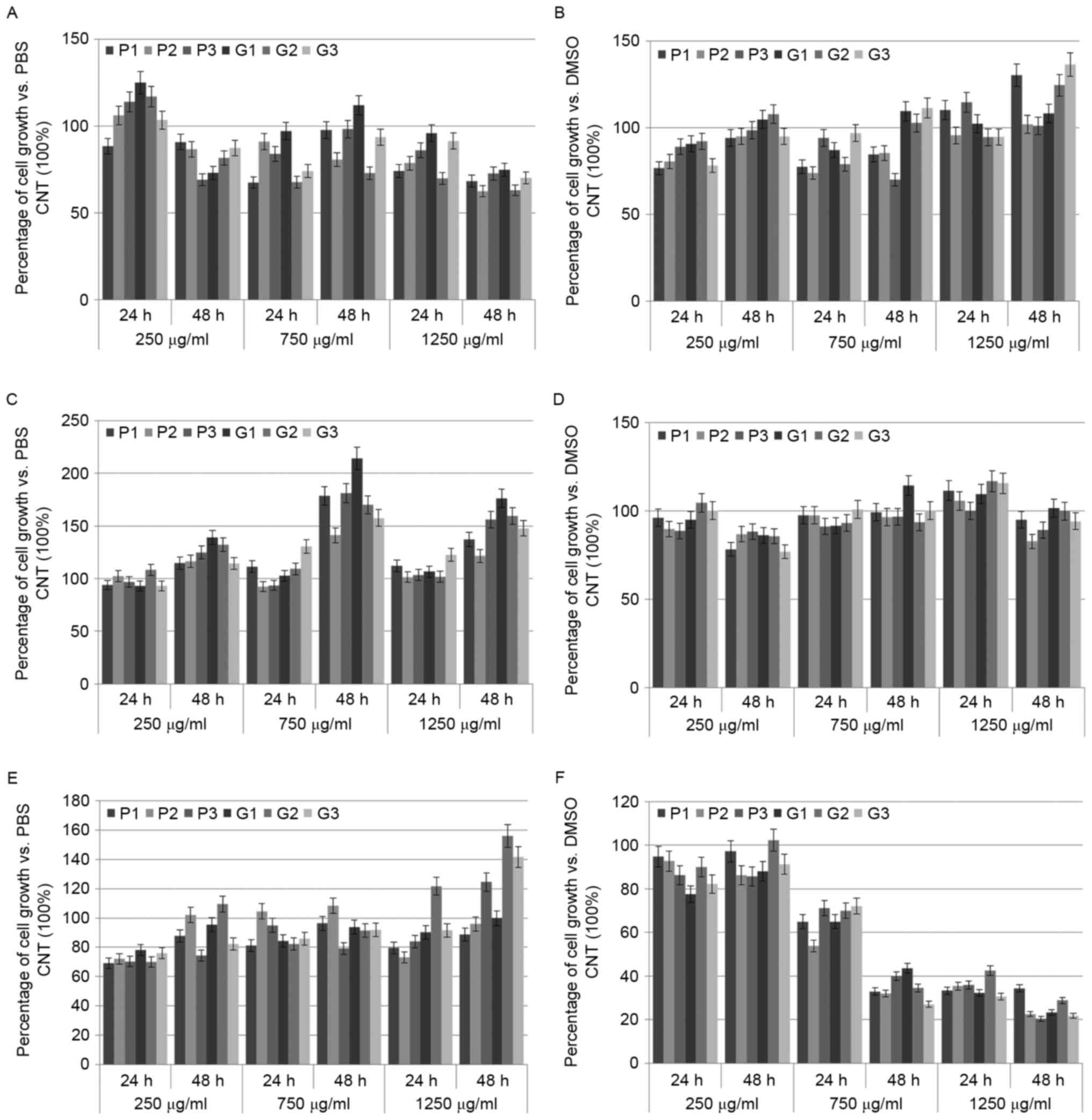 | Figure 3.RJ bioactivity on mammalian cells.
The graphs represent the results obtained by MMT assay performed on
C2C12 (A and B), PC3 (C and D) and SH-SY5Y (E and F) cells after
treatment, for 24 and 48 h, with hydrophilic (A, C and E) or
lipophilic (B, D and F) extract of RJs (P1, P2, P3, G1, G2 and G3)
at different concentrations (250, 750 and 1250 µg per ml of culture
medium). Results are reported as percentage of cell growth compared
to the respective control (PBS or DMSO). Data are expressed as mean
of three independent measurements ± s.d. p-values vs. control:
p<0.05 for (A, B, D and E); p<0.03 for (C); p<0.01 for
(F). |
In C2C12, the hydrophilic extracts of the RJs,
except that P1, slightly induced an increase of cell proliferation
after 24 h of incubation with the lowest dose (maximum value
+24.95% for G1). On the other hand, all the other treatments, at 24
and 48 h with the remaining concentrations, showed a weak reduction
of cell growth, reaching the best antiproliferative effect
(−37.53%) using the P2 hydrophilic fraction at 1250 µg/ml for 48 h.
Singularly, G1 sample determined an increase of C2C12 cells after
48 h of exposure to 750 µg/ml (Fig.
3A).
All lipophilic portions of the RJs caused a slight
decrease of C2C12 proliferation after 24 h of exposure with 250 and
750 µg of extract per ml of culture medium (maximum value −26.01%
for P2 at 750 µg/ml). The same treatments, after 48 h, showed a
rescue of the cell growth (maximum value +36.51% for G3 at 750
µg/ml), except the P3 sample. At the highest concentration, the RJ
fractions did not exercise a significant variation of the cell
reproduction at 24 h, while, at 48 h, P1, G2 and G3 samples induced
hyperproliferative phenomena (maximum value +36.51% for G3)
(Fig. 3B).
PC3 cells did not show any substantial modification
of their cell growth after exposure, for 24 h, to all doses of the
hydrophilic fractions, except G3 (+30.46% at 750 µg/ml; +22.41% at
1250 µg/ml). In contrast, after 48 h, all treatments caused an
increase of the cell count. In particular, G1 was the most
proliferative extract, amounting to +75.95% and +114.14% at 750 and
1250 µg/ml, respectively (Fig.
3C).
In PC3 cells, all lipophilic extracts of the RJs did
not induce significant changes in the proliferation rate. In
general, results of all treatments at 250 µg/ml, for 24 h, at 750
µg/ml, for 24 and 48 h, and at 1250 µg/ml, for 48 h were very
similar to the control, expect that G1 (+14.20%) and P2 (−17.41%)
at 750 and 1250 µg/ml, in that order, after 48 h. On the other
hand, we observed that the lowest dose of treatment with all RJ
fractions caused, after 48 h, a weak antiproliferative effect
(maximum value −24.00% for G3), while the highest one determined,
at 24 h, a feeble pro-proliferative effect (maximum value +16.63
for G2) (Fig. 3D).
SH-SY5Y cells reduced their proliferation after
exposure, for 24 h, to the lowest dose of hydrophilic extracts
(maximum value −30.83% for P1). All the other treatments at 250
µg/ml, for 48 h, at 750 µg/ml, for 24 and 48 h, and at 1250 µg/ml,
for 24 h, determined a variable decrease of the cell growth
(maximum value −26.83% for P2 at 1250 µg/ml), except that P2 and G2
extracts in some cases. After 48 h, the highest doses of P1, P2 and
G1 samples did not produce any significant alteration compared to
the control, while P3, G2 and G3 ones induced an increase of the
cell proliferation of +24.74%, +55.97% and +41.72%, respectively
(Fig. 3E).
Surprisingly, all lipophilic fractions of the RJs
caused a strong reduction of SH-SY5Y cell growth, in a dose- and
time-dependent manner. In particular, after treatment at 1250 µg/ml
for 48 h, the cell proliferation was reduced by RJ extracts in a
range which varied between −65.62% (P1 sample) and −79.53% (P3
sample) (Fig. 3D).
Finally, to facilitate the comprehension of the
biological effect exerted by all RJs on the mammalian cell lines
and individuate the general trend of their treatments, the mean of
the results previously obtained after exposure with the different
RJ fractions, in each condition, was calculated (Fig. 4).
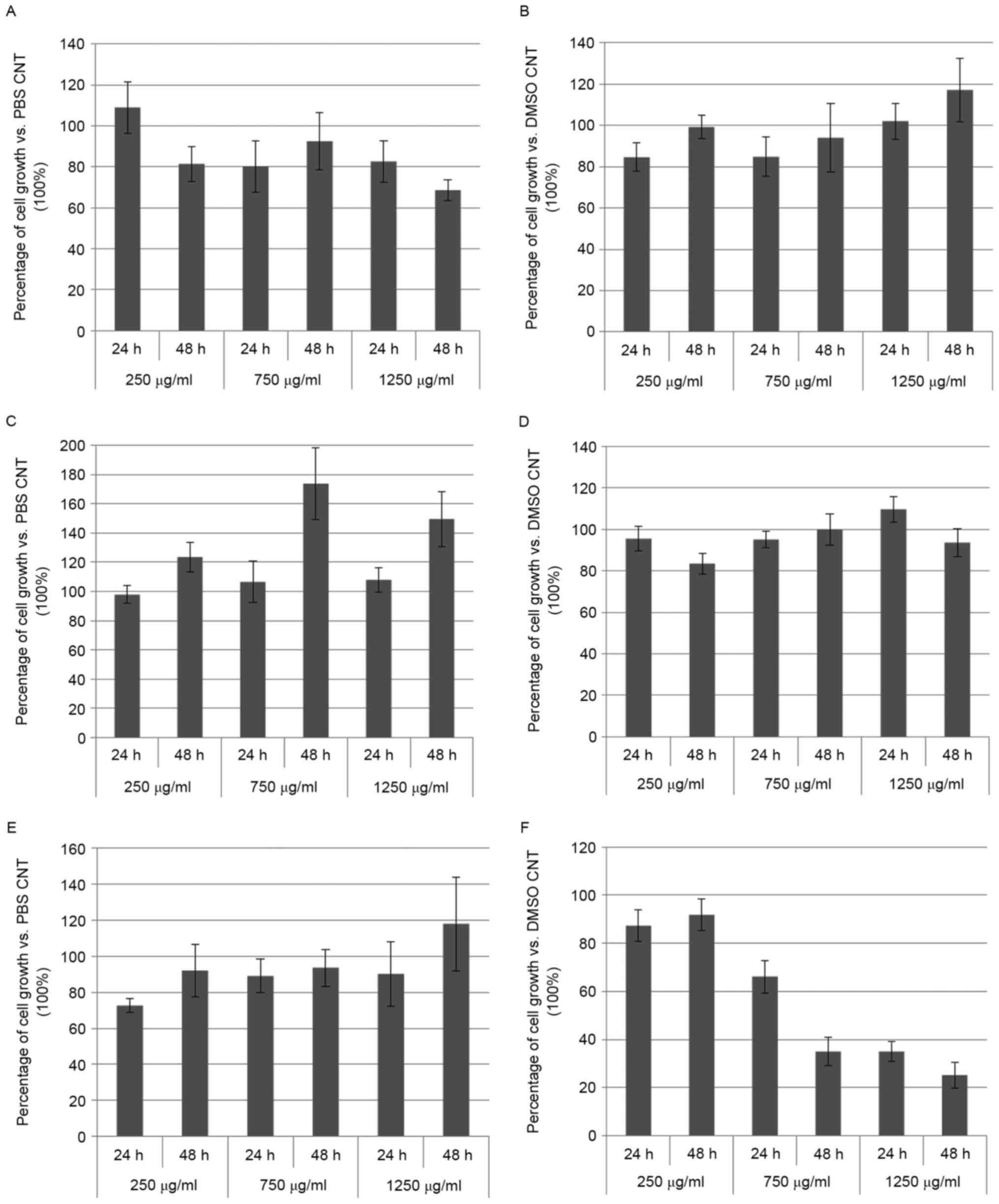 | Figure 4.Resuming diagrams of the MTT results
reported in Figure 3. Each graph
represent the mean value ± s.d. of the six biological effects
induced by the hydrophilic (A, C and E) or lipophilic (B, D and F)
extracts of the RJs (P1, P2, P3, G1, G2 and G3) on C2C12 (A and B),
PC3 (C and D) and SH-SY5Y (E and F) cells, after treatment (for 24
and 48 h) at different concentrations (250, 750 and 1250 µg per ml
of culture medium). Results were reported as percentage of cell
growth compared to the respective control (PBS or DMSO); p-values
vs. control: p<0.05 for (A, B, D and E); p<0.02 for (C);
p<0.01 for (F). |
Discussion
We studied the biochemical composition of six
different RJs, with the purpose of understanding if these matrixes
were characterized by homogeneous and similar chemical profiles or
by specific and typical spectra. To do it, a high-throughput GC-MS
analysis was carried out both on methanol and on diethyl ether
extracts of the RJs (Fig. 1),
detecting a total of 348 and 276 different compounds, respectively
(Table I). Literature data usually
report a substantially lower number of molecules associated to RJ
(11,26), indicating that the present research
can be considered a very in-depth and original work which does not
limit itself to the identification of the main chemical markers of
the RJ but reveals all its components. In general, the amount of
substances that were peculiar of only one sample (class 4 for each
type of extraction, Table I) was
always greater than those commonly detectable in two or more RJs
(classes 3, 2 and 1), suggesting that these bee products are almost
impossible to standardize. In fact, RJ composition is strongly
dependent on several factors, such as environment conditions,
genetics and physiology of the bee colony, plant biodiversity
distributed around the hive (33).
The chromatographic study revealed that the acids,
including free fatty acids, were the most abundant compounds in all
RJs, as widely reported in literature (34,35).
In particular, 10-HDA and its saturated form, considered as
authenticity and freshness markers of RJs (36), were detected in all matrixes, except
in G2 and G3, suggesting that these molecules cannot be used as
absolute parameters for RJ certification and/or that G2 and G3
samples were partially spoiled (deriving from large distribution
network, see Materials and methods). In the RJs, we also detected
high concentrations of plant phenolic derivatives (i.e.
m-phenethyl-benzonitrile) and alcans (i.e. eicosane) with
antimicrobial and antioxidant properties, in coherence with other
works (37,38). On the other hand, low doses of
sugars and peptides could be revealed only in methanol
extracts.
Another innovative aspect of the current work was
the fractionation of the RJ proteins according to their
lipophilicity. All RJs showed an higher content of hydrosoluble
proteins than liposoluble ones (Table
II). Thanks to one-dimensional SDS-PAGEs (Fig. 2), we were also able to separate and
distinguish these proteins in all samples. In particular, MPRJ1
appeared as the most abundant of the hydrophilic proteins, followed
by MPRJ2 and 3. On the contrary, MPRJ5 was the only one which could
be found in the lipophilic protein extracts. Finally, MPRJ4 was not
detectable in any fractions, as expected due to the low expression
level of its mRNA in nurse bees (39). The multiple isoforms and fragments
of the RJ proteins could be easily explained by the existence of
several repeated regions into their amino acidic sequences and by
degradation processes, respectively (32). In general, our results did not
reveal qualitative differences in the protein profiles of the six
samples, as happened in GC-MS analysis, possibly because the
protein composition of the RJ is influenced by fewer factors than
its chemical assortment.
Since only a few scientific works describe the
antineoplastic properties of the RJ, we focused our research on
in vitro analysis of the biological effect of RJs on cell
growth of three different mammalian cell lines (C2C12, PC3 and
SH-SY5Y). The choice of these specific cell models was encouraged
by the lack of studies on RJ bioactivity on them. Moreover, for the
first time in literature, the cell treatments were carried out
using, separately, both hydrophilic and lipophilic fraction of the
RJs, at various concentrations and times. With respect to the
control, each fraction of RJ exercised a typical effect on the
growth rate of the treated cells (Fig.
3). However, in most cases, we observed that extracts of
different RJs surprisingly induced a similar proliferative trend on
the same cells; although each RJ had a peculiar biochemical profile
(Table I) and protein content
(Table II).
In general, the hydrophilic fraction of the RJs
caused a small decrease of C2C12 proliferation, while the
respective lipophilic portion determined just a slight increase of
the cell growth, both in a time- and dose-dependent manner
(Fig. 4A and B). On PC3, the
hydrophilic extracts caused a time-dependent over-proliferation,
while the lipophilic ones did not show any significant effect
(Fig. 4C and D). Finally, the
hydrophilic samples did not effectively influence SH-SY5Y growth
rate, whereas antiproliferative effects were extraordinarily
revealed by the lipophilic fractions, according to concentration
and exposure of the treatments (Fig. 4E
and F). This last outcome could suggest a potential medical
application of the lipophilic portion of the RJ as antineoplastic
treatment for human neuroblastoma.
Similar results were already described in Tamura
et al (25), where the
authors reported that lipid components of the RJ presented
antineoplastic property against slow-growing cancers (i.e. P388 and
L1210 murine lymphocytic leukemias, 180 murine sarcoma), such as
SH-SY5Y neuroblastoma we studied. Obviously, other in vitro
and in vivo studies should be carried out to confirm this
hypothesis. Finally, the use of RJ to prevent the onset and slow
down the growth of the neuroblastoma could also be supported by the
evidence, documented in Hashimoto et al (40) and Hattori et al (41), which proved that RJ fatty acids (in
particular 10-HAD) showed neurotrophic and differentiative activity
on murine neuronal cells.
It is quite difficult to establish which RJ compound
exerts antiproliferative effects, since RJs usually possess very
different compositions, as demonstrated in the present study.
However, we hypothesize that this specific bioactivity is strongly
linked to lipophilic molecules, such as those detected in all our
samples (eicosane; dodecane, 2,6,11-trimethyl; octacosyl
trifluoroacetate; phenantrenemetil-2 phenilcinnamato; class 1,
Table I). The predominant
biological role that literature assigned to 10-HAD (and its
saturated form) in the RJ was unexpectedly not confirmed in the
current research. In fact, all RJs induced SH-SY5Y growth
inhibition, although some of them were lacking this fatty acid.
Previous evidence let us to believe that all RJ compounds work in
synergy to reach the final observed effect.
In conclusion, all lipophilic fractions extracted
from chemically different RJs surprisingly shared a common
antitumoral feature against human neuroblastoma.
Acknowledgements
The present study was supported and financed by
Regione Lazio (FILAS-RU-2014-1122) through the SMART CAMPUS PROGRAM
‘Analisi qualità delle materie prime, origine e verifica di
contaminazione di alimenti vegetali’ - codice F1-2016-0069 (CUP:
E82I15000980002).
References
|
1
|
Haydak MH: Honey bee nutrition. Annu Rev
Entomol. 15:143–156. 1970. View Article : Google Scholar
|
|
2
|
Contessi A: Le api biologia, allevamento,
prodotti. 4th. Edagricole, Bologna: 2016, (In Italian).
|
|
3
|
Nagai T and Inoue R: Preparation and
functional properties of water and alkaline extract of royal jelly.
Food Chem. 84:181–186. 2004. View Article : Google Scholar
|
|
4
|
Hanes J and Simúth J: Identification and
partial characterization of major royal jelly protein of honeybee
(Apis mellifera). J Apic Res. 31:22–26. 1992. View Article : Google Scholar
|
|
5
|
Schmitzová J, Klaudiny J, Albert S,
Schröder W, Schreckengost W, Hanes J, Júdová J and Simúth J: A
family of major royal jelly proteins of the honeybee Apis
mellifera L. Cell Mol Life Sci. 54:1020–1030. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Boselli E, Caboni MF, Sabatini AG,
Marcazzan GL and Lercker G: Determination and changes of free amino
acids in royal jelly during storage. Apidologie (Celle).
34:129–137. 2003. View Article : Google Scholar
|
|
7
|
Fujita T, Kozuka-Hata H, Ao-Kondo H,
Kunieda T, Oyama M and Kubo T: Proteomic analysis of the royal
jelly and characterization of the functions of its derivation
glands in the honeybee. J Proteome Res. 12:404–411. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lercker G, Savioli S, Vecchi MA, Sabatini
AG, Nanetti A and Piana L: Carbohydrate determination of royal
jelly by high resolution gas chromatography (HRGC). Food Chem.
19:255–264. 1986. View Article : Google Scholar
|
|
9
|
Lercker G, Capella P, Conte LS, Ruini F
and Giordani G: Components of royal jelly: I. Identification of the
organic acids. Lipids. 16:912–919. 1981. View Article : Google Scholar
|
|
10
|
Caboni MF, Sabatini AG and Lercker G: La
gelatina reale: origine, proprietà e composizione/Royal
jelly:origin, properties and composition. APOidea. 1:72–79.
2004.
|
|
11
|
Melliou E and Chinou I: Chemistry and
bioactivity of royal jelly from Greece. J Agric Food Chem.
53:8987–8992. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gómez-Caravaca AM, Gómez-Romero M,
Arráez-Román D, Segura-Carretero A and Fernández-Gutiérrez A:
Advances in the analysis of phenolic compounds in products derived
from bees. J Pharm Biomed Anal. 41:1220–1234. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mohamed AA, Galal AA and Elewa YH:
Comparative protective effects of royal jelly and cod liver oil
against neurotoxic impact of tartrazine on male rat pups brain.
Acta Histochem. 117:649–658. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Viuda-Martos M, Ruiz-Navajas Y,
Fernández-López J and Pérez-Alvarez JA: Functional properties of
honey, propolis, and royal jelly. J Food Sci. 73:R117–R124. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Guo H, Saiga A, Sato M, Miyazawa I,
Shibata M, Takahata Y and Morimatsu F: Royal jelly supplementation
improves lipoprotein metabolism in humans. J Nutr Sci Vitaminol
(Tokyo). 53:345–348. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Münstedt K, Bargello M and Hauenschild A:
Royal jelly reduces the serum glucose levels in healthy subjects. J
Med Food. 12:1170–1172. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cihangir E, Orhan D, Ercument O and Yasam
B: The effects of royal jelly on autoimmunity in Grave's disease.
Endocrinologia. 30:175–183. 2006.
|
|
18
|
Majtán J, Kovácová E, Bíliková K and
Simúth J: The immunostimulatory effect of the recombinant apalbumin
1-major honeybee royal jelly protein-on TNFalpha release. Int
Immunopharmacol. 6:269–278. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bincoletto C, Eberlin S, Figueiredo CA,
Luengo MB and Queiroz ML: Effects produced by Royal Jelly on
haematopoiesis: Relation with host resistance against Ehrlich
ascites tumour challenge. Int Immunopharmacol. 5:679–688. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hidaka S, Okamoto Y, Uchiyama S, Nakatsuma
A, Hashimoto K, Ohnishi ST and Yamaguchi M: Royal jelly prevents
osteoporosis in rats: Beneficial effects in ovariectomy model and
in bone tissue culture model. Evid Based Complement Alternat Med.
3:339–348. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yatsunami K and Echigo T: Antibacterial
action of royal jelly. Bull Fac Agric Tamagawa Univ. 25:13–22.
1985.
|
|
22
|
Fujiwara S, Imai J, Fujiwara M, Yaeshima
T, Kawashima T and Kobayashi K: A potent antibacterial protein in
royal jelly. Purification and determination of the primary
structure of royalisin. J Biol Chem. 265:11333–11337.
1990.PubMed/NCBI
|
|
23
|
Taniguchi Y, Kohno K, Inoue S, Koya-Miyata
S, Okamoto I, Arai N, Iwaki K, Ikeda M and Kurimoto M: Oral
administration of royal jelly inhibits the development of atopic
dermatitis-like skin lesions in NC/Nga mice. Int Immunopharmacol.
3:1313–1324. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shirzad M, Kordyazdi R, Shahinfard N and
Nikokar M: Does Royal jelly affect tumor cells. J HerbMed
Pharmacol. 2:45–48. 2013.
|
|
25
|
Tamura T, Fujii A and Kuboyama N:
Antitumor effects of royal jelly (RJ). Nippon Yakurigaku Zasshi.
89:73–80. 1987.(In Japanese). View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Isidorov VA, Bakier S and Grzech I: Gas
chromatographic-mass spectrometric investigation of volatile and
extractable compounds of crude royal jelly. J Chromatogr B Analyt
Technol Biomed Life Sci. 885–886:109–116. 2012. View Article : Google Scholar
|
|
27
|
Gismondi A, Canuti L, Grispo M and Canini
A: Biochemical composition and antioxidant properties of
Lavandula angustifolia Miller essential oil are shielded by
propolis against UV radiations. Photochem Photobiol. 90:702–708.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Giovannini D, Gismondi A, Basso A, Canuti
L, Braglia R, Canini A, Mariani F and Cappelli G: Lavandula
angustifolia Mill. essential oil exerts antibacterial and
anti-inflammatory effect in macrophage mediated immune response to
Staphylococcus aureus. Immunol Invest. 45:11–28. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li J, Wang T, Zhang Z and Pan Y: Proteomic
analysis of royal jelly from three strains of western honeybees
(Apis mellifera). J Agric Food Chem. 55:8411–8422. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bradford MM: A rapid and sensitive method
for the quantitation of microgram quantities of protein utilizing
the principle of protein-dye binding. Anal Biochem. 72:248–254.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gismondi A, Lentini A, Tabolacci C,
Provenzano B and Beninati S: Transglutaminase-dependent
antiproliferative and differentiative properties of nimesulide on
B16-F10 mouse melanoma cells. Amino Acids. 38:257–262. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Scarselli R, Donadio E, Giuffrida MG,
Fortunato D, Conti A, Balestreri E, Felicioli R, Pinzauti M,
Sabatini AG and Felicioli A: Towards royal jelly proteome.
Proteomics. 5:769–776. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Howe SR, Dimick PS and Benton AW:
Composition of freshly harvested and commercial royal jelly. J Apic
Res. 24:52–61. 1985. View Article : Google Scholar
|
|
34
|
Kodai T, Umebayashi K, Nakatani T,
Ishiyama K and Noda N: Compositions of royal jelly II. Organic acid
glycosides and sterols of the royal jelly of honeybees (Apis
mellifera). Chem Pharm Bull (Tokyo). 55:1528–1531. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bogdanov S: Royal Jelly, Bee Brood:
Composition, Health, Medicine: A Review. Bee Product Sci. 1–36.
2011.
|
|
36
|
Antinelli JF, Zeggane S, Ico R Dav,
Rognone C, Faucon JP and Lizzani L: Evaluation of
(E)-10-hydroxydec-2-enoic acid as a freshness parameter for royal
jelly. Food Chem. 80:85–89. 2003. View Article : Google Scholar
|
|
37
|
Matsuka M: Content of benzoic acid in
royal jelly and propolis. Mitsubachi Kagaku. 14:79–80. 1993.
|
|
38
|
Rizwan K, Zubair M, Rasool N, Riaz M,
Zia-Ul-Haq M and de Feo V: Phytochemical and biological studies of
Agave attenuata. Int J Mol Sci. 13:6440–6451. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Klaudiny J, Hanes J, Kulifajová J, Albert
Š and Šimúth J: Molecular cloning of two cDNAs from the head of the
nurse honeybee (Apis mellifera L.) for coding related
proteins of royal jelly. J Apic Res. 33:105–111. 1994.doi:
10.1080/00218839.1994.11100857. View Article : Google Scholar
|
|
40
|
Hashimoto M, Kanda M, Ikeno K, Hayashi Y,
Nakamura T, Ogawa Y, Fukumitsu H, Nomoto H and Furukawa S: Oral
administration of royal jelly facilitates mRNA expression of glial
cell line-derived neurotrophic factor and neurofilament H in the
hippocampus of the adult mouse brain. Biosci Biotechnol Biochem.
69:800–805. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hattori N, Nomoto H, Fukumitsu H, Mishima
S and Furukawa S: Royal jelly and its unique fatty acid,
10-hydroxy-trans-2-decenoic acid, promote neurogenesis by neural
stem/progenitor cells in vitro. Biomed Res. 28:261–266. 2007.
View Article : Google Scholar : PubMed/NCBI
|